Abstract
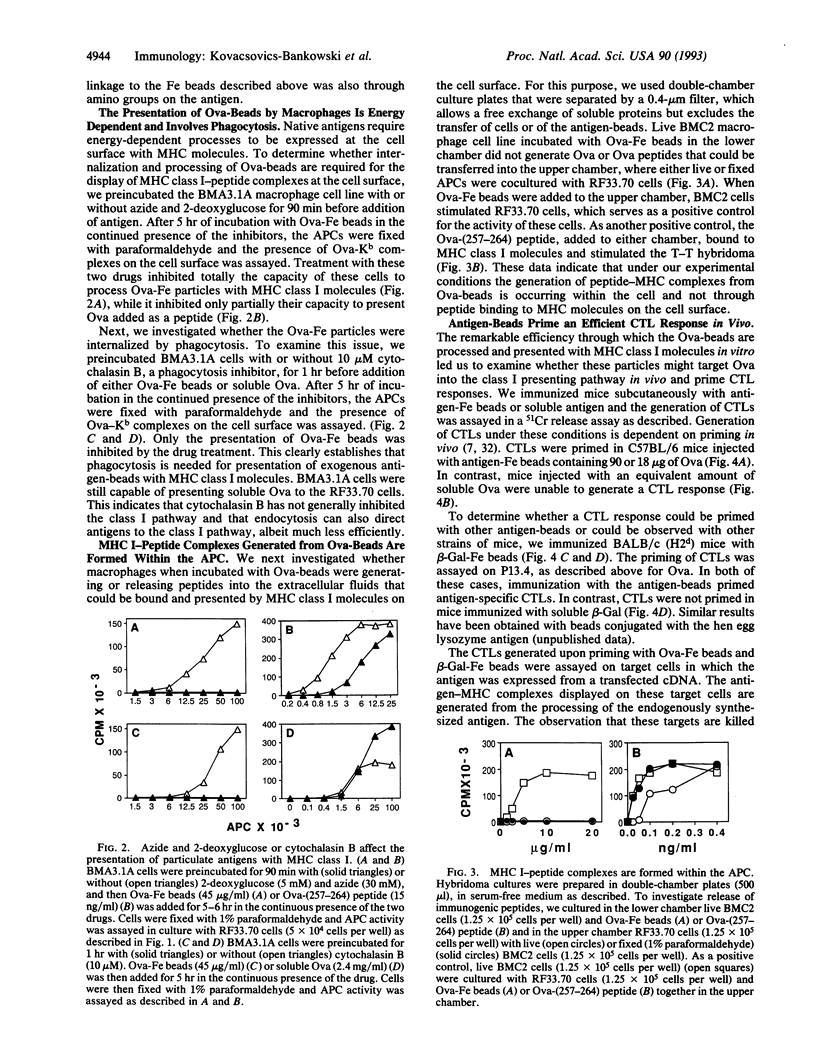
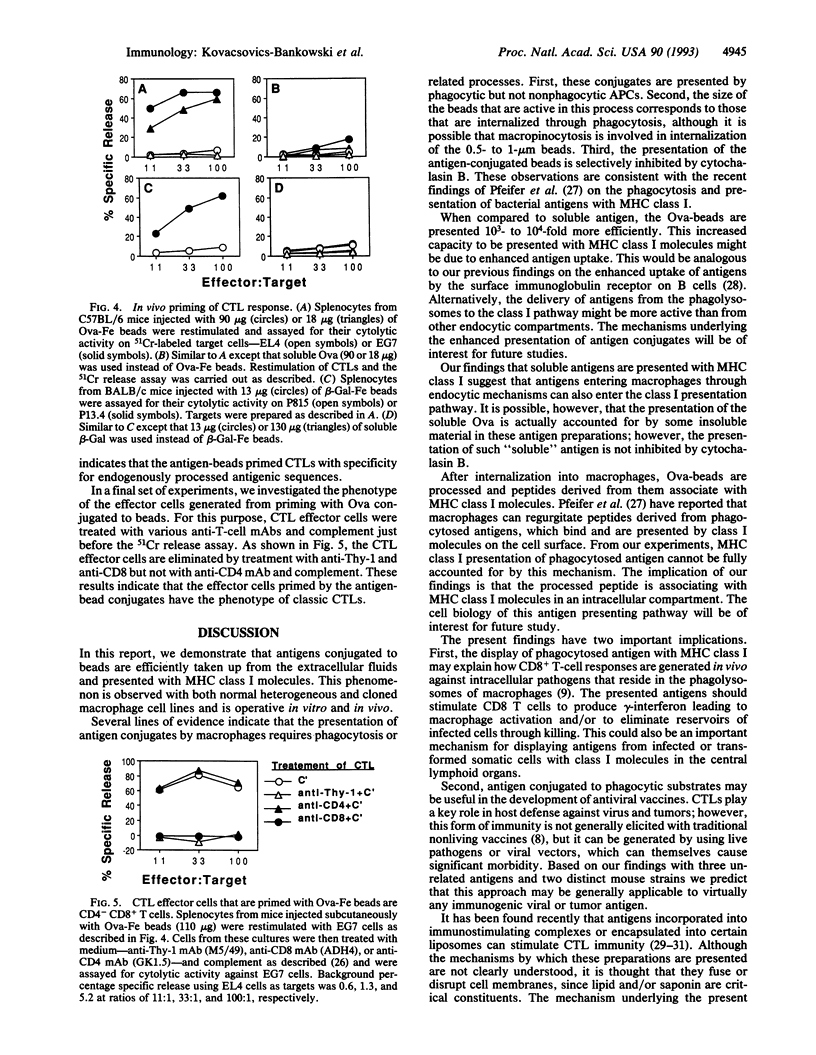
Antigens in extracellular fluids can be processed and presented with major histocompatibility complex (MHC) class I molecules by a subset of antigen presenting cells (APCs). Chicken egg ovalbumin (Ova) linked to beads was presented with MHC class I molecules by these cells up to 10(4)-fold more efficiently than soluble Ova. This enhanced presentation was observed with covalently or noncovalently linked Ova and with beads of different compositions. A key parameter in the activity of these conjugates was the size of the beads. The APC that is responsible for this form of presentation is a macrophage. These cells internalize the antigen constructs through phagocytosis, since cytochalasin B inhibited presentation. Processing of the antigen and association with MHC class I molecules appears to occur intracellularly as presentation was observed under conditions where there was no detectable release of peptides into the extracellular fluids. When injected in vivo in C57BL/6 mice, Ova-beads, but not soluble Ova, primed CD4- CD8+ cytotoxic T lymphocytes (CTLs). Similar results were obtained in BALB/c mice immunized with beta-galactosidase-beads. The implications of these findings for development of nonliving vaccines that stimulate CTL immunity are discussed.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bevan M. J. Cross-priming for a secondary cytotoxic response to minor H antigens with H-2 congenic cells which do not cross-react in the cytotoxic assay. J Exp Med. 1976 May 1;143(5):1283–1288. doi: 10.1084/jem.143.5.1283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bjorkman P. J., Saper M. A., Samraoui B., Bennett W. S., Strominger J. L., Wiley D. C. The foreign antigen binding site and T cell recognition regions of class I histocompatibility antigens. Nature. 1987 Oct 8;329(6139):512–518. doi: 10.1038/329512a0. [DOI] [PubMed] [Google Scholar]
- Braciale T. J., Morrison L. A., Sweetser M. T., Sambrook J., Gething M. J., Braciale V. L. Antigen presentation pathways to class I and class II MHC-restricted T lymphocytes. Immunol Rev. 1987 Aug;98:95–114. doi: 10.1111/j.1600-065x.1987.tb00521.x. [DOI] [PubMed] [Google Scholar]
- Carbone F. R., Bevan M. J. Class I-restricted processing and presentation of exogenous cell-associated antigen in vivo. J Exp Med. 1990 Feb 1;171(2):377–387. doi: 10.1084/jem.171.2.377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carter V. C., Schaffer P. A., Tevethia S. S. The involvement of herpes simplex virus type 1 glycoproteins in cell-mediated immunity. J Immunol. 1981 May;126(5):1655–1660. [PubMed] [Google Scholar]
- Collins D. S., Findlay K., Harding C. V. Processing of exogenous liposome-encapsulated antigens in vivo generates class I MHC-restricted T cell responses. J Immunol. 1992 Jun 1;148(11):3336–3341. [PubMed] [Google Scholar]
- Davignon D., Martz E., Reynolds T., Kürzinger K., Springer T. A. Lymphocyte function-associated antigen 1 (LFA-1): a surface antigen distinct from Lyt-2,3 that participates in T lymphocyte-mediated killing. Proc Natl Acad Sci U S A. 1981 Jul;78(7):4535–4539. doi: 10.1073/pnas.78.7.4535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Debrick J. E., Campbell P. A., Staerz U. D. Macrophages as accessory cells for class I MHC-restricted immune responses. J Immunol. 1991 Nov 1;147(9):2846–2851. [PubMed] [Google Scholar]
- Dialynas D. P., Wilde D. B., Marrack P., Pierres A., Wall K. A., Havran W., Otten G., Loken M. R., Pierres M., Kappler J. Characterization of the murine antigenic determinant, designated L3T4a, recognized by monoclonal antibody GK1.5: expression of L3T4a by functional T cell clones appears to correlate primarily with class II MHC antigen-reactivity. Immunol Rev. 1983;74:29–56. doi: 10.1111/j.1600-065x.1983.tb01083.x. [DOI] [PubMed] [Google Scholar]
- Doherty P. C., Knowles B. B., Wettstein P. J. Immunological surveillance of tumors in the context of major histocompatibility complex restriction of T cell function. Adv Cancer Res. 1984;42:1–65. doi: 10.1016/s0065-230x(08)60455-8. [DOI] [PubMed] [Google Scholar]
- Fast L. D., Fan D. P. An adherent, radiosensitive accessory cell necessary for mouse cytolytic T lymphocyte response to viral antigen. J Immunol. 1981 Mar;126(3):1114–1119. [PubMed] [Google Scholar]
- Grant E. P., Rock K. L. MHC class I-restricted presentation of exogenous antigen by thymic antigen-presenting cells in vitro and in vivo. J Immunol. 1992 Jan 1;148(1):13–18. [PubMed] [Google Scholar]
- Hosken N. A., Bevan M. J., Carbone F. R. Class I-restricted presentation occurs without internalization or processing of exogenous antigenic peptides. J Immunol. 1989 Feb 15;142(4):1079–1083. [PubMed] [Google Scholar]
- Jin Y., Shih W. K., Berkower I. Human T cell response to the surface antigen of hepatitis B virus (HBsAg). Endosomal and nonendosomal processing pathways are accessible to both endogenous and exogenous antigen. J Exp Med. 1988 Jul 1;168(1):293–306. doi: 10.1084/jem.168.1.293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaufmann S. H. CD8+ T lymphocytes in intracellular microbial infections. Immunol Today. 1988 Jun;9(6):168–174. doi: 10.1016/0167-5699(88)91292-3. [DOI] [PubMed] [Google Scholar]
- Moore M. W., Carbone F. R., Bevan M. J. Introduction of soluble protein into the class I pathway of antigen processing and presentation. Cell. 1988 Sep 9;54(6):777–785. doi: 10.1016/s0092-8674(88)91043-4. [DOI] [PubMed] [Google Scholar]
- Pfeifer J. D., Wick M. J., Roberts R. L., Findlay K., Normark S. J., Harding C. V. Phagocytic processing of bacterial antigens for class I MHC presentation to T cells. Nature. 1993 Jan 28;361(6410):359–362. doi: 10.1038/361359a0. [DOI] [PubMed] [Google Scholar]
- Reddy R., Zhou F., Nair S., Huang L., Rouse B. T. In vivo cytotoxic T lymphocyte induction with soluble proteins administered in liposomes. J Immunol. 1992 Mar 1;148(5):1585–1589. [PubMed] [Google Scholar]
- Rock K. L., Benacerraf B., Abbas A. K. Antigen presentation by hapten-specific B lymphocytes. I. Role of surface immunoglobulin receptors. J Exp Med. 1984 Oct 1;160(4):1102–1113. doi: 10.1084/jem.160.4.1102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rock K. L., Fleischacker C., Gamble S. Peptide-priming of cytolytic T cell immunity in vivo using beta 2-microglobulin as an adjuvant. J Immunol. 1993 Feb 15;150(4):1244–1252. [PubMed] [Google Scholar]
- Rock K. L., Gamble S., Rothstein L. Presentation of exogenous antigen with class I major histocompatibility complex molecules. Science. 1990 Aug 24;249(4971):918–921. doi: 10.1126/science.2392683. [DOI] [PubMed] [Google Scholar]
- Rock K. L., Rothstein L., Gamble S., Fleischacker C. Characterization of antigen-presenting cells that present exogenous antigens in association with class I MHC molecules. J Immunol. 1993 Jan 15;150(2):438–446. [PubMed] [Google Scholar]
- Rock K. L., Rothstein L., Gamble S. Generation of class I MHC-restricted T-T hybridomas. J Immunol. 1990 Aug 1;145(3):804–811. [PubMed] [Google Scholar]
- Rock K. L. The role of Ia molecules in the activation of T lymphocytes. I. The activation of an IL 1-dependent IL 2-producing T cell hybridoma by Con A requires an interaction, which is not H-2-restricted, with an Ia-bearing accessory cell. J Immunol. 1982 Oct;129(4):1360–1366. [PubMed] [Google Scholar]
- Staerz U. D., Karasuyama H., Garner A. M. Cytotoxic T lymphocytes against a soluble protein. Nature. 1987 Oct 1;329(6138):449–451. doi: 10.1038/329449a0. [DOI] [PubMed] [Google Scholar]
- Takahashi H., Takeshita T., Morein B., Putney S., Germain R. N., Berzofsky J. A. Induction of CD8+ cytotoxic T cells by immunization with purified HIV-1 envelope protein in ISCOMs. Nature. 1990 Apr 26;344(6269):873–875. doi: 10.1038/344873a0. [DOI] [PubMed] [Google Scholar]
- Townsend A., Bodmer H. Antigen recognition by class I-restricted T lymphocytes. Annu Rev Immunol. 1989;7:601–624. doi: 10.1146/annurev.iy.07.040189.003125. [DOI] [PubMed] [Google Scholar]
- Watson J. Continuous proliferation of murine antigen-specific helper T lymphocytes in culture. J Exp Med. 1979 Dec 1;150(6):1510–1519. doi: 10.1084/jem.150.6.1510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yewdell J. W., Bennink J. R. Cell biology of antigen processing and presentation to major histocompatibility complex class I molecule-restricted T lymphocytes. Adv Immunol. 1992;52:1–123. doi: 10.1016/s0065-2776(08)60875-5. [DOI] [PubMed] [Google Scholar]
- Yewdell J. W., Bennink J. R., Hosaka Y. Cells process exogenous proteins for recognition by cytotoxic T lymphocytes. Science. 1988 Feb 5;239(4840):637–640. doi: 10.1126/science.3257585. [DOI] [PubMed] [Google Scholar]
- Zhou F., Rouse B. T., Huang L. Induction of cytotoxic T lymphocytes in vivo with protein antigen entrapped in membranous vehicles. J Immunol. 1992 Sep 1;149(5):1599–1604. [PubMed] [Google Scholar]



