Abstract
Purpose
BRAF inhibitors (BRAFi) extend survival in BRAF mutant melanoma but can promote the growth of Ras mutant neoplasms. This study determined if gastrointestinal polyps found in BRAFi-treated patients harbored Ras mutations.
Experimental Design
Colonic and gastric polyps were identified and resected from BRAFi-treated melanoma patients. Next generation sequencing (NGS) was performed on polyps. The ability of BRAFi to promote polyp formation was functionally characterized in Apc Min +/− mice. MAPK and beta catenin pathway activity was assessed by immunohistochemistry in mouse and human polyps.
Results
14 patients treated with BRAFi underwent endoscopy to assess for polyps. Seven out of 7 patients >40 years of age and treated for >2 years were found to have colonic tubular adenomas with 4 out of the 7 patients having 5 or more polyps. One patient presented with bleeding from hyperplastic gastric polyps that recurred 6 months after BRAFi rechallenge. NGS performed on polyps found no mutations in MAPK pathway genes, but found APC mutations in all tubular adenomas. A significant increase in the number of polyps was observed in BRAFi-treated compared to control-treated Apc Min +/− mice (20.8 ± 9.2 v. 12.8 ± 0.1; p=0.016). No polyps were observed in BRAFi-treated wild type mice.
Conclusion
BRAF inhibitors may increase the risk of developing hyperplastic gastric polyps and colonic adenomatous polyps. Due to the risk of gastrointestinal bleeding and the possibility of malignant transformation, further studies are needed to determine whether or not endoscopic surveillance should be recommended for patients treated with BRAF inhibitors.
Introduction
BRAF inhibitors, including vemurafenib and dabrafenib, extend survival in Stage IV BRAFV600 mutant melanoma patients (1, 2), and produce a 45% 2 year survival rate (3, 4). While the median progression-free survival is approximately 7 months (1, 4, 5), in some cases patients have been treated for 3–5 years continuously with BRAF inhibitors. Early in the development of BRAF inhibitors, treatment-associated cutaneous squamous cell carcinoma (SCC) raised concerns regarding oncogenic risks. In phase II trials of BRAF inhibitors, 10–26% of patients developed cutaneous SCC or keratoacanthoma (4, 5). Molecular characterization of these SCCs found that some tumors harbored HRAS mutations, (6, 7). BRAF inhibition in HRAS mutant/BRAF wild type cutaneous SCC cells leads to paradoxical increase in mitogen active protein kinase (MAPK) signaling (8). Given the potential paradoxical activation of MAPK signaling especially in the presence of RAS mutations, there is concern that accelerated growth of other more life-threatening neoplasms is possible in patients treated with BRAF inhibitors. Reports of the progression of a preexisting NRAS mutant chronic myelomonocytic leukemia in a melanoma patient treated with vemurafenib (9), of the progression of a KRAS mutant colon cancer (10), and the development of a KRAS mutant pancreatic cancer(11) in two separate patients treated with combined BRAF and MEK inhibition (dabrafenib and trametinib) underscore this possibility. Furthermore, the concern BRAF inhibitor associated neoplasms is increased given the adjuvant studies of vemurafenib or dabrafenib in resected stage II and III melanoma.
Here we report several patients with advanced BRAFV600 mutant melanoma who were treated long-term with BRAF inhibitors, and were found to have intestinal polyps. Genetic characterization of these intestinal polyps revealed no mutations in MAPK pathway genes, however mutations in the APC (adenomatous polyposis coli) gene, commonly associated with colonic neoplasms, were detected in all cases. BRAF inhibitor treatment significantly increased the number of intestinal polyps in Apc Min +/− but not wild type mice, providing further evidence that BRAF inhibitors may promote the progression of existing intestinal polyps.
Methods
Patients and Lesion Samples
Patients participated in the phase I trial of vemurafenib (NCT00405587), the phase II study of vemurafenib (NCT00949702), the vemurafenib expanded access protocol (NCT01248936), the phase I trial of dabrafenib (NCT00880321) or received commercial drug. All patients had BRAFV600E metastatic melanoma and received either 720 mg or 960 mg of vemurafenib or 150 mg dabrafenib twice daily. Patients provided written informed consent for the molecular analysis of lesions obtained during treatment. Esophagogastroduodenoscopy (EGD) and colonoscopy were performed in the standard manner under conscious sedation.
Molecular analysis of tumor specimens
DNA was extracted from formalin fixed and paraffin embedded (FFPE) tissue sections of polyps and was sequenced by next generation sequencing (NGS) on the Ion Torrent (AmpliSeq™ Cancer Hotspot panel v.2, Life Technologies, Carlsbad, CA), and MiSeq (illumina TruSeq Cancer Hotspot panel, San Diego, CA) platforms. Library preparation for Ion Torrent sequencing of 50 genes was performed on a 318 chip using 10 to 15 ng of DNA and the Ion PGM sequencer (Life Technologies, Carlsbad, CA). Library preparation for MiSeq sequencing of 47 genes (MiSeq reagent kit v2; Illumina, San Diego, CA) using 250 ng of genomic DNA, on the MiSeq platform. Ion Torrent data were analyzed with the Ion Torrent Suite v. 3.4 (Life Technologies). MiSeq sequencing data was analyzed using an in-house analysis pipeline (12).
For more information including genes sequenced and methods to detect amplifications see Supplemental Methods.
Studies in Mice
Animal procedures were performed in accordance with guidelines from the local animal ethics committee. Two cohorts with 15 C57BL/6J-APCmin/+ mice (Jackson Laboratory, Maine) each were fed control chow or PLX4720 (the laboratory tool compound for vemurafenib)-infused (high dose) chow (13) for 28 days. Upon sacrifice, small intestine and colon were cut open length-wise, and polyp number and size were evaluated in a blinded fashion for proximal small intestine (PSI), distal small intestine (DSI), and colon using a Nikon SMZ645 microscope. Wild type C57Bl6 mice were treated with control and PLX chow for 6 months and sacrificed. All gastrointestinal tracts were formalin fixed, and paraffin embedded. Immunohistochemistry for phospho-ERK, and beta-catenin, was performed as previously described (14) using the following antibodies: Phospho-ERK (Cell Signaling 4370); beta-catenin (Cell Signaling 9562). Stained slides were evaluated for histological features of mouse intestinal polyps, the number of polyps, and nuclear phospho-ERK and β-catenin were scored in a blinded fashion. For the latter, a polyp with a well oriented section, permitting evaluation of luminal and basal epithelium and with the highest proportion of nuclear staining was given an estimated percentage of positive nuclei in the polyp.
Results
Characteristics of melanoma patients who developed polyps
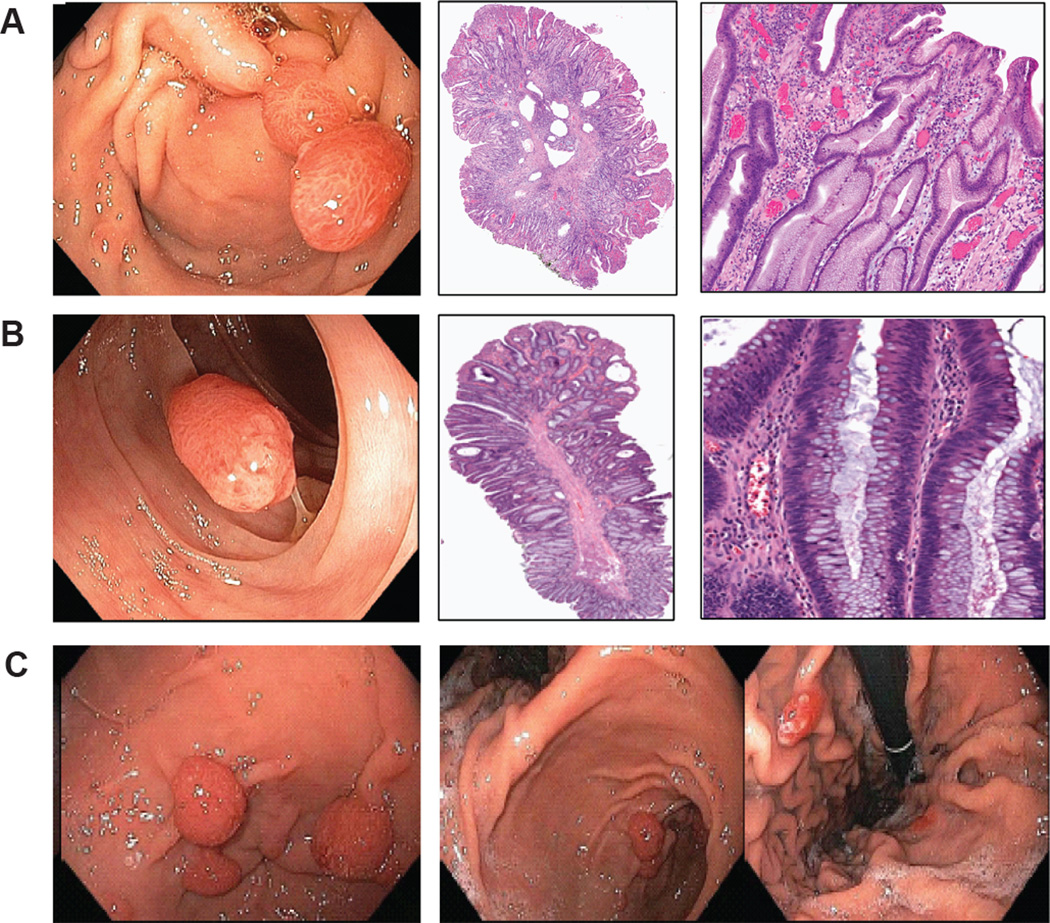
In the phase I trial of vemurafenib, 48 patients were treated with therapeutic doses of vemurafenib (15). . Four of the eight patients treated continuously for more than 2 years underwent EGD and colonoscopy (Table 1). The first patient (UPENN1) presented with hemodynamically unstable gastrointestinal bleeding 2.5 years after starting vemurafenib. He had a negative EGD and colonoscopy immediately prior to starting therapy. EGD demonstrated 6 gastric polyps (Figure 1A) and a duodenal ulcer (not shown). The polyps were resected and histological analysis demonstrated hyperplastic gastric polyps. Colonoscopy demonstrated 5 polyps. Histological examination demonstrated typical adenomatous polyps with features of tubular adenomas (Figure 1B). The patient developed anemia and fatigue 6 months after rechallenge with vemurafenib. Repeat EGD, colonoscopy, and histological analysis demonstrated recurrence of 6 bleeding hyperplastic gastric polyps (Figure 1C) and no recurrence of colonic polyps. Patient UPENN2 was asymptomatic when she underwent endoscopy and was found to have 7 colonic polyps (6 tubular adenomas and 1 hyperplastic polyp) after 2.25 years of vemurafenib (Supplemental Figure 1). She resumed vemurafenib treatment after resection of polyps and had repeat EGD and colonoscopies after one year, which showed no recurrent or new polyps. Patient MSKCC1 underwent colonoscopy due to persistent diarrhea after 2.76 years of vemurafenib and was found to have 10 colonic polyps (7 tubular adenomas and 3 hyperplastic polyps). Like patient UPENN1, this patient had a negative colonoscopy prior to enrollment on the phase I study, increasing the likelihood that the 10 polyps were associated with drug therapy. Patient PMAC1, who unlike the other patients was under the age of 40, was found to have no polyps despite 3.5 years of vemurafenib therapy.
Table 1.
Characteristics of Patients treated with BRAF inhibitor who underwent GI endoscopy
| Patient | Age, years |
BRAF inhibitor |
Time on BRAF inhibitor, years |
Family History of colon cancer |
Prior colonoscopy |
Number of colonic polyps |
Number of gastric polyps |
|---|---|---|---|---|---|---|---|
| Vemurafenib Phase I patients | |||||||
| UPENN 1 | 65 | VEM | 2.5 | None | Yes, no prior polyps | 5 TA | 5 HP |
| UPENN 2 | 61 | VEM | 2.25 | None | None | 6 TA, 1 HP | 0 |
| MSKCC1 | 51 | VEM | 2.67 | None | Yes, no prior polyps | 7 TA, 3HP | 0 |
| PMAC1 | 37 | VEM | 3.5 | None | None | 0 | 0 |
| Other BRAF inhibitor treated patients undergoing endoscopy | |||||||
| UPENN 3 | 55 | VEM | 0.32 | No | None | 0 | 6 HP |
| UPENN4 | 58 | VEM | 0.63 | Mother and maternal GM | None | 0 | 0 |
| MSKCC2 | 69 | VEM | 0.67 | Father | Yes, 1 adenoma 1.3 years prior to VEM | 1 TA | 0 |
| UPENN5 | 42 | VEM | 0.76 | None | None | 0 | 0 |
| MSKCC3 | 32 | VEM | 0.83 | None | None | 0 | 0 |
| UPENN6 | 52 | VEM | 1.88 | None | None | 0 | 0 |
| VAN 1 | 59 | VEM | 2 | None | None | 1 TA | 0 |
| VAN 2 | 64 | VEM | 2.5 | Mother | None | 3 TA | 0 |
| VAN3 | 48 | VEM | 2.75 | Mother | none | 1 HP | 0 |
| WMD1 | 61 | DAB | 5 | Brother | None | 8 TA | NA |
UPENN: University of Pennsylvania; MSKCC: Memorial Sloan Kettering Cancer Center: PMAC: Peter Macallum Cancer Center; VAN: Vanderbilt-Ingram Cancer Center; WMD1: Westmead Hospital; VEM: vemurafenib; DAB: dabrafenib; GM: Grandmother; NA: Not available; TA: tubular adenoma; HP: hyperplastic polyp
Figure 1. Multiple colonic adenomas and recurrent gastric polyps in a BRAF mutant melanoma patient treated with long term BRAF inhibitor.
Representative endoscopic or histological H&E images of (A) one of six gastric hyperplastic polyps; Magnification: 10X (middle); 100X (right) ; (B) one of 5 colonic tubular adenomas; Magnification: 10X (middle); 200X (right) (C) Recurrent gastric polyps 6 months after resection of original polyps and rechallenge with vemurafenib.
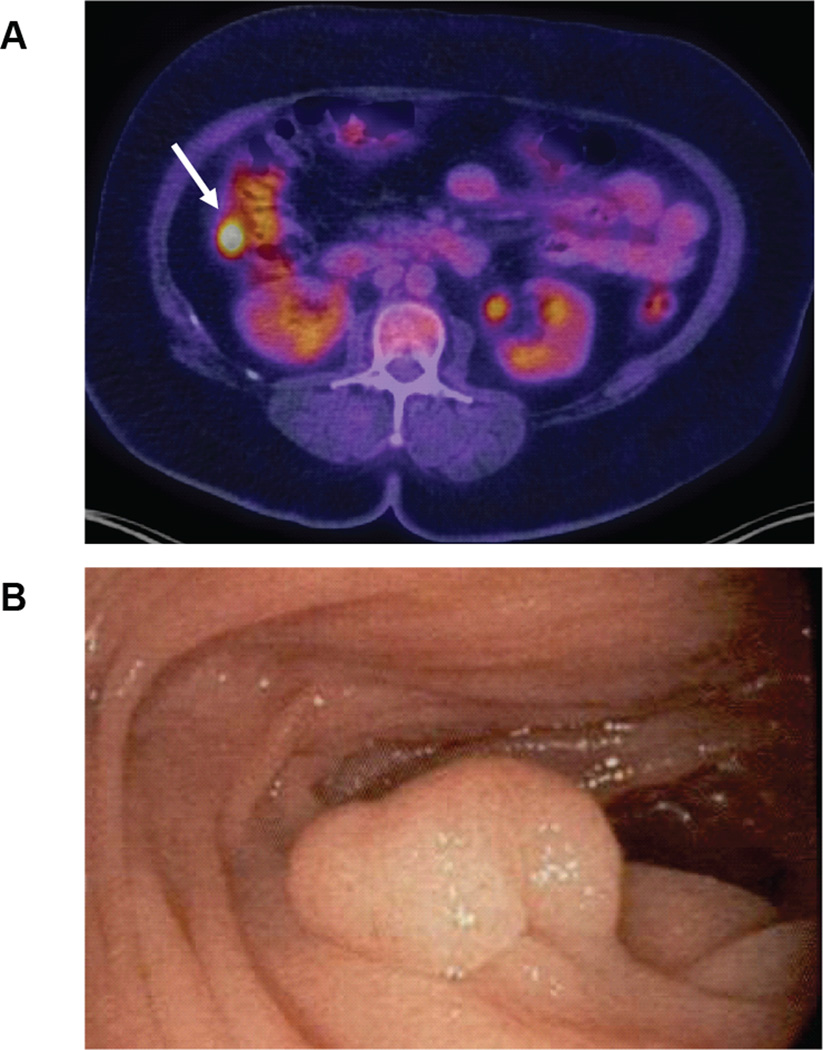
EGD and colonoscopy findings that were available for additional patients that were treated with BRAF inhibitors are presented in Table 1. The histology for nearly every colonic polyp was tubular adenoma. In most cases patients underwent endoscopy for either symptoms of dyspepsia, colitis, or for routine colon cancer screening. Patient WMD1 from Australia was treated with dabrafenib for 5 years, and on surveillance 18-fluorodeoxy (FDG) positron emission tomography/computed tomography (PET/CT) scan (Figure 2A), an FDG avid lesion in the transverse colon was found and interpreted as a potential melanoma metastases. Colonoscopy revealed 8 colonic polyps which were in some cases > 10 mm in size, including a 15 mm transverse colon polyp which corresponded to the FDG avid lesion on the PET/CT scan (Figure 2B). In this small cohort of patients, those who were > 40 years of age and who had been treated with BRAF inhibitors for > 2 years all were found to have colonic polyps. Once polyps reach larger proportions (>10 mm) they may be detected radiographically and may be mistaken for melanoma progression. However, a possible limiting factor for this observation is that 7/10 patients did not have a baseline colonoscopy prior to the start of BRAF inhibitor therapy and in two of the remaining three patients, the prior colonoscopy was more than one year prior to the start of BRAF inhibitor therapy. Additional confounding factors such as family history and advanced age may have played a significant role in these observations.
Figure 2. 18-Fluorodeoxyglocuse (FDG) avid colonic polyp mistaken for melanoma metastases.
(A) Cross sectional image of a FDG PET/CT obtained from a BRAF mutant melanoma patient with a complete response to dabrafenib after 5 years of therapy. The intense uptake of radiotracer in a colonic lesion (arrow) resulted in (B) a colonoscopy which demonstrated a 15 mm colonic tubular adenoma.
Genetic characterization of polyps
A stepwise accumulation of somatic mutations in the APC, KRAS, DCC, SMAD4, and TP53 genes has been described to explain the transformation of normal colonic epithelium to adenomatous polyps and adenocarcinoma (16). APC mutations are found in early adenomas, KRAS mutations in late adenomas and TP53 mutations in adenocarcinoma. There is very little known about the pathogenesis of hyperplastic gastric polyps, but TP53 and PIK3CA mutations may be present in dysplastic hyperplastic polyps (17, 18). We performed NGS on patient polyps using both the AmpliSeq and TruSeq cancer panels on the Ion Torrent (n=12), and Illumina (n=7) sequencing platforms respectively (Table 2), expecting to see somatic mutations in RAS genes. To increase yield in individual patients, DNA extracted from small polyps was combined for sequencing assays. In general there was agreement between the 2 platforms as far as mutated genes. In colonic polyps no mutations in MAPK pathway genes (e.g. RAS, RAF) were found, but all adenomas tested had APC mutations, with the most common mutation being truncation mutations in the beta-catenin binding domain (R1450X). We found 2/8 tubular adenomas harbored previously described mutations in the GSK3B (glycogen synthase kinase-3B) phosphorylation site of beta-catenin (19). One patient’s adenoma had a low allelic frequency of 2 pathogenic TP53 mutations more typical of adenocarcinomas rather than adenomas. In this sample there was no histological evidence of adenocarcinoma. No somatic mutations were found in gastric hyperplastic polyps, except a KRAS mutation, which given the low allele frequency may be a spurious result.
Table 2.
Disease associated somatic mutations in intestinal polyps from patients on BRAF inhibitors
| Patient | Sample | Gene | Mutation (AA Change) |
Allele Freq. (%) |
Total Coverage (fold) |
|---|---|---|---|---|---|
| Colonic Polyps | |||||
| MSKCC1 | Pooled TA |
APC APC |
P1441A T1448S |
15 30 |
473 518 |
| UPENN1 | Pooled TA |
CTNNB1 APC |
S45F G1106X |
49 8 |
6118 3905 |
| UPENN1 | Large TA | APC APC |
G1106X Q1338X |
27 29 |
4379 23882 |
| UPENN2 | Pooled TA |
TP53 TP53 TP53 APC |
R181H Y234C R175H 1450X |
5 4 5 30 |
1166 4463 4523 2485 |
| UPENN2 | Large TA | APC | 1450X | 41 | 2275 |
| VAN1 | Pooled TA |
CTNNB1 APC APC |
S45P P1441A P1422T |
54 9 25 |
1529 679 673 |
| VAN1 | Pooled TA | APC | P1450X | 16 | 11170 |
| VAN2 | Pooled TA | APC | Q1477X | 10 | 14079 |
| Gastric polyps | |||||
| UPENN1 | Gastric Polyp |
None | |||
| UPENN1 | Gastric Polyp |
None | |||
| UPENN1 | Gastric Polyp |
KRAS | G13C | 2 | 1480 |
| UPENN1 | Gastric Polyp |
None | |||
UPENN: University of Pennsylvania; MSKCC: Memorial Sloan Kettering Cancer Center; VAN: Vanderbilt-Ingram Cancer Center; TA: Tubular adenoma; AA: amino acid;
BRAF inhibition increases the number of polyps in Apc Min +/− mice
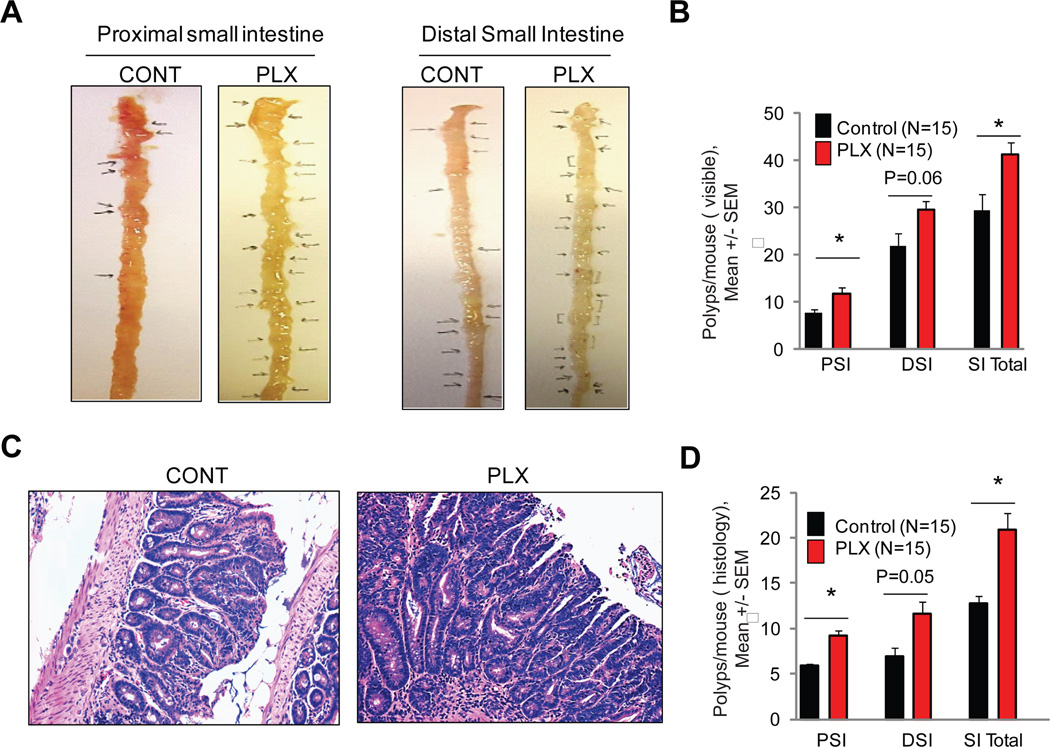
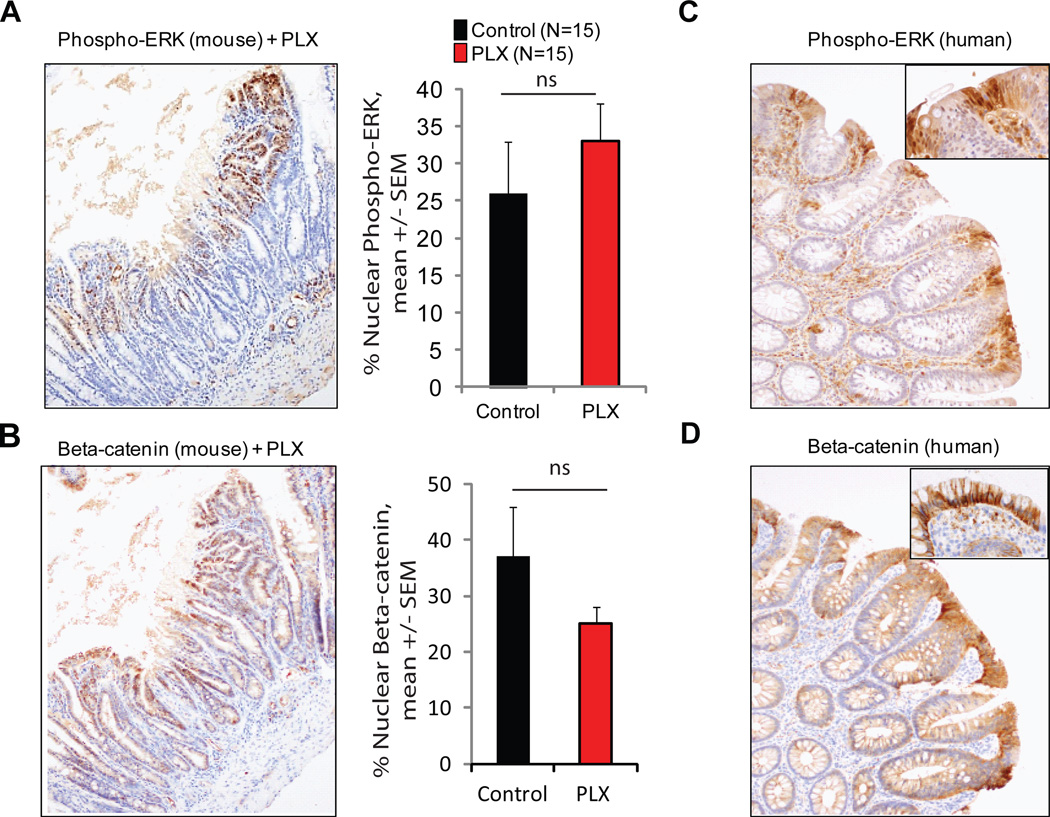
C57BL/6J-Apc Min +/− mice harbor a truncation in the Apc gene in the β-catenin binding motif, similar to the adenomas we found in melanoma patients treated with BRAF inhibitors. These mice develop adenomatous polyps that are localized to the small intestine rather than the colon, in contrast to humans where the APC mutant polyps are typically found in the colon. This genotype produces 100% penetrance leading to uniform anemia and death by 3 months (20). Examination of the GI tract of Apc Min +/− mice (N=15 each) fed control or PLX4720 ( BRAF inhibitor) chow for 28 days found no polyps in the stomach, few in the colon, with the majority residing in the small intestine (Figure 3A). The mean number of visible polyps in the small intestine was significantly increased in PLX4720 fed mice compared to control mice, in the proximal small intestine and in the total intestine (Figure 3B). There was a non-significant but consistent increase in polyps in the distal small intestine as well. No significant difference in the incidence of colon polyps was observed. Histological examination of polyps (Figure 3C) found the number of polyps was increased (Figure 3D) in mice fed PLX4720 chow compared to control chow in the proximal (mean ± SEM 9.2 ± 0.5 v. 5.8 ± 0.2 polyps, p=0.004) and total small intestine (20.8 ± 1.9 v.12.8 ± 0.1, p= 0.016). Invasive adenocarcinoma of the colon was found in 1/15 mice fed PLX4720 chow and 0/15 mice treated with control chow. It is known that Apc min +/− polyps occasionally progress to invasive adenocarcinoma without any other intervention in this model. To determine if PLX4720 can induce polyp formation in the absence of Apc mutations in mice, cohorts of C57Bl6 mice (WT mice) were fed control (N=3) or PLX4720 (N=4) chow for 6 months. No polyps were found in the intestines of mice fed PLX or control chow suggesting that that BRAF inhibition interacts specifically with the APC mutation to accelerate polyposis. Immunohistochemistry against phospho-ERK and beta-catenin in Apc Min +/− mouse polyps demonstrated nuclear phospho-ERK and beta-catenin that colocalized to the epithelial cells in the most proliferative segments of adenomatous tissue. No significant difference in the percentage of adenomatous epithelium with nuclear localization of phospho-ERK or beta-catenin in Apc Min +/− mice fed control chow versus PLX4720 chow were found (Figure 4A, B). Unlike the mouse tissue, human polyps obtained from BRAF mutant melanoma patients treated with BRAF inhibitors had almost no nuclear beta-catenin, and nuclear phospho-ERK was present in a patchy distribution (Figure 4C, D). Taken together these data indicate that that APC mutation can confer sensitivity to BRAF inhibitor-mediated acceleration of polyposis in the absence of RAS mutation.
Figure 3. Effects of BRAF inhibitor therapy on the number of intestinal polyps in APCMin/+ mice.
(A). Representative photographs of proximal and distal small intestine. Arrows indicate scored polyps. (B) Visual scoring of polyps in resected proximal (PSI) and distal (DSI) small intestines in 15 APC/min mice fed control chow compared to 15 APC/min mice fed chow containing the equivalent of 60 mg/kg/day PLX4720 (C) Representative hematoxylin and eosin stained sections of APCMin/+ polyps in mice fed control (CONT) and PLX4720 (PLX) chow; Original magnification 100X. (D) Histological scoring of these GI tracts. *p<0.05.
Figure 4. Immunohistochemistry against phospho-ERK and beta-catenin in mouse and human polyps.
(A-B) Representative images and % nuclear staining in Apc Min +/− mouse polyps (A) phospho-ERK; (B) Beta –catenin; ns: not significant. (C-D) Representative images of human tubular adenoma from a BRAF mutant melanoma patient treated with vemurafenib (C) phospho-ERK (D) Beta-catenin . Original magnification 100X (A-B); 200X (C-D).
Discussion
Here we describe the first report of a MAPK targeted therapy potentially accelerating neoplastic growth in benign gastric epithelium, and in APC mutant colonic tissue lacking MAPK mutations. One patient had a low frequency TP53 mutation present in her polyps, suggesting that the eventual malignant transformation of these adenomas is possible. Since BRAF inhibitor-associated colonic adenomas undetectable by CT scanning could potentially progress to frank colon adenocarcinoma if left untreated, we view this finding as a significant and unanticipated adverse event. It is important to note that we did not find any histology consistent with adenocarcinoma in any of the cases described in this report and clinical trials of single agent BRAF inhibitor have not reported any cases of adenocarcinoma during the specified followup period. The high number of polyps and the recurrence of the gastric polyps in one patient that was rechallenged with vemurafenib provided some clinical evidence of a potential association. Despite this observation, colonic adenomas are common in patients >50 years of age. One study encompassing 4 million patients found that detection rates of adenomas especially non-advanced adenomas (<5mm) have increased significantly over the years 2003–2012 to >30% in men and >20% in women in 2012 (21). The projected annual transition rates from advanced adenomas to CRC strongly increase with age from 2.6% in 55–65 year old and 5.1–5.6% in >80 year old men or women (22). Other studies which include both advanced and non-advanced adenomas, have estimated 5% of adenomas progress to colon cancer over 7–10 years (23). While multiple adenomas are common in the general population, the incidence of ≥5 adenomas was estimated in one study to be 0.4%, and the risk of progression to colon cancer if more than 5 polyps were observed was 24% in this study (24). While this is an older study, in a more modern series of 889 patients, 5 or more polyps were found in 4% of patients, and multiple adenomatous polyps, or a polyp > 10 mm significantly increased the odds of finding concurrent high grade dysplasia or adenocarcinoma (25). Due to the limitation of followup times in any endoscopy study, the precise risk and the rate of transformation from colonic adenoma to adenocarcinoma is difficult to ascertain. In our study 4/14 patients treated with BRAF inhibitor had 5 or more colonic polyps, further supporting the potential association between BRAF inhibitors and polyp development. It is clear that the patients described here are a highly selected population and the rate of multiple polyps could be much lower in a larger population.
Gastric polyps are found in only 6% of all endoscopies. Multiple gastric polyps are found in 1.5% of all endoscopies (26). While PPI therapy is a risk factor for developing gastric polyps, 7% of PPI patients develop gastric polyps after an average of 32 months , and 90% of PPI-associated polyps are fundic gland polyps not hyperplastic polyps like those observed here (27). While BRAF inhibitor associated gastric polyps may have a low malignant potential, our experience indicates they can be associated with clinically significant gastrointestinal bleeding.
In practice common hyperplastic gastric polyps often don’t spontaneously bleed. And those that are found are often left in place. The added burden and risk of screening for and resecting these lesions in patients treated with BRAF inhibitors with no evidence of bleeding needs to be considered carefully against the prognosis of their malignancy.
The next generation sequencing platforms used in this study were able to rule out most deleterious mutations in MAPK genes such as BRAF, CRAF and NRAS, but this platform is not designed to definitively detect functional amplifications of MAPK genes that may in fact interact with APC mutations. Nevertheless, the finding of APC mutations in the absence of RAS or other MAPK mutations provided the opportunity to test the causative association between BRAF inhibitors and polyp formation using wild type mice and the Apc Min +/− mouse model. The lack of polyp formation in wild type mice treated for 6 months with BRAF inhibitor suggests BRAF inhibitors do not initiate polyp development but could increase the rate of polyp growth in epithelium that has lost APC function. Although a high percentage of neoplastic cells with nuclear localization of phospho-ERK were not observed in adenomas from either patients or mice treated with BRAF inhibitors, it is possible that paradoxical MAPK activation took place at earlier time points during therapy. The lack of a high percentage of nuclear beta catenin is consistent with a previous report that indicates that APC loss by itself is insufficient for beta-catenin nuclear localization, which requires concurrent MAPK activation (28). Additional laboratory studies in genetically engineered cell lines will be necessary to define the mechanistic interaction between MAPK and beta catenin signaling in this context.
Additional clinical studies are ongoing to determine the rate of polyp formation and the demographics of patients most likely to develop these polyps. As the combination of BRAF and MEK inhibition was found to be superior to BRAF inhibitor alone (29) and is becoming more widely adopted as a standard of care in melanoma, studies are needed to determine if dual inhibition of the MAPK pathway in this manner would have any effect on the likelihood of developing colonic and/or gastric polyps. Importantly, in preclinical models of HRAS mutant BRAF WT cutaneous squamous carcinoma, BRAFi-associated paradoxical MAPK activation and growth of squamous tumors could be blunted by adding a MEK inhibitor to the BRAF inhibitor (7). This finding was recapitulated in the randomized phase III trial of dabrafenib and trametinib versus dabrafenib alone, where the incidence of squamous cell carcinoma was significantly decreased in the combination arm that included the MEK inhibitor(30). Before these studies are complete it is premature to recommend widespread serial colonoscopy in patients treated with BRAF inhibitors, especially since no cases of frank adenocarcinoma were observed in this cohort of patients. However, given the risk of gastrointestinal bleeding, judicious surveillance in asymptomatic patients over the age of 40 with a family history of colon cancer, or a personal history of polyps and more than two years of BRAF inhibitor therapy may be considered reasonable.
Supplementary Material
Statement of Translational Relevance.
BRAF inhibitors can promote the growth of cutaneous squamous cell carcinoma, and possibly other Ras-driven neoplasms. We describe a high incidence of multiple gastrointestinal polyps in patients treated with BRAF inhibitors for >2 years. Next generation sequencing determined that these colonic polyps harbored adenomatous polyposis coli (APC) mutations only. BRAF inhibition significantly increased the number of polyps in APC Min +/− mice. Gastrointestinal polyps are often undetected on cancer imaging studies, and pose a risk of bleeding and transformation into adenocarcinoma. For these reasons endoscopy needs to be considered in patients undergoing BRAF inhibitor therapy. No cases of invasive cancer were observed, and the development of polyps did not impact the effective use of BRAF inhibitors in these patients. This is the first example in which neoplastic growth potentially associated with BRAF inhibitors was observed in the absence of a MAPK gene mutation and in the presence of APC mutations.
Acknowledgements
Funding: Funding for this work was provided in part by Genentech through a sponsored research agreement with University of Pennsylvania (RKA). Additional resources used included the Molecular Pathology and Imaging, Molecular Biology and Gene Expression, and Transgenic and Chimeric Mouse Cores of the University of Pennsylvania Molecular Studies in Digestive and Liver Diseases Center (NIH/NIDDK P30 DK050306). DNA sequencing was performed using the Abramson Cancer Center Genomics Core services (P30 CA016520).
Footnotes
Conflict of Interest Disclosure: RKA received grant funding from Genentech for this project. GRG is an employee of Genentech. There are no other conflicts of interest to disclose.
References
- 1.Chapman PB, Hauschild A, Robert C, Haanen JB, Ascierto P, Larkin J, et al. Improved survival with vemurafenib in melanoma with BRAF V600E mutation. N Engl J Med. 2011;364:2507–2516. doi: 10.1056/NEJMoa1103782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hauschild A, Grob JJ, Demidov LV, Jouary T, Gutzmer R, Millward M, et al. Dabrafenib in BRAF-mutated metastatic melanoma: a multicentre, open-label, phase 3 randomised controlled trial. Lancet. 2012;380:358–365. doi: 10.1016/S0140-6736(12)60868-X. [DOI] [PubMed] [Google Scholar]
- 3.Puzanov I AR, McArthur GA, Flaherty KT,Chapman PB,Sosman JA, Ribas A, Shackleton M, Hwug PCB, Nolop K, Lin PS, Kim KB. Long-term outcome in BRAFV600E melanoma patients treated with vemurafenib: Patterns of disease progression and clinical management of limited progression. European Journal of Cancer. 2015 May 13; doi: 10.1016/j.ejca.2015.04.010. pii: S0959-8049(15)00346-9. doi:10.1016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sosman JA, Kim KB, Schuchter L, Gonzalez R, Pavlick AC, Weber JS, et al. Survival in BRAF V600-mutant advanced melanoma treated with vemurafenib. N Engl J Med. 2012;366:707–714. doi: 10.1056/NEJMoa1112302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ascierto PA, Minor D, Ribas A, Lebbe C, O'Hagan A, Arya N, et al. Phase II trial (BREAK-2) of the BRAF inhibitor dabrafenib (GSK2118436) in patients with metastatic melanoma. J Clin Oncol. 2013;31:3205–3211. doi: 10.1200/JCO.2013.49.8691. [DOI] [PubMed] [Google Scholar]
- 6.Oberholzer PA, Kee D, Dziunycz P, Sucker A, Kamsukom N, Jones R, et al. RAS mutations are associated with the development of cutaneous squamous cell tumors in patients treated with RAF inhibitors. J Clin Oncol. 2012;30:316–321. doi: 10.1200/JCO.2011.36.7680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Su F, Viros A, Milagre C, Trunzer K, Bollag G, Spleiss O, et al. RAS mutations in cutaneous squamous-cell carcinomas in patients treated with BRAF inhibitors. N Engl J Med. 2012;366:207–215. doi: 10.1056/NEJMoa1105358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Weeraratna AT. RAF around the edges--the paradox of BRAF inhibitors. N Engl J Med. 2012;366:271–273. doi: 10.1056/NEJMe1111636. [DOI] [PubMed] [Google Scholar]
- 9.Callahan MK, Rampal R, Harding JJ, Klimek VM, Chung YR, Merghoub T, et al. Progression of RAS-mutant leukemia during RAF inhibitor treatment. N Engl J Med. 2012;367:2316–2321. doi: 10.1056/NEJMoa1208958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Andrews MC, Behren A, Chionh F, Mariadason J, Vella LJ, Do H, et al. BRAF inhibitor-driven tumor proliferation in a KRAS-mutated colon carcinoma is not overcome by MEK1/2 inhibition. J Clin Oncol. 2013;31:e448–e451. doi: 10.1200/JCO.2013.50.4118. [DOI] [PubMed] [Google Scholar]
- 11.Carlino MS, Kwan V, Miller DK, Saunders CA, Yip D, Nagrial AM, et al. New RAS-Mutant Pancreatic Adenocarcinoma With Combined BRAF and MEK Inhibition for Metastatic Melanoma. 2015 Apr 10;33(11) doi: 10.1200/JCO.2013.51.5783. [DOI] [PubMed] [Google Scholar]
- 12.Daber R, Sukhadia S, Morrissette JJ. Understanding the limitations of next generation sequencing informatics, an approach to clinical pipeline validation using artificial data sets. Cancer Genet. 2013;206:441–448. doi: 10.1016/j.cancergen.2013.11.005. [DOI] [PubMed] [Google Scholar]
- 13.Ma XH, Piao SF, Dey S, McAfee Q, Karakousis G, Villanueva J, et al. Targeting ER stress-induced autophagy overcomes BRAF inhibitor resistance in melanoma. J Clin Invest. 2014;124:1406–1417. doi: 10.1172/JCI70454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Abraham RM, Karakousis G, Acs G, Ziober AF, Cerroni L, Mihm MC, Jr, et al. Lymphatic invasion predicts aggressive behavior in melanocytic tumors of uncertain malignant potential (MELTUMP) The American journal of surgical pathology. 2013;37:669–675. doi: 10.1097/PAS.0b013e318288ff47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Flaherty KT, Puzanov I, Kim KB, Ribas A, McArthur GA, Sosman JA, et al. Inhibition of mutated, activated BRAF in metastatic melanoma. N Engl J Med. 2010;363:809–819. doi: 10.1056/NEJMoa1002011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Rustgi AK. The genetics of hereditary colon cancer. Genes Dev. 2007;21:2525–2538. doi: 10.1101/gad.1593107. [DOI] [PubMed] [Google Scholar]
- 17.Jain R, Chetty R. Gastric hyperplastic polyps: a review. Dig Dis Sci. 2009;54:1839–1846. doi: 10.1007/s10620-008-0572-8. [DOI] [PubMed] [Google Scholar]
- 18.Murakami K, Mitomi H, Yamashita K, Tanabe S, Saigenji K, Okayasu I. p53, but not c-Ki-ras, mutation and down-regulation of p21WAF1/CIP1 and cyclin D1 are associated with malignant transformation in gastric hyperplastic polyps. Am J Clin Pathol. 2001;115:224–234. doi: 10.1309/VLF5-UCNH-XQM2-X410. [DOI] [PubMed] [Google Scholar]
- 19.Samowitz WS, Powers MD, Spirio LN, Nollet F, van Roy F, Slattery ML. Beta-catenin mutations are more frequent in small colorectal adenomas than in larger adenomas and invasive carcinomas. Cancer Res. 1999;59:1442–1444. [PubMed] [Google Scholar]
- 20.Jacoby RF, Marshall DJ, Newton MA, Novakovic K, Tutsch K, Cole CE, et al. Chemoprevention of spontaneous intestinal adenomas in the Apc Min mouse model by the nonsteroidal anti-inflammatory drug piroxicam. Cancer Res. 1996;56:710–714. [PubMed] [Google Scholar]
- 21.Brenner H, Altenhofen L, Kretschmann J, Rosch T, Pox C, Stock C, et al. Trends in Adenoma Detection Rates During the First 10 y of the German Screening Colonoscopy Program. Gastroenterology. 2015 doi: 10.1053/j.gastro.2015.04.012. [DOI] [PubMed] [Google Scholar]
- 22.Brenner H, Hoffmeister M, Stegmaier C, Brenner G, Altenhofen L, Haug U. Risk of progression of advanced adenomas to colorectal cancer by age and sex: estimates based on 840,149 screening colonoscopies. Gut. 2007;56:1585–1589. doi: 10.1136/gut.2007.122739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Heitman SJ, Ronksley PE, Hilsden RJ, Manns BJ, Rostom A, Hemmelgarn BR. Prevalence of adenomas and colorectal cancer in average risk individuals: a systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2009;7:1272–1278. doi: 10.1016/j.cgh.2009.05.032. [DOI] [PubMed] [Google Scholar]
- 24.Schuman BM, Simsek H, Lyons RC. The association of multiple colonic adenomatous polyps with cancer of the colon. Am J Gastroenterol. 1990;85:846–849. [PubMed] [Google Scholar]
- 25.Kurome M, Kato J, Nawa T, Fujimoto T, Yamamoto H, Shiode J, et al. Risk factors for high-grade dysplasia or carcinoma in colorectal adenoma cases treated with endoscopic polypectomy. Eur J Gastroenterol Hepatol. 2008;20:111–117. doi: 10.1097/MEG.0b013e3282f1cbef. [DOI] [PubMed] [Google Scholar]
- 26.Carmack SW, Genta RM, Schuler CM, Saboorian MH. The current spectrum of gastric polyps: a 1-year national study of over 120,000 patients. Am J Gastroenterol. 2009;104:1524–1532. doi: 10.1038/ajg.2009.139. [DOI] [PubMed] [Google Scholar]
- 27.el-Zimaity HM, Jackson FW, Graham DY. Fundic gland polyps developing during omeprazole therapy. Am J Gastroenterol. 1997;92:1858–1860. [PubMed] [Google Scholar]
- 28.Phelps RA, Chidester S, Dehghanizadeh S, Phelps J, Sandoval IT, Rai K, et al. A two-step model for colon adenoma initiation and progression caused by APC loss. Cell. 2009;137:623–634. doi: 10.1016/j.cell.2009.02.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Flaherty KT, Infante JR, Daud A, Gonzalez R, Kefford RF, Sosman J, et al. Combined BRAF and MEK inhibition in melanoma with BRAF V600 mutations. N Engl J Med. 2012;367:1694–1703. doi: 10.1056/NEJMoa1210093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Robert C, Karaszewska B, Schachter J, Rutkowski P, Mackiewicz A, Stroiakovski D, et al. Improved overall survival in melanoma with combined dabrafenib and trametinib. N Engl J Med. 2015;372:30–39. doi: 10.1056/NEJMoa1412690. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.