Abstract
Background
Rotavirus gastroenteritis is a major cause of mortality among children <2 years of age. Disease burden data are important for introducing and sustaining new rotavirus vaccines in immunization programs.
Methods
We analyzed population-based infectious disease surveillance data from 2007 to 2010 from Kenyan sites in rural and urban slum areas. Stool specimens were collected from patients of all ages presenting to study clinics with diarrheal disease and tested for rotavirus by enzyme immunoassay. Incidence rates were adjusted using data on healthcare utilization (from biweekly home visits) and proportion of stools collected at study clinics from patients meeting case definitions.
Results
Rotavirus was detected in 285 (9.0%) of 3174 stools tested, including 122 (11.9%) from children <5 years of age and 162 (7.6%) from participants ≥5 years of age. Adjusted incidence rates for infants were 13,419 and 12,135 per 100,000 person-years of observation in rural and urban areas, respectively. Adjusted incidence rates were high in adults across age ranges. The rates suggest that annually, among children <5 years of age, there are >54,500 cases of rotavirus-associated gastroenteritis in rural Nyanza Province and >16,750 cases in Nairobi urban slums.
Conclusions
Community-based surveillance in urban and rural Kenya suggests that rotavirus plays an important role as a cause of acute gastroenteritis in adults, as well as in children. In addition to substantially preventing illness and complications from diarrheal disease in children, rotavirus infant immunization has the potential of indirectly preventing diarrheal disease in older children and adults, assuming children are the predominant sources of transmission.
Keywords: rotavirus, gastroenteritis, diarrhea, population-based incidence, rates, children, adults, Kenya, urban, rural, vaccine, impact
Interest in precise data on burden of rotavirus gastroenteritis has grown due to the recent availability of safe and efficacious new generation rotavirus vaccines.1,2 In addition to direct protection, data from high and middle income countries that have introduced rotavirus vaccines suggest that immunization of infants may provide indirect protection by reducing transmission of rotavirus to older children and adults.3,4 While data are available on rotavirus disease burden in children <5 years of age from multiple settings,5 most of these data are hospital-based, which does not provide a representative and accurate disease incidence data burden in areas where health care utilization is low. In addition, there is limited information on incidence rates for rotavirus-associated diarrhea in older children and in adults, particularly in developing countries.6,7
The Kenya Medical Research Institute (KEMRI) and the US Centers for Disease Control and Prevention (CDC) collaborate on population-based surveillance for infectious disease syndromes and their etiologies in a rural setting and in an urban slum in Kenya; adults in both areas have high (15–20%) HIV seroprevalence.8 The surveillance platform has provided opportunities to assess the burden of a variety of syndromes. We assessed incidence and characterized the epidemiology of rotavirus-associated gastroenteritis by age group in these 2 locations. These data may serve as a baseline to monitor impact of introduction (anticipated in 2014) of rotavirus vaccines, including in older children and adults.
METHODS
Study Sites
KEMRI and CDC have collaborated on population-based infectious disease surveillance (PBIDS) since late 2005 in 2 sites in Kenya: Lwak (within Rarieda District in Siaya County), a rural location in Nyanza province in Western Kenya (Fig. 1), and an urban setting in Kibera, a slum in Nairobi.9
FIGURE 1.
Map of Kenya with insets showing location of study sites.
Surveillance Procedures
The Kenya PBIDS system for diarrheal disease, febrile illness, pneumonia and jaundice surveillance has been described elsewhere [9]. Briefly, trained community interviewers visited participants every 2 weeks, collecting data on illnesses and deaths during the 2 weeks before visit; after September 1, 2009, household visits were increased in frequency to every week to allow for more intensive data collection during the pandemic of influenza A H1N1.10 Surveillance participants have free access to a well-staffed and supplied field clinic centrally located within 5 km and 1 km of all residences in Lwak and Kibera, respectively.
PBIDS is carried out within 2 of 12 villages in Kibera: Gatwikera and Soweto West. The average population of the surveillance area is 28,500 in a 0.37 km2 area (population density = 77,000 persons/km2). Kibera lacks adequate sanitation facilities; human and animal wastes drain into open sewage runoff. The Lwak surveillance area is 100 km2 with average population of 25,000 (325 people/km2), and the area is holoendemic for malaria. The 2 areas are 340 km (6–8 travel hours) apart by road. Kibera is at approximately 1600 m altitude and the elevation of Lwak is 1000 m. Surveillance participants included all people living in consenting households within the surveillance areas for a minimum of 4 months (8 consecutive biweekly home visits) between January 1, 2007, and December 31, 2010 (the study period).
For acute diarrheal disease surveillance, we collected stool specimens from consenting patients presenting to the study clinics during the study period. We sampled patients presenting with diarrhea, defined as ≥3 loose stools within a 24-hour period. To minimize burden on the laboratory, we sampled no more than 5 children and 5 adults a day (from each site) who had diarrhea without any sign of dehydration or dysentery. However, there were no restrictions on the number of patients sampled who presented with diarrhea and had signs of dehydration (defined as having ≥1 of the following signs and symptoms: drinking eagerly or unable to drink or breast-feed, vomits everything, slow skin pinch return ≥2 seconds, irritability, sunken eyes, lethargy or unconsciousness) or had evidence of dysentery (defined as reported or visible blood in at least 1 stool during the 24 hours before clinic visit).
A case of rotavirus gastroenteritis was defined as a patient with a diarrheal illness who had rotavirus detected in a stool specimen. To assess the proportion of pathogens detected from patients with diarrhea which was likely associated with illness, we also collected stool specimens from “control” residents of the surveillance area presenting to the clinic without fever, respiratory symptoms or diarrhea during the past 2 weeks, as previously described.11,12 Each month, we attempted to enroll a target number of controls for a variety of age groups based on a frequency distribution of ages of patients who presented to the clinics with respiratory or diarrheal disease and for adults, HIV status. We compared proportions of cases and noncases by age group who had rotavirus detected in stool. Odds ratios (OR) and 95% confidence intervals (CIs) were calculated using unconditional logistic regression, adjusting for age group. We used the OR to calculate pathogen-attributable fractions (PAF), which estimate the proportion of cases positive for rotavirus in which it is the likely cause of illness.11–13 PAF were calculated as (OR-1)/OR.
Diagnostics
We provided consenting participants meeting the stool collection criteria with a labeled stool container and instructions on how to sample their stool. Stool specimens were collected from patients’ homes within 4 hours of clinic visit, if they were unable to produce stool at the clinic. A sample of whole stool underwent routine enteric microbiology testing14; in addition, portions were aliquoted into 2 vials, 1 for rotavirus testing and the other was archived. In Kibera, stool specimens were kept at −20°C at the field laboratory and were transported daily to the KEMRI/CDC Nairobi laboratory, where they were stored at −80°C until testing. Specimens were batch shipped to the KEMRI/CDC Enterics laboratory in Kisumu, Kenya in a dry shipper (at −80°C) for rotavirus testing. Specimens collected in Lwak were transferred to the KEMRI/CDC Kisumu laboratory on a daily basis with cool packs and stored at −80°C until testing.
At the KEMRI/CDC Enterics laboratory in Kisumu, specimen identification numbers were entered into a log book and into a computerized database, and specimens were stored at 2–8°C. Specimens were batch tested weekly using a commercially available enzyme immunoassay kit (Rotaclone, Meridian Premier, Charlotte, NC) for detection of rotavirus antigen in the human fecal specimens, as previously described.15
Analysis
Data were analyzed using SAS, version 9.1 (Cary, NC). We calculated crude incidence rates as the number of rotavirus gastroenteritis cases among PBIDS participants per 100,000 participant person-years of observation (PYO). PYO were calculated by totaling person-days for all people who met the residence requirement during each biweekly round and dividing the total number of person-days by 365.25. A participant who moved away from the surveillance area for 4 consecutive months was not counted in the denominator (for calculation of PYO) during or after those 4 months. If that person moved back into the surveillance area, he/she would not be counted in denominator (or numerator) until documented to be living in the surveillance area for 4 months and reconsented to participate in the study.
We used 2 incidence rate adjustments to account for the possibility of cases missed due to gaps in case detection and diagnosis, as previously described.16 The first (adjustment 1) was based on patients who visited the clinic and did not have a stool specimen collection done despite meeting the criteria for stool collection. The usual reason given for not having a stool specimen obtained was that the patient was not able or willing to produce a stool specimen at the clinic or during the required time after clinic visit (see above). For this extrapolation, we divided the number of participants meeting stool-sampling criteria who had a stool specimen obtained by the total number of all surveillance participants presenting to the clinic meeting stool specimen collection criteria. The resulting proportion was divided into the crude incidence rate to account for those meeting stool specimen collection criteria who were not cultured.
A second adjustment (adjustment 2) was based on dividing the number of patients who had a diarrheal illness (based on data from the home visits) and who visited the study clinic by the total number of patients who had diarrheal illness and visited any clinic (including nonstudy clinics where information and specimens are not collected for this surveillance)—thus, the denominator would include patients who felt they were sick enough to visit a clinic but did not have the opportunity for stool specimen collection. This figure was divided into adjustment 1 to yield adjustment 2. This adjusted rate was then multiplied by the PAF (see above) to achieve the final adjusted rate.
Because of the likelihood that healthcare-seeking and stool-sampling patterns vary by type of diarrheal syndrome, adjustments 1 and 2 and PAF correction were initially done separately for diarrhea without dehydration, diarrhea with evidence of dehydration and dysentery (blood in stool). The adjusted rates for each diarrheal syndrome by age group were then summed to provide the final adjusted rates for rotavirus diarrhea by age group for Kibera and Lwak. CIs for the crude incidence rates were calculated using the Exact method.17 CIs for the final adjusted incidence rates were calculated using the same adjustments; that is, the lower and upper ends of the exact CIs were divided by adjustment 1 and then by adjustment 2, and then those figures were multiplied by the PAF.
We used incidence rates from this study to estimate the annual number of cases of rotavirus gastroenteritis occurring among people within the same age groups based on 2009 census data in rural Nyanza Province and in urban Nairobi.18 This project was approved by the Ethical Review Committee of KEMRI (Protocol #932 and #1899) and Institutional Review Board of CDC (Protocol #4566).
RESULTS
We enrolled 48,337 and 35,063 persons in Kibera and Lwak in the study between 2007 and 2010, who contributed 110,592 and 102,217 PYO, respectively. The overall crude incidence rate of rotavirus gastroenteritis was 153/100,000 PYO (Kibera) and 113/100,000 PYO (Lwak). In Lwak, crude incidence was highest among children <12 months of age (671/100,000 PYO); in Kibera, toddlers, 12–23 months of age, had the highest rate (504/100,000 PYO; Table 1 and Table 2). In Kibera, rotavirus was detected in 170 (7.8%) of 2166 stools from patients with diarrhea compared with 6 (2.1%) of 289 control-patients (PAF = 0.75). In Lwak, rotavirus was detected in 115 (11.4%) of 1008 stools collected from patients with diarrhea, compared with 9 (1%) of 737 control-patients (PAF = 0.9). Thus, rural Lwak residents had an increased likelihood that an episode of acute gastroenteritis was associated with detection of rotavirus in stool when compared with urban Kibera residents (relative risk = 1.46; 95% CI = 1.16–1.83) and given differences in PAF, a greater likelihood that rotavirus was the cause of the episode of gastroenteritis. The difference in rate of detection of rotavirus in stools from control residents of Kibera when compared with control residents in Lwak was not statistically significant. Mild diarrhea accounted for many incidences in both sites; however, in Lwak, a greater proportion of rotavirus gastroenteritis was associated with dehydration than in Kibera for all age groups, especially so for children <24 months of age (Table 3 and Table 4).
TABLE 1.
Detection of Rotavirus in Stool From Cases and Controls by Age Group, Kibera (Urban Slum Site)
| Cases
|
Controls
|
PAF | |||
|---|---|---|---|---|---|
| Number of Stool Specimens Processed | Rotavirus Detected n (%) | Number of Stool Specimens Processed | Rotavirus Detected n (%) | ||
| Age | |||||
| <12 months | 166 | 22 (13.3) | 25 | 1 (4) | 0.73 |
| 12–23 months | 184 | 28 (15.2) | 8 | 0 (0) | 1.00* |
| 24–59 months | 412 | 23 (5.6) | 20 | 0 (0) | 1.00* |
| <5 years | 762 | 73 (9.6) | 53 | 1 (1.9) | 0.82 |
| 5–9 years | 229 | 12 (5.2) | 30 | 1 (3.3) | 0.38 |
| 10–17 years | 260 | 18 (6.9) | 53 | 1 (1.9) | 0.74 |
| 18–34 years | 585 | 43 (7.4) | 101 | 1 (0.9) | 0.87 |
| 35–49 years | 259 | 21 (8.1) | 46 | 2 (4.3) | 0.48 |
| 50+ years | 58 | 2 (3.4) | 6 | 0 (0) | 1.00* |
| ≥5 years | 1391 | 96 (6.9) | 236 | 5 (2.1) | 0.72 |
| Total | 2166 | 170 (7.8)† | 289 | 6 (2.1) | 0.75 |
| Sex | |||||
| Male | 1053 | 77 (7.3) | 139 | 3 (2.2) | 0.71 |
| Female | 1113 | 92 (8.3) | 151 | 2 (1.3) | 0.85 |
| Year | |||||
| 2007 | 200 | 20 (10.0) | 0 | 0 (0.0) | 1.00* |
| 2008 | 435 | 18 (4.1) | 29 | 3 (10.3) | 1.69 |
| 2009 | 730 | 62 (8.5) | 162 | 1 (0.6) | 0.94 |
| 2010 | 801 | 70 (8.7) | 99 | 2 (2.0) | 0.79 |
PAF not calculable, thus otherwise specified as 1.
Age information was not available for 13 patients, including 1 patient with rotavirus detected in stool.
TABLE 2.
Detection of Rotavirus in Stool From Cases and Controls by Age Group, Lwak (Rural Site)
| Cases
|
Controls
|
PAF | |||
|---|---|---|---|---|---|
| Number of Stool specimens Processed | Rotavirus Detected n (%) | Number of Stool Specimens Processed | Rotavirus Detected n (%) | ||
| Age | |||||
| <12 months | 107 | 23 (21.5) | 48 | 1 (2.1) | 0.92 |
| 12–23 months | 85 | 20 (23.5) | 52 | 1 (1.9) | 0.94 |
| 24–59 months | 75 | 6 (8) | 87 | 1 (1.1) | 0.87 |
| <5 years | 267 | 49 (18.4) | 187 | 3 (1.6) | 0.93 |
| 5–9 years | 53 | 3 (5.7) | 118 | 1 (0.8) | 0.86 |
| 10–17 years | 122 | 11 (9) | 117 | 0 (0) | 1.00* |
| 18–34 years | 252 | 31 (12.3) | 152 | 3 (2) | 0.86 |
| 35–49 years | 165 | 11 (6.7) | 98 | 1 (1) | 0.86 |
| 50+ years | 149 | 10 (6.7) | 65 | 1 (1.5) | 0.78 |
| ≥5 years | 741 | 66 (8.9) | 550 | 6 (1.1) | 0.89 |
| Total | 1008 | 115 (11.4) | 737 | 9 (1.2) | 0.90 |
| Sex | |||||
| Male | 454 | 48 (10.6) | 262 | 3 (1.1) | 0.90 |
| Female | 554 | 67 (12.1) | 475 | 6 (1.3) | 0.91 |
| Year | |||||
| 2007 | 276 | 39 (14.1) | 0 | 0 (−) | 1.00* |
| 2008 | 251 | 22 (8.8) | 69 | 2 (2.9) | 0.69 |
| 2009 | 267 | 29 (10.9) | 311 | 4 (1.3) | 0.89 |
| 2010 | 214 | 25 (11.7) | 357 | 3 (0.8) | 0.94 |
PAF not calculable, thus otherwise specified as 1.
TABLE 3.
Crude and Adjusted Incidence Rates for Rotavirus Gastroenteritis (GE), Kibera (January 1, 2007, to December 31, 2010)
| Syndrome | Rotavirus GE Cases |
PYO | Crude IR |
95% CI
|
% GE Sampled at Clinic |
Adjusted
|
% GE Cases Visiting Field Clinic |
Adjusted
|
PAF | Final Adjusted
|
95% CI
|
||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lower | Upper | Adjustment 1 Cases |
Rate 1 | Adjustment 2 Cases |
Rate 2 | Adjustment 3 Cases |
Rate 3 | Lower | Upper | ||||||||
| Age | |||||||||||||||||
| <12 months | Overall | 14 | 3047 | 459.5 | 272.1 | 775.8 | 321 | 10,549.4 | 451 | 14,799.2 | 0.82 | 370 | 12,135 | 10,959 | 13,438 | ||
| Dehydration | 4 | 3047 | 131.3 | 49.3 | 349.8 | 10.3 | 39 | 1274.5 | 69.5 | 56 | 1833.9 | 0.82 | 46 | 1504 | 1126 | 2009 | |
| Dysentery | 2 | 3047 | 65.6 | 16.4 | 262.5 | 23.2 | 9 | 283.3 | 100 | 9 | 283.3 | 0.82 | 7 | 232 | 111 | 485 | |
| Mild Diarrhea | 8 | 3047 | 262.6 | 131.3 | 525.0 | 2.9 | 274 | 8991.6 | 70.9 | 386 | 12,682.0 | 0.82 | 317 | 10,399 | 9315 | 11,610 | |
| 12–23 months | Overall | 21 | 4167 | 504.0 | 328.6 | 772.9 | 479 | 11,483.1 | 652 | 15,642.4 | 0.82 | 534 | 12,827 | 11,784 | 13,962 | ||
| Dehydration | 4 | 4167 | 96.0 | 36.0 | 255.8 | 9.1 | 44 | 1060.7 | 73.9 | 60 | 1435.3 | 0.82 | 49 | 1177 | 890 | 1557 | |
| Dysentery | 3 | 4167 | 72.0 | 23.2 | 223.2 | 29.8 | 10 | 241.4 | 100 | 10 | 241.4 | 0.82 | 8 | 198 | 100 | 392 | |
| Mild Diarrhea | 14 | 4167 | 336.0 | 199.0 | 567.3 | 3.3 | 424 | 10,181.0 | 72.9 | 582 | 13,965.7 | 0.82 | 477 | 11,452 | 10,469 | 12,527 | |
| 24–59 months | Overall | 21 | 12,553 | 167.3 | 109.1 | 256.6 | 332 | 2641.6 | 388 | 3093.6 | 0.82 | 318 | 2537 | 2273 | 2831 | ||
| Dehydration | 1 | 12,553 | 8.0 | 1.1 | 56.6 | 10.4 | 10 | 76.7 | 79.2 | 12 | 96.8 | 0.82 | 10 | 79 | 43 | 148 | |
| Dysentery | 3 | 12,553 | 23.9 | 7.7 | 74.1 | 33.7 | 9 | 70.9 | 88.9 | 10 | 79.8 | 0.82 | 8 | 65 | 33 | 130 | |
| Mild Diarrhea | 17 | 12,553 | 135.4 | 84.2 | 217.8 | 5.4 | 313 | 2494.0 | 85.5 | 366 | 2917.0 | 0.82 | 300 | 2392 | 2136 | 2678 | |
| <5 years | Overall | 56 | 19,767 | 283.3 | 218.0 | 368.1 | 1092 | 5522.9 | 1419 | 7179.4 | 0.82 | 1164 | 5887 | 5558 | 6235 | ||
| Dehydration | 9 | 19,767 | 45.5 | 23.7 | 87.5 | 9.9 | 91 | 459.9 | 74.4 | 122 | 618.1 | 0.82 | 100 | 507 | 417 | 617 | |
| Dysentery | 8 | 19,767 | 40.5 | 20.2 | 80.9 | 31 | 26 | 130.6 | 94.1 | 27 | 138.7 | 0.82 | 22 | 114 | 75 | 172 | |
| Mild Diarrhea | 39 | 19,767 | 197.3 | 144.2 | 270.0 | 4 | 975 | 4932.5 | 76.8 | 1270 | 6422.5 | 0.82 | 1041 | 5266 | 4956 | 5596 | |
| 5–9 years | Overall | 11 | 16,764 | 65.6 | 36.3 | 118.5 | 78 | 464.1 | 94 | 560.0 | 0.72 | 68 | 403 | 318 | 512 | ||
| Dehydration | 1 | 16,764 | 6.0 | 0.8 | 42.3 | 37.5 | 3 | 15.9 | 80.2 | 3 | 19.8 | 0.72 | 2 | 14 | 4 | 51 | |
| Dysentery | 3 | 16,764 | 17.9 | 5.8 | 55.5 | 43.8 | 7 | 40.9 | 80 | 9 | 51.1 | 0.72 | 6 | 37 | 17 | 81 | |
| Mild Diarrhea | 7 | 16,764 | 41.8 | 19.9 | 87.6 | 10.3 | 68 | 407.4 | 83.3 | 82 | 489.0 | 0.72 | 59 | 352 | 273 | 454 | |
| 10–17 years | Overall | 14 | 17,203 | 81.4 | 48.2 | 137.4 | 77 | 446.0 | 93 | 539.8 | 0.72 | 67 | 389 | 306 | 494 | ||
| Dehydration | 0 | 17,203 | 0.0 | - | - | 28.6 | 0 | 0.0 | 81.5 | 0 | 0.0 | 0.72 | 0 | 0 | - | - | |
| Dysentery | 3 | 17,203 | 17.4 | 5.6 | 54.1 | 53.4 | 6 | 32.7 | 100 | 6 | 32.7 | 0.72 | 4 | 24 | 9 | 62 | |
| Mild Diarrhea | 11 | 17,203 | 63.9 | 35.4 | 115.5 | 15.5 | 71 | 413.3 | 81.5 | 87 | 507.2 | 0.72 | 63 | 365 | 285 | 468 | |
| 18–34 years | Overall | 35 | 40,046 | 87.4 | 62.8 | 121.7 | 154 | 385.6 | 200 | 500.4 | 0.72 | 144 | 360 | 306 | 424 | ||
| Dehydration | 3 | 40,046 | 7.5 | 2.4 | 23.2 | 19.2 | 16 | 39.0 | 66.8 | 23 | 58.3 | 0.72 | 17 | 42 | 26 | 68 | |
| Dysentery | 11 | 40,046 | 27.5 | 15.2 | 49.6 | 59.3 | 19 | 46.3 | 66.7 | 28 | 69.4 | 0.72 | 20 | 50 | 32 | 78 | |
| Mild Diarrhea | 21 | 40,046 | 52.4 | 34.2 | 80.4 | 17.5 | 120 | 300.3 | 80.6 | 149 | 372.6 | 0.72 | 107 | 268 | 222 | 324 | |
| 35–49 years | Overall | 14 | 13,600 | 102.9 | 61.0 | 173.8 | 52 | 383.3 | 83 | 606.6 | 0.72 | 59 | 437 | 339 | 563 | ||
| Dehydration | 0 | 13,600 | 0.0 | - | - | 9.1 | 0 | 0.0 | 64.6 | 0 | 0.0 | 0.72 | 0 | 0 | - | - | |
| Dysentery | 6 | 13,600 | 44.1 | 19.8 | 98.2 | 59.1 | 10 | 74.7 | 50 | 20 | 149.4 | 0.72 | 15 | 108 | 64 | 180 | |
| Mild Diarrhea | 8 | 13,600 | 58.8 | 29.4 | 117.6 | 19.1 | 42 | 308.6 | 67.5 | 62 | 457.2 | 0.72 | 45 | 329 | 246 | 441 | |
| 50+ years | Overall | 2 | 3213 | 62.2 | 15.6 | 248.9 | 8 | 249.0 | 10 | 308.8 | 0.72 | 7 | 222 | 107 | 463 | ||
| Dehydration | 0 | 3213 | 0.0 | - | - | 0 | 0 | 0.0 | 100 | 0 | 0.0 | 0.72 | 0 | 0 | - | - | |
| Dysentery | 1 | 3213 | 31.1 | 4.4 | 221.0 | 50 | 2 | 62.2 | 82.4 | 2 | 75.5 | 0.72 | 2 | 54 | 12 | 240 | |
| Mild Diarrhea | 1 | 3213 | 31.1 | 4.4 | 221.0 | 17.2 | 6 | 180.7 | 77.5 | 7 | 233.2 | 0.72 | 5 | 168 | 72 | 390 | |
| ≥5 years | Overall | 76 | 90,826 | 83.7 | 66.8 | 104.8 | 376 | 413.6 | 481 | 530.1 | 0.72 | 347 | 382 | 344 | 424 | ||
| Dehydration | 4 | 90,826 | 4.4 | 1.7 | 11.7 | 23.0 | 17 | 19.2 | 72.8 | 24 | 26.4 | 0.72 | 17 | 19 | 12 | 30 | |
| Dysentery | 24 | 90,826 | 26.4 | 17.7 | 39.4 | 55.0 | 44 | 48.1 | 70.6 | 62 | 68.1 | 0.72 | 45 | 49 | 37 | 66 | |
| Mild Diarrhea | 48 | 90,826 | 52.8 | 39.8 | 70.1 | 15.3 | 315 | 346.3 | 79.5 | 396 | 435.6 | 0.72 | 285 | 314 | 279 | 352 | |
| Total | Overall* | 169 | 110,592 | 152.8 | 131.4 | 177.7 | 1467 | 1326.8 | 1777 | 1606.6 | 0.72 | 1279 | 1157 | 1095 | 1222 | ||
| Dehydration | 13 | 110,592 | 11.8 | 6.8 | 20.2 | 11.1 | 117 | 106.0 | 73.9 | 159 | 143.4 | 0.72 | 114 | 103 | 86 | 124 | |
| Dysentery | 32 | 110,592 | 28.9 | 20.5 | 40.9 | 44.8 | 71 | 64.5 | 82.4 | 87 | 78.3 | 0.72 | 62 | 56 | 44 | 72 | |
| Mild Diarrhea | 87 | 110,592 | 78.7 | 63.8 | 97.1 | 7.3 | 1187 | 1073.2 | 77.5 | 1531 | 1384.8 | 0.72 | 1103 | 997 | 940 | 1058 | |
| Sex | |||||||||||||||||
| Male | Overall | 57 | 56,433 | 101.0 | 77.9 | 130.9 | 570 | 1010.3 | 753 | 1334.5 | 0.71 | 535 | 948 | 871 | 1031 | ||
| Dehydration | 4 | 56,433 | 7.1 | 2.7 | 18.9 | 10.5 | 38 | 67.6 | 73.7 | 52 | 91.8 | 0.71 | 37 | 65 | 47 | 90 | |
| Dysentery | 17 | 56,433 | 30.1 | 18.7 | 48.5 | 41.6 | 41 | 72.4 | 76.5 | 53 | 94.6 | 0.71 | 38 | 67 | 49 | 92 | |
| Mild Diarrhea | 36 | 56,433 | 63.8 | 46.0 | 88.4 | 7.3 | 491 | 870.3 | 75.8 | 648 | 1148.1 | 0.71 | 460 | 815 | 744 | 893 | |
| Female | Overall | 75 | 54,159 | 138.5 | 110.4 | 173.7 | 803 | 1482.1 | 1014 | 1871.9 | 0.85 | 862 | 1591 | 1488 | 1701 | ||
| Dehydration | 9 | 54,159 | 16.6 | 8.6 | 31.9 | 11.7 | 77 | 141.7 | 74.2 | 103 | 190.9 | 0.85 | 88 | 162 | 132 | 200 | |
| Dysentery | 15 | 54,159 | 27.7 | 16.7 | 45.9 | 48.2 | 31 | 57.5 | 88.2 | 35 | 65.2 | 0.85 | 30 | 55 | 39 | 79 | |
| Mild Diarrhea | 51 | 54,159 | 94.2 | 71.6 | 123.9 | 7.3 | 695 | 1282.9 | 79.4 | 875 | 1615.8 | 0.85 | 744 | 1373 | 1278 | 1476 | |
| Dehydration | 8 | 27,702 | 28.9 | 14.4 | 57.7 | 14.5 | 55 | 198.9 | 73.5 | 75 | 270.6 | 0.94 | 70 | 254 | 201 | 321 | |
| Dysentery | 11 | 27,702 | 39.7 | 22.0 | 71.7 | 60.3 | 18 | 65.8 | 88.9 | 21 | 74.1 | 0.94 | 19 | 70 | 45 | 109 | |
| Mild Diarrhea | 31 | 27,702 | 111.9 | 78.7 | 159.1 | 10.1 | 308 | 1113.5 | 78.4 | 393 | 1420.3 | 0.94 | 370 | 1335 | 1206 | 1478 | |
TABLE 4.
Crude and Adjusted Incidence Rates for Rotavirus Gastroenteritis (GE), Lwak (January 1, 2007, to December 31, 2010)
| Syndrome | Rotavirus cases |
PYO | Crude IR |
95% CI
|
% GE | Adjusted
|
% GE Cases Visiting Field Clinic |
Adjusted
|
PAF | Final Adjusted
|
95% CI
|
||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lower | Upper | Sampled at Clinic |
Cases 1 | Rate 1 | Cases 2 | Rate 2 | Cases 3 | Rate 3 | Lower | Upper | |||||||
| Age | |||||||||||||||||
| <12 months | Overall | 23 | 3426 | 671 | 446.1 | 1010.2 | 267 | 7803.6 | 498 | 14,549.5 | 0.93 | 462 | 13 494 | 12 318 | 14 782 | ||
| Dehydration | 16 | 3426 | 467 | 286.1 | 762.3 | 15.8 | 101 | 2955.2 | 60.2 | 168 | 4910.7 | 0.93 | 156 | 4 555 | 3 893 | 5 328 | |
| Dysentery | 0 | 3426 | 0 | 0.0 | 0.0 | 12.5 | 0 | 0.0 | 61.5 | 0 | 0.0 | 0.93 | 0 | 0 | 0 | 0 | |
| Mild diarrhea | 7 | 3426 | 204 | 97.4 | 428.6 | 4.2 | 166 | 4848.4 | 50.3 | 330 | 9638.7 | 0.93 | 306 | 8 940 | 7 992 | 9 999 | |
| 12–23 months | Overall | 20 | 3242 | 617 | 398.0 | 956.2 | 277 | 8547.2 | 524 | 16,158.0 | 0.93 | 486 | 14 986 | 13 711 | 16 380 | ||
| Dehydration | 14 | 3242 | 432 | 255.8 | 729.2 | 15.7 | 89 | 2745.8 | 60.8 | 146 | 4514.8 | 0.93 | 136 | 4 187 | 3 539 | 4 954 | |
| Dysentery | 0 | 3242 | 0 | 0.0 | 0.0 | 8.0 | 0 | 0.0 | 100 | 0 | 0.0 | 0.93 | 0 | 0 | 0 | 0 | |
| Mild diarrhea | 6 | 3242 | 185 | 83.1 | 412.0 | 3.2 | 188 | 5801.3 | 49.8 | 377 | 11,643.2 | 0.93 | 350 | 10 799 | 9 725 | 11 991 | |
| 24–59 months | Overall | 6 | 9275 | 65 | 29.1 | 144.0 | 94 | 1018.3 | 177 | 1907.9 | 0.93 | 164 | 1 770 | 1 519 | 2 062 | ||
| Dehydration | 2 | 9275 | 22 | 5.4 | 86.2 | 15.3 | 13 | 141.4 | 56.2 | 23 | 251.5 | 0.93 | 22 | 233 | 153 | 356 | |
| Dysentery | 0 | 9275 | 0 | 0.0 | 0.0 | 13.9 | 0 | 0.0 | 75.0 | 0 | 0.0 | 0.93 | 0 | 0 | 0 | 0 | |
| Mild diarrhea | 4 | 9275 | 43 | 16.2 | 114.9 | 4.9 | 81 | 876.9 | 52.9 | 154 | 1656.4 | 0.93 | 142 | 1 536 | 1 304 | 1 810 | |
| <5 years | Overall | 49 | 15,943 | 307 | 232.3 | 406.7 | 621 | 3893.0 | 1165 | 7308.7 | 0.93 | 1081 | 6 779 | 6 386 | 7 195 | ||
| Dehydration | 32 | 15,943 | 201 | 141.9 | 283.8 | 15.7 | 204 | 1281.2 | 59.3 | 345 | 2161.5 | 0.93 | 320 | 2 005 | 1 797 | 2 237 | |
| Dysentery | 0 | 15,943 | 0 | 0.0 | 0.0 | 11.9 | 0 | 0.0 | 68.2 | 0 | 0.0 | 0.93 | 0 | 0 | 0 | 0 | |
| Mild diarrhea | 17 | 15,943 | 107 | 66.3 | 171.5 | 4.1 | 416 | 2611.9 | 50.7 | 821 | 5147.1 | 0.93 | 761 | 4 774 | 4 446 | 5 125 | |
| 5–9 years | Overall | 3 | 19,443 | 15 | 5.0 | 47.8 | 42 | 215.1 | 90 | 460.4 | 0.89 | 79 | 408 | 328 | 509 | ||
| Dehydration | 0 | 19,443 | 0 | 0.0 | 0.0 | 12.9 | 0 | 0.0 | 54.1 | 0 | 0.0 | 0.89 | 0 | 0 | 0 | 0 | |
| Dysentery | 0 | 19,443 | 0 | 0.0 | 0.0 | 20.0 | 0 | 0.0 | 40.0 | 0 | 0.0 | 0.89 | 0 | 0 | 0 | 0 | |
| Mild diarrhea | 3 | 19,443 | 15 | 5.0 | 47.8 | 7.2 | 42 | 215.1 | 46.7 | 90 | 460.4 | 0.89 | 79 | 408 | 328 | 509 | |
| 10–17 years | Overall | 11 | 18,818 | 58 | 32.4 | 105.6 | 72 | 384.5 | 121 | 645.4 | 0.89 | 108 | 573 | 474 | 692 | ||
| Dehydration | 0 | 18,818 | 0 | 0.0 | 0.0 | 31.4 | 0 | 0.0 | 54.8 | 0 | 0.0 | 0.89 | 0 | 0 | 0 | 0 | |
| Dysentery | 6 | 18,818 | 32 | 14.3 | 71.0 | 23.0 | 26 | 138.6 | 80.0 | 33 | 173.3 | 0.89 | 29 | 154 | 107 | 221 | |
| Mild diarrhea | 5 | 18,818 | 27 | 11.1 | 63.8 | 10.8 | 46 | 245.9 | 52.1 | 89 | 472.1 | 0.89 | 79 | 419 | 336 | 522 | |
| 18–34 years | Overall | 32 | 23,725 | 135 | 95.4 | 190.7 | 148 | 624.8 | 272 | 1146.8 | 0.89 | 241 | 1 017 | 897 | 1 154 | ||
| Dehydration | 3 | 23,725 | 13 | 4.1 | 39.2 | 21.2 | 14 | 59.6 | 60.7 | 23 | 98.1 | 0.89 | 21 | 87 | 57 | 134 | |
| Dysentery | 9 | 23,725 | 38 | 19.7 | 72.9 | 40.5 | 22 | 93.6 | 73.7 | 30 | 127.0 | 0.89 | 27 | 113 | 77 | 165 | |
| Mild diarrhea | 20 | 23,725 | 84 | 54.4 | 130.7 | 17.9 | 112 | 471.7 | 51.2 | 219 | 921.7 | 0.89 | 194 | 818 | 710 | 941 | |
| 35–49 years | Overall | 11 | 10,312 | 107 | 59.1 | 192.6 | 42 | 411.8 | 76 | 736.3 | 0.89 | 67 | 653 | 515 | 830 | ||
| Dehydration | 0 | 10,312 | 0 | 0.0 | 0.0 | 19.5 | 0 | 0.0 | 57.0 | 0 | 0.0 | 0.89 | 0 | 0 | 0 | 0 | |
| Dysentery | 6 | 10,312 | 58 | 26.1 | 129.5 | 35.6 | 17 | 163.6 | 61.9 | 27 | 264.2 | 0.89 | 24 | 234 | 157 | 349 | |
| Mild diarrhea | 5 | 10,312 | 48 | 20.2 | 116.5 | 19.5 | 26 | 248.2 | 52.6 | 49 | 472.1 | 0.89 | 43 | 419 | 311 | 564 | |
| 50+ years | Overall | 10 | 13,977 | 72 | 38.5 | 133.0 | 52 | 374.1 | 99 | 705.8 | 0.89 | 88 | 626 | 508 | 772 | ||
| Dehydration | 4 | 13,977 | 29 | 10.7 | 76.3 | 18.6 | 21 | 153.7 | 54.1 | 40 | 284.3 | 0.89 | 35 | 252 | 181 | 351 | |
| Dysentery | 3 | 13,977 | 21 | 6.9 | 66.6 | 24.5 | 12 | 87.6 | 57.1 | 21 | 153.4 | 0.89 | 19 | 136 | 87 | 213 | |
| Mild diarrhea | 3 | 13,977 | 21 | 6.9 | 66.6 | 16.2 | 19 | 132.8 | 49.5 | 37 | 268.0 | 0.89 | 33 | 238 | 169 | 334 | |
| ≥5 years | Overall | 67 | 86,274 | 78 | 61.1 | 98.7 | 363 | 420.6 | 679 | 786.7 | 0.89 | 602 | 698 | 644 | 756 | ||
| Dehydration | 7 | 86,274 | 8 | 3.9 | 17.0 | 20.3 | 35 | 40.0 | 56.4 | 61 | 71.0 | 0.89 | 54 | 63 | 48 | 82 | |
| Dysentery | 24 | 86,274 | 28 | 18.6 | 41.5 | 31.6 | 76 | 88.1 | 64.5 | 118 | 136.6 | 0.89 | 105 | 121 | 100 | 147 | |
| Mild diarrhea | 36 | 86,274 | 42 | 30.1 | 57.8 | 14.3 | 252 | 292.5 | 50.5 | 500 | 579.1 | 0.89 | 443 | 514 | 468 | 564 | |
| Total | Overall | 116 | 102,217 | 113 | 94.6 | 136.1 | 864 | 845.2 | 1614 | 1578.9 | 0.90 | 1459 | 1 427 | 1 356 | 1502 | ||
| Dehydration | 39 | 102,217 | 38 | 27.9 | 52.2 | 17.2 | 227 | 222.0 | 57.8 | 393 | 384.3 | 0.90 | 355 | 347 | 313 | 386 | |
| Dysentery | 24 | 102,217 | 23 | 15.7 | 35.0 | 28.6 | 84 | 82.1 | 65.3 | 128 | 125.7 | 0.90 | 116 | 114 | 95 | 136 | |
| Mild diarrhea | 53 | 102,217 | 52 | 39.6 | 67.9 | 9.6 | 553 | 541.2 | 50.6 | 1093 | 1068.9 | 0.90 | 988 | 966 | 908 | 1029 | |
| Sex | |||||||||||||||||
| Male | Overall | 48 | 48,253 | 99 | 75.0 | 132.0 | 395 | 819.5 | 764 | 1583.6 | 0.90 | 689 | 1 428 | 1 326 | 1539 | ||
| Dehydration | 14 | 48,253 | 29 | 17.2 | 49.0 | 15.6 | 90 | 185.6 | 57.5 | 156 | 322.9 | 0.90 | 141 | 291 | 247 | 344 | |
| Dysentery | 9 | 48,253 | 19 | 9.7 | 35.8 | 29.7 | 30 | 62.9 | 64.3 | 47 | 97.8 | 0.90 | 43 | 88 | 65 | 119 | |
| Mild diarrhea | 25 | 48,253 | 52 | 35.0 | 76.7 | 9.1 | 276 | 571.0 | 49.1 | 561 | 1162.9 | 0.90 | 506 | 1 049 | 961 | 1144 | |
| Female | Overall | 68 | 53,964 | 126 | 99.4 | 159.8 | 465 | 862.0 | 845 | 1565.5 | 0.91 | 766 | 1 420 | 1 323 | 1524 | ||
| Dehydration | 25 | 53,964 | 46 | 31.3 | 68.6 | 18.8 | 133 | 245.9 | 58.0 | 229 | 424.0 | 0.91 | 208 | 385 | 3356 | 441 | |
| Dysentery | 15 | 53,964 | 28 | 16.8 | 46.1 | 27.9 | 54 | 99.8 | 66.1 | 81 | 151.0 | 0.91 | 74 | 137 | 109 | 172 | |
| Mild diarrhea | 28 | 53,964 | 52 | 35.8 | 75.1 | 10.1 | 279 | 516.2 | 52.1 | 534 | 990.5 | 0.91 | 485 | 898 | 822 | 982 | |
Incidence rates (IR) per 100,000 PYO.
Adjustment 1 based on percentage of patients who met sample collection criteria who provided a stool sample (see Methods).
Adjustment 2 (total adjustment) based on the patients with diarrhea or dysentery during home visit, who visited the field clinic (adjusting the adjustment 1 rates—see methods).
The overall adjusted incidence rates were 1157/100,000 PYO and 1427/100,000 PYO in Kibera and Lwak, respectively, with highest rates in children 12–23 months of age followed by infants (<12 months of age; Table 3 and Table 4). Rotavirus gastroenteritis incidence rates were >220/100,000 PYO for each age group ≥5 years of age with the highest rate (437/100,000 PYO) in Kibera in persons 35–49 years of age, which was about one-sixth of the rate calculated for children 24–59 months of age (Table 3). In Lwak, the highest rates in adults were in the 18–34 year age group (982/100,000 PYO), which was more than half of the rate calculated for 24–59 month olds (Table 4).
HIV test results were available from a limited number of patients. In Kibera, among 39 rotavirus cases ≥18 years old tested for HIV antibodies, 9 (23.1%) were HIV-infected, compared with 133 (23.9%) of 557 nonrotavirus-associated diarrhea cases tested for HIV infection. In Lwak, among 29 rotavirus cases ≥18 years old, tested for HIV antibodies, 10 (34.5%) were HIV-infected compared with 154 (50.2%) of 307 nonrotavirus-associated diarrhea cases tested for HIV infection (relative risk = 0.55; 95% CI = 0.26–1.15).
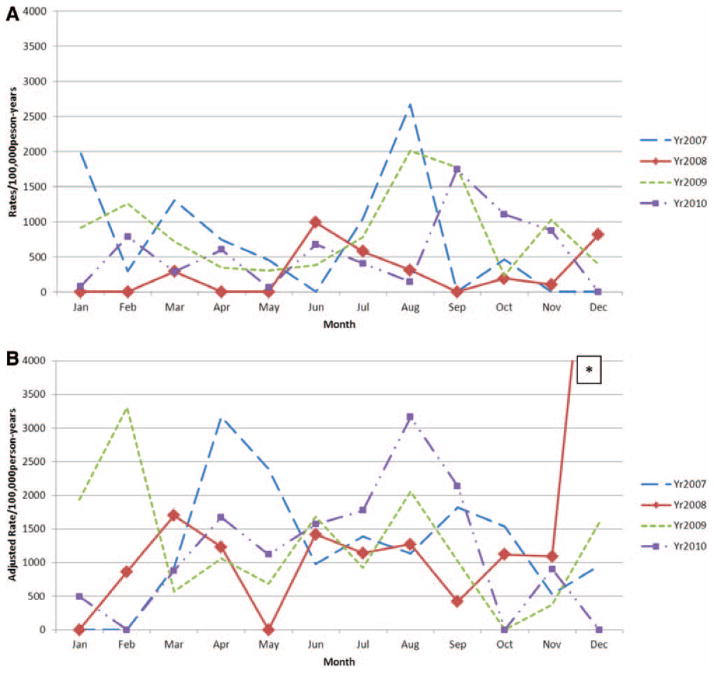
Annual crude rotavirus rates varied across the 4 years of observation from 45 to 195/100,000 PYO in Kibera and from 85 to 160/100,000 PYO in Lwak. Throughout the 4-year period, rotavirus gastroenteritis occurred year round without consistent, specific high incidence peaks, although in Kibera, peaks occurred during the cool, dry months of July and August for 3 of the 4 years (Fig. 2).
FIGURE 2.
Adjusted annual incidence of rotavirus gastroenteritis by month: A) Kibera (2007–2010) and B) Lwak (2007–2010). *Actual value of this off-scale data point (December 2008) is 8000/100,000 person years of observation.
DISCUSSION
This study provides community population-based incidence data for rotavirus gastroenteritis in children and adults as a result of rigorously defined denominators and active surveillance to identify cases and to adjust incidence based on the proportion of cases likely missed. The incidence rates can be used to estimate absolute numbers of cases for people living in similar settings, using population census data. While demonstration of high rates in children is not unexpected, the study also showed that rotavirus is an important cause of gastroenteritis in adults in these settings in Kenya.
Rotavirus crude incidence rates varied substantially annually over the 4-year course of this study. This confirms earlier reports of annual incidence variation19 and highlights the importance of multiple years of surveillance (and use of alternative approaches, like case-control studies, cluster designed interventions and severity-impact investigations) to assess and document impact of rotavirus vaccine postintroduction. For example, if rotavirus vaccine had been introduced in 2007, it would have been tempting to ascribe the 53% reduction in incidence (based on surveillance data from a national surveillance system) in Lwak when comparing 2008 data with 2007 data, as evidence of vaccine impact; conversely, in Kibera, the crude rate increased 4-fold from 2008 to 2009; if vaccine introduction had occurred in 2008, there might have been premature and incorrect conclusions of ineffectiveness (or increased associated risk) of the immunization program, potentially negatively impacting continued enthusiasm.
Rotavirus is often described as an “egalitarian” virus, infecting people regardless of their location or socioeconomic status,20 although severity and fatal outcome are dictated by comorbidities and access to health care (including oral rehydration solutions).5,19 Despite vastly different ecologies found in the sparsely populated rural western Kenya and densely populated urban Nairobi, interestingly, the overall rates of rotavirus disease in the 2 areas are quite similar. The higher incidence we observed of dehydration-associated rotavirus gastroenteritis in the rural area might reflect differential access to rehydration in the 2 areas.
A value of population-based incidence rates is that one can estimate numbers of people affected by a disease and project the number of illness episodes that can be prevented by applying interventions of known effectiveness. For instance, the 2009 census showed that there were 5.4 million people living in Nyanza Province (where the rural Lwak surveillance site is located), including 810,000 children <5 years of age. Given an adjusted annual incidence rate of rotavirus gastroenteritis in Lwak of 6779 cases per 100,000 children <5 years of age, we estimate that there are >54,500 cases per year in children in the Province (which represents 13.5% of the national population). If we assume the efficacy of rotavirus immunization in this setting to be approximately 60%2 and an 80% vaccine coverage rate, then about 26,000 cases would be preventable per year in Nyanza Province alone. Likewise, there is an estimated 1.9 million people living in urban slums in Nairobi with an estimated 285,000 children <5 years of age. The adjusted incidence rate in Kibera for children <5 years of age of 5887 cases per 100,000 population per year, translates to >16,750 cases per year. Using the same vaccine efficacy and estimated coverage rates as above, >8000 cases of rotavirus gastroenteritis could be prevented per year in <5 years of age in Nairobi urban slums.
Based on data from a variety of studies and sites, it would be possible to estimate preventable burden from the estimated proportion of rotavirus gastroenteritis that requires hospitalization and the proportion of rotavirus gastroenteritis associated with mortality. Our study was not designed to assess mortality from rotavirus or to calculate economic impact contributing to disease burden calculations. Given that rotavirus-associated mortality is associated with poor or delayed access to rehydration options and that provision of good quality health care is a component of participation in the PBIDS from which the incidence rates are calculated, we did not expect to observe many deaths. However, most children in Kenya do not have this level of access to health care, so the rates of rotavirus illness determined during this study could be used to estimate mortality, as has been done during global estimates of rotavirus-associated burden.5,19,20 Having estimates of overall burden provide a tangible way for policy-makers to assess likely impact of interventions, and for donors and vaccine developers to consider potential impact of more effective vaccine regimens.
HIV has been known to increase the incidence, shedding and severity of infections due to a variety of pathogens21–23 and studies have suggested that HIV-infected adults have an increased risk for rotavirus gastroenteritis.24,25 While surprisingly high rates of rotavirus-associated diarrhea were shown in our study, rotavirus-infected adults were not more likely to be infected with HIV than adults with diarrhea due to other causes; thus, it did not appear that HIV infection was a specific risk factor for rotavirus infection. Nonetheless, the HIV-infection rates for adults with diarrhea (with or without rotavirus as an etiology) were higher than the seroprevalences of HIV infection in both areas, consistent with evidence that HIV infection is associated with greater risk of illness from a variety of gastrointestinal pathogens.
More data are needed to assess the impact (including severity and outcome) of rotavirus gastroenteritis in adults, especially in areas with high HIV seroprevalence. Vaccinating children will be key in directly reducing childhood associated morbidity and death, and there may be an indirect benefit of reducing infections in older children and adults.25
While this study was unique in terms of its community-based methods identifying rotavirus gastroenteritis in the urban and rural communities we surveyed, and for providing reliable basis for rate adjustments, limitations may have led to underestimates or overestimates of the burden of disease. Because of transport requirements to the Kisumu laboratory, stool specimens were often processed >24 hours after collection. In calculating adjusted incidence rates, we assumed that persons with diarrhea who reported visiting any clinic had similar rotavirus gastroenteritis incidence rates as those seen in the study field clinic. We also assumed that severity of illness for patients with diarrhea (and the 3 categories of dehydration, dysentery and mild diarrhea) who provided stool for testing were not different from those of patients who were not sampled. These assumptions would be erroneous if there was a systematic bias with regard to whether persons with moderate to severe diarrhea went to the designated field clinic and whether patients and clinicians were more aggressive about sampling, based on the type or severity of diarrhea. It also should be noted that freezing and batch testing of specimens may have reduced sensitivity of the assay.
The findings of this study provide evidence for high community-based incidence of rotavirus gastroenteritis, as well as an important role of rotavirus as a cause of diarrheal disease in adults, providing the basis for evaluating indirect protection in older children and adults when rotavirus immunization programs are implemented. High incidence rates are shown in both rural and urban slum settings, supporting expectations for substantial impact of universal rotavirus immunization of infants and toddlers in Kenya, and likely elsewhere in Africa. In addition, year to year variation of rotavirus-associated diarrhea incidence highlights that evaluation of impact of rotavirus immunization will require multiple years of observation.
Footnotes
At the time of publication, W. Mwiti is an employee of GlaxoSmithKline Limited East Africa. His employment began after the investigative and analytic phases relevant to this paper and during the final stages of manuscript preparation. During this time W. Mwiti had limited input to the content of this paper. The other authors have no funding or conflicts of interest statement to disclose.
References
- 1.Madhi SA, Cunliffe NA, Steele D, et al. Effect of human rotavirus vaccine on severe diarrhea in African infants. N Engl J Med. 2010;362:289–298. doi: 10.1056/NEJMoa0904797. [DOI] [PubMed] [Google Scholar]
- 2.Armah GE, Sow SO, Breiman RF, et al. Efficacy of pentavalent rotavirus vaccine against severe rotavirus gastroenteritis in infants in developing countries in sub-Saharan Africa: a randomised, double-blind, placebo-controlled trial. Lancet. 2010;376:606–614. doi: 10.1016/S0140-6736(10)60889-6. [DOI] [PubMed] [Google Scholar]
- 3.Desai R, Oliveira LH, Parashar UD, et al. Reduction in morbidity and mortality from childhood diarrhoeal disease after species A rotavirus vaccine introduction in Latin America - a review. Mem Inst Oswaldo Cruz. 2011;106:907–911. doi: 10.1590/s0074-02762011000800002. [DOI] [PubMed] [Google Scholar]
- 4.Van Effelterre T, Soriano-Gabarró M, Debrus S, et al. A mathematical model of the indirect effects of rotavirus vaccination. Epidemiol Infect. 2010;138:884–897. doi: 10.1017/S0950268809991245. [DOI] [PubMed] [Google Scholar]
- 5.Parashar UD, Burton A, Lanata C, et al. Global mortality associated with rotavirus disease among children in 2004. J Infect Dis. 2009;200(suppl 1):S9–S15. doi: 10.1086/605025. [DOI] [PubMed] [Google Scholar]
- 6.Krishnan T, Sen A, Choudhury JS, et al. Emergence of adult diarrhoea rotavirus in Calcutta, India. Lancet. 1999;353:380–381. doi: 10.1016/s0140-6736(05)74954-0. [DOI] [PubMed] [Google Scholar]
- 7.Anderson EJ, Weber SG. Rotavirus infection in adults. Lancet Infect Dis. 2004;4:91–99. doi: 10.1016/S1473-3099(04)00928-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Dalal W, Feikin DR, Amolloh M, et al. Home-based HIV testing and counseling in rural and urban Kenyan communities. J Acquir Immune Defic Syndr. 2013;62:e47–e54. doi: 10.1097/QAI.0b013e318276bea0. [DOI] [PubMed] [Google Scholar]
- 9.Feikin DR, Olack B, Bigogo GM, et al. The burden of common infectious disease syndromes at the clinic and household level from population-based surveillance in rural and urban Kenya. PLoS One. 2011;6:e16085. doi: 10.1371/journal.pone.0016085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kim CY, Breiman RF, Cosmas L, et al. Secondary household transmission of 2009 pandemic influenza A (H1N1) virus among an urban and rural population in Kenya, 2009–2010. PLoS One. 2012;7:e38166. doi: 10.1371/journal.pone.0038166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Feikin DR, Njenga MK, Bigogo G, et al. Viral and bacterial causes of severe acute respiratory illness among children less than 5 years old in a high malaria prevalence area of Western Kenya, 2007–2010. Pediatr Infect Dis J. 2013;32:e14–19. doi: 10.1097/INF.0b013e31826fd39b. [DOI] [PubMed] [Google Scholar]
- 12.Feikin DR, Njenga MK, Bigogo G, et al. Etiology and incidence of viral and bacterial acute respiratory illness among older children and adults in rural Western Kenya, 2007–2010. PLoS One. 2012;7:e43656. doi: 10.1371/journal.pone.0043656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Singleton RJ, Bulkow LR, Miernyk K, et al. Viral respiratory infections in hospitalized and community control children in Alaska. J Med Virol. 2010;82:1282–1290. doi: 10.1002/jmv.21790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Njuguna HN, Cosmas L, Williamson J, et al. Use of population-based surveillance to define the high incidence of shigellosis in an urban slum in Nairobi, Kenya. PLoS One. 2013;8:e58437. doi: 10.1371/journal.pone.0058437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dennehy PH, Gauntlett DR, Spangenberger SE. Choice of reference assay for the detection of rotavirus in fecal specimens: electron microscopy versus enzyme immunoassay. J Clin Microbiol. 1990;28:1280–1283. doi: 10.1128/jcm.28.6.1280-1283.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Breiman RF, Cosmas L, Njuguna H, et al. Population-based incidence of typhoid fever in an urban informal settlement and a rural area in Kenya: implications for typhoid vaccine use in Africa. PLoS One. 2012;7:e29119. doi: 10.1371/journal.pone.0029119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kirkwood BR, Sterne JAC. Essentials of Medical Statistics. Oxford: Blackwell Science; 2006. [Google Scholar]
- 18.Kenya National Bureau of Statistics. Kenya census. [Accessed November 12, 2013];Summary tables. 2009 Available at: http://www.knbs.or.ke/Census%20Results/KNBS%20Brochure.pdf.
- 19.Parashar UD, Hummelman EG, Bresee JS, et al. Global illness and deaths caused by rotavirus disease in children. Emerg Infect Dis. 2003;9:565–572. doi: 10.3201/eid0905.020562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gomez JA, Sordo ME, Gentile A. Epidemiologic patterns of diarrheal disease in Argentina: estimation of rotavirus disease burden. Pediatr Infect Dis J. 2002;21:843–850. doi: 10.1097/00006454-200209000-00012. [DOI] [PubMed] [Google Scholar]
- 21.Weinstock DM, Gubareva LV, Zuccotti G. Prolonged shedding of multidrug-resistant influenza A virus in an immunocompromised patient. N Engl J Med. 2003;348:867–868. doi: 10.1056/NEJM200302273480923. [DOI] [PubMed] [Google Scholar]
- 22.Klimov AI, Rocha E, Hayden FG, et al. Prolonged shedding of amantadine-resistant influenzae A viruses by immunodeficient patients: detection by polymerase chain reaction-restriction analysis. J Infect Dis. 1995;172:1352–1355. doi: 10.1093/infdis/172.5.1352. [DOI] [PubMed] [Google Scholar]
- 23.Pozio E, Rezza G, Boschini A, et al. Clinical cryptosporidiosis and human immunodeficiency virus (HIV)-induced immunosuppression: findings from a longitudinal study of HIV-positive and HIV-negative former injection drug users. J Infect Dis. 1997;176:969–975. doi: 10.1086/516498. [DOI] [PubMed] [Google Scholar]
- 24.Anderson EJ, Katz BZ, Polin JA, et al. Rotavirus in adults requiring hospitalization. J Infect. 2012;64:89–95. doi: 10.1016/j.jinf.2011.09.003. [DOI] [PubMed] [Google Scholar]
- 25.Groome MJ, Madhi SA. Five-year cohort study on the burden of hospitalisation for acute diarrhoeal disease in African HIV-infected and HIV-uninfected children: potential benefits of rotavirus vaccine. Vaccine. 2012;30(suppl 1):A173–A178. doi: 10.1016/j.vaccine.2011.08.004. [DOI] [PubMed] [Google Scholar]