Abstract
The study of T cell responses and their consequences during allo-antigen recognition requires a model that enables one to distinguish between donor and host T cells, to easily monitor the graft, and to adapt the system in order to answer different immunological questions. Medawar and colleagues established allogeneic tail-skin transplantation in mice in 1955. Since then, the skin transplantation model has been continuously modified and adapted to answer specific questions. The use of tail-skin renders this model easy to score for graft rejection, requires neither extensive preparation nor deep anesthesia, is applicable to animals of all genetic background, discourages ischemic necrosis, and permits chemical and biological intervention.
In general, both CD4+ and CD8+ allogeneic T cells are responsible for the rejection of allografts since they recognize mismatched major histocompatibility antigens from different mouse strains. Several models have been described for activating allogeneic T cells in skin-transplanted mice. The identification of major histocompatibility complex (MHC) class I and II molecules in different mouse strains including C57BL/6 mice was an important step toward understanding and studying T cell-mediated alloresponses. In the tail-skin transplantation model described here, a three-point mutation (I-Abm12) in the antigen-presenting groove of the MHC-class II (I-Ab) molecule is sufficient to induce strong allogeneic CD4+ T cell activation in C57BL/6 mice. Skin grafts from I-Abm12 mice on C57BL/6 mice are rejected within 12-15 days, while syngeneic grafts are accepted for up to 100 days. The absence of T cells (CD3-/- and Rag2-/- mice) allows skin graft acceptance up to 100 days, which can be overcome by transferring 2 x 104 wild type or transgenic T cells. Adoptively transferred T cells proliferate and produce IFN-γ in I-Abm12-transplanted Rag2-/- mice.
Keywords: Immunology, Issue 89, Tail-skin transplantation, I-Abm12 mismatch, CD4+ T cell, ABM, Rejection, Tolerance
Introduction
Transplantation of solid organs such as skin, heart and kidneys is now a standard procedure in medical practice worldwide1. Successfully transplanted organs can be rejected by activation of the recipient immune system, which recognizes the major histocompatibility antigens of the donor. Therefore transplanted patients need treatment with immunosuppressive drugs2. Allogeneic skin transplantation in mice was established by Medawar and colleagues in 1955 and was helpful for identifying the targeted molecules later described as major histocompatibility complex (MHC) class I and II. Since then, the skin transplantation model has been continuously modified and adapted to study the role of T cell subsets and the relevance of chemical and biological intervention in suppressing graft rejection2-4. Skin from the ear and trunk are more difficult to prepare and are more susceptible to hypoxia and necrosis than tail-skin5; however, the transplantation procedure is similar. In addition the monitoring of tail-skin transplants is easy due to the characteristic hair texture of the skin.
This article provides a detailed procedure for MHC class II mismatch tail-skin transplantation that allows for the study of different aspects of CD4+ T cell-mediated allograft rejection and tolerance in mice. The natural three-point mutation in the MHC class II molecule I-Ab (called I-Abm12)6-9 is sufficient to induce rejection of skin allografts in C57BL/6 mice8. The I-Abm12 molecule activates CD4+ T cells with various αβ-T cell receptor (TcR) chains from C57BL/6 mice, among which Vα2Vβ8-TcR-specific T cells were identified in order to generate a TcR-transgenic mouse10. The adoptive transfer of Vα2Vβ8-TcR-specific T cells has been used to establish a rejection model in immunodeficient C57BL/6 Rag2-/- mice transplanted with I-Abm12 skin.
Genetic differences between donor and recipient impact the outcome of transplant acceptance and rejection. There are different types of transplants: autografts are transplants from the recipient individual itself; syngrafts and allografts are transplants from genetically identical and genetically unrelated individuals respectively. Acceptance of different allogeneic organ transplants has been demonstrated by chemical and biological intervention in patients and mouse models11,3,4. In a basic approach, anti-CD3 antibody-treated C57BL/6 mice showed prolonged survival of I-Abm12 tail-skin (unpublished data). Depletion of CD4+ and CD8+ T cells before transplantation in recipient mice resulted in acceptance of MHC class I and II mismatched grafts (rev. in 12). Interestingly, rejection of skin grafts depends on the presence of CD4+ T cells (rev. in 12). In this model, targeting specific interactions between different immune cells by blocking costimulatory molecules with antibodies or suppression with regulatory T cells might induce tolerance (unpublished data). Indeed, blocking both CD40 and CD28 led to long-term skin allograft tolerance13,14.
Tail-skin transplantation is easy to perform and easy to monitor compared to transplantation of other organs. In addition, tail-skin transplants are easy to prepare and are less susceptible to ischemia than other skin tissues. In contrast to injected anesthetics, the use of anesthetic gas (isofluorane) during transplantation shortens both the procedure and recipient recovery time. Curling of the tail-skin transplant, which may lead to incomplete wound healing and graft rejection, is prevented by application of tissue adhesive. Furthermore, the I-Abm12 tail-skin transplantation model exclusively activates CD4+ T cells in both immunocompetent and immunodeficient mice (of the same genetic background) facilitating the interpretation of the results.
This protocol describes a reliable, reproducible and easily monitored mouse model that allows for chemical and biological intervention. The model is intended for investigating rejection and tolerance induction of tail-skin transplants.
Protocol
In this video publication and protocol, all animal procedures were conducted in accordance to the animal protocol approved by the Cantonal Authority Basel-Stadt, Switzerland. Perform all procedures in sterile conditions where possible.
1. Preparation of Surgery
Autoclave all surgical instruments and gauze before use.
Heat the warm pad and organize surgical instruments on the table (Table of Materials/Methods).
Open the finger strip bandages. Apply petroleum jelly on the wound pad with a cotton swab (make sure the gauze is completely covered).
Weigh the mice and administer analgesia Buprenorphine 0.05 mg/kg body weight. Note: this will provide analgesia immediately after transplantation.
2. Preparation of the Tail-skin for Transplantation
Euthanize donor mice (e.g., I-Abm12) per CO2 asphyxiation and disinfect the tails with disinfection solution before excision.
Position the tail with the black stripe down on a clean plastic board and cut it longitudinally throughout the middle line, making a superficial incision involving only the skin layer.
Pull off the skin from the tail using gripping forceps.
Put the tail-skin in a 10 cm cell culture dish filled with 10 ml HBSS.
3. Surgical Procedure
Anesthetize recipient mice (e.g., C57BL/6) by inhalation of a 3% isofluorane solution in a transparent induction box. Note: It takes 3-5 min for the mouse to reach surgical plane anesthesia. Use vet ointment on eyes to prevent dryness while under anesthesia.
Place mouse on the warm pad and apply 1.5% isofluorane via a mouth mask and asses each animal reflex by toe pinching.
Shave the dorsal site of the recipient mice (e.g., C57BL/6) and remove contaminating hairs using a dry gauze swab.
Disinfect shaved transplantation site with aseptic solution.
Pinch the skin with forceps in the middle/right site of the dorsum and use curved scissors to make a round incision of about 1 x 0.6 cm.
If bleeding occurs, then clean the wound with a sterile cotton swab.
Evaluate the size of the incision and cut a transplant of the estimated size from the tail-skin using the scalpel.
Trim corners to round the transplant.
Load the transplant on the scalpel, dry excess of HBSS on a sterile cotton swab and place it in the graft bed of the recipient mouse.
Avoid overlapping of donor and host skin and apply tissue adhesive on the contact zone exclusively.
Apply a plaster without creases around the mouse waist.
Apply adhesive non-elastic bandage to fix the plaster to the mouse (3 mm shifted).
Incise with the scissors about 3 mm in the upper ventral site of the bandage. Note: This is to avoid trapping of the mouse’s teeth during the recovery phase.
Remove the anesthetic-mouth-mask and keep the transplanted mice on the heating pad until awake.
Return transplanted mice to their cages only when fully recovered and assess their mobility to ensure that the bandage is not too tight.
Sterilize all surgical instruments with 70% ethanol before proceeding with next surgery.
4. Post-operative Care
Add paracetamol-containing syrup to the drinking water (4 mg/ml) and administer it for 7 days.
Return mouse to the animal room once it has recovered.
Closely monitor mice for clinical signs including depression or other behavioral changes (eating or immobility), abnormal appearance (lack of grooming) or posture (pilo-erection or hunched posture) for the time the bandage is in place (1 week).
5. Bandage Removal (6-7 Days Post-transplantation)
Anesthetize mice by inhalation of a 3% Isofluorane solution in a transparent induction box (see above).
Cut bandage and plaster with artery scissors and carefully remove them. Note: avoid stretching of the transplant site.
Monitor the transplanted mice for clinical signs (see 4.3) and score for rejection of skin grafts (Table 1). Note: The graft score system takes advantage of the different hair textures on the tail-skin compared to the dorsal-skin. The appearance of the transplant and its rejection is categorized in 3 grades. Allogeneic skin (MHC II mismatched, e.g., I-Abm12) grafts of C57BL/6 immunocompetent mice are usually rejected within 12-15 days post transplantation (own data and 15-20).
Representative Results
In a first approach C57BL/6 mice were transplanted with I-Abm12 allografts and I-Ab syngrafts. After bandage removal, grafts manifest signs of wound healing without closure of the contact zone in C57BL/6 mice (Figure 1A). After bandage removal, CD4+ T cell-mediated inflammation led to the appearance of necrotic areas (red spots) and rejection of I-Abm12 allografts in C57BL/6 mice within 13 days after transplantation (Figure 1B-C, filled symbols), while syngrafts were tolerated for up to 100 days (Figure 1B-C, open symbols). These data show the progression of acute rejection of I-Abm12 grafts in immunocompetent C57BL/6 mice.
Various studies demonstrate that rejection of MHC class II mismatched tail-skin depends on the presence of allogeneic CD4+ T cells (rev. in12 ). Indeed, T cell deficient CD3ε-/- mice and T- and B-cells deficient Rag2-/- mice showed complete wound healing and tolerated the I-Abm12 grafts up to 100 days (Figure 2), whereas C57BL/6 mice rejected grafts within 13 days (Figure 2). The acceptance of I-Abm12 mismatched tail-skin allows for the study of allogeneic CD4+ T cells and their effector functions in a mouse model without severe disease pathologies.
To break the established tolerance in CD3ε-/- and Rag2-/- mice, CD4+ T cells were adoptively transferred: 2 x 104 polyclonal C57BL/6 CD4+ T cells were sufficient to induce rejection of I-Abm12 grafts within 14 days (data not shown). Transfer of 50,000 and 100,000 C57BL/6 CD4+ T cells did not accelerate the graft rejection process in CD3ε-/- and Rag2-/- mice. These data underline the role of allogeneic CD4+ T cells in the acute skin graft rejection model.
The advantage of this tail-skin transplantation model is that functional analysis of CD4+ T cells can be easily done by adoptive transfer of antigen-specific CD4+ T cells. The transgenic ABM mouse9 is considered an ideal tool to delineate the activation and the phenotypic behavior of the population without the influence of non-specific CD4+ T cells. The transfer of 2 x 104 antigen-specific ABM cells was sufficient to induce rejection of I-Abm12 grafts in Rag2-/- mice within 13 days (Figure 3A-B). In addition, ABM cells are easy to isolate and can be tracked using the expression of the Vα2Vβ8-TcR chains (Figure 3C). ABM cell proliferation and production of cytokines in I-Abm12 transplanted Rag2-/- mice were investigated. ABM cells proliferated extensively as determined by eFluor670 dilution assay (Figure 3D) and up to 30% of the ABM cells produced IFN-γ upon PMA-stimulation (Figure 3E).
Taken together, these data show that relatively few allogeneic CD4+ T cells are required to induce rejection of I-Abm12 skin grafts, rendering this model ideal for the analysis of allogeneic T cells with minimal severe pathology.
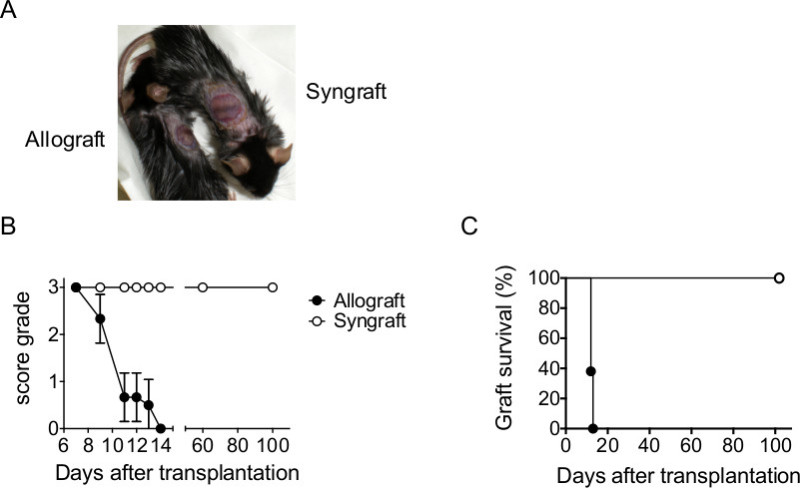 Figure 1. Transplantation of mice with allogeneic and syngeneic tail-skin. (A) C57BL/6 mice were transplanted with allogeneic (I-Abm12) and syngeneic (I-Ab) tail-skin (n = 5 per group). Pictures illustrate allografts (left) and syngrafts (right) after bandage removal on day 7. (B) Scoring and (C) survival of skin grafts from day 7 until day of rejection or final monitoring (day 100). Please click here to view a larger version of this figure.
Figure 1. Transplantation of mice with allogeneic and syngeneic tail-skin. (A) C57BL/6 mice were transplanted with allogeneic (I-Abm12) and syngeneic (I-Ab) tail-skin (n = 5 per group). Pictures illustrate allografts (left) and syngrafts (right) after bandage removal on day 7. (B) Scoring and (C) survival of skin grafts from day 7 until day of rejection or final monitoring (day 100). Please click here to view a larger version of this figure.
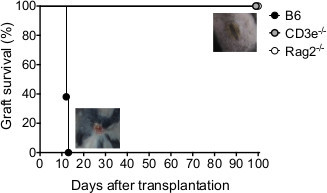 Figure 2. Allografts are tolerated in the absence of T cells in mice. Survival of Rag2-/-- I-Abm12- mouse skin grafts transplanted in C57BL/6, CD3ε-/- and Rag2-/- mice (n = 4-6 per group) and illustration of rejected and accepted grafts. Please click here to view a larger version of this figure.
Figure 2. Allografts are tolerated in the absence of T cells in mice. Survival of Rag2-/-- I-Abm12- mouse skin grafts transplanted in C57BL/6, CD3ε-/- and Rag2-/- mice (n = 4-6 per group) and illustration of rejected and accepted grafts. Please click here to view a larger version of this figure.
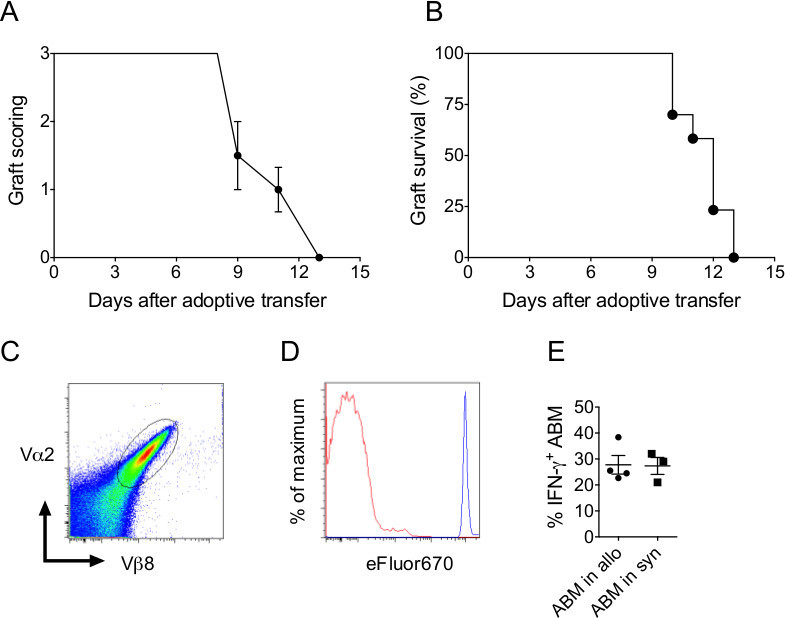 Figure 3. Anti-I-Abm12 specific CD4+ T cells proliferate and produce IFN-γ during graft rejection.(A) Scoring and (B) survival of I-Abm12 tail-skin grafts in Rag2-/- mice (n = 5) adoptively transferred with 2 x 104 antigen-specific ABM cells. (C) Vα2Vβ8-staining of ABM cells isolated from lymph nodes of transgenic ABM mice. (D) eFluor670 dilution of proliferating ABM cells and (E) IFN-γ production of PMA-stimulated ABM cells isolated from lymph nodes of I-Abm12 skin grafted Rag2-/- mice on day 9 after adoptive ABM cell transfer (n = 5 mice). Please click here to view a larger version of this figure.
Figure 3. Anti-I-Abm12 specific CD4+ T cells proliferate and produce IFN-γ during graft rejection.(A) Scoring and (B) survival of I-Abm12 tail-skin grafts in Rag2-/- mice (n = 5) adoptively transferred with 2 x 104 antigen-specific ABM cells. (C) Vα2Vβ8-staining of ABM cells isolated from lymph nodes of transgenic ABM mice. (D) eFluor670 dilution of proliferating ABM cells and (E) IFN-γ production of PMA-stimulated ABM cells isolated from lymph nodes of I-Abm12 skin grafted Rag2-/- mice on day 9 after adoptive ABM cell transfer (n = 5 mice). Please click here to view a larger version of this figure.
| Graft Score | Description |
| 3+ | No red areas and presence of a shiny black stripe |
| 2+ | Appearance of small red areas, loss of a shiny black stripe and dryness |
| 1+ | Appearance of large red areas, no black stripe and shrinkage |
| 0 | Graft rejection defined as 80% necrosis |
Table 1. Graft score system. The appearance of the transplant and its rejection is categorized in 3 grades, taking advantage of the different hair textures on the tail- and dorsal-skin.
Discussion
Skin transplantation is a widely used method for studying rejection and tolerance dependent on T cells. Since the skin transplantation model was established, several adaptations and changes have been applied. In the described procedure, I-Abm12 tail-skin transplantation is performed using anesthetic gas (isofluorane). The use of gas anesthesia decreases the time of execution and mice recovery, which diminishes the stress on transplanted mice. The procedure employs tissue adhesive to fix the tail-skin, which tends to curl more than ear or dorsal-skin tissue. Moreover it is important to flatten the tail-skin before cutting and trimming the edges as this reduces curling and improves positioning of the tail-skin on the dorsal excision site.
To assess graft rejection or acceptance it is required to prepare the tail-skin with the black stripe (e.g., in C57BL/6 mice) in the middle of the graft to facilitate scoring of transplants based on hair texture differences.
The mutation of the MHC Class II I-Abm12 molecule exclusively activates CD4+ T cells in immunocompetent and immunodeficient mice (of the same genetic background). On the one hand this facilitates the interpretation of the results, but on the other hand it limits the method to CD4+ T cells studies. To extend T cell activation to other T cell subsets, tail-skin from other donors, e.g., same genetic background including I-Abm1 mice and different genetic background including Balb/c or DBA mice can be transplanted.
In the acute rejection studies, we benefit from the naturally occurring mutation in the MHC class II groove and the activation of allogeneic CD4+ T cells to study organ rejection in mice. C57BL/6 mice rejected I-Abm12 skin grafts within 14 days after transplantation, which is consistent with most studies using I-Abm12 skin transplants15-20. In addition the acute rejection model is widely used to test the allogeneic potential of CD4+ T cells from gene-deficient mice. Indeed, IFN-γ-/- mice showed delayed I-Abm12 skin graft rejection (unpublished data and16). Blocking IFN-γ by administration of blocking antibody similarly delayed rejection15,17,18. This indicates a key role of IFN-γ in initiating MHC class II expression on Ag-presenting and endothelial cells (rev. in 12). Since perforin-deficient mice did not delay I-Abm12 skin graft rejection21, the role of cytokines seems to be more important for rejection. Indeed, IL-6 administration resulted in prolonged graft survival, possibly by downregulating IFN-γ-production15. In addition, neutrophils seem to play a major role in the rejection of I-Abm12 skin grafts in IL-4-deficient mice19 indicating a fine-tuning of innate and adaptive immune cell derived cytokines. This hypothesis is strengthened by the observation that ABT-100 blocks cytokine secretion and delays I-Abm12 graft rejection in mice20. Administration of anti-CD3 antibody delays I-Abm12 skin graft rejection in immunocompetent mice (unpublished data). Moreover, we showed that T cell deficient CD3ε-/- and Rag2-/- mice accept the allogeneic skin grafts for up to 100 days. Tolerance in these mice can be easily reverted/broken by adoptive transfer of CD4+ T cells from immunocompetent C57BL/6 mice. Transfer of 2 x 104 CD4+ T cells or antigen-specific ABM cells is sufficient to induce I-Abm12 graft rejection around day 14. Isolated Vα2Vβ8-positive ABM cells showed extensive proliferation and IFN-γ production in the lymph nodes before rejection, however rejection was dependent on I-Abm12 expression in the skin graft.
In summary, this mouse model is an excellent way to study activation of low T cell numbers during rejection or tolerance. The tail-skin transplantation is easy to perform and can be applied to mice with different genetic backgrounds to study the allogeneic responses of T cells without incurring a severe physiological effect. Moreover, this model is ideal for interventions with chemical and biological substances that induce or break skin graft tolerance.
Disclosures
The authors declare that they have no competing financial interests.
Acknowledgments
This work was supported by SNF grants PPOOA-_119204 and PPOOP3_144918 to S.W.R. We thank E. Palmer and B.T.H. Hausmann for mice and technical expertise.
References
- Mahillo B, Carmona M, Álvarez M, White S, Noel L, Matesanz R. Global Data in Organ Donation and Transplantation. transplantation. 2011;92(10):1069–1074. doi: 10.1097/TP.0b013e31823360b1. [DOI] [PubMed] [Google Scholar]
- Halleck F, et al. New perspectives of immunosuppression. Transplantation proceedings. 2013;45(3):1224–1231. doi: 10.1016/j.transproceed.2013.02.033. [DOI] [PubMed] [Google Scholar]
- Wood KJ, Bushell A, Jones ND. Immunologic unresponsiveness to alloantigen in vivo. a role for regulatory T cells. Immunological reviews. 2011;241(1):119–132. doi: 10.1111/j.1600-065X.2011.01013.x. [DOI] [PubMed] [Google Scholar]
- Sykes M. Immune tolerance: mechanisms and application in clinical transplantation. Journal of Internal Medicine. 2007;262(3):288–310. doi: 10.1111/j.1365-2796.2007.01855.x. [DOI] [PubMed] [Google Scholar]
- McFarland HI, Rosenberg AS. Skin allograft rejection. Current protocols in immunology. 2009. [DOI] [PubMed]
- McKenzie IF, Morgan GM, Sandrin MS, Michaelides MM, Melvold RW, Kohn HI. B6.C-H-2bm12. A new H-2 mutation in the I region in the mouse. The Journal of experimental medicine. 1979;150(6):1323–1338. doi: 10.1084/jem.150.6.1323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McIntyre KR, Seidman JG. Nucleotide sequence of mutant I-A beta bm12 gene is evidence for genetic exchange between mouse immune response genes. Nature. 1984;308(5959):551–553. doi: 10.1038/308551a0. [DOI] [PubMed] [Google Scholar]
- Stuart PM, Beck-Maier B, Melvold RW. Provocation of skin graft rejection across murine class II differences by non--bone-marrow-derived cells. Transplantation. 1984;37(4):393–396. doi: 10.1097/00007890-198404000-00016. [DOI] [PubMed] [Google Scholar]
- Hausmann B, Palmer E. Positive selection through a motif in the alphabeta T cell receptor. Science. 1998;281(5378):835–838. doi: 10.1126/science.281.5378.835. [DOI] [PubMed] [Google Scholar]
- Bill J, Ronchese F, Germain RN, Palmer E. The contribution of mutant amino acids to alloantigenicity. The Journal of experimental medicine. 1989;170(3) doi: 10.1084/jem.170.3.739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Monaco AP. Immunosuppression and tolerance for clinical organ allografts. Current Opinion in Immunology. 1989;1(6):1174–1177. doi: 10.1016/0952-7915(89)90012-5. [DOI] [PubMed] [Google Scholar]
- Rosenberg AS, Singer A. Cellular basis of skin allograft rejection: an in vivo model of immune-mediated tissue destruction. Annual Review of Immunology. 1992;10:333–358. doi: 10.1146/annurev.iy.10.040192.002001. [DOI] [PubMed] [Google Scholar]
- Kingsley CI, Nadig SN, Wood KJ. Transplantation tolerance: lessons from experimental rodent models. Transplant international : official journal of the European Society for Organ Transplantation. 2007;20(10):828–841. doi: 10.1111/j.1432-2277.2007.00533.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Larsen CP, et al. Long-term acceptance of skin and cardiac allografts after blocking CD40 and CD28 pathways. Nature. 1996;381(6581):434–438. doi: 10.1038/381434a0. [DOI] [PubMed] [Google Scholar]
- Tomura M, Nakatani I, Murachi M, Tai XG, Toyo-oka K, Fujiwara H. Suppression of allograft responses induced by interleukin-6, which selectively modulates interferon-gamma but not interleukin-2 production. Transplantation. 1997;64(5):757–763. doi: 10.1097/00007890-199709150-00017. [DOI] [PubMed] [Google Scholar]
- Ring GH, et al. Interferon-gamma is necessary for initiating the acute rejection of major histocompatibility complex class II-disparate skin allografts. Transplantation. 1999;67(10):1362–1365. doi: 10.1097/00007890-199905270-00012. [DOI] [PubMed] [Google Scholar]
- Rosenberg AS, Finbloom DS, Maniero TG, Vander Meide PH, Singer A. Specific prolongation of MHC class II disparate skin allografts by in vivo administration of anti-IFN-gamma monoclonal antibody. Journal of immunology. 1950;144(12):4648–4650. [PubMed] [Google Scholar]
- Goes N, Sims T, Urmson J, Vincent D, Ramassar V, Halloran PF. Disturbed MHC regulation in the IFN-gamma knockout mouse. Evidence for three states of MHC expression with distinct roles for IFN-gamma. Journal of immunology. 1995;155(10):4559–4566. [PubMed] [Google Scholar]
- Surquin M, et al. IL-4 deficiency prevents eosinophilic rejection and uncovers a role for neutrophils in the rejection of MHC class II disparate skin grafts. Transplantation. 2005;80(10):1485–1492. doi: 10.1097/01.tp.0000176486.01697.3f. [DOI] [PubMed] [Google Scholar]
- Gaylo AE, Laux KS, Batzel EJ, Berg ME, Field KA. Delayed rejection of MHC class II-disparate skin allografts in mice treated with farnesyltransferase inhibitors. Transplant immunology. 2009;20(3):163–170. doi: 10.1016/j.trim.2008.09.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bose A, Inoue Y, Kokko KE, Lakkis FG. Cutting edge: perforin down-regulates CD4 and CD8 T cell-mediated immune responses to a transplanted organ. Journal of immunology. 2003;170(4):1611–1614. doi: 10.4049/jimmunol.170.4.1611. [DOI] [PubMed] [Google Scholar]


