Summary
Background
Diabetes has been defined on the basis of different biomarkers, including fasting plasma glucose (FPG), 2-h plasma glucose in an oral glucose tolerance test (2hOGTT), and HbA1c. We assessed the effect of different diagnostic definitions on both the population prevalence of diabetes and the classification of previously undiagnosed individuals as having diabetes versus not having diabetes in a pooled analysis of data from population-based health examination surveys in different regions.
Methods
We used data from 96 population-based health examination surveys that had measured at least two of the biomarkers used for defining diabetes. Diabetes was defined using HbA1c (HbA1c ≥6·5% or history of diabetes diagnosis or using insulin or oral hypoglycaemic drugs) compared with either FPG only or FPG-or-2hOGTT definitions (FPG ≥7·0 mmol/L or 2hOGTT ≥11·1 mmol/L or history of diabetes or using insulin or oral hypoglycaemic drugs). We calculated diabetes prevalence, taking into account complex survey design and survey sample weights. We compared the prevalences of diabetes using different definitions graphically and by regression analyses. We calculated sensitivity and specificity of diabetes diagnosis based on HbA1c compared with diagnosis based on glucose among previously undiagnosed individuals (ie, excluding those with history of diabetes or using insulin or oral hypoglycaemic drugs). We calculated sensitivity and specificity in each survey, and then pooled results using a random-effects model. We assessed the sources of heterogeneity of sensitivity by meta-regressions for study characteristics selected a priori.
Findings
Population prevalence of diabetes based on FPG-or-2hOGTT was correlated with prevalence based on FPG alone (r=0·98), but was higher by 2–6 percentage points at different prevalence levels. Prevalence based on HbA1c was lower than prevalence based on FPG in 42·8% of age–sex–survey groups and higher in another 41·6%; in the other 15·6%, the two definitions provided similar prevalence estimates. The variation across studies in the relation between glucose-based and HbA1c-based prevalences was partly related to participants' age, followed by natural logarithm of per person gross domestic product, the year of survey, mean BMI, and whether the survey population was national, subnational, or from specific communities. Diabetes defined as HbA1c 6·5% or more had a pooled sensitivity of 52·8% (95% CI 51·3–54·3%) and a pooled specificity of 99·74% (99·71–99·78%) compared with FPG 7·0 mmol/L or more for diagnosing previously undiagnosed participants; sensitivity compared with diabetes defined based on FPG-or-2hOGTT was 30·5% (28·7–32·3%). None of the preselected study-level characteristics explained the heterogeneity in the sensitivity of HbA1c versus FPG.
Interpretation
Different biomarkers and definitions for diabetes can provide different estimates of population prevalence of diabetes, and differentially identify people without previous diagnosis as having diabetes. Using an HbA1c-based definition alone in health surveys will not identify a substantial proportion of previously undiagnosed people who would be considered as having diabetes using a glucose-based test.
Funding
Wellcome Trust, US National Institutes of Health.
Introduction
Diabetes prevalence and diabetes-related deaths are rising in most parts of the world, at least partly fuelled by the worldwide increase in excess weight and adiposity.1, 2, 3, 4, 5 This trend has created concerns about the health and functional consequences for patients, and costs for health systems.6, 7, 8 Tracking the epidemic and the progress of programmes aimed at reducing diabetes and its complications requires consistent and comparable measurement of the prevalence of diabetes and the coverage of drug and lifestyle interventions that slow diabetes progression and decrease the risk of complications.
Different biomarkers have been used to define diabetes, including fasting plasma glucose (FPG), 2-h plasma glucose in an oral glucose tolerance test (2hOGTT), and, more recently, HbA1c.9, 10, 11, 12, 13, 14, 15 Population-based health surveys in different countries and at different times have also used different biomarkers for glycaemia and diabetes, and thus define diabetes differently. The variety of biomarkers and definitions creates a challenge in consistently analysing diabetes prevalence across countries and over time, and in measuring what proportion of people with diabetes are diagnosed and receive effective treatments for diabetes and its complications.1, 16, 17 Therefore, there is a need to understand how the use of different biomarkers and definitions affects the identification of diabetes cases and the resulting estimates of population prevalence. This need is particularly pressing because two of the nine global targets for non-communicable diseases set after the 2011 United Nations high-level meeting on non-communicable diseases require estimates of diabetes prevalence: to halt the rise in the prevalence of diabetes, and to achieve a 50% coverage of drug treatment and counselling, including glycaemic control, to prevent coronary heart disease and stroke in people at high risk of cardiovascular disease.4, 18 Diabetes is also one of the four main non-communicable diseases for which there is a global target of 25% reduction in premature mortality by 2025 compared with 2010.4, 18
Research in context.
Evidence before this study
We reviewed studies included in the NCD Risk Factor Collaboration databases for comparisons of various diabetes definitions. We also searched PubMed with the term ((A1c[Title/Abstract]) AND Sensitivity[Title/Abstract]) AND Specificity[Title/Abstract]) on April 13, 2015. We also searched the references of recent reviews and guidelines. We found some studies on the classification of individuals as having diabetes or on comparison of prevalence estimates based on different definitions in specific cohorts, especially for HbA1c compared with either fasting plasma glucose (FPG) or 2-h oral glucose tolerance test (2hOGTT). Most of these analyses were based on a single cohort and very few covered different world regions. Two pooled analyses of Asian and European cohorts, and a study in the Pacific and Indian Ocean islands, assessed how the prevalence of diabetes and the classification of individuals as having diabetes versus not having diabetes changed depending whether diabetes was based on FPG or on 2hOGTT. There is no pooling study for HbA1c and we identified only one review of data from six countries. Other studies compared different diabetes definitions among people with specific pre-existing diseases—eg, heart disease and tuberculosis. We also found some prospective studies that assessed how HbA1c predicts future incidence of diabetes or cardiovascular diseases with mixed results.
Added value of this study
This study is the first pooling of a large number of population-based data from different world regions that addresses how different definitions of diabetes affect both the total prevalence, and the identification of previously undiagnosed individuals. By pooling a large number of data sources, the overall meta-analytical finding overcomes between-study variation, which can be probed in meta-regressions. Furthermore, by having a large number of studies, and age–sex groups within each study, we were able to develop regressions to convert across different diabetes definitions, which is essential for enhancing comparability over time and across countries in surveillance.
Implications of all the available evidence
The use of HbA1c in surveillance requires further consideration in terms of how it predicts, and helps prevent, diabetes complications and sequelae. As such studies are done, and to maximise comparability of results across surveys, the best approach in population-based health surveys is to measure FPG and define diabetes as FPG 7·0 mmol/L or more or history of diagnosis with diabetes or using insulin or oral hypoglycaemic drugs, as used in the global monitoring framework for prevention and control of non-communicable diseases. When HbA1c is used, it would be valuable to also measure FPG in a subsample of participants to provide information about how the two tests relate. The conversion regressions developed here can be used to convert prevalence based on FPG to that based on FPG-or-2hOGTT.
Some studies have analysed the classification of individuals as having diabetes or compared prevalence estimates based on different definitions in specific cohorts, especially for HbA1c compared with either FPG or 2hOGTT.19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61 Most of these analyses were based on a single cohort and very few covered different geographical regions. Two pooled analyses of Asian and European cohorts, and a study in the Pacific and Indian Ocean islands, assessed how the prevalence of diabetes and the classification of individuals as having diabetes versus not having diabetes changed depending on whether diabetes was defined by FPG or 2hOGTT.62, 63, 64, 65, 66 There is no pooling study for HbA1c, which can be measured easily in population-based surveys without the need for overnight fasting and has been approved by the American Diabetes Association and WHO as a diagnostic test for diabetes.11, 14 However, a review of data from six countries reported that the sensitivity of diabetes diagnosis based on HbA1c compared with FPG ranged from 17% to 78%,67 raising concerns about ethnic variation of HbA1c-based definition.17
We assessed the effect of diagnostic definitions both on the identification of diabetes in previously undiagnosed individuals and on the population prevalence estimates for diabetes in a pooled analysis of data from population-based health examination surveys in different world regions.
Methods
Study design
We aimed to answer two questions. First, how does the estimated prevalence of diabetes in a population change when the new definition of diabetes based on HbA1c is used compared with earlier definitions based on blood glucose? Second, how does the new definition of diabetes based on HbA1c compare with earlier definitions in identifying previously undiagnosed people with diabetes, as measured by the sensitivity and specificity of the new definition with respect to the previous ones? We further assessed whether sensitivity varied by the characteristics of the study population, because this possible variation is a source of concern about the generalisability of HbA1c as a diagnostic and surveillance measure.17, 67, 68, 69, 70
For the HbA1c-based definition of diabetes, we used HbA1c of 6·5% or more, or history of diagnosis with diabetes or using insulin or oral hypoglycaemic drugs.11 For definitions based on blood glucose, we used either the American Diabetes Association definition of FPG of 7·0 mmol/L or more, or history of diagnosis with diabetes or using insulin or oral hypoglycaemic drugs (which is also used in the global monitoring framework for prevention and control of non-communicable diseases),12, 18 or the WHO definition of FPG of 7·0 mmol/L or more, or 2hOGTT of 11·1 mmol/L or more, or history of diabetes or using insulin or oral hypoglycaemic drugs.9, 10
Data sources
We used population-based data collated by the NCD Risk Factor Collaboration (NCD-RisC), a worldwide network of health researchers and practitioners who, together with WHO, have collated a large database of population-based health examination surveys and epidemiological studies of cardiometabolic risk factors. All data sources were checked by at least two independent reviewers as being representative of a national, subnational, or community population, and for study quality indicators such as fasting duration and the protocol for OGTT. We excluded surveys that had not used a standard glucose load for OGTT. Within each survey, we included participants aged 18 years and older who were not pregnant and had fasted at least for 6 h before measurement as a part of the survey instructions. We excluded HbA1c data from before the year 2000 to minimise the use of non-standard assays.71 We also excluded surveys that had measured a biomarker only among participants with a high value of another—eg, studies in which FPG was only measured in participants with HbA1c above a prespecified value, because the relation between the two measurements might be different in this prescreened group compared with the whole sample. The appendix shows details of individual surveys.
We restricted the analysis of sensitivity and specificity to people without a history of diabetes diagnosis, because previous diagnosis and the use of drug treatments probably affect the concentrations of biomarkers used to diagnose diabetes. History of diabetes diagnosis was established with survey-specific questions, such as “have you ever been told by a doctor or other health professional that you have diabetes?” or the combination of “do you now have, or have you ever had diabetes?” and “were you told by a doctor that you had diabetes?”. We also excluded follow-up surveys of closed cohorts from the analysis of sensitivity and specificity because active surveillance within a cohort shifts participants from undiagnosed to diagnosed status at each follow-up, thus affecting the composition of undiagnosed cases.
Statistical analysis
We calculated diabetes prevalence by sex and age group, taking into account complex survey design and survey sample weights when relevant. We excluded age–sex groups with fewer than 25 participants when calculating prevalence because the sampling error of estimated prevalence can bias the associations between prevalences based on different definitions. Some surveys had measured HbA1c or FPG in all participants, but had not measured 2hOGTT among people with diagnosed diabetes. These previously diagnosed participants were included in calculation of diabetes prevalence because their exclusion would underestimate diabetes prevalence. Furthermore, some surveys measured 2hOGTT in only a subset of people without history of diabetes diagnosis, generally for logistical or cost reasons. Simply combining these participants with previously diagnosed participants might overestimate diabetes prevalence based on 2hOGTT. To account for these missing measurements, and to avoid overestimation of diabetes prevalence, we recalculated the survey sample weights for these participants as the original sample weights divided by weighted proportion of non-diabetic participants with data. This approach is similar to that used in the US National Health and Nutrition Examination Survey for their 2hOGTT sample weights.72 A similar approach was taken in a few surveys that had measured HbA1c in all participants, but had not measured FPG among people diagnosed with diabetes.
We compared graphically the prevalences of diabetes using different definitions. We also did regression analyses of the relation between diabetes defined (1) on the basis of FPG-or-2hOGTT versus on the basis of FPG only and (2) on the basis of HbA1c versus on the basis of FPG. We did not do a regression for diabetes prevalence based on HbA1c versus prevalence based on FPG-or-2hOGTT because very few surveys had data for both 2hOGTT and HbA1c, leading to unstable regression coefficients. We probit-transformed diabetes prevalence because it provided better fit to the data and it avoids predicting prevalences that are less than 0 or greater than 1. We considered regression models with alternative covariates and specifications, and chose the best model using the Bayesian information criterion, which measures the relative goodness of fit of a model; it rewards how well the model fits the data but discourages overfitting.73 The regressions included age (mean age of each age–sex group); the years over which each survey collected data (as the midyear of the period of data collection; appendix); national income (natural logarithm of per person gross domestic product) in the survey country and year; whether the study was representative of a national, subnational, or community population; and mean BMI for each age–sex group. Sex was excluded from the regressions on the basis of the Bayesian information criterion. The regression of diabetes prevalence based on HbA1c against diabetes prevalence based on FPG, for which there were more data, also included terms for geographical region as random effects on the basis of Bayesian information criterion; these random effects account for differences in the relationship by region. Two regions consisted of high-income countries, as in previous global analyses5, 74—high-income Asia Pacific (consisting of Japan, Singapore, and South Korea) and high-income western countries (consisting of countries in Australasia, North America, and western Europe). The other countries were divided based on their geography into central and eastern Europe; central Asia, Middle East and north Africa; east and southeast Asia; south Asia; Latin America and the Caribbean; and sub-Saharan Africa.
We plotted the residuals of the regression models against the main independent variable (probit-transformed FPG-based prevalence), and found no evidence of heteroscedasticity in the residuals. We also report the univariate and semipartial R2 for each of the variables in the regression model. Univariate R2 measures how much of the variance is explained by each independent variable. Semipartial R2 measures the contribution of each variable to the total explained variance, conditional on the presence of the other model variables.75
We calculated sensitivity and specificity of diagnosis separately in each survey, and then pooled the sensitivities and specificities across surveys with a random-effects model.76 We examined the sources of heterogeneity in sensitivity and specificity with metaregressions and a-priori selected study characteristics: mean age, proportion of male participants, midyear of study data collection period; sample size; prevalence of undiagnosed diabetes in the survey; whether the survey was representative of a national, subnational, or community population; geographical region; national income in the survey country and year; and mean haemoglobin concentration in the survey country and year. We did the analyses with Stata (version 12.2) and R (version 3.0.3).
Role of the funding source
The funders had no role in study design, data collection, analysis, or interpretation, or writing of the report. SF, YL, and BZ had full access to all the data. ME was responsible for submitting the Article for publication.
Results
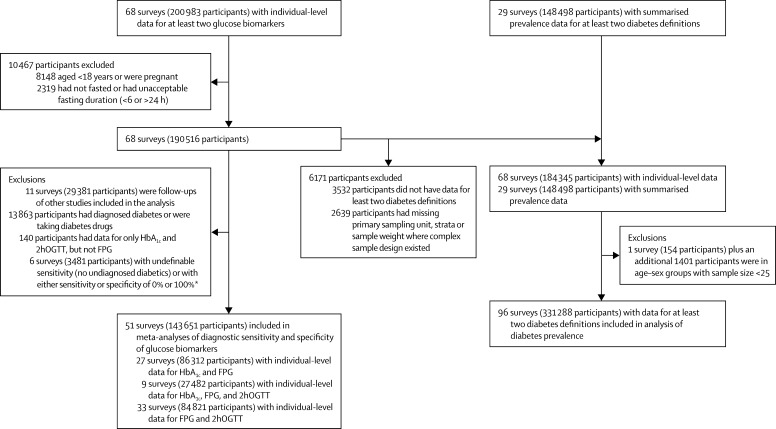
After exclusions, we included 96 population-based health examination surveys of 331 288 participants (figure 1). 46 surveys were from Australia, USA, and western Europe; 18 from east and southeast Asia; ten from Latin America and the Caribbean; seven from Oceania; six from sub-Saharan Africa; five from south Asia; three from the Middle East and north Africa; and one from central and eastern Europe. All 96 studies measured FPG; 47 also measured 2hOGTT and 63 measured HbA1c (appendix). 14 of these studies measured all three biomarkers. All but three studies of the 47 studies used for comparing prevalence based on FPG alone versus based on FPG-or-2hOGTT measured FPG in a laboratory; two of the remaining studies used a portable unit, and we did not have information for the remaining study. All studies measured 2hOGTT in a laboratory. All but one of the 63 studies used for comparing glucose-based and HbA1c-based prevalences measured glucose in a laboratory; the remaining study measured FPG with a portable unit. An enzymatic method was used to measure FPG in 65 of the 92 studies that had measured FPG in a laboratory, but we had no information for the remaining 27 studies. In all 63 studies, HbA1c was measured in a laboratory; in 40 of these studies, the measurements were done by chromatography or immunoassay. No information was available for the remaining 23. Such a dominance of laboratory-based measurements prevented us from assessing the role of measurement method as a source of variation because laboratory-based methods are equally acceptable, especially for glucose.77
Figure 1.
Study and data inclusion
FPG=fasting plasma glucose. 2hOGTT=2-h oral glucose tolerance test. *The meta-analyses used inverse of variance as survey weights; sensitivity or specificity of either 0% or 100% would make the corresponding variance zero, and therefore the inverse of variance infinite.
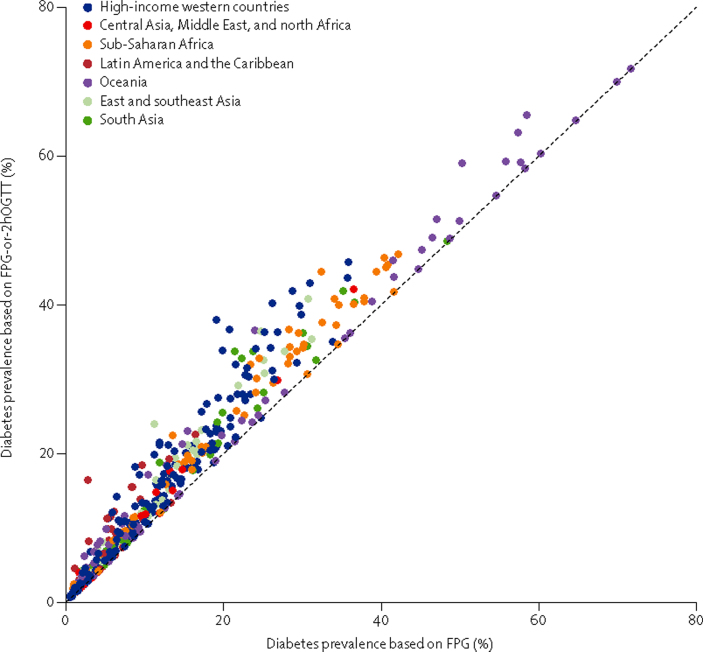
Diabetes prevalence ranged from 0% in people younger than 40 years of age in some surveys to about 70% in middle-aged and older adults in Nauru (figure 2). Prevalence of diabetes based on FPG alone was lower than that based on FPG-or-2hOGTT, by 2–6 percentage points at different prevalence levels, although prevalences estimated using these two glucose-based measures were highly correlated (r=0·98; figure 2). Table 1, Table 2 show results of the regression analyses. After accounting for prevalence based on FPG, prevalence based on FPG-or-2hOGTT increased with age—ie, prevalence based on FPG-or-2hOGTT rose more sharply with age than did prevalence based on FPG only.65, 80, 81
Figure 2.
Prevalence of diabetes defined by FPG-or-2hOGTT versus by FPG only
FPG-or-2hOGTT definition was FPG 7·0 mmol/L or more, or 2hOGTT 11·1 mmol/L or more, or history of diabetes or using insulin or oral hypoglycaemic drugs. FPG only definition was FPG 7·0 mmol/L or more, or history of diabetes or using insulin or oral hypoglycaemic drugs. Each point shows one age–sex group in one survey. Table 1 shows the relation summarised as regression coefficients. FPG=fasting plasma glucose. 2hOGTT=2-h oral glucose tolerance test.
Table 1.
Regression coefficients for the relation between probit-transformed prevalence of diabetes based on FPG-or-2hOGTT versus diabetes based on FPG only
| Coefficient (95% CI) | p value | Univariate R2* | Semipartial R2† | ||
|---|---|---|---|---|---|
| Intercept | 0·135 (−0·020 to 0·290) | 0·0872 | NA | NA | |
| Probit-transformed prevalence of diabetes based on FPG | 0·903 (0·880 to 0·927) | <0·0001 | 0·963 | 0·368 | |
| Mean age of age–sex group (per 10 years older) | 0·048 (0·039 to 0·056) | <0·0001 | 0·444 | 0·008 | |
| Study midyear (per one more recent year since 1976) | −0·001 (−0·002 to 0·000) | 0·1643 | 0·003 | <0·001 | |
| Natural logarithm of per person gross domestic product | −0·033 (−0·046 to −0·019) | <0·0001 | 0·004 | 0·001 | |
| Mean BMI | 0·000 (−0·004 to 0·004) | 0·9057 | 0·092 | <0·001 | |
| Study representativeness | .. | .. | 0·021 | 0·001 | |
| National | Reference | .. | .. | .. | |
| Subnational | −0·031 (−0·070 to 0·008) | 0·1141 | .. | .. | |
| Community | −0·070 (−0·101 to −0·039) | <0·0001 | .. | .. | |
FPG=fasting plasma glucose. 2hOGTT=2-h oral glucose tolerance test.
Calculated by regressing against each independent variable alone; equals the square of the correlation coefficient.
Shows how much R2 decreases if that independent variable is removed from the full model; the overall R2 for the model was 0·973.
Table 2.
Regression coefficients for the association between probit-transformed prevalence of diabetes based on HbA1c and probit-transformed prevalence based on FPG
| Coefficient (95% CI) | p value* | Univariate R2† | Semipartial R2‡ | ||
|---|---|---|---|---|---|
| Intercept | −1·761 (−2·229 to −1·266) | <0·0001 | NA | NA | |
| Probit-transformed prevalence of diabetes based on FPG | 0·799 (0·763 to 0·835) | <0·0001 | 0·915 | 0·075 | |
| Mean age of age–sex group (per 10 years older) | 0·052 (0·042 to 0·062) | <0·0001 | 0·601 | 0·011 | |
| Study midyear (per one more recent year since 2000) | 0·012 (0·009 to 0·015) | <0·0001 | 0·014 | 0·006 | |
| Natural logarithm of per person gross domestic product | 0·076 (0·035 to 0·114) | 0·0001 | 0·052 | 0·003 | |
| Mean BMI | 0·018 (0·010 to 0·027) | <0·0001 | 0·022 | 0·002 | |
| Study representativeness | .. | .. | 0·013 | 0·004 | |
| National | Reference | .. | .. | .. | |
| Subnational | −0·004 (−0·047 to 0·040) | 0·8758 | .. | .. | |
| Community | 0·090 (0·060 to 0·119) | <0·0001 | .. | .. | |
The appendix shows regional random effects. FPG=fasting plasma glucose.
p values using likelihood ratio test, which compares the likelihood of the models with and without the variable of interest.78
Calculated by regressing against each independent variable alone, without the regional random effect; equals the square of the correlation coefficient.
Is the decrease of R2 if one of the independent variables is removed from the full model; however, traditional R2 is not clearly defined for mixed-effect models, we have used the conditional R2 that describes the proportion of variance explained by both fixed and random factors.79 The overall conditional R2 for the model was 0·949.
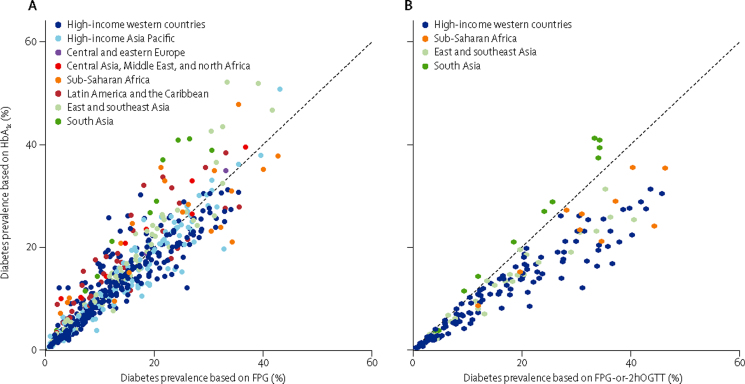
HbA1c-based prevalences were lower than those based on FPG for 42·8% of age–sex–survey groups and higher in another 41·6%; in the other 15·6%, the two definitions gave similar prevalences (figure 3). In the regression analysis, prevalence based on HbA1c was on average slightly lower than prevalence based on FPG (table 2). The most important determinant of variation between these two prevalences was age, with some effect from national income, mean BMI, year of survey, and whether the survey was representative of a national, subnational, or community population. After accounting for prevalence based on FPG, prevalence based on HbA1c increased with age, national income, mean BMI, and the year of survey. After accounting for prevalence based on FPG, HbA1c-based prevalence was higher in south Asia than in other regions, and was lower in high-income regions than in other regions (appendix).
Figure 3.
Prevalence of diabetes defined by HbA1c only versus prevalence defined by (A) FPG only, and (B) FPG-or-2hOGTT
HbA1c definition was HbA1c 6·5% or more, or history of diabetes, or using insulin or oral hypoglycaemic drugs. FPG only definition was FPG 7·0 mmol/L or more, or history of diabetes or using insulin or oral hypoglycaemic drugs. FPG-or-2hOGTT definition was FPG 7·0 mmol/L or more, or 2hOGTT 11·1 mmol/L or more, or history of diabetes or using insulin or oral hypoglycaemic drugs. Each point shows one age–sex group in one survey. Table 2 shows the relations summarised as regression coefficients. FPG=fasting plasma glucose. 2hOGTT=2-h oral glucose tolerance test.
Diabetes defined as HbA1c of 6·5% or more had a pooled sensitivity of 52·8% (95% CI 51·3–54·3) compared with a definition of FPG of 7·0 mmol/L or more for diagnosing participants without a previous diagnosis of diabetes. This finding suggests that 47·2% of participants without a previous diagnosis of diabetes who would be considered to have diabetes based on their FPG concentration would not be considered to have diabetes with an HbA1c test (table 3). The sensitivity of HbA1c varied substantially across studies (I2 of 97·6%), ranging from 13·0% to 93·2% (appendix pp 11–12). HbA1c had even lower sensitivity when compared with defining diabetes based on FPG-or-2hOGTT (30·5%, 95% CI 28·7–32·3). None of the preselected study-level characteristics explained the heterogeneity in the sensitivity of HbA1c versus FPG (all p values >0·06; table 4). Pooled specificity of HbA1c was 99·74% (95% CI 99·71–99·78) relative to FPG and 99·69% (99·63–99·76) relative to FPG-or-2hOGTT, suggesting few false positives compared with glucose-based definitions.
Table 3.
Pooled sensitivity and specificity of diabetes diagnosis using different definitions among participants without diagnosed diabetes
| Number of surveys | Sensitivity | Specificity | |||
|---|---|---|---|---|---|
| (%; 95% CI) | I2 | (%; 95% CI) | I2 | ||
| HbA1cvs FPG | 27 | 52·82 (51·33–54·30) | 97·6% | 99·74 (99·71–99·78) | 98·2% |
| HbA1cvs 2hOGTT | 9 | 37·16 (35·05–39·28) | 97·6% | 99·84 (99·79–99·89) | 97·3% |
| HbA1cvs FPG-or-2hOGTT | 9 | 30·46 (28·66–32·25) | 97·9% | 99·69 (99·63–99·76) | 98·0% |
| FPG vs 2hOGTT | 33 | 54·42 (53·26–55·57) | 96·9% | 98·90 (98·83–98·97) | 94·4% |
The appendix shows detailed results of these meta-analyses. Diabetes was defined as HbA1c ≥6·5%, FPG ≥7·0 mmol/L, and 2hOGTT ≥11·1 mmol/L. FPG=fasting plasma glucose. 2hOGTT=2-h oral glucose tolerance test.
Table 4.
Univariate metaregression coefficients for sensitivity of HbA1c versus FPG in participants without diagnosed diabetes
| Mean difference in sensitivity (percentage points; 95% CI) | p value | ||
|---|---|---|---|
| Mean age (per 10 years older) | −4·1 (−12·7 to 4·5) | 0·3361 | |
| Percent male participants (per 10% more male) | 4·6 (−9·0 to 18·2) | 0·4901 | |
| Study midyear (per one more recent year) | 1·2 (−0·9 to 3·2) | 0·2566 | |
| Region | .. | 0·2097 | |
| High-income western countries | Reference group | .. | |
| East, south, and southeast Asia | 21·0 (−0·3 to 42·2) | .. | |
| Latin America and the Caribbean | 8·5 (−17·9 to 34·9) | .. | |
| Sub-Saharan Africa | 17·6 (−14·1 to 49·2) | .. | |
| Study representativeness | .. | 0·0915 | |
| National | Reference group | .. | |
| Subnational | 1·7 (−28·6 to 31·9) | .. | |
| Community | 21·4 (2·1 to 40·8) | .. | |
| Prevalence of undiagnosed diabetes (percentage point higher undiagnosed diabetes) | −0·7 (−4·0 to 2·6) | 0·6780 | |
| Sample size (per 1000 participants without diagnosed diabetes) | −1·6 (−4·6 to 1·4) | 0·2730 | |
| Natural logarithm of per person gross domestic product | −6·5 (−17·6 to 4·6) | 0·2410 | |
| Mean haemoglobin (per g/L)* | −2·0 (−4·1 to 0·2) | 0·0677 | |
We used a HbA1c definition of 6·5% or more and a FPG definition of 7·0 mmol/L or more. FPG=fasting plasma glucose.
Reliable mean haemoglobin data were available only for women of child-bearing age.82 The national mean for each country-year was used for both men and women; restricting the analysis to women led to similar results, with a mean difference of −2·1 (−4·5 to 0·3, p=0·0929).
Lowering the threshold for diabetes by HbA1c from 6·5% to 6·3% (a cutoff suggested by some studies49, 50) increased sensitivity compared with the FPG-based definition from 52·8% to 64·3% while maintaining a high specificity at 99·53%. Lowering it further to 6·1% increased sensitivity to 72·8% but the specificity would drop to 99·08%, resulting in more false positives. Follow-up studies are needed to establish how these cutoffs predict complications and sequelae in newly diagnosed patients.83, 84
Discussion
In this large international pooled analysis of population-based health examination surveys, we found that the use of different biomarkers and definitions for diabetes can lead to different estimates of population prevalence of diabetes, with the highest prevalence when diabetes is defined on the basis of FPG-or-2hOGTT and the lowest when based on HbA1c alone. For example, at an FPG-based prevalence of 10%, similar to the age-standardised global prevalence of diabetes in adults aged 25 years and older in 2008,1 prevalence based on FPG-or-2hOGTT would be about 13% according to the relation in figure 2. The variation across studies in the relation between glucose-based and HbA1c-based prevalences was partly related to age, followed by national income, mean BMI, the year of survey, and whether the survey population was national, subnational, or from specific communities. The reasons for additional regional effects—higher HbA1c-based prevalence in south Asia and lower prevalence in high-income regions than in other regions after accounting for prevalence based on FPG—are unknown, but they might be a result of true physiological differences; for example, related to red blood cell turnover (itself related to anaemia and iron status), which affects HbA1c, or related to glucose dysregulation during fasting and non-fasting which are captured by HbA1c.85 Establishing these reasons requires multicentre studies with consistent methods and protocols and data for phenotypical factors that might affect the relation between glucose and HbA1c. For now, they are unexplained empirical results that should be taken into account when using surveys from different regions.
Similarly, different definitions identified different people without a previous diagnosis as having diabetes. Specifically, use of an HbA1c-based definition would not identify almost half of the undiagnosed cases that could be detected with a FPG test, and more than three-quarters of undiagnosed cases that would be detected by FPG and 2hOGTT combined, but it would lead to few false positives compared with glucose-based definitions. Inversely, using a glucose-based test alone would not identify some people who would be considered as having diabetes with HbA1c.
Our results, based on a large number of surveys from different regions, are consistent with previous smaller studies that compared different diabetes definitions. Diabetes prevalence based on FPG-or-2hOGTT was higher than prevalence based on FPG alone by 18% in an analysis of 13 European cohorts and by 6% in an analysis of 11 Asian cohorts.63, 64 A previous comparison of diabetes prevalence across six studies, including two analysed here, reported that diagnostic sensitivity for HbA1c compared with 2hOGTT ranged from 17% to 78%,67 which is consistent with the results of our analysis. However, this study also found surprisingly low specificities for HbA1c compared with ours.67 Other single-cohort studies also generally reported low but variable sensitivities and high specificities for HbA1c relative to blood glucose. Several studies86, 87, 88, 89 assessed the optimal cutoff for HbA1c in different populations and all reported values lower than 6·5%, which is consistent with our finding that lowering the threshold would increase sensitivity while preserving high specificity. One small study90 examined the effect of anaemia on diagnostic accuracy of HbA1c and reported higher sensitivity (than with FPG) in patients with anaemia, which is consistent with our results.
Our analysis, which focused on questions that are relevant for population-based surveillance of diabetes and monitoring treatment coverage, has several strengths. We pooled data from a large number of population-based surveys from different world regions, thereby increasing both the precision of our estimates and their generalisability compared with analyses of one or a small number of cohorts. We used consistent eligibility and inclusion criteria, and assessed whether the surveys met these criteria. In particular, we only used surveys that had rigorous protocols for fasting duration and for OGTT. Furthermore, most surveys measured glucose and HbA1c in a laboratory. We also assessed the sources of heterogeneity in how diagnostic criteria compare across surveys, which could not be done in previous analyses because they included few surveys.
Our results should be interpreted with some limitations in mind. We had few studies from some regions including sub-Saharan Africa, south Asia, the Middle East and north Africa, and central and eastern Europe. We analysed the surveys with consistent methods but surveys might have differed in details such as the exact limit for fasting duration beyond the 6-h limit imposed by us. Because HbA1c measurement has changed over time,91, 92, 93, 94, 95, 96, 97, 98, 99 and to minimise the use of non-standard assays, we did not include any HbA1c data from before the year 2000.71 Despite this exclusion, and the fact that all of our surveys had measured HbA1c in a laboratory, HbA1c measurements can vary between laboratories and instruments,100 about which we did not have complete data. For the same reason, we could not standardise the HbA1c data to account for different assays and instruments used in measurement. Nutritional status—especially iron deficiency—anaemia, malaria and other parasitic diseases, living at high altitudes, and high prevalence of haemoglobinopathies can affect HbA1c,101 but could not be assessed as a source of heterogeneity beyond their effects through mean haemoglobin concentration. Similarly, data for glucose can be affected by unrecorded factors such as inaccurate information about fasting, fluctuations in diet and physical activity in days before measurement, and how samples were handled, including time between drawing blood and laboratory analysis and the type of tube used for collecting and storing blood.
Although we assessed the role of geographical region, we did not have data for the ethnic composition of participants in each survey. By their nature, health examination surveys used for population-based surveillance use a single measurement for each participant, whereas diagnosis in a clinical setting might repeat the measurements based on the first test. The use of a single test is affected by within-individual and even within-laboratory variation, and could lead to misclassification of some individuals.99 Finally, we did not have longitudinal follow-up data to assess sensitivity and specificity for diagnosis using one definition (or one cutoff value of HbA1c) compared to another or for development of diabetes complications and sequelae that contribute the bulk of the public health burden of diabetes. Such data are not available in population-based surveys because surveys are typically cross-sectional.
There is no gold standard definition that captures the phenotypic complexity of diabetes and the risk of its microvascular and macrovascular complications, although 2hOGTT is often treated as the most reliable test.15, 102, 103 In clinical practice, physicians follow an analytical process to diagnose diabetes, in which different sequences of glucose biomarkers are used depending on factors such as a patient's age and symptoms; those with high levels of one biomarker (eg, HbA1c) might be asked to have additional measurements of the same or a different biomarker, and be monitored over time to decide on the best course of treatment. The process might vary from patient to patient to account for their unique characteristics, and might further vary from physician to physician based on available infrastructure and medical resources. In surveillance using population-based surveys, which provides evidence for policies and programmes related to whole populations, repeated measurements are virtually impossible. Therefore, considerations about diabetes definition and diagnosis are different from those of clinical practice, and the emphasis is on comparability of definitions over time and across populations. Our results provide much needed empirical evidence for planning global surveillance of diabetes and coverage of its interventions. Specifically, despite its relative ease of use, using HbA1c alone in health surveys might miss some previously undiagnosed people who would be considered as having diabetes using a glucose-based test, and thus could benefit from lifestyle and treatment interventions. Even so, 2hOGTT is difficult to measure even in a clinical setting, let alone in population-based surveys. Of 493 worldwide population-based diabetes data sources between 1975 and 2014 in the NCD-RisC databases, 448 had measured FPG but only 59 had measured 2hOGTT; 33% of surveys before 1990 had 2hOGTT and only 11% did after 1990. Therefore, a strategy for consistent and comparable surveillance is to use FPG in population-based surveys, be it national or multicountry survey programmes such as the WHO STEPS surveys, and define diabetes based on FPG. Data such as those in figure 2 and table 1 can then be used to relate prevalences based on FPG to those based on FPG-or-2hOGTT. The use of HbA1c in surveillance requires further consideration of how it predicts and helps prevent diabetes complications and sequelae. When HbA1c is used, FPG should ideally also be measured in a subsample of participants to provide information about how the two tests relate.
Correspondence to: Prof Majid Ezzati, Imperial College London, London W2 1PG, UK majid.ezzati@imperial.ac.uk
Acknowledgments
Acknowledgments
This study was funded by the Wellcome Trust and US National Institutes of Health (DK090435). The authors alone are responsible for the views expressed in this Article and they do not necessarily represent the views, decisions, or policies of the institutions with which they are affiliated.
Contributors
GD and ME designed the study and oversaw research. Members of the Country and Regional Data Group collected and reanalysed data, and checked pooled data for accuracy of information about their study and other studies in their country. Members of the Pooled Analysis and Writing Group collated data, checked all data sources in consultation with the Country and Regional Data Group, analysed pooled data, and prepared results. GD and ME wrote the first draft of the report with input from other members of Pooled Analysis and Writing Group. Members of Country and Regional Data Group commented on draft report.
NCD Risk Factor Collaboration (NCD-RisC)
Pooled Analysis and Writing (*equal contribution; listed alphabetically)—Goodarz Danaei (Harvard T H Chan School of Public Health, USA)*; Saman Fahimi (Harvard T H Chan School of Public Health, USA)*; Yuan Lu (Harvard T H Chan School of Public Health, USA)*; Bin Zhou (Imperial College London, UK)*; Kaveh Hajifathalian (Harvard T H Chan School of Public Health, USA); Mariachiara Di Cesare (Imperial College London, UK); Wei-Cheng Lo (National Taiwan University, Taiwan); Barbara Reis-Santos (Universidade Federal de Pelotas, Brazil); Melanie J Cowan (World Health Organization, Switzerland); Jonathan E Shaw (Baker IDI Heart and Diabetes Institute, Australia); James Bentham (Imperial College London, UK); John K Lin (University of California San Francisco, USA); Honor Bixby (Imperial College London, UK); Dianna Magliano (Baker IDI Heart and Diabetes Institute, Australia); Pascal Bovet (University of Lausanne, Switzerland; Ministry of Health, Seychelles); J Jaime Miranda (Universidad Peruana Cayetano Heredia, Peru); Young-Ho Khang (Seoul National University, South Korea); Gretchen A Stevens (World Health Organization, Switzerland); Leanne M Riley (World Health Organization, Switzerland); Mohammed K Ali (Emory University, USA); Majid Ezzati (Imperial College London, UK).
Country and Regional Data (*equal contribution; listed alphabetically)—Ziad A Abdeen (Al-Quds University, Palestine)*; Khalid Abdul Kadir (Monash University Malaysia, Malaysia)*; Niveen M Abu-Rmeileh (Birzeit University, Palestine)*; Benjamin Acosta-Cazares (Instituto Mexicano del Seguro Social, Mexico)*; Wichai Aekplakorn (Mahidol University, Thailand)*; Carlos A Aguilar-Salinas (Instituto Nacional de Ciencias Médicas y Nutricion, Mexico)*; Alireza Ahmadvand (Tehran University of Medical Sciences, Iran)*; Mohannad Al Nsour (Eastern Mediterranean Public Health Network, Jordan)*; Ala'a Alkerwi (Luxembourg Health Institute, Luxembourg)*; Philippe Amouyel (Lille University and Hospital, France)*; Lars Bo Andersen (University of Southern Denmark, Denmark)*; Sigmund A Anderssen (Norwegian School of Sport Sciences, Norway)*; Dolores S Andrade (Universidad de Cuenca, Ecuador)*; Ranjit Mohan Anjana (Madras Diabetes Research Foundation, India)*; Hajer Aounallah-Skhiri (National Institute of Public Health, Tunisia)*; Tahir Aris (Ministry of Health Malaysia, Malaysia)*; Nimmathota Arlappa (Indian Council of Medical Research, India)*; Dominique Arveiler (Strasbourg University and Hospital, France)*; Felix K Assah (Health of Populations in Transition Research Group, Cameroon)*; Mária Avdicová (Regional Authority of Public Health, Banska Bystrica, Slovakia)*; Nagalla Balakrishna (Indian Council of Medical Research, India)*; Piotr Bandosz (Medical University of Gdansk, Poland)*; Carlo M Barbagallo (University of Palermo, Italy)*; Alberto Barceló (Pan American Health Organization, USA)*; Anwar M Batieha (Jordan University of Science and Technology, Jordan)*; Louise A Baur (University of Sydney, Australia)*; Habiba Ben Romdhane (University Tunis El Manar, Tunisia)*; Antonio Bernabe-Ortiz (Universidad Peruana Cayetano Heredia, Peru)*; Santosh K Bhargava (Sunder Lal Jain Hospital, India)*; Yufang Bi (Shanghai Jiao-Tong University School of Medicine, China)*; Peter Bjerregaard (University of Southern Denmark, Denmark; University of Greenland, Greenland)*; Cecilia Björkelund (University of Gothenburg, Sweden)*; Margaret Blake (NatCen Social Research, UK)*; Anneke Blokstra (National Institute for Public Health and the Environment, Netherlands)*; Simona Bo (University of Turin, Italy)*; Bernhard O Boehm (Nanyang Technological University, Singapore)*; Carlos P Boissonnet (Centro de Educación Médica e Investigaciones Clínicas, Argentina)*; Pascal Bovet (University of Lausanne, Switzerland; Ministry of Health, Seychelles)*; Imperia Brajkovich (Universidad Central de Venezuela, Venezuela)*; Juergen Breckenkamp (Bielefeld University, Germany)*; Lizzy M Brewster (University of Amsterdam, Netherlands)*; Garry R Brian (The Fred Hollows Foundation New Zealand, New Zealand)*; Graziella Bruno (University of Turin, Italy)*; Anna Bugge (University of Southern Denmark, Denmark)*; Antonio Cabrera de León (Canarian Health Service, Spain)*; Gunay Can (Istanbul University, Turkey)*; Ana Paula C Cândido (Universidade Federal de Juiz de Fora, Brazil)*; Vincenzo Capuano (Reparto di Cardiologia ed UTIC di Mercato S., Italy)*; Maria J Carvalho (University of Porto, Portugal)*; Felipe F Casanueva (Santiago de Compostela University, Spain)*; Carmelo A Caserta (Associazione Calabrese di Epatologia, Italy)*; Katia Castetbon (French Institute for Health Surveillance, France)*; Snehalatha Chamukuttan (India Diabetes Research Foundation, India)*; Nishi Chaturvedi (University College London, UK)*; Chien-Jen Chen (Academia Sinica, Taiwan)*; Fangfang Chen (Capital Institute of Pediatrics, China)*; Shuohua Chen (Kailuan General Hospital, China)*; Ching-Yu Cheng (Duke-NUS Graduate Medical School, Singapore)*; Angela Chetrit (The Gertner Institute for Epidemiology and Health Policy Research, Israel)*; Shu-Ti Chiou (Ministry of Health and Welfare, Taiwan)*; Yumi Cho (Korea Centers for Disease Control and Prevention, South Korea)*; Jerzy Chudek (Medical University of Silesia, Poland)*; Renata Cifkova (Charles University in Prague, Czech Republic)*; Frank Claessens (Katholieke Universiteit Leuven, Belgium)*; Hans Concin (Agency for Preventive and Social Medicine, Austria)*; Cyrus Cooper (University of Southampton, UK)*; Rachel Cooper (University College London, UK)*; Simona Costanzo (IRCCS Istituto Neurologico Mediterraneo Neuromed, Italy)*; Dominique Cottel (Institut Pasteur de Lille, France)*; Chris Cowell (Westmead University of Sydney, Australia)*; Ana B Crujeiras (CIBERobn, Spain)*; Graziella D'Arrigo (National Research Council, Italy)*; Jean Dallongeville (Institut Pasteur de Lille, France)*; Rachel Dankner (The Gertner Institute for Epidemiology and Health Policy Research, Israel)*; Luc Dauchet (Lille University Hospital, France)*; Giovanni de Gaetano (IRCCS Istituto Neurologico Mediterraneo Neuromed, Italy)*; Stefaan De Henauw (Ghent University, Belgium)*; Mohan Deepa (Madras Diabetes Research Foundation, India)*; Abbas Dehghan (University Medical Center Rotterdam, Netherlands)*; Klodian Dhana (University Medical Center Rotterdam, Netherlands)*; Augusto F Di Castelnuovo (IRCCS Istituto Neurologico Mediterraneo Neuromed, Italy)*; Shirin Djalalinia (Tehran University of Medical Sciences, Iran)*; Kouamelan Doua (Ministère de la Santé et de la Lutte contre le Sida, Côte d'Ivoire)*; Wojciech Drygas (The Cardinal Wyszynski Institute of Cardiology, Poland)*; Yong Du (Robert Koch Institute, Germany)*; Eruke E Egbagbe (University of Benin College of Medical Sciences, Nigeria)*; Robert Eggertsen (University of Gothenburg, Sweden)*; Jalila El Ati (National Institute of Nutrition and Food Technology, Tunisia)*; Roberto Elosua (Institut Hospital del Mar d'Investigacions Mèdiques, Spain)*; Rajiv T Erasmus (University of Stellenbosch, South Africa)*; Cihangir Erem (Karadeniz Technical University, Turkey)*; Gul Ergor (Dokuz Eylul University, Turkey)*; Louise Eriksen (University of Southern Denmark, Denmark)*; Jorge Escobedo-de la Peña (Instituto Mexicano del Seguro Social, Mexico)*; Caroline H Fall (MRC Lifecourse Epidemiology Unit, UK)*; Farshad Farzadfar (Tehran University of Medical Sciences, Iran)*; Francisco J Felix-Redondo (Centro de Salud Villanueva Norte, Spain)*; Trevor S Ferguson (The University of the West Indies, Jamaica)*; Daniel Fernández-Bergés (Hospital Don Benito-Villanueva de la Serena, Spain)*; Marika Ferrari (National Research Institute on Food and Nutrition, Italy)*; Catterina Ferreccio (Pontificia Universidad Católica de Chile, Chile)*; Joseph D Finn (University of Manchester, UK)*; Bernhard Föger (Agency for Preventive and Social Medicine, Austria)*; Leng Huat Foo (Universiti Sains Malaysia, Malaysia)*; Heba M Fouad (World Health Organization Regional Office for the Eastern Mediterranean, Egypt)*; Damian K Francis (The University of the West Indies, Jamaica)*; Maria do Carmo Franco (Federal University of São Paulo, Brazil)*; Oscar H Franco (University Medical Center Rotterdam, Netherlands)*; Guillermo Frontera (Hospital Universitario Son Espases, Spain)*; Takuro Furusawa (Kyoto University, Japan)*; Zbigniew Gaciong (Medical University of Warsaw, Poland)*; Andrzej Galbarczyk (Jagiellonian University Medical College, Poland)*; Sarah P Garnett (University of Sydney, Australia)*; Jean-Michel Gaspoz (Geneva University Hospitals, Switzerland)*; Magda Gasull (CIBER en Epidemiología y Salud Pública, Spain)*; Louise Gates (Australian Bureau of Statistics, Australia)*; Johanna M Geleijnse (Wageningen University, Netherlands)*; Anoosheh Ghasemain (Tehran University of Medical Sciences, Iran)*; Simona Giampaoli (Istituto Superiore di Sanita, Italy)*; Francesco Gianfagna (University of Insubria, Italy)*; Jonathan Giovannelli (Lille University Hospital, France)*; Marcela Gonzalez Gross (Universidad Politécnica de Madrid, Spain)*; Juan P González Rivas (The Andes Clinic of Cardio-Metabolic Studies, Venezuela)*; Mariano Bonet Gorbea (National Institute of Hygiene, Epidemiology and Microbiology, Cuba)*; Frederic Gottrand (Université de Lille 2, France)*; Janet F Grant (The University of Adelaide, Australia)*; Tomasz Grodzicki (Jagiellonian University Medical College, Poland)*; Anders Grøntved (University of Southern Denmark, Denmark)*; Grabriella Gruden (University of Turin, Italy)*; Dongfeng Gu (National Center of Cardiovascular Diseases, China)*; Ong Peng Guan (Singapore Eye Research Institute, Singapore)*; Ramiro Guerrero (Universidad Icesi, Colombia)*; Idris Guessous (Geneva University Hospitals, Switzerland)*; Andre L Guimaraes (State University of Montes Claros, Brazil)*; Laura Gutierrez (Institute for Clinical Effectiveness and Health Policy, Argentina)*; Rebecca Hardy (University College London, UK)*; Rachakulla Hari Kumar (Indian Council of Medical Research, India)*; Jiang He (Tulane University, USA)*; Christin Heidemann (Robert Koch Institute, Germany)*; Ilpo Tapani Hihtaniemi (Imperial College London, UK)*; Sai Yin Ho (University of Hong Kong, China)*; Suzanne C Ho (The Chinese University of Hong Kong, China)*; Albert Hofman (University Medical Center Rotterdam, Netherlands)*; Andrea R V Russo Horimoto (Heart Institute, Brazil)*; Claudia M Hormiga (Fundación Oftalmológica de Santander, Colombia)*; Bernardo L Horta (Universidade Federal de Pelotas, Brazil)*; Leila Houti (University of Oran 1, Algeria)*; Abdullatif S Hussieni (Birzeit University, Palestine)*; Inge Huybrechts (International Agency for Research on Cancer, France)*; Nahla Hwalla (American University of Beirut, Lebanon)*; Licia Iacoviello (IRCCS Istituto Neurologico Mediterraneo Neuromed, Italy)*; Anna G Iannone (Reparto di Cardiologia ed UTIC di Mercato S., Italy)*; Mohsen M Ibrahim (Cairo University, Egypt)*; Nayu Ikeda (National Institute of Health and Nutrition, Japan)*; M Arfan Ikram (University Medical Center Rotterdam, Netherlands)*; Vilma E Irazola (Institute for Clinical Effectiveness and Health Policy, Argentina)*; Muhammad Islam (Aga Khan University, Pakistan)*; Masanori Iwasaki (Niigata University, Japan)*; Jeremy M Jacobs (Hadassah University Medical Center, Israel)*; Tazeen Jafar (Duke-NUS Graduate Medical School, Singapore)*; Grazyna Jasienska (Jagiellonian University Medical College, Poland)*; Chao Qiang Jiang (University of Hong Kong, China)*; Jost B Jonas (Ruprecht-Karls-University of Heidelberg, Germany)*; Pradeep Joshi (World Health Organization Country Office, India)*; Anthony Kafatos (University of Crete, Greece)*; Ofra Kalter-Leibovici (The Gertner Institute for Epidemiology and Health Policy Research, Israel)*; Amir Kasaeian (Tehran University of Medical Sciences, Iran)*; Joanne Katz (Johns Hopkins Bloomberg School of Public Health, USA)*; Prabhdeep Kaur (National Institute of Epidemiology, India)*; Maryam Kavousi (University Medical Center Rotterdam, Netherlands)*; Roya Kelishadi (Isfahan University of Medical Sciences, Iran)*; Andre P Kengne (South African Medical Research Council, South Africa)*; Mathilde Kersting (Research Institute of Child Nutrition, Germany)*; Yousef Saleh Khader (Jordan University of Science and Technology, Jordan)*; Young-Ho Khang (Seoul National University, South Korea)*; Stefan Kiechl (Medical University Innsbruck, Austria)*; Jeongseon Kim (National Cancer Center, South Korea)*; Yutaka Kiyohara (Kyushu University, Japan)*; Patrick Kolsteren (Institute of Tropical Medicine, Belgium)*; Paul Korrovits (Tartu University Clinics, Estonia)*; Seppo Koskinen (National Institute for Health and Welfare, Finland)*; Wolfgang Kratzer (University Hospital Ulm, Germany)*; Daan Kromhout (Wageningen University, Netherlands)*; Krzysztof Kula (Medical University of Łodz, Poland)*; Pawel Kurjata (The Cardinal Wyszynski Institute of Cardiology, Poland)*; Catherine Kyobutungi (African Population and Health Research Center, Kenya)*; Carl Lachat (Ghent University, Belgium)*; Youcef Laid (National Institute of Public Health, Algeria)*; Tai Hing Lam (University of Hong Kong, China)*; Orlando Landrove (Ministerio de Salud Pública, Cuba)*; Vera Lanska (Institute for Clinical and Experimental Medicine Prague, Czech Republic)*; Georg Lappas (Sahlgrenska Academy, Sweden)*; Avula Laxmaiah (Indian Council of Medical Research, India)*; Catherine Leclercq (Food and Agriculture Organization, Italy)*; Jeannette Lee (National University of Singapore, Singapore)*; Jeonghee Lee (National Cancer Center, South Korea)*; Terho Lehtimäki (Tampere University Hospital, Finland)*; Rampal Lekhraj (Universiti Putra Malaysia, Malaysia)*; Luz M León-Muñoz (Universidad Autónoma de Madrid, Spain)*; Yanping Li (Harvard T H Chan School of Public Health, USA)*; Wei-Yen Lim (National University of Singapore, Singapore)*; M Fernanda Lima-Costa (Oswaldo Cruz Foundation Rene Rachou Research Institute, Brazil)*; Hsien-Ho Lin (National Taiwan University, Taiwan)*; Xu Lin (University of Chinese Academy of Sciences, China)*; Lauren Lissner (University of Gothenburg, Sweden)*; Roberto Lorbeer (University Medicine Greifswald, Germany)*; José Eugenio Lozano (Consejería de Sanidad Junta de Castilla y León, Spain)*; Annamari Lundqvist (National Institute for Health and Welfare, Finland)*; Per Lytsy (University of Uppsala, Sweden)*; Guansheng Ma (Peking University, China)*; George L L Machado-Coelho (Universidade Federal de Ouro Preto, Brazil)*; Suka Machi (The Jikei University School of Medicine, Japan)*; Stefania Maggi (National Research Council, Italy)*; Dianna Magliano (Baker IDI Heart and Diabetes Institute, Australia)*; Marcia Makdisse (Hospital Israelita Albert Einstein, Brazil)*; Kodavanti Mallikharjuna Rao (Indian Council of Medical Research, India)*; Yannis Manios (Harokopio University of Athens, Greece)*; Enzo Manzato (University of Padova, Italy)*; Paula Margozzini (Pontificia Universidad Católica de Chile, Chile)*; Pedro Marques-Vidal (Lausanne University Hospital, Switzerland)*; Reynaldo Martorell (Emory University, USA)*; Shariq R Masoodi (Sher-i-Kashmir Institute of Medical Sciences, India)*; Tandi E Matsha (Cape Peninsula University of Technology, South Africa)*; Jean Claude N Mbanya (University of Yaoundé 1, Cameroon)*; Shelly R McFarlane (The University of the West Indies, Jamaica)*; Stephen T McGarvey (Brown University, USA)*; Stela McLachlan (University of Edinburgh, UK)*; Breige A McNulty (University College Dublin, Ireland)*; Sounnia Mediene-Benchekor (University of Oran 1, Algeria)*; Aline Meirhaeghe (INSERM, France)*; Ana Maria B Menezes (Universidade Federal de Pelotas, Brazil)*; Shahin Merat (Tehran University of Medical Sciences, Iran)*; Indrapal I Meshram (Indian Council of Medical Research, India)*; Jie Mi (Capital Institute of Pediatrics, China)*; Juan Francisco Miquel (Pontificia Universidad Católica de Chile, Chile)*; J Jaime Miranda (Universidad Peruana Cayetano Heredia, Peru)*; Mostafa K Mohamed (Ain Shams University, Egypt)*; Kazem Mohammad (Tehran University of Medical Sciences, Iran)*; Viswanathan Mohan (Madras Diabetes Research Foundation, India)*; Muhammad Fadhli Mohd Yusoff (Ministry of Health Malaysia, Malaysia)*; Niels C Møller (University of Southern Denmark, Denmark)*; Denes Molnar (University of Pécs, Hungary)*; Charles K Mondo (Mulago Hospital, Uganda)*; Luis A Moreno (Universidad de Zaragoza, Spain)*; Karen Morgan (PU-RCSI School of Medicine, Malaysia)*; George Moschonis (Harokopio University of Athens, Greece)*; Malgorzata Mossakowska (International Institute of Molecular and Cell Biology, Poland)*; Aya Mostafa (Ain Shams University, Egypt)*; Jorge Mota (University of Porto, Portugal)*; Maria L Muiesan (University of Brescia, Italy)*; Martina Müller-Nurasyid (Helmholtz Zentrum München, Germany)*; Jaakko Mursu (University of Eastern Finland, Finland)*; Gabriele Nagel (University of Ulm, Germany)*; Jana Námešná (Regional Authority of Public Health, Banska Bystrica, Slovakia)*; Ei Ei K Nang (National University of Singapore, Singapore)*; Vinay B Nangia (Suraj Eye Institute, India)*; Eva Maria Navarrete-Muñoz (CIBER de Epidemiología y Salud Pública, Spain)*; Ndeye Coumba Ndiaye (INSERM, France)*; Flavio Nervi (Pontificia Universidad Católica de Chile, Chile)*; Nguyen D Nguyen (University of Pharmacy and Medicine of Ho Chi Minh City, Vietnam)*; Ramfis E Nieto-Martínez (Universidad Centro-Occidental Lisandro Alvarado, Venezuela)*; Guang Ning (Shanghai Jiao-Tong University School of Medicine, China)*; Toshiharu Ninomiya (Kyushu University, Japan)*; Marianna Noale (National Research Council, Italy)*; Davide Noto (University of Palermo, Italy)*; Angélica M Ochoa-Avilés (Universidad de Cuenca, Ecuador)*; Kyungwon Oh (Korea Centers for Disease Control and Prevention, South Korea)*; Altan Onat (Istanbul University, Turkey)*; Clive Osmond (MRC Lifecourse Epidemiology Unit, UK)*; Johanna A Otero (Fundación Oftalmológica de Santander, Colombia)*; Luigi Palmieri (Istituto Superiore di Sanita, Italy)*; Songhomitra Panda-Jonas (Ruprecht-Karls-University of Heidelberg, Germany)*; Francesco Panza (Unversity of Bari, Italy)*; Mahboubeh Parsaeian (Tehran University of Medical Sciences, Iran)*; Sergio Viana Peixoto (Oswaldo Cruz Foundation Rene Rachou Research Institute, Brazil)*; Alexandre C Pereira (Heart Institute, Brazil)*; Annette Peters (Helmholtz Zentrum München, Germany)*; Niloofar Peykari (Tehran University of Medical Sciences, Iran)*; Aida Pilav (Federal Ministry of Health, Bosnia and Herzegovina)*; Freda Pitakaka (University of New South Wales, Australia)*; Aleksandra Piwonska (The Cardinal Wyszynski Institute of Cardiology, Poland)*; Jerzy Piwonski (The Cardinal Wyszynski Institute of Cardiology, Poland)*; Pedro Plans-Rubió (Public Health Agency of Catalonia, Spain)*; Miquel Porta (Hospital del Mar Medical Research Institute-IMIM, Spain)*; Marileen L P Portegies (University Medical Center Rotterdam, Netherlands)*; Hossein Poustchi (Tehran University of Medical Sciences, Iran)*; Rajendra Pradeepa (Madras Diabetes Research Foundation, India)*; Jacqueline F Price (University of Edinburgh, UK)*; Margus Punab (Tartu University Clinics, Estonia)*; Radwan F Qasrawi (Al-Quds University, Palestine)*; Mostafa Qorbani (Alborz University of Medical Sciences, Iran)*; Olli Raitakari (University of Turku, Finland)*; Sudha Ramachandra Rao (National Institute of Epidemiology, India)*; Ambady Ramachandran (India Diabetes Research Foundation, India)*; Rafel Ramos (Institut Universitari d'Investigació en Atenció Primària Jordi Gol, Spain)*; Sanjay Rampal (University of Malaya, Malaysia)*; Wolfgang Rathmann (German Diabetes Center, Germany)*; Josep Redon (University of Valencia, Spain)*; Paul Ferdinand M Reganit (University of the Philippines, Philippines)*; Fernando Rigo (Health Center San Agustin, Spain)*; Sian M Robinson (University of Southampton, UK)*; Cynthia Robitaille (Public Health Agency of Canada, Canada)*; Laura A Rodríguez (Universidad Industrial de Santander, Colombia)*; Fernando Rodríguez-Artalejo (Universidad Autónoma de Madrid, Spain)*; María del Cristo Rodriguez-Perez (Canarian Health Service, Spain)*; Rosalba Rojas-Martinez (Instituto Nacional de Salud Pública, Mexico)*; Dora Romaguera (Centre for Research in Environmental Epidemiology, Spain)*; Annika Rosengren (Sahlgrenska Academy, Sweden)*; Adolfo Rubinstein (Institute for Clinical Effectiveness and Health Policy, Argentina)*; Ornelas Rui (University of Madeira, Portugal)*; Blanca Sandra Ruiz-Betancourt (Instituto Mexicano del Seguro Social, Mexico)*; Marcin Rutkowski (Medical University of Gdansk, Poland)*; Charumathi Sabanayagam (Singapore Eye Research Institute, Singapore)*; Harshpal S Sachdev (Sitaram Bhartia Institute of Science and Research, India)*; Olfa Saidi (University Tunis El Manar, Tunisia)*; Sibel Sakarya (Marmara University, Turkey)*; Benoit Salanave (French Institute for Health Surveillance, France)*; Jukka T Salonen (University of Helsinki, Finland)*; Massimo Salvetti (University of Brescia, Italy)*; Jose Sánchez-Abanto (National Institute of Health, Peru)*; Renata Nunes dos Santos (University of São Paulo, Brazil)*; Rute Santos (University of Porto, Portugal)*; Luis B Sardinha (Universidade de Lisboa, Portugal)*; Marcia Scazufca (University of São Paulo, Brazil)*; Herman Schargrodsky (Hospital Italiano de Buenos Aires, Argentina)*; Christa Scheidt-Nave (Robert Koch Institute, Germany)*; Jonathan E Shaw (Baker IDI Heart and Diabetes Institute, Australia)*; Kenji Shibuya (The University of Tokyo, Japan)*; Youchan Shin (Singapore Eye Research Institute, Singapore)*; Rahman Shiri (Finnish Institute of Occupational Health, Finland)*; Rosalynn Siantar (Singapore Eye Research Institute, Singapore)*; Abla M Sibai (American University of Beirut, Lebanon)*; Mary Simon (India Diabetes Research Foundation, India)*; Judith Simons (St Vincent's Hospital, Australia)*; Leon A Simons (University of New South Wales, Australia)*; Michael Sjostrom (Karolinska Institute, Sweden)*; Jolanta Slowikowska-Hilczer (Medical University of Łodz, Poland)*; Przemyslaw Slusarczyk (International Institute of Molecular and Cell Biology, Poland)*; Liam Smeeth (London School of Hygiene & Tropical Medicine, UK)*; Marieke B Snijder (University of Amsterdam, Netherlands)*; Vincenzo Solfrizzi (University of Bari, Italy)*; Emily Sonestedt (Lund University, Sweden)*; Aicha Soumare (University of Bordeaux, France)*; Jan A Staessen (University of Leuven, Belgium)*; Jostein Steene-Johannessen (Norwegian School of Sport Sciences, Norway)*; Peter Stehle (Bonn University, Germany)*; Aryeh D Stein (Emory University, USA)*; Jochanan Stessman (Hadassah University Medical Center, Israel)*; Doris Stöckl (Helmholtz Zentrum München, Germany)*; Jakub Stokwiszewski (National Institute of Hygiene, Poland)*; Maria Wany Strufaldi (Federal University of São Paulo, Brazil)*; Chien-An Sun (Fu Jen Catholic University, Taiwan)*; Johan Sundström (Uppsala University, Sweden)*; Paibul Suriyawongpaisal (Mahidol University, Thailand)*; Rody G Sy (University of the Philippines, Philippines)*; E Shyong Tai (National University of Singapore, Singapore)*; Mohammed Tarawneh (Ministry of Health, Jordan)*; Carolina B Tarqui-Mamani (National Institute of Health, Peru)*; Lutgarde Thijs (University of Leuven, Belgium)*; Janne S Tolstrup (University of Southern Denmark, Denmark)*; Murat Topbas (Karadeniz Technical University, Turkey)*; Maties Torrent (Area de Salut de Menorca, Spain)*; Pierre Traissac (Institut de Recherche pour le Développement, France)*; Oanh T H Trinh (University of Pharmacy and Medicine of Ho Chi Minh City, Vietnam)*; Marshall K Tulloch-Reid (The University of the West Indies, Jamaica)*; Tomi-Pekka Tuomainen (University of Eastern Finland, Finland)*; Maria L Turley (Ministry of Health, New Zealand)*; Christophe Tzourio (University of Bordeaux, France)*; Peter Ueda (Harvard T H Chan School of Public Health, USA)*; Flora M Ukoli (Meharry Medical College, USA)*; Hanno Ulmer (Medical University of Innsbruck, Austria)*; Gonzalo Valdivia (Pontificia Universidad Católica de Chile, Chile)*; Irene G M van Valkengoed (Academic Medical Center of University of Amsterdam, Netherlands)*; Dirk Vanderschueren (Katholieke Universiteit Leuven, Belgium)*; Diego Vanuzzo (Centro di Prevenzione Cardiovascolare, Italy)*; Tomas Vega (Consejería de Sanidad, Junta de Castilla y León, Spain)*; Gustavo Velasquez-Melendez (Universidade Federal de Minas Gerais, Brazil)*; Giovanni Veronesi (University of Insubria, Italy)*; Monique Verschuren (National Institute for Public Health and the Environment, Netherlands)*; Jesus Vioque (Universidad Miguel Hernandez, Spain)*; Jyrki Virtanen (University of Eastern Finland, Finland)*; Sophie Visvikis-Siest (INSERM, France)*; Bharathi Viswanathan (Ministry of Health, Seychelles)*; Peter Vollenweider (Lausanne University Hospital, Switzerland)*; Sari Voutilainen (University of Eastern Finland, Finland)*; Alisha N Wade (University of the Witwatersrand, South Africa)*; Aline Wagner (University of Strasbourg, France)*; Janette Walton (University College Cork, Ireland)*; Wan Nazaimoon Wan Mohamud (Institute for Medical Research, Malaysia)*; Ming-Dong Wang (Public Health Agency of Canada, Canada)*; Ya Xing Wang (Beijing Tongren Hospital, China)*; S Goya Wannamethee (University College London, UK)*; Deepa Weerasekera (Ministry of Health, New Zealand)*; Peter H Whincup (St George's, University of London, UK)*; Kurt Widhalm (Medical University of Vienna, Austria)*; Andrzej Wiecek (Medical University of Silesia, Poland)*; Rainford J Wilks (The University of the West Indies, Jamaica)*; Johann Willeit (Medical University Innsbruck, Austria)*; Bogdan Wojtyniak (National Institute of Hygiene, Poland)*; Tien Yin Wong (Duke-NUS Graduate Medical School, Singapore)*; Jean Woo (The Chinese University of Hong Kong, China)*; Mark Woodward (University of Sydney, Australia; University of Oxford, UK)*; Aleksander Giwercman Wu (Lund University, Sweden)*; Frederick C Wu (University of Manchester, UK)*; Shou Ling Wu (Kailuan General Hospital, China)*; Haiquan Xu (Institute of Food and Nutrition Development of Ministry of Agriculture, China)*; Xiaoguang Yang (Chinese Center for Disease Control and Prevention, China)*; Xingwang Ye (University of Chinese Academy of Sciences, China)*; Akihiro Yoshihara (Niigata University, Japan)*; Novie O Younger-Coleman (The University of the West Indies, Jamaica)*; Sabina Zambon (University of Padova, Italy)*; Abdul Hamid Zargar (Center for Diabetes and Endocrine Care, India)*; Tomasz Zdrojewski (Medical University of Gdansk, Poland)*; Wenhua Zhao (Chinese Center for Disease Control and Prevention, China)*; Yingfeng Zheng (Singapore Eye Research Institute, Singapore)*.
Declaration of interests
JJM has received funding from Medtronics Foundation outside the submitted work. DM has received grants to her institution from Novartis Pharmaceutical, Novo Nordisk Pharmaceutical, Pharmacia and Upjohn, Pfizer, Sanofi Synthelabo, and Servier Laboratories. JES has received grants to his institution from Abbott, Alphapharm, AstraZeneca, Aventis Pharmaceutical, Eli Lilly, GlaxoSmithKline, Janssen-Cilag, Merck Lipha, Merck Sharp & Dohme, Novartis Pharmaceutical, Novo Nordisk Pharmaceutical, Pharmacia and Upjohn, Pfizer, Sanofi Synthelabo, and Servier Laboratories. All other Pooled Analysis and Writing Group members report no competing interests.
Supplementary Material
How do different methods for diagnosing diabetes affect estimates of prevalence and how is this likely to affect global estimates of this disease?
References
- 1.Danaei G, Finucane MM, Lu Y. National, regional, and global trends in fasting plasma glucose and diabetes prevalence since 1980: systematic analysis of health examination surveys and epidemiological studies with 370 country-years and 2·7 million participants. Lancet. 2011;378:31–40. doi: 10.1016/S0140-6736(11)60679-X. [DOI] [PubMed] [Google Scholar]
- 2.Finucane MM, Stevens GA, Cowan MJ. National, regional, and global trends in body-mass index since 1980: systematic analysis of health examination surveys and epidemiological studies with 960 country-years and 9·1 million participants. Lancet. 2011;377:557–567. doi: 10.1016/S0140-6736(10)62037-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Stevens GA, Singh GM, Lu Y. National, regional, and global trends in adult overweight and obesity prevalences. Popul Health Metr. 2012;10:22. doi: 10.1186/1478-7954-10-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kontis V, Mathers CD, Rehm J. Contribution of six risk factors to achieving the 25×25 non-communicable disease mortality reduction target: a modelling study. Lancet. 2014;384:427–437. doi: 10.1016/S0140-6736(14)60616-4. [DOI] [PubMed] [Google Scholar]
- 5.Global Burden of Metabolic Risk Factors for Chronic Diseases Collaboration Cardiovascular disease, chronic kidney disease, and diabetes mortality burden of cardiometabolic risk factors from 1980 to 2010: a comparative risk assessment. Lancet Diabetes Endocrinol. 2014;2:634–647. doi: 10.1016/S2213-8587(14)70102-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Zimmet PZ, Magliano DJ, Herman WH, Shaw JE. Diabetes: a 21st century challenge. Lancet Diabetes Endocrinol. 2014;2:56–64. doi: 10.1016/S2213-8587(13)70112-8. [DOI] [PubMed] [Google Scholar]
- 7.Yach D, Stuckler D, Brownell KD. Epidemiologic and economic consequences of the global epidemics of obesity and diabetes. Nat Med. 2006;12:62–66. doi: 10.1038/nm0106-62. [DOI] [PubMed] [Google Scholar]
- 8.International Diabetes Federation . IDF Diabetes Atlas. 6th edn. International Diabetes Federation; Brussels: 2013. [Google Scholar]
- 9.WHO . Definition, diagnosis and classification of diabetes mellitus and its complications: report of a WHO consultation. Part 1: Diagnosis and classification of diabetes mellitus. World Health Organization; Geneva: 1999. [Google Scholar]
- 10.WHO . Definition and diagnosis of diabetes mellitus and intermediate hyperglycemia: report of a WHO/IDF consultation. World Health Organization; Geneva: 2006. [Google Scholar]
- 11.WHO . Use of glycated haemoglobin (HbA1c) in the diagnosis of diabetes mellitus: abbreviated report of a WHO consultation. World Health Organization; Geneva: 2011. [PubMed] [Google Scholar]
- 12.Expert Committee on the Diagnosis and Classification of Diabetes Mellitus Report of the expert committee on the diagnosis and classification of diabetes mellitus. Diabetes Care. 2003;26 (suppl 1):S5–S20. doi: 10.2337/diacare.26.2007.s5. [DOI] [PubMed] [Google Scholar]
- 13.American Diabetes Association Standards of medical care in diabetes—2014. Diabetes Care. 2014;37 (suppl 1):S14–S80. doi: 10.2337/dc14-S014. [DOI] [PubMed] [Google Scholar]
- 14.American Diabetes Association Diagnosis and classification of diabetes mellitus. Diabetes Care. 2014;37 (suppl 1):S81–S90. doi: 10.2337/dc14-S081. [DOI] [PubMed] [Google Scholar]
- 15.Barrett-Connor E. The oral glucose tolerance test, revisited. Eur Heart J. 2002;23:1229–1231. doi: 10.1053/euhj.2002.3243. [DOI] [PubMed] [Google Scholar]
- 16.Tobias M. Global control of diabetes: information for action. Lancet. 2011;378:3–4. doi: 10.1016/S0140-6736(11)60604-1. [DOI] [PubMed] [Google Scholar]
- 17.Hare MJ, Shaw JE, Zimmet PZ. Current controversies in the use of haemoglobin A1c. J Intern Med. 2012;271:227–236. doi: 10.1111/j.1365-2796.2012.02513.x. [DOI] [PubMed] [Google Scholar]
- 18.WHO . Global action plan for the prevention and control of non-communicable diseases 2013–2020. World Health Organization; Geneva: 2013. [Google Scholar]
- 19.Shibata K, Suzuki S, Sato J. Diagnostic accuracy of glycohemoglobin A1c (HbA1c) for postprandial hyperglycemia was equivalent to that of fasting blood glucose. J Clin Epidemiol. 2005;58:1052–1057. doi: 10.1016/j.jclinepi.2005.01.019. [DOI] [PubMed] [Google Scholar]
- 20.Kim HK, Bae SJ, Choe J. Impact of HbA1c criterion on the detection of subjects with increased risk for diabetes among health check-up recipients in Korea. Diabetes Metab J. 2012;36:151–156. doi: 10.4093/dmj.2012.36.2.151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Mo M, Zhong W, Zhao G. Combining glycosylated hemoglobin A1c and fasting plasma glucose for diagnosis of type 2 diabetes in Chinese adults. BMC Endocr Disord. 2013;13:44. doi: 10.1186/1472-6823-13-44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wiener K, Roberts NB. The relative merits of haemoglobin A1c and fasting plasma glucose as first-line diagnostic tests for diabetes mellitus in non-pregnant subjects. Diabet Med. 1998;15:558–563. doi: 10.1002/(SICI)1096-9136(199807)15:7<558::AID-DIA669>3.0.CO;2-Q. [DOI] [PubMed] [Google Scholar]
- 23.Vlaar EM, Admiraal WM, Busschers WB. Screening South Asians for type 2 diabetes and prediabetes: (1) comparing oral glucose tolerance and haemoglobin A1c test results and (2) comparing the two sets of metabolic profiles of individuals diagnosed with these two tests. BMC Endocr Disord. 2013;13:8. doi: 10.1186/1472-6823-13-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Mannucci E, Ognibene A, Sposato I. Fasting plasma glucose and glycated haemoglobin in the screening of diabetes and impaired glucose tolerance. Acta Diabetol. 2003;40:181–186. doi: 10.1007/s00592-003-0109-8. [DOI] [PubMed] [Google Scholar]
- 25.Carson AP, Reynolds K, Fonseca VA, Muntner P. Comparison of A1C and fasting glucose criteria to diagnose diabetes among US adults. Diabetes Care. 2010;33:95–97. doi: 10.2337/dc09-1227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Adamu AN. Comparative performance of HbA1c 6.5% for FPG >/=7.0 vs 2hr PG>/=11.1 criteria for diagnosis of type 2 diabetes. Afr Health Sci. 2011;11:421–426. [PMC free article] [PubMed] [Google Scholar]
- 27.Cederberg H, Saukkonen T, Laakso M. Postchallenge glucose, A1C, and fasting glucose as predictors of type 2 diabetes and cardiovascular disease: a 10-year prospective cohort study. Diabetes Care. 2010;33:2077–2083. doi: 10.2337/dc10-0262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kramer CK, Araneta MR, Barrett-Connor E. A1C and diabetes diagnosis: The Rancho Bernardo Study. Diabetes Care. 2010;33:101–103. doi: 10.2337/dc09-1366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Li HY, Ma WY, Wei JN. Hemoglobin A1c for the diagnosis of diabetes: To replace or to guide oral glucose tolerance tests? J Diabetes Investig. 2012;3:259–265. doi: 10.1111/j.2040-1124.2011.00181.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Pajunen P, Peltonen M, Eriksson JG. HbA(1c) in diagnosing and predicting Type 2 diabetes in impaired glucose tolerance: the Finnish Diabetes Prevention Study. Diabet Med. 2011;28:36–42. doi: 10.1111/j.1464-5491.2010.03183.x. [DOI] [PubMed] [Google Scholar]
- 31.Piette JD, Milton EC, Aiello AE, Mendoza-Avelares MO, Herman WH. Comparison of three methods for diabetes screening in a rural clinic in Honduras. Rev Panam Salud Publica. 2010;28:49–57. doi: 10.1590/s1020-49892010000700008. [DOI] [PubMed] [Google Scholar]
- 32.Pinelli NR, Jantz AS, Martin ET, Jaber LA. Sensitivity and specificity of glycated hemoglobin as a diagnostic test for diabetes and prediabetes in Arabs. J Clin Endocrinol Metab. 2011;96:E1680–E1683. doi: 10.1210/jc.2011-1148. [DOI] [PubMed] [Google Scholar]
- 33.Selvin E, Steffes MW, Gregg E, Brancati FL, Coresh J. Performance of A1C for the classification and prediction of diabetes. Diabetes Care. 2011;34:84–89. doi: 10.2337/dc10-1235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Olson DE, Rhee MK, Herrick K, Ziemer DC, Twombly JG, Phillips LS. Screening for diabetes and pre-diabetes with proposed A1C-based diagnostic criteria. Diabetes Care. 2010;33:2184–2189. doi: 10.2337/dc10-0433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Almoosawi S, Cole D, Nicholson S. Biomarkers of diabetes risk in the National Diet and Nutrition Survey rolling programme (2008–2011) J Epidemiol Community Health. 2014;68:51–56. doi: 10.1136/jech-2013-202885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Droumaguet C, Balkau B, Simon D. Use of HbA1c in predicting progression to diabetes in French men and women: data from an Epidemiological Study on the Insulin Resistance Syndrome (DESIR) Diabetes Care. 2006;29:1619–1625. doi: 10.2337/dc05-2525. [DOI] [PubMed] [Google Scholar]
- 37.Engelgau MM, Thompson TJ, Herman WH. Comparison of fasting and 2-hour glucose and HbA1c levels for diagnosing diabetes. Diagnostic criteria and performance revisited. Diabetes Care. 1997;20:785–791. doi: 10.2337/diacare.20.5.785. [DOI] [PubMed] [Google Scholar]
- 38.Hjellestad ID, Astor MC, Nilsen RM, Softeland E, Jonung T. HbA(1)c versus oral glucose tolerance test as a method to diagnose diabetes mellitus in vascular surgery patients. Cardiovasc Diabetol. 2013;12:79. doi: 10.1186/1475-2840-12-79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Hu YM, Liu W, Chen YW. Combined use of fasting plasma glucose and glycated hemoglobin A1c in the screening of diabetes and impaired glucose tolerance. Acta Diabetol. 2010;47:231–236. doi: 10.1007/s00592-009-0143-2. [DOI] [PubMed] [Google Scholar]
- 40.Balkau B, Soulimane S, Lange C. Are the same clinical risk factors relevant for incident diabetes defined by treatment, fasting plasma glucose, and HbA1c? Diabetes Care. 2011;34:957–959. doi: 10.2337/dc10-1581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Soulimane S, Simon D, Shaw J. HbA1c, fasting plasma glucose and the prediction of diabetes: Inter99, AusDiab and D.E.S.I.R. Diabetes Res Clin Pract. 2012;96:392–399. doi: 10.1016/j.diabres.2011.06.003. [DOI] [PubMed] [Google Scholar]
- 42.Soulimane S, Simon D, Shaw JE. Comparing incident diabetes as defined by fasting plasma glucose or by HbA(1c). The AusDiab, Inter99 and DESIR studies. Diabet Med. 2011;28:1311–1318. doi: 10.1111/j.1464-5491.2011.03403.x. [DOI] [PubMed] [Google Scholar]
- 43.de Vegt F, Dekker JM, Stehouwer CD, Nijpels G, Bouter LM, Heine RJ. Similar 9-year mortality risks and reproducibility for the World Health Organization and American Diabetes Association glucose tolerance categories: the Hoorn Study. Diabetes Care. 2000;23:40–44. doi: 10.2337/diacare.23.1.40. [DOI] [PubMed] [Google Scholar]
- 44.Wild SH, Smith FB, Lee AJ, Fowkes FG. Criteria for previously undiagnosed diabetes and risk of mortality: 15-year follow-up of the Edinburgh Artery Study cohort. Diabet Med. 2005;22:490–496. doi: 10.1111/j.1464-5491.2004.01433.x. [DOI] [PubMed] [Google Scholar]
- 45.Zhang YH, Ma WJ, Thomas GN. Diabetes and pre-diabetes as determined by glycated haemoglobin A1c and glucose levels in a developing southern Chinese population. PLoS One. 2012;7:e37260. doi: 10.1371/journal.pone.0037260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Cowie CC, Rust KF, Byrd-Holt DD. Prevalence of diabetes and high risk for diabetes using A1C criteria in the U.S. population in 1988-2006. Diabetes Care. 2010;33:562–568. doi: 10.2337/dc09-1524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Ito C, Maeda R, Ishida S, Sasaki H, Harada H. Correlation among fasting plasma glucose, two-hour plasma glucose levels in OGTT and HbA1c. Diabetes Res Clin Pract. 2000;50:225–230. doi: 10.1016/s0168-8227(00)00187-x. [DOI] [PubMed] [Google Scholar]
- 48.Kharroubi AT, Darwish HM, Abu Al-Halaweh AI, Khammash UM. Evaluation of glycated hemoglobin (HbA1c) for diagnosing type 2 diabetes and prediabetes among Palestinian Arab population. PLoS One. 2014;9:e88123. doi: 10.1371/journal.pone.0088123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Bao Y, Ma X, Li H. Glycated haemoglobin A1c for diagnosing diabetes in Chinese population: cross sectional epidemiological survey. BMJ. 2010;340:c2249. doi: 10.1136/bmj.c2249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Davidson MB. Diagnosing diabetes with glucose criteria: worshiping a false God. Diabetes Care. 2011;34:524–526. doi: 10.2337/dc10-1689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Nazir A, Papita R, Anbalagan VP, Anjana RM, Deepa M, Mohan V. Prevalence of diabetes in Asian Indians based on glycated hemoglobin and fasting and 2-H post-load (75-g) plasma glucose (CURES-120) Diabetes Technol Ther. 2012;14:665–668. doi: 10.1089/dia.2012.0059. [DOI] [PubMed] [Google Scholar]
- 52.Rathmann W, Kowall B, Tamayo T. Hemoglobin A1c and glucose criteria identify different subjects as having type 2 diabetes in middle-aged and older populations: The KORA S4/F4 Study. Ann Med. 2012;44:170–177. doi: 10.3109/07853890.2010.531759. [DOI] [PubMed] [Google Scholar]
- 53.Blunt BA, Barrettconnor E, Wingard DL. Evaluation of fasting plasma-glucose as screening-test for Niddm in older adults—Rancho Bernardo Study. Diabetes Care. 1991;14:989–993. doi: 10.2337/diacare.14.11.989. [DOI] [PubMed] [Google Scholar]
- 54.Nakagami T, Tominaga M, Nishimura R. Is the measurement of glycated hemoglobin A1c alone an efficient screening test for undiagnosed diabetes? Japan National Diabetes Survey. Diabetes Res Clin Pract. 2007;76:251–256. doi: 10.1016/j.diabres.2006.09.015. [DOI] [PubMed] [Google Scholar]
- 55.Munera-Jaramillo MI, Restrepo-Lozada MA, Gomez-Bahamon LM, Mesa-Suarez Ddel R, Ramirez-Puerta BS. Glycosylated haemoglobin A1c compared to fasting plasma glucose in outpatients referred to a medical laboratory. Rev Salud Publica (Bogota) 2011;13:980–989. doi: 10.1590/s0124-00642011000600010. (in Spanish). [DOI] [PubMed] [Google Scholar]
- 56.Araneta MR, Grandinetti A, Chang HK. A1C and diabetes diagnosis among Filipino Americans, Japanese Americans, and Native Hawaiians. Diabetes Care. 2010;33:2626–2628. doi: 10.2337/dc10-0958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Homko CJ, Zamora LC, Kerper MM, Mozzoli M, Kresge K, Boden G. A single a1c >= 6.5% accurately identifies type 2 diabetes/impaired glucose tolerance in African Americans. J Prim Care Community Health. 2012;3:235–238. doi: 10.1177/2150131911435526. [DOI] [PubMed] [Google Scholar]
- 58.Khoo J, Tay TL, Foo JP. Sensitivity of A1C to diagnose diabetes is decreased in high-risk older Southeast Asians. J Diabetes Complications. 2012;26:99–101. doi: 10.1016/j.jdiacomp.2012.02.007. [DOI] [PubMed] [Google Scholar]
- 59.Peter A, Fritsche A, Stefan N, Heni M, Haring HU, Schleicher E. Diagnostic value of hemoglobin A1c for type 2 diabetes mellitus in a population at risk. Exp Clin Endocrinol Diabetes. 2011;119:234–237. doi: 10.1055/s-0030-1270440. [DOI] [PubMed] [Google Scholar]
- 60.van 't Riet E, Alssema M, Rijkelijkhuizen JM, Kostense PJ, Nijpels G, Dekker JM. Relationship between A1C and glucose levels in the general Dutch population: the new Hoorn study. Diabetes Care. 2010;33:61–66. doi: 10.2337/dc09-0677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Lipska KJ, De Rekeneire N, Van Ness PH. Identifying dysglycemic states in older adults: implications of the emerging use of hemoglobin A1c. J Clin Endocrinol Metab. 2010;95:5289–5295. doi: 10.1210/jc.2010-1171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.DECODE Study Group Will new diagnostic criteria for diabetes mellitus change phenotype of patients with diabetes? Reanalysis of European epidemiological data. BMJ. 1998;317:371–375. doi: 10.1136/bmj.317.7155.371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.DECODE Study Group Age- and sex-specific prevalences of diabetes and impaired glucose regulation in 13 European cohorts. Diabetes Care. 2003;26:61–69. doi: 10.2337/diacare.26.1.61. [DOI] [PubMed] [Google Scholar]
- 64.Qiao Q, Hu G, Tuomilehto J. Age- and sex-specific prevalence of diabetes and impaired glucose regulation in 11 Asian cohorts. Diabetes Care. 2003;26:1770–1780. doi: 10.2337/diacare.26.6.1770. [DOI] [PubMed] [Google Scholar]
- 65.Shaw JE, de Courten M, Boyko EJ, Zimmet PZ. Impact of new diagnostic criteria for diabetes on different populations. Diabetes Care. 1999;22:762–766. doi: 10.2337/diacare.22.5.762. [DOI] [PubMed] [Google Scholar]
- 66.DECODE Study Group Is fasting glucose sufficient to define diabetes? Epidemiological data from 20 European studies. Diabetologia. 1999;42:647–654. doi: 10.1007/s001250051211. [DOI] [PubMed] [Google Scholar]
- 67.Christensen DL, Witte DR, Kaduka L. Moving to an A1C-based diagnosis of diabetes has a different impact on prevalence in different ethnic groups. Diabetes Care. 2010;33:580–582. doi: 10.2337/dc09-1843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Ziemer DC, Kolm P, Weintraub WS. Glucose-independent, black-white differences in hemoglobin A1c levels: a cross-sectional analysis of 2 studies. Ann Intern Med. 2010;152:770–777. doi: 10.7326/0003-4819-152-12-201006150-00004. [DOI] [PubMed] [Google Scholar]
- 69.Kumar PR, Bhansali A, Ravikiran M. Utility of glycated hemoglobin in diagnosing type 2 diabetes mellitus: a community-based study. J Clin Endocrinol Metab. 2010;95:2832–2835. doi: 10.1210/jc.2009-2433. [DOI] [PubMed] [Google Scholar]
- 70.Selvin E, Steffes MW, Ballantyne CM, Hoogeveen RC, Coresh J, Brancati FL. Racial differences in glycemic markers: a cross-sectional analysis of community-based data. Ann Intern Med. 2011;154:303–309. doi: 10.1059/0003-4819-154-5-201103010-00004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.American Diabetes Association Implications of the Diabetes Control and Complications Trial. Diabetes Care. 2000;23 (suppl 1):S24–S26. [PubMed] [Google Scholar]
- 72.Cowie CC, Rust KF, Ford ES. Full accounting of diabetes and pre-diabetes in the U.S. population in 1988-1994 and 2005-2006. Diabetes Care. 2009;32:287–294. doi: 10.2337/dc08-1296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Schwarz G. Estimating dimension of a model. Ann Stat. 1978;6:461–464. [Google Scholar]
- 74.Di Cesare M, Khang YH, Asaria P. Inequalities in non-communicable diseases and effective responses. Lancet. 2013;381:585–597. doi: 10.1016/S0140-6736(12)61851-0. [DOI] [PubMed] [Google Scholar]
- 75.Cohen J, Cohen P, West SG, Aiken LS. Applied multiple regression/correlation analysis for the behavioral sciences. Routledge; 2013. [Google Scholar]
- 76.DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7:177–188. doi: 10.1016/0197-2456(86)90046-2. [DOI] [PubMed] [Google Scholar]
- 77.Sacks DB, Arnold M, Bakris GL. Guidelines and recommendations for laboratory analysis in the diagnosis and management of diabetes mellitus. Clin Chem. 2011;57:e1–e47. doi: 10.1373/clinchem.2010.161596. [DOI] [PubMed] [Google Scholar]
- 78.Snijders TAB, Bosker RJ. Multilevel Analysis: An Introduction to Basic and Advanced Multilevel Modeling. SAGE Publications; 2011. [Google Scholar]
- 79.Nakagawa S, Schielzeth H. A general and simple method for obtaining R2 from generalized linear mixed-effects models. Methods Ecol Evol. 2013;4:133–142. [Google Scholar]
- 80.Qiao Q, Tuomilehto J, Balkau B. Are insulin resistance, impaired fasting glucose and impaired glucose tolerance all equally strongly related to age? Diabet Med. 2005;22:1476–1481. doi: 10.1111/j.1464-5491.2005.01655.x. [DOI] [PubMed] [Google Scholar]
- 81.Zamboni M, Armellini F, Harris T. Effects of age on body fat distribution and cardiovascular risk factors in women. Am J Clin Nutr. 1997;66:111–115. doi: 10.1093/ajcn/66.1.111. [DOI] [PubMed] [Google Scholar]
- 82.Stevens GA, Finucane MM, De-Regil LM. Global, regional, and national trends in haemoglobin concentration and prevalence of total and severe anaemia in children and pregnant and non-pregnant women for 1995-2011: a systematic analysis of population-representative data. Lancet Glob Health. 2013;1:e16–e25. doi: 10.1016/S2214-109X(13)70001-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Colagiuri S, Lee CM, Wong TY. Glycemic thresholds for diabetes-specific retinopathy: implications for diagnostic criteria for diabetes. Diabetes Care. 2011;34:145–150. doi: 10.2337/dc10-1206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Emerging Risk Factors Collaboration Glycated hemoglobin measurement and prediction of cardiovascular disease. JAMA. 2014;311:1225–1233. doi: 10.1001/jama.2014.1873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Hare MJ, Magliano DJ, Zimmet PZ. Glucose-independent ethnic differences in HbA1c in people without known diabetes. Diabetes Care. 2013;36:1534–1540. doi: 10.2337/dc12-1210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Choi SH, Kim TH, Lim S, Park KS, Jang HC, Cho NH. Hemoglobin A1c as a diagnostic tool for diabetes screening and new-onset diabetes prediction: a 6-year community-based prospective study. Diabetes Care. 2011;34:944–949. doi: 10.2337/dc10-0644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Lee H, Oh JY, Sung YA. Optimal hemoglobin A1C cutoff value for diagnosing type 2 diabetes mellitus in Korean adults. Diabetes Res Clin Pract. 2013;99:231–236. doi: 10.1016/j.diabres.2012.09.030. [DOI] [PubMed] [Google Scholar]
- 88.Nomura K, Inoue K, Akimoto K. A two-step screening, measurement of HbA1c in association with FPG, may be useful in predicting diabetes. PLoS One. 2012;7:e36309. doi: 10.1371/journal.pone.0036309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Kim JM, Hong JW, Won JC. Glycated hemoglobin value for fasting plasma glucose of 126 mg/dL in Korean: The 2011 Korea National Health and Nutrition Examination Survey. Diabetes Metab J. 2014;38:480–483. doi: 10.4093/dmj.2014.38.6.480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Son JI, Rhee SY, Woo JT. Hemoglobin a1c may be an inadequate diagnostic tool for diabetes mellitus in anemic subjects. Diabetes Metab J. 2013;37:343–348. doi: 10.4093/dmj.2013.37.5.343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.The Diabetes Control and Complications Trial Research Group The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. The Diabetes Control and Complications Trial Research Group. N Engl J Med. 1993;329:977–986. doi: 10.1056/NEJM199309303291401. [DOI] [PubMed] [Google Scholar]
- 92.UK Prospective Diabetes Study (UKPDS) Group Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33) Lancet. 1998;352:837–853. [PubMed] [Google Scholar]
- 93.Little RR. Glycated hemoglobin standardization—National Glycohemoglobin Standardization Program (NGSP) perspective. Clin Chem Lab Med. 2003;41:1191–1198. doi: 10.1515/CCLM.2003.183. [DOI] [PubMed] [Google Scholar]
- 94.Hoelzel W, Weykamp C, Jeppsson JO. IFCC reference system for measurement of hemoglobin A1c in human blood and the national standardization schemes in the United States, Japan, and Sweden: a method-comparison study. Clin Chem. 2004;50:166–174. doi: 10.1373/clinchem.2003.024802. [DOI] [PubMed] [Google Scholar]
- 95.American Diabetes Association. European Association for the Study of Diabetes. International Federation of Clinical Chemistry and Laboratory Medicine. International Diabetes Federation Consensus statement on the worldwide standardisation of the HbA1c measurement. Diabetologia. 2007;50:2042–2043. doi: 10.1007/s00125-007-0789-7. [DOI] [PubMed] [Google Scholar]
- 96.Hanas R, John G. 2010 consensus statement on the worldwide standardization of the hemoglobin A1c measurement. Diabet Med. 2010;27:737–738. doi: 10.1111/j.1464-5491.2010.03033.x. [DOI] [PubMed] [Google Scholar]
- 97.Hanas R, John WG. 2013 update on the worldwide standardization of the HbA1c measurement. Diabet Med. 2013;30:885–886. doi: 10.1111/dme.12204. [DOI] [PubMed] [Google Scholar]
- 98.National Glycohemoglobin Standardization Program International Federation of Clinical Chemistry (IFCC) Standardization of HbA1c. http://www.ngsp.org/docs/IFCCstd.pdf (accessed Dec 29, 2014).
- 99.Simon D, Senan C, Balkau B, Saint-Paul M, Thibult N, Eschwege E. Reproducibility of HbA1c in a healthy adult population: the Telecom Study. Diabetes Care. 1999;22:1361–1363. doi: 10.2337/diacare.22.8.1361. [DOI] [PubMed] [Google Scholar]
- 100.Little RR, Rohlfing CL, Sacks DB. Status of hemoglobin A1c measurement and goals for improvement: from chaos to order for improving diabetes care. Clin Chem. 2011;57:205–214. doi: 10.1373/clinchem.2010.148841. [DOI] [PubMed] [Google Scholar]
- 101.Hardikar PS, Joshi SM, Bhat DS. Spuriously high prevalence of prediabetes diagnosed by HbA(1c) in young indians partly explained by hematological factors and iron deficiency anemia. Diabetes Care. 2012;35:797–802. doi: 10.2337/dc11-1321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.DECODE study group Glucose tolerance and mortality: comparison of WHO and American Diabetes Association diagnostic criteria. Lancet. 1999;354:617–621. [PubMed] [Google Scholar]
- 103.Nakagami T, DECODA Study Group Hyperglycaemia and mortality from all causes and from cardiovascular disease in five populations of Asian origin. Diabetologia. 2004;47:385–394. doi: 10.1007/s00125-004-1334-6. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
How do different methods for diagnosing diabetes affect estimates of prevalence and how is this likely to affect global estimates of this disease?