Abstract
Tanshinone IIA (Tan IIA) (C19H18O3) is one of the major active lipophilic components in a conventional Chinese medicine called danshen, and it has long been used in the People’s Republic of China and other neighboring countries to treat patients suffering from inflammatory bowel disease (IBD). Previous experiments by many teams determined which mechanism of Tan IIA is relevant to the treatment of IBD associated with inflammation and the pregnane X receptor (PXR). The current study demonstrated that Tan IIA is an efficacious PXR agonist and its ability to induce CYP3A4 mRNA and protein expression was mediated by the transactivation of PXR, a known target of abrogating inflammation in IBD. Clinical symptoms in mice and histological assessment data suggested that administration of Tan IIA in mice demonstrated significant protection and showed that in DSS-induced IBD it acts in a concentration-dependent manner. PXR-silenced mice treated with Tan IIA demonstrated low protection against DSS-induced mouse IBD and exacerbated the severity of IBD compared with wild-type mice; PXR-silenced mice demonstrated the necessity for PXR in Tan IIA-mediated upregulation of xenobiotic metabolism genes. The IBD treatment effects of Tan IIA are partially due to PXR-mediated upregulation of xenobiotic metabolism and downregulation of inflammatory mediators. The novel findings reported here may contribute to the effective utilization of Tan IIA and its derivatives as a PXR ligand in the treatment of human IBD. This suggests that Tan IIA may have considerable clinical utility.
Keywords: dashen, ulcerative colitis, Crohn’s disease, tanshinone IIA, pregnane X receptor, inflammatory bowel disease, dextran sodium sulfate
Introduction
Inflammatory bowel disease (IBD) is an idiopathic disease caused by a dysregulated immune response to host intestinal microflora. The two major types of IBD are ulcerative colitis (UC) and Crohn’s disease. IBD afflicts millions of individuals throughout the world and has shown a steep rise in incidence, becoming one of the five most prevalent gastrointestinal diseases in the US.1–3 Three characteristics define the etiology of IBD: 1) genetic predisposition; 2) an altered, dysregulated immune response; and 3) an altered response to gut microorganisms.4–6 However, the etiology of IBD is still not completely understood.
Evidence from several studies has suggested that the detoxification properties of pregnane X receptor (PXR) and its target genes are necessary to maintaining the integrity of the intestinal epithelial barrier.4,7 PXR, confirmed as a member of the nuclear receptor superfamily in 1998, is involved in the absorption, distribution, metabolism, and elimination of xenobiotics. It has also been implicated in the pathogenesis of IBD. In as early as 2004, Langmann et al, in addition to other authors, found low levels of PXR expression in the intestines of UC patients.4,7–10 In 2006, the hypothesis that PXR is a key regulator of intestinal integrity in IBD was strengthened by a study by Dring et al.11,12 One study has demonstrated that PXR activation ameliorates dextran sodium sulfate (DSS)-induced colitis in mice via nuclear factor-κB (NF-κB) target gene expression, suggesting that PXR may modulate intestinal inflammation.8 PXR is abundantly expressed in the mammalian liver and small intestine and seems to be critical to intestinal integrity. It acts by regulating the xenobiotic metabolizing enzyme CYP3A4 and the P-glycoprotein encoded by the multidrug resistance 1 gene (MDR1), which is mainly involved in cellular transport.8,9,13–15
Tanshinone IIA (Tan IIA), one of the main natural active ingredients purified from Salvia miltiorrhiza radix, also known as danshen in Chinese,16 is widely used for treating cerebral and cardiovascular diseases such as stroke, coronary heart disease, and hyperlipidemia.17–19 In an in vitro study, Tan IIA has been shown to suppress production of nitric oxide and expression of inducible nitric oxide synthase (iNOS) and interleukin (IL)-1β in activated RAW 264.7 cells.20 An in vivo study showed that Tan IIA induces activation of the cytochrome P450 mixed-function oxidase system in C57B/6J mice.21,22 Tan IIA has also been shown to be beneficial in specific cases of IBD when used alongside traditional Chinese medicine treatments.23 This study also reported that Tan IIA can ameliorate trinitrobenzene sulfonic acid-induced colitis in a dose-dependent manner. Experiments identified a mechanism by which Tan IIA might ameliorate IBD associated with inflammation. However, the role of Tan IIA in irritable bowel syndrome and IBD therapy and its mechanisms of action are not well understood.
Previous studies demonstrated that Tan IIA was an efficacious PXR agonist capable of activating human PXR to induce the expression of CYP3A4 in human hepatoma cells.24–26 PXR also plays a key role in the treatment of IBD. It was therefore hypothesized that PXR might play a crucial role in Tan IIA-mediated protection of DSS-induced IBD. Small interfering RNA (siRNA) is a powerful tool for specific gene silencing during gene therapy.27–29 siRNAs inhibit specific gene expression by inducing the sequence-specific degradation of homologous mRNA, which knocks down the gene product at the posttranscriptional level.30–32 In the current study, wild-type (WT) and PXR-knockdown mice were used to assess the effects of PXR on the Tan IIA treatment of DSS-induced IBD.14 Results indicated that Tan IIA could ameliorate DSS-induced IBD in a dose-dependent manner, and the effects were exerted by the activation of PXR and inhibition of the expression of inflammatory mediator genes in vivo.6,8
Materials and methods
Drugs and reagents
Tan IIA (>98% purity) was obtained from the National Institute for the Control of Pharmaceutical and Biological Products (Beijing, People’s Republic of China). Rifampicin (RIF) and pregnenolone 16a-carbonitrile (PCN) were purchased from Sigma-Aldrich (St Louis, MO, USA). DSS (molecular weight 36–50 kDa) was purchased from MP Biomedical LLC (Solon, OH, USA). Gibco® G418 and Dulbecco’s Modified Eagle’s Medium (DMEM), minimum essential medium (MEM), and fetal bovine serum (FBS) are Hyclone products obtained from Thermo Fisher Scientific (Waltham, MA, USA). TransScript™ First-Step quantitative reverse transcriptase polymerase chain reaction (qRT-PCR) SuperMix and Dual-Luciferase reporter assay systems were purchased from Promega Corporation (Fitchburg, WI, USA). An electrophoretic mobility shift assay (EMSA) kit with LightShift® chemiluminescent was purchased from Pierce (Rockford, IL, USA). Applied Biosystems Fast SYBR@ Green Master Mix was purchased from Thermo Fisher Scientific. Polyclonal anti-human anti-PXR (H-11: sc-48340), anti-human CYP3A4 (HL3: sc53850), polyclonal anti-mouse PXR (R-14: sc-7739), mouse monoclonal immunoglobulin (Ig)G, goat polyclonal anti-actin antibodies, and the PXR siRNA (mouse; sc-44058) were purchased from Santa Cruz Biotechnology, Inc. (Dallas, TX, USA). The tissue protein extraction kit was obtained from Cowin Biotech Company Ltd. (Beijing, People’s Republic of China). BCA was purchased from Applygen Technologies Inc. (Beijing, People’s Republic of China). All other chemicals were of the highest quality commercially available. Three individual siRNA duplexes (sc-44058A, sc-44058B, and sc-44058C are listed in Table 1) were used to independently verify the PXR mouse gene silencing results and were synthesized and modified with 2′-O-Methyl oligo at Shanghai GenePharma Company Ltd. (Shanghai, People’s Republic of China).
Table 1.
siRNA synthesized at Shanghai GenePharma Company Ltd
| siRNA | Primer sequence |
|---|---|
| sc-44058A | 5′-CCAAUGGCUACCACUUCAAtt-3′ |
| 5′-UUGAAGUGGUAGCCAUUGGtt-3′ | |
| sc-44058B | 5′-CUAGACAGAUGGACAUACUtt-3′ |
| 5′-AGUAUGUCCAUCUGUCUAGtt-3′ | |
| sc-44058C | 5′-GAACCCUGUAACAGUUGUAtt-3′ |
| 5′-UACAACUGUUACAGGGUUCtt-3′ |
Note: Shanghai GenePharma Company Ltd., Shanghai, People’s Republic of China.
Abbreviation: siRNA, small interfering RNA.
Cell cultures and treatment
A secreted luciferase reporter gene, with two-distal enhancer and proximal promoter ligated in tandem was constructed in-house. Plasmids containing the proximal and distal CYP3A4 promoter regions and a human PXR expression vector were stably transfected into HepG2 cells. Stably transfected HepG2 cells were accurate and reliable, and they predicted the ability of CYP3A4 induction or inhibition by direct supernatant detection. The stably transfected HepG2 cell line was cultured in MEM supplemented with 10% FBS, 1% nonessential amino acids, and 500 mg/mL G418.
LS174T cells (American Type Culture Collection, Manassas, VA, USA) were cultured in DMEM supplemented with 10% FBS. Cells were maintained at 37°C in an incubator with a saturated humid atmosphere containing 95% air and 5% CO2. No ethics statement was required from the institutional review board for the use of these cell lines.
RNA isolation and qRT-PCR analysis
LS174T cells were maintained in 6-well plates until 80% confluence and then treated with various concentrations of Tan IIA (2.5, 5, 10, and 20 μM) in serum-free medium for various periods of time to assess the specific induction of CYP3A4 and PXR expression. Cells were also treated with 20 μM Tan IIA for 0, 3, 6, 12, 24, 48, 60, and 72 hours, and total cellular RNA was isolated using Invitrogen TRIzol reagent (Thermo Fisher Scientific) according to the manufacturer’s instructions. RIF (10 μM) served as a positive control, and dimethyl sulfoxide (DMSO) was used as the negative control.
The quality of the RNA was confirmed by an A260/A280 ratio of >1.8 and an RNA integrity number ≥6.5. Complementary DNA (cDNA) was synthesized using a Transcriptor™ first-strand cDNA synthesis kit. qRT-PCR reactions were performed on an ABI StepOne Plus™ Real Time PCR instrument using SYBR Green PCR Master Mix. The amplification reactions were performed as follows: 20 seconds at 95°C, and 40 cycles at 95°C for 3 seconds, and 60°C for 30 seconds. The primers used in the current study are listed in Table 2. The quantity of each transcript was calculated as described in the instrument manual and was normalized to the amount of the housekeeping gene β-actin.
Table 2.
Primer used in real time PCR analysis
| Gene | Primer sequence |
|---|---|
| CYP3A4 | 5′-CACAGATCCCCCTGAAATTAAGCTTA-3′ |
| 5′-AAAATTCAGGCTCCACTTACGGTG-3′ | |
| PXR | 5′-TGGGTGACACCTCCGAGA-3′ |
| 5′-TAGGGAGACAGGCCAGCA-3′ | |
| β-actin | 5′-CTACAATGAGCTGCGTGTGG-3′ |
| 5′-TAGCTCTTCTCCAGGGAGGA-3′ |
Abbreviations: CYP, cytochrome P450; PXR, pregnane X receptor.
Preparation of total protein and Western blotting
The expression of CYP3A4 and PXR was determined by Western blotting. LS174T cells were treated with Tan IIA (2.5, 5, 10, or 20 μM) or RIF (10 μM) for 48 hours. The cells were then harvested in RIPA lysis buffer (100 μL per million cells). Protein concentrations were determined using a BCA protein quantification kit with bovine serum albumin as the standard. A total of 25–50 mg of whole cell lysate was separated by 12% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and was subsequently transferred electrophoretically to a polyvinylidene fluoride membrane. After transfer, the membrane was incubated at 4°C for 1 hour in Tween Tris-buffered saline containing 5% skimmed milk. The membranes were then incubated with human anti-human PXR (H-11: sc-48340; dilution 1:500) or anti-human CYP3A4 (HL3: sc53850; 1:800) antibodies overnight at 4°C. The samples were subsequently incubated with monoclonal IgG (1:2,000) secondary antibody for 2 hours, and visualized on film using a Santa Cruz ECL detection system. β-actin served as a loading control, and RIF and DMSO served as the positive and negative controls, respectively. Densitometric analyses were performed to semiquantify protein expression.
Reporter gene assays
Approximately 200,000 stably transfected HepG2 cells were seeded into each well of 96-well plates for 24 hours. RIF (10 μM) or Tan IIA (2.5, 5, 10, 20, or 40 μM) were added with fresh medium, and cells were incubated for an additional 24 or 48 hours. The cells were washed once with phosphate-buffered saline (PBS) and harvested at the indicated times. The signal from Tan IIA- and RIF-treated cells was normalized to that of vehicle-treated cells to determine the degree of activation.
Preparation of nuclear extract and electrophoretic mobility shift assays
EMSAs were performed as reported previously. Briefly, LS174T cells were treated for 24 hours with DMSO, Tan IIA, and RIF in growth media. Nuclear proteins were isolated using NE-PER® nuclear and cytoplasmic extraction reagent kits (Pierce). EMSAs were conducted with a LightShift® chemiluminescent EMSA kit. Commercially synthesized oligonucleotides were used for EMSA (Invitrogen; Thermo Fisher Scientific). The following double-stranded oligonucleotides of specific response elements of CYP3A4 and proximal and distal promoter oligonucleotides were labeled with 5′-biotin (5′-GAATGAACTTGCTGACCCTCTATATGAA CTCAAAGGAGGTCAGTG-3′ and its complement). The binding reactions were carried out for 30 minutes and reaction mixtures contained 5 μg nuclear protein. To determine the specificity of oligonucleotide binding, a 200-fold excess of unlabeled pregnane X response element (PXRE) was added to extracts of Tan IIA- and RIF-treated cells for competition analysis. DNA-protein complexes were separated under nondenaturing conditions on a 6% polyacrylamide gel using 0.5× TBE (45 mM Tris, pH 7.5, 45 mM boric acid, and 2 mM ethylenediaminetetraacetic acid) as a running buffer, and the results were recorded using a chemiluminescent nucleic acid detection module kit (Pierce).
Animals and treatments
Seven-week-old male BALB/c mice weighing 20–25 g were obtained from the animal center of the Academy of Military Medical Sciences and fed at the specific pathogen-free (SPF) level animal center of the Academy of Military Medical Sciences (Beijing, People’s Republic of China). The mice were kept in an SPF animal facility at a constant temperature of 23°C and relative humidity of 40%–60%. The protocol was approved by the Committee on the Ethics of Animal Experiments of the animal center of the Academy of Military Medical Sciences (IACUC of AMMS 2013013). The animal experiments were conducted in accordance with the Guiding Principles in the Use of Animals in Toxicology. All surgeries were performed under sodium pentobarbital anesthesia, and all reasonable effort was made to minimize suffering. The mice were acclimatized to laboratory conditions for 3 days before the study. DSS was administered to induce colitis in the mice, and PCN served as the positive control. PXR siRNA mouse was hypothesized to inhibit PXR expression in mice.
Mice were randomly divided into 6 groups (n=8 per group): vehicle, DSS, and Tan IIA or PCN and DSS cotreatment groups. Animals were treated with Tan IIA (5, 10, or 20 mg/kg/day) or PCN (45 mg/kg/day) for 10 days by intraperitoneal injection, and the agents were dissolved in camellia oil. From the 4th day, mice were fed 4% (wt/vol) DSS dissolved in sterile, distilled water (vehicle control) ad libitum for 7 days. During the same period, the Tan IIA and PCN administration continued until the end of the DSS treatment.
For siRNA experiments, mice were randomly divided into six groups (n=4 per group): vehicle, lithocholic acid, negative control siRNA (siRNA-NC), PXR siRNA, PXR siRNA with Tan IIA, and PXR siRNA with PCN. The PXR siRNA was a pool of three target-specific 20–25 nucleotide siRNAs designed to knock down the PXR gene expression. The three synthetic PXR siRNAs with 2′-Methyl oligo modification were mixed and delivered in vivo using a modified hydrodynamic transfection method, in which 100 μg siRNA dissolved in 0.9 mL PBS was rapidly injected into the tail vein.33 Mice in the vehicle, DSS, and PXR siRNA-NC groups were injected with equal volumes of PBS or siRNA-NC. The animals were then treated with control or chemical inducers (20 mg/mg Tan IIA or 45 mg/kg PCN) for 10 days via intraperitoneal injection. Mice were fed 4% (wt/vol) DSS dissolved in sterile, distilled water (vehicle control) ad libitum from the 4th day onward. The siRNA injection was repeated once every 3 days (on days 1, 4, and 7).
Food was removed from all animals 4 hours before they were killed. The colons were quickly excised, cleaned thoroughly with ice-cold PBS, and preserved in liquid nitrogen until use. Pieces of colons were fixed in 10% formalin for histological analysis.
Histological assessment of colitis
Daily changes in body weight and clinical signs of colitis, such as rectal bleeding, diarrhea, and bloody stool, were assessed and scored from 0 to 4. An occult blood kit was used for the examination of rectal bleeding and an injury severity score was presented on an ordinal scale of 1–4. For macroscopic colonic damage, colons were opened longitudinally, flushed with PBS, and fixed in 10% buffered formalin. Colons were Swiss rolled for examination of the entire length of the colon and processed in paraffin. A single pathologist, who was blinded to the treatment, duration, and genotype of the sample, examined and scored representative colons sections from three mice per group. Hematoxylin and eosin (H&E)-stained 5 μm sections were assessed according to a standard staining protocol. Histology scores were assessed according to morphological criteria and the ratio of the proportion of the colons affected by individual changes to the total area of the colons sectioned. Histological scores were presented on an ordinal scale of 1–4.
RNA and total protein preparation and analysis
RNA was isolated from mouse colon tissues using a tissue protein extraction kit according to the manufacturer’s instructions. A qRT-PCR assay was performed as described above. The primer sequences are provided in Table 3. Colon proteins (40–100 μg) were separated by 10%–15% SDS-PAGE and analyzed as described above using PXR (R-14: sc-7739, dilution 1:500) primary antibody.
Table 3.
Primer used in real time PCR analysis
| Gene | Primer sequence |
|---|---|
| GAPDH | 5′-AACGACCCCTTCATTGAC-3′ |
| 5′-TCCACGACATACTCAGCAC-3′ | |
| PXR | 5′-GTTCAAGGGCGTCATCAACT-3′ |
| 5′-TTCTGGAAGCCACCATTAGG-3′ | |
| CYP3A11 | 5′-CTCAATGGTGTGTATATCCCC-3′ |
| 5′-CCGATGTTCTTAGACACTGCC-3′ | |
| CYP3A13 | 5′-AGCAGTGCTCTTTCCTTTGC-3′ |
| 5′-CCCTTTGGGAATGAATAGCC-3′ | |
| MDR1α | 5′-GTCCCAACTGGGATATTGTACA-3′ |
| 5′-AGGTGCCCATGTCTGAGTAA-3′ | |
| GSTα1 | 5′-CCCCTTTCCCTCTGCTGAAG-3′ |
| 5′-TGCAGCTTCACTGAATCTTGAAAG-3′ | |
| TNF-α | 5′-GGCAGGTCTACTTTGGAGTCATTGC-3′ |
| 5′-ACATTCGAGGCTCCAGTGAATTCGG-3′ | |
| IL-6 | 5′-CTGGTGACAACCACGGCCTTCCCTA-3′ |
| 5′-ATGCTTAGGCATAACGCACTAGGTT-3′ | |
| iNOS | 5′-CCTTGTTCAGCTACGCCTTC-3′ |
| 5′-AAGGCCAAACACAGCATACC-3′ | |
| MCP | 5′-CCCCAGTCACCTGCTGTTAT-3′ |
| 5′-TCCTGAACCCACTTCTGCTT-3′ |
Abbreviations: CYP, cytochrome P450; GSTα1, glutathione S-transferase alpha 1; IL-6, interleukin 6; iNOS, inducible nitrogen oxide synthase; MDR1α, multidrug resistance 1 alpha; PXR, pregnane X receptor; TNF-α, tumor necrosis factor-alpha; MCP, monocyte chemoattractant protein.
Statistical analysis
Experimental values are expressed as mean ± standard deviation. Statistical analyses were performed using one way analysis of variance (ANOVA). Daily changes in body weight and clinical signs of colitis, such as rectal bleeding and diarrhea, were performed using repeated measures ANOVA. A value of P<0.01 was considered to be statistically significant.
Results
Concentration and time-dependent induction of PXR and xenobiotic detoxification genes expression by Tan IIA
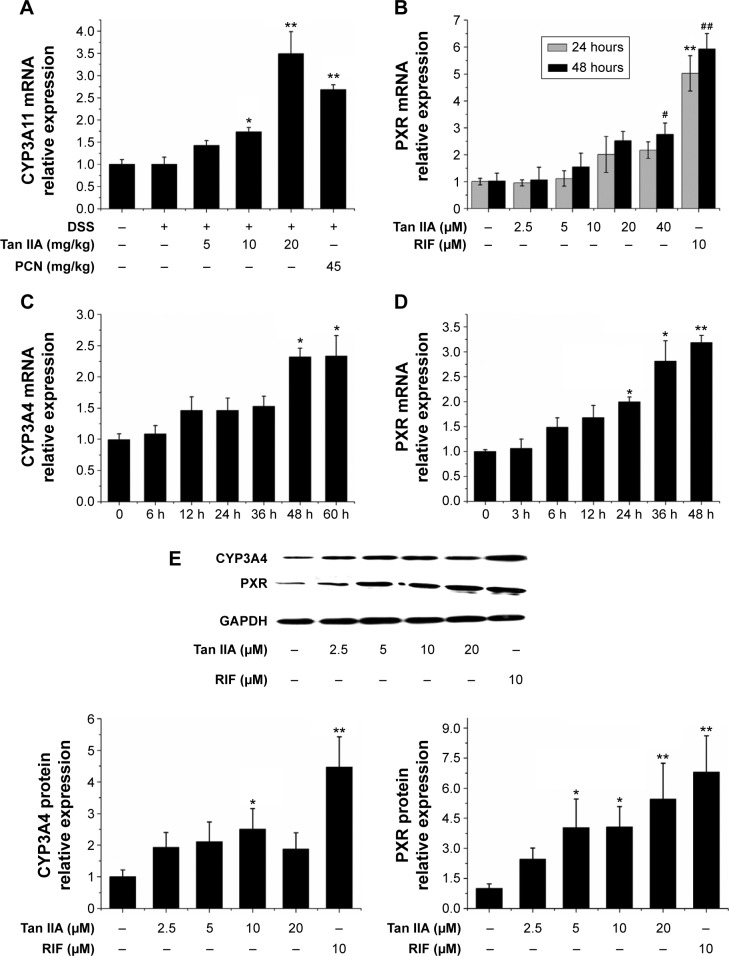
To investigate the effect of Tan IIA treatment on CYP3A4 and PXR transcription, qRT-PCR was used to quantify CYP3A4 and PXR mRNA levels in LS174T cells treated with 2.5, 5, 10, or 20 μM Tan IIA or 10 μM RIF for 24 and 48 hours. As shown in Figure 1A and B, CYP3A4 and PXR levels were significantly higher in RIF-treated HepG2 cells than in vehicle controls. This indicated that LS174T cells were found to be a sensitive and selective model for assessing the regulation of CYP3A4 and PXR transcription. CYP3A4 and PXR levels increased in a concentration-dependent manner with doses of 2.5–20 μM in Tan IIA-treated cells. Expression peaked at 20 μM. Twenty micromoles of Tan IIA was used to assess Tan IIA-induced CYP3A4 and PXR mRNA expression over time (Figure 1C and D). Tan IIA-induced CYP3A4 expression in a time-dependent manner from 0 to 60 hours. These results confirmed that Tan IIA could induce CYP3A4 and PXR mRNA expression in LS174T cells.
Figure 1.
Effects of Tan IIA on mRNA and protein expression in LS174T cells.
Notes: (A) Concentration-dependent effects of Tan IIA on CYP3A4 mRNA levels in LS174T cells treated for 24 and 48 hours; (B) Concentration-dependent effects of Tan IIA on PXR mRNA levels in LS174T cells treated for 24 and 48 hours; (C) Time-dependent effects of Tan IIA on CYP3A4 mRNA levels in LS174T cells; (D) Time-dependent effects of Tan IIA on PXR mRNA levels in LS174T cells; (E) Concentration-dependent effects of Tan IIA on CYP3A4 and PXR protein levels in LS174T cells; RIF served as the positive control, and the housekeeping gene β-actin served as the loading control; Data are presented as the means ± SD of three independent experiments, and the bands are from a representative experiment; Asterisks indicate a statistically significant difference compared with untreated cells; *P<0.05 and **P<0.01 versus control group (24 hours); #P<0.05 and ##P<0.01 versus control group (48 hours).
Abbreviations: CYP, cytochrome P450; mRNA, messenger RNA; DSS, dextran sodium sulfate; PCN, pregnenolone 16a-carbonitrile; PXR, pregnane X receptor; RIF, rifampicin; SD, standard deviation; Tan IIA, tanshinone IIA; GAPDH, glyceraldehyde phosphate dehydrogenase.
Western blotting was used to determine whether the sustained induction of CYP3A4 and PXR mRNA expression by Tan IIA was accompanied by corresponding changes in protein expression. As shown in Figure 1E, the expression of CYP3A4 and PXR was much higher in RIF-treated cells than in controls. A concentration-dependent elevation was also observed in cells treated with 2.5–20 μM Tan IIA. The extent of the increased CYP3A4 and PXR protein levels with RIF and Tan IIA was consistent with the altered mRNA levels. Taken together, these results suggest that Tan IIA-induced CYP3A4 expression in HepG2 cells, possibly by increasing the expression of PXR.
Tan IIA and stably transfected HepG2 cells
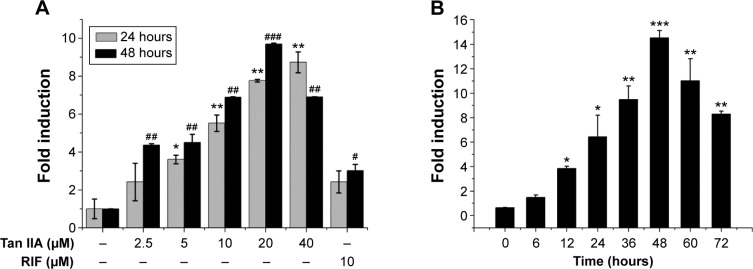
PXR is an important transcription factor. It regulates CYP3A4 gene expression by binding to its promoter. A stably transfected HepG2 cell line in vitro screening system based on the PXR-CYP3A4 pathway was used to investigate other possible mechanisms underlying the induction of CYP3A4 by Tan IIA and to assess the ability of Tan IIA to transactivate PXR. The potential capacity of Tan IIA to induce CYP3A4 was also screened using this method. The cells were then treated with 10 μM RIF or 2.5, 5, 10, 20, or 40 μM Tan IIA for 24 and 48 hours. As shown in Figure 2A, RIF- and Tan IIA-treated cells exhibited increased PXR transactivation. PXR transactivation in cells exposed to 10 μM RIF, a known PXR activator and CYP3A4 inducer, was greater (>2-fold) than vehicle controls. In addition, cells exposed to Tan IIA had significantly increased induction (2.4–9.7-fold) than vehicle controls. These effects were concentration-dependent within a range of 2.5–20 μM. The time-dependence of the PXR activation was investigated using 20 μM Tan IIA. A 1.4–14.5-fold increase in luciferase activity was observed from 0 to 48 hours; activity peaked at 48 hours, then decreased thereafter (Figure 2B).
Figure 2.
Effects of Tan IIA on PXR activation in transfected 9+ PXR cells.
Notes: Stably transfected HepG2 cells were transfected with expression vectors encoding human PXR (pCMX-hPXR) and a reporter plasmid (pGL3-P3A4); (A) Cells treated with vehicle (0.1% DMSO), RIF (10 μM) or increasing concentrations of Tan IIA (2.5–40 μM) for 24 and 48 hours prior to harvest; (B) Cell lysates were harvested 0 to 72 hours after treatment with 20 μM Tan IIA; Firefly luciferase activity was determined and normalized against Renilla luciferase activity; The values are expressed as the mean of the fold increase in activity compared with DMSO-treated cells from three independent experiments; Asterisks indicate statistically significant differences compared with untreated cells; *P<0.05, **P<0.01, ***P<0.001 versus control group (24 hours); #P<0.05, ##P<0.01, ###P<0.001 versus control group (48 hours).
Abbreviations: DMSO, dimethyl sulfoxide; PXR, pregnane X receptor; RIF, rifampicin; Tan IIA, tanshinone IIA.
Tan IIA and PXR activation as assessed using ligand-dependent EMSA
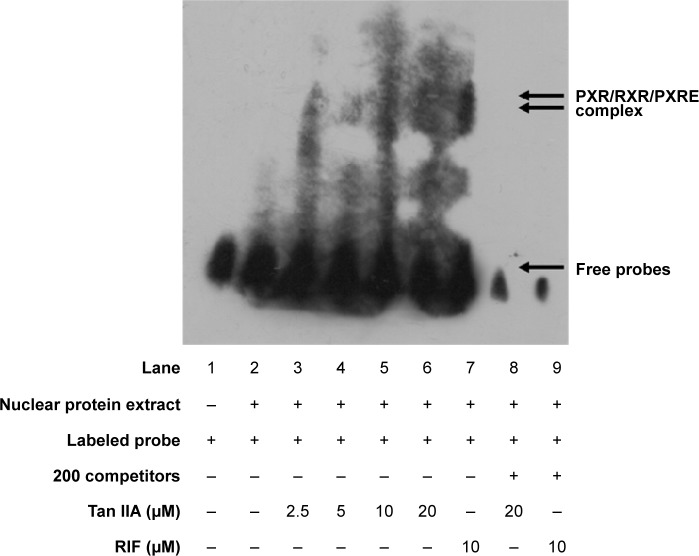
The interactions between the PXR heterodimer and nuclear receptor-binding motif of CYP3A4 were examined via EMSA using RXR proteins translated in vitro in the presence of Tan IIA. LS174T cells were treated with the indicated concentrations of Tan IIA for 24 hours. Then nuclear extraction was performed, and the resulting extracts were subjected to an EMSA assay. As shown in Figure 3, 10 μM RIF (positive control) was associated with visibly better DNA-binding by PXR to oligonucleotides containing PXRE compared to the solvent control (Figure 3, lanes 2 and 9). Significantly enhanced binding of PXR to the PXRE consensus sequence was also observed for Tan IIA-treated cells, with a concentration-dependent increase over the range of 2.5–20 μM. The maximum increase was seen at 20 μM (Figure 3, lane 6). The specificity of this band as a PXR-mediated gel shift was confirmed by nuclear extracts from cells treated with 20 μM Tan IIA (Figure 3, lane 8). The binding shift was abolished in the presence of excess concentrations of unlabeled PXRE. As indicated by qRT-PCR, Western-blot analysis, and the reporter assay, induction of human CYP3A4 at the mRNA and protein levels, and luciferase reporter gene activity of Tan IIA was mediated through activation of PXR and initiated by PXR-PXRE binding.
Figure 3.
Tan IIA, RIF, and the DNA-binding activity of nuclear pregnane X receptor.
Notes: The double arrows indicate the specific binding of the PXR complex to PXRE, which competes against the presence of 200-fold excess of unlabeled PXRE (lanes 8 and 9); The single arrow indicates the free probes; Only the labeled probes were added to the reaction system (lane 1).
Abbreviations: PXR, pregnane X receptor; PXRE, pregnane X response element; RXR, retinoid X receptor; RIF, rifampicin; Tan IIA, tanshinone IIA.
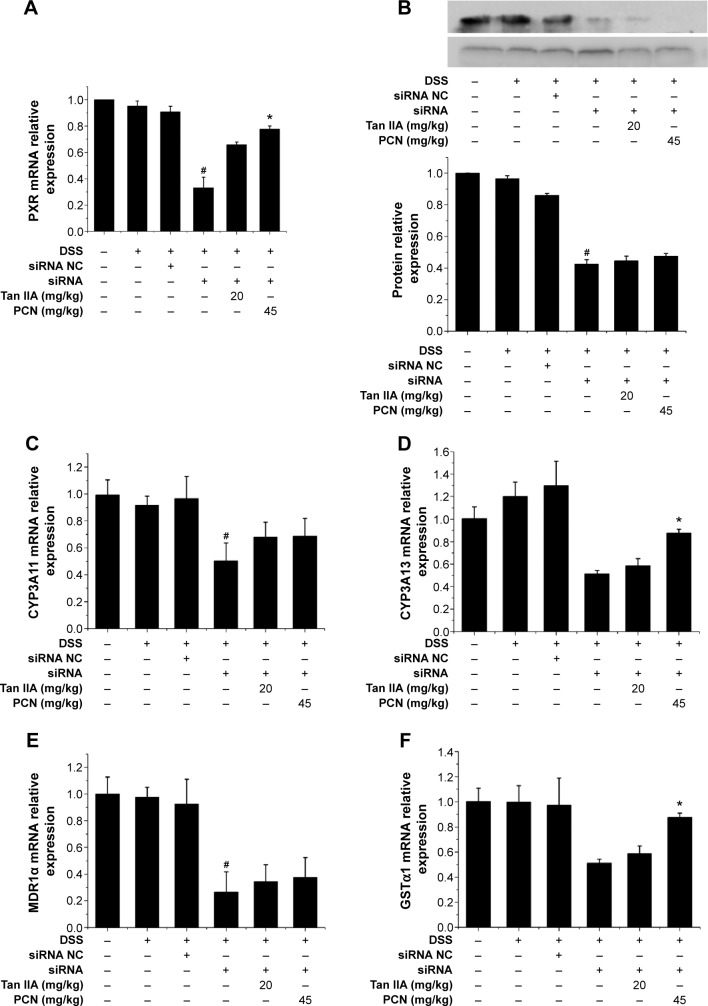
Figure 10.
Effects of Tan IIA on the expression of PXR target genes in PXR-silenced mice.
Notes: qRT-PCR and Western blotting were performed to measure the expression of PXR from colon tissue isolated from vehicle-, DSS-, siRNA-, siRNA NC-, Tan IIA-, and PCN-treated PXR lacking mice; (A) mRNA expression of PXR; (B) Protein expression of PXR, (C) CYP3A11, (D) CYP3A13, (E) MDR1α, and (F) GSTα1 expression was normalized to β-actin; Each bar represents the means ± SD (n=4); *P<0.01 versus PXR siRNA-treated group. #P<0.01 versus vehicle-treated group.
Abbreviations: CYP, cytochrome P450; DSS, dextran sodium sulfate; GSTα1, glutathione S-transferases alpha 1; MDR1α, multidrug resistance 1 alpha; mRNA, messenger RNA; PXR, pregnane X receptor; qRT-PCR, quantitative reverse transcriptase polymerase chain reaction; SD, standard deviation; siRNA, small interfering RNA; NC, negative control; Tan IIA, tanshinone IIA.
Tan IIA treatment and amelioration of DSS-induced IBD in vivo
Tan IIA treatment and DSS-induced IBD
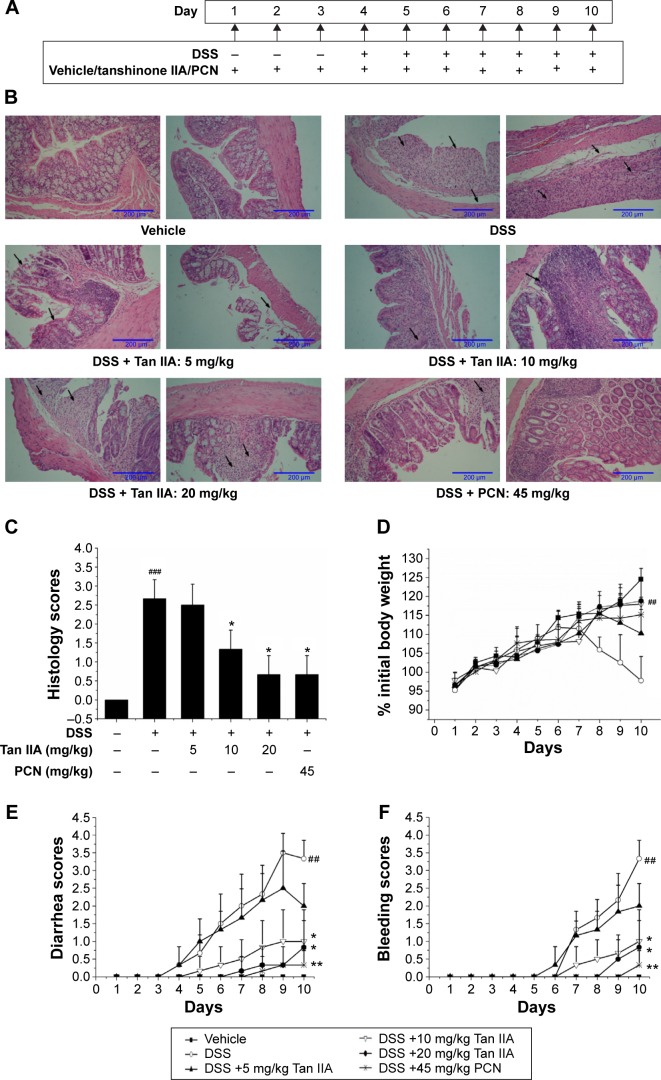
The DSS model of IBD closely simulates the conditions of the human body. Oral administration of DSS in mice induces colitis resembling human UC, and the inflammation was mainly localized to the colon. To define the treatment function of Tan IIA against the DSS-induced IBD model, the DSS-induced IBD was simultaneously treated with Tan IIA or PCN following the dosing scheme in Figure 4A. Mice were then killed, and their colons were analyzed. The histological assessments clearly indicated that the severe injury was caused by DSS and DSS-induced colonic injury was less severe in PCN- or Tan IIA-treated mice (Figure 4B and C). H&E-stained colon sections showed large necrotic foci in mice treated with DSS, and this pattern was both significantly concentration-dependent and significantly attenuated by coadministration of Tan IIA (5, 10, and 20 mg/kg) and PCN (45 mg/kg).
Figure 4.
Clinical assessment of dextran sulfate sodium (DSS)-induced inflammatory bowel disease in vehicle-, Tan IIA-, and PCN-treated mouse model.
Notes: (A) Dosing scheme; (B) Representative hematoxylin and eosin stained colon sections (left 20×, right 40× magnification); areas of severe colon necrosis are marked with arrows; (C) Histology scores, as assessed according to morphological criteria and the proportion of the colon that was affected by the individual changes relative to the total area of the colon section; (D) Changes in body weight after DSS induction of colitis; (E) Diarrhea scores; (F) Bleeding scores; (G) Macroscopic observation of colon length; (H) Colon lengths; DSS-stimulated elevated histology scores, diarrhea, and bleeding while the body weight and colon lengths were significantly reduced by cotreatment with different doses of Tan IIA or 45 mg/kg PCN; the injury has been ameliorated in dose-dependent manner; Each bar represents the means ± SD (n=10). *P<0.01, **P<0.001 versus DSS-treated group. #P<0.05, ##P<0.01, ###P<0.001 versus vehicle-treated group.
Abbreviations: PCN, pregnenolone 16a-carbonitrile; SD, standard deviation; Tan IIA, tanshinone IIA.
Clinical symptoms in mice (body weight loss, diarrhea, bleeding, and colon shortening) were also analyzed as shown in Figure 4D–H. Significant differences were observed in the severity of DSS-induced groups of mice, as indicated by major clinical symptoms. PCN was associated with significantly better scores than the DSS group. The clinical parameters associated with colitis showed statistically significantly better values after Tan IIA pretreatment than in the case of DSS administered alone, and Tan IIA-treated mice showed a concentration-dependent increase in all scores over the range of 5–20 mg/kg. Taken together, these data clearly demonstrate that mice treated with Tan IIA or PCN demonstrated significant protection against DSS-induced IBD in a concentration-dependent manner compared with DSS treatment alone.
Tan IIA, activation of PXR target genes, and downregulation of inflammatory mediator expression in colonic tissue
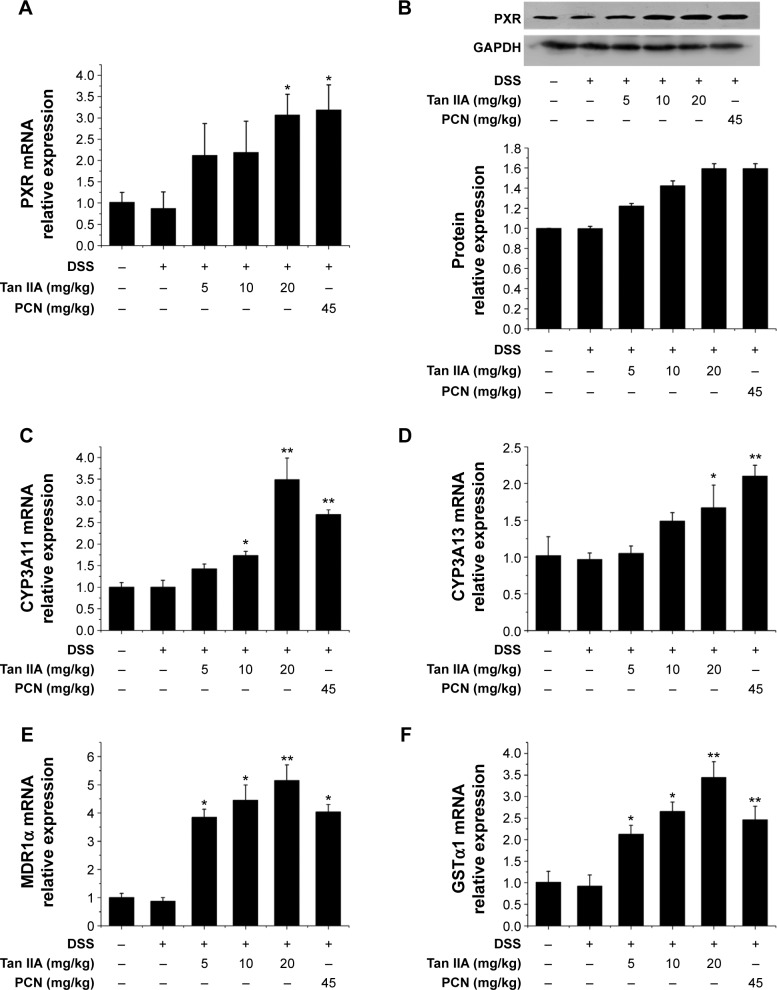
PXR acts as the key treatment target of IBD. It is here hypothesized that the effects of Tan IIA treatment in DSS-induced colitis might be linked to PXR activation. The colon gene and protein expression profiles of PXR were examined using qRT-PCR and Western blotting. Figure 5A and B demonstrates that DSS slightly inhibited PXR expression. Tan IIA cotreatment reversed DSS-induced decreases of PXR to normal levels. This reversal was further enhanced by Tan IIA in a concentration-dependent manner. PXR, which is a xenobiotic sensor nuclear receptor, upregulates the expression of xenobiotic oxidation and conjugation enzymes and transporters involved in the elimination of potentially harmful chemicals from the body. It has been hypothesized that the detoxification properties of PXR and its target genes are necessary to maintaining the integrity of the intestinal epithelial barrier and, for this reason, they contribute to the anti-inflammatory effect in the intestines. Therefore, expression of the metabolism enzymes (eg, Cyp3a11 and Cyp3a13 MDR1, Gsta1, etc) was also assessed. As shown in Figure 5C–F, DSS slightly decreased the levels of Cyp3a11, Cyp3a13, Mdr1, and Gsta1 mRNA, but expression recovered and increased dramatically upon cotreatment with Tan IIA. In addition, PCN cotreatment exerted effects similar to those of Tan IIA. Taken together, these data suggest that the upregulation of xenobiotic detoxification genes might contribute to the effects of Tan IIA in abrogating experimental IBD. For the best treatment effect, 20 mg/kg Tan IIA was chosen for the following study.
Figure 5.
Tan IIA altered the expression profile of PXR target genes in a DSS-induced mouse model of inflammatory bowel disease.
Notes: Expression of mRNAs encoding PXR, CYP3A11, CYP3A13, MDR1α, and GSTα1 was determined by qRT-PCR from colon epithelial cells isolated from vehicle-, Tan IIA-, and PCN-treated mice; (A) PXR gene expression in mice model; (B) PXR protein expression in mice model; (C) CYP3A11; (D) CYP3A13; (E) MDR1α; (F) GSTα1; Expression was normalized to β-actin; Each bar represents the means ± SD (n=4); *P<0.01, **P<0.001 versus DSS-treated group.
Abbreviations: CYP, cytochrome P450; DSS, dextran sodium sulfate; GSTα1, glutathione S-transferases alpha 1; MDR1α, multidrug resistance 1 alpha; mRNAs, messenger RNAs; PCN, pregnenolone 16a-carbonitrile; PXR, pregnane X receptor; qRT-PCR, quantitative reverse transcriptase polymerase chain reaction; SD, standard deviation; Tan IIA, tanshinone IIA; GAPDH, glyceraldehyde phosphate dehydrogenase.
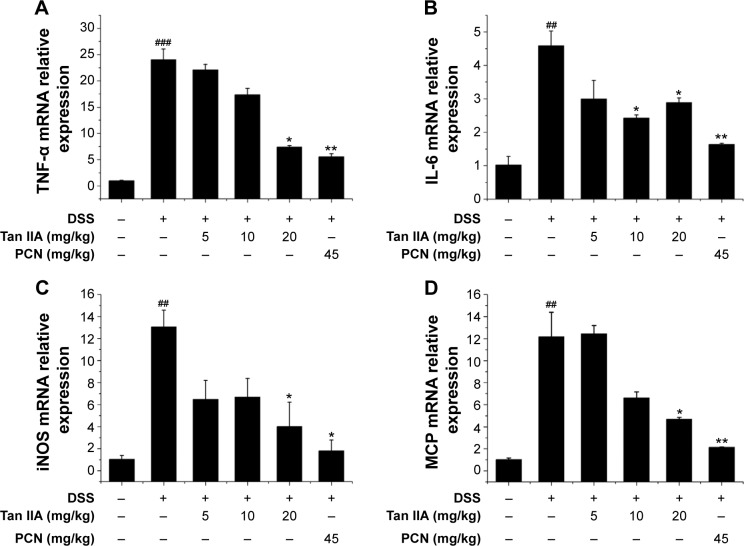
Inflammatory mediators are the central transcription factors in the regulation of proinflammatory signaling pathways involved in IBD. Previous studies demonstrated that PXR activation decreases the susceptibility of mice to DSS-induced colitis via suppression of inflammatory mediators, and activation of PXR can attenuate NF-κB signaling.5 It is here reasoned that the anti-inflammatory effect of Tan IIA in response to DSS-induced IBD was associated with the blockage of NF-κB activation. To elucidate the mechanism underling the role of Tan IIA in DSS-induced colitis, several inflammatory mediator genes in the colon were subjected to qRT-PCR analysis. As shown in Figure 6, results indicate that mRNA expression of TNFα, IL-6, iNOS, and monocyte chemoattractant protein (MCP) was markedly induced in the inflamed colons of mice that had been exposed to DSS. In contrast, the increases in inflammatory mediators after DSS treatment were significantly lower in mice treated with Tan IIA or PCN. These data suggested that Tan IIA ameliorates experimental colitis through repression of inflammatory mediators.
Figure 6.
Cytokine expression in colonic tissue after DSS-induced inflammatory bowel disease (IBD) in vehicle-, Tan IIA-, and PCN-treated mice.
Notes: (A) TNF-α, (B) IL-6, (C) iNOS, and (D) MCP mRNA expression was assessed by qRT-PCR in colon RNA isolated from DSS-induced IBD in vehicle-, Tan IIA-, and PCN-treated mice; Expression was normalized to β-actin; Each bar represents the mean ± SD (n=4); *P<0.05 and **P<0.01 versus DSS-treated group; ##P<0.05 and ###P<0.001 versus vehicle-treated group.
Abbreviations: DSS, dextran sodium sulfate; IL-6, interleukin 6; iNOS, inducible nitric oxide synthase; mRNA, messenger RNA; PCN, pregnenolone 16a-carbonitrile; qRT-PCR, quantitative reverse transcriptase polymerase chain reaction; SD, standard deviation; Tan IIA, tanshinone IIA; TNF-α, tumor necrosis factor alpha; MCP, monocyte chemoattractant protein.
PXR-dependency of Tan IIA and intestinal inflammation in a mouse model of colitis
siRNA knockdown of gene and protein expression of PXR in a PXR-free mouse colon model
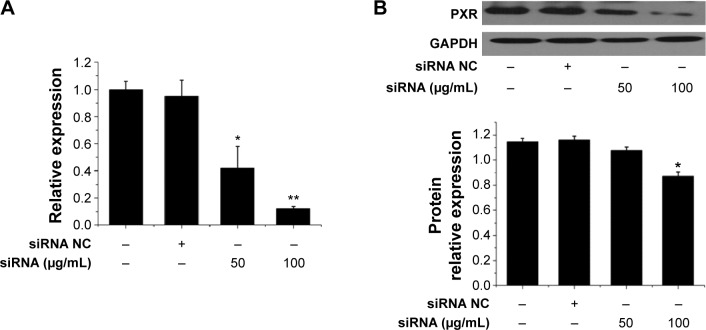
To further investigate the mechanisms underlying the effects of Tan IIA during DSS-induced IBD, the effects of Tan IIA were assessed in PXR-silenced mice. A modified hydrodynamic tail vein injection was used to deliver synthetic siRNA into mouse hepatocytes in vivo. PXR protein expression in the colon was then measured by immunoblotting 72 hours after injection. Data indicated that PXR siRNA reduced PXR protein expression by ≈75% relative to vehicle and negative siRNA injection controls in colon tissues (Figure 7A and B). This indicated that PXR siRNA could reduce the expression of PXR in colon tissues effectively.
Figure 7.
Effects of siRNA on PXR gene and protein expression in mice model.
Notes: PXR expression in colon tissues was assessed using qRT-PCR in vehicle-, siRNA NC-, and siRNA-treated mice; (A) mRNA expression was assessed by qRT-PCR; (B) Protein expression was assessed by Western blot; Expression was normalized to β-actin; Each bar represents the means ± SD (n=4); *P<0.01, **P<0.001 versus PXR siRNA-treated group.
Abbreviations: mRNA, messenger RNA; NC, negative control; PXR, pregnane X receptor; qRT-PCR, quantitative reverse transcriptase polymerase chain reaction; SD, standard deviation; siRNA, small interfering RNA; GAPDH, glyceraldehyde phosphate dehydrogenase.
Tan IIA and DSS-induced IBD model in a PXR-free mouse colon model
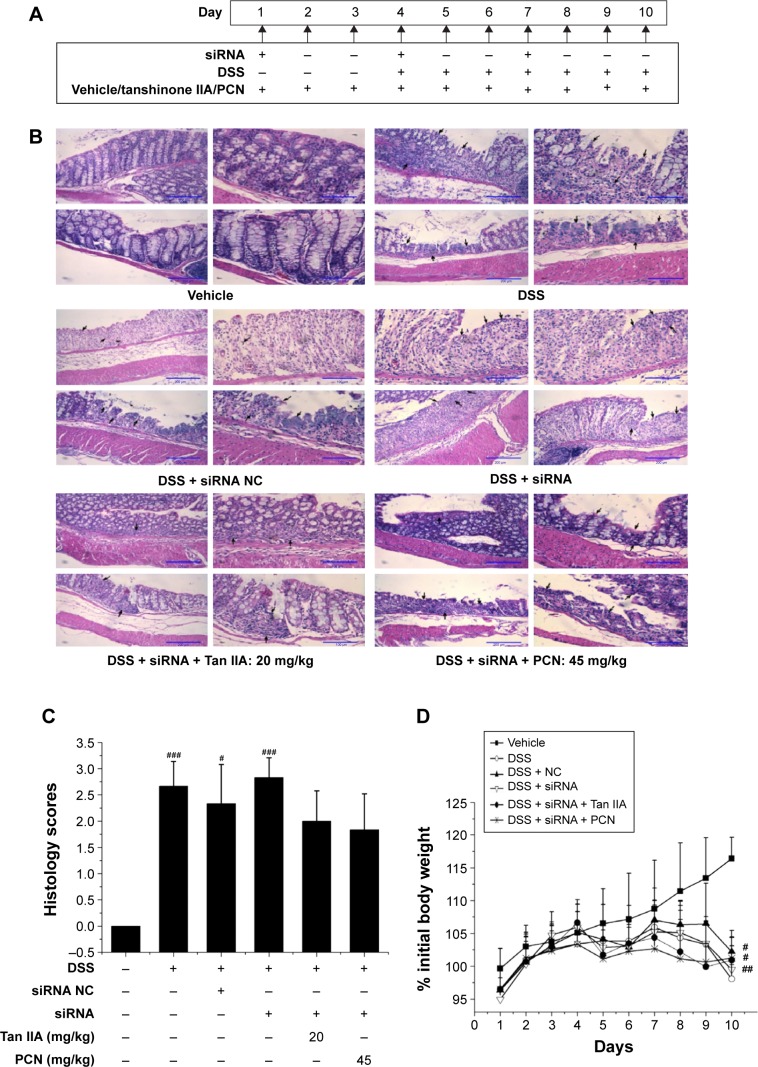
To assess the role of PXR in the effects of Tan IIA on DSS-induced IBD, the PXR gene was silenced by PXR siRNA transfection. The model was evaluated in mice treated with PXR siRNA and negative siRNA. Mice with reduced PXR expression were simultaneously treated with Tan IIA or PCN for 10 days and then given oral DSS for the next 7 days. siRNA-mediated gene suppression in mouse colons in vivo was examined in a preliminary experiment after 72 hours, and siRNA injections were repeated once every 72 hours for 10 consecutive days following the dosing scheme, as shown in Figure 8A.
Figure 8.
Clinical assessment of dextran sulfate sodium (DSS)-induced inflammatory bowel disease (IBD) in vehicle-, DSS-, siRNA-, siRNA NC-, Tan IIA-, and PCN-treated PXR lacking mouse model.
Notes: (A) Dosing scheme; (B) Representative hematoxylin and eosin stained colon sections (20× magnification); areas of severe colon necrosis are marked with arrows; (C) Histology scores, as assessed according to morphological criteria and the proportion of the colon that was affected by the individual changes relative to the total area of the colon section; (D) Changes in body weight after DSS induction of colitis; (E) Diarrhea scores; (F) Bleeding scores; (G) Macroscopic observation of colon length; (H) Colon lengths; Mice treated with DSS or DSS combined with negative siRNA were accompanied by worse clinical symptoms than the vehicle group, and mice with silenced PXR expression cotreated with 20 mg/kg Tan IIA or 45 mg/kg PCN could slightly aggravate severity in DSS-induced IBD; Each bar represents the means ± SD (n=10). *P<0.01 versus PXRsirna-treated group. #P<0.05, ##P<0.01, ###P<0.001 versus vehicle-treated group.
Abbreviations: NC, negative control; PCN, pregnenolone 16a-carbonitrile; PXR, pregnane X receptor; SD, standard deviation; siRNA, small interfering RNA; Tan IIA, tanshinone IIA.
As shown in Figure 8B and C, histological analysis of H&E-stained colon sections revealed large-scaled necrotic foci in mice treated with DSS or DSS combined with negative siRNA. Mice with reduced PXR expression had slightly aggravated pathological changes in the colon tissue. In addition, PXR-knockdown mice that were cotreated with 20 mg/kg Tan IIA or 45 mg/kg PCN exhibited fewer subcapsular foci of white discoloration in the colon than PXR-silenced mice. However, although all mice exhibited some reduction in colon damage, the pathological changes in the colon could still be distinguished clearly, and there remained significant and severe multifocal necrosis and biliary proliferation.
Consistent with the histological findings, the major clinical symptoms (body weight loss, diarrhea, bleeding, and shortening of the colon) results demonstrated that mice treated with DSS or DSS combined with negative siRNA had worse clinical symptoms than the vehicle group. As shown in Figure 8D–H, mice with reduced PXR expression slightly aggravated the severity of DSS-induced IBD and this damage was slightly augmented by cotreatment with 20 mg/kg Tan IIA or 45 mg/kg PCN.
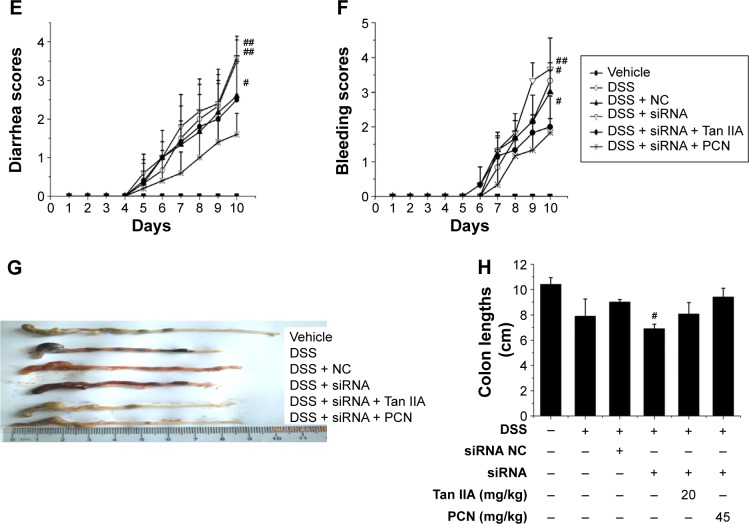
Clinical symptoms from two experiments are contrasted in Figure 9, pathology, body weight loss, diarrhea, bleeding, and shortening of the colon. The data show a marked statistical distinction between PXR-silenced mice and WT mice. These data demonstrate that mice with silenced PXR expression cotreated with 20 mg/kg Tan IIA or 45 mg/kg PCN showed slightly aggravated DSS-induced IBD, but the pathological changes could still be distinguished clearly and were statistically significantly more severe than those of WT mice cotreated with 20 mg/kg Tan IIA or 45 mg/kg PCN. For this reason, the therapeutic effect was significantly less pronounced in PXR-silenced mice than in WT mice.
Figure 9.
Comparison of clinical symptoms of dextran sulfate sodium (DSS)-induced inflammatory bowel disease (IBD) treated with 20 mg/kg Tan IIA or 45 mg/kg PCN in WT and PXR siRNA-treated mouse model.
Notes: (A) Histology scores and (B) changes in body weight after DSS induction of colitis; (C) Diarrhea scores; (D) Bleeding scores; Marked statistical distinction can be found between PXR-silenced mice and WT mice; Mice with silenced PXR expression cotreated with 20 mg/kg Tan IIA or 45 mg/kg PCN could slightly aggravate severity in DSS-induced IBD, and the therapeutic effect was significantly reduced in PXR-silenced mice compared with WT mice; Each bar represents the means ± SD (n=10); ##P<0.05, ###P<0.001 versus vehicle-treated group; *P<0.01 represents WT versus PXR siRNA-treated mouse model group.
Abbreviations: PCN, pregnenolone 16a-carbonitrile; PXR, pregnane X receptor; SD, standard deviation; siRNA, small interfering RNA; Tan IIA, tanshinone IIA; WT, wild-type.
Expression of Tan IIA on PXR and inflammatory mediators in a DSS-induced IBD model and in a PXR-free mouse colon model
The PXR expression profile was analyzed by qRT-PCR. The PXR gene was efficiently silenced to approximately 33.1% than that of negative siRNA groups, as shown in Figure 10A. PXR-silenced mice coadministered Tan IIA and PCN saw expression increase by 65.8% and 77.7%, respectively, indicating that the PXR gene still had some function. To confirm the qRT-PCR results indicating the effects of Tan IIA on PXR, the expression of PXR protein was detected using Western-blot analysis, as shown in Figure 10B. The data also demonstrated the success of PXR silencing.
As demonstrated in Figure 10C–F, the mRNA levels of Cyp3a11, Cyp3a13, Mdr1, and Gsta1 were still far lower in the PXR-silenced mice than in negative mouse group and normal group, though Tan IIA and PCN cotreatment was found to induce the expression of PXR, Cyp3a11, Cyp3a13, Mdr1, and Gsta1 genes. The expression of PXR, Cyp3a11, Cyp3a13, Mdr1, and Gsta1 genes demonstrated similar effects in histological assessments and clinical symptoms.
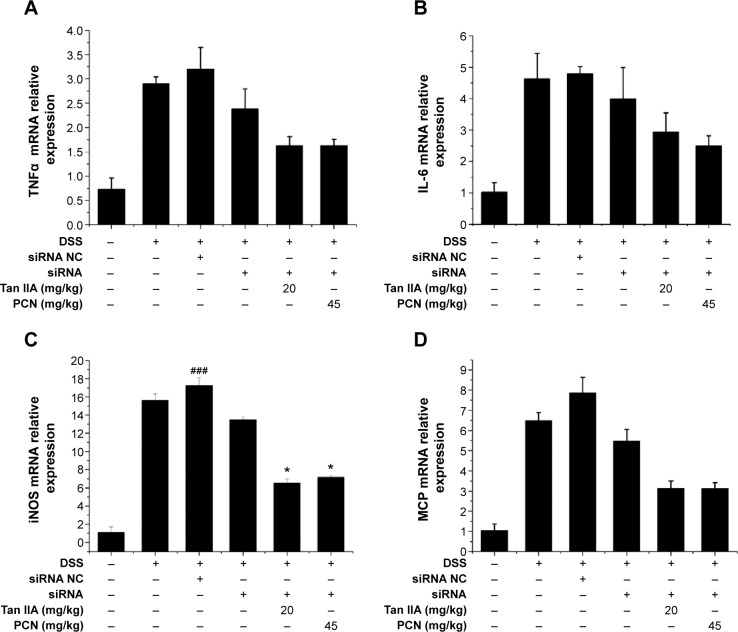
To further elucidate the mechanisms underlying the effects of Tan IIA in DSS-induced colitis in PXR-silenced mice, qRT-PCR analyses of several inflammatory mediator genes in the colon were performed. As shown in Figure 11, results indicate that mRNA expression of TNFα, IL-6, iNOS, and MCP was markedly induced in the inflamed colons of mice exposed to DSS-, siRNA-DSS, and siRNA NC-DSS. Levels of inflammatory mediators were lower in Tan IIA and PCN cotreatment siRNA-DSS PXR-silenced mice, and the effects of treatment were markedly decreased. These data suggest that the effects of Tan IIA on IBD may be partially due to PXR-mediated upregulation of xenobiotic metabolism and downregulation of inflammatory mediators and not due to the general anti-inflammatory effects in DSS-induced IBD.
Figure 11.
Tan IIA altered the expression profile of cytokine expression in colon tissue harvested from vehicle-, DSS-, siRNA-, siRNA NC-, Tan IIA-, and PCN-treated PXR lacking mice.
Notes: (A) TNFα, (B) IL-6, (C) iNOS, and (D) MCP mRNA expression was assessed by qRT-PCR; Expression was normalized to β-actin; Each bar represents the means ± SD (n=4); ###P<0.001 versus vehicle-treated group; *P<0.01 versus PXR siRNA-treated group.
Abbreviations: DSS, dextran sodium sulfate; IL-6, interleukin 6; iNOS, inducible nitric oxide synthase; mRNA, messenger RNA; NC, negative control; PCN, pregnenolone 16a-carbonitrile; PXR, pregnane X receptor; qRT-PCR, quantitative reverse transcriptase polymerase chain reaction; SD, standard deviation; siRNA, small interfering RNA; Tan IIA, tanshinone IIA; TNFα, tumor necrosis factor alpha; MCP, monocyte chemoattractant protein.
Discussion
These experiments identified the mechanism underlying the action of Tan IIA in the treatment of IBD associated with inflammation and the PXR.
Clinical symptoms in mice (ie, body weight loss, diarrhea, bleeding, and colon shortening) and histological assessment data suggested that administration of Tan IIA in mice offered significant protection and may have had a therapeutic role against DSS-induced IBD. They also showed that its effects are concentration-dependent. PXR-silenced mice treated with Tan IIA experienced little protection against DSS-induced mouse IBD and suffered more severe IBD than WT mice; PXR-silenced mice exposed to siRNA demonstrated the necessity of PXR in Tan-IIA-mediated upregulation of xenobiotic metabolism genes. The IBD treatment effects of Tan IIA are partially due to PXR-mediated upregulation of xenobiotic metabolism, and downregulation of expression of inflammatory mediators are not due to the general anti-inflammatory effects in DSS-induced IBD. The novel findings presented here may contribute to the effective utilization of Tan IIA or its derivatives as a PXR ligand in the treatment of human IBD. These results suggest the clinical utility of Tan IIA.
Tan IIA is one of the major lipophilic active components in a conventional Chinese medicine called danshen. The current results on mRNA, protein expression, and luciferase reporter gene activity suggest that Tan IIA is an efficacious PXR agonist, and, like most CYP3A4 inducers, the induction of CYP3A4 by Tan IIA is mediated by the transactivation of PXR in a visible concentration- and time-dependent manner. In addition, Tan IIA has been shown to be beneficial in specific cases of IBD in traditional Chinese medicine.34 Previous studies have reported that Tan IIA can ameliorate trinitrobenzene sulfonic acid-induced colitis in a dose-dependent manner.23 In most cases, first-line therapy includes immune-suppressing drugs, and studies of mechanisms are mainly concerned with anti-inflammatory activity.
The etiology of IBD is still not completely understood. However, it is widely accepted that the development of IBD is associated with the interplay of genetic, bacterial, and environmental factors and dysregulation of the intestinal immune system.35,36 The treatment for IBD is highly individualized. Studies have indicated that PXR can be involved in IBD. PXR may also be a suitable target for abrogating inflammation in IBD. PXR is a ligand-activated transcription factor that is important for induction of drug transport and metabolism, especially the induction of cytochrome P450 CYP3A4, which is involved in the metabolism of many clinically used drugs. Gene expression analysis of colon tissues from patients with UC and IBD demonstrated significantly less expression of PXR and its target genes than in normal intestinal samples.10 Specific polymorphisms in the PXR locus that are associated with decreases in PXR activity have been found to be correlated with an increased susceptibility to IBD.11
Chemically induced animal models of IBD are common, particularly models involving DSS colitis. In the current study, acute colitis was induced using DSS. The preventive and therapeutic role of Tan IIA and its mechanism were assessed in DSS-induced IBD models. The clinical symptoms in mice (body weight loss, diarrhea, bleeding, and colon shortening) demonstrated that mice treated with Tan IIA or PCN had significant protection, that Tan IIA acts in a concentration-dependent manner, and that Tan IIA may have a therapeutic role in DSS-induced IBD. PXR siRNA was found to reduce the expression of PXR in the colon effectively, confirming that the experiments were valid and pharmacologically significant. PXR-silenced mice treated with Tan IIA or PCN demonstrated little protection against DSS-induced mouse IBD and had more severe of IBD and less of a response to treatment than WT mice. The results suggest that the effects of Tan IIA had been impaired and that it was unable to exert efficient action in the absence of PXR. For this reason, WT and PXR-silenced mice were assessed to establish the preventive and therapeutic roles of Tan IIA in DSS-induced IBD models.
Previous studies indicated that Tan IIA contributes to the maintenance of the integrity of the intestinal barrier by regulating the metabolism of xenobiotics and by increasing the expression and activity of PXR and PXR-regulated genes.37,38 It also indicated that there were less mRNA levels of Cyp3a11, Cyp3a13, Mdr1, and Gsta1 expression in PXR-silenced mice than in negative siRNA groups. Tan IIA and PCN cotreatment induced more gene expression than in the PXR-silenced mice, but levels were still far below normal. The slight amelioration of IBD induced by Tan IIA and PCN may be due to PXR not being completely silenced. PXR-silenced has some physiological and pathological significance and could activate metabolic enzymes. PXR is a xenobiotic sensor nuclear receptor, and it activates upregulation of the expression of xenobiotic oxidation and conjugation enzymes and transporters involved in the elimination of potentially harmful chemicals from the body. It has been hypothesized that the detoxification properties of PXR and its target genes are necessary to maintaining the integrity of the intestinal epithelial barrier and that, in this way, they contribute to the anti-inflammatory effect in intestines.39 Thus, the upregulation of xenobiotic detoxification genes might contribute to the effects of Tan IIA in abrogating experimental IBD.
Assessments of gene expression of inflammatory mediators showed that mRNA expression of TNFα, IL-6, iNOS, and MCP were markedly induced in the inflamed colons of mice that had been exposed to DSS. In contrast, the increases in inflammatory mediators after DSS treatment were significantly less pronounced in mice treated with Tan IIA and PCN. Because PXR-silenced mice treated with Tan IIA and PCN still had high levels of inflammatory mediator expression, it is here hypothesized that the anti-inflammatory effect of Tan IIA in response to DSS-induced IBD may be associated with the blockage of inflammatory mediator activation. In this way, the effects of Tan IIA on IBD might be mediated partially by the upregulation of the PXR nuclear receptor, Cyp3a11, Cyp3a13, and Mdr1. Downregulation of inflammatory mediators and Tan IIA inhibited the NF-κB signaling cascade in a human PXR-dependent manner.
NF-κB is the central transcription factor in the regulation of proinflammatory signaling pathways involved in IBD.9,40 Previous studies have demonstrated that PXR activation decreases the susceptibility of mice to DSS-induced colitis via suppression of NF-κB and that activation of PXR can attenuate NF-κB signaling.5 Treatment with the NF-κB pathway activator leads to a marked increase in NF-κB reporter activity, and over-expression of human PXR inhibits NF-κB reporter activity. PXR inhibits the proinflammatory transcription factor NF-κB, indicating that some mechanism may link PXR signaling to inflammation.5,41,42 Activation of mouse PXR ameliorates DSS-induced IBD via inhibition of NF-κB. In previous works, cryptotanshinone (CTN) significantly increased the survival rate against lipopolysaccharide (LPS) challenge in d-galactosamine (GalN)-sensitized mice.43,44 Park et al reported that CTN-containing purified extract of S. miltiorrhiza protects hepatocytes from GalN-induced liver toxicity in vitro.45 The same group also demonstrated that CTN can protect mice from GalN/LPS-induced fulminant hepatic failure. They found that CTN inhibited GalN/LPS-induced expression of proinflammatory cytokines via down regulation of NF-κB. It is here hypothesized that the ameliorating effect of Tan IIA on DSS-induced IBD is via PXR-mediated upregulation of xenobiotic metabolism and downregulation of NF-κB signaling.
This is the first report showing that the IBD treatment effects of Tan IIA are partially due to PXR-mediated upregulation of xenobiotic metabolism and downregulation of NF-κB signaling and not as a general anti-inflammatory in DSS-induced IBD. However, the specific mechanism is likely to be governed by a number of factors, including the age, strain, and sex of the mice, as well as by their housing conditions and the duration and dose of DSS administered. In summary, these findings may contribute to the effective utilization of Tan IIA or its derivatives as a PXR ligand in the treatment of human IBD and indicate the clinical utility of Tan IIA.
Acknowledgments
Grants from the National Natural Science Foundation of China (grant 81073149), National Basic Research Program of China (“973 Program”) (grants 2011CB505304, 2012CB518402), and Scientific and Technological Major Special Project-Major Creation of New Drugs (grant 2009ZX09501-304) supported this study.
Footnotes
Disclosure
The authors report no conflicts of interest in this work.
References
- 1.Long MD, Crandall WV, Leibowitz IH, et al. ImproveCareNow Collaborative for Pediatric IBD Prevalence and epidemiology of overweight and obesity in children with inflammatory bowel disease. Inflamm Bowel Dis. 2011;17(10):2162–2168. doi: 10.1002/ibd.21585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Podolsky DK. Inflammatory bowel disease. N Engl J Med. 2002;347(6):417–429. doi: 10.1056/NEJMra020831. [DOI] [PubMed] [Google Scholar]
- 3.Vermeire S, Van Assche G, Rutgeerts P. Inflammatory bowel disease and colitis: new concepts from the bench and the clinic. Curr Opin Gastroenterol. 2011;27(1):32–37. doi: 10.1097/MOG.0b013e3283412e87. [DOI] [PubMed] [Google Scholar]
- 4.Cheng J, Shah YM, Ma X, et al. Therapeutic role of rifaximin in inflammatory bowel disease: clinical implication of human pregnane X receptor activation. J Pharmacol Exp Ther. 2010;335(1):32–41. doi: 10.1124/jpet.110.170225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Xie W, Tian Y. Xenobiotic receptor meets NF-kappaB, a collision in the small bowel. Cell Metab. 2006;4(3):177–178. doi: 10.1016/j.cmet.2006.08.004. [DOI] [PubMed] [Google Scholar]
- 6.Yan FP, Zheng K, Wang L, et al. Measurement of thickness and refractive index of Zn1-xMgxO film grown on sapphire substrate by molecular beam epitaxy. Acta Phys Sin-Ch Ed. 2007;56(7):4127–4131. [Google Scholar]
- 7.Langmann T, Schmitz G. Loss of detoxification in inflammatory bowel disease. Nat Clin Pract Gastroenterol Hepatol. 2006;3(7):358–359. doi: 10.1038/ncpgasthep0545. [DOI] [PubMed] [Google Scholar]
- 8.Dou W, Zhang J, Zhang E, et al. Chrysin ameliorates chemically induced colitis in the mouse through modulation of a PXR/NF-κB signaling pathway. J Pharmacol Exp Ther. 2013;345(3):473–482. doi: 10.1124/jpet.112.201863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Andersen V, Christensen J, Ernst A, et al. Polymorphisms in NF-κB, PXR, LXR, PPARγ and risk of inflammatory bowel disease. World J Gastroenterol. 2011;17(2):197–206. doi: 10.3748/wjg.v17.i2.197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Langmann T, Moehle C, Mauerer R, et al. Loss of detoxification in inflammatory bowel disease: dysregulation of pregnane X receptor target genes. Gastroenterology. 2004;127(1):26–40. doi: 10.1053/j.gastro.2004.04.019. [DOI] [PubMed] [Google Scholar]
- 11.Dring MM, Goulding CA, Trimble VI, et al. The pregnane X receptor locus is associated with susceptibility to inflammatory bowel disease. Gastroenterology. 2006;130(2):341–348. doi: 10.1053/j.gastro.2005.12.008. quiz 592. [DOI] [PubMed] [Google Scholar]
- 12.Wistuba W, Gnewuch C, Liebisch G, Schmitz G, Langmann T. Lithocholic acid induction of the FGF19 promoter in intestinal cells is mediated by PXR. World J Gastroenterol. 2007;13(31):4230–4235. doi: 10.3748/wjg.v13.i31.4230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Glas J, Seiderer J, Fischer D, et al. Pregnane X receptor (PXR/NR1I2) gene haplotypes modulate susceptibility to inflammatory bowel disease. Inflamm Bowel Dis. 2011;17(9):1917–1924. doi: 10.1002/ibd.21562. [DOI] [PubMed] [Google Scholar]
- 14.Dou W, Mukherjee S, Li H, et al. Alleviation of gut inflammation by Cdx2/Pxr pathway in a mouse model of chemical colitis. PLoS One. 2012;7(7):e36075. doi: 10.1371/journal.pone.0036075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Qiu F, Zhang R, Sun J, et al. Inhibitory effects of seven components of danshen extract on catalytic activity of cytochrome P450 enzyme in human liver microsomes. Drug Metab Dispos. 2008;36(7):1308–1314. doi: 10.1124/dmd.108.021030. [DOI] [PubMed] [Google Scholar]
- 16.Sung HJ, Choi SM, Yoon Y, An KS. Tanshinone IIA, an ingredient of Salvia miltiorrhiza BUNGE, induces apoptosis in human leukemia cell lines through the activation of caspase-3. Exp Mol Med. 1999;31(4):174–178. doi: 10.1038/emm.1999.28. [DOI] [PubMed] [Google Scholar]
- 17.Cheng TO. Danshen: a versatile Chinese herbal drug for the treatment of coronary heart disease. Int J Cardiol. 2006;113(3):437–438. doi: 10.1016/j.ijcard.2005.10.026. [DOI] [PubMed] [Google Scholar]
- 18.Liu YW, Huang YT. Inhibitory effect of tanshinone IIA on rat hepatic stellate cells. PLoS One. 2014;9(7):e103229. doi: 10.1371/journal.pone.0103229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Chiu SC, Huang SY, Chen SP, Su CC, Chiu TL, Pang CY. Tanshinone IIA inhibits human prostate cancer cells growth by induction of endoplasmic reticulum stress in vitro and in vivo. Prostate Cancer Prostatic Dis. 2013;16(4):315–322. doi: 10.1038/pcan.2013.38. [DOI] [PubMed] [Google Scholar]
- 20.Jang SI, Jeong SI, Kim KJ, et al. Tanshinone IIA from Salvia miltiorrhiza inhibits inducible nitric oxide synthase expression and production of TNF-alpha, IL-1beta and IL-6 in activated RAW 264.7 cells. Planta Med. 2003;69(11):1057–1059. doi: 10.1055/s-2003-45157. [DOI] [PubMed] [Google Scholar]
- 21.Ueng YF, Kuo YH, Wang SY, Lin YL, Chen CF. Induction of CYP1A by a diterpene quinone tanshinone IIA isolated from a medicinal herb Salvia miltiorrhiza in C57BL/6J but not in DBA/2J mice. Life Sci. 2004;74(7):885–896. doi: 10.1016/j.lfs.2003.07.035. [DOI] [PubMed] [Google Scholar]
- 22.Kuo YH, Lin YL, Don MJ, Chen RM, Ueng YF. Induction of cytochrome P450-dependent monooxygenase by extracts of the medicinal herb Salvia miltiorrhiza. J Pharm Pharmacol. 2006;58(4):521–527. doi: 10.1211/jpp.58.4.0012. [DOI] [PubMed] [Google Scholar]
- 23.Bai A, Lu N, Guo Y, Fan X. Tanshinone IIA ameliorates trinitrobenzene sulfonic acid (TNBS)-induced murine colitis. Dig Dis Sci. 2008;53(2):421–428. doi: 10.1007/s10620-007-9863-8. [DOI] [PubMed] [Google Scholar]
- 24.Honkakoski P, Sueyoshi T, Negishi M. Drug-activated nuclear receptors CAR and PXR. Ann Med. 2003;35(3):172–182. doi: 10.1080/07853890310008224. [DOI] [PubMed] [Google Scholar]
- 25.Yu C, Ye S, Sun H, et al. PXR-mediated transcriptional activation of CYP3A4 by cryptotanshinone and tanshinone IIA. Chem Biol Interact. 2009;177(1):58–64. doi: 10.1016/j.cbi.2008.08.013. [DOI] [PubMed] [Google Scholar]
- 26.Cheng J, Ma X, Gonzalez FJ. Pregnane X receptor- and CYP3A4-humanized mouse models and their applications. Br J Pharmacol. 2011;163(3):461–468. doi: 10.1111/j.1476-5381.2010.01129.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Tompkins SM, Lo CY, Tumpey TM, Epstein SL. Protection against lethal influenza virus challenge by RNA interference in vivo. Proc Natl Acad Sci U S A. 2004;101(23):8682–8686. doi: 10.1073/pnas.0402630101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Robson MG, Sacks SH. RNA interference – a new experimental and therapeutic tool. Curr Opin Nephrol Hypertens. 2005;14(6):558–560. doi: 10.1097/01.mnh.0000168937.41270.34. [DOI] [PubMed] [Google Scholar]
- 29.Song E, Lee SK, Wang J, et al. RNA interference targeting Fas protects mice from fulminant hepatitis. Nat Med. 2003;9(3):347–351. doi: 10.1038/nm828. [DOI] [PubMed] [Google Scholar]
- 30.Zamore PD, Tuschl T, Sharp PA, Bartel DP. RNAi: double-stranded RNA directs the ATP-dependent cleavage of mRNA at 21 to 23 nucleotide intervals. Cell. 2000;101(1):25–33. doi: 10.1016/S0092-8674(00)80620-0. [DOI] [PubMed] [Google Scholar]
- 31.Bernstein E, Caudy AA, Hammond SM, Hannon GJ. Role for a bidentate ribonuclease in the initiation step of RNA interference. Nature. 2001;409(6818):363–366. doi: 10.1038/35053110. [DOI] [PubMed] [Google Scholar]
- 32.Lin SL, Sukasweang S, Chuong CM, Rasheed S, Ying SY. D-RNAi (messenger RNA-antisense DNA interference) as a novel defense system against cancer and viral infections. Curr Cancer Drug Targets. 2001;1(3):241–247. doi: 10.2174/1568009013334151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Zhang X, Yang J, Guo Y, et al. Functional proteomic analysis of nonalcoholic fatty liver disease in rat models: enoyl-coenzyme a hydratase down-regulation exacerbates hepatic steatosis. Hepatology. 2010;51(4):1190–1199. doi: 10.1002/hep.23486. [DOI] [PubMed] [Google Scholar]
- 34.Gionchetti P, Rizzello F, Lammers KM, et al. Antibiotics and probiotics in treatment of inflammatory bowel disease. World J Gastroentero. 2006;12(21):3306–3313. doi: 10.3748/wjg.v12.i21.3306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kaser A, Zeissig S, Blumberg RS. Inflammatory bowel disease. Annu Rev Immunol. 2010;28:573–621. doi: 10.1146/annurev-immunol-030409-101225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Sliva D, Loganathan J, Jiang J, et al. Mushroom Ganoderma lucidum prevents colitis-associated carcinogenesis in mice. PLoS One. 2012;7(10):e47873. doi: 10.1371/journal.pone.0047873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Mencarelli A, Migliorati M, Barbanti M, et al. Pregnane-X-receptor mediates the anti-inflammatory activities of rifaximin on detoxification pathways in intestinal epithelial cells. Biochem Pharmacol. 2010;80(11):1700–1707. doi: 10.1016/j.bcp.2010.08.022. [DOI] [PubMed] [Google Scholar]
- 38.Jiang H, Chen K, He J, et al. Association of pregnane X receptor with multidrug resistance-related protein 3 and its role in human colon cancer chemoresistance. J Gastrointest Surg. 2009;13(10):1831–1838. doi: 10.1007/s11605-009-0964-x. [DOI] [PubMed] [Google Scholar]
- 39.Cheng J, Shah YM, Gonzalez FJ. Pregnane X receptor as a target for treatment of inflammatory bowel disorders. Trends Pharmacol Sci. 2012;33(6):323–330. doi: 10.1016/j.tips.2012.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Zhang L, Ravipati AS, Koyyalamudi SR, et al. Antioxidant and anti-inflammatory activities of selected medicinal plants containing phenolic and flavonoid compounds. J Agric Food Chem. 2011;59(23):12361–12367. doi: 10.1021/jf203146e. [DOI] [PubMed] [Google Scholar]
- 41.Mencarelli A, Distrutti E, Renga B, Palladino G, Barbanti M, Fiorucci S. Inhibition of NF-kB by a PXR-dependent pathway mediates counter-regulatory activities of rifaximin on innate immunity in intestinal epithelial cells. Gastroenterology. 2011;140(5):S639. doi: 10.1016/j.ejphar.2011.06.058. [DOI] [PubMed] [Google Scholar]
- 42.Mencarelli A, Renga B, Palladino G, et al. Inhibition of NF-κB by a PXR-dependent pathway mediates counter-regulatory activities of rifaximin on innate immunity in intestinal epithelial cells. Eur J Pharmacol. 2011;668(1–2):317–324. doi: 10.1016/j.ejphar.2011.06.058. [DOI] [PubMed] [Google Scholar]
- 43.Li X, Lian LH, Bai T, et al. Cryptotanshinone inhibits LPS-induced proinflammatory mediators via TLR4 and TAK1 signaling pathway. Int Immunopharmacol. 2011;11(11):1871–1876. doi: 10.1016/j.intimp.2011.07.018. [DOI] [PubMed] [Google Scholar]
- 44.Tang S, Shen XY, Huang HQ, et al. Cryptotanshinone suppressed inflammatory cytokines secretion in RAW264.7 macrophages through inhibition of the NF-κB and MAPK signaling pathways. Inflammation. 2011;34(2):111–118. doi: 10.1007/s10753-010-9214-3. [DOI] [PubMed] [Google Scholar]
- 45.Park EJ, Zhao YZ, Kim YC, Sohn DH. Preventive effects of a purified extract isolated from Salvia miltiorrhiza enriched with tanshinone I, tanshinone IIA and cryptotanshinone on hepatocyte injury in vitro and in vivo. Food Chem Toxicol. 2009;47(11):2742–2748. doi: 10.1016/j.fct.2009.08.007. [DOI] [PubMed] [Google Scholar]