Abstract
Background
Plasma cell myeloma is the most common primary bone malignancy in adults. However, liver involvement in the form of an initial and asymptomatic nodular plasmacytoma is exceedingly rare.
Case Report
A 64-year-old male was found to have a right hepatic lobe nodule on a routine abdominal ultrasound prior to bariatric surgery. Liver biopsy revealed a plasma cell neoplasm that, given the location of the lesion, was favored to represent a lymphoma with prominent plasmacytic differentiation. Positron emission tomography (PET) demonstrated a hypermetabolic hepatic mass and identified multiple destructive bony lesions. Biopsy of a clavicular lesion revealed sheets of plasma cells and confirmed the diagnosis of multiple myeloma. The patient underwent 6 cycles of chemotherapy with cyclophosphamide, bortezomib, and dexamethasone before transitioning to lenalidomide and dexamethasone because of early disease progression. Although the patient had International Staging System I (low-risk) disease, his disease demonstrated an aggressive clinical course and resistance to multiple lines of therapy.
Conclusion
Extramedullary nodular hepatic plasmacytoma is exceedingly rare. Nevertheless, extramedullary plasmacytomas should be included in the differential diagnosis of patients with indistinct hepatic lesions visualized on computed tomography scan, especially if PET scans show associated bony lesions. In general, extramedullary plasmacytomas are a poor prognostic sign and a harbinger of an aggressive clinical course in the context of multiple myeloma.
Keywords: Hypercalcemia, liver neoplasms, multiple myeloma, neoplasms–plasma cell, plasmacytoma
INTRODUCTION
Multiple myeloma (MM) is a malignant proliferation of clonal plasma cells, characterized by infiltration of bone marrow and overproduction of monoclonal immunoglobulins (Igs) and/or free light chains.1 The incidence of extramedullary disease with newly diagnosed MM is variable, ranging from 7%-18%.2 Extramedullary plasmacytomas arise most commonly from direct extension of primary bone tumors, but rarely they may also result from hematogenous spread involving distant organs. Plasmacytoma involvement of the gastrointestinal system—more specifically, presentation as an asymptomatic nodular hepatic lesion—is exceedingly rare.3 We report the case of a patient with an incidental nodular hepatic lesion who was ultimately diagnosed with MM.
CASE REPORT
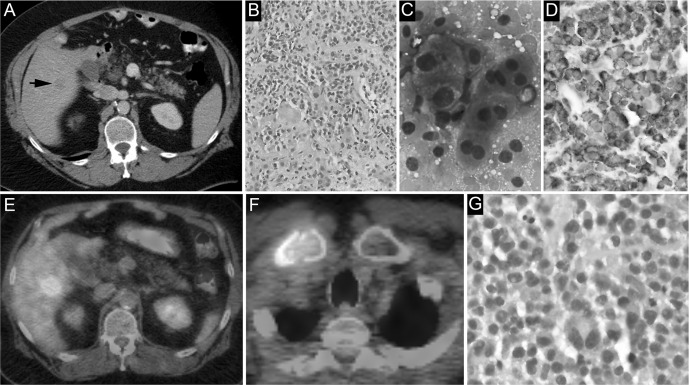
A 64-year-old male with a medical history significant for obesity and remote gastric stapling underwent preoperative workup for bariatric surgery revision. Routine ultrasound revealed an incidental 2.2 cm solid right hepatic lobe lesion, barely visible on contrast-enhanced computed tomography (CT) scan (Figure, A). The patient reported right shoulder pain, fatigue, and intentional weight loss, and his physical examination was unremarkable. Laboratory workup at this time was significant for mild hypercalcemia (10.3 mg/dL). An ultrasound-guided biopsy of the hepatic lesion performed 1 week later showed a plasma cell neoplasm. Although a lymphoid component was not identified by morphology or immunohistochemistry, given the location of the lesion and the lack of other identifiable lesions on CT scan, it was considered a primary hepatic lymphoma with plasmacytic differentiation (Figure, B-D).
Figure.
A: The lesion is barely visible on contrast-enhanced computed tomography scan (arrow). B: Core sections of an ultrasound-guided liver biopsy show confluent expansile sheets of plasma cells (hematoxylin and eosin [H&E] stain, ×20). C: Touch imprints of the liver biopsy reveal discohesive plasmacytoid cells and naked nuclei (Diff-Quik stain, ×100). D: Chromogenic in situ hybridization analysis demonstrates cytoplasmic kappa light chain restriction (immunohistochemical stain, ×40). E: Metabolically active lesions in the right hepatic lobe can be seen on positron emission tomography. F: Metabolically active lesions in the right clavicle can be seen on positron emission tomography. G: Core sections of right clavicular bone marrow biopsy show sheets of plasma cells (H&E stain, ×40).
A subsequent positron emission tomography (PET) scan showed a hypermetabolic hepatic mass and destructive bony lesions in the clavicle, manubrium, right third rib, pelvis, and sacrum (Figure, E and F). Biopsy of the clavicular lesion revealed confluent sheets of plasma cells (Figure, G). Tissue immunohistochemistry and the concurrent flow cytometry study showed kappa-restricted plasma cells without associated clonal lymphoid populations. A myeloma fluorescence in situ hybridization panel revealed an isolated translocation (11;14). Serum and urine protein electrophoresis did not detect a monoclonal protein. Ig quantification showed IgM <16.9 mg/dL (normal 40-230 mg/dL), IgG 769.0 mg/dL (normal 700-1,600 mg/dL), and IgA 94.3 mg/dL (normal 70-400 mg/dL). Serum free light chain assay revealed increased kappa light chains (1,030.5 mg/L, normal 3.3-19.4 mg/L) and an increased kappa/lambda light chain ratio (112.01, normal 0.26-1.65). Additional laboratory investigations were significant for anemia (hemoglobin 11.7 g/dL), preserved renal function (creatinine 0.7 mg/dL), and increased β2 microglobulin (3 mg/L, normal 1.09-2.53 mg/L). Alanine aminotransferase, aspartate aminotransferase, alkaline phosphatase, lactate dehydrogenase, and albumin were all within normal limits. The patient was therefore diagnosed with kappa light chain MM with extramedullary involvement of the liver. The International Staging System (ISS) prognostic stage was I (usually indicative of low-risk/good prognosis disease).
Systemic chemotherapy was initiated with cyclophosphamide, bortezomib, and dexamethasone (CyBorD regimen).4 The patient completed 6 cycles, initially showing a partial response followed by early disease progression evidenced by new lesions on PET scan and marked rise in the kappa light chain level.
His chemotherapy regimen was changed to lenalidomide and dexamethasone5 but was discontinued after 3 cycles because of disease progression. Although the patient had ISS stage I disease, his disease demonstrated an aggressive clinical course, resistant to multiple subsequent lines of therapy. The patient's care was later transferred to an outside facility for ongoing chemotherapy with a plan for autologous stem cell transplant. Despite several cycles of chemotherapy, his disease continued to progress, and he ultimately died after a complicated hospital course for neutropenic fever.
DISCUSSION
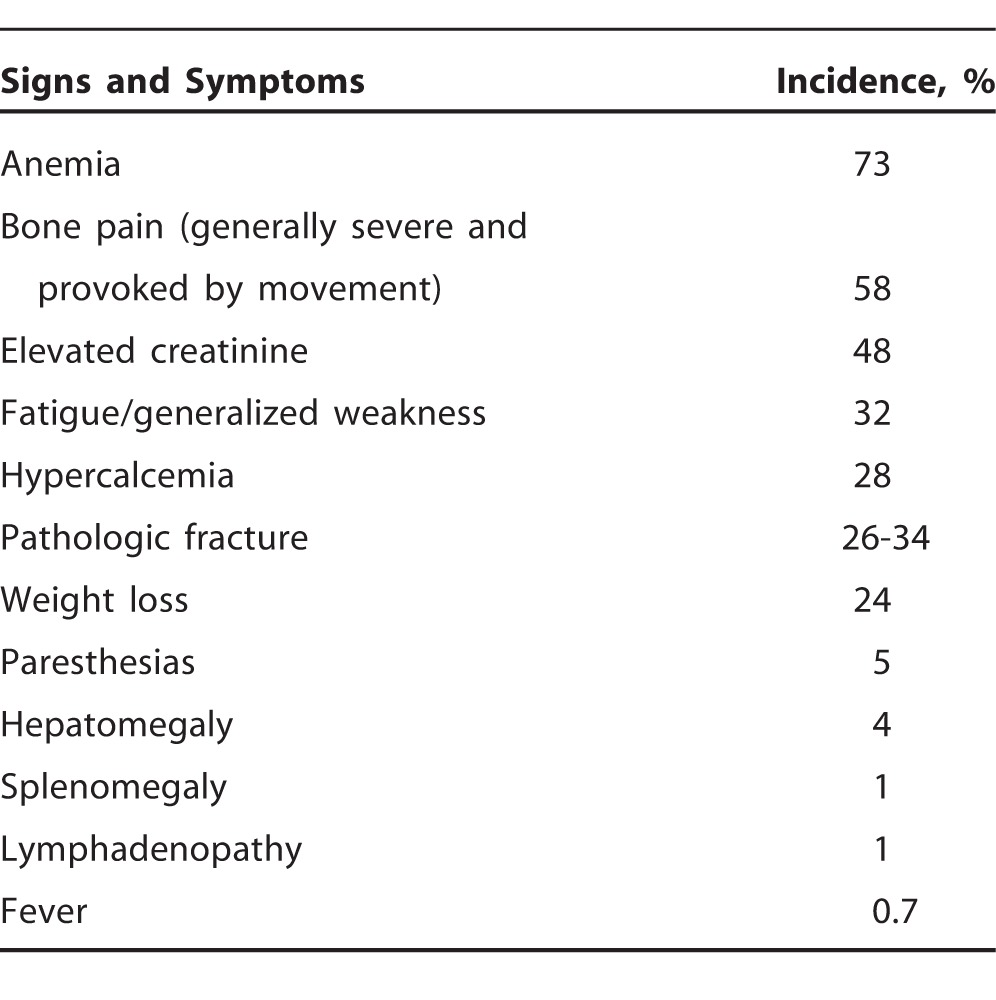
MM is the most common primary bone malignancy in adults.6 Uncontrolled proliferation of clonal plasma cells and overproduction of monoclonal protein lead to organ damage and result in the common clinical manifestations of MM, including hypercalcemia, renal insufficiency, anemia, and bone lesions.1,2 The incidence of presenting clinical manifestations in patients with MM varies (Table 1). Anemia and bone pain are the 2 most common presenting signs and symptoms.7,8 Bony lesions commonly involve long bones, ribs, skull, and pelvis, possibly leading to further complications including pathologic fractures. As in our patient, hypercalcemia is a common presenting manifestation of MM, and in asymptomatic patients, it may be one of the few subtle abnormalities pointing toward the diagnosis.
Table 1.
Extramedullary spread of MM can be detected at diagnosis but is much more common in the later stages of the disease.9 Our patient illustrates an atypical initial presentation of MM as an incidentally discovered solitary hepatic mass. The most common mechanism of extramedullary spread is through local growth from focal bone sites into surrounding soft tissue.2 Alternatively, distant metastasis of malignant plasma cells can spread hematogenously, most commonly to organs of the reticuloendothelial system, but in the terminal stages of the disease any organ may be involved. A study of 64 patients with MM from 1956-1970 reported hepatomegaly in 58%, splenomegaly in 25%, ascites in 14%, and abnormal liver function tests in ~90% of the cases at presentation.10 Hepatic infiltration by plasma cells was present during autopsy in up to 40% of these patients, most commonly as diffuse sinusoidal infiltrates followed by discrete portal aggregates and masses. Tumors were found in 16% of the cases, but in contrast with our case, the lesions were multiple 1-2 cm nodules distributed throughout the hepatic parenchyma mimicking metastatic disease. These findings would be very unusual today, likely because of much earlier diagnosis and more effective therapies.
More recent studies report much lower frequencies of liver involvement; Perez-Soler et al11 reviewed 128 patients with MM in a series published in 1985. Histologic study of the liver was available for 21 patients. A diffuse infiltrative pattern of plasma cells was observed in 10 patients, but no cases of nodular liver infiltration were seen. Talamo et al3 reported 9 cases of nodular liver infiltration in a cohort of 2,584 patients with MM (0.35%) in 2006. These findings underscore the clinical rarity of involvement of the liver in MM patients in present times and the overall rarity of isolated plasmacytomas of the liver that have only been described as case reports.
We performed a literature review and summarized all available studies (n=13) describing MM with nodular hepatic involvement (Tables 2 and 3).12-24 In the majority of cases (n=8), liver involvement manifested with abdominal pain and/or other gastrointestinal symptoms. Six cases demonstrated abnormalities in liver function tests, with alkaline phosphatase most commonly elevated. Liver function tests were not reported for 3 cases. Distribution of the monoclonal protein was varied: IgG kappa (n=4), IgA kappa (n=3), IgA lambda (n=1), lambda light chain (n=3), and kappa light chain (n=2). Ultrasound and CT were the usual imaging modalities utilized for detection of the hepatic lesions. Only 2 patients had solitary nodular liver lesions, while the remaining patients presented with multiple lesions. Hepatic plasmacytomas were frequently (n=6) described as hypoechoic on ultrasound or hypoattenuating on CT. In all but 1 case, diagnosis of hepatic involvement was based on histologic analysis of tissue samples. Identification of bony lytic lesions was typically achieved using skeletal surveys. PET scan was used in 1 case, similar to our case. In 4 cases, nodular hepatic plasmacytomas presented during a period of relapse after initial diagnosis of MM. In all other cases, hepatic involvement was discovered at the time of MM diagnosis. Finally, the documented clinical course varied among cases and included complete remission (n=2), initial clinical improvement without further follow-up reported (n=3), aggressive course with relapsing disease (n=3), and death (n=3). In 2 cases, the clinical course and response to treatment were not reported.
Table 2.
Summary of Case Reports Describing Multiple Myeloma with Nodular Hepatic Involvement: Patient Characteristics
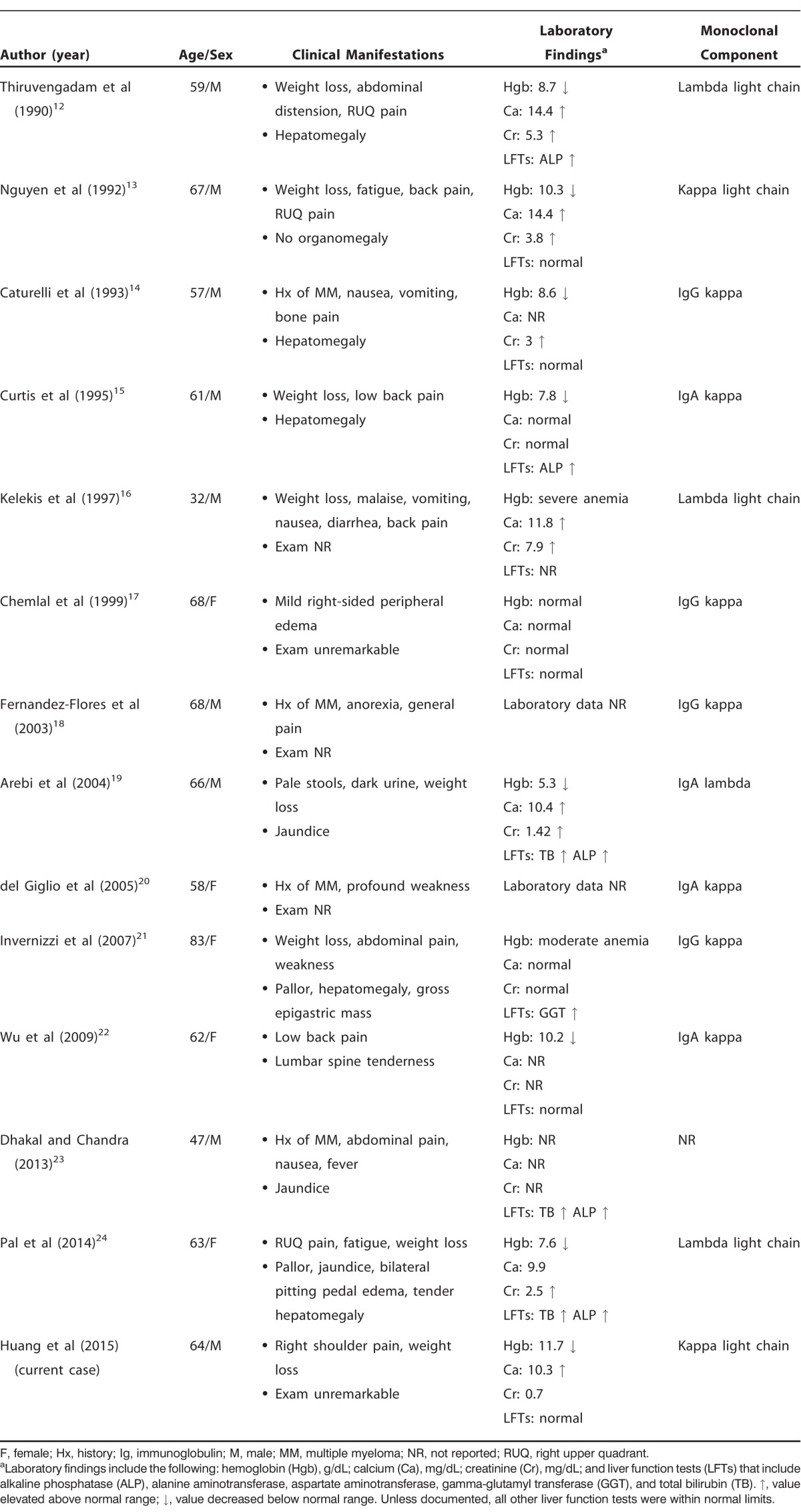
Table 3.
Summary of Case Reports Describing Multiple Myeloma with Nodular Hepatic Involvement: Imaging, Pathology, and Treatment
Conventional skeletal radiography has traditionally been used to identify bone disease in patients with MM.25 Skeletal radiography is still widely used today in clinical practice and is generally the recommended first-line imaging modality in the diagnosis and staging of MM.26 However, with the introduction and widespread availability of newer imaging methods, including CT, magnetic resonance imaging (MRI), 18F-fluorodeoxyglucose PET with CT (PET-CT), the diagnostic criteria and staging of MM have been substantially updated. Compared to skeletal radiography, these newer imaging techniques provide increased sensitivity for the detection of bony lytic lesions.25 CT and MRI are generally performed to (1) further evaluate lytic lesions that are ambiguous or equivocal on plain radiography, (2) assess the nature and extent of soft tissue disease, (3) evaluate suspected spinal cord compression (MRI preferred), and (4) differentiate between malignant and benign vertebral compression fractures.27 PET-CT or MRI is routinely performed in patients with a suspected diagnosis of smoldering (asymptomatic) MM or solitary plasmacytoma,25,28,29 as these diagnostic modalities can detect abnormal infiltrative processes of the bone marrow in the absence of bone lesions. These diagnostic modalities are also important in the diagnosis of MM relapse because the lytic bone lesions frequently persist after successful therapy.
The presence of extramedullary disease is often associated with light chain–secreting MM and a higher number of immature or plasmablastic plasma cells,30 but most of the cases we reviewed (8/13) had paraproteins with complete Igs. Studies comparing baseline characteristics between patients with and without extramedullary disease at diagnosis showed that nonsecretory disease and lambda chain expression were more common in extramedullary MM.9,31 Nonsecretory multiple myeloma (NSMM) is a rare subtype of MM defined by the absence of monoclonal Igs on serum and urine electrophoresis.32 However, in the majority of patients with presumed NSMM, monoclonal free light chains can now be detected with newer sensitive automated serum assays,33 similar to our case, indicating that these presumed NSMM cases likely represent light chain–only myelomas in which the abnormal protein was not detected in 24-hour urine protein electrophoresis, possibly because of technical reasons. Nevertheless, the secretion of an incomplete Ig may imply additional abnormalities and possible mutations in the genes involved in their synthesis compared to regular MM. These additional abnormalities and genetic mutations are likely related to the reported higher incidence of extramedullary disease at diagnosis in light chain MM. A shift in secretion from intact Igs to free light chains (ie, light chain escape) has been associated with progression of MM in the form of extramedullary metastasis.30 Furthermore, patients with incomplete Ig synthesis or light chain–secreting MM may have a higher incidence of renal failure, anemia, bone disease, amyloidosis, and extramedullary plasmacytomas.34 This in turn may translate into a more aggressive disease course with worse outcomes, as in our case. In our patient, confirmatory diagnosis of MM after the discovery of an extramedullary plasmacytoma was crucial and held significant prognostic implications. Most extramedullary plasmacytomas that are not associated with bone marrow disease represent marginal zone lymphomas with extensive plasmacytic differentiation and usually pursue an indolent clinical course limited to solitary lesions.35 Conversely, extramedullary plasmacytomas in the context of MM are indicative of advanced disease and portend a poor prognosis.9 In our case, PET imaging proved to be a highly sensitive technique that identified occult bony lesions and ultimately helped confirm the diagnosis of MM by identifying a bone lesion amenable to biopsy.
The presence of extramedullary involvement in MM at any time during the course of disease has been associated with shorter progression-free survival and shorter overall survival.9 Moreover, a series of 24 patients with MM and extramedullary gastrointestinal involvement showed a strong association with adverse biological features and short remission periods.3
Stem cell transplantation (SCT) after primary induction chemotherapy remains an important component in the treatment of newly diagnosed MM.36 Forms of SCT include autologous, tandem, or allogeneic SCT. The standard approach involves early or delayed autologous SCT. However, alternative approaches such as allogeneic SCT may be considered in highly select patients or clinical trials.36 Early treatment with high-dose therapy or autologous SCT has been shown to provide similar overall survival and progression-free survival irrespective of the presence or absence of extramedullary disease.9,31 Newer agents, including immunomodulatory drugs (eg, thalidomide, lenalidomide, and pomalidomide) and proteasome inhibitors (eg, bortezomib and carfilzomib), are now used earlier in the treatment of MM, as well as in relapsed disease. However, data are scarce on the efficacy of these drugs in newly diagnosed patients with extramedullary MM. Our patient demonstrated initial treatment response before showing early disease progression despite treatment with an ordinarily highly effective multidrug regimen including cyclophosphamide, bortezomib, and dexamethasone. Our case illustrates the clinical aggressiveness of a light chain MM with extramedullary involvement that progressed despite several lines of multiagent chemotherapy. The patient's progressive disease and rapid decline in functional status prevented SCT and ultimately led to his death.
CONCLUSION
We describe the case of a single hepatic nodule discovered incidentally during routine preoperative ultrasound that unexpectedly proved to represent MM with extramedullary involvement. In our review of the literature, we found only 2 other cases of MM with a solitary liver plasmacytoma at presentation. Although rare, an extramedullary plasmacytoma should be included in the differential diagnosis of radiologically indistinct hepatic lesions, especially in the context of hypercalcemia and concurrent bone lesions. The presence of extramedullary plasmacytoma is indicative of advanced disease and poor prognosis in the setting of MM. Our case also demonstrates that PET scan is a highly sensitive modality for identifying plasmacytomas and occult bony lesions in cases with uncommon presentations.
ACKNOWLEDGMENTS
The authors have no financial or proprietary interest in the subject matter of this article.
This article meets the Accreditation Council for Graduate Medical Education and the American Board of Medical Specialties Maintenance of Certification competencies for Patient Care and Medical Knowledge.
REFERENCES
- 1.Raab MS, Podar K, Breitkreutz I, Richardson PG, Anderson KC. Multiple myeloma. Lancet. 2009 Jul 25;374(9686):324–339. doi: 10.1016/S0140-6736(09)60221-X. [DOI] [PubMed] [Google Scholar]
- 2.Bladé J, Fernández de Larrea C, Rosiñol L, Cibeira MT, Jiménez R, Powles R. Soft-tissue plasmacytomas in multiple myeloma: incidence, mechanisms of extramedullary spread, and treatment approach. J Clin Oncol. 2011 Oct 1;29(28):3805–3812. doi: 10.1200/JCO.2011.34.9290. [DOI] [PubMed] [Google Scholar]
- 3.Talamo G, Cavallo F, Zangari M, et al. Clinical and biological features of multiple myeloma involving the gastrointestinal system. Haematologica. 2006 Jul;91(7):964–967. [PubMed] [Google Scholar]
- 4.Khan ML, Reeder CB, Kumar SK, et al. A comparison of lenalidomide/dexamethasone versus cyclophosphamide/lenalidomide/dexamethasone versus cyclophosphamide/bortezomib/dexamethasone in newly diagnosed multiple myeloma. Br J Haematol. 2012 Feb;156(3):326–333. doi: 10.1111/j.1365-2141.2011.08949.x. [DOI] [PubMed] [Google Scholar]
- 5.Weber DM, Chen C, Niesvizky R, et al. Lenalidomide plus dexamethasone for relapsed multiple myeloma in North America. N Engl J Med. 2007 Nov 22;357(21):2133–2142. doi: 10.1056/NEJMoa070596. [DOI] [PubMed] [Google Scholar]
- 6.Resnick D. Tumors and tumor-like lesions of bone: imaging and pathology of specific lesions. In: Resnick D, editor. Bone and Joint Imaging. 2nd ed. Philadelphia, PA: W.B. Saunders;; 1996. pp. 991–1063. In. ed. [Google Scholar]
- 7.Nau KC, Lewis WD. Multiple myeloma: diagnosis and treatment. Am Fam Physician. 2008 Oct 1;78(7):853–859. [PubMed] [Google Scholar]
- 8.Kyle RA, Gertz MA, Witzig TE, et al. Review of 1027 patients with newly diagnosed multiple myeloma. Mayo Clin Proc. 2003 Jan;78(1):21–33. doi: 10.4065/78.1.21. [DOI] [PubMed] [Google Scholar]
- 9.Varettoni M, Corso A, Pica G, Mangiacavalli S, Pascutto C, Lazzarino M. Incidence, presenting features and outcome of extramedullary disease in multiple myeloma: a longitudinal study on 1003 consecutive patients. Ann Oncol. 2010 Feb;21(2):325–330. doi: 10.1093/annonc/mdp329. [DOI] [PubMed] [Google Scholar]
- 10.Thomas FB, Clausen KP, Greenberger NJ. Liver disease in multiple myeloma. Arch Intern Med. 1973 Aug;132(2):195–202. [PubMed] [Google Scholar]
- 11.Perez-Soler R, Esteban R, Allende E. Tornos Salomo C, Julia A, Guardia J. Liver involvement in multiple myeloma. Am J Hematol. 1985 Sep;20(1):25–29. doi: 10.1002/ajh.2830200105. [DOI] [PubMed] [Google Scholar]
- 12.Thiruvengadam R, Penetrante RB, Goolsby HJ, Silk YN, Bernstein ZP. Multiple myeloma presenting as space-occupying lesions of the liver. Cancer. 1990 Jun 15;65(12):2784–2786. doi: 10.1002/1097-0142(19900615)65:12<2784::aid-cncr2820651229>3.0.co;2-3. [DOI] [PubMed] [Google Scholar]
- 13.Nguyen BD, Dash N, Lupetin AR. MR imaging of hepatic plasmacytoma: a case report. Clin Imaging. 1992 Apr-Jun;16(2):98–101. doi: 10.1016/0899-7071(92)90120-x. [DOI] [PubMed] [Google Scholar]
- 14.Caturelli E, Squillante MM, Castelvetere M, Falcone A, Musto P. Myelomatous nodular lesions of the liver: diagnosis by ultrasound-guided fine-needle biopsy. J Clin Ultrasound. 1993 Feb;21(2):133–137. doi: 10.1002/jcu.1870210211. [DOI] [PubMed] [Google Scholar]
- 15.Curtis JM, Pellegrini V, Tappin JA. Case report: multiple myeloma—a rare presentation. Clin Radiol. 1995 Jan;50(1):63–64. doi: 10.1016/s0009-9260(05)82970-8. [DOI] [PubMed] [Google Scholar]
- 16.Kelekis NL, Semelka RC, Warshauer DM, Sallah S. Nodular liver involvement in light chain multiple myeloma: appearance on US and MRI. Clin Imaging. 1997 May-Jun;21(3):207–209. doi: 10.1016/s0899-7071(96)00022-8. [DOI] [PubMed] [Google Scholar]
- 17.Chemlal K, Couvelard A, Grange MJ, et al. Nodular lesions of the liver revealing multiple myeloma. Leuk Lymphoma. 1999 Apr;33(3-4):389–392. doi: 10.3109/10428199909058441. [DOI] [PubMed] [Google Scholar]
- 18.Fernandez-Flores A, Fortes J, Smucler A, Orduña M, Pol A. Involvement of the liver by multiple myeloma as nodular lesions: a case diagnosed by fine-needle aspiration and immunocytochemistry. Diagn Cytopathol. 2003 Nov;29(5):280–282. doi: 10.1002/dc.10367. [DOI] [PubMed] [Google Scholar]
- 19.Arebi N, Patel B, Aqel NM, Pitcher MC. IgA multiple myeloma presenting as non-obstructive jaundice. Postgrad Med J. 2004 Aug;80(946):489–490. doi: 10.1136/pgmj.2002.002147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.del Giglio A, Weinschenker P, Manhani AR, Carbonell AL, Mitteldorf CA. Hepatic plasmacytosis as a manifestation of relapse in multiple myeloma treated with thalidomide. South Med J. 2005 Feb;98(2):238–240. doi: 10.1097/01.SMJ.0000152542.36111.6F. [DOI] [PubMed] [Google Scholar]
- 21.Invernizzi R, Maffè GC, Travaglino E, Pagani E, Pieresca C. Nodular lesions of the liver in multiple myeloma. Haematologica. 2007 Jul;92(7):e81. doi: 10.3324/haematol.11908. [DOI] [PubMed] [Google Scholar]
- 22.Wu XN, Zhao XY, Jia JD. Nodular liver lesions involving multiple myeloma: a case report and literature review. World J Gastroenterol. 2009 Feb 28;15(8):1014–1017. doi: 10.3748/wjg.15.1014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Dhakal A, Chandra A. A multiple myeloma patient presenting with multiple hepatic masses. J Med Cases. 2013 Dec;4(10):673–675. [Google Scholar]
- 24.Pal S, Chattopadhyay B, Chatterjee A, Bhattacharya B. Lambda light chain myeloma presenting as nodular hepatic lesion: a clinical rarity. J Cancer Res Ther. 2014 Jan-Mar;10(1):191–193. doi: 10.4103/0973-1482.131409. [DOI] [PubMed] [Google Scholar]
- 25.Rajkumar SV, Dimopoulos MA, Palumbo A, et al. International Myeloma Working Group updated criteria for the diagnosis of multiple myeloma. Lancet Oncol. 2014 Nov;15(12):e538–e548. doi: 10.1016/S1470-2045(14)70442-5. [DOI] [PubMed] [Google Scholar]
- 26.Zamagni E, Cavo M. The role of imaging techniques in the management of multiple myeloma. Br J Haematol. 2012 Dec;159(5):499–513. doi: 10.1111/bjh.12007. [DOI] [PubMed] [Google Scholar]
- 27.Dimopoulos M, Kyle R, Fermand JP, et al. Consensus recommendations for standard investigative workup: report of the International Myeloma Workshop Consensus Panel 3. Blood. 2011 May 5;117(18):4701–4705. doi: 10.1182/blood-2010-10-299529. [DOI] [PubMed] [Google Scholar]
- 28.Dimopoulos M, Terpos E, Comenzo RL, et al. International myeloma working group consensus statement and guidelines regarding the current role of imaging techniques in the diagnosis and monitoring of multiple myeloma. Leukemia. 2009 Sep;23(9):1545–1556. doi: 10.1038/leu.2009.89. [DOI] [PubMed] [Google Scholar]
- 29.Kato T, Tsukamoto E, Nishioka T, et al. Early detection of bone marrow involvement in extramedullary plasmacytoma by whole-body F-18 FDG positron emission tomography. Clin Nucl Med. 2000 Nov;25(11):870–873. doi: 10.1097/00003072-200011000-00003. [DOI] [PubMed] [Google Scholar]
- 30.Dawson MA, Patil S, Spencer A. Extramedullary relapse of multiple myeloma associated with a shift in secretion from intact immunoglobulin to light chains. Haematologica. 2007 Jan;92(1):143–144. doi: 10.3324/haematol.10297. [DOI] [PubMed] [Google Scholar]
- 31.Wu P, Davies FE, Boyd K, et al. The impact of extramedullary disease at presentation on the outcome of myeloma. Leuk Lymphoma. 2009 Feb;50(2):230–235. doi: 10.1080/10428190802657751. [DOI] [PubMed] [Google Scholar]
- 32.Lopes da Silva R, Monteiro A, Veiga J. Non-secretory multiple myeloma relapsing as extramedullary liver plasmacytomas. J Gastrointesin Liver Dis. 2011 Mar;20(1):81–83. [PubMed] [Google Scholar]
- 33.Drayson M, Tang LX, Drew R, Mead GP, Carr-Smith H, Bradwell AR. Serum free light-chain measurements for identifying and monitoring patients with nonsecretory multiple myeloma. Blood. 2001 May 1;97(9):2900–2902. doi: 10.1182/blood.v97.9.2900. [DOI] [PubMed] [Google Scholar]
- 34.Zhan JJ, Sun WJ, Huang ZX, et al. Light chain multiple myeloma, clinic features, responses to therapy and survival in a long-term study. World J Surg Oncol. 2014 Jul 28;12:234. doi: 10.1186/1477-7819-12-234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Fischbach W, Goebeler-Kolve ME, Dragosics B, Greiner A, Stolte M. Long term outcome of patients with gastric marginal zone B cell lymphoma of mucosa associated lymphoid tissue (MALT) following exclusive Helicobacter pylori eradication therapy: experience from a large prospective series. Gut. 2004 Jan;53(1):34–37. doi: 10.1136/gut.53.1.34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kyle RA, Rajkumar SV. Multiple myeloma. N Engl J Med. 2004 Oct 28;351(18):1860–1873. doi: 10.1056/NEJMra041875. Erratum in: N Engl J Med. 2005 Mar 17;352(11):1163. [DOI] [PubMed] [Google Scholar]