Abstract
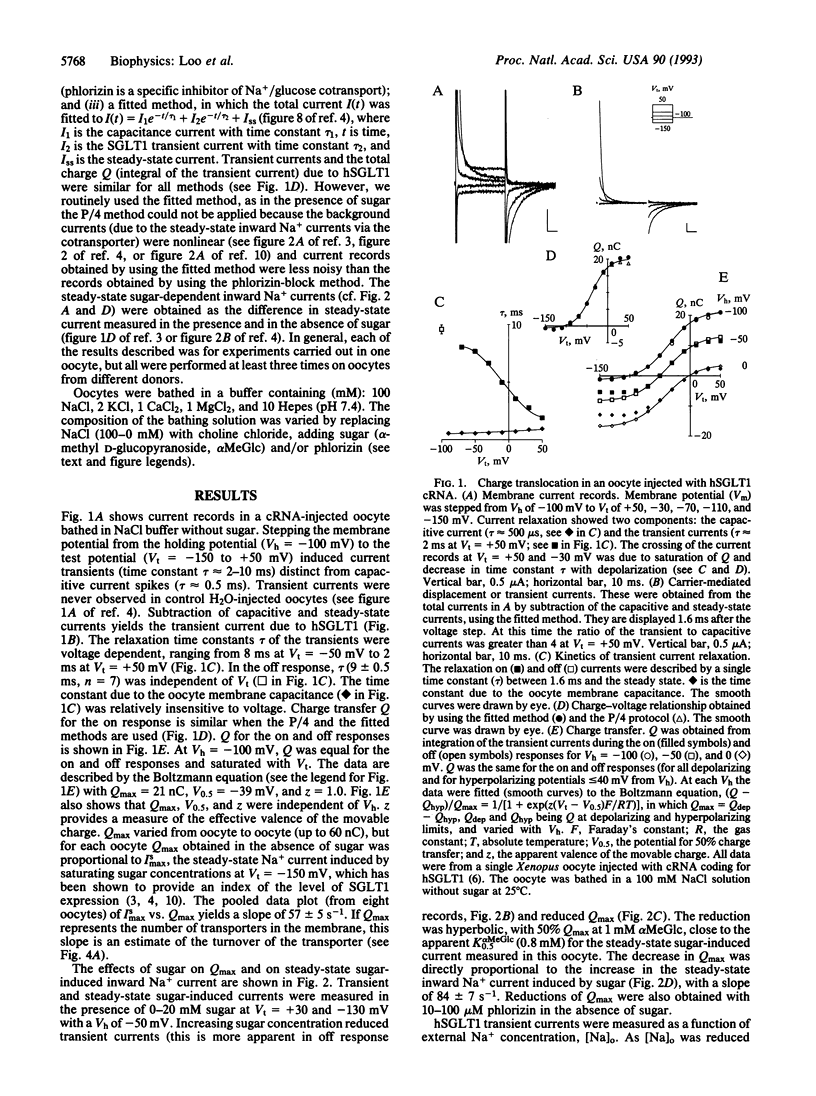
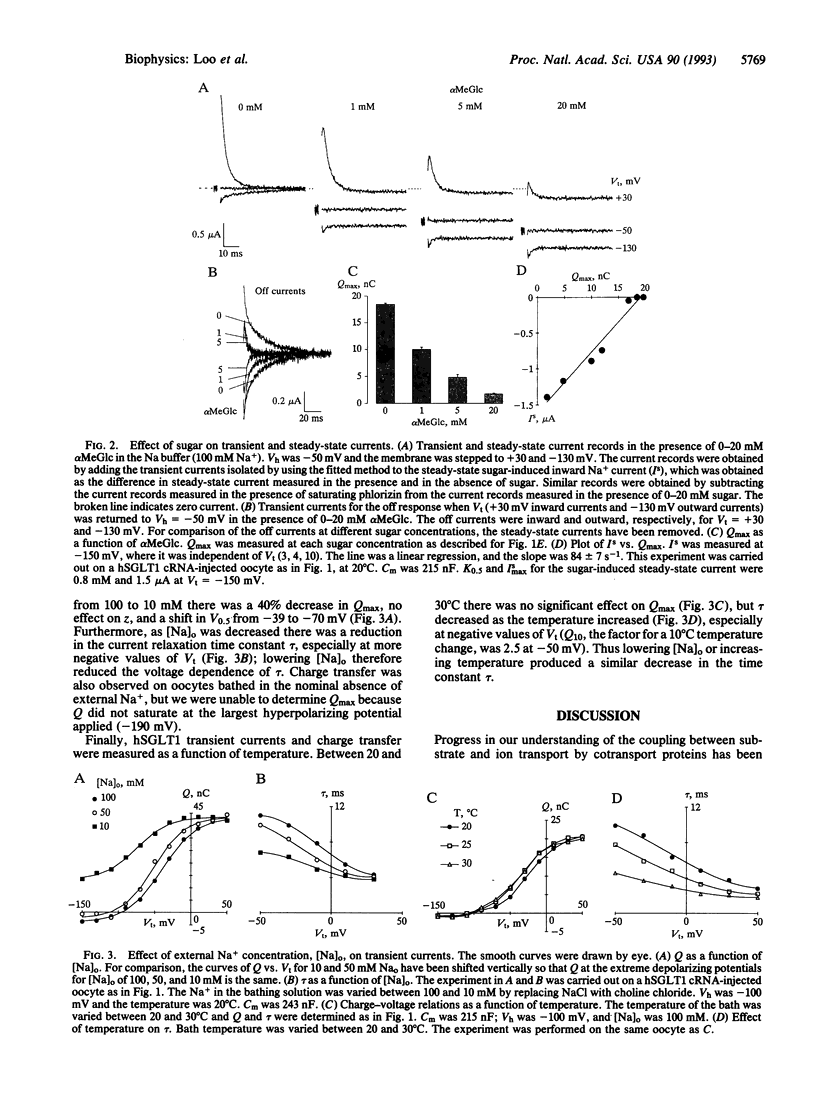
An important class of integral membrane proteins, cotransporters, couple solute transport to electrochemical potential gradients; e.g., the Na+/glucose cotransporter uses the Na+ electrochemical potential gradient to accumulate sugar in cells. So far, kinetic analysis of cotransporters has mostly been limited to steady-state parameters. In this study, we have examined pre-steady-state kinetics of Na+/glucose cotransport. The cloned human transporter (hSGLT1) was expressed in Xenopus oocytes, and voltage-clamp techniques were used to monitor current transients after step changes in membrane potential. Transients exhibited a voltage-dependent time constant (tau) ranging between 2 and 10 ms. The charge movement Q was fitted to a Boltzmann relation with maximal charge Qmax of approximately 20 nC, apparent valence z of 1, and potential V0.5 of -39 mV for 50% Qmax. Lowering external Na+ from 100 to 10 mM reduced Qmax 40%, shifted V0.5 from -39 to -70 mV, had no effect on z, and reduced the voltage dependence of tau. Qmax was independent of, but tau was dependent on, temperature (a 10 degrees C increase increased tau by a factor of approximately 2.5 at -50 mV). Addition of sugar or phlorizin reduced Qmax. Analyses of hSGLT1 pre-steady-state kinetics indicate that transfer upon a step of membrane potential in the absence of sugar is due to two steps in the reaction cycle: Na+ binding/dissociation (30%) and reorientation of the protein in the membrane field (70%). The rate-limiting step appears to be Na+ binding/dissociation. Qmax provides a measure of transporter density (approximately 10(4)/microns 2). Charge transfer measurements give insight into the partial reactions of the Na+/glucose cotransporter, and, combined with genetic engineering of the protein, provide a powerful tool for studying transport mechanisms.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Armstrong C. M., Bezanilla F. Currents related to movement of the gating particles of the sodium channels. Nature. 1973 Apr 13;242(5398):459–461. doi: 10.1038/242459a0. [DOI] [PubMed] [Google Scholar]
- Bezanilla F., Armstrong C. M. Inactivation of the sodium channel. I. Sodium current experiments. J Gen Physiol. 1977 Nov;70(5):549–566. doi: 10.1085/jgp.70.5.549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bezanilla F., Perozo E., Papazian D. M., Stefani E. Molecular basis of gating charge immobilization in Shaker potassium channels. Science. 1991 Nov 1;254(5032):679–683. doi: 10.1126/science.1948047. [DOI] [PubMed] [Google Scholar]
- Birnir B., Loo D. D., Wright E. M. Voltage-clamp studies of the Na+/glucose cotransporter cloned from rabbit small intestine. Pflugers Arch. 1991 Mar;418(1-2):79–85. doi: 10.1007/BF00370455. [DOI] [PubMed] [Google Scholar]
- Griffith J. K., Baker M. E., Rouch D. A., Page M. G., Skurray R. A., Paulsen I. T., Chater K. F., Baldwin S. A., Henderson P. J. Membrane transport proteins: implications of sequence comparisons. Curr Opin Cell Biol. 1992 Aug;4(4):684–695. doi: 10.1016/0955-0674(92)90090-y. [DOI] [PubMed] [Google Scholar]
- Hediger M. A., Turk E., Wright E. M. Homology of the human intestinal Na+/glucose and Escherichia coli Na+/proline cotransporters. Proc Natl Acad Sci U S A. 1989 Aug;86(15):5748–5752. doi: 10.1073/pnas.86.15.5748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwon H. M., Yamauchi A., Uchida S., Preston A. S., Garcia-Perez A., Burg M. B., Handler J. S. Cloning of the cDNa for a Na+/myo-inositol cotransporter, a hypertonicity stress protein. J Biol Chem. 1992 Mar 25;267(9):6297–6301. [PubMed] [Google Scholar]
- Mager S., Naeve J., Quick M., Labarca C., Davidson N., Lester H. A. Steady states, charge movements, and rates for a cloned GABA transporter expressed in Xenopus oocytes. Neuron. 1993 Feb;10(2):177–188. doi: 10.1016/0896-6273(93)90309-f. [DOI] [PubMed] [Google Scholar]
- Nakao M., Gadsby D. C. Voltage dependence of Na translocation by the Na/K pump. Nature. 1986 Oct 16;323(6089):628–630. doi: 10.1038/323628a0. [DOI] [PubMed] [Google Scholar]
- Parent L., Supplisson S., Loo D. D., Wright E. M. Electrogenic properties of the cloned Na+/glucose cotransporter: I. Voltage-clamp studies. J Membr Biol. 1992 Jan;125(1):49–62. doi: 10.1007/BF00235797. [DOI] [PubMed] [Google Scholar]
- Parent L., Supplisson S., Loo D. D., Wright E. M. Electrogenic properties of the cloned Na+/glucose cotransporter: II. A transport model under nonrapid equilibrium conditions. J Membr Biol. 1992 Jan;125(1):63–79. doi: 10.1007/BF00235798. [DOI] [PubMed] [Google Scholar]
- Taglialatela M., Toro L., Stefani E. Novel voltage clamp to record small, fast currents from ion channels expressed in Xenopus oocytes. Biophys J. 1992 Jan;61(1):78–82. doi: 10.1016/S0006-3495(92)81817-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Umbach J. A., Coady M. J., Wright E. M. Intestinal Na+/glucose cotransporter expressed in Xenopus oocytes is electrogenic. Biophys J. 1990 Jun;57(6):1217–1224. doi: 10.1016/S0006-3495(90)82640-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaughan P. C., McLarnon J. G., Loo D. D. Voltage dependence of chloride current through Xenopus muscle membrane in alkaline solutions. Can J Physiol Pharmacol. 1980 Sep;58(9):999–1010. doi: 10.1139/y80-153. [DOI] [PubMed] [Google Scholar]
- Wright E. M., Hager K. M., Turk E. Sodium cotransport proteins. Curr Opin Cell Biol. 1992 Aug;4(4):696–702. doi: 10.1016/0955-0674(92)90091-p. [DOI] [PubMed] [Google Scholar]