Abstract
Objective: To investigate the effect of isoliquiritigenin on the activity of DNA topoisomerase (TOP I) and its inhibitory effect on the growth of U87 glioma cells. Methods: This study investigated the inhibitory effect of isoliquiritigenin on the growth of U87 glioma cells and its cytotoxicity by MTT method and determined the effect of isoliquiritigenin on TOP I activity by agarose gel electrophoresis. On this basis, we studied the interaction between isoliquiritigenin and TOP I and DNA. Finally, we further discussed the effect of isoliquiritigenin on the activity of Caspase 3, the apoptosis protein of U87 glioma cells. Results: Isoliquiritigenin could inhibit the growth of U87 glioma cells (half inhibitory concentration IC50: 0.221 mM) and is of low cytotoxicity to normal cells. Agarose gel electrophoresis showed that isoliquiritigenin had significant inhibitory effect on TOP I activity. Molecular simulation results indicated that isoliquiritigenin took priority of binding to the active center of TOP I, and formed hydrogen bonds with the catalytic site Try723. Finally, Caspase 3 activity detection results suggested that isoliquiritigenin could significantly increase the activity of Caspase 3 (P < 0.05). Conclusion: Isoliquiritigenin had a reversible inhibitory effect on TOP I activity, reduced the rate of single strand DNA unwinding in tumor cells, and thus played an important role in inducing the apoptosis of U87 glioma cells.
Keywords: Isoliquiritigenin, U87 glioma cell, DNA topoisomerase, inhibitory effect, apoptosis
Introduction
DNA topoisomerases refers to a group of important enzymes which adjust DNA’s topological structure by cutting, transferring and re-connecting with DNA chains. They are crucial in adjusting dynamic changes of the spatial structure and controlling the physiological function of nucleic acids. DNA topoisomerases can be classified into two groups, namely, Topoisomerase I (TOP I) and Topoisomerase II (TOP II) [1]. TOP I can induce DNA single-stranded break, while TOP II can induce DNA double-stranded break in the main chain and another DNA double strand pass through the break point. In tumor cells, especially in patients with ovarian cancer, colon cancer and cervical cancer, etc., the amount of TOP I is much higher than that of normal cells. Therefore, TOP I is considered as an important target in the investigation of antitumor drugs. Camptothecin (CPT), as the only TOP I inhibitor used clinically, is of high toxicity, severe drug resistance and unstable in-vivo metabolism [2]. Hence, screening an effective TOP I inhibitor with low toxicity has become an important way to inhibit the growth and metastasis of tumor cells.
Isoliquiritigenin (the structural formula is presented in Figure 1), belongs to isoflavone compounds, a type of flavonoid, and can be found in various leguminous plants [3-7]. Pharmacological studies indicate that isoliquiritigenin is of strong antiviral, anti-inflammatory, anti-angiogenic, hypnosis and other bioactivities, among which its anti-tumor effect has become a hotspot in recent years. By focusing on the reduction of TOP I activity induced by isoliquiritigenin, this article investigated its inhibitory effect on the growth of U87 glioma cells and its low toxicity in normal cells. Moreover, through analyzing DNA binding mechanism to TOP I by molecular simulation, we eventually know the inhibitory mechanism of isoliquiritigenin and its mechanism of inducing the apoptosis of U87 glioma cells, which will provide an experimental basis for utilizing it as an effective anti-tumor drug with low toxicity.
Figure 1.
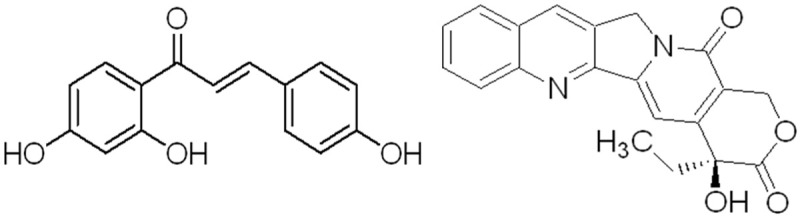
The molecular structural formula of isoliquiritigenin (left) and CPT (right).
Materials and methods
Investigational product
The isoliquiritigenin (20 mg, standard) is purchased from Aladdin Industrial Corporation (batch No. I111284). The standard stock solution is prepared with ethanol solution (100 μmol/L). Although high-concentration ethanol may have an influence on the structure and function of proteins, the concentration of ethanol used in this experiment is controlled within 1.0%, which will not affect TOP I and U87 glioma cells. CPT (20 mg, standard) is also purchased from Aladdin Industrial Corporation (batch No.: C111281).
Cells
Normal brain cells and U87 glioma cells are purchased from JRDUN Biotechnogy (Shanghai) Co. Ltd and are cultured in this laboratory.
Reagents
The Caspase-GlooR3 Assay Kit is purchased from Sigma-Aldrich (Shanghai) Trading Co., Ltd., RPMI1640 culture medium from Thermo-Fisher Biochemical Products (Beijing) Co., Ltd., Characterized fetal calf/calf serum from PAA in Australia, trypsin from Gibco in the US, dimethyl sulfoxide (DMSO) from Guangzhou Ruishu Biotechnology Co., Ltd., methyl thiazolyl tetrazolium (MTT, 5 × 103 mg/L, prepared with PBS) from Beijing Dingguo Changsheng Biotechnology Co., Ltd., negative supercoiled pBR322DNA (Toyobo in Japan) and ethidium bromide (EB, Biosharp Co.).
Instruments
Millipore Simplicity Water Purification System (Merck Millipore, France); ELIASA SpectraMax Plus384 (Molecular Devices); pHS-3C acidimeter (Shanghai REX Instrument Factory); EPS-300 electrophoresis apparatus and other devices used for electrophoresis (Tanon Science & Technology Co., Ltd., Shanghai); Bio-Rad680 enzyme-linked detector.
A test investigating the inhibitory effect of isoliquiritigenin on the growth of U87 glioma cells
[8] In this test, CPT was used as a positive control. Isoliquiritigenin of different concentrations were mixed with U87 glioma cells and normal brain cells. The mixture was incubated in 5% CO2 for 48 h under 37°C. Then, 50 μL staining solution of 0.5% MTT was added and incubated for another 4 h. After that, it was centrifuged at 2500 rpm for 30 min. The culture medium was then discarded and 150 µL DMSO solution was added. After we mixing them well by oscillator, the light absorption value was measured at 570 nm with ELIASA (OD value). Cell growth inhibition ratio was calculated as follows:
Inhibition rate (%) = (Blank OD value-Inhibitor OD value/Blank OD value) × 100%
Effect of isoliquiritigenin on TOP I activity (detected by agarose gel electrophoresis)
[9] The buffer system included 2 μL TOP I buffer solution, 2 μL 0.1% BSA, 0.5 U TOP I and 0.252 μg negative supercoiled pBR322DNA; positive control tube (0.3 μL CPT DMSO solution) and 0.3 μL isoliquiritigenin. Each tube was diluted with distilled water to 20 μL, mixed well and placed in a incubator under 37°C for reaction for 30 min. Then, 2.5 μL 5 × loading buffer of stop buffer precooled under 4°C was added to terminate the reaction. Afterwards, in a 1% agarose gel, it underwent electrophoresis for an hour with an electrophoresis apparatus at 120 V. At last, the resultant was stained with ethidium bromide (EB, Biosharp Co.) for 20-30 min, photographed under UV light at 260 nm and observed.
Effect of isoliquiritigenin on the activity of Caspase 3 (the apoptosis protein of U87 glioma cells)
[10] A Caspase-GlooR3 Assay Kit was used in this test. For detailed procedure, please refer to the product specification. The following part is a brief summary. Before analysis, reagents and a 96-well plate was placed under room temperature for balancing. Afterwards, U87 glioma cells were inoculated into each well at 15 × 104 and isoliquiritigenin at three different concentrations (0.5, 5, 10 μmol/L) was added and stood for 24 h, 48 h and 72 h. Then, the same volume of Caspase-Glo was added into the cell culture (1:1), which was then mildly mixed in an oscillator, shaken at 400 rpm for 30 s and incubated at room temperature for 60 min. During this test, casepase3 could catalyze Ac-DEVD-pNA to produce yellow pNA, which showed UV absorption at about 405 nm. Hence, the activity of caspase 3 could be detected by determining the absorbency at 405 nm. ELIASA was also used and the results were compared with those of the positive control CPT.
Molecular docking
It was conducted by using AUTODOCK4.0 molecular simulation software for the docking between isoliquiritigenin and TOP I and DNA. TOP I (PDB: 1T8I) and DNA (PDB: 453D) were obtained from RCSB Protein Data Bank (http://www.rcsb.org/pdb), while the three-dimensional molecular crystal of isoliquiritigenin was drawn by Sybyl × 1.1 (Tripos Inc., St. Louis, USA). Before docking, the ligand isoliquiritigenin and receptors TOP I and DNA were optimized in terms of structure and energy (dehydrated, hydrogenated and powered up). The parameter autogrid box was set and Lamarckian genetic algorithm (LGA) was selected for the calculation of autodock. Other parameters used the default setting. After docking for a hundred times, analysis for output results was completed by PyMoL software.
Statistical method
SPSS11.01 statistical software was used. Factor data was input into the statistical system after being quantified into enumeration data. χ2 test was applied for enumeration data, x̅±s was applied for measurement data, and t test was adopted for comparing these two groups. P < 0.05 means that the difference is of statistical significance.
Results
Inhibitory effect of isoliquiritigenin on the growth of U87 glioma cells
Figure 2 suggested that the half inhibitory concentration IC50 of the positive control CPT and isoliquiritigenin was 0.509 ± 0.36 μmol/L and 6.265 ± 0.89 μmol/L, respectively. It revealed that the inhibitory effect of isoliquiritigenin on the growth of U87 glioma cells was slightly lower than that of CPT, but it was also good.
Figure 2.
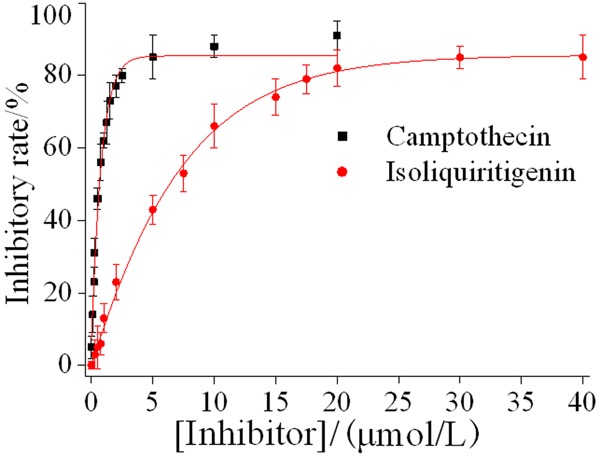
The inhibitory effect of isoliquiritigenin on the growth of U87 glioma cells.
Effect of isoliquiritigenin on TOP I activity
The effect could be found by analyzing the DNA electrophoretogram and its bands (Figure 3). The figure indicated that the supercoiled DNA (pBR322 DNA) turned into linear DNA under the effect of TOP I. Along with increased isoliquiritigenin concentration (1-8), the activity of TOP I was gradually inhibited. When the concentration of isoliquiritigenin reached up to 2.5 μmol/L (4), the inhibitory effect became obvious, which was manifested by ringlike gaps and decreased linear DNA. Compared with CPT (C, 2 μmol/L), the inhibitory effect of isoliquiritigenin on TOP I was good.
Figure 3.
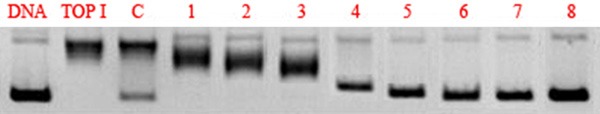
Electrophoretogram of the inhibitory effect of isoliquiritigenin on TOP I. C (isoliquiritigenin) 1-8 = 0.5, 1.0, 2.0, 2.5, 5.0, 7.5, 10, 15 and 20 μmol/L.
Caspase 3 activity assay
The results of cell activity assay suggested that after being treated with low-dose isoliquiritigenin (0.5 µmol/L) for 24, 48 and 72 hours, compared with the control group treated with DMSO, the activity of Caspase 3 was not increased significantly, while isoliquiritigenin (5, 10 µmol/L) and CPT (5 µmol/L) increased Caspase 3 activity evidently (P < 0.05). Please see Table 1.
Table 1.
Results of Caspase 3 activity assay (x(-) ± s, n = 6)
| Group | Dose (µmol/L) | 0 h | 24 h | 48 h | 72 h |
|---|---|---|---|---|---|
| DMSO blank | 11.43 ± 0.65 | 12.18 ± 0.96 | 14.23 ± 1.33 | 15.34 ± 1.43 | |
| Camptothecin | 5 | 11.98 ± 0.77 | 18.17 ± 1.71** | 35.65 ± 4.12** | 58.94 ± 5.13** |
| Isoliquiritigenin | 0.5 | 11.27 ± 1.43 | 12.44 ± 0.97 | 16.12 ± 1.84 | 16.54 ± 1.54 |
| Isoliquiritigenin | 5 | 11.82 ± 0.89 | 18.65 ± 1.47* | 26.28 ± 1.99** | 48.17 ± 4.73** |
| Isoliquiritigenin | 10 | 11.15 ± 0.56 | 20.12 ± 2.35** | 36.55 ± 3.15** | 67.76 ± 4.26** |
Compared with the DMSO blank group;
P < 0.05;
P < 0.01.
Molecular docking of isoliquiritigenin and TOP I
From results of one hundred running, the author selected the low-energy regions with most docking as the study object, which was also the best binding regions of isoliquiritigenin and TOP I. In Figures 4 and 5, it was shown that isoliquiritigenin was bound to the active central pocket of TOP I, which as around amino acid residues (Arg364, Asn352, Tyr723 and Thr718) of the catalytic center [11] and formed a hydrogen bond with Tyr723.
Figure 4.
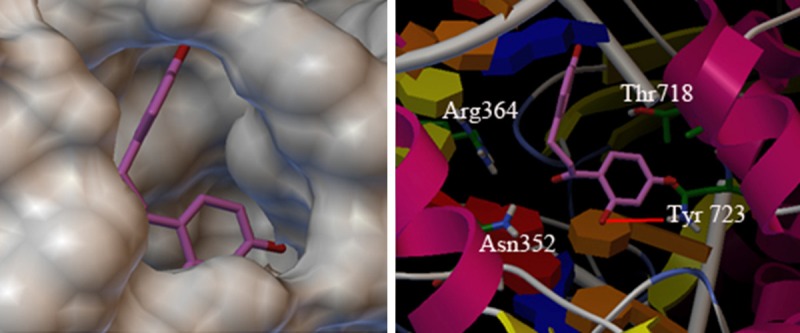
Molecular docking of isoliquiritigenin and TOP I.
Figure 5.
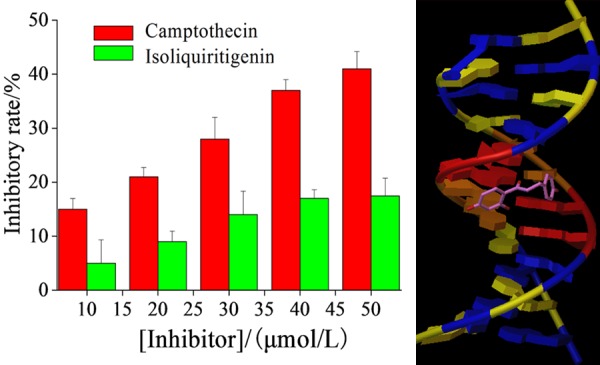
Toxicity toward normal brain cells.
Toxicity of isoliquiritigenin toward normal brain cells
Figure 5 (left) showed the toxicity of the positive control CPT and isoliquiritigenin toward normal brain cells after 24 hours. It revealed that the toxicity of isoliquiritigenin toward normal cells was much lower than that of CPT. Besides, it also indicated that isoliquiritigenin could be well inserted into the A-T base pair of DNA, which made the bond of normal cells’ DNA stronger so that it became less prone to canceration [12].
Discussion
CPT can induce apoptosis of tumor cells and TOP I is its major target spot. In this study, the inhibitory effect of isoliquiritigenin on the growth of U87 glioma cells was investigated by using these cells as target cells. Results indicated that isoliquiritigenin had this inhibitory effect which was dose dependent. In addition, it was found that the effect was weaker than that of CPT. The reason was that the inhibitory effect of CPT on TOP I was irreversible to some extent because it could bind covalently to enzymes, inactivating these enzymes irreversibly. This is harmful to normal human cells and may cause serious complications [2]. In contrast, the enzyme inhibition test and molecular docking showed that isoliquiritigenin could bind to TOP I reversibly and competitively and inhibit effectively at last. Studies conducted at home and abroad also indicated [13-18] that when inhibitors were of reversible competitive inhibitory effects for enzymes associated with diseases, the dose of the drug could be controlled effectively according to the activity of enzymes so as to reduce the activity of enzymes reversibly with high selectivity rather than inactivate them completely which might cause other adverse reactions. In addition, we also found that the toxicity of isoliquiritigenin was much lower than that of CPT and isoliquiritigenin could insert into normal DNAs without destroying them. In this way, its low toxicity greatly decreased the side effects it might cause in human body.
During apoptosis, the terminal factor of integrated enzyme reaction consisted of caspas-3 and caspase [19], in which caspase 3 was the key enzyme for apoptosis. Results of our study suggested that after being treated with isoliquiritigenin, caspase-3 activity increased constantly and it was time dependent. This means that isoliquiritigenin can activate caspase-3, regulate the integrated enzyme reaction efficaciously, and finally lead to the apoptosis of U87 glioma cells.
Disclosure of conflict of interest
None.
References
- 1.Giles GI, Sharma RP. Toporisomerase enzymes as therapeutic targets for cancer chemotherapy. Med Chem. 2005;1:283–94. doi: 10.2174/1573406054368738. [DOI] [PubMed] [Google Scholar]
- 2.Basili S, Moro S. Novel camptothecin derivatives as topoisomerase I inhibitors. Expert Opin Ther Pat. 2009;19:555–74. doi: 10.1517/13543770902773437. [DOI] [PubMed] [Google Scholar]
- 3.Chin YW, Jung HA, Liu Y, Su BN, Castoro JA, Keller WJ, Pereira MA, Kinghorn AD. Antioxidant constituents of the roots and stolons of licorice (Glycyrrhiza glabra) J Agric Food Chem. 2007;55:4691–7. doi: 10.1021/jf0703553. [DOI] [PubMed] [Google Scholar]
- 4.Gong H, Zhang BK, Yan M, Fang PF, Li HD, Hu CP, Yang Y, Cao P, Jiang P, Fan XR. A protective mechanism of licorice (Glycyrrhiza uralensis): Isoliquiritigenin stimulates detoxification system via Nrf2 activation. J Ethnopharmacol. 2015;162:134–9. doi: 10.1016/j.jep.2014.12.043. [DOI] [PubMed] [Google Scholar]
- 5.Zhao Z, Park SM, Guan L, Wu Y, Lee JR, Kim SC, Kim YW, Zhao R. Isoliquiritigenin attenuates oxidative hepatic damage induced by carbon tetrachloride with or without buthionine sulfoximine. Chem Biol Interact. 2015;225:13–20. doi: 10.1016/j.cbi.2014.10.030. [DOI] [PubMed] [Google Scholar]
- 6.Wong TY, Lin SM, Poon CH, Leung LK. The licorice flavonoid isoliquiritigenin reduces DNA-binding activity of AhR in MCF-7 cells. Chem Biol Interact. 2014;221:70–6. doi: 10.1016/j.cbi.2014.07.015. [DOI] [PubMed] [Google Scholar]
- 7.Zhao H, Zhang X, Chen X, Li Y, Ke Z, Tang T, Chai H, Guo AM, Chen H, Yang J. Isoliquiritigenin, a flavonoid from licorice, blocks M2 macrophage polarization in colitis-associated tumorigenesis through downregulating PGE2 and IL-6. Toxicol Appl Pharmacol. 2014;279:311–21. doi: 10.1016/j.taap.2014.07.001. [DOI] [PubMed] [Google Scholar]
- 8.Chen L, Lin YL, Peng GQ, Li F. Structural basis for multifunctional roles of mammalian aminopeptidase N. Proc Natl Acad Sci U S A. 2012;109:17966–71. doi: 10.1073/pnas.1210123109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kruczynski A, Barret JM, Van Hille B, Chansard N, Astruc J, Menon Y, Duchier C, Créancier L, Hill BT. Decreased nucleotide excision repair activity and alterations of topoisomerase IIalpha are associated with the in vivo resistance of a P388 leukemia subline to F11782, a novel catalytic inhibitor of topoisomerases I and II. Clin Cancer Res. 2004;10:3156–68. doi: 10.1158/1078-0432.ccr-1305-2. [DOI] [PubMed] [Google Scholar]
- 10.Yoshimori A, Oyama T, Takahashi S, Abe H, Kamiya T, Abe T, Tanuma S. Structure-activity relationships of the thujaplicins for inhibition of human tyrosinase. Bioorg Med Chem. 2014;22:6193–200. doi: 10.1016/j.bmc.2014.08.027. [DOI] [PubMed] [Google Scholar]
- 11.Qiu L, Chen QH, Zhang JX, Zhong X, Zhou JJ, Guo YJ, et al. Inhibitory effects of α-cyano-4-hydroxycinnamic acid on the activity of mushroom tyrosinase. Food Chem. 2009;112:609–613. [Google Scholar]
- 12.Zhang GW, Fu P, Wang L, Hu M. Molecular Spectroscopic Studies of Farrerol International with Calf Thymus DNA. J Agric Food Chem. 2011;59:8944–8952. doi: 10.1021/jf2019006. [DOI] [PubMed] [Google Scholar]
- 13.Skit M, Benedik E, Podlipnik C, Ulrih NP. Interactions of different polyphenols with bovine serum albumin using fluorescence quenching and molecular docking. Food Chem. 2012;135:2418–2424. doi: 10.1016/j.foodchem.2012.06.114. [DOI] [PubMed] [Google Scholar]
- 14.Park H, Hwang KY, Oh KH, Kim YH, Lee JY, Kim K. Discovery of novel α-glucosidase inhibitors based on the virtual screening with the homology-modeled protein structure. Bioorg Med Chem. 2008;16:284–92. doi: 10.1016/j.bmc.2007.09.036. [DOI] [PubMed] [Google Scholar]
- 15.Brindis F, Rodríguez R, Bye R, González-Andrade M, Mata R. (Z)-3-Butylidenephthalide from Ligusticum porteri, an α-Glucosidase Inhibitor. J Nat Prod. 2011;74:314–320. doi: 10.1021/np100447a. [DOI] [PubMed] [Google Scholar]
- 16.Ross PD, Subramanian S. Thermodynamics of protein association reactions: forces contributing to stability. Biochemistry. 1981;20:3096–3102. doi: 10.1021/bi00514a017. [DOI] [PubMed] [Google Scholar]
- 17.Park H, Hwang KY, Kim YH, Oh KH, Lee JY, Kim K. Discovery and biological evaluation of novel α-glucosidase inhibitors with in vivo antidiabetic effect. Bioorg Med Chem Lett. 2008;18:3711–3715. doi: 10.1016/j.bmcl.2008.05.056. [DOI] [PubMed] [Google Scholar]
- 18.Yan J, Zhang G, Hu Y, Ma Y. Effect of luteolin on xanthine oxidase: Inhibition kinetics and interaction mechanism merging with docking simulation. Food Chem. 2013;141:3766–3773. doi: 10.1016/j.foodchem.2013.06.092. [DOI] [PubMed] [Google Scholar]
- 19.Yao G, Yang L, Hu Y, Liang J, Liang J, Hou Y. None phenol-induced thymocyte apoptosis Involved caspase-3 activation and mitochondrial depolarization. Mol Immunol. 2006;43:915–926. doi: 10.1016/j.molimm.2005.06.031. [DOI] [PubMed] [Google Scholar]


