Abstract:
Anticoagulation for the open heart surgery patient undergoing cardiopulmonary bypass (CPB) is achieved with the use of heparin. The industry standard of activated clotting time (ACT) was used to measure the effect of heparin. The commonly acceptable target time of anticoagulation adequacy is 480 seconds or greater. Some patients, however, exhibit resistance to standard dosing of heparin and do not reach target anticoagulation time (480 seconds). Antithrombin III deficiency has been previously cited as the cause of heparin resistance. Early detection of heparin resistance (HR) may avoid both the delayed start of CPB and inadequate anticoagulation, if emergency bypass is required. An anticoagulation sensitivity test (AST) was developed by adding 12 units of porcine mucosa heparin to the ACT tube (International Technidyne, celite type). Before anticoagulation, 4 mL of blood was drawn from the patient arterial line. Following the manufacturer’s instructions, 2 mL of blood was added to each tube (ACT-baseline and ACT-AST). Three minutes after anticoagulation with 4 mg heparin/kg body weight, a second sample (ACT-CPB) was taken to determine anticoagulation adequacy. The ACT times of each sample were recorded for 300 procedures occurring during 2004 and were retrospectively reviewed. Heparin resistance occurred in ∼20% of the patients (n = 61). In 54 patients, heparin resistance was predicted by the ACT-AST. This was determined by the presence of an ACT-AST time and an ACT-CPB that were both <480 seconds. The positive predictive value was 90%, with a false positive rate of 3%. Heparin resistance occurs in patients undergoing CPB. We describe a simple and reliable test to avoid the delays of assessing anticoagulation for CPB (90% positive predictive value). Depending on program guidelines, patients can be given additional heparin or antithrombin III derivatives to aid in anticoagulation. An additional ACT must be performed and reach target times before CPB initiation. Testing of patient blood before the time of incision for sensitivity to heparin is a way to avoid a delay that can be critical in the care of the patient. Commercial tests are available, but efficacy data are limited, and they lead to added inventory expense. This method of titrating a diluted heparin additive, mixed with patient blood in a familiar ACT test, has proven to be an inexpensive and reliable test to predict patient’s sensitivity to heparin.
Keywords: heparin resistance, antithrombin III, anticoagulation, cardiopulmonary bypass
Anticoagulation for the open heart surgery patient is an essential modality for any procedure involving cardiopulmonary bypass (CPB), and it is achieved with the use of heparin. To measure the effect of heparin, the activated clotting time (ACT) test, an industry standard, is performed. The ACT test is a measure of the intrinsic pathway of coagulation that detects the presence of fibrin formation. The test is reproducible over a wide range of doses from 1 to 5 U/mL. The correlation of variance (CV), an indication of test variability, is <10% as required for in vitro test devices (1). This test is simple to perform, and cardiac surgery groups have reliably used it for >30 years. Although the test is affected by temperature and hemodilution, these changes affect the coagulation cascade concomitantly.
The commonly acceptable target time of anticoagulation adequacy is 480 seconds or greater to provide adequate anticoagulation and prevention of fibrin formation. This was based on previous work by Bull and others (2,3). Some patients, however, exhibit resistance to standard dosing of heparin and do not reach target anticoagulation time (480 seconds). The most common correlates of heparin resistance (HR) include previous heparin and/or nitroglycerin drips and decreased antithrombin III levels (3–5). More recently, however, antithrombin III deficiency has been cited as the cause of HR. HR is a serious condition when necessary anticoagulation is required for CPB. Therefore, early detection of this condition may avoid both the delayed start of CPB and inadequate anticoagulation, if emergency bypass is required. HR has been defined as the failure of 500–600 IU/kg of body weight of heparin to prolong the activated clotting time (ACT) to >480 seconds (6,7).
Data were collected from adult patients who were not separated regarding sex, age, or body surface area from the three hospitals in a major metropolitan area that our team provides perfusion coverage. One is a university-based academic medical center and the other two are smaller community hospital programs. HR occurred less frequently in the university program than in the community-based programs. A higher majority of patients are on heparin drips in the community-based programs.
To avoid these delays and predict which patients would have HR, we attempted to evaluate available heparin dose response (HDR) systems. No local perfusion teams could confidently recommend an HDR. The systems were primarily being used for protamine titration. Our review of available literature revealed conflicting information regarding the reliability of various systems for heparin monitoring. Dumond et al. (8) described an HDR, but concluded that it was unable to accurately calculate heparin loading doses or significantly improve the ability to predict HR in patients anticoagulated with porcine heparin. Clark et al. (9) found that the Hemostasis Management System (HMS; Medtronic Hemotec, Englewood, CO) offered the least variability vs. two other methods studied. Hardy et al. (10), however, found that this system could not be recommended for determining heparin levels because it did not correlate with laboratory determination of plasma heparin concentration.
Intuitively, we decided to develop a test to predict a patient’s response to heparin bolus based on a modified celite ACT, termed an anticoagulation sensitivity test (AST), by creating an in vitro test that would mimic heparinized patient blood. To determine the actual units per milliliter of fully heparinized patient blood, we took the patient’s heparin dose (units) divided by the blood volume (Table 1).
Table 1.
Determination of heparin level in blood for AST heparin.
| Weight—70 kg |
| Heparin dose—400 U/kg |
| Total dose = weight (70 kg) × heparin dose (400 U/kg) = 28,000 units heparin |
| Blood volume = 70 mL/kg × 70 kg = 4900 mL of blood |
| Heparin concentration in heparinized patient (units of heparin/kg) = 28,000 units divided by 4900 mL (blood volume) = 5.7 units of heparin U/mL blood |
For our test we used a concentration of 6 U/mL blood in the AST-ACT tube.
MATERIALS AND METHODS
Test Preparation
After Investigational Review Board approval for exemption from review, a log book containing only ACT data was reviewed for ACT information relevant to this audit. No other patient information was available for review. An AST was developed by adding 12 units of porcine mucosa heparin to the ACT tube (International Technidyne, celite type). To achieve this dose, 6000 units (6 mL)of porcine heparin was added to 44 mL of saline solution in a 60-mL syringe. For each test, 1 mL was removed through a 1-mL tuberculin syringe. From this tuberculin syringe, 0.1 mL (12 U heparin) was injected into the AST test tube. This addition of heparin to the ACT tube would mimic a full heparin dose of ∼6 U/mL of blood. This is slightly higher than our standard dose of heparin. From our calculations, a dose of 4 mg/kg (400 U/kg) translates to a heparin blood level of ∼5.7 U/mL of patient blood.
Anticoagulation Testing
Before anticoagulation and aprotinin, if used, 4 mL of blood was drawn from the patient arterial line. Following the manufacturer’s instructions, 2 mL of blood was added to the standard ACT tube. In the ACT-AST tube, 12 U of heparin was also added in addition to the 2 mL of blood required for the test. Three minutes after anticoagulation with 4 mg heparin/kg body weight, a second sample (ACT-CPB) was taken to determine anticoagulation adequacy. The ACT times of each sample were recorded for 300 procedures and were retrospectively reviewed.
RESULTS
A test was considered positive if the AST was <480 seconds, indicating the presence of HR. Alternatively, a negative test implied that the AST was >480 seconds and that the patient was not resistant to heparin. According to the AST, heparin resistance occurred in ∼20% of the patients (n = 61). In 54 patients, HR was predicted by the ACT-AST. This was determined by the presence of an ACT-AST time and an ACT-CPB that were both <480 seconds. A false positive would be an AST that did not reach 480 seconds but the ACT-CPB was >480 seconds. The false-positive rate was 3%, and the positive and negative predictive values were 89% and 90%, respectively (Table 2).
Table 2.
Test results from the AST and actual pre-bypass ACT
| Heparin resistant | Non-heparin resistant | Total | |
|---|---|---|---|
| AST | |||
| Positive | 54 | 7 | 61 |
| Negative | 25 | 214 | 239 |
| Total | 79 | 221 | 300 |
A positive test was an ACT or AST of 480 seconds or less. ACT is activated clotting time. AST is anticoagulation sensitivity test.
DISCUSSION
Heparin resistance occurs in patients undergoing CPB (11). In our setting of university medical centers and community hospitals, HR occurred in 8%–25%. We describe a simple and reliable test to avoid the delays of assessing anticoagulation for CPB. Depending on program guidelines, patients can be given additional heparin, fresh frozen plasma, or recombinant antithrombin III derivatives to aid in anticoagulation. An additional ACT must be performed and reach target times before CPB initiation. Testing of patient blood before the time of incision for sensitivity to heparin is a way to avoid a delay that can be critical in the care of the patient. Commercial tests are available, but efficacy data are limited, and they lead to added inventory expense.
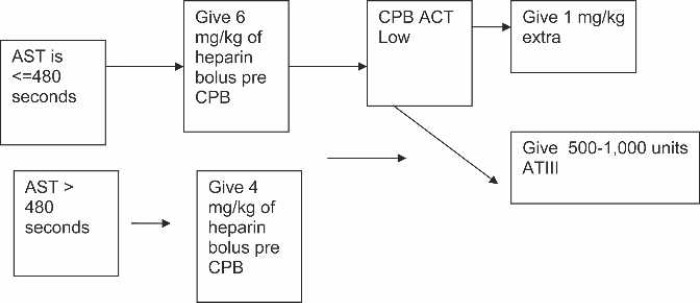
Originally, we developed this assay as part of a performance improvement project to reduce heparinization delays related to HR. We tried the AST at lower levels (i.e., 4 and 5 U/mL). At these levels, we found increased false-positive results. A level of 6 U/mL is higher than our standard heparin levels, but it avoids many false-positive results. We have not tried higher levels because of our belief that we would miss patients who have HR. The next step is to determine whether patients with a negative test result (AST < 480 seconds) should be treated with increased heparin boluses for bypass anticoagulation or given both antithrombin III concentrate and heparin bolus (Figure 1). This method of titrating a diluted heparin additive, mixed with patient blood in a familiar ACT test, has proven to be an inexpensive and reliable test to predict a patient’s sensitivity to heparin.
Figure 1.
Decision tree for treating HR before CPB.
REFERENCES
- 1.Zucker ML, Jobes C, Siegal M, et al. Activated clotting time (ACT) testing: analysis of reproducibility. J Extra Corpor Technol. 1999;31:130–4. [PubMed] [Google Scholar]
- 2.Bull BS, Korpman RA, Huse WM, et al. Heparin therapy during extracorporeal circulation: I Problems inherent in existing heparin protocols. J Thorac Cardiovasc Surg. 1975;69:674–82. [PubMed] [Google Scholar]
- 3.Young JA, Kisker CT, Doty DB.. Adequate anticoagulation during cardiopulmonary bypass determined by activated clotting time and appearance of fibrin monomer. Ann Thorac Surg. 1978;26:231–40. [DOI] [PubMed] [Google Scholar]
- 4.Dietrich W, Spannagl M, Schramm W, et al. The influence of preoperative anticoagulation on heparin response during cardiopulmonary bypass. J Thorac Cardiovasc Surg. 1991;102:505–14. [PubMed] [Google Scholar]
- 5.Habbab MA, Haft JI.. Heparin resistance induced by intravenous nitroglycerin. A word of caution when both drugs are used concomitantly. Arch Intern Med. 1987;147:857–60. [PubMed] [Google Scholar]
- 6.Esposito RA, Culliford AT, Colvin SB, et al. Heparin resistance during cardiopulmonary bypass: the role of heparin pretreatment. J Thorac Cardiovasc Surg. 1983;85:346–53. [PubMed] [Google Scholar]
- 7.Lemmer JH, Despotis GJ.. Antithrombin III concentrate to treat heparin resistance in patients undergoing cardiac surgery. J Thorac Cardiovasc Surg. 2002;123:213–7. [DOI] [PubMed] [Google Scholar]
- 8.Dumond M, Dumond D, Cook C, et al. A clinical evaluation of the correlation and reproducibility of three automated devices for measurement of activated clotting times. J Extra Corpor Technol. 1990;22:30–4. [Google Scholar]
- 9.Clark TD, Ecklund JM, McCall MM, Wolk DR, Riley JB.. Comparison of three methods to estimate heparin loading dose for cardiopulmonary bypass. J Extra Corpor Technol. 1996;28:67–70. [PubMed] [Google Scholar]
- 10.Hardy J, Belisle S, Robitaille D, Perrault J, Roy M, Gagnon L.. Measurement of heparin concentration in whole blood with the Hepcon/HMS device does not agree with laboratory determination of plasma heparin concentration using a chromagenic substrate for activated factor X. J Thorac Cardiovasc Surg. 1996;112:154–61. [DOI] [PubMed] [Google Scholar]
- 11.Avidan MS, Levy JH, Scholz J, et al. A phase III, double-blind, placebo-controlled, multicenter study on the efficacy of recombinant human antithrombin in heparin-resistant patients scheduled to undergo cardiac surgery necessitating cardiopulmonary bypass. Anesthesiology. 2005;102:276–84. [DOI] [PubMed] [Google Scholar]