Abstract
In the last decade, numerous studies of immunotherapy for malignant glioma (glioblastoma multiforme) have brought new knowledge and new hope for improving the prognosis of this incurable disease. Some clinical trials have reached Phase III, following positive outcomes in Phase I and II, with respect to safety and immunological end points. Results are encouraging especially when considering the promise of sustained efficacy by inducing antitumor immunological memory. Progress in understanding the mechanisms of tumor-induced immune suppression led to the development of drugs targeting immunosuppressive checkpoints, which are used in active clinical trials for glioblastoma multiforme. Insights related to the heterogeneity of the disease bring new challenges for the management of glioma and underscore a likely cause of therapeutic failure. An emerging therapeutic strategy is represented by a combinatorial, personalized approach, including the standard of care: surgery, radiation, chemotherapy with added active immunotherapy and multiagent targeting of immunosuppressive checkpoints.
Keywords: : clinical trials, dendritic cells, gene therapy, glioma, immune checkpoints blockade, immunotherapy, vaccination
It has long been known that immunosuppressive treatment regimens or diseases accompanied by an immunosuppressive state are associated with increased incidence of malignancy [1] and that tumors progress more slowly and can even be rejected when an immune response is elicited [2,3]. These observations have led to formulating the concept of immune surveillance [4,5], stipulating that immune mechanisms are responsible for the continuous monitoring and elimination of cells displaying neoplastic mutations. Cancer cells, glioblastoma cells included, activate mechanisms to evade immune surveillance through immunosuppressive cytokines and cells, which act upon all aspects of the immune defense to hinder the recognition and immune eradication of tumor cells.
In the context of the tumor-induced immunosuppressive environment, CD4+ T-lymphocytes either do not recognize the tumor antigens, recognize them but become anergic or differentiate into Tregs, which suppress the immune response further [6]. In patients with glioma, characterized by generalized lymphopenia, the proportion of CD4+FoxP3+ Tregs has been shown to be markedly increased and to correlate with a decreased proliferative capacity of CD4+ T cells. Depletion of Tregs restored proliferation of CD4+ T cells and this was accompanied by a decrease in Th-2 (IL-4 and IL-10) and an increase of Th-1 (IL-6, IL-2, TNF-α and IFN-γ) cytokines [7]. CD8+ cytotoxic T cells (CTLs) are functionally impaired either directly by tumor cells or indirectly through inflammatory molecules in the tumor microenvironment or through interactions with altered antigen-presenting cells (APCs), Tregs or myeloid-derived suppressor cells [8]. Decreased expression of HLA class I molecules by glioma cells, leading to impaired antigen presentation and lysis by CTLs, has been correlated with increased grade of malignancy [9]. Also, decreased expression of the costimulatory molecule B7 leads to poor costimulation and T-cell anergy [10], whereas high expression of B7-H1 (PD-L1) on tumor cells inhibits the function of CD4+ and CD8+ cells [11] and induces apoptosis of CTLs [12]. Glioma cells also express soluble Fas ligand, responsible for the death of antigen-stimulated CTLs [13]. Myeloid-derived suppressor cells, a heterogeneous population of immature myeloid cells, have also been shown to accumulate in the blood of glioma patients and inhibit T-cell function, effect mediated through their production of arginase 1 [14,15]. Natural killer (NK) cells are unable to activate their cytotoxic mechanisms in the absence of activating receptors on tumor cells [16–19], which are downregulated in glioma patients [20]. Also, glioma cells express surface proteins, like galectin-1, which inhibit NK-mediated immune surveillance [21]. Finally, type II NK T cells (NKT cells) contribute to the immunosuppressive tumor microenvironment through secretion of anti-inflammatory cytokines like TGF-β and IL-13 [22]. It is now evident that successful immunotherapy for glioma needs to address the mechanisms of tumor-induced immune suppression in addition to being mindful of the unique environment of the brain, which, unlike other organs, has minimal tolerance for inflammation.
Glioblastoma (GBM [WHO grade IV]) is the deadliest and most common form of glioma (0.59–3.69 new cases in 100,000 every year) [23]. The current standard of care (SOC): surgery, radiation and temozolomide [24] can only offer patients a median survival of 14.6 months after diagnosis and a 5-year survival rate of 0.05–4.7% [25]. Interestingly, an increasing number of studies (reviewed by [25]) indicate that an overactive immune system, as found in asthma, hay fever, eczema and food allergies, reduces the risk of developing GBM, suggesting that immune therapies for GBM could be a successful avenue to improve patient outcome. Recent accumulating evidence has highlighted the heterogeneous and evolving nature of GBM [26–29], and suggests that this heterogeneity represents a key to treatment failure. Harnessing the power of the dynamic, versatile and continuously adapting immune system to aid in the treatment of a moving target, like GBM, represents a challenging but worthwhile pursuit.
Numerous preclinical studies have demonstrated that several immunotherapeutic strategies can be successful in animal models of GBM, including: gene therapy [30], passive immunotherapy with antibodies against tumor antigens [31], adoptive T-cell transfer with T cells activated against tumor antigens or engineered to express chimeric antigen receptors (CARs) [32–34], immunomodulatory strategies aimed at inhibiting the immune checkpoints used by tumors to escape from immune surveillance [35,36] as well as active immunotherapy, employing peptide or dendritic cell (DC) vaccines (summarized in Table 1 [37–76]), to elicit immune reactivity against tumors and induce immunological memory capable of preventing recurrence of the disease.
Table 1. . Summary of preclinical studies of dendritic cell vaccination previously reported in the literature.
| Study (year) | Cytokines used for DC differentiation | Antigen loading on DC | Vaccine injection site | Adjuvant therapy | Cell line | Host | Tumor injection site | Treatment initiation | Day of treatment | Survival >60 days | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Liau (1999) |
GM-CSF, IL-4 |
Acid-eluted tumor peptide |
s.c |
None |
9L |
Fischer 344 rats |
Intracranial |
7 |
7, 14, 21 |
30% |
[55] |
| Akasaki (2001) |
GM-CSF, IL-4 |
DC chemically fused with irradiated tumor cells |
s.c. |
rIL-12 (i.p, d5, 7, 9, 11, 13, 15, 17, 19) |
SR-B10.A |
B10.A mice |
Intracranial |
5 |
5, 12 |
50% |
[37] |
| Aoki (2001) |
GM-CSF |
Sonicated tumor cells |
i.p. |
SUV (small unilaminar vesicle, in vitro) |
GL261 |
C57BL/6 mice |
Intracranial |
7 |
7, 14, 21 |
80% (d28) |
[39] |
| Ni (2001) |
GM-CSF with myc and raf |
Irradiated tumor cells (apoptosis) |
i.p. |
None |
GL261 |
C57BL/6 mice |
Intracranial |
3 |
3, 7, 10, 14 |
40% |
[56] |
| Insug (2002) |
GM-CSF, IL-4 |
RNA (transfection) |
i.p. |
rIL-12 (i.p., d1–5) |
GL261 |
C57BL/6 mice |
Intracranial |
-21 |
-21, -14, -7 |
100% |
[48] |
| Witham (2002) |
GM-CSF, IL-4, Flt3L |
UCN-01-treated tumor cells (apoptosis) |
s.c |
None |
9L |
Fischer 344 rats |
Intracranial |
0 |
0, 1, 4 |
0% |
[61] |
| Yamanaka (2002) |
GM-CSF, IL-4 |
None |
i.t. |
SFV-mediated IL-12 |
B16 |
C57BL/6 mice |
Intracranial |
7 |
7, 14, 21 |
40% |
[63] |
| Prins (2003) |
GM-CSF, IL-4 |
hgp100/TRP-2 peptides |
i.d. |
None |
GL26 |
C57BL/6 mice |
Intracranial |
0 |
0 |
75% |
[58] |
| Takagi (2003) |
GM-CSF, IL-4 |
DC chemically fused with irradiated tumor cells |
s.c. |
None |
SR-B10.A |
B10.A mice |
Flank |
0 |
0, 7 |
40% (40d) |
[60] |
| Driessens (2004) |
GM-CSF |
Irradiated tumor cells (apoptosis) |
s.c |
GM-CSF-secreting tumor |
9L |
Fischer 344 rats |
Intracranial |
4 |
4, 11, 18 |
60% |
[44] |
| Herrlinger (2004) |
(GM-CSF, IL-4) |
Iradiated, GM-CSF and IL-4 transfected GL261 |
s.c. |
GM-CSF, IL-4 |
GL261 |
C57BL/6 mice |
Intracranial |
3 |
3, 10 |
100% |
[47] |
| Saito (2004) |
GM-CSF, IL-4 |
Sonicated tumor cells |
i.t., s.c. |
pSV2muIFN-β(d7) |
GL261 |
C57BL/6 mice |
Intracranial |
3 |
3 |
50% |
[59] |
| Zhang (2004) |
GM-CSF, IL-4 |
RNA (coincubation) |
s.c. |
None |
G422 |
Kuming mice |
Intracranial |
4 |
4, 11, 18 |
0% |
[64] |
| Zhu (2005) |
GM-CSF, IL-4 |
Heat-treated tumor cells (apoptosis) |
s.c. |
None |
C6 |
Wister rats |
Intracranial |
ND |
Five-times weekly |
0% |
[65] |
| Pellegatta (2005) |
GM-CSF, IL-4 |
Freeze-thawed tumor cells (necrosis) |
s.c. |
None |
GL261 |
C57BL/6 mice |
Intracranial |
0 |
0, 7, 14 |
75% |
[57] |
| Kjaergaard (2005) |
GM-CSF, IL-4 |
DC electronically fused with irradiated tumor cells |
intrasplenic |
OX40R mAb |
GL261 |
C57BL/6 mice |
Intracranial |
7 |
7 |
80% |
[53] |
| Kuwashima (2005) |
GM-CSF, IL-4 |
Irradiated tumor cells with IL-4 transfection (apoptosis) |
i.t., s.c. (DC, tumor) |
IFN-α-producing DC, IL-4-secreting tumor |
GL261 |
C57BL/6 mice |
Intracranial |
4 |
4 (d3, 5, 7 for tumor vaccine) |
55% |
[54] |
| Clavreul (2006) |
(GM-CSF) |
Irradiated tumor cells (apoptosis) |
s.c. |
GM-CSF |
F98 |
Fischer 344 rats |
Intracranial |
4 |
4 |
0% (n.s.) |
[43] |
| Kim (2006) |
GM-CSF, IL-4 |
Freeze-thawed tumor cells (necrosis) |
s.c. |
rIL-12-producing DC |
GL26 |
C57BL/6 mice |
Intracranial |
-21 |
-21, -14, -7 |
70% |
[50] |
| Jouanneau (2006) |
GM-CSF, IL-4, Flt3L; Ribomunyl, IFN-γ |
Freeze-thawed tumor cells (necrosis) |
i.p. |
None |
GL26 |
C57BL/6 mice |
Intracranial |
-14 |
-14, -7 |
37.50% |
[49] |
| Ciesielski (2006) |
GM-CSF |
Human survivin (transfection) |
s.c. |
None |
GL261 |
C57BL/6 mice |
Intracranial |
-4 |
-4, +3, +10 |
0% |
[41] |
| Cho (2007) |
GM-CSF, IL-4; TNF-α, PGE2 |
Survivin fused to protein transduction domain of HIVtat |
s.c. |
None |
GL26 |
C57BL/6 mice |
Flank |
10 |
10 |
75% |
[40] |
| Kim (2007) |
GM-CSF, IL-4 |
TAT–survivin |
s.c. |
TMZ (d3–6, i.p.) |
GL26 |
C57BL/6 mice |
Intracranial |
13 |
13 |
40% |
[52] |
| Amano (2007) |
GM-CSF, IL-4 |
RHAMM–mRNA (transfection) |
i.p. |
None |
KR158B |
C57BL/6 mice |
Intracranial |
3 |
3, 10 |
15% |
[38] |
| Kim (2007) |
GM-CSF, IL-4 |
mTERT–mRNA (transfection) |
i.m. |
None |
GL26 |
C57BL/6 mice |
Intracranial |
-14 |
-14, -7 |
85% |
[51] |
| Grauer (2007) |
GM-CSF, IL-4 |
Freeze-thawed tumor cells (necrosis) |
s.c. |
Anti-CD25 mAbs |
GL261 |
C57BL/6 mice |
Intracranial |
-14 |
-14, -7 |
80% |
[46] |
| Ciesielski (2008) |
GM-CSF with myc and raf |
Survivin 53–67 peptide |
s.c. |
None |
GL261 |
C57BL/6 mice |
Intracranial |
4 |
4, 11, 18 |
25% |
[42] |
| Xu (2009) |
GM-CSF, IL-4 |
Irradiated tumor cells (apoptosis) |
s.c. |
None |
9L |
Fischer 344 rats |
Intracranial |
7 |
7, 4, 21 |
30% |
[62] |
| Fujita (2009) |
GM-CSF; LPS; poly-ICLC, IFN-γ, IFN-α, IL-4 |
Garc177–85 + EphA2682–689 |
i.t., s.c. |
None |
GL261 |
C57BL/6 mice |
Intracranial |
0 |
0, 10 |
75% |
[45] |
| Maes (2009) |
GM-CSF |
RNA of tumor cells (elactroporation) |
s.c. |
Anti-CD25 mAbs (day-21) |
GL261 |
C57BL/6 mice |
Intracranial |
-7 |
-7 |
100% |
[66] |
| Jiang (2009) |
GM-CSF, IL-4 |
Freeze-thawed tumor cells (necrosis) |
s.c. |
CXCL10/IP-10 (d5,10,15) |
GL261 |
C57BL/6 mice |
Intracranial |
1 |
1, 8, 15 |
60% |
[67] |
| Saka (2010) |
GM-CSF, IL-4 |
IL13ra2-mRNA (transfection) |
s.c. |
None |
KR158B |
C57BL/6 mice |
Intracranial |
3 |
3, 10 |
0% |
[68] |
| Kim (2010) |
GM-CSF, IL-4 |
TMZ and radiation-treated tumor cells (apoptosis) |
s.c. |
TMZ (d2–6, i.p.) |
GL26 |
C57BL/6 mice |
Intracranial |
4 |
4, 11, 18 |
70% |
[69] |
| Xiao (2011) |
GM-CSF, IL-4 |
RNA of tumor stem cells (transfection) |
s.c. |
None |
9L |
Fischer 344 rats |
Intracranial |
3 |
3, 10, 17 |
0% |
[70] |
| Mineharu (2011) |
hFlt3L, IL-6 |
Ad-TK/GCV-treated tumor cells (autophagy/apoptosis) |
i.t., s.c. |
CpG2006 (s.c., d10), Ad-TK/Flt3L (i.t., d10) |
CNS1/9L |
Lewis/Fisher rat |
Intracranial |
10/9 |
10, 17, 24/9, 16, 23 |
90%/83.3% |
[71] |
| Feng (2012) |
GM-CSF, IL-4; TNF-α, LPS |
Freeze-thawed tumor cells (necrosis) |
s.c. |
T cell (tail vein, d3) |
C6 |
Wister rats |
Intracranial |
3 |
3, 7, 14 |
50% |
[72] |
| Zhang (2013) |
GM-CSF, IL-4 |
Freeze-thawed tumor cells (necrosis) |
i.p. |
COX-2 inhibitor |
C6 |
Wistar rats |
Intracranial |
3 |
3, 10, 17 |
50% |
[73] |
| Peng (2014) |
GM-CSF, IL-4 |
Ad-CPEB4 (transfection) |
s.c. |
None |
GL261 |
C57BL/6 mice |
Flank |
7 |
7, 14 |
Not described |
[74] |
| Bu (2015) |
GM-CSF, IL-4; TNF-α |
Exoxome from DCs pulsed with chaperon-rich tumor lysate |
i.d. |
None |
GL261 |
C57BL/6 mice |
Intracranial |
3 |
3, 6, 9, 12 (every 3 days) |
60% |
[75] |
| Li (2015) | GM-CSF | EphA1-PE38 | i.t. | None | C6 | Wistar rats | Intracranial | 10 | 10, 13, 16, 19 (every 3 days) | Not given | [76] |
DC: Dendritic cell; Flt3L: Fms-like tyrosine kinase 3 ligand; GCV: Gancyclovir; GM-CSF represents granulocyte macrophage colony-stimulating factor; i.d.: Intradermal; IL: Interleukin; i.p.: Intraperitoneal; i.t.: Intratumoral; LPS: Lipopolysaccharide; mAb: Monoclonal antibody; s.c.: Subcutaneous; TK: Thymidine kinase; TMZ: Temozolomide.
Taken into the clinic, many Phase I and II clinical trials for GBM using immunotherapy in combination with SOC have come to completion in the last 5 years. Results have shown that immunological approaches are generally safe, with minimal side effects and able to elicit specific immune responses and in some cases improve progression-free survival (PFS) and overall survival (OS) [77–85]. Studies employed gene therapy [86–93], DC vaccines, which vary primarily in the agents used to prime the DCs for antigen presentation: either GBM-associated antigens (GAA) [81,94], autologous tumor lysates [78,82,94–97] or RNA from GBM stem cells [85], with or without adjuvants aimed to activate Toll-like receptors (TLRs). A few trials tested the effect of antigenic stimulation with peptides or tumor cells [77,79,98–102] and some analyzed the effect of autologous T-cell transfer on eliciting an antiglioma immune response [103,104] or the effect of specific antibodies against GBM receptors or to deplete Tregs [105,106].
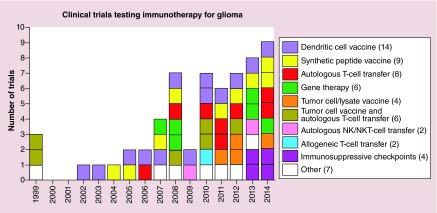
A growing interest for immunotherapeutic approaches for GBM is illustrated by the increasing annual number of funded clinical trials worldwide (ClinicalTrials.org database, Figure 1). Most of the trials involved active immunotherapy using different vaccination strategies or gene therapy. Following promising results in the treatment metastatic melanoma, where antibodies against CTLA-4 and PD-1 have shown an increase in OS [107], or induce tumor regression [108], an important new addition to the anti-GBM toolbox is represented by drugs targeting immunosuppressive checkpoints. It is currently thought that immunotherapeutic strategies most likely to succeed will entail a combination of active vaccination and immune checkpoint inhibition [109]. In this review, the authors highlight advances in immune treatment strategies for GBM during the past 5 years and present some of the ongoing challenges and future perspectives in the field. The authors apologize to all scientists whose work they were unable to mention due to limitations in space. New insights will soon be brought about by the results of open clinical trials testing immunotherapy regimens for GBM (Table 2).
Figure 1. . Timeline of clinical trials for glioma using immunotherapy.
A search for ‘immunotherapy’ and ‘glioma’ in the ClinicalTrials.gov database (March 2015) yields a list of 61 clinical trials: 14 with dendritic cell vaccines, 9 testing synthetic peptide vaccines, 8 using autologous T-cell transfer, 6 gene therapy, 4 with tumor cell lysate vaccine combined with T-cell transfer, 2 with autologous NK or NKT cell transfer, 2 with allogeneic T-cell transfer, 4 targeting immunosuppressive checkpoints and 7 using other immune treatment strategies. Limitations of the search engine may not allow a comprehensive listing; nonetheless, the graph illustrates the extensive interest in antiglioma vaccines and an emerging trend of testing immunosuppressive checkpoints.
NK: Natural killer; NKT: NK T cell.
Table 2. . Open clinical trials of immunotherapy in glioblastomas.
|
NCT number, date first received, category |
Phase |
Study title |
Diagnosis |
Intervention |
Outcome measures |
|||
|---|---|---|---|---|---|---|---|---|
| Safety Toxicity | Efficacy | Immunologic analyses | Other | |||||
|
NCT00045968 September 17 2002 DC vaccine |
III |
DCVax®-L to treat newly diagnosed GBM |
Newly diagnosed GBM |
DC vaccine made with dendritic cells differentiated from the patients own PBMC primed with autologous tumor lysate |
Y |
PFS and OS |
Analyze immune response |
|
|
NCT00626483 February 28 2008 DC vaccine |
I |
Basiliximab in treating patients with newly diagnosed glioblastoma multiforme undergoing targeted immunotherapy and temozolomide-caused lymphopenia |
GBM seropositive or seronegative for CMV |
pp65-lysosomal-associated membrane protein (LAMP) mRNA-loaded DCs with GM-CSF in combination with basiliximab, an anti CD25 monoclonal antibody used to deplete regulatory T cells |
Y |
PFS |
Functional capacity of T-reg cells, vaccine-induced cellular or humoral immune responses development of autoimmunity, phenotype of NK cells |
Characterize immunologic infiltrates in recurrent tumors |
|
NCT00634231 5 March 2008 Gene therapy |
I |
A Phase I study of AdV-tk + prodrug therapy and radiation therapy for pediatric brain tumors |
GBM and ependymoma |
AdV-tk + valacyclovir in combination with standard of care radiation |
Y |
PFS, OS |
Analyze immunologic function |
|
|
NCT01326104 22 July 2010 Autologous T cells w DC vaccine |
I/II |
Vaccine immunotherapy for recurrent medulloblastoma and primitive neuroectodermal tumor |
Medulloblastoma, neuroectodermal tumor |
One time iv. administration of autologous ex vivo-expanded autologous T-cell transfer (TTRNA-xALT) in combination with 3 weekly doses tumor RNA-loaded DCs (TTRNA-DCs) |
Y |
PFS, OS |
Magnitude and persistence of antitumor humoral and cellular immunity, cytokine production, profile of lymphocytes |
Identify potential tumor-specific antigens as vaccine candidates |
| NCT0140067 19 July 2011 Tumor lysate vaccine |
I |
Imiquimod/brain tumor initiating cell (BTIC) vaccine in brain stem glioma |
Diffuse intrinsic pontine glioma |
BTIC (brain tumor initiating cells) lysate vaccine and the TLR7 agonist Imiquimod in combination with radiation therapy |
Y |
PFS |
|
|
|
NCT01657734 7 September 2011 Other |
|
Multimodal MR imaging in patients with glioblastoma treated with dendritic cell therapy |
GBM following immunotherapy |
Observational |
N |
N |
N |
MRI spectroscopy, perfusion imaging and diffusion imaging to characterize inflammatory response, metabolism and tissue structure |
|
NCT01454596 6 October 2011 Autologous T cells ahIL-2 and chemotherapy |
I |
CAR T-cell receptor immunotherapy targeting EGFRvIII for patients with malignant gliomas expressing EGFRvIII |
EGFRvIII positice glioma and gliosarcoma with failed prior radiotherapy with or without prior chemotherapy |
Autologous T-cell transfer with retrovirally transduced PBLs with EGFRvIII CAR, combined with IL-2 and the chemotherapeutics: fludarabine and cyclophosphamide |
Y |
PFS |
N |
|
|
NCT01522820 25 January 2012 Autologous T cells with DC vaccine |
I |
A Phase I clinical trial of mTOR inhibition with rapamycin for enhancing intranodal DC vaccine-induced antitumor immunity in patients with NY-ESO-1 expressing solid tumors |
NY-ESO-1 expressing solid tumors |
Intranodal administration of the CDX-1401 vaccine (a fusion protein consisting of a fully human monoclonal antibody with specificity for the dendritic cell receptor DEC-205 linked to the NY-ESO-1 tumor antigen) with or without the mTOR inhibitor: rapamycin |
Y |
PFS |
NY-ESO-1-specific cellular and humoral immunity |
|
|
NCT01567202 26 March 2012 DC vaccine |
III |
A triple-blind randomized clinical study of vaccination with dendritic cells loaded with glioma stem-like cells-associated antigens against brain GBM |
GBM |
SOC (surgery, radiation, temozolomide) with or without DC vaccine generated from PBMC primed with stem-like cell-associated antigens (SAA) derived from the patients’ own tumor |
N |
PFS, OS |
N |
|
|
NCT01702792 3 October 2012 Other |
I |
Vaccination of patients with newly diagnosed GBM using autologous tumor lysate and montanide emulsion for derivation of tumor-specific hybridomas |
Newly diagnosed GBM |
Derivation of tumor-specific hybridomas |
Y |
N |
N |
Number of hybridoma clones that produce glioma-specific antibodies |
|
NCT01678352 30 August 2012 Tumor lysate vaccine |
0 |
Imiquimod and tumor lysate vaccine immunotherapy in adults with high risk or recurrent grade II gliomas |
WHO grade II glioma |
BTIC lysate vaccine and the TLR7 agonist imiquimod |
Y |
N |
Induction of BTIC lysate-specific T-cell response |
|
|
NCT01795313 12 February 2013 Peptide vaccine |
|
Immunotherapy for recurrent ependymomas in children using HLA-A2-restricted tumor antigen peptides in combination with imiquimod |
Recurrent ependymoma in HLA-2A-positive pediatric patients |
Vaccine with HLA-2A restricted peptides and imiquimod as adjuvant |
Y |
|
Tumor-associated antigen-specific T-cell responses |
|
|
NCT01811992 13 February 2013 Gene therapy |
I |
Combined cytotoxic and immune-stimulatory therapy for glioma |
Malignant glioma, GBM |
Dose escalation of Ad-hCMV-TK and Ad-hCMV-Flt3L |
Y |
PFS, postoperative survival |
Adenioviral antibodies |
MRI and clinical assessment |
|
NCT01952769 15 September 2013 Immunosuppressive checkpoint blockade |
I/II |
A Phase I/II clinical trial of CT-011 (pidilizumab) in diffuse intrinsic pontine glioma and relapsed glioblastoma multiforme |
DIPG, relapsed GBM |
Administration of CT-011 (pidilizumab, anti PD-L1 antibody) in patients with DIPG or recurrent GBM |
Y |
PFS, OS, ORR |
N |
Response on imaging and neurological examination |
|
NCT01956734 27 September 2013 Gene therapy |
I |
Virus DNX2401 and temozolomide in recurrent glioblastoma |
GBM recurrent tumor |
Stereotaxic, intratumoral injection of the oncolytic virus DNX2401 in combination with temozolomide |
Y |
PFS, OS12 |
Immunogenicity |
Biomarkers and tumor genetics |
|
NCT02017249 7 November 2013 Other |
I |
Double-blinded randomized placebo-controlled trial analyzing the efficacy of oral arginine to improve immune function in GBM |
GBM |
Arginine supplement will be administered orally three times per day for 7 days before surgery and 7 days after surgery |
|
|
Change in immune function (>25% increase in functional response of peripheral T cells) |
|
|
NCT02010606 2 December 2013 DC vaccine |
I |
Phase I trial of vaccination with autologous DCs pulsed with lysate derived from an allogeneic glioblastoma stem-like cell line for patients with newly diagnosed or recurrent glioblastoma |
Newly diagnosed or recurrent GBM |
SOC with DC vaccine primed with autologous tumor cell lysate and optional bevacizumab |
Y |
|
Immune response: T-cell activity |
Tumor stem cell antigen expression |
|
NCT02017717 17 December 2013 Immunosuppressive checkpoint blockade |
III |
A randomized study of nivolumab versus bevacizumab and a safety study of nivolumab in adult subjects with recurrent GBM (CheckMate 143) |
Recurrent GBM |
Administration of nivolumab (anti PD-L1 antibody) versus bevacizumab and nivolumab or nivolumab in combination with ipilimumab (anti-CTLA antibody) in adult subjects with recurrent GBM |
Y |
OS, PFS, ORR |
N |
|
|
NCT02049489 26 January 2014 DC vaccine |
I |
A study of ICT-121 dendritic cell vaccine in recurrent glioblastoma |
GBM |
Repeated administration of ICT-121 DC vaccine prepared from autologous DCs pulsed with purified CD133 peptides to target GSC |
Y |
ORR OS, PFS |
Cytotoxic T-cell response to the ICT-121 vaccine epitopes |
Antigen expression, culture of CD133+ neurospheres |
|
NCT02060955 10 February 2014 Autologous T cells |
II |
Randomized Phase II multicenter study to investigate efficacy of autologous lymphoid effector cells specific against tumor cells (ALECSAT) in patients with GBM measured compared with avastin/irinotecan |
GBM |
Autologous T-cell transfer with ex vivo antitumor activated and expanded lymphoid effector cells (ALECSAT administered at week 4, 9, 14, 26 and 46) compared with standard bevacizumab/irinotecan treatment |
Y |
OS, PFS |
N |
|
|
NCT02078648 14 February 2014 Peptide vaccine |
I/II |
Safety and efficacy study of SL-701, a glioma-associated antigen vaccine to treat recurrent GBM |
Adult GBM |
Multivalent SL-701 synthetic peptide vaccine in combination in combination with GM-CSF and imiquimod (TLR7 agonist) |
Y |
PFS/OS |
N |
|
|
NCT02122822 23 April 2014 Tumor lysate vaccine |
I |
Research for immunotherapy of glioblastoma with autologous heat shock protein gp96 |
Newly diagnosed supratentorial glioma |
Heat shock protein gp96-peptide complex made from a person's tumor cells |
Y |
PFS/OS |
Antigen-specific T-cell responses (ELISPOT) |
|
|
NCT02193347 2 July 2014 Peptide vaccine |
I |
Patients with IDH1-positive recurrent grade II glioma enrolled in a safety and immunogenicity study of tumor-specific peptide vaccine |
IDH1-positive recurrent grade II glioma |
PEPIDH1M vaccine (a peptide vaccine derived from the mutated IDH1R132H) with tetanus-toxoid preconditioning and temozolomide |
Y |
N |
IFN ELISPOT on peptide-stimulated lymphocytes |
|
|
NCT02197169 20 July 2014 Gene therapy |
Ib |
DNX-2401 with Interferon gamma (IFN-γ) for recurrent glioblastoma or gliosarcoma brain tumors |
Glioblastoma, gliosarcoma |
Modified adenovirus DNX-2401, an oncolytic adenovirus with or without IFN |
Y |
PFS |
IImmunological and biological effects after DNX-2401 with interferon gamma |
|
|
NCT02208362 1 August 2014 Autologous T cells |
I |
Genetically modified T cells in treating patients with recurrent or refractory malignant glioma |
Refractory malignant glioma |
Intratumoral, intracavitary autologous T-cell transfer with central memory enriched T cells transduced with lentivirus to express an IL13Rα2-specific, hinge-optimized, 41BB-costimulatory chimeric receptor and a truncated CD19 |
Y |
PFS, OS |
T-cell detection in tumors |
Tumor size, intracerebral inflammation, IL13Ra2 expression on tumor cells |
|
NCT02311582 4 December 2014 Immunosuppressive checkpoint blockade |
I/II |
A Phase I and open label, randomized, controlled Phase II study testing the safety, toxicities and efficacy of MK-3475 in combination with MRI-guided laser ablation in recurrent malignant gliomas |
Malignant glioma |
Administration of MK-3475 (anti PD-L1 antibody) in combination with MRI-guided laser ablation |
Y |
PFS, OS |
Antiglioma immune response before and after treatment. Correlate intratumoral expression of PD-L1 with number of glioma cell-specific cytotoxic T cells and OS |
Identify PD-1-dependent biomarkers |
|
NCT02149225 16 May 2014 Personalized peptide vaccine |
I |
Trial of actively personalized peptide vaccinations plus immunomodulators in patients with newly diagnosed GBM concurrent with temozolomide maintenance |
GBM |
Administration of APVAC1 vaccine plus poly-ICLC and GM-CSF or APVAC2 vaccine plus poly-ICLC and GM-CSF |
Y |
PFS, OS |
Frequency of peptide-specific CD8 T cells, No. of T-cell responses per peptides vaccinated No. of patients with at least one and two vaccine-induced T-cell responses Average number of immune responses per patient |
Frequency of immune cell populations in the blood. Serum and plasma immunological proteins. Noncellular parameters measured from tumor, plasma or serum. Cellular parameters from PBMCs, leukapheresis samples or isolated TILs |
| NCT02336165 23 December 2014 Immunosuppressive checkpoint blockade | II | Phase II study to evaluate the clinical efficacy and safety of MEDI4736 in patients with glioblastoma | GBM | Administration of MEDI4736 (a human monoclonal antibody directed against PD-L1) in combination with radiotherapy and bevacizumab | Y | OS, PFS, ORR | Biological activity of MEDI4736, as assessed by immunologic markers | Pharmacokinetic profile of MEDI4736: half-life, Tmax, Cmax |
Worldwide, there are currently (ClinicalTrials.gov, 10 March 2015) 29 open clinical trials assessing immunotherapeutic approaches for the treatment of glioblastoma: therapeutic strategies include: DC vaccines (5 trials), autologous T cell transfer (3), autologous T cell transfer with DC vaccine therapy (1), synthetic peptide vaccines (5), tumor lysate vaccines (3), gene therapy (4) and targeting immunosuppressive checkpoints (4). In addition, one study is looking at generating anti-GBM antibodies with hybridomas, one study is an MRI observational study following DC vaccination and one is looking at arginine as immunostimulant.
BTIC: Brain tumor-initiating cell; CAR: Chimeric antigen receptor; CMV: Cytomegalovirus; DC: Dendritic cell; DIPG: Diffuse intrinsic pontine glioma; GBM: Glioblastoma multiforme; ORR: Overall response rate; OS: Overall survival; PBMC: Peripheral blood mononuclear cell; PFS: Progression-free survival.
Targeting immunosuppressive checkpoints
Immune checkpoints are negative-regulatory signaling mechanisms responsible for maintaining self tolerance and preventing autoimmune reactions, which attenuate the strength and duration of normal T-cell-mediated immune responses. It has become apparent that diverse cancers, including GBM, co-opt the physiological function of immune checkpoints to greatly diminish T-cell-mediated antitumor immunity [11,36,110,111].
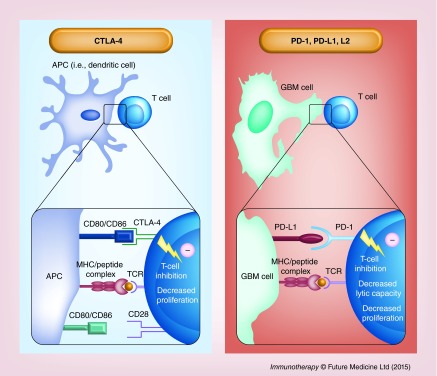
Two major checkpoints have been identified as immune escape mechanisms in both rodent and human cancers: CTLA-4/CD152 and PD-1/CD279 [110]. Both attenuate T-cell activation and promote T-cell anergy, however, they differ in their spatial and temporal activity. CTLA-4 is a powerful inhibitory T-cell receptor which preferentially binds to B7.1/CD80 and B7.2/CD86, ligands expressed on the surface of APCs, precluding their binding to the T-cell costimulatory receptor CD28 and thus inhibiting T-cell proliferation and cytokine production [112]. The CTLA-4 immune checkpoint occurs early in the immune response, during the priming phase and acts primarily within secondary lymphoid organs. Conversely, PD-1 signaling takes place directly within the tumor microenvironment, during the effector phase, by interacting with one of two currently identified PD-1 ligands: PD-L1/B7-H1/CD274 [113] or PD-L2/B7-DC/CD273 [112] expressed on the surface of cancer cells. Engagement of PD-1 ligands with the PD-1 T-cell receptor also leads to T-cell inhibition by blocking cell proliferation and inhibiting cytokine production [113,114]. CTLA-4 and PD-1 are both commensurately upregulated at the T-cell surface in response to proinflammatory cues. A diagrammatic summary of the CTLA-4 and PD-1 immune checkpoints is shown in Figure 2.
Figure 2. . CTLA-4 and PD-L1 immune checkpoints in glioma immune escape.
The CTLA-4 immune checkpoint (left panel) occurs during the priming phase of the immune response, primarily within secondary lymphoid organs. The inhibitory CTLA-4 T-cell receptor binds with higher affinity to the CD80/86 ligands on the surface of APCs and prevents their binding to and signaling through the costimulatory receptor CD28. This leads to decreased T-cell activation and proliferation in the context of antigen presenting MHC class I. PD-1 signaling (right panel) occurs during the effector phase of the immune response within the tumor microenvironment. The PD-1 receptor on the T-cell surface interacts with one of two PD-1 ligands that are expressed on the surface of tumor cells: PD-L1 or PD-L2. This interaction, in the context of tumor antigen presenting MHC class I, decreases the T-cell tumor lytic capacity and induces T-cell anergy.
APC: Antigen-presenting cell.
Cytotoxic T-lymphocyte-associated protein 4
A few human monoclonal antibodies targeting CTLA-4 (e.g., ipilimumab and tremelimumab) have been evaluated for safety and efficacy in human cancer patients: ipilimumab has been shown to improve OS in patients with metastatic melanoma in a Phase III clinical trial [107]; however, another Phase III trial using tremelimumab failed to show significant survival advantage [115]. Several Phase I and II studies of solid tumors using these antibodies show promise of improved PFS [116] but can also elicit severe immune adverse effects [117].
Preclinical investigations using anti-CTLA-4 antibodies against primary brain cancers have demonstrated significant increases in animal survival. For example, Fecci et al. have shown that administration of anti-CTLA-4 antibodies results in long-term survival in 80% of immunocompetent mice bearing syngeneic SMA-560 intracranial tumors [111]. The treatment normalized systemic CD4+ T-cell counts and decreased the number of Tregs (CD4+/CD25+/Foxp3+/GITR+) [111]. Intratumoral administration of IL-12 in combination with Anti-CTLA-4 antibodies leads to the eradication of intracranial GL261 gliomas, increasing the number of CD8+ effector cells and reducing Foxp3+ Tregs within the tumor microenvironment [118]. Also, vaccination with GM- CSF-expressing glioma cells, when combined with anti-CTLA-4 antibodies, has been shown to be more effective against established murine GL261 intracranial tumors than either treatment alone [119].
Programmed cell death protein 1
Promising results in the treatment of metastatic melanoma and other solid tumors have been demonstrated in many trials using the anti-PD-1 human antibodies: nivolumab and pembrolizumab [108,120,121]. Preclinical glioma studies using anti-PD-1 antibodies have also shown effectiveness. The combination of anti-PD-1 antibodies and radiotherapy has been shown to double median survival and elicit long-term survival in 15–40% of mice bearing GL261 gliomas compared with either treatment alone [122]. The authors show that the tumor microenvironment was infiltrated by CD8+/IFN-γ+/TNF-α+ CTLs along with reductions in tumor-infiltrating CD4+/Foxp3+ Tregs, thus suggesting that the mechanisms targeted by immune checkpoint blocking antibodies in animal studies are similar to those in human clinical trials. Recently, an investigation by Wainwright et al. has demonstrated that combinatorial targeting of immune checkpoints in the murine GL261 glioma model is more effective than single agent treatment, a strategy which could carry high potential value for future clinical trials for GBM [123]. The establishment of long-term antitumor immunological memory in many of these preclinical studies as demonstrated by the failure of tumors to grow in response to tumor rechallenge suggests a potential added benefit of immune checkpoint blockade in the prevention of tumor recurrence.
A few clinical trials have recently started to test the safety and efficacy of immune checkpoint blockade in GBM patients. A randomized Phase III study is aimed to test nivolumab versus bevacizumab in adult patients with recurrent GBM (CheckMate 143, ClinicalTrials Identifier: NCT02017717). One of the arms of this trial will test the combination therapy of nivolimab and ipilimumab. Three Phase I/II studies will analyze pembrolizumab with or without bevacizumab (NCT02337491) or pembrolizumab in combination with MRI-guided laser ablation (NCT02311582) in patients with recurrent GBM and will test the effect of anti-PD-1 antibody, pidilizumab against diffuse intrinsic pontine glioma and recurrent GBM (NCT01952769). MEDI4736, a human anti-PD-L1 antibody, is currently being tested in combination with radiotherapy and bevacizumab in the treatment of GBM (NCT02336165). A kinase inhibitor for TGFβRI (galunisertib) will be evaluated in combination with nivolumab in several advanced solid tumors, including GBM in a Phase Ib/II safety study (NCT02423343).
Enthusiasm over the use of immune checkpoint blockade as a powerful immunotherapeutic strategy in the fight against cancer has been undermined by a relatively high frequency of immune-related adverse effects in the form of gastrointestinal, dermatological, hepatic and endocrinological toxicities [124] which, in extreme cases, have led to treatment-related death [125,126]. It is thought that immune-related adverse effects associated with immune checkpoint blockade are due to aberrant infiltration of activated CD4+ and CD8+ T cells into normal tissues together with elevated levels of proinflammatory cytokines [127]. One study found grade 3–4 toxicities in 41 of 296 patients, with three treatment-related deaths attributed to pneumonitis in response to treatment to the PD-1 inhibitor nivolumab [121]. Newer agents targeting cognate PD-1 ligands (PD-1Ls) have now been tested in NSCLC, renal cell cancer and melanoma (NCT00729664) and show durable tumor regression with less grade 3 or 4 adverse events compared with anti-CTLA-4 and anti-PD-1 blockade [128].
Immune stimulatory gene therapy
Clinical trials targeting gliomas with gene therapy started in the early 1990s [93] and have employed a variety of vehicles to deliver genes, such as viral vectors, synthetic nanoparticles and liposomes, neural and mesenchymal or embryonic stem cells [87,88,92,93,129] These gene therapeutic approaches have aimed to introduce suicide genes or oncolytic viruses into the tumor cells and/or to induce the expression of immunomodulatory cytokines to aid the antitumor immune response. More recently, genetic techniques have been used to generate lymphocytes expressing CARs [33,34,130–132], with great selectivity and cytotoxicity against tumor cells. While viral-mediated suicide gene therapy and oncolytic viral therapy are not generally considered ‘classical’ immunotherapeutic approaches, accumulating data demonstrated that, in some cases, viral/gene therapy-mediated tumor cell death qualifies as immunogenic cell death initiating ER stress, expression of calreticulin on the cell surface, release of damage-associated molecular patterns like HMGB1 [133,134] and ATP and of pathogen-associated molecular patterns from the viral vector/oncolytic virus, leading to enhanced antigen presentation and antitumor immune response [135,136].
Suicide gene therapy involves the introduction of viral genes into the tumor cells, most commonly thymidine kinase (TK), resulting in the conversion of a systemically administered prodrug: gancyclovir (GCV) into a toxic metabolite within tumor cells and leading to tumor cell death [93,137]. In addition, vectors for gene therapy can be modified to express immunostimulatory molecules, which will aid in the fight against GBM. A number of studies using replication-deficient viruses have been conducted for GBM [92,138], including a bicistronic system that carries IL-2 and TK [90] and Flt3L and TK [139].
Oncolytic viral therapy utilizes replication-competent viral vectors, able to selectively replicate in tumor cells, induce tumor cell lysis and spread of viruses to adjacent cells [87,93,140]. Additionally, the nonlytic viruses can express therapeutic genes in target cells. Oncolytic herpes simplex virus (HSV), measles virus, poliovirus, Newcastle disease virus and conditionally replicating adenovirus are all being tested at various stages in GBM therapy [88]. G207 is a conditionally replicating mutant HSV that has an impaired RR enzyme allowing it to replicate only in dividing cells. Since HSV is a human pathogen with neurotropic properties, this genetic manipulation provides tumor selectivity with safety. Phase I and Ib clinical studies using G207 showed no treatment-related toxicity with repeated doses and direct injection into the resected cavity [141,142]. Promising therapeutic responses were also identified in 8 out of 21 patients in the Phase I study and 3 out of 6 patients in the Phase Ib trial. Another genetically modified mutant HSV, HSV1716 was also tested in a Phase I study, demonstrating no toxicity [91]. In a subsequent Phase Ib study, no treatment-related toxicity was observed and evidence of viral replication was seen in tumor biopsies by histological examination. Second-generation oncolytic HSV vectors are also in preclinical development. Such vectors have been engineered to express therapeutic transgenes such as TNF-α, VEGF and IL-4 [93]. The two commonly tested conditionally replicating Ads in glioma are ONYX-015 and Ad5Delta24 [137]. ONYX-015 has a deletion in the early region E1B-55kD protein that normally binds to inactivated p53 in the infected cells preventing them from undergoing apoptosis. Absence of this protein allows the virus to only replicate in p53-deficient tumor cells. Phase I studies with ONYX-015 in patients with recurrent glioma that were injected with various doses of ONYX-015 in the resection cavity showed neither treatment-related toxicity nor clinical benefit [143,144]. The Ad5Delta24 can selectively replicate in glioma cells because of a deletion in the viral protein E1A and preclinical studies have shown therapeutic efficacy against glioma xenografts [145]. However, cancer cells with intact Rb protein are refractory to Ad5Delta24. Ad5Delta24 was further modified (Ad5Delta24-RGD) to increase its targeting to tumor cells and is currently under a Phase I study. Genetically modified variants of measles virus such as MV-Edm, that have a high affinity for cellular CD46 receptors abundantly expressed on tumor cells, have also been tested in preclinical settings with favorable therapeutic success [146,147]. MV-Edm-CEA is also in a Phase I study. Specific targeting to tumor cells has also been achieved by the creation of variants expressing glioma-specific ligands such as IL-13 against IL-13Rα2 [148] and glycoproteins gD and gB to mediate HSV infection exclusively through recognition of EGFR on glioblastoma cells [149].
Immune stimulatory gene therapy serves to generate a robust immune response against glioma-specific antigens. Cytokine-mediated gene therapies also aim to achieve higher local concentrations of cytokines/chemokines and reduce systemic toxicity. Cytokine gene therapy with IFN-β has shown efficacy in preclinical studies with human xenografts and in mouse models of glioma, demonstrating augmentation of T-helper cell response, DC activity, NK cells and prolonged survival [150–152]. IFN-β also induces MHC I expression and therefore enhances the CTL response. A Phase I clinical trial with liposome-mediated IFN-β gene injection into the resection cavity demonstrated minimal toxicity with more than 50% tumor reduction (T1-weighted MRI) in two out of five patients for at least 16 months [153]. Another dose escalation study with Ad-IFN-β in 11 patients with GBM also showed therapeutic efficacy with no adverse effects except in one patient [154].
IFN-γ-mediated gene therapy has not shown efficacy when administered alone [137]; however, in combination with TNF-α, it enhanced the survival of glioma bearing animals along with an increase in T-cell recruitment to the tumor [155]. In other studies, IL-2 or IL-12 gene therapy resulted in growth inhibition of a rat glioma [156]. A combination of IL-2 with TK (GCV) in 12 patients with recurrent glioma using RV (retrovirus)-mediated gene therapy showed 12-month PFS and OS rates of 14 and 25%, respectively, with minor side effects [86]. Clinical trials using IL-4/TK gene-modified autologous glioma cells or fibroblasts and a replicative oncolytic HSV carrying IL-12 gene therapy are underway [89].
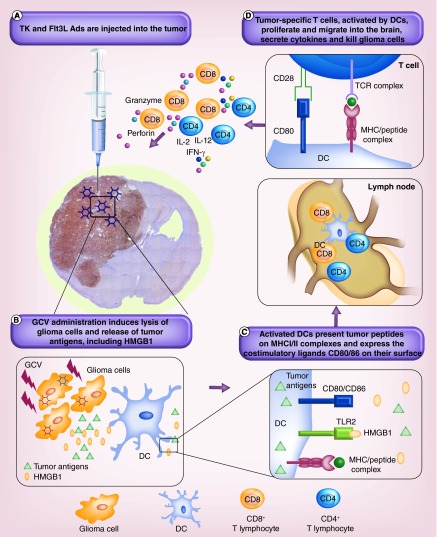
Flt3L was initially characterized as the cytokine that resulted in enhanced myelopoiesis and B lymphopoiesis and subsequently it was shown to have a potent effect on the generation of both myeloid- and lymphoid-derived DC populations in mice [157]. Our group has pioneered the development and efficacy testing of Ad-mediated delivery of recombinant human Flt3L (Ad-Flt3L) in preclinical models of glioma [158]. Administration of Ad-Flt3L significantly inhibited tumor growth and increased survival in a dose-dependent manner. We have also developed a conditionally cytotoxic-immune stimulatory gene therapy that delivers TK and FLT3L using Ads [159,160]. Tumor cells are selectively killed by the TK/GCV administration. Flt3L serves to increase the recruitment of APCs to the tumor microenvironment which take up antigens released by the dying tumor cells and subsequently induce tumor-specific T-cell responses. Dying tumor cells also produce HMGB1, which activates TLR2 on APCs. Our experiments showed that release of HMGB1 and activation of TLR2 were crucial for the TK-Flt3L-induced antiglioma response [133]. This gene therapy approach has demonstrated tumor regression, long-term survival and immunological memory in several transplantable, orthotropic syngeneic models of GBM in mice and rats [159–162]. Based on the excellent success seen in preclinical testing with the TK-Flt3L gene therapy, a Phase I clinical trial was launched in 2013, using this cytotoxic immune stimulatory approach (NCT01611992). A diagram of this therapeutic mechanism is presented in Figure 3.
Figure 3. . Gene therapy for glioma with Ad-TK and Ad-Flt3L.
(A) Thymidine kinase and Flt3L-expressing adenoviruses are injected directly into the tumor. (B) Following GCV administration, TK will convert GCV to GCV-triphosphate that is incorporated into the DNA of actively proliferating cells, that is, tumor cells, causing them to undergo apoptosis and release tumor antigens, including HMGB1. Flt3L entering systemic circulation will induce the trafficking of DCs into the tumor and their exposure to tumor antigens. (C) DCs exposed to tumor antigens process them into peptides presented on MHC class I/II molecules and increase the expression of the costimulatory molecules CD80/86 on their surface. (D) Activated DCs travel to the draining lymph nodes where they present antigenic peptides in combination with the costimulatory signals to naive CD4+ and CD8+ T cells, inducing clonal expansion and maturation of glioma-specific T cells, secretion of stimulatory cytokines, trafficking into the tumor and cytolytic killing of glioma cells.
DC: Dendritic cell; GCV: Gancyclovir.
Active immunotherapy
DC vaccines
DCs, the most effective APCs, can prime both CD4+ T helper and CD8+ cytotoxic cells [163,164] and also function as strong activators of NK and NKT cells [165,166]. To stimulate T cells and generate a specific and efficient antitumor immune response and induce immunological memory, DCs need to deliver three signals: signal (1), represented by the by cross-presentation of antigenic peptides via MHC molecules; signal (2), by the interaction of costimulatory molecules, CD80 and CD86 with the CD28 receptor located on T cells [167,168] and signal (3) by immunostimulatory cytokines such as IL-12 and IL-2 secreted by DCs and activated CD4+ T cells [169].
Preclinical studies
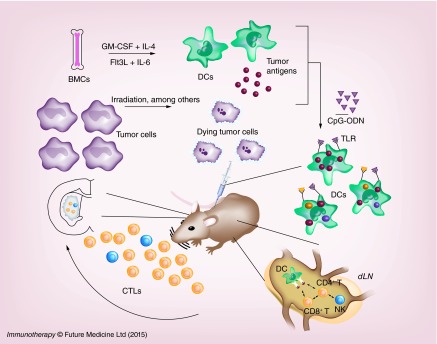
Since 1999, numerous preclinical studies (summarized in Table 1 [37–76]) have analyzed the efficiency of DC vaccines in the treatment of glioma using rodent syngeneic models. The first step in any vaccination protocol is represented by the generation of sufficient number of DCs, typically from bone marrow cells, induced to differentiate with specific cytokines like GM-CSF and/or Flt3L, pulsed with GAAs (tumor lysate or tumor-specific peptide/mRNA epitopes) and injected (most often intradermally or subcutaneously) into animals either prior to or after tumor inoculation (Figure 4). Oftentimes, adjuvants like CpG oligonucleotides or lipopolysaccharide are coinjected in order to increase the expression of maturation markers: CD80 and CD86 on DCs. Preclinical studies have aimed to optimize several factors shown to be critical in the efficacy of DC vaccination: DC differentiation, antigen loading, administration route and adjuvant treatment.
Figure 4. . Dendritic cell vaccination for glioma therapy.
Mononuclear cells extracted from bone marrow are cultured with GM-CSF and IL-4 or Flt3L and IL-6 to induce their differentiation into DCs. Tumor cells are killed by irradiation or other cytotoxic stimuli to generate tumor antigens. DCs are then pulsed with tumor antigens by co-culturing DCs with whole tumor lysates, purified and injected subcutaneously or intradermally as a vaccine together with costimulatory agents such as CpG oligonucleotide (CpG-ODN). CpG-ODNs stimulate DCs thorough signaling via Toll-like receptors. Injected DCs migrate to lymph nodes, where they encounter CD8+ and CD4+ T cells. MHC-antigen complexes are recognized by T-cell receptors and IL-12 secreted by the DCs further activates CD8+ T cells to become antigen-specific CTLs. CTLs migrate into the tumor where they attack and lyse tumor cells.
CTL: Cytotoxic T-lymphocyte; DC: Dendritic cell; GCV: Gancyclovir; TK: Thymidine kinase.
DC differentiation
Bone marrow-derived mononuclear cells can be induced to differentiate into DCs through the actions of two main cytokines, GM-CSF (used in combination with IL-4) and Flt3L (used with IL-6) [170]. It has been shown that alpha-type 1-polarized DCs induce larger numbers of antitumor CTLs, secrete increased amounts of IL-12 and are resistant to immunosuppression by Tregs [171]. DCs induced by Flt3L + IL-4 are of the α1 type, whereas DCs generated with GMCSF are not, but can be converted into α1-DCs with lipopolysaccharide, IFN-γ, IFN-α IL-4 and poly- ICLC [172].
DC loading with tumor antigens
Most commonly, whole tumor lysates have been used to load DCs with GAAs. These lysates can be generated with methods which induce necrosis or apoptosis: acid elution, freeze-thawing, irradiation, temozolomide or thymidine-kinase + GCV treatment [71,173]. Alternatively, tumor RNA and tumor-specific peptides/mRNA including ephrin (Eph)-A1, EphA2, IL-13rα2, survivin, gp100 and TRP-2 have been used to pulse DCs, or DCs have been directly fused to tumor cells [45,46,68,76]. Using multiple epitopes to pulse DCs decreases the risk of developing immune tolerance through immunoediting. Genetically engineering DCs to express common glioma antigens permit multiple epitope presentation of GAAs, irrespective of the patient's human leukocyte antigen (HLA) type.
DC vaccination route
The route of DC administration represents a critical aspect of vaccination strategies. It has been shown that increased proximity to the tumor site negatively impacts the efficacy of DC vaccinations due to increased immunosuppression [174]. In addition, intratumoral administration of immunostimulatory cytokines like IL-12, IFN-γ and Flt3L enhances the efficacy of subcutaneous DC administration [45,59,71,175], suggesting that a combinatorial approach, intratumoral, for cytokines and systemic, at the distance from the tumor, for DCs, may represent an optimal strategy for vaccination.
Adjuvant treatment
In animal models, several proinflammatory cytokines have been used to improve the therapeutic efficacy of DC vaccination, including IL-12, IFN-α, IL-4, IFN-β, CXCL10 and Flt3L (Table 1). It was thought that chemotherapy negatively impacts immunotherapy for glioma; however, it has been shown that temozolomide does not inhibit but rather enhances the effect of DC vaccination [52] and that it does not impair the efficiency of immunomodulatory gene therapy with Ad-TK+GCV and Ad-Flt3L [134]. Activation by costimulatory molecules is critical to avoid T-cell anergy [176]. OX40 receptor (CD134) is a costimulatory molecule that is expressed on activated CD4+ and transiently expressed on activated CD8+ T cells. Stimulatory anti-OX40 receptor antibodies have been shown to enhance the therapeutic efficacy of DC vaccination in a mouse glioma model [53].
Clinical studies
In clinical trials, DCs are induced to differentiate from peripheral blood mononuclear cells using most commonly GM-CSF and IL-4. DCs are then loaded with specific antigens, transfected with tumor RNA or fused with tumor cells and induced to mature using combinations of cytokines like: IL-6, TNF-α, PGE2 and IL-1β or TNF-α, IL-1β, IFN-α, IFN-γ and poly[I:C] [95]. Mature DCs can be administered directly to the patient or frozen down for future use. Oftentimes, the number of cells produced is limited and it is critical to harvest the peripheral blood mononuclear cell prior to beginning of any treatment, to avoid collecting blood when the patients become lymphopenic.
The most advanced current clinical trial with DC vaccines for glioblastomas (NCT00045968) using an autologous DC vaccine (DCVax-L), prepared by pulsing DCs with proteins from the patient's own tumor, has reached Phase III. In earlier phases of the trial, it has been shown that the vaccine is safe and elicits systemic antitumor CTL response, yet, clinical benefit was very limited. This was attributed to actively progressing tumors and high expression of TGFβ2 in the tumor [96,97]. A study using autologous DC vaccines in combination with TLR agonists as adjuvants: Imiquimod or poly-ICLC has shown that patients who had tumors with a mesenchymal, but not proneural gene expression pattern responded to treatment as demonstrated by increased OS compared with controls with the same genetic signature [82].
Another study of autologous DC vaccine pulsed with six GAA peptides (HER2, TRP-2, gp100, MAGE-1, IL13Rα2 and AIM-2) resulted in response to treatment of 33% of the 21 patients enrolled. PFS and OS correlated with the quantitative expression of MAGE1 and AIM2 from resected tumors [81]. The tumors of all patients expressed at least three antigens and 75% expressed all six antigens. The median PFS in newly diagnosed patients was 15.9 months and the median OS was 38.4 months. Furthermore, production of IFN-γ and TNF-α in stimulated CD8+ T cells was increased and correlated with survival. Expression of CD133 has been shown to increase in recurrent GBM, indicative of resistance to treatment and worse clinical outcome [177,178]. Following the GAA vaccine, there was a decrease in CD133 expression in patients with recurrent GBM, suggesting that the vaccine therapy resulted in a cytotoxic attack on glioma stem cells.
The treatment of 22 patients with recurrent GBM with α-type 1-polarized DCs (αDC1) pulsed with the GAA peptides: EphA2, IL13Rα2, YKL-40 and gp100 combined with administration of poly-ICLC showed the induction of a positive immune response at least against one of the peptides in 58% of patients, an increase in type 1 cytokines (IFN-α1 and CXCL10) and that production of IL-12 correlated with time to progression [179].
When comparing two Phase I clinical trials with DC vaccines pulsed either with autologous tumor lysate (NCT00068510) or with the GAA: TRP-2, gp100, HER-2/neu and survivin (NCT00612001), there were no significant differences between the trials with respect to frequency of helper, CTLs, B cells or NK cells; however, the GAA trial had a relative increase in activated NK cells (CD3-CD4+CD16+ CD25+) as well as an increased ratio of Tregs when compared with the tumor lysate-pulsed DC vaccine group [94]. This was associated with decreased survival in the GAA trial. Decreased Treg populations postvaccination correlated with increased survival in both groups, recommending the use of monitoring Treg populations in clinical trials of immunotherapy for GBM.
A randomized Phase II clinical trial using SOC with added antiangiogenic therapy with bevacizumab and DC vaccine (AVO113), generated by priming DCs with autologous tumor antigens, showed an increase in median OS (535 days +/- 155) in the combined bevacizumab and vaccine group compared with the vaccine group (438 days +/- 205) or bevacizumab alone group (406 days +/- 224) [78], suggesting a possible mechanism of decreased immunosuppression induced by the anti-VEGF antibodies.
Targeting GSCs with active immunotherapy represents an attractive therapeutic strategy considering that GSCs are resistant to the conventional approaches of radiotherapy and chemotherapy. Many studies have focused on identifying cellular markers of GSC, notably CD133, but also EGFRvIII, HER2, IL13Rα2 and L1-CAM, yet these markers are also found on neural stem cells and other nontumor cells [180]. A recently completed Phase I/II trial (NCT00846456) using DCs transfected with autologous GSC mRNA (extracted from glioma neurospheres), demonstrated induction of T-lymphocyte proliferation in response to tumor lysate, hTERT or survivin peptides. The vaccinated patients had significantly longer median PFS than the matched controls (694 days compared with 236 days) and also a longer median OS (759 days) than the controls [85]. This study is encouraging and highlights the benefit of identifying GSCs by their sphere-forming ability rather than by particular surface markers.
Amplifications of the EGFR gene with gain of function represent the most common (40%) genetic alteration in GBM [181]. A mutant form of the EGFR gene, EGFRvIII, found in ˜20–30% of GBM patients, expresses a truncated, constitutively active form of the receptor which results in increased proliferation and survival advantage of GBM tumor cells [182]. Another transforming mutation is EGFRvIV, with a deletion in the C-terminal domain. These mutations are very specific to glioma cells and hence an attractive target for therapy. Numerous preclinical studies demonstrated the efficiency of targeting the EGFRvIII or wild-type EGFR with peptide vaccines [183] or targeted antibodies [184] and led to development of a clinical trial with autologous DC vaccines pulsed with the EGFRvIII keyhole limpet hemocyanin (KLH)-conjugated specific peptide (PEPvIII-KLH/CDX-110), which showed safety and efficacy in eliciting an antitumor immune response and improved survival in GBM patients who express the respective variant [185].
Peptide vaccines
Peptide vaccines offer advantages compared with DC vaccines, as they do not require generation of activated and mature autologous DCs, a process that may not be feasible in all patients. It is important that the peptides are tumor-specific and that immune stimulatory strategies (immune adjuvants, cytokines: IL-2, GM-CSF) are coopted to ensure the proper priming and maturation of the endogenous APCs.
Following promising results with the DC vaccine pulsed with the EGFRvIII peptide, a subsequent Phase II multicenter study (ACTIVATE, ACTII) applied the PEPvIII-KLH/CDX-100 vaccine (Rindopepimut/CDX-110) concurrent with temozolomide, without the accompanying DCs, in patients with newly diagnosed EGFRvIII-positive GBM [186]. This study showed that 6 out of 14 patients analyzed developed EGFRvIII-specific antibody responses which correlated positively with OS, the median OS (26.0 months) being higher than in the matched historical control group (15 months) and that at recurrence 82% of patients lost EGFRvIII expression, demonstrating treatment-induced tumor immunoediting and immune escape [185,186]. A subsequent Phase II multicenter single-arm trial (ACTIII), aimed to confirm previous results using the same therapeutic approach, showed a median OS of 21.8 months, specific anti-EGFRvIII antibody titers in 85% of patients and decrease in EGFRvIII immunoreactivity in 4/6 (67%) tumor samples [84]. A current Phase III multicenter clinical trial (ACTIV, NCT01480479) is testing the efficacy of (CDX-110, Rintega, CellDex therapeutics), GM-CSF, temozolomide and KLH for the treatment of adult patients with EGFRvIII-positive glioblastomas. Another Phase II study is looking at the effects of combining rindopepimut, GM-CSF and bevacizumab for the treatment of relapsed EGFRvIII-positive glioma (NCT01498328).
Given the risk of immunoediting following single-peptide vaccinations, many investigators are aiming to produce effective combinations of GBM-specific peptides to induce robust antitumor immune responses and prevent the induction of immune tolerance. A pilot study of 26 pediatric brain stem and high-grade gliomas used a combination of three GAA peptides: EphA2, IL-13Rα2 and survivin, together with a pan HLA-DR tetanus toxoid peptide and the TLR3 agonist poly[I:C] administered intradermally in HLA-A2-positive children. This study showed that the vaccines were well tolerated, induced specific anti-GAA immune responses (by ELISPOT) and favorable clinical responses [102]. Some patients presented initial pseudoprogression, as evidenced by worsening symptoms and transient increased edema, evidenced on MRI scans, due to tumor infiltration with immune cells following the vaccine. However, patients showing pseudoprogression survived longer, suggesting that this may be a favorable prognostic marker for treatment efficacy.
In adult patients with high-risk low-grade glioma (LGG), a study using vaccinations with eight courses of intramuscular administration of the GAAs: IL13Rα2, EphA2, WT1 and Survivin emulsified with the adjuvant Montanide-ISA-51 demonstrated robust IFNγ ELISPOT responses against at least 3 out of 4 peptides in 14 out of 22 patients and median PFS of 17 months in newly diagnosed patients and 12 months in recurrent LGG. Results are encouraging and warrant further studies using this approach in patients with LGG (WHO grade II), in which the slower course of disease progression allows for repeated vaccinations with improved outcome [101].
Numerous other glioma-specific peptides have been identified (reviewed in [187]) and there is great potential in finding combinations of peptides which may be used to generate an ‘off-the-shelf’ vaccine that will be effective in a broad range of glioma patients. Ongoing clinical trials are testing a proprietary combination of 11 HLA-A2-restricted tumor-associated peptides IMA950 alone (NCT020278648) or in combination with GM-CSF (NCT01222221) or poly-ICLC (NCT01920191).
In Europe, the Glioma Actively Personalized Vaccine Consortium aims to rapidly personalize peptide vaccines, within a few months after the initial surgery, using next-generation sequencing, mass spectrometry and computational medicine algorithms. A current Phase I clinical trial in newly diagnosed GBM patients (NCT02149225) will test the safety profile of patient-tailored APVAC vaccines when administered with immunomodulators concurrent with temozolomide. The frequency of antigen-specific CD8+ T cells will be monitored, as well as immune cell populations in the blood and tumor together with a panel of serum cytokines/proteins to identify biomarkers of immune response to the vaccine.
Tumor cell vaccines
Results from a recent Phase I/II prospective clinical trial using temozolomide, fractionated radiotherapy and autologous, formalin-fixed tumor cell vaccine in 24 patients with newly diagnosed GBM show promising results with treated patients, exhibiting a median PFS of 8.2 months and OS of 22.2 months [79]. Interestingly, the median PFS in patients with a delayed-type hypersensitivity response at the third vaccination of greater than 10 mm was significantly higher (OS = 29.5 months) compared with patients with smaller delayed-type hypersensitivity, in congruence with the emerging concept that atopic reactions represent a favorable prognostic sign for immunotherapy against GBM.
The heat shock protein chaperone gp96 plays a crucial role in folding, assembly and export of TLRs and in binding to the CD91 receptor on APCs [188,189]. Activation of DCs, with tumor-associated peptides and proteins, is greatly enhanced when coupled to heatshock protein–peptide gp96. A recent Phase II clinical trial of adult recurrent GBM in which vaccines were made by isolating tumor gp96 complexes from autologous-resected tumors (HSPPC-96) demonstrated their safety and encouraged further studies of efficacy [77]. The median OS was 42.6 weeks, the 6 months survival: 90.2%, and 12 month survival of 29.3%, with poorer outcome among patients with lymphopenia. In the previous Phase I trial, with the same vaccine, significant peripheral immune responses were demonstrated in 11 out of 12 patients treated and also significant infiltration of tumor tissue with CD3+, CD8+ and CD56+ IFN-γ-positive cells. The median survival of responders was 47 weeks, longer than the 16 weeks of the single nonresponder [98]. A follow-up randomized Phase II open clinical trial (NCT01814813) will compare the efficacy of the HSPPC-96 autologous vaccine with or without bevacizumab therapy in recurrent, resectable GBMs.
Passive immunotherapy
Antibodies
Antibodies against EGFR (nimotuzumab) in combination with radiation and chemotherapy (vinorelbin) have been used in a study of pediatric diffuse intrinsic pontine gliomas, which presents with overexpression and amplification ERBB1/EGFR [105]. Results recently published from this trial show that this treatment increased median OS (15 months) when compared with a study of nimotuzumab and radiation alone (OS = 9.4). To overcome the immunosuppressive effect of tumor-induced Tregs, a randomized, placebo-controlled pilot study combined a selective antibody for the high-affinity IL-2Rα (daclizumab) with a vaccine (ZAP IT) targeting EGFRvIII, in temozolomide-treated GBM patients [106]. The study shows that one administration of daclizumab reduced the number of Tregs, without markedly affecting the number or activation of CD4+ or CD8+ T cells and favorably influenced the production of antibodies against EGFRvIII.
Autologous T-cell transfer & CAR-modified lymphocytes
Adoptive T-cell therapy was first used in the treatment of melanoma patients [32]. Treatment efficacy is, however, limited by the immunosuppressive tumor environment and the fact that not all patients have resectable tumors to be used in the production of tumor-specific lymphocytes [190]. A Phase I clinical trial in patients with recurrent GBM- and cytomegalovirus-positive serology, autologous T-cells transfer with cytomegalovirus-specific peptides-stimulated T cells, four out of ten patients who completed a minimum of three T-cell infusions showed PFS during the extent of the study. The median survival was 403 days and the median PFS was 246 days. The clinical outcome was, however, not correlated with antigen-specific T-cell number or functional phenotype [103], as indicated by expression of CD103, characteristic of recent thymic emigrant status of vaccine-induced CD8+ T cells [191].
Genetically altering lymphocytes to express CARs provides a feasible and useful method of passive immunotherapy against tumors. CARs consist of the antigen-binding region of a monoclonal antibody fused with the signal transduction domain of CD3ζ or FCεRIγ (gamma subunit of the Fc region of the immunoglobulin E receptor I) [192]. They permit independence from MHC I expression on tumors and increased penetration and persistence into the tumor microenvironment when compared with monoclonal antibodies.
CARs have been tested in clinical trials for neuroblastoma, renal cell carcinoma and B-cell malignancies [193–195]. While therapies did show therapeutic efficacy, serious adverse effects were also observed in some cases, possibly because of the expression of the targeted antigen on normal tissues. It is therefore essential to select targets that are highly specific to tumor cells. IL-13Rα2 is a cell surface receptor specific for glioma [196] and thus represents a good target for immunotherapy. Preclinical data with IL-13 zetakine CAR to target IL-13Rα2 showed elimination of human xenografts in mice [132]. The clinical trial testing safety and feasibility of this therapeutic approach in patients with recurrent GBM has recently been published [104], showing that the approach is feasible with minimal adverse effects and that two out of three patients who received repeated intracranial infusion of IL13-zetakine+CTLs developed transient antiglioma responses, suggested by increased regions of tumor necrosis visualized on MRI in one patient and decreased expression of IL-13Rα2 in another.
HER2 is expressed by up to 80% of GBMs and absent from the normal brain [197], and hence a good target for therapy. It has been shown that HER2-positive autologous GBM cells can induce a specific T-cell response with increased production of IFN-γ and IL-2 and result in tumor regression in a xenograft GBM mouse model [130]. Furthermore, these T cells can kill CD133 HER-2-positive glioma stem cells. A Phase I safety/efficacy clinical trial using HER-2-specific CARs will test this treatment in patients with glioblastoma (NCT02442297).
Immune stimulatory adjuvants
Pathogen-associated molecular patterns bind to TLRs and have a high capacity to stimulate cell-mediated immunity by increasing the production of immunostimulatory cytokines and increasing expression of costimulatory molecules on APCs. Compounds most commonly investigated as adjuvants for cancer vaccines are: polyriboinosinic–polyribocytidylic acid (poly[I:C]) and its derivative poly-ICLC, (poly[I:C] stabilized with poly-L-lysine and carboxymethylcellulose) (synthetic analogs of viral dsRNA polymers), TLR3 agonists which have been shown to enhance antitumor response by activating NK and T cells [198,199], CpG oligonucleotides (CpG ODN), strong activators of both the native and adaptive immune system [200] and TLR7 agonists, like Imiquimod [201,202].
A prospective Phase II clinical trial of pediatric glioma treated with poly-ICLC has proven to be well tolerated by children with no observed dose-limiting toxicities, five out of ten children showing long-term stable disease. The authors conclude that the results justify biomarker studies for personalization of poly-ICLC as a single agent or adjuvant. Another Phase II study of poly-ICLC and radiotherapy with concurrent and adjuvant temozolomide in newly diagnosed GBM concluded that poly-ICLC may improve the efficacy of radiotherapy and temozolamide treatment without added toxicity [203].
CpG ODNs (TLR9 agonists) have shown promise in many preclinical studies and also in Phase I clinical studies of glioma. A multicenter Phase II clinical study testing intratumoral administration of CpG-ODN for patients with recurrent GBM, showed little beneficial effect in this use of single-agent CpG-ODN [200,204]. Several trials use CpG-ODNs in combination with other immunotherapeutic agents.
Another immunostimulatory adjuvant, tetanus toxoid, has recently been shown to improve the efficacy of DC vaccination and to prolong the survival of glioblastoma patients, with more than 50% surviving longer than 40 months. In mice, it was shown that tetanus toxoid-enhanced DC migration and suppressed tumor growth in a CCL3-dependent manner [80].
Ongoing challenges
During the last 5 years, much progress has been made in refining immunotherapeutic approaches for the treatment of GBM and it is likely that soon, immunotherapy will be included in the SOC for GBM, next to maximal possible surgical resection, radiation and chemotherapy. It has become clear, however, that many details of immunotherapeutic protocols still need to be carefully assessed. With current approaches, the OS is still very short. A unique challenge of tumors localized in the brain is brought about by the minimal tolerance to inflammation, difficult to avoid when attempting immunostimulatory treatment strategies; hence, a delicate balance needs to be met between enhancing immune-mediated tumor killing and limiting brain inflammation. A great need exists to identify biomarkers with prognostic value and of clinical efficacy to guide the therapeutic intervention. Clinical trials do not have a standardized protocol to analyze the antitumor immune response and this makes it difficult to interpret the results and to compare one trial to the next. Given the tremendous heterogeneity of clinical presentations, the intrinsic heterogeneity of each individual tumor, as well as the immunoediting following various therapeutic interventions, it is apparent that a single ‘magic bullet’, a ‘one-size-fits-all’ approach will not be forthcoming. Personalized medicine with ongoing monitoring of tumor progression and immunological end points using a dynamically tailored therapeutic approach could bring great promise to the management of GBM.
Conclusion and future perspective
Current open clinical trials of immunotherapy and GBM illustrate a predominance of studies of DC vaccines in various combinatorial treatment strategies and an emerging popularity of studies with antibodies targeting immunosuppressive checkpoints (ClinicalTrials.org, Table 2). Combinatorial approaches in preclinical trials of GBM show benefit when targeting multiple immune checkpoints [123] or when adding cytokine therapy [118] and are likely to result in better outcomes when translated to the clinic. Ongoing clinical trials are testing many combination therapies. Antibodies against CTLA-4 and PD-1 are administered with bevacizumab, and/or radiotherapy or kinase inhibitors (NCT02017717, NCT02337491, NCT02336165, NCT02423343). Suicide gene therapy is combined with radiation therapy or immune-stimulatory therapy (NCT01811992, NCT00634231). Oncolytic viral therapy is used together with temozolomide or IFN-γ (NCT01956734, NCT02197169). Peptide or DC vaccines are tested with GMCSF, temozolomide, KLH or bevacizumab (NCT01920191, NCT01480479, NCT01498328) and autologous T-cell transfer with DC vaccine or IL-2 and chemotherapy (NCT01326104, NCT01522820, NCT01454596). These trials will soon shed light on the most promising avenues to pursue further.
Criteria for radiographic response assessment to immunotherapeutics have been recently defined [205]; however, it has become clear that the neuro-oncology field would benefit from guidelines tailored to the unique characteristics of brain tumors. The immunotherapy Response Assessment in Neuro-Oncology (iRANO) criteria are currently being discussed [206] and are meant to guide therapeutic decisions and prevent premature termination of immunotherapeutic treatment due to pseudoprogression in patients responding to the treatment. An open observational clinical trial (NCT01657734) is using advanced multimodal imaging techniques: MRI spectroscopy, perfusion imaging and diffusion imaging in patients with glioblastoma treated with DC therapy to characterize inflammatory response, metabolism and tissue structures and will be instrumental in further shaping the iRANO criteria.
Increased efforts are dedicated to establish reliable biomarkers to improve the assessment of response to treatment and guide further therapeutic decisions. Advances in technology: quantitative, high-sensitivity and resolution flow cytometry of rare populations, high-throughput microscopy of tumor infiltrating lymphocytes in situ, whole transcriptome profiling of tumor and immune genes, high-throughput sequencing of antigen-specific receptors of whole lymphocyte populations (TCRseq and BCRseq) permit nowadays the accumulation of large datasets, which allow for multidimensional profiling of tumors and immunological parameters [207]. This will enable a more in depth analysis of response to treatment and will likely improve therapeutic decisions and clinical outcome of GBM toward the development of optimal personalized medicine.
Executive summary.
Immunotherapy for glioblastoma multiforme (GBM) is increasingly viewed as the fourth arm in the standard of care (SOC), as several clinical studies have shown safety, increased progression-free survival (PFS) and the promise of long-term efficacy through eliciting antitumor immunological memory. Successful immunotherapy for glioma needs to address tumor-induced immune suppression in addition to being mindful of the minimal tolerance for inflammation in the brain.
Targeting immunosuppressive checkpoints
CTLA-4: receptor expressed on T-lymphocytes, inhibits costimulatory molecules on antigen-presenting cells, which results in T-cell anergy. Blocking CTLA-4 with specific antibodies (iT-cell counts and decreased the number of Tregpilimumab, tremelimumab) increases the survival of patients with metastatic melanoma and survival of animals in preclinical glioma studies, increasing antitumor CD8 cytotoxicity and decreasing accumulation of Tregs. Current clinical trials are testing these antibodies in glioma patients.
PD-1 ligands (PD-L1 and PD-L2) expressed by glioma cells bind to PD-1 receptors on T cells, blocking their proliferation and cytokine production. Anti-PD-1 antibodies (nivolumab and pembrolizumab) have shown efficacy in the treatment of metastatic melanoma and xenograft animal models of glioma. In preclinical trials, decreased Treg infiltration, increased cytotoxic antitumor immunity and establishment of immunologic memory have been demonstrated. Clinical trials are underway to test these antibodies in glioma patients.
Immunostimulatory gene therapy
Viral-mediated gene therapy induces immunogenic cell death of tumor cells which enhances antigen presentation and antitumor immune responses.
Suicide gene therapy is the most common strategy for gene therapy in glioma. Thymidine kinase introduced into the tumor by viruses (or other vectors) will convert an administered prodrug (ganciclovir), into a toxic analog, which induces apoptosis of dividing tumor cells.
Oncolytic viral therapy induces tumor cell death through viruses genetically modified to selectively replicate within tumor cells and can also express therapeutic genes like TNFα, IL-4. Viruses most commonly used are oncolytic HSV, RVs, measles virus (MV-Edm) and conditionally replicating adenovirus (ONYX-015, Ad5Delta24).
Immunomodulatory gene therapy aims to create an immunostimulatory tumor microenvironment within the brain. Clinical trials are testing combinations of suicide gene therapy or oncolytic viral therapy with immunomodulating cytokine gene therapy (IFNβ, TNFα, IFNγ, IL-2, IL-12 and Flt3L).
Active immunotherapy
Dendritic cell (DC) vaccines, generated from autologous peripheral blood mononuclear cells expanded ex vivo and induced to differentiate into DCs, pulsed with various tumor antigens and reintroduced into the patient, represent the most common strategy for active immunotherapy and personalized medicine. A Phase III multicenter randomized clinical trial with autologous DC vaccines is currently underway (NCT00045968).
Peptide vaccines offer advantages over cell-based vaccine with added convenience of an ‘off the shelf’ product. Many glioma-specific peptides have been identified and tested in combination or alone in many studies. The most commonly tested are EGFRvIII, EphA2, IL13Rα2, survivin, WT2725, gp100, YKL-40, MAGE-1 in various combinations and added immune stimulatory strategies using cytokines (IL-2, GMCSF) and adjuvants to activate TLR receptors (CpG, poly[I:C]). Limitations are restricted to the HLA type of the patients, most commonly used are HLA-A2-restricted peptides. A Phase I clinical trial for personalized peptide vaccines is conducted in Europe by the Glioma Actively Personalized Vaccine Consortium.
Tumor cell vaccines using autologous tumor lysates show increased efficacy especially in patients undergoing delayed-type hypersensitivity reactions following vaccination. Glioma peptides coupled to the heat shock protein chaperon gp96 have an increased capacity to activate DCs and elicit-specific peripheral immune responses. Clinical trials using this strategy have shown increased PFS.
Passive immunotherapy
Antibodies.
Autologous T-cell transfer with T cells stimulated with cytomegalovirus peptides in patients with CMV-positive serology has shown increased PFS in a Phase I trial.
Chimeric antigen receptor-modified lymphocytes. This strategy allows the generation of clonally expanded T cells modified to express receptors, which will recognize tumor cells independent of their MHC I expression. This approach is limited to very specific tumor antigens (like EGFRvIII and IL- 13Rα2), which are not expressed on normal cells, as to not induce severe autoimmune adverse effects.
Immune stimulatory adjuvants. These compounds are very useful in nonspecifically stimulating cell-mediated immunity through the activation of TLRs. Compounds used are: poly:[I:C], poly-ICLC, CpG- ODNs, imiquimod, tetanus toxoid.
Ongoing challenges
The survival of patients treated with SOC and effective immunotherapy is still very short.
Immunoediting and the intrinsic heterogeneity of GBMs allow tumor cells to escape treatment.
Clinical trials using combinatorial approaches with SOC and antibodies to target immunosuppressive checkpoints are underway and show promise.
Care needs to be taken when severe autoimmune adverse reactions develop; however, in many cases delayed hypersensitivity reaction may represent a positive prognostic factor.
A great need exists to identify biomarkers able to guide therapeutic strategies and monitor efficacy of immunotherapeutic approaches.
Future perspective
Ongoing clinical trials are testing combinations of immunotherapeutic approaches together with SOC and will soon shed light on the most promising avenues to pursue further.
The immunotherapy Response Assessment in Neuro-Oncology criteria are currently being discussed and will improve the therapeutic decision making process to prevent premature termination of immunotherapeutic treatment due to pseudoprogression.
Advances in technology allow for multidimensional profiling of tumors and immunological parameters which will enable a global analysis of response to treatment and will likely improve therapeutic decisions and clinical outcome of GBM through the development of optimal personalized medicine.
Acknowledgements
We thank M Dahlgren and M Dzaman for their administrative and technical support.
Footnotes
Financial & competing interests disclosure
This work was supported by NIH/National Institute of Neurological Disorders & Stroke (NIH/NINDS) Grants RO1-NS094804, R01-NS074387, R01-NS057711, R21-NS091555, University of Michigan U042841 to MG Castro; NIH/NINDS Grants R01-NS054193, R01- NS061107, R01-NS082311 and R21-NS084275 to PR Lowenstein; the Michigan Institute for Clinical and Health Research, NIH 2UL1-TR000433; University of Michigan Cancer Biology Training Grant, NIH/NCI (National Cancer Institute) T32-CA009676; University of Michigan Training in Clinical and Basic Neuroscience, NIH/NINDS T32-NS007222 and the University of Michigan Medical Scientist Training Program, NIH/NIGMS (National Institute of General Medicine Sciences) T32. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
References
Papers of special note have been highlighted as: • of interest; •• of considerable interest
- 1.Doll R, Kinlen L. Immunosurveillance and cancer: epidemiological evidence. Br. Med. J. 1970;4(5732):420–422. doi: 10.1136/bmj.4.5732.420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Greene HS. Heterotransplantation of tumors. Ann. NY Acad. Sci. 1957;69(4):818–829. doi: 10.1111/j.1749-6632.1957.tb49720.x. [DOI] [PubMed] [Google Scholar]
- 3.Baldwin RW, Robins RA. Factors interfering with immunological rejection of tumours. Br. Med. Bull. 1976;32(2):118–123. doi: 10.1093/oxfordjournals.bmb.a071342. [DOI] [PubMed] [Google Scholar]
- 4.Burnet FM. Immunological aspects of malignant disease. Lancet. 1967;1(7501):1171–1174. doi: 10.1016/s0140-6736(67)92837-1. [DOI] [PubMed] [Google Scholar]
- 5.Vesely MD, Kershaw MH, Schreiber RD, Smyth MJ. Natural innate and adaptive immunity to cancer. Annu. Rev. Immunol. 2011;29:235–271. doi: 10.1146/annurev-immunol-031210-101324. [DOI] [PubMed] [Google Scholar]
- 6.Facciabene A, Motz GT, Coukos G. T-regulatory cells: key players in tumor immune escape and angiogenesis. Cancer Res. 2012;72(9):2162–2171. doi: 10.1158/0008-5472.CAN-11-3687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Fecci PE, Mitchell DA, Whitesides JF, et al. Increased regulatory T-cell fraction amidst a diminished CD4 compartment explains cellular immune defects in patients with malignant glioma. Cancer Res. 2006;66(6):3294–3302. doi: 10.1158/0008-5472.CAN-05-3773. [DOI] [PubMed] [Google Scholar]; • Demonstrates that glioma patients present with generalized lymphopenia and that the proportion of Tregs (CD4+FoxP3+) was 2.63-times higher in glioma patients than in normal controls and correlated with decreased proliferating capacity of CD4+ T cells. Depletion of Tregs restored the functional impairment of CD4+ T cells and this was accompanied by a decrease in Th-2 and an increase of Th-1 cytokines. This is the first study to underscore the role of Tregs in facilitating immune evasion by glioblastoma multiformes (GBMs).
- 8.Crespo J, Sun H, Welling TH, Tian Z, Zou W. T cell anergy, exhaustion, senescence, and stemness in the tumor microenvironment. Curr. Opin. Immunol. 2013;25(2):214–221. doi: 10.1016/j.coi.2012.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Mehling M, Simon P, Mittelbronn M, et al. WHO grade associated downregulation of MHC class I antigen-processing machinery components in human astrocytomas: does it reflect a potential immune escape mechanism? Acta Neuropathol. 2007;114(2):111–119. doi: 10.1007/s00401-007-0231-8. [DOI] [PubMed] [Google Scholar]
- 10.Anderson RC, Anderson DE, Elder JB, et al. Lack of B7 expression, not human leukocyte antigen expression, facilitates immune evasion by human malignant gliomas. Neurosurgery. 2007;60(6):1129–1136. doi: 10.1227/01.NEU.0000255460.91892.44. discussion 1136. [DOI] [PubMed] [Google Scholar]
- 11.Wintterle S, Schreiner B, Mitsdoerffer M, et al. Expression of the B7-related molecule B7-H1 by glioma cells: a potential mechanism of immune paralysis. Cancer Res. 2003;63(21):7462–7467. [PubMed] [Google Scholar]
- 12.Dong H, Strome SE, Salomao DR, et al. Tumor-associated B7-H1 promotes T-cell apoptosis: a potential mechanism of immune evasion. Nat. Med. 2002;8(8):793–800. doi: 10.1038/nm730. [DOI] [PubMed] [Google Scholar]
- 13.Didenko VV, Ngo HN, Minchew C, Baskin DS. Apoptosis of T lymphocytes invading glioblastomas multiforme: a possible tumor defense mechanism. J. Neurosurg. 2002;96(3):580–584. doi: 10.3171/jns.2002.96.3.0580. [DOI] [PMC free article] [PubMed] [Google Scholar]; • In this study it is demonstrated that apoptotic T cells infiltrated in human GBM express Fas and are localized in close proximity or direct contact to Fas-L expressing tumor cells, suggesting a mechanism of immune evasion by GBM through the Fas/FasL pathway induced killing of T cells.
- 14.Raychaudhuri B, Rayman P, Ireland J, et al. Myeloid-derived suppressor cell accumulation and function in patients with newly diagnosed glioblastoma. Neuro Oncol. 2011;13(6):591–599. doi: 10.1093/neuonc/nor042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sippel TR, White J, Nag K, et al. Neutrophil degranulation and immunosuppression in patients with GBM: restoration of cellular immune function by targeting arginase I. Clin. Cancer Res. 2011;17(22):6992–7002. doi: 10.1158/1078-0432.CCR-11-1107. [DOI] [PubMed] [Google Scholar]
- 16.Larsen SK, Gao Y, Basse PH. NK cells in the tumor microenvironment. Crit. Rev. Oncog. 2014;19(1–2):91–105. doi: 10.1615/critrevoncog.2014011142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Levy EM, Roberti MP, Mordoh J. Natural killer cells in human cancer: from biological functions to clinical applications. J. Biomed. Biotechnol. 2011;2011:676198. doi: 10.1155/2011/676198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Vitale M, Cantoni C, Pietra G, Mingari MC, Moretta L. Effect of tumor cells and tumor microenvironment on NK-cell function. Eur. J. Immunol. 2014;44(6):1582–1592. doi: 10.1002/eji.201344272. [DOI] [PubMed] [Google Scholar]
- 19.Waldhauer I, Steinle A. NK cells and cancer immunosurveillance. Oncogene. 2008;27(45):5932–5943. doi: 10.1038/onc.2008.267. [DOI] [PubMed] [Google Scholar]
- 20.Crane CA, Han SJ, Barry JJ, Ahn BJ, Lanier LL, Parsa AT. TGF-beta downregulates the activating receptor NKG2D on NK cells and CD8+ T cells in glioma patients. Neuro Oncol. 2010;12(1):7–13. doi: 10.1093/neuonc/nop009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Baker GJ, Chockley P, Yadav VN, et al. Natural killer cells eradicate galectin-1-deficient glioma in the absence of adaptive immunity. Cancer Res. 2014;74(18):5079–5090. doi: 10.1158/0008-5472.CAN-14-1203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Robertson FC, Berzofsky JA, Terabe M. NKT cell networks in the regulation of tumor immunity. Front. Immunol. 2014;5:543. doi: 10.3389/fimmu.2014.00543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ostrom QT, Gittleman H, Farah P, et al. CBTRUS statistical report: primary brain and central nervous system tumors diagnosed in the United States in 2006–2010. Neuro Oncol. 2013;15(Suppl. 2):ii1–ii56. doi: 10.1093/neuonc/not151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Stupp R, Hegi ME, Gilbert MR, Chakravarti A. Chemoradiotherapy in malignant glioma: standard of care and future directions. J. Clin. Oncol. 2007;25(26):4127–4136. doi: 10.1200/JCO.2007.11.8554. [DOI] [PubMed] [Google Scholar]
- 25.Ostrom QT, Bauchet L, Davis FG, et al. The epidemiology of glioma in adults: a “state of the science” review. Neuro Oncol. 2014;16(7):896–913. doi: 10.1093/neuonc/nou087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Burrell RA, Mcgranahan N, Bartek J, Swanton C. The causes and consequences of genetic heterogeneity in cancer evolution. Nature. 2013;501(7467):338–345. doi: 10.1038/nature12625. [DOI] [PubMed] [Google Scholar]
- 27.Fisher R, Pusztai L, Swanton C. Cancer heterogeneity: implications for targeted therapeutics. Br. J. Cancer. 2013;108(3):479–485. doi: 10.1038/bjc.2012.581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Patel AP, Tirosh I, Trombetta JJ, et al. Single-cell RNA-seq highlights intratumoral heterogeneity in primary glioblastoma. Science. 2014;344(6190):1396–1401. doi: 10.1126/science.1254257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sottoriva A, Spiteri I, Piccirillo SG, et al. Intratumor heterogeneity in human glioblastoma reflects cancer evolutionary dynamics. Proc. Natl Acad. Sci. USA. 2013;110(10):4009–4014. doi: 10.1073/pnas.1219747110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kane JR, Miska J, Young JS, Kanojia D, Kim JW, Lesniak MS. Sui generis: gene therapy and delivery systems for the treatment of glioblastoma. Neuro Oncol. 2015;17(Suppl. 2):ii24–ii36. doi: 10.1093/neuonc/nou355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Chen KS, Mitchell DA. Monoclonal antibody therapy for malignant glioma. Adv. Exp. Med. Biol. 2012;746:121–141. doi: 10.1007/978-1-4614-3146-6_10. [DOI] [PubMed] [Google Scholar]
- 32.Morgan RA, Dudley ME, Wunderlich JR, et al. Cancer regression in patients after transfer of genetically engineered lymphocytes. Science. 2006;314(5796):126–129. doi: 10.1126/science.1129003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Morgan RA, Johnson LA, Davis JL, et al. Recognition of glioma stem cells by genetically modified T cells targeting EGFRvIII and development of adoptive cell therapy for glioma. Hum. Gene Ther. 2012;23(10):1043–1053. doi: 10.1089/hum.2012.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ohno M, Natsume A, Ichiro Iwami K, et al. Retrovirally engineered T-cell-based immunotherapy targeting type III variant epidermal growth factor receptor, a glioma-associated antigen. Cancer Sci. 2010;101(12):2518–2524. doi: 10.1111/j.1349-7006.2010.01734.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Callahan MK, Postow MA, Wolchok JD. CTLA-4 and PD-1 pathway blockade: combinations in the clinic. Front. Oncol. 2014;4:385. doi: 10.3389/fonc.2014.00385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Postow MA, Callahan MK, Wolchok JD. Immune checkpoint blockade in cancer therapy. J. Clin. Oncol. 2015;33(17):1974–1982. doi: 10.1200/JCO.2014.59.4358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Akasaki Y, Kikuchi T, Homma S, Abe T, Kofe D, Ohno T. Antitumor effect of immunizations with fusions of dendritic and glioma cells in a mouse brain tumor model. J. Immunother. 2001;24(2):106–113. [PubMed] [Google Scholar]
- 38.Amano T, Kajiwara K, Yoshikawa K, et al. Antitumor effects of vaccination with dendritic cells transfected with modified receptor for hyaluronan-mediated motility mRNA in a mouse glioma model. J. Neurosurg. 2007;106:638–645. doi: 10.3171/jns.2007.106.4.638. [DOI] [PubMed] [Google Scholar]
- 39.Aoki H, Mizuno M, Natsume A, et al. Dendritic cells pulsed with tumor extract-cationic liposome complex increase the induction of cytotoxic T lymphocytes in mouse brain tumor. Cancer Immunol. Immunother. 2001;50:463–468. doi: 10.1007/s002620100220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Cho HI, Kim EK, Park SY, Lee SK, Hong YK, Kim TG. Enhanced induction of anti-tumor immunity in human and mouse by dendritic cells pulsed with recombinant TAT fused human survivin protein. Cancer Lett. 2007;258:189–198. doi: 10.1016/j.canlet.2007.08.023. [DOI] [PubMed] [Google Scholar]
- 41.Ciesielski MJ, Apfel L, Barone TA, Castro CA, Weiss TC, Fenstermaker RA. Antitumor effects of a xenogeneic survivin bone marrow derived dendritic cell vaccine against murine GL261 gliomas. Cancer Immunol. Immunother. 2006;55:1491–1503. doi: 10.1007/s00262-006-0138-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Ciesielski MJ, Kozbor D, Castanaro CA, Barone TA, Fenstermaker RA. Therapeutic effect of a T helper cell supported CTL response induced by a survivin peptide vaccine against murine cerebral glioma. Cancer Immunol. Immunother. 2008;57:1827–1835. doi: 10.1007/s00262-008-0510-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Clavreul A, Delhaye M, Jadaud E, Menei P. Effects of syngeneic cellular vaccinations alone or in combination with GM-CSF on the weakly immunogenic F98 glioma model. J. Neurooncol. 2006;79:9–17. doi: 10.1007/s11060-005-9115-8. [DOI] [PubMed] [Google Scholar]
- 44.Driessens G, Hamdane M, Cool V, Velu T, Bruyns C. Highly successful therapeutic vaccinations combining dendritic cells and tumor cells secreting granulocyte macrophage colony-stimulating factor. Cancer Res. 2004;64:8435–8442. doi: 10.1158/0008-5472.CAN-04-0774. [DOI] [PubMed] [Google Scholar]
- 45.Fujita M, Zhu X, Ueda R, et al. Effective immunotherapy against murine gliomas using type 1 polarizing dendritic cells-significant roles of CXCL10. Cancer Res. 2009;69:1587–1595. doi: 10.1158/0008-5472.CAN-08-2915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Grauer OM, Sutmuller RPM, Van Maren W, et al. Elimination of regulatory T cells is essential for an effective vaccination with tumor lysate-pulsed dendritic cells in a murine glioma model. Int. J. Cancer. 2008;122:1794–1802. doi: 10.1002/ijc.23284. [DOI] [PubMed] [Google Scholar]
- 47.Herrlinger U, Aulwurm S, Strik H, Weit S, Naumann U, Weller M. MIP-1α antagonizes the effect of a GM-CSF-enhanced subcutaneous vaccine in a mouse glioma model. J. Neurooncol. 2004;66:147–154. doi: 10.1023/b:neon.0000013497.04322.fc. [DOI] [PubMed] [Google Scholar]
- 48.Insug O, Ku G, Ertl HCJ, Blaszczyk-Thurin M. A dendritic cell vaccine induces protective immunity to intracranial growth of glioma. Anticancer Res. 2002;22(2A):613–621. [PubMed] [Google Scholar]
- 49.Jouanneau E, Poujol D, Gulia S, et al. Dendritic cells are essential for priming but inefficient for boosting antitumour immune response in an orthotopic murine glioma model. Cancer Immunol. 2006;55:254–267. doi: 10.1007/s00262-005-0040-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Kim CH, Hong MJ, Park SD, et al. Enhancement of anti-tumor immunity specific to murine glioma by vaccination with tumor cell lysate-pulsed dendritic cells engineered to produce interleukin-12. Cancer Immunol. Immunother. 2006;55(11):1309–1319. doi: 10.1007/s00262-006-0134-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Kim CH, Yoon JS, Sohn HJ, et al. Direct vaccination with pseudotype baculovirus expressing murine telomerase induces anti-tumor immunity comparable with RNA-electroporated dendritic cells in a murine glioma model. Cancer Lett. 2007;250:276–283. doi: 10.1016/j.canlet.2006.10.022. [DOI] [PubMed] [Google Scholar]
- 52.Kim C-H, Woo S-J, Park J-S, et al. Enhanced antitumour immunity by combined use of temozolomide and TAT-survivin pulsed dendritic cells in a murine glioma. Immunology. 2007;122:615–622. doi: 10.1111/j.1365-2567.2007.02680.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Kjaergaard J, Wang L-X, Kuriyama H, Shu S, Plautz GE. Active immunotherapy for advanced intracranial murine tumors by using dendritic cell-tumor cell fusion vaccines. J. Neurosurg. 2005;103:156–164. doi: 10.3171/jns.2005.103.1.0156. [DOI] [PubMed] [Google Scholar]
- 54.Kuwashima N, Nishimura F, Eguchi J, et al. Delivery of dendritic cells engineered to secrete IFN-alpha into central nervous system tumors enhances the efficacy of peripheral tumor cell vaccines: dependence on apoptotic pathways. J. Immunol. 2005;175:2730–2740. doi: 10.4049/jimmunol.175.4.2730. [DOI] [PubMed] [Google Scholar]
- 55.Liau LM, Black KL, Prins RM, et al. Treatment of intracranial gliomas with bone marrow-derived dendritic cells pulsed with tumor antigens. J. Neurosurg. 1999;90:1115–1124. doi: 10.3171/jns.1999.90.6.1115. [DOI] [PubMed] [Google Scholar]
- 56.Ni HT, Spellman SR, Jean WC, Hall WA, Low WC. Immunization with dendritic cells pulsed with tumor extract increases survival of mice bearing intracranial gliomas. J. Neurooncol. 2001;51:1–9. doi: 10.1023/a:1006452726391. [DOI] [PubMed] [Google Scholar]
- 57.Pellegatta S, Finocchiaro G. Cell therapies in neuro-oncology. Neurol. Sci. 2005;26:43–45. doi: 10.1007/s10072-005-0405-x. [DOI] [PubMed] [Google Scholar]
- 58.Prins RM, Odesa SK, Liau LM. Immunotherapeutic targeting of shared melanoma-associated antigens in a murine glioma model. Cancer Res. 2003;63:8487–8491. [PubMed] [Google Scholar]
- 59.Saito R, Mizuno M, Nakahara N, et al. Vaccination with tumor cell lysate-pulsed dendritic cells augments the effect of IFN-β gene therapy for malignant glioma in an experimental mouse intracranial glioma. Int. J. Cancer. 2004;111:777–782. doi: 10.1002/ijc.20331. [DOI] [PubMed] [Google Scholar]
- 60.Takagi Y, Kikuchi T, Niimura M, Ohno T. Effects of glucocorticoids on antitumor effects of immunizations with fusions of dendritic and tumor cells. Anticancer Res. 2003;23:2553–2558. [PubMed] [Google Scholar]
- 61.Witham TF, Erff ML, Okada H, et al. 7-Hydroxystaurosporine-induced apoptosis in 9L glioma cells provides an effective antigen source for dendritic cells and yields a potent vaccine strategy in an intracranial glioma model. Neurosurgery. 2002;50:1327–1335. doi: 10.1097/00006123-200206000-00025. [DOI] [PubMed] [Google Scholar]
- 62.Xu Q, Liu G, Yuan X, et al. Antigen-specific T-cell response from dendritic cell vaccination using cancer stem-like cell-associated antigens. Stem Cells. 2009;27:1734–1740. doi: 10.1002/stem.102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Yamanaka R, Zullo S, Ramsey J, et al. Marked enhancement of antitumor immune responses in mouse brain tumor models by genetically modified dendritic cells producing Semliki Forest virus–mediated interleukin-12. J. Neurosurg. 2002;97:611–618. doi: 10.3171/jns.2002.97.3.0611. [DOI] [PubMed] [Google Scholar]
- 64.Zhang Z, Tang L-L, Zhan R-Y, Tong Y, Yao H-P, Du L-A. Immunotherapy of intracranial G422 glioblastoma with dendritic cells pulsed with tumor extract or RNA. J. Zhejiang Univ. Sci. 2004;5:1298–1303. doi: 10.1631/jzus.2004.1298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Zhu X, Lu C, Xiao B, Qiao J, Sun Y. An experimental study of dendritic cells-mediated immunotherapy against intracranial gliomas in rats. J. Neurooncol. 2005;74:9–17. doi: 10.1007/s11060-004-3339-x. [DOI] [PubMed] [Google Scholar]
- 66.Maes W, Rosas GG, Verbinnen B, et al. DC vaccination with anti-CD25 treatment leads to long-term immunity against experimental glioma. Neuro Oncol. 2009;11:529–542. doi: 10.1215/15228517-2009-004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Jiang X-B, Lu X-L, Hu P, Liu R-E. Improved therapeutic efficacy using vaccination with glioma lysate-pulsed dendritic cells combined with IP-10 in murine glioma. Vaccine. 2009;27:6210–6216. doi: 10.1016/j.vaccine.2009.08.002. [DOI] [PubMed] [Google Scholar]
- 68.Saka M, Amano T, Kajiwara K, et al. Vaccine therapy with dendritic cells transfected with Il13ra2 mRNA for glioma in mice. J. Neurosurg. 2010;113:270–279. doi: 10.3171/2009.9.JNS09708. [DOI] [PubMed] [Google Scholar]
- 69.Kim T-G, Kim C-H, Park J-S, et al. Immunological factors relating to the antitumor effect of temozolomide chemoimmunotherapy in a murine glioma model. Clin. Vaccine Immunol. 2010;17:143–153. doi: 10.1128/CVI.00292-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Xiao Z-Y, Tang H, Xu Z-M, et al. An experimental study of dendritic cells transfected with cancer stem-like cells RNA against 9L brain tumors. Cancer Biol. Ther. 2011;11:974–980. doi: 10.4161/cbt.11.11.15705. [DOI] [PubMed] [Google Scholar]
- 71.Mineharu Y, King GD, Muhammad AKMG, et al. Engineering the brain tumor microenvironment enhances the efficacy of dendritic cell vaccination: implications for clinical trial design. Clin. Cancer Res. 2011;17:4705–4718. doi: 10.1158/1078-0432.CCR-11-0915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Feng E, Gao H, Su W, Yu C. Immunotherapy of rat glioma without accumulation of CD4(+)CD25(+)FOXP3(+) regulatory T cells. Neural Regen. Res. 2012;7:1498–1506. doi: 10.3969/j.issn.1673-5374.2012.19.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Zhang H, Tian M, Xiu C, Wang Y, Tang G. Enhancement of antitumor activity by combination of tumor lysate-pulsed dendritic cells and celecoxib in a rat glioma model. Oncol. Res. 2013;20:447–455. doi: 10.3727/096504013x13685487925176. [DOI] [PubMed] [Google Scholar]
- 74.Peng W, Nan Z, Liu Y, et al. Dendritic cells transduced with CPEB4 induced antitumor immune response. Exp. Mol. Pathol. 2014;97:273–278. doi: 10.1016/j.yexmp.2014.06.001. [DOI] [PubMed] [Google Scholar]
- 75.Bu N, Wu H, Zhang G, et al. Exosomes from dendritic cells loaded with chaperone-rich cell lysates elicit a potent T cell immune response against intracranial glioma in mice. J. Mol. Neurosci. 2015;56(3):631–643. doi: 10.1007/s12031-015-0506-9. [DOI] [PubMed] [Google Scholar]
- 76.Li M, Wang B, Wu Z, et al. A novel recombinant protein of ephrinA1–PE38/GM-CSF activate dendritic cells vaccine in rats with glioma. Tumor Biol. 2015;36(7):5497–5503. doi: 10.1007/s13277-015-3217-5. [DOI] [PubMed] [Google Scholar]
- 77.Bloch O, Crane CA, Fuks Y, et al. Heat-shock protein peptide complex-96 vaccination for recurrent glioblastoma: a Phase II, single-arm trial. Neuro Oncol. 2014;16(2):274–279. doi: 10.1093/neuonc/not203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Buchroithner J, Pichler J, Marosi C, et al. Vascular endothelia growth factor targeted therapy may improve the effect of dendritic cell-based cancer immune therapy. Int. J. Clin. Pharmacol. Ther. 2014;52(1):76–77. doi: 10.5414/CPXCES13EA02. [DOI] [PubMed] [Google Scholar]
- 79.Ishikawa E, Muragaki Y, Yamamoto T, et al. Phase I/IIa trial of fractionated radiotherapy, temozolomide, and autologous formalin-fixed tumor vaccine for newly diagnosed glioblastoma. J. Neurosurg. 2014;121(3):543–553. doi: 10.3171/2014.5.JNS132392. [DOI] [PubMed] [Google Scholar]
- 80.Mitchell DA, Batich KA, Gunn MD, et al. Tetanus toxoid and CCL3 improve dendritic cell vaccines in mice and glioblastoma patients. Nature. 2015;519:366–369. doi: 10.1038/nature14320. [DOI] [PMC free article] [PubMed] [Google Scholar]; • Demonstrates increased efficiency of dendritic cell (DC) vaccination when the vaccine site was preconditioned with tetanus toxoid. This led to more than 50% of patients surviving longer than 40 months. In mice, it was shown that tetanus toxoid-enhanced DC migration to the lymph nodes draining the vaccine site and that this therapy suppressed tumor growth in a CCL3-dependent manner.
- 81.Phuphanich S, Wheeler CJ, Rudnick JD, et al. Phase I trial of a multi-epitope-pulsed dendritic cell vaccine for patients with newly diagnosed glioblastoma. Cancer Immunol. Immunother. 2013;62(1):125–135. doi: 10.1007/s00262-012-1319-0. [DOI] [PMC free article] [PubMed] [Google Scholar]; • Report of a Phase I clinical trial using autologous DC vaccine pulsed with six GAA (HER2, TRP-2, gp100, MAGE-1, IL13Ra2 and AIM-2) demonstrating response to treatment of 33% of the 21 patients enrolled, increased production of IFN-γ and TNF-α in stimulated CD8+ T cells, which correlated with survival, decreased expression of CD133 in recurrent GBM patients, suggesting that the treatment killed GSC cells. The median progression-free survival was 15.9 months and the median overall survival was 38.4 months, larger than other studies.
- 82.Prins RM, Soto H, Konkankit V, et al. Gene expression profile correlates with T-cell infiltration and relative survival in glioblastoma patients vaccinated with dendritic cell immunotherapy. Clin. Cancer Res. 2011;17(6):1603–1615. doi: 10.1158/1078-0432.CCR-10-2563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Riedmann EM. Agenus cancer vaccine extends survival of GBM patients. Hum. Vaccin. Immunother. 2014;10(2):252–253. [PubMed] [Google Scholar]
- 84.Schuster J, Lai RK, Recht LD, et al. A Phase II, multicenter trial of rindopepimut (CDX-110) in newly diagnosed glioblastoma: the ACT III study. Neuro Oncol. 2015;17(6):854–861. doi: 10.1093/neuonc/nou348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Vik-Mo EO, Nyakas M, Mikkelsen BV, et al. Therapeutic vaccination against autologous cancer stem cells with mRNA-transfected dendritic cells in patients with glioblastoma. Cancer Immunol. Immunother. 2013;62(9):1499–1509. doi: 10.1007/s00262-013-1453-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Colombo F, Barzon L, Franchin E, et al. Combined HSV-TK/IL-2 gene therapy in patients with recurrent glioblastoma multiforme: biological and clinical results. Cancer Gene Ther. 2005;12(10):835–848. doi: 10.1038/sj.cgt.7700851. [DOI] [PubMed] [Google Scholar]
- 87.Kroeger KM, Muhammad AK, Baker GJ, et al. Gene therapy and virotherapy: novel therapeutic approaches for brain tumors. Discov. Med. 2010;10(53):293–304. [PMC free article] [PubMed] [Google Scholar]
- 88.Kwiatkowska A, Nandhu MS, Behera P, Chiocca EA, Viapiano MS. Strategies in gene therapy for glioblastoma. Cancers (Basel) 2013;5(4):1271–1305. doi: 10.3390/cancers5041271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Okada H, Pollack IF, Lotze MT, et al. Gene therapy of malignant gliomas: a Phase I study of IL-4-HSV-TK gene-modified autologous tumor to elicit an immune response. Hum. Gene Ther. 2000;11(4):637–653. doi: 10.1089/10430340050015824. [DOI] [PubMed] [Google Scholar]
- 90.Palu G, Cavaggioni A, Calvi P, et al. Gene therapy of glioblastoma multiforme via combined expression of suicide and cytokine genes: a pilot study in humans. Gene Ther. 1999;6(3):330–337. doi: 10.1038/sj.gt.3300805. [DOI] [PubMed] [Google Scholar]
- 91.Rampling R, Cruickshank G, Papanastassiou V, et al. Toxicity evaluation of replication-competent herpes simplex virus (ICP 34.5 null mutant 1716) in patients with recurrent malignant glioma. Gene Ther. 2000;7(10):859–866. doi: 10.1038/sj.gt.3301184. [DOI] [PubMed] [Google Scholar]
- 92.Sonabend AM, Ulasov IV, Lesniak MS. Gene therapy trials for the treatment of high-grade gliomas. Gene Ther. Mol. Biol. 2007;11(A):79–92. [PMC free article] [PubMed] [Google Scholar]
- 93.Tobias A, Ahmed A, Moon KS, Lesniak MS. The art of gene therapy for glioma: a review of the challenging road to the bedside. J. Neurol. Neurosurg. Psychiatry. 2013;84(2):213–222. doi: 10.1136/jnnp-2012-302946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Prins RM, Wang X, Soto H, et al. Comparison of glioma-associated antigen peptide-loaded versus autologous tumor lysate-loaded dendritic cell vaccination in malignant glioma patients. J. Immunother. 2013;36(2):152–157. doi: 10.1097/CJI.0b013e3182811ae4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Akiyama Y, Oshita C, Kume A, et al. Alpha-type-1 polarized dendritic cell-based vaccination in recurrent high-grade glioma: a Phase I clinical trial. BMC Cancer. 2012;12:623. doi: 10.1186/1471-2407-12-623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Liau LM, Prins RM, Kiertscher SM, et al. Dendritic cell vaccination in glioblastoma patients induces systemic and intracranial T-cell responses modulated by the local central nervous system tumor microenvironment. Clin. Cancer Res. 2005;11(15):5515–5525. doi: 10.1158/1078-0432.CCR-05-0464. [DOI] [PubMed] [Google Scholar]
- 97.Yu JS, Liu G, Ying H, Yong WH, Black KL, Wheeler CJ. Vaccination with tumor lysate-pulsed dendritic cells elicits antigen-specific, cytotoxic T-cells in patients with malignant glioma. Cancer Res. 2004;64(14):4973–4979. doi: 10.1158/0008-5472.CAN-03-3505. [DOI] [PubMed] [Google Scholar]
- 98.Crane CA, Han SJ, Ahn B, et al. Individual patient-specific immunity against high-grade glioma after vaccination with autologous tumor derived peptides bound to the 96 KD chaperone protein. Clin. Cancer Res. 2013;19(1):205–214. doi: 10.1158/1078-0432.CCR-11-3358. [DOI] [PubMed] [Google Scholar]
- 99.Izumoto S, Tsuboi A, Oka Y, et al. Phase II clinical trial of Wilms tumor 1 peptide vaccination for patients with recurrent glioblastoma multiforme. J. Neurosurg. 2008;108(5):963–971. doi: 10.3171/JNS/2008/108/5/0963. [DOI] [PubMed] [Google Scholar]
- 100.Morita S, Oka Y, Tsuboi A, et al. A Phase I/II trial of a WT1 (Wilms’ tumor gene) peptide vaccine in patients with solid malignancy: safety assessment based on the Phase I data. Jpn J. Clin. Oncol. 2006;36(4):231–236. doi: 10.1093/jjco/hyl005. [DOI] [PubMed] [Google Scholar]
- 101.Okada H, Butterfield LH, Hamilton RL, et al. Induction of robust type-I CD8+ T-cell responses in WHO grade 2 low-grade glioma patients receiving peptide-based vaccines in combination with poly-ICLC. Clin. Cancer Res. 2015;21(2):286–294. doi: 10.1158/1078-0432.CCR-14-1790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Pollack IF, Jakacki RI, Butterfield LH, et al. Antigen-specific immune responses and clinical outcome after vaccination with glioma-associated antigen peptides and polyinosinic-polycytidylic acid stabilized by lysine and carboxymethylcellulose in children with newly diagnosed malignant brainstem and nonbrainstem gliomas. J. Clin. Oncol. 2014;32(19):2050–2058. doi: 10.1200/JCO.2013.54.0526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Schuessler A, Smith C, Beagley L, et al. Autologous T-cell therapy for cytomegalovirus as a consolidative treatment for recurrent glioblastoma. Cancer Res. 2014;74(13):3466–3476. doi: 10.1158/0008-5472.CAN-14-0296. [DOI] [PubMed] [Google Scholar]
- 104.Brown CE, Badie B, Barish ME, et al. Bioactivity and safety of IL13Ralpha2-redirected chimeric antigen receptor CD8+ T cells in patients with recurrent glioblastoma. Clin. Cancer Res. 2015;21(18):4062–4072. doi: 10.1158/1078-0432.CCR-15-0428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Massimino M, Biassoni V, Miceli R, et al. Results of nimotuzumab and vinorelbine, radiation and re-irradiation for diffuse pontine glioma in childhood. J. Neurooncol. 2014;118(2):305–312. doi: 10.1007/s11060-014-1428-z. [DOI] [PubMed] [Google Scholar]
- 106.Sampson JH, Schmittling RJ, Archer GE, et al. A pilot study of IL-2Ralpha blockade during lymphopenia depletes regulatory T-cells and correlates with enhanced immunity in patients with glioblastoma. PLoS ONE. 2012;7(2):e31046. doi: 10.1371/journal.pone.0031046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Hodi FS, O'Day SJ, Mcdermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N. Engl. J. Med. 2010;363(8):711–723. doi: 10.1056/NEJMoa1003466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Hamid O, Robert C, Daud A, et al. Safety and tumor responses with lambrolizumab (anti-PD-1) in melanoma. N. Engl. J. Med. 2013;369(2):134–144. doi: 10.1056/NEJMoa1305133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Bloch O. Immunotherapy for malignant gliomas. Cancer Treat. Res. 2015;163:143–158. doi: 10.1007/978-3-319-12048-5_9. [DOI] [PubMed] [Google Scholar]
- 110.Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat. Rev. Cancer. 2012;12(4):252–264. doi: 10.1038/nrc3239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Fecci PE, Ochiai H, Mitchell DA, et al. Systemic CTLA-4 blockade ameliorates glioma-induced changes to the CD4+ T cell compartment without affecting regulatory T-cell function. Clin. Cancer Res. 2007;13(7):2158–2167. doi: 10.1158/1078-0432.CCR-06-2070. [DOI] [PubMed] [Google Scholar]
- 112.Chambers CA, Kuhns MS, Egen JG, Allison JP. CTLA-4-mediated inhibition in regulation of T cell responses: mechanisms and manipulation in tumor immunotherapy. Annu. Rev. Immunol. 2001;19:565–594. doi: 10.1146/annurev.immunol.19.1.565. [DOI] [PubMed] [Google Scholar]
- 113.Freeman GJ, Long AJ, Iwai Y, et al. Engagement of the PD-1 immunoinhibitory receptor by a novel B7 family member leads to negative regulation of lymphocyte activation. J. Exp. Med. 2000;192(7):1027–1034. doi: 10.1084/jem.192.7.1027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Chemnitz JM, Parry RV, Nichols KE, June CH, Riley JL. SHP-1 and SHP-2 associate with immunoreceptor tyrosine-based switch motif of programmed death 1 upon primary human T cell stimulation, but only receptor ligation prevents T cell activation. J. Immunol. 2004;173(2):945–954. doi: 10.4049/jimmunol.173.2.945. [DOI] [PubMed] [Google Scholar]
- 115.Ribas A, Kefford R, Marshall MA, et al. Phase III randomized clinical trial comparing tremelimumab with standard-of-care chemotherapy in patients with advanced melanoma. J. Clin. Oncol. 2013;31(5):616–622. doi: 10.1200/JCO.2012.44.6112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Lynch TJ, Bondarenko I, Luft A, et al. Ipilimumab in combination with paclitaxel and carboplatin as first-line treatment in stage IIIB/IV non-small-cell lung cancer: results from a randomized, double-blind, multicenter Phase II study. J. Clin. Oncol. 2012;30(17):2046–2054. doi: 10.1200/JCO.2011.38.4032. [DOI] [PubMed] [Google Scholar]
- 117.Yang JC, Hughes M, Kammula U, et al. Ipilimumab (anti-CTLA4 antibody) causes regression of metastatic renal cell cancer associated with enteritis and hypophysitis. J. Immunother. 2007;30(8):825–830. doi: 10.1097/CJI.0b013e318156e47e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Vom Berg J, Vrohlings M, Haller S, et al. Intratumoral IL-12 combined with CTLA-4 blockade elicits T cell-mediated glioma rejection. J. Exp. Med. 2013;210(13):2803–2811. doi: 10.1084/jem.20130678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Agarwalla P, Barnard Z, Fecci P, Dranoff G, Curry WT., Jr. Sequential immunotherapy by vaccination with GM-CSF-expressing glioma cells and CTLA-4 blockade effectively treats established murine intracranial tumors. J. Immunother. 2012;35(5):385–389. doi: 10.1097/CJI.0b013e3182562d59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Robert C, Long GV, Brady B, et al. Nivolumab in previously untreated melanoma without BRAF mutation. N. Engl. J. Med. 2015;372(4):320–330. doi: 10.1056/NEJMoa1412082. [DOI] [PubMed] [Google Scholar]
- 121.Topalian SL, Hodi FS, Brahmer JR, et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N. Engl. J. Med. 2012;366(26):2443–2454. doi: 10.1056/NEJMoa1200690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Zeng J, See AP, Phallen J, et al. Anti-PD-1 blockade and stereotactic radiation produce long-term survival in mice with intracranial gliomas. Int. J. Radiat. Oncol. Biol. Phys. 2013;86(2):343–349. doi: 10.1016/j.ijrobp.2012.12.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Wainwright DA, Chang AL, Dey M, et al. Durable therapeutic efficacy utilizing combinatorial blockade against IDO, CTLA-4, and PD-L1 in mice with brain tumors. Clin. Cancer Res. 2014;20(20):5290–5301. doi: 10.1158/1078-0432.CCR-14-0514. [DOI] [PMC free article] [PubMed] [Google Scholar]; • Shows that combinatorial therapeutic inhibition of IDO, CTLA-4 and PD-L1 induces 100% long-term survival in a mouse model of well-established GBMs. This was accompanied by a decrease in tumor-infiltrating Tregs, coincident with a significant increase in CD8+IFNγ+ T cells and illustrates the benefits of multiagent targeting of immunosuppressive agents for the treatment of GBM.
- 124.Gelao L, Criscitiello C, Esposito A, Goldhirsch A, Curigliano G. Immune checkpoint blockade in cancer treatment: a double-edged sword cross-targeting the host as an “innocent bystander”. Toxins (Basel) 2014;6(3):914–933. doi: 10.3390/toxins6030914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Weber JS, Kahler KC, Hauschild A. Management of immune-related adverse events and kinetics of response with ipilimumab. J. Clin. Oncol. 2012;30(21):2691–2697. doi: 10.1200/JCO.2012.41.6750. [DOI] [PubMed] [Google Scholar]
- 126.Slovin SF, Higano CS, Hamid O, et al. Ipilimumab alone or in combination with radiotherapy in metastatic castration-resistant prostate cancer: results from an open-label, multicenter Phase I/II study. Ann. Oncol. 2013;24(7):1813–1821. doi: 10.1093/annonc/mdt107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Kaehler KC, Piel S, Livingstone E, Schilling B, Hauschild A, Schadendorf D. Update on immunologic therapy with anti-CTLA-4 antibodies in melanoma: identification of clinical and biological response patterns, immune-related adverse events, and their management. Semin. Oncol. 2010;37(5):485–498. doi: 10.1053/j.seminoncol.2010.09.003. [DOI] [PubMed] [Google Scholar]
- 128.Brahmer JR, Tykodi SS, Chow LQ, et al. Safety and activity of anti-PD-L1 antibody in patients with advanced cancer. N. Engl. J. Med. 2012;366(26):2455–2465. doi: 10.1056/NEJMoa1200694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Juratli TA, Schackert G, Krex D. Current status of local therapy in malignant gliomas – a clinical review of three selected approaches. Pharmacol. Ther. 2013;139(3):341–358. doi: 10.1016/j.pharmthera.2013.05.003. [DOI] [PubMed] [Google Scholar]
- 130.Ahmed N, Salsman VS, Kew Y, et al. HER2-specific T cells target primary glioblastoma stem cells and induce regression of autologous experimental tumors. Clin. Cancer Res. 2010;16(2):474–485. doi: 10.1158/1078-0432.CCR-09-1322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Balyasnikova IV, Ferguson SD, Sengupta S, Han Y, Lesniak MS. Mesenchymal stem cells modified with a single-chain antibody against EGFRvIII successfully inhibit the growth of human xenograft malignant glioma. PLoS ONE. 2010;5(3):e9750. doi: 10.1371/journal.pone.0009750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Kahlon KS, Brown C, Cooper LJ, Raubitschek A, Forman SJ, Jensen MC. Specific recognition and killing of glioblastoma multiforme by interleukin 13-zetakine redirected cytolytic T cells. Cancer Res. 2004;64(24):9160–9166. doi: 10.1158/0008-5472.CAN-04-0454. [DOI] [PubMed] [Google Scholar]
- 133.Curtin JF, Liu N, Candolfi M, et al. HMGB1 mediates endogenous TLR2 activation and brain tumor regression. PLoS Med. 2009;6(1):e10. doi: 10.1371/journal.pmed.1000010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Candolfi M, Yagiz K, Wibowo M, et al. Temozolomide does not impair gene therapy-mediated antitumor immunity in syngeneic brain tumor models. Clin. Cancer Res. 2014;20(6):1555–1565. doi: 10.1158/1078-0432.CCR-13-2140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Workenhe ST, Mossman KL. Oncolytic virotherapy and immunogenic cancer cell death: sharpening the sword for improved cancer treatment strategies. Mol. Ther. 2014;22(2):251–256. doi: 10.1038/mt.2013.220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Krysko DV, Garg AD, Kaczmarek A, Krysko O, Agostinis P, Vandenabeele P. Immunogenic cell death and DAMPs in cancer therapy. Nat. Rev. Cancer. 2012;12(12):860–875. doi: 10.1038/nrc3380. [DOI] [PubMed] [Google Scholar]
- 137.Okura H, Smith C, Rutka J. Gene therapy for malignant glioma. Mol. Cell Ther. 2014;2(1):1–19. doi: 10.1186/2052-8426-2-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Oldfield EH, Ram Z, Culver KW, Blaese RM, Devroom HL, Anderson WF. Gene therapy for the treatment of brain tumors using intra-tumoral transduction with the thymidine kinase gene and intravenous ganciclovir. Hum. Gene Ther. 1993;4(1):39–69. doi: 10.1089/hum.1993.4.1-39. [DOI] [PubMed] [Google Scholar]
- 139.Puntel M, Muhammad AK, Candolfi M, et al. A novel bicistronic high-capacity gutless adenovirus vector that drives constitutive expression of herpes simplex virus type 1 thymidine kinase and tet-inducible expression of Flt3L for glioma therapeutics. J. Virol. 2010;84(12):6007–6017. doi: 10.1128/JVI.00398-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Grandi P, Peruzzi P, Reinhart B, Cohen JB, Chiocca EA, Glorioso JC. Design and application of oncolytic HSV vectors for glioblastoma therapy. Expert Rev. Neurother. 2009;9(4):505–517. doi: 10.1586/ern.09.9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Markert JM, Liechty PG, Wang W, et al. Phase Ib trial of mutant herpes simplex virus G207 inoculated pre-and post-tumor resection for recurrent GBM. Mol. Ther. J. Am. Soc. Gene Ther. 2009;17(1):199–207. doi: 10.1038/mt.2008.228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Markert JM, Medlock MD, Rabkin SD, et al. Conditionally replicating herpes simplex virus mutant, G207 for the treatment of malignant glioma: results of a Phase I trial. Gene Ther. 2000;7(10):867–874. doi: 10.1038/sj.gt.3301205. [DOI] [PubMed] [Google Scholar]
- 143.Kirn D, Hermiston T, Mccormick F. ONYX-015: clinical data are encouraging. Nat. Med. 1998;4(12):1341–1342. doi: 10.1038/3902. [DOI] [PubMed] [Google Scholar]
- 144.Chiocca EA, Abbed KM, Tatter S, et al. A Phase I open-label, dose-escalation, multi-institutional trial of injection with an E1B-attenuated adenovirus, ONYX-015, into the peritumoral region of recurrent malignant gliomas, in the adjuvant setting. Mol. Ther. J. Am. Soc. Gene Ther. 2004;10(5):958–966. doi: 10.1016/j.ymthe.2004.07.021. [DOI] [PubMed] [Google Scholar]
- 145.Fueyo J, Gomez-Manzano C, Alemany R, et al. A mutant oncolytic adenovirus targeting the Rb pathway produces anti-glioma effect in vivo . Oncogene. 2000;19(1):2–12. doi: 10.1038/sj.onc.1203251. [DOI] [PubMed] [Google Scholar]
- 146.Anderson BD, Nakamura T, Russell SJ, Peng KW. High CD46 receptor density determines preferential killing of tumor cells by oncolytic measles virus. Cancer Res. 2004;64(14):4919–4926. doi: 10.1158/0008-5472.CAN-04-0884. [DOI] [PubMed] [Google Scholar]
- 147.Wollmann G, Ozduman K, van den Pol AN. Oncolytic virus therapy for glioblastoma multiforme: concepts and candidates. Cancer J. 2012;18(1):69–81. doi: 10.1097/PPO.0b013e31824671c9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Allen C, Paraskevakou G, Iankov I, et al. Interleukin-13 displaying retargeted oncolytic measles virus strains have significant activity against gliomas with improved specificity. Mol. Ther. J. Am. Soc. Gene Ther. 2008;16(9):1556–1564. doi: 10.1038/mt.2008.152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Uchida H, Marzulli M, Nakano K, et al. Effective treatment of an orthotopic xenograft model of human glioblastoma using an EGFR-retargeted oncolytic herpes simplex virus. Mol. Ther. 2013;21(3):561–569. doi: 10.1038/mt.2012.211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Qin XQ, Beckham C, Brown JL, Lukashev M, Barsoum J. Human and mouse IFN-beta gene therapy exhibits different anti-tumor mechanisms in mouse models. Mol. Ther. 2001;4(4):356–364. doi: 10.1006/mthe.2001.0464. [DOI] [PubMed] [Google Scholar]
- 151.Qin XQ, Tao N, Dergay A, et al. Interferon-beta gene therapy inhibits tumor formation and causes regression of established tumors in immune-deficient mice. Proc. Natl Acad. Sci. USA. 1998;95(24):14411–14416. doi: 10.1073/pnas.95.24.14411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Mizuno M, Yoshida J. Effect of human interferon beta gene transfer upon human glioma, transplanted into nude mouse brain, involves induced natural killer cells. Cancer Immunol. Immunother. 1998;47(4):227–232. doi: 10.1007/s002620050525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Yoshida J, Mizuno M, Fujii M, et al. Human gene therapy for malignant gliomas (glioblastoma multiforme and anaplastic astrocytoma) by in vivo transduction with human interferon beta gene using cationic liposomes. Hum. Gene Ther. 2004;15(1):77–86. doi: 10.1089/10430340460732472. [DOI] [PubMed] [Google Scholar]
- 154.Chiocca EA, Smith KM, McKinney B, et al. A Phase I trial of Ad.hIFN-beta gene therapy for glioma. Mol. Ther. 2008;16(3):618–626. doi: 10.1038/sj.mt.6300396. [DOI] [PubMed] [Google Scholar]
- 155.Ehtesham M, Samoto K, Kabos P, et al. Treatment of intracranial glioma with in situ interferon-gamma and tumor necrosis factor-alpha gene transfer. Cancer Gene Ther. 2002;9(11):925–934. doi: 10.1038/sj.cgt.7700516. [DOI] [PubMed] [Google Scholar]
- 156.Chen B, Timiryasova TM, Andres ML, et al. Evaluation of combined vaccinia virus-mediated antitumor gene therapy with p53, IL-2, and IL-12 in a glioma model. Cancer Gene Ther. 2000;7(11):1437–1447. doi: 10.1038/sj.cgt.7700252. [DOI] [PubMed] [Google Scholar]
- 157.Maraskovsky E, Brasel K, Teepe M, et al. Dramatic increase in the numbers of functionally mature dendritic cells in Flt3 ligand-treated mice: multiple dendritic cell subpopulations identified. J. Exp. Med. 1996;184(5):1953–1962. doi: 10.1084/jem.184.5.1953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Ali S, Curtin JF, Zirger JM, et al. Inflammatory and anti-glioma effects of an adenovirus expressing human soluble Fms-like tyrosine kinase 3 ligand (hsFlt3L): treatment with hsFlt3L inhibits intracranial glioma progression. Mol. Ther. 2004;10(6):1071–1084. doi: 10.1016/j.ymthe.2004.08.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Ali S, King GD, Curtin JF, et al. Combined immunostimulation and conditional cytotoxic gene therapy provide long-term survival in a large glioma model. Cancer Res. 2005;65(16):7194–7204. doi: 10.1158/0008-5472.CAN-04-3434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.King GD, Muhammad AK, Larocque D, et al. Combined Flt3L/TK gene therapy induces immunological surveillance which mediates an immune response against a surrogate brain tumor neoantigen. Mol. Ther. 2011;19(10):1793–1801. doi: 10.1038/mt.2011.77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Candolfi M, Curtin JF, Yagiz K, et al. B cells are critical to T-cell-mediated antitumor immunity induced by a combined immune-stimulatory/conditionally cytotoxic therapy for glioblastoma. Neoplasia. 2011;13(10):947–960. doi: 10.1593/neo.11024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.King GD, Muhammad AK, Curtin JF, et al. Flt3L and TK gene therapy eradicate multifocal glioma in a syngeneic glioblastoma model. Neuro Oncol. 2008;10(1):19–31. doi: 10.1215/15228517-2007-045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Rock KL, Clark K. Analysis of the role of MHC class II presentation in the stimulation of cytotoxic T lymphocytes by antigens targeted into the exogenous antigen-MHC class I presentation pathway. J. Immunol. 1996;156(10):3721–3726. [PubMed] [Google Scholar]
- 164.Rock KL, Gamble S, Rothstein L. Presentation of exogenous antigen with class I major histocompatibility complex molecules. Science. 1990;249:918–921. doi: 10.1126/science.2392683. [DOI] [PubMed] [Google Scholar]
- 165.Gustafsson K, Junevik K, Werlenius O, Holmgren S, Karlsson-Parra A, Andersson P-O. Tumour-loaded α-type 1-polarized dendritic cells from patients with chronic lymphocytic leukaemia produce a superior NK-, NKT- and CD8+ T cell-attracting chemokine profile. Scand. J. Immunol. 2011;74:318–326. doi: 10.1111/j.1365-3083.2011.02580.x. [DOI] [PubMed] [Google Scholar]
- 166.Shimizu K, Fujii S-I. DC therapy induces long-term NK reactivity to tumors via host DC. Eur. J. Immunol. 2009;39:457–468. doi: 10.1002/eji.200838794. [DOI] [PubMed] [Google Scholar]
- 167.Lafferty KJ, Warren HS, Woolnough JA. A mediator acting as a costimulator for the development of cytotoxic responses in vitro . Adv. Exp. Med. Biol. 1979;114:497–501. doi: 10.1007/978-1-4615-9101-6_82. [DOI] [PubMed] [Google Scholar]
- 168.Capece D, Verzella D, Fischietti M, Zazzeroni F, Alesse E. Targeting costimulatory molecules to improve antitumor immunity. J. Biomed. Biotech. 2012;2012:1–17. doi: 10.1155/2012/926321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Joffre O, Nolte MA, Spörri R, Reis E Sousa C. Inflammatory signals in dendritic cell activation and the induction of adaptive immunity. Immunol. Rev. 2009;227:234–247. doi: 10.1111/j.1600-065X.2008.00718.x. [DOI] [PubMed] [Google Scholar]
- 170.Cohen PA, Koski GK, Czerniecki BJ, et al. STAT3- and STAT5-dependent pathways competitively regulate the pan-differentiation of CD34pos cells into tumor-competent dendritic cells. Blood. 2008;112:1832–1843. doi: 10.1182/blood-2007-12-130138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Mailliard RB, Wankowicz-Kalinska A, Cai Q, et al. Alpha-type-1 polarized dendritic cells: a novel immunization tool with optimized CTL-inducing activity. Cancer Res. 2004;64:5934–5937. doi: 10.1158/0008-5472.CAN-04-1261. [DOI] [PubMed] [Google Scholar]
- 172.Okada H. Brain tumor immunotherapy with type-1 polarizing strategies. Ann. NY Acad. Sci. 2009;1174:18–23. doi: 10.1111/j.1749-6632.2009.04932.x. [DOI] [PubMed] [Google Scholar]
- 173.Mineharu Y, Castro MG, Lowenstein PR, Sakai N, Miyamoto S. Dendritic cell-based immunotherapy for glioma: multiple regimens and implications in clinical trials. Neurol. Med. Chir. (Tokyo) 2013;53:741–754. doi: 10.2176/nmc.ra2013-0234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Ohlfest JR, Andersen BM, Litterman AJ, et al. Vaccine injection site matters: qualitative and quantitative defects in CD8 T cells primed as a function of proximity to the tumor in a murine glioma model. J. Immunol. 2013;190(2):613–620. doi: 10.4049/jimmunol.1201557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Yamanaka R, Zullo SA, Tanaka R, Ramsey J, Blaese M, Xanthopoulos KG. Induction of a therapeutic antitumor immunological response by intratumoral injection of genetically engineered Semliki Forest virus to produce interleukin-12. Neurosurg. Focus. 2000;9:e7. doi: 10.3171/foc.2000.9.6.8. [DOI] [PubMed] [Google Scholar]
- 176.Jenkins M, Schwartz R. Antigen presentation by chemically modified splenocytes induces antigen-specific T cell unresponsiveness in vitro and in vivo . J. Exp. Med. 1987;165:302–319. doi: 10.1084/jem.165.2.302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Zeppernick F, Ahmadi R, Campos B, et al. Stem cell marker CD133 affects clinical outcome in glioma patients. Clin. Cancer Res. 2008;14(1):123–129. doi: 10.1158/1078-0432.CCR-07-0932. [DOI] [PubMed] [Google Scholar]
- 178.Pallini R, Ricci-Vitiani L, Montano N, et al. Expression of the stem cell marker CD133 in recurrent glioblastoma and its value for prognosis. Cancer. 2011;117(1):162–174. doi: 10.1002/cncr.25581. [DOI] [PubMed] [Google Scholar]
- 179.Okada H, Kalinski P, Ueda R, et al. Induction of CD8+ T-cell responses against novel glioma-associated antigen peptides and clinical activity by vaccinations with {alpha}-type 1 polarized dendritic cells and polyinosinic-polycytidylic acid stabilized by lysine and carboxymethylcellulose in patients with recurrent malignant glioma. J. Clin. Oncol. 2011;29(3):330–336. doi: 10.1200/JCO.2010.30.7744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Esparza R, Azad TD, Feroze AH, Mitra SS, Cheshier SH. Glioblastoma stem cells and stem cell-targeting immunotherapies. J. Neurooncol. 2015;123(3):449–457. doi: 10.1007/s11060-015-1729-x. [DOI] [PubMed] [Google Scholar]
- 181.Gan HK, Kaye AH, Luwor RB. The EGFRvIII variant in glioblastoma multiforme. J. Clin. Neurosci. 2009;16(6):748–754. doi: 10.1016/j.jocn.2008.12.005. [DOI] [PubMed] [Google Scholar]
- 182.Heimberger AB, Suki D, Yang D, Shi W, Aldape K. The natural history of EGFR and EGFRvIII in glioblastoma patients. J. Transl. Med. 2005;3:38. doi: 10.1186/1479-5876-3-38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Heimberger AB, Crotty LE, Archer GE, et al. Epidermal growth factor receptor VIII peptide vaccination is efficacious against established intracerebral tumors. Clin. Cancer Res. 2003;9(11):4247–4254. [PubMed] [Google Scholar]
- 184.Mishima K, Johns TG, Luwor RB, et al. Growth suppression of intracranial xenografted glioblastomas overexpressing mutant epidermal growth factor receptors by systemic administration of monoclonal antibody (mAb) 806, a novel monoclonal antibody directed to the receptor. Cancer Res. 2001;61(14):5349–5354. [PubMed] [Google Scholar]
- 185.Babu R, Adamson DC. Rindopepimut: an evidence-based review of its therapeutic potential in the treatment of EGFRvIII-positive glioblastoma. Core Evid. 2012;7:93–103. doi: 10.2147/CE.S29001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Sampson JH, Heimberger AB, Archer GE, et al. Immunologic escape after prolonged progression-free survival with epidermal growth factor receptor variant III peptide vaccination in patients with newly diagnosed glioblastoma. J. Clin. Oncol. 2010;28(31):4722–4729. doi: 10.1200/JCO.2010.28.6963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Reardon DA, Wucherpfennig KW, Freeman G, et al. An update on vaccine therapy and other immunotherapeutic approaches for glioblastoma. Expert Rev. Vaccines. 2013;12(6):597–615. doi: 10.1586/erv.13.41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Caudill MM, Li Z. HSPPC-96: a personalised cancer vaccine. Expert Opin. Biol. Ther. 2001;1(3):539–547. doi: 10.1517/14712598.1.3.539. [DOI] [PubMed] [Google Scholar]
- 189.Binder RJ, Srivastava PK. Essential role of CD91 in re-presentation of gp96-chaperoned peptides. Proc. Natl Acad. Sci. USA. 2004;101(16):6128–6133. doi: 10.1073/pnas.0308180101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Chandramohan V, Mitchell DA, Johnson LA, Sampson JH, Bigner DD. Antibody, T-cell and dendritic cell immunotherapy for malignant brain tumors. Future Oncol. 2013;9(7):977–990. doi: 10.2217/fon.13.47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Jouanneau E, Black KL, Veiga L, et al. Intrinsically de-sialylated CD103(+) CD8 T cells mediate beneficial anti-glioma immune responses. Cancer Immunol. Immunother. 2014;63(9):911–924. doi: 10.1007/s00262-014-1559-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Ikeda H, Shiku H. Antigen-receptor gene-modified T cells for treatment of glioma. Adv. Exp. Med. Biol. 2012;746:202–215. doi: 10.1007/978-1-4614-3146-6_16. [DOI] [PubMed] [Google Scholar]
- 193.Chekmasova AA, Rao TD, Nikhamin Y, et al. Successful eradication of established peritoneal ovarian tumors in SCID-Beige mice following adoptive transfer of T cells genetically targeted to the MUC16 antigen. Clin. Cancer Res. 2010;16(14):3594–3606. doi: 10.1158/1078-0432.CCR-10-0192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Kochenderfer JN, Yu Z, Frasheri D, Restifo NP, Rosenberg SA. Adoptive transfer of syngeneic T cells transduced with a chimeric antigen receptor that recognizes murine CD19 can eradicate lymphoma and normal B cells. Blood. 2010;116(19):3875–3886. doi: 10.1182/blood-2010-01-265041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Zhao Y, Wang QJ, Yang S, et al. A herceptin-based chimeric antigen receptor with modified signaling domains leads to enhanced survival of transduced T lymphocytes and antitumor activity. J. Immunol. 2009;183(9):5563–5574. doi: 10.4049/jimmunol.0900447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Mintz A, Gibo DM, Slagle-Webb B, Christensen ND, Debinski W. IL-13Ralpha2 is a glioma-restricted receptor for interleukin-13. Neoplasia. 2002;4(5):388–399. doi: 10.1038/sj.neo.7900234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Liu G, Ying H, Zeng G, Wheeler CJ, Black KL, Yu JS. HER-2, gp100, and MAGE-1 are expressed in human glioblastoma and recognized by cytotoxic T cells. Cancer Res. 2004;64(14):4980–4986. doi: 10.1158/0008-5472.CAN-03-3504. [DOI] [PubMed] [Google Scholar]
- 198.Ammi R, de Waele J, Willemen Y, et al. Poly(I:C) as cancer vaccine adjuvant: knocking on the door of medical breakthroughs. Pharmacol. Ther. 2015;146:120–131. doi: 10.1016/j.pharmthera.2014.09.010. [DOI] [PubMed] [Google Scholar]
- 199.Black PL, Hartmann D, Pennington R, et al. Effect of tumor burden and route of administration on the immunotherapeutic properties of polyinosinic-polycytidylic acid stabilized with poly-L-lysine in carboxymethyl cellulose [Poly(I,C)-LC] Int. J. Immunopharmacol. 1992;14(8):1341–1353. doi: 10.1016/0192-0561(92)90005-6. [DOI] [PubMed] [Google Scholar]
- 200.Ursu R, Carpentier AF. Immunotherapeutic approach with oligodeoxynucleotides containing CpG motifs (CpG-ODN) in malignant glioma. Adv. Exp. Med. Biol. 2012;746:95–108. doi: 10.1007/978-1-4614-3146-6_8. [DOI] [PubMed] [Google Scholar]
- 201.Gaspari AA, Tyring SK, Rosen T. Beyond a decade of 5% imiquimod topical therapy. J. Drugs Dermatol. 2009;8(5):467–474. [PubMed] [Google Scholar]
- 202.Xiong Z, Ohlfest JR. Topical imiquimod has therapeutic and immunomodulatory effects against intracranial tumors. J. Immunother. 2011;34(3):264–269. doi: 10.1097/CJI.0b013e318209eed4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Rosenfeld MR, Chamberlain MC, Grossman SA, et al. A multi-institution Phase II study of poly-ICLC and radiotherapy with concurrent and adjuvant temozolomide in adults with newly diagnosed glioblastoma. Neuro Oncol. 2010;12(10):1071–1077. doi: 10.1093/neuonc/noq071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Carpentier A, Metellus P, Ursu R, et al. Intracerebral administration of CpG oligonucleotide for patients with recurrent glioblastoma: a Phase II study. Neuro Oncol. 2010;12(4):401–408. doi: 10.1093/neuonc/nop047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Wolchok JD, Hoos A, O'Day S, et al. Guidelines for the evaluation of immune therapy activity in solid tumors: immune-related response criteria. Clin. Cancer Res. 2009;15(23):7412–7420. doi: 10.1158/1078-0432.CCR-09-1624. [DOI] [PubMed] [Google Scholar]
- 206.Reardon DA, Okada H. Re-defining response and treatment effects for neuro-oncology immunotherapy clinical trials. J. Neurooncol. 2015;123(3):339–346. doi: 10.1007/s11060-015-1748-7. [DOI] [PubMed] [Google Scholar]
- 207.Sims JS, Ung TH, Neira JA, Canoll P, Bruce JN. Biomarkers for glioma immunotherapy: the next generation. J. Neurooncol. 2015;123(3):359–372. doi: 10.1007/s11060-015-1746-9. [DOI] [PMC free article] [PubMed] [Google Scholar]