Abstract
Vitamin D deficiency has been associated with increased risk of prostate cancer (PC) in epidemiologic and prospective studies. An association has also been made between high dietary calcium and increased PC risk. In this study, we evaluated the effect of dietary vitamin D and calcium on the growth of human androgen-insensitive prostate tumor in an athymic mouse model. We observed highest tumor growth in the normal calcium - vitamin D-deficient group, while tumor growth between the normal calcium - vitamin D-sufficient, high calcium - vitamin D-sufficient and high calcium - vitamin D-deficient diet-groups did not significantly differ but was significantly lower than that in the normal calcium - vitamin D-deficient group. Our results suggest an important role of dietary vitamin D as a preventive agent in androgen-insensitive PC.
Keywords: Androgen-insensitive prostate cancer, vitamin D, calcium, mouse tumor model, DU-145 cells
Prostate Cancer (PC) is the most prevalent cancer among men, and the second leading cause of cancer death among men in the US (1). Although PC occurs more often in elderly men, the incidence of prostatic carcinoma is also significant among young men. Thus, as the average life expectancy continues to increase and as the elderly population grows, there is a greater need for effective preventative measures, such as dietary intervention, as well as nutritional strategies for PC, particularly androgen-insensitive and metastatic PC.
Vitamin D (vitamin D2 of plant origin and vitamin D3 of animal origin) is a naturally occurring molecule that is biosynthesized in the skin. In 1989, Garland et al. carried out an eight-year prospective study among 25,620 healthy adults to demonstrate that if the initial level of serum 25-hydroxyvitamin D is at least 20 ng/ml, there is a 50% reduced risk of developing colon cancer (2). Since this observation, other investigators have confirmed the effect of latitude and vitamin D intake on reducing risk of various types of cancer, including malignancy of the prostate (3–9).
Several epidemiological and dietary studies have suggested a correlation between calcium intake and PC risk. Because high levels of calcium reduce the renal production of 1,25-dihydroxyvitamin D (1,25(OH)2D, the dihydroxy metabolite of vitamin D, the active form of the vitamin D hormone) it was suggested that high calcium levels may diminish the beneficial effect of vitamin D on some types of cancer including PC (10, 11). Bao et al. hypothesized that vitamin D has an antioxidant effect via up-regulation of glucose-6-phosphate-dehydrogenase and glutathione levels (12). Guzey et al. found that vitamin D up-regulates pro-apoptotic genes in PC cells (13). Moreno et al. observed that vitamin D reduces cyclooxygenase-2 and this inhibition leads to a reduction of angiogenesis (14). Based on these studies, it was concluded that decreased production of 1,25(OH)2D in a high calcium diet and/or its higher rate of catabolism may contribute significantly towards PC risk. Thus it was suggested that high intake of milk and dairy products may increase the risk of developing PC (15, 16).
In contrast, Huncharek et al. carried out a meta-analysis of 45 observational studies and showed that there is no correlation between dairy, vitamin D, calcium intake and PC risks (17). Tseng et al. also failed to find significant correlation between vitamin D intake (dietary and supplemental) and PC, although a weak but significant correlation with low-fat milk intake and PC was observed in the same study (18). In another study, an association between calcium intake and PC risk was observed in cases where calcium intake exceeded 2,000 mg/day (19). In contrast, Allen et al., in a large prospective study consisting of 2,727 cases of PC, found a positive correlation between increase in PC incidence with an increased intake of calcium from dairy products, but not calcium from other foods (20). But Park et al. examined over 82,000 men via a detailed food frequency questionnaire and found no correlation between calcium intake and vitamin D levels and PC risk (21).
In essence, careful review of the available literature suggests that the relationship between increased calcium intake and an increased risk of PC is rather inconclusive. In order to address this controversy we chose an athymic mouse tumor model of human androgen-insensitive PC to study the effect of dietary vitamin D and calcium on the growth of PC. Results of this study and their implications in the dietary intake of calcium and vitamin D in the growth of prostate tumor, and possibly its prevention are discussed here.
Materials and Methods
Diets
Specialized animal diets were purchased from Dyets, Inc. (Bethlehem, PA, USA) with defined compositions (normal calcium, NCa2+; high calcium, HCa2+; vitamin D, D): I, NCa2+ +D: D (5000 IU/kg), Ca2+ (0.9%), phosphorus (P, 0.7%), II, NCa2+-D: D (none), Ca2+ (0.9%), P (0.7%), III, HCa2+ +D: D (5000 IU/kg), Ca2+ (2.0%), P (0.7%), and IV, HCa2+ -D: D (none), Ca2+ (2.0%), P (1.25%).
Animals
Six week-old, male nu/nu athymic mice, weighing approximately 30 g (Charles River Laboratories Inc., Wilmington, MA, USA) were maintained under standard light and dark cycles with the specialized animal chow and water ad libitum unless specified otherwise. Approval from the Animal Safety Board of Boston University School of Medicine was obtained prior to animal experiments, which were carried out with strict adherence to established animal safety rules.
Establishment of diets in mice
Fifty-two mice were randomized into four groups of 13 each. Animals in each group were maintained on one of the specific diets listed above. Four weeks after the introduction of the custom diets, blood samples from all the mice were obtained via mandibular vein and sera were produced. The sera samples were subjected to 25-hydroxyvitamin D [25(OH)D] analysis by a method established in our laboratory (22).
Cell culture and implantation of tumor
DU-145 cells (ATCC, Manasas, VA, USA) were grown in Dulbecco’s Modified Eagle Medium (DMEM) with 10% fetal bovine serum and other additives (according to manufacturer’s specification) until approximately 80% confluency. They were trypsinized, washed with phosphate saline buffer (PBS), re-suspended in PBS and counted in a hemocytometer. The cell density of the suspension was adjusted by re-suspension in equal parts of PBS and matrigel (BD Biosciences, Franklin Lakes, NJ, USA) so that there were approximately 4×106 cells/100 μl of suspension. The cell suspension was injected under the skin (right flank) of the animals with a 25-gauge hypodermic needle. Animals were observed daily for tumor growth.
Tumor measurements
Tumor volume was measured with a caliper using the formula: volume=length (longest measurement)/2 × length × width. Measurement started when the tumor size reached approximately 3.5 mm3 (approximately 29 days post-injection). Body weights and tumor measurements were made three times a week until the 76 day post injection, when mice were sacrificed and blood samples were collected via cardiac puncture and sera made and stored to be assayed for calcium (QuantiChrom Calcium Assay Kit, BioAssays Systems, Hayward, CA, USA) and 25(OH)D (22).
Results and Discussion
A link between PC risk and dietary intake of calcium and vitamin D is confusing and inconclusive, as discussed earlier. Therefore, we set out to determine the effect of dietary calcium and vitamin D in a mouse model of human androgen-insensitive PC. Androgens have been positively implicated in the growth and progression of prostate tumor. As a result, anti-hormone treatment is one of the first-line therapies of prostate cancer. But, within a relatively short period of time, hormone-sensitive tumors become hormone insensitive. Therefore, we hypothesized that an androgen-insensitive PC model would be most appropriate to study the effect of dietary calcium and vitamin D on tumor growth, because in general, the effect of a dietary component needs a long time to take hold, mirroring slow development on prostate tumor and eventual androgen insensitivity.
In our studies, we first established a significant decline in the vitamin D status of animals fed vitamin D-deficient diets prior to implantation of tumor by measuring serum 25(OH)D levels. The serum 25(OH)D level in NCa2++D group (38.3±4.8 ng/ml) is significantly higher (p<0.05) than its vitamin D-deficient counterpart, NCa2+ -D (10.7±1.3 ng/ml). The serum 25(OH)D level of the HCa2+ +D group (27.0± 4.0 ng/ml) was also significantly higher (p<0.05) than its vitamin D-deficient counterpart (HCa2+ -D=10.6±3.7 ng/ml). These results established that the mice fed a vitamin D deficient diet had a low serum 25(OH)D level, consistent with vitamin D deficiency, i.e. <20 ng/ml (23).
At the termination of the study, mice fed the normal calcium-vitamin D-deficient diet had an undetectable level of serum 25(OH)D (<5 ng/ml), while the corresponding vitamin D-sufficient group (NCa2+ +D) had an average serum 25(OH)D value of 28.07±2.2 ng/ml. A similar trend existed in the high calcium diet groups (HCa2+ +D=18.67± 2.2 ng/ml vs. HCa2+ –D=7.13±0.5 ng/ml). Our laboratory reported that LNCaP prostate cancer cells, transfected with 1,25-dihydroxyvitamin D-1α-hydroxylase are capable of converting 25(OH)D3 to 1,25(OH)2D3, bypassing the renal route (24). This is a probable explanation, why mice fed the HCa2+ –D diet still had a detectable level of 25(OH)D at the end of the study.
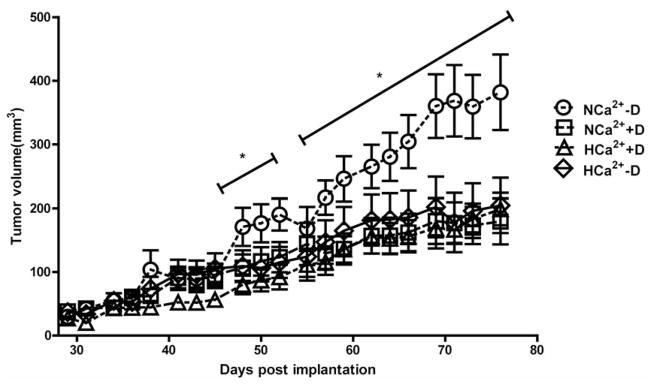
Tumors from the NCa2+ –D group had an average volume of 381±59 mm3 compared to those of the NCa2+ +D group, which had an average volume of 179±36 mm3. Tumor volumes of HCa2++D and HCa2+ -D diet groups averaged 199±25 mm3 and 204±43 mm3, respectively. Overall, tumor volume from the NCa2+ -D group was significantly higher (p<0.05) than the tumor volumes of three other groups, which were not significantly different from one another.
Vitamin D is biologically inert, and it undergoes stepwise metabolic activation, first to 25(OH)D in the liver, and then to 1,25(OH)2D in the kidney. 1,25(OH)2D is the most active vitamin D metabolite in terms of inhibiting the growth of tumor cells in vitro, as well as reducing tumor growth in vivo (25). Therefore, the tumor inhibitory effect of dietary vitamin D is most likely due to its conversion to 1,25(OH)2D. In our study, the NCa2+ –D group, deprived of the beneficial effect of vitamin D in inhibiting tumor growth, showed highest tumor growth (Figure 1). Recent evidence suggests that calcium, particularly in high concentration, may cause cytotoxicity via necrosis or apoptosis (26). Therefore, in mice fed with the high calcium diet with or without vitamin D (HCa2+ +D, HCa2+ -D) cell growth inhibitory effect of high calcium may have primarily contributed to the reduction of tumor growth, because high calcium is known to reduce the renal production of 1,25(OH)2D.
Figure 1.
Tumor growth as a function of time. Starting at day 48, the NCa2+ –D group had a significantly larger tumor volume compared to the other groups (*p<0.05) and this trend continued until the end of the experiment, when this group had the highest tumor volume compared to all other groups.
We chose androgen-insensitive DU-145 tumor as our model for reasons specified earlier. However, several studies have demonstrated that DU-145 cells are unresponsive towards 1,25(OH)2D treatment due to up-regulation of 1,25-dihydroxyvitamin D-24-hydroxylase, the enzyme responsible for initiating the catabolic degradation of 1,25(OH)2D andreducing its half-life (27, 28). Results delineated in this communication raise an interesting possibility that increased production of this enzyme observed in cell-culture models may not be operative in vivo.
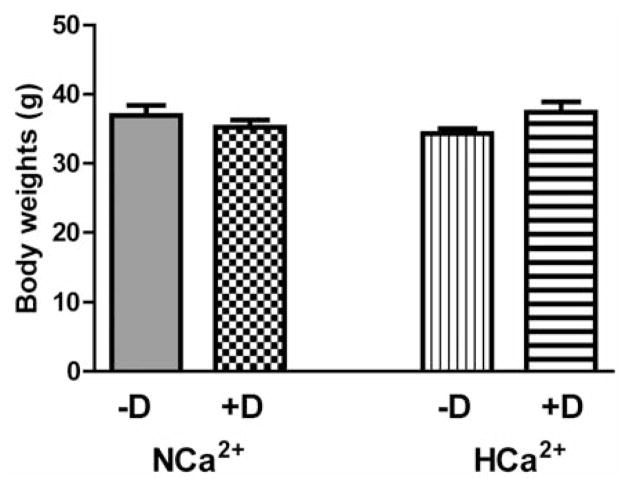
A potential problem with the chemopreventive use of vitamin D includes its calcemic toxicity either via a cumulative path due to repeated/daily use at high doses, or a very large bolus due to accidental misuse (29, 30). Such toxicity is often reflected in the rise of the serum calcium level, and/or a negative impact on body weight. At the termination of our study, HCa2+ –D, HCa2+ +D and NCa2+ +D groups had serum calcium levels of 12.7±.4 mg/dl, 12.0±0.9 and 12.6±1.1 mg/dl, respectively, slightly above the normal limit of 10–12 mg/dl. However, calcium level in the NCa2+ –D group was 9.4±1.0 mg/ml (p<0.001) which was significantly lower than in other groups. As another indicator of the lack of toxicity, weights of the animals at the end of the experiment showed no significant difference among the groups (Figure 2).
Figure 2.
The weights of the mice at the end of the experiment. There was no significant difference among the groups (n=10).
1,25(OH)2D has been shown to act through p21CIP/WAF and p27 pathways resulting in the inhibition of G1/S phase transition in the cell cycle (31–33). It has also been shown to induce p21 in a p53-dependent manner, that results in G0/G1 senescence in a variety of cell types, e.g. breast, prostate, kidney (34–36). On the other hand, growth inhibition of tumor cells by 1,25(OH)2D has been shown to involve apoptosis, and modulation of BCL pathway markers (37). However, gene analysis for p21, p27, p53, BAX and BCL-2 in tumor samples as well as immunohistochemical analysis of tumor slices did not show any significant difference among the groups. Therefore, molecular basis for the results of our diet study remains an open question.
In summary, results of our study suggest that a normal calcium, vitamin D-deficient diet may be detrimental towards the growth of androgen-insensitive prostate tumor, further emphasizing the role of dietary vitamin D in the prevention of PC.
Acknowledgments
Elizabeth Genova is acknowledged for technical assistance. This work was supported by a grant from the National Dairy Council (DMI 1253).
References
- 1.Howlader N, Noone AM, Krapcho M, Neyman N, Aminou R, Waldron W, Altekruse SF, Kosary CL, Ruhl J, Tatalovich Z, Cho H, Mariotto A, Eisner MP, Lewis DR, Chen HS, Feuer EJ, Cronin KA, Edwards BK, editors. SEER Cancer Statistics Review, 1975–2008. National Cancer Institute; Bethesda, MD: 2011. http://seer.cancer.gov/csr/1975_2008/, based on November 2010 SEER data submission, posted to the SEER web site. [Google Scholar]
- 2.Garland CF, Garland FC, Shaw EK, Comstock GW, Helsing KJ, Gorham ED. Serum 25-hydroxyvitamin D and colon cancer: Eight-year prospective study. Lancet. 1989;18:1176–1178. doi: 10.1016/s0140-6736(89)91789-3. [DOI] [PubMed] [Google Scholar]
- 3.Garland CF, Garland FC, Gorham ED, Raffa J, editors. Sunlight, vitamin D, and mortality from breast and colorectal cancer in Italy: Biologic Effects of Light. New York: Walter de Gruyter & Co; 1992. pp. 39–43. [Google Scholar]
- 4.Garland FC, Garland CF, Gorham ED, Young JF. Geographic variation in breast cancer mortality in the United States: A hypothesis involving exposure to solar radiation. Preventive Med. 1990;19:614–622. doi: 10.1016/0091-7435(90)90058-r. [DOI] [PubMed] [Google Scholar]
- 5.Hanchette CL, Schwartz GG. Geographic patterns of prostate cancer mortality. Cancer. 1992;70:2861–2869. doi: 10.1002/1097-0142(19921215)70:12<2861::aid-cncr2820701224>3.0.co;2-g. [DOI] [PubMed] [Google Scholar]
- 6.Holick MF. Vitamin D: The underappreciated D-lightful hormone that is important for skeletal and cellular health. Curr Opin Endocrinol Diab. 2002;9:87–98. [Google Scholar]
- 7.Giovannucci E, Liu Y, Rimm EB, Hollis BW, Fuchs CS, Stampfer MJ, Willett WC. Prospective study of predictors of vitamin D status and cancer incidence and mortality in men. J Natl Cancer Inst. 2006;98:451–459. doi: 10.1093/jnci/djj101. [DOI] [PubMed] [Google Scholar]
- 8.Giovannucci E, Liu Y, Stampfer MJ, Willett WC. A prospective study of calcium intake and incident and fatal prostate cancer. Cancer Epidemiol Biomarkers Prev. 2006;15:203–210. doi: 10.1158/1055-9965.EPI-05-0586. [DOI] [PubMed] [Google Scholar]
- 9.Wei EK, Giovannucci E, Fuchs CS, Willett WC, Mantzoros CS. Low plasma adiponectin levels and risk of colorectal cancer in men: a prospective study. J Natl Cancer Inst. 2005;97:1688–1694. doi: 10.1093/jnci/dji376. [DOI] [PubMed] [Google Scholar]
- 10.Bonjour JP, Chevalley T, Farellone P. Calcium intake and vitamin D metabolism and action, in healthy conditions and in prostate cancer. Br J Nutr. 2007;97:611–616. doi: 10.1017/S0007114507657961. [DOI] [PubMed] [Google Scholar]
- 11.Lou YR, Qiao S, Talonpoika R, Syvälä H, Tuohimaa P. The role of Vitamin D3 metabolism in prostate cancer. J Steroid Biochem Mol Biol. 2004;92:317–325. doi: 10.1016/j.jsbmb.2004.10.007. [DOI] [PubMed] [Google Scholar]
- 12.Bao BY, Ting HJ, Hsu JW, Lee YF. Protective role of 1 alpha, 25-dihydroxyvitamin D3 against oxidative stress in nonmalignant human prostate epithelial cells. Int J Cancer. 2008;122:2699–2706. doi: 10.1002/ijc.23460. [DOI] [PubMed] [Google Scholar]
- 13.Guzey M, Kitada S, Reed JC. Apoptosis induction by 1alpha,25-dihydroxyvitamin D3 in prostate cancer. Mol Cancer Ther. 2002;1:667–677. [PubMed] [Google Scholar]
- 14.Moreno J, Krishnan AV, Feldeman D. Molecular mechanisms mediating the anti-proliferative effects of vitamin D in prostate cancer. J Steroid Biochem Biol. 2005;97:31–36. doi: 10.1016/j.jsbmb.2005.06.012. [DOI] [PubMed] [Google Scholar]
- 15.Rodriguez C, McCullough ML, Mondul AM, Jacobs EJ, Fakhrabadi-Shokoohi D, Giovannucci EL, Thun MJ, Calle EE. Calcium, dairy products, and risk of prostate cancer in a prospective cohort of United States men. Cancer Epidemiol Biomarkers Prev. 2003;12:597–603. [PubMed] [Google Scholar]
- 16.Tavani A, Bertuccio P, Bosetti C, Talamini R, Negri E, Franceschi S, Montella M, La Vecchia C. Dietary intake of calcium, vitamin D, phosphorus and the risk of prostate cancer. Eur Urol. 2005;48:27–33. doi: 10.1016/j.eururo.2005.03.023. [DOI] [PubMed] [Google Scholar]
- 17.Huncharek M, Muscat J, Kupelnick B. Dairy products, dietary calcium and vitamin d intake as risk factors for prostate cancer: a meta-analysis of 26,769 cases from 45 observational studies. Nutr Cancer. 2008;60:421–441. doi: 10.1080/01635580801911779. [DOI] [PubMed] [Google Scholar]
- 18.Tseng M, Breslow RA, Graubard BI, Ziegler RG. Dairy, calcium, and vitamin D intakes and prostate cancer risk in the National Health and Nutrition Examination Epidemiologic Follow-up Study cohort. Am J Clin Nutr. 2005;81:1147–1154. doi: 10.1093/ajcn/81.5.1147. [DOI] [PubMed] [Google Scholar]
- 19.Ahn J, Albanes D, Peters U, Schatzkin A, Lim U, Freedman M, Chatterjee N, Andriole GL, Leitzmann MF, Hayes RB. Prostate, Lung, Colorectal, and Ovarian Trial Project Team. Dairy products, calcium intake, and risk of prostate cancer in the prostate, lung, colorectal, and ovarian cancer screening trial. Cancer Epidemiol Biomarkers Prev. 2007;16:2623–2630. doi: 10.1158/1055-9965.EPI-07-0601. [DOI] [PubMed] [Google Scholar]
- 20.Allen NE, Key TJ, Appleby PN, Travis RC, Roddam AW, Tjønneland A, Johnsen NF, Overvad K, Linseisen J, Rohrmann S, Boeing H, Pischon T, Bueno-de-Mesquita HB, Kiemeney L, Tagliabue G, Palli D, Vineis P, Tumino R, Trichopoulou A, Kassapa C, Trichopoulos D, Ardanaz E, Larrañaga N, Tormo MJ, González CA, Quirós JR, Sánchez MJ, Bingham S, Khaw KT, Manjer J, Berglund G, Stattin P, Hallmans G, Slimani N, Ferrari P, Rinaldi S, Riboli E. Animal foods, protein, calcium and prostate cancer risk: the European Prospective Investigation into Cancer and Nutrition. Br J Cancer. 2008;98:1574–1581. doi: 10.1038/sj.bjc.6604331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Park SY, Murphy SP, Wilkens LR, Stram DO, Henderson BE, Kolonel LN. Calcium, vitamin D, and dairy product intake and prostate cancer risk: the Multiethnic Cohort Study. Am J Epidemiol. 2007;166:1259–1269. doi: 10.1093/aje/kwm269. [DOI] [PubMed] [Google Scholar]
- 22.Chen TC, Turner AK, Holick MF. Methods for the determination of the circulating concentration of 25-hydroxyvitamin D. J Nutr Biochem. 1990;1:315–319. doi: 10.1016/0955-2863(90)90067-u. [DOI] [PubMed] [Google Scholar]
- 23.Holick MF, Binkley NC, Bischoff-Ferrari HA, Gordon CM, Hanley DA, Heaney RP, Murad MH, Weaver CM. Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96:1911–1930. doi: 10.1210/jc.2011-0385. [DOI] [PubMed] [Google Scholar]
- 24.Whitlatch LW, Young MV, Schwartz GG, Flanagan JN, Burnstein KL, Lokeshwar BL, Rich ES, Holick MF, Chen TC. 25-Hydroxyvitamin D-1alpha-hydroxylase activity is diminished in human prostate cancer cells and is enhanced by gene transfer. J Steroid Biochem Mol Biol. 2002;81:135–140. doi: 10.1016/s0960-0760(02)00053-5. [DOI] [PubMed] [Google Scholar]
- 25.Bouillon R, Carmeliet G, Verlinden L, van Etten E, Verstuyf A, Luderer HF, Lieben L, Mathieu C, Demay M. Vitamin D and human health: lessons from vitamin D receptor null mice. Endocr Rev. 2008;6:726–776. doi: 10.1210/er.2008-0004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Orrenius S, Zhivotovsky B, Nicotera P. Regulation of cell death: the calcium-apoptosis link. Nat Rev Mol Cell Biol. 2003;4:552–565. doi: 10.1038/nrm1150. [DOI] [PubMed] [Google Scholar]
- 27.Ly LH, Zhao XY, Holloway L, Feldman D. Liarozole acts synergistically with 1alpha,25-dihydroxyvitamin D3 to inhibit growth of DU-145 human prostate cancer cells by blocking 24-hydroxylase activity. Endocrinol. 1999;140:2071–2076. doi: 10.1210/endo.140.5.6698. [DOI] [PubMed] [Google Scholar]
- 28.Krishnan AV, Peehl DM, Feldman D. Inhibition of prostate cancer growth by vitamin D: Regulation of target gene expression. J Cell Biochem. 2003;88:363–371. doi: 10.1002/jcb.10334. [DOI] [PubMed] [Google Scholar]
- 29.Querfeld U, Mak RH. Vitamin D deficiency and toxicity in chronic kidney disease: in search of the therapeutic window. Pediatr Nephrol. 2010;25:2413–2430. doi: 10.1007/s00467-010-1574-2. [DOI] [PubMed] [Google Scholar]
- 30.Heaney RP, Vieth R, Hollis BW. Vitamin D efficacy and safety. Arch Intern Med. 2011;171:266. doi: 10.1001/archinternmed.2010.528. [DOI] [PubMed] [Google Scholar]
- 31.Zhuang SH, Burnstein KL. Antiproliferative effect of 1alpha,25-dihydroxyvitamin D3 in human prostate cancer cell line LNCaP involves reduction of cyclin-dependent kinase 2 activity and persistent G1 accumulation. Endocrinol. 1998;139:1197–1207. doi: 10.1210/endo.139.3.5770. [DOI] [PubMed] [Google Scholar]
- 32.Polek TC, Stewart LV, Ryu EJ, Cohen MB, Allegretto EA, Weigel NL. p53 Is required for 1,25-dihydroxyvitamin D3-induced G0 arrest but is not required for G1 accumulation or apoptosis of LNCaP prostate cancer cells. Endocrinol. 2003;144:50–60. doi: 10.1210/en.2001-210109. [DOI] [PubMed] [Google Scholar]
- 33.Verlinden L, Verstuyf A, Convents R, Marcelis S, Van Camp M, Bouillon R. Action of 1,25(OH)2D3 on the cell cycle genes, cyclin D1, p21 and p27 in MCF-7 cells. Mol Cell Endocrinol. 1998;142:57–65. doi: 10.1016/s0303-7207(98)00117-8. [DOI] [PubMed] [Google Scholar]
- 34.Blutt SE, McDonnell TJ, Polek TC, Weigel N. Calcitriol-induced apoptosis in LNCaP cells is blocked by overexpression of BCL-2. Endocrinol. 2000;141:10–17. doi: 10.1210/endo.141.1.7289. [DOI] [PubMed] [Google Scholar]