Abstract
Purpose
Diminished ovarian reserve (DOR) is characterized by poor fertility outcomes, and it represents a major challenge in reproductive medicine. Although consensus exists on the concept of DOR, its definition remains blurry. DOR has to be distinguished from premature ovarian failure (POF) and poor ovarian responders (POR), who are clearly defined.
Methods
We performed a PubMed search with the terms “diminished ovarian reserve” and “in vitro fertilization (IVF)” to assess the homogeneity of the definition of DOR.
Results
Out of 121 articles, 14 gave a definition for DOR. Only one definition was used by two different teams (basal follicle-stimulating hormone (FSH) value >10 IU/l) and eight teams used 11 different definitions. Among those, four definitions did not include antral follicular count (AFC) and seven studies did. Two definitions included the results from a previous cycle.
Conclusions
The heterogeneity in the definition of DOR used in these studies contributes to confusing results. Hence, there is a need for a clear definition of DOR. It appears that AFC and anti-Müllerian hormone (AMH) serum levels are the most relevant criteria. One option could be the use of the following definition: (i) woman with any of the risk factors for POR and/or (ii) an abnormal ovarian reserve test (i.e., antral follicular count (AFC) <5–7 follicles or AMH <0.5–1.1 ng/ml). This hypothesis requires validation.
Keywords: Diminished ovarian reserve, IVF, AMH, AFC
Diminished ovarian reserve (DOR) is characterized by poor fertility outcomes even when assisted reproductive techniques (ART) are used and represents a major challenge in reproductive medicine. Although consensus exists on the concept of DOR, its definition remains blurry.
It is important to distinguish DOR from premature ovarian failure (POF) and poor ovarian responders (POR), the latter two having been clearly defined.
POF, also called premature menopause or primary ovarian insufficiency, is related to cessation of ovarian function before the age of 40. It is defined by a triad of signs: (i) amenorrhea for at least 4 months, (ii) decreased estradiol serum concentrations, and (iii) elevated follicle-stimulating hormone (FSH) serum concentrations (more than 40 IU/l in at least two samples a few weeks apart) [1, 2]. POF-related infertility cannot be treated by intraconjugal assisted reproductive techniques.
POR, on the other hand, is a condition that seems closer to DOR. The Bologna ESHRE consensus defines women as “poor ovarian responders” when at least two of the following three characteristics are present: (i) advanced maternal age (≥40 years) or any of the risk factors for POR, (ii) a previous poor ovarian response (≤3 oocytes with a conventional stimulation protocol), and (iii) an abnormal ovarian reserve test (i.e., antral follicular count (AFC) <5–7 follicles or AMH <0.5–1.1 ng/ml) [3]. Consequently, to be included in this definition, a woman must be over 40 years or have previously undergone at least one controlled ovarian hyperstimulation cycle. Thus, a young infertile woman with markers of poor ovarian reserve who has never undergone ART does not meet the Bologna criteria.
Identification of women with DOR, prior to a controlled ovarian stimulation, would enable more individualization of treatment and protocol selection as well as better information of couples on the chances of pregnancy. In daily practice, clinicians have gained experience from ART in the “older woman” (>38) and have extrapolated results to treat DOR-associated infertility. In “older women”, in vitro fertilization (IVF) allows better pregnancy rates than intrauterine insemination (IUI) [4–6]. Indeed, FORT-T trial has demonstrated that IVF offered higher pregnancy rate than IUI in patients aged between 38 and 42 years without other infertility factors than age.
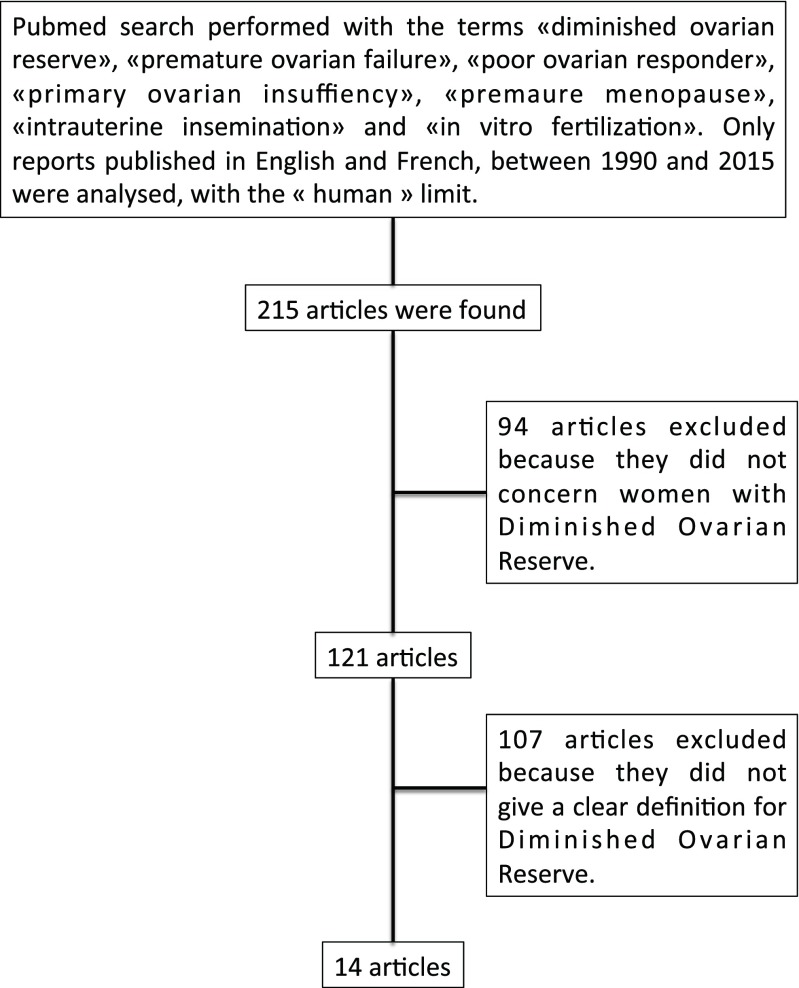
We performed a PubMed search with the terms “diminished ovarian reserve,” “premature ovarian failure,” “poor ovarian responder,” “primary ovarian insufficiency,” “premature menopause,” “intrauterine insemination,” and “in vitro fertilization (IVF)” with the “human” limit, to assess the homogeneity of the definition of DOR. Only reports published in English and in French from 1990 to 2015 were analyzed. Out of the 215 articles found, we excluded those not specifically concerning women with DOR (based on title or abstract). Out of the 121 remaining articles, we excluded those not giving a clear definition of DOR (after reading the full text). After selection, we found 14 articles that gave a definition for DOR (Fig. 1).
Fig. 1.
Flow chart of the search methodology
Only one definition was used by two different teams:
Eight teams used 11 different definitions, including various cutoffs for markers, and heterogeneous combinations of markers:
- Among those, four definitions did not include AFC:
- Basal FSH value >10 IU/l or an FSH/luteinizing hormone (LH) ratio >3 [11].
- Abnormal FSH and/or AMH levels, outside of the 95 % confidence interval for age and/or because of age above 40 years [12].
- FSH >9.9 IU/l or E2 >74.9 pg/ml or based on a prior cycle performance if oocyte or embryo yields were considered inadequate: fewer than seven oocytes or fewer than four embryos [13].
- Seven studies included pre-treatment AFC:
- Serum AMH <1 ng/ml or serum FSH >15 IU/l and AFC <4 on day 2 of the menstrual cycle [14].
- AFC <5 or AMH <1.1 ng/ml and a previous poor ovarian response [15].
- Day 3 serum FSH >12 mIU/ml and AFC ≤3 [16].
- AFC <6 per ovary [17].
- Day 3 FSH level >10 IU/l or an FSH/(LH) >3 or AFC <5 or previous poor ovarian response (<5 oocytes retrieved) [18].
- Day 3 FSH level ≥12 IU/l and AFC ≥5 [19].
- AFC ≤10 and AMH ≤5 pmol/L (0.7 ng/ml) [20].
In addition, two definitions included the results from a previous treatment cycle [13, 18].
These results are summarized in Table 1.
Table 1.
Markers used in the definitions of diminished ovarian reserve in the studies
| FSH | Estradiol | AFC | AMH | FSH/LH ratio | Previous IVF outcomes | |
|---|---|---|---|---|---|---|
| Merhi et al. [7] | >10 IU/l | |||||
| Buyuk et al. [8] | >10 IU/l | |||||
| Gleicher et al. [9, 10] | >7 IU/l under 33 years >7.9 IU/l between 33 and 37 years >8.4 IU/l between 38 and 40 years |
<0.8 ng/ml | ||||
| Lu et al. [11] | >10 IU/l | >3 | ||||
| Gleicher et al. [12] | Outside the 95 % CI for age | Outside the 95 % CI for age | ||||
| Gleicher and Barad [13] | >9.9 IU/l | >74.9 IU/l | <7 oocytes or <4 embryos | |||
| Kara et al. [14] | >15 IU/l | <4 | <1 ng/ml | |||
| Yilmaz et al. [15] | <5 | <1.1 ng/ml | ≤3 oocytes | |||
| Check et al. [16] | >12 IU/l | ≤3 | ||||
| Kumbak et al. [17] | <6 per ovary | |||||
| Zhang et al. [18] | >10 IU/l | <5 | >3 | <5 oocytes | ||
| Check et al. [19] | >12 IU/l | ≥5 | ||||
| Jayaprakasan et al. [20] | ≤10 | ≤5 pmol/l (0.7 ng/ml) |
FSH follicle-stimulating hormone, AFC antral follicular count, AMH anti-Müllerian hormone, LH luteinizing hormone, IVF in vitro fertilization, CI confidence interval
It is obvious that the considerable heterogeneity in the definition of DOR used in these studies contributes to confusing results. Consequently, it is extremely difficult to draw conclusions on IVF outcomes in the population of women with DOR. Moreover, in specifically DOR-related infertility (i.e., without male or tubal factor), no data enable us to assert what is the best treatment for the couple (IVF, IUI, controlled ovarian stimulation (COS) alone) or what the probabilities of success are for each technique. Hence, there is a need for a clear and consensual definition of DOR. In addition a consistent definition in this situation will allow clinical trials to share the same inclusion/exclusion criteria.
Among the various parameters used to define DOR, it appears that FSH, AFC, and AMH serum levels are the most evaluated and thus frequently used criteria, as an extrapolation of all available data on diminished ovarian reserve.
Definition of cutoff values is the first challenge. Although even the best ovarian reserve marker at the best cutoff values is associated with a false positive rate of 10–20 % [21, 22], both AMH and AFC appear to be considered as the most reliable and accurate markers of ovarian reserve [22, 23]. In the ESHRE consensus regarding POR, AMH and AFC are rather considered as post hoc test to confirm DOR after a first ovarian stimulation resulting in poor outcomes [3]. Consequently, one option could be the use of the ESHRE consensus values used for POR as an a priori definition of DOR, excluding previous stimulation results and age over 40. Thus, (i) any of the risk factors for POR and/or (ii) an abnormal ovarian reserve test (i.e., antral follicular count (AFC) <5–7 follicles or anti-Müllerian hormone (AMH) <0.5–1.1 ng/ml) could be suggested.
Theoretically, cutoffs for AMH and AFC might be stratified by age. Indeed, both AMH and AFC decrease with age-specific levels, and the true issue is to identify among an age-specific level patients who had a significant decrease in ovarian reserve. However, data providing the predictive values of AMH and AFC on success rates are not stratified by age. A recent meta-analysis showed that AMH, independently of age, might be helpful when counseling couples before undergoing infertility treatment [24]. However predictive accuracy was low (OR = 2.39) for predicting live birth after assisted conception [24]. In addition, in the clinical practice, stratification of AFC and AMH by age to define cutoffs would be time-consuming and complex. Another issue that needs to be raised is the quantitative approach to what is a “normal ovarian reserve” stratified by age. So far, no such data is available. Therefore, there is a need of quantitative criteria stratified by age, to define ovarian reserve in the general population, as well as in infertile women with other infertility factors than altered ovarian function. This issue is particularly important for women with single ovary. Moreover, in this specific population, it is necessary to better define normality of ovarian volume, AFC, AMH, and additional biological markers such as FSH/E2 ratio.
It should be kept in mind as well that several AMH assays are available and this contributes to the hetereogeneity of the data. Most of the recent studies measured AMH using the Diagnostic Systems Laboratories Inc. (DSL, Webster, TX, USA) assay, the Immunotech-Beckman Coulter (IBC, Marseille, France) assay, or the Beckman Coulter Generation II assay. Homogenization of technical aspects in the definition markers thus appears necessary, and this should be the subject of a consensus among reproductive medicine specialists and biologists. Nevertheless, the predictive accuracy of AMH for live birth was established, regardless of the assay used [24].
In conclusion, to date, there are no studies that specifically deal with the issue of the appropriate treatment for women with DOR-associated infertility. An international consensus on the definition of DOR, including markers, cutoffs, and assay techniques, is thus strongly needed as well as studies focusing on this specific population using consensual criteria. The specific assay and standard utilized should be included in any definition and may need to change as improvements occur. We suggest that (i) any of the risk factors for POR and/or (ii) an abnormal ovarian reserve test (i.e., antral follicular count (AFC) <5–7 follicles or anti-Müllerian hormone (AMH) <0.5–1.1 ng/ml) could be used and that the choice of the assay should be defined in a consensus meeting.
Footnotes
Capsule
Diminished ovarian reserve could be defined as: (i) any of the risk factors for poor ovarian response and/or (ii) an abnormal ovarian reserve test (i.e., antral follicular count (AFC) <5–7 follicles or anti-Müllerian hormone (AMH) <0.5–1.1 ng/ml).
References
- 1.De Vos M, Devroey P, Fauser BCJM. Primary ovarian insufficiency. Lancet Lond Engl. 2010;376(9744):911–921. doi: 10.1016/S0140-6736(10)60355-8. [DOI] [PubMed] [Google Scholar]
- 2.Goswami D, Conway GS. Premature ovarian failure. Hum Reprod Update. 2005;11(4):391–410. doi: 10.1093/humupd/dmi012. [DOI] [PubMed] [Google Scholar]
- 3.Ferraretti AP, La Marca A, Fauser BCJM, Tarlatzis B, Nargund G, Gianaroli L, et al. ESHRE consensus on the definition of “poor response” to ovarian stimulation for in vitro fertilization: the Bologna criteria. Hum Reprod Oxf Engl. 2011;26(7):1616–1624. doi: 10.1093/humrep/der092. [DOI] [PubMed] [Google Scholar]
- 4.Goldman MB, Thornton KL, Ryley D, Alper MM, Fung JL, Hornstein MD, et al. A randomized clinical trial to determine optimal infertility treatment in older couples: the Forty and Over Treatment Trial (FORT-T) Fertil Steril. 2014;101(6):1574.e1–2–81.e1–2. doi: 10.1016/j.fertnstert.2014.03.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wiser A, Shalom-Paz E, Reinblatt SL, Son W-Y, Das M, Tulandi T, et al. Ovarian stimulation and intrauterine insemination in women aged 40 years or more. Reprod Biomed Online. 2012;24(2):170–173. doi: 10.1016/j.rbmo.2011.11.007. [DOI] [PubMed] [Google Scholar]
- 6.Harris ID, Missmer SA, Hornstein MD. Poor success of gonadotropin-induced controlled ovarian hyperstimulation and intrauterine insemination for older women. Fertil Steril. 2010;94(1):144–148. doi: 10.1016/j.fertnstert.2009.02.040. [DOI] [PubMed] [Google Scholar]
- 7.Merhi Z, Zapantis A, Berger DS, Jindal SK. Determining an anti-Mullerian hormone cutoff level to predict clinical pregnancy following in vitro fertilization in women with severely diminished ovarian reserve. J Assist Reprod Genet. 2013;30(10):1361–1365. doi: 10.1007/s10815-013-0077-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Buyuk E, Seifer DB, Illions E, Grazi RV, Lieman H. Elevated body mass index is associated with lower serum anti-mullerian hormone levels in infertile women with diminished ovarian reserve but not with normal ovarian reserve. Fertil Steril. 2011;95(7):2364–2368. doi: 10.1016/j.fertnstert.2011.03.081. [DOI] [PubMed] [Google Scholar]
- 9.Gleicher N, Weghofer A, Barad DH. Improvement in diminished ovarian reserve after dehydroepiandrosterone supplementation. Reprod Biomed Online. 2010;21(3):360–365. doi: 10.1016/j.rbmo.2010.04.006. [DOI] [PubMed] [Google Scholar]
- 10.Gleicher N, Ryan E, Weghofer A, Blanco-Mejia S, Barad DH. Miscarriage rates after dehydroepiandrosterone (DHEA) supplementation in women with diminished ovarian reserve: a case control study. Reprod Biol Endocrinol RBE. 2009;7:108. doi: 10.1186/1477-7827-7-108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lu Q, Shen H, Li Y, Zhang C, Wang C, Chen X, et al. Low testosterone levels in women with diminished ovarian reserve impair embryo implantation rate: a retrospective case-control study. J Assist Reprod Genet. 2014;31(4):485–491. doi: 10.1007/s10815-014-0186-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gleicher N, Kim A, Weghofer A, Shohat-Tal A, Lazzaroni E, Lee H-J, et al. Starting and resulting testosterone levels after androgen supplementation determine at all ages in vitro fertilization (IVF) pregnancy rates in women with diminished ovarian reserve (DOR) J Assist Reprod Genet. 2013;30(1):49–62. doi: 10.1007/s10815-012-9890-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gleicher N, Barad D. “Ovarian age-based” stimulation of young women with diminished ovarian reserve results in excellent pregnancy rates with in vitro fertilization. Fertil Steril. 2006;86(6):1621–1625. doi: 10.1016/j.fertnstert.2006.04.046. [DOI] [PubMed] [Google Scholar]
- 14.Kara M, Aydin T, Aran T, Turktekin N, Ozdemir B. Does dehydroepiandrosterone supplementation really affect IVF-ICSI outcome in women with poor ovarian reserve? Eur J Obstet Gynecol Reprod Biol. 2014;173:63–65. doi: 10.1016/j.ejogrb.2013.11.008. [DOI] [PubMed] [Google Scholar]
- 15.Yilmaz N, Uygur D, Inal H, Gorkem U, Cicek N, Mollamahmutoglu L. Dehydroepiandrosterone supplementation improves predictive markers for diminished ovarian reserve: serum AMH, inhibin B and antral follicle count. Eur J Obstet Gynecol Reprod Biol. 2013;169(2):257–260. doi: 10.1016/j.ejogrb.2013.04.003. [DOI] [PubMed] [Google Scholar]
- 16.Check JH, Summers-Chase D, Yuan W, Horwath D, Wilson C. Effect of embryo quality on pregnancy outcome following single embryo transfer in women with a diminished egg reserve. Fertil Steril. 2007;87(4):749–756. doi: 10.1016/j.fertnstert.2006.11.001. [DOI] [PubMed] [Google Scholar]
- 17.Kumbak B, Oral E, Kahraman S, Karlikaya G, Karagozoglu H. Young patients with diminished ovarian reserve undergoing assisted reproductive treatments: a preliminary report. Reprod Biomed Online. 2005;11(3):294–299. doi: 10.1016/S1472-6483(10)60836-X. [DOI] [PubMed] [Google Scholar]
- 18.Zhang HH, Xu PY, Wu J, Zou WW, Xu XM, Cao XY, et al. Dehydroepiandrosterone improves follicular fluid bone morphogenetic protein-15 and accumulated embryo score of infertility patients with diminished ovarian reserve undergoing in vitro fertilization: a randomized controlled trial. J Ovarian Res. 2014;7:93. doi: 10.1186/s13048-014-0093-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Check JH, Amui J, Choe JK, Cohen R. The effect of a rise or fall of serum estradiol the day before oocyte retrieval in women aged 40–42 with diminished egg reserve. Clin Exp Obstet Gynecol. 2015;42(3):282–284. [PubMed] [Google Scholar]
- 20.Jayaprakasan K, Narkwichean A, Maalouf WE, Campbell BK. Efficacy of dehydroepiandrosterone to overcome the effect of ovarian ageing (DITTO): a proof of principle randomised controlled trial protocol. BMJ Open. 2014;4(10):e005767. doi: 10.1136/bmjopen-2014-005767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Broekmans FJ, Kwee J, Hendriks DJ, Mol BW, Lambalk CB. A systematic review of tests predicting ovarian reserve and IVF outcome. Hum Reprod Update. 2006;12(6):685–718. doi: 10.1093/humupd/dml034. [DOI] [PubMed] [Google Scholar]
- 22.La Marca A, Sighinolfi G, Radi D, Argento C, Baraldi E, Artenisio AC, et al. Anti-Mullerian hormone (AMH) as a predictive marker in assisted reproductive technology (ART) Hum Reprod Update. 2010;16(2):113–130. doi: 10.1093/humupd/dmp036. [DOI] [PubMed] [Google Scholar]
- 23.Broer SL, Mol BWJ, Hendriks D, Broekmans FJM. The role of antimullerian hormone in prediction of outcome after IVF: comparison with the antral follicle count. Fertil Steril. 2009;91(3):705–714. doi: 10.1016/j.fertnstert.2007.12.013. [DOI] [PubMed] [Google Scholar]
- 24.Iliodromiti S, Kelsey TW, Wu O, Anderson RA, Nelson SM. The predictive accuracy of anti-Müllerian hormone for live birth after assisted conception: a systematic review and meta-analysis of the literature. Hum Reprod Update. 2014;20(4):560–570. doi: 10.1093/humupd/dmu003. [DOI] [PubMed] [Google Scholar]