Abstract
Microbial processes within deep-sea hydrothermal plumes affect ocean biogeochemistry on global scales. In rising hydrothermal plumes, a combination of microbial metabolism and particle formation processes initiate the transformation of reduced chemicals like hydrogen sulfide, hydrogen, methane, iron, manganese and ammonia that are abundant in hydrothermal vent fluids. Despite the biogeochemical importance of this rising portion of plumes, it is understudied in comparison to neutrally buoyant plumes. Here we use metagenomics and bioenergetic modeling to describe the abundance and genetic potential of microorganisms in relation to available electron donors in five different hydrothermal plumes and three associated background deep-sea waters from the Eastern Lau Spreading Center located in the Western Pacific Ocean. Three hundred and thirty one distinct genomic ‘bins' were identified, comprising an estimated 951 genomes of archaea, bacteria, eukarya and viruses. A significant proportion of these genomes is from novel microorganisms and thus reveals insights into the energy metabolism of heretofore unknown microbial groups. Community-wide analyses of genes encoding enzymes that oxidize inorganic energy sources showed that sulfur oxidation was the most abundant and diverse chemolithotrophic microbial metabolism in the community. Genes for sulfur oxidation were commonly present in genomic bins that also contained genes for oxidation of hydrogen and methane, suggesting metabolic versatility in these microbial groups. The relative diversity and abundance of genes encoding hydrogen oxidation was moderate, whereas that of genes for methane and ammonia oxidation was low in comparison to sulfur oxidation. Bioenergetic-thermodynamic modeling supports the metagenomic analyses, showing that oxidation of elemental sulfur with oxygen is the most dominant catabolic reaction in the hydrothermal plumes. We conclude that the energy metabolism of microbial communities inhabiting rising hydrothermal plumes is dictated by the underlying plume chemistry, with a dominant role for sulfur-based chemolithoautotrophy.
Introduction
Deep-sea hydrothermal vent plumes occur at mid-ocean ridges and back arc-basins throughout the world's oceans, where chemically reduced hydrothermal vent fluids are mixed with cold, oxic deep-ocean water. The enrichment of electron donors such as H2S, H2, CH4, NH3, Mn2+ and Fe2+ in plumes fuels chemosynthetic microbial metabolisms (Jannasch and Mottl, 1985; Distel et al., 1988; de Angelis et al., 1993; Lam et al., 2004; Dick et al., 2009b; Petersen et al., 2011). Hydrothermal plumes influence the broader oceans because biogeochemical processes in plumes control the availability and fate of trace metals and essential micronutrients (Kadko, 1993; Toner et al., 2009; Tagliabue et al., 2010; Breier et al., 2012), and primary production in plumes may serve as a significant source of carbon to the deep oceans (McCollom, 2000). The microbial ecology of hydrothermal plumes shares many similarities with other marine environments where primary production is linked to oxidation of reduced sulfur species, ammonia, and hydrocarbons, including the pelagic ocean water column, oxygen minimum zones and deep-sea hydrocarbon plumes (DeLong et al., 2006; Aristegui et al., 2009; Canfield et al., 2010; Reinthaler et al., 2010; Swan et al., 2011; Dick et al., 2013; Rivers et al., 2013).
Recent studies have begun to elucidate the importance and role of microorganisms and metabolisms that operate within hydrothermal plumes. Surveys of the small subunit (SSU) ribosomal RNA (rRNA) genes using tag pyrosequencing and clone libraries have revealed the composition of plume microbial communities (Sunamura et al., 2004; Dick and Tebo, 2010; German et al., 2010; Sylvan et al., 2012). Metagenomic, metatranscriptomic and metaproteomic methods have provided insights into the roles of dominant organisms involved in oxidation of sulfur, hydrogen, methane and ammonia in hydrothermal plumes such as SUP05 Gammaproteobacteria (Anantharaman et al., 2013), Methylococcaceae Gammaproteobacteria (Li et al., 2014a), Marine Group I Thaumarchaea (Baker et al., 2012) and SAR324 Deltaproteobacteria (Sheik et al., 2014). New studies also show that rare members of the plume microbial community such as Alteromonadaceae Gammaproteobacteria (Li et al., 2014) and Nitrospirae (Baker et al., 2013) are potentially keystone species with roles in iron uptake and nitrite oxidation, respectively. Although these studies have expanded our knowledge of hydrothermal plume microbiology, key questions remain unanswered. Little is known about the distribution of microbial groups and associated metabolic functional capacities across geochemical and physical gradients spanning different hydrothermal plumes as well as within the same plume.
The Eastern Lau Spreading Center (ELSC) is a deep-sea hydrothermal system located in the Lau Basin, a back-arc basin in the Western Pacific Ocean. In contrast to mid-ocean ridges, back-arc basins exhibit greater geologic diversity and are characterized by asymmetrical seafloor spreading. At ELSC, these characteristics manifest as steep gradients in the chemistry of underlying rocks, tectonic characteristics and hydrothermal activity along a north–south axis. Spreading rates are fast in the north (~97 mm per year) and slower in the south (40 mm per year) (Zellmer and Taylor, 2001). A deep axial valley (~2640 m) with basaltic underlying rock in the north transitions into a shallow axial ridge with andesitic underlying rocks (~1877 m) in the south (Martinez et al., 2006). Six different vent fields have been identified along the ELSC, and the chemistry of hydrothermal fluids exhibits significant inter-field variability, with properties similar to mid-ocean ridges in the north and highly elevated concentrations of H2S, Fe and Mn coupled with lower pH towards the south (Ferrini et al., 2008; Mottl et al., 2011). Recently, Flores et al. (2012) surveyed seafloor hydrothermal deposits at ELSC and concluded that they host microbial communities similar to mid-ocean ridges except for the Mariner vent field, where the community was heavily influenced by its unique geochemistry. Sylvan et al. (2013) noted that the microbial diversity of low temperature hydrothermal deposits along ELSC displayed significant heterogeneity that followed host rock composition. Since gradients in the geochemistry of hydrothermal vent fluids manifest themselves in the geochemistry of hydrothermal plumes, the ELSC provides an opportunity to study the impacts of vent geology and geochemistry on the composition of microbial communities in plumes.
We recently analyzed the overall microbial community diversity of hydrothermal plumes across the ELSC by high-throughput tag sequencing of 92 plume and background samples collected from various elevations at five of the six vent fields (Supplementary Table 1; Sheik et al., 2015). This study showed that although ELSC plumes contain a mixture of vent-associated and pelagic ocean microorganisms, ELSC plume communities are more similar to pelagic than to vent-associated communities. In a different study, we identified bacterial sulfur oxidation genes on novel viruses that putatively infect globally distributed sulfur-oxidizing SUP05 bacteria (Anantharaman et al., 2014). Here, we follow up these studies by using shotgun metagenomic sequencing to characterize the metabolic functions of microorganisms in hydrothermal plumes at the five different ELSC vent fields and in three surrounding deep background waters. Metagenomic analyses and thermodynamic-bioenergetic models show that despite the microbial and geochemical variability observed previously, the most abundant energy metabolisms and community composition across ELSC plumes are strikingly similar, being dominated by diverse populations of sulfur-oxidizing chemolithoautotrophs. The ELSC plumes also host heterotrophs and chemolithoautotrophs that utilize hydrogen, ammonia, methane and nitrite as electron donors. Minor variations between ELSC sites reflect differences in underlying plume chemistry.
Materials and methods
Sample collection
Samples were collected from five different hydrothermal vent fields (Kilo Moana, Tahi Moana, Abe, Tui Malila and Mariner, from north to south) during cruises TN235 and TN236 aboard the R/V Thomas G. Thompson in May–July 2009. Details of samples and the sampling locations are provided in Supplementary Table 1. A total of 92 hydrothermal plume and background deep-sea samples were collected. Of these, 78 samples were collected from the rising plume and background deep sea using a suspended particle rosette sampler (SUPR) (Breier et al., 2009) mounted on remotely operated vehicle ROV Jason II, while 14 samples were collected with 20 l Niskin bottles via water rosette hydrocasts. Water samples collected with SUPR (10–60 l) were filtered in situ on to 0.8-μm, 37-mm polycarbonate Supor membranes (Pall Corporation, Port Washington, NY, USA) and preserved shipboard in RNAlater (Ambion, Austin, TX, USA). Water samples collected by hydrocasts were pressure filtered with N2 gas shipboard on to 0.2-μm, 47-mm polycarbonate membranes and preserved in RNAlater. To compare these two sampling strategies, we collected a single above plume background sample on both a 0.8- and 0.2-μm filter on Dive J2-445. Comparison of the microbial communities on these two filters was performed using 16 s rRNA surveys, and no substantial differences were observed between these two filter sizes (Sheik et al., 2015).
Extraction of nucleic acids and DNA sequencing
DNA was extracted from ¼ filters as described previously (Dick and Tebo, 2010). Multiple displacement amplification of genomic DNA was performed using the illustra Ready-To-Go GenomiPhi V3 DNA Amplification Kit (GE Healthcare, Piscataway, NJ, USA). Shotgun sequencing of DNA was performed with Illumina HiSeq2000 PE 100 (Illumina, Inc., San Diego, CA, USA) at the University of Michigan DNA Sequencing Core. Comparison of normalized metagenomic abundance to 16 s rRNA abundance show similar abundance patterns (Sheik et al., 2015), suggesting that any possible multiple displacement amplification bias (Pinard et al., 2006) did not obscure the major trends.
De novo genomic assembly and annotation
Raw shotgun sequencing reads were deprelicated (100% identity over 100% of length) and trimmed using the adaptive read trimmer, Sickle (https://github.com/najoshi/sickle). Samples from the five vent sites (Kilo Moana, Abe, Mariner, Tahi Moana and Tui Malila) were each assembled de novo to obtain five separate site-specific assemblies. Whole genome de novo assemblies were performed using IDBA-UD (Peng et al., 2012) with the following parameters: —mink 50, —maxk 92, —step 4, —min_contig 500. rRNA reads were identified using RiboPicker (Schmieder et al., 2012) with a custom database (5s + 16s + 23s rRNA) and assembled separately using IDBA-UD with the following parameters: —mink 50, —maxk 92, —step 4. To check the veracity of the generated contigs, de novo whole-genome assemblies were repeated using Velvet (Zerbino and Birney, 2008) in an iterative manner by removing the reads used in formation of contigs at the higher kmer (kmer 91 to 51, steps of 4) using the following parameters: -exp_cov auto –ins_length 214–223 -ins_length_sd 20 –read_trkg yes –min_contig_lgth 2500. This was followed by further refinement with MetaVelvet (Namiki et al., 2012) (kmer 91 to 51, steps of 4) using the following parameters: -scaffolding yes, -min_contig_lgth 2500. (Li et al., 2009). Referenced contigs were visualized for evenness of mapped read coverage to identify potential chimeric regions using Integrative Genome Viewer (IGV; Thorvaldsdóttir et al., 2013). All major trends were similar across both sets of assemblies. All data presented in this paper are from the assemblies generated by IDBA-UD.
Read mapping
Paired-end sequencing reads were mapped to assembled contigs using the Burrows–Wheeler Aligner (BWA version 0.7.5a; Li and Durbin, 2009) to generate read coverage information for assembled contigs. First, individual forward and reverse reads for each sample were mapped to the assembled contigs using the BWA-ALN algorithm implemented using default parameters except for a modified maxDiff parameter (aln, -n 0.02). Second, paired forward and reverse read alignments were generated in the SAM format using the BWA-SAMPE algorithm with default parameters. The mapped read counts were extracted using SAMtools 0.1.17 (Li et al., 2009).
Binning and conserved gene analysis
All resulting contigs were assigned into putative taxonomic groups by binning with emergent self-organizing maps (ESOM) with a combination of tetranucleotide frequency (Dick et al., 2009a) and coverage of contigs across the five different assemblies (cutoffs: minimum contig size=4 Kb, maximum contig size=8 Kb) as determined by read mapping. All tetramers containing start and stop codons were removed prior to analysis. All resultant bins were manually evaluated for accuracy, completeness and to estimate number of genome equivalents using the distribution of conserved phylogenetic markers described previously (Ciccarelli et al., 2006). Gene calling and annotations were done through the DOE Joint Genome Institute (JGI) Integrated Microbial Genomes metagenomics expert review (IMG-MER) pipeline (Markowitz et al., 2008).
Identification of bins
All bins were identified to appropriate taxonomic levels in the following order of workflow: (i) identification of SSU rRNA genes on contigs by BLASTN (Altschul et al., 1990) against the Silva SSU Database version 111 (Pruesse et al., 2007); (ii) ESOM binning with reference genomes; (iii) taxonomic clustering of annotated genes by protein UBLAST with the IMG database (>50% Markowitz et al., 2008) of identified open reading frames. Bins in which a majority (>50%) of the predicted open reading frames could not be identified to any taxonomic level or clustered with viruses were annotated as putative extrachromosomal elements (plasmids/phages/prophages). Number of genome equivalents for bacteria and archaea were estimated using the average number of conserved phylogenetic markers. Number of genome equivalents for bins containing extrachromosomal elements was estimated by the presence of contigs from each of the five rising plumes sampled (for example, if bin A contained contigs from two different rising plumes, then the number of genome equivalents was 2).
Functional gene analysis
Genetic potential of individual bins was investigated by using reciprocal protein blasts with custom databases, NCBI-nr (cutoff: e-value<1e−5), and verified by protein functional classification using InterProScan 5 (Jones et al., 2014). Abundance of genes for oxidation of electron donors was calculated by recruitment of individual reads from each of the 12 samples using BLASTX with custom protein databases (cutoffs: minimum alignment length>60 bp; e-value<1e−5; bitscore>50). Custom protein databases were built from all predicted open reading frames (not only binned scaffolds) from our ELSC scaffolds and closely related genes from public databases (as determined by blastP against NCBI-nr). All our custom databases are available at http://www.earth.lsa.umich.edu/geomicrobiology/Lau.html. All gene abundances were normalized to account for variations in gene length and dataset size.
Sequence alignment and phylogeny
Alignment of SSU rRNA, particulate hydrocarbon monooxygenase and Ni-Fe hydrogenase sequences was performed by MUSCLE (Edgar, 2004) with reference sequences using default parameters followed by manual refinement. Phylogenetic analysis of Ni-Fe hydrogenase genes and particulate hydrocarbon monooxygenase genes was inferred by Maximum Likelihood implemented in RaxML (Stamatakis, 2006) using the PROTGAMMAGTR algorithms and bootstrapped 1000 times. The phylogeny of SSU rRNA genes was inferred by maximum likelihood implemented in RaxML using the GAMMAGTR algorithm and bootstrapped 1000 times.
Bioenergetic and thermodynamic modeling
Equilibrium thermodynamic reaction path modeling was used to predict mineral precipitation, chemical concentrations and activity coefficients resulting from the mixing of sea water with end-member fluid from the hydrothermal vents (Mottl et al., 2011). Supplementary Table S2 lists the end-member fluid concentrations utilized in our model. Our approach follows those of previous studies (Janecky and Seyfried, 1984; Bowers et al., 1985; McCollom, 2000). This specific plume thermodynamic model implementation has been described previously (Anantharaman et al., 2013; Anantharaman et al., 2014).
Results
To investigate links between microbial metabolic potential and geochemistry in hydrothermal plumes, we analyzed a total of 12 samples, comprising 9 plume and 3 background deep-sea samples from 5 different vent sites along a north–south axis at ELSC (Kilo Moana, Tahi Moana, Abe, Tui Malila and Mariner) (Supplementary Table 1). Optical backscatter was used to identify hydrothermal plumes and verify the absence of hydrothermal influence on background samples. To characterize the background sea water microbial community, we collected three distinct types of background samples: (1) near bottom/seafloor backgrounds were collected with SUPR away from the hydrothermal vent fields; (2) above neutrally buoyant plume backgrounds were collected with SUPR continuously pumping through the water depth interval of 700–1300 m; and (3) below neutrally buoyant plume backgrounds were collected by hydrocasts.
Metagenomic sequencing, de novo assembly and binning
Shotgun sequencing of community genomic DNA on 6 lanes of Illumina HiSeq2000 produced a total of 1026 438 887 paired-end reads (read length=100 bp, total reads=2052 877 774, average insert-size=421 bp). About 90.1% of reads were above Q30, and the mean quality score was 35.18. De novo assemblies of quality filtered reads generated a total of 1 091 773 contigs and 1511 701 270 bp of consensus sequence ranging from 33.9 to 53.8% of reads from a specific vent site (Supplementary Table 3). Prediction of open reading frames resulted in 1 697 862 putative genes (Supplementary Table 4).
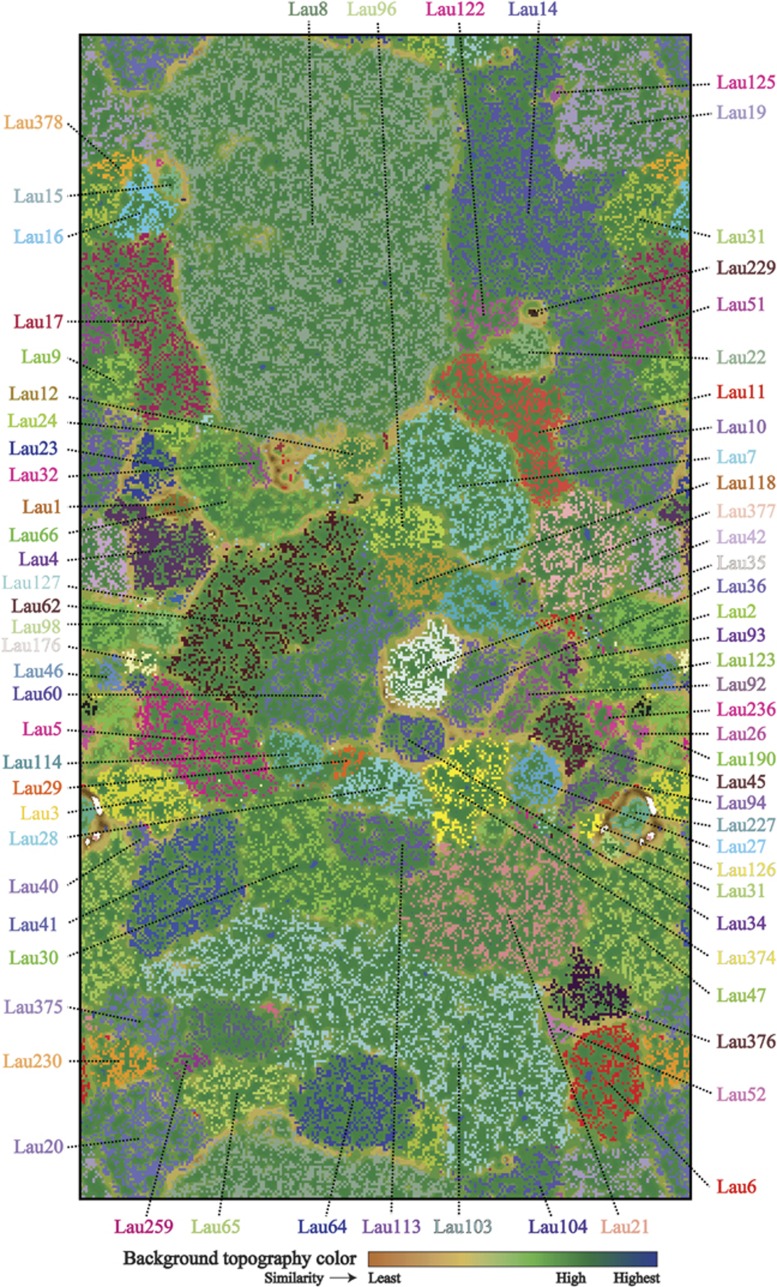
Tetranucleotide-based ESOM binning was performed on all contigs of length >4000 bp (62 135 contigs). All five assemblies were binned together to identify similar organisms across sites. Putative taxonomic assignment by SSU rRNA genes, binning with reference genomes, and taxonomic profiling of annotated genes yielded a total of 331 distinct genomic bins comprising 98 bacteria, 7 archaea, 1 eukarya (Figure 1,Supplementary Table 5) and 225 putative extrachromosomal elements (plasmids/phages/prophages) (Supplementary Figure S1, Supplementary Table 6) representing 32.89% of assembled scaffolds. In total, these bins represent an estimated total of 951 genomes sequences, ranging in completeness from partial to near-complete (Supplementary Table 7).
Figure 1.
Assignment of assembled contigs from ELSC to specific bacterial, archaeal and eukaryotic populations using ESOM implemented with tetranucleotide frequencies. Each point on the map represents a contig (>4 kb) or contig fragment generated in silico (4–8 kb). All identified bins are uniquely color coded as indicated. The ESOM map displayed is tiled and torroidal (that is, continuous from top to bottom and left to right).
The hydrothermal plumes of ELSC were dominated by microbial populations described previously from the hydrothermal plumes of Guaymas Basin: SUP05 Gammaproteobacteria, Alteromonadaceae Gammaproteobacteria, Marine Group I Thaumarchaea and SAR324 Deltaproteobacteria (Dick and Tebo, 2010; Baker et al., 2012; Lesniewski et al., 2012; Anantharaman et al., 2013; Sheik et al., 2014; Figure 2, Supplementary Figure S2). In addition, we assembled essentially complete, near-complete and partial genomes for novel organisms previously unknown in the pelagic water column or known only through SSU rRNA genes retrieved from hydrothermal plumes, seafloor or hydrothermal vent chimney communities (Supplementary Table 5). This included Aquificae (genomic bins Lau227 and Lau237), Planctomycetes (Lau7, Lau35, Lau36, Lau94 and Lau96), Cyanobacteria (Lau27), Melainabacteria (Lau2), Candidate Division TM7 (Saccharibacteria) (Lau1), Chloroflexi (Lau3 and Lau12), Marine Group A (Lau47, Lau103 and Lau104), Nitrospinae (Lau17), Nitrospirae (Lau44), Poribacteria (Lau21 and Lau377), Verrucomicrobia (Lau158, Lau184, Lau190 and Lau191), Acidobacteria (Lau40), Epsilonproteobacteria (Lau15, Lau129 and Lau229) and Gemmatimonadetes (Lau45) (Teske et al., 2002; Walsh et al., 2009; Swan et al., 2011; Yamamoto and Takai, 2011; Flores et al., 2012; Sylvan et al., 2012; Wright et al., 2012; Allers et al., 2013; Di Rienzi et al., 2013; Sylvan et al., 2013). Searches for eukaryotic SSU rRNA genes revealed the presence of novel populations of Archaeplastida, DH147-EKD23, Alveolata (Ciliophora, Dinoflagellata, OLI11255 and Protalveolata), Rhizaria and Opisthokonta (Fungi and Metazoa). However, we conclusively identified only one bin of Eukarya, the largest bin (Lau8) in the metagenome, putatively identified as a Bathymodiolinae mussel with highest SSU rRNA similarity (99.9%) to Bathymodiolus tangaroa from the Western Pacific Ocean (Supplementary Figure S3; Jones et al., 2006). This bin contained a total of 54 Mb of consensus sequence on 9599 contigs, and was represented most abundantly in the Abe hydrothermal plume metagenome.
Figure 2.
Normalized abundance, energy and carbon metabolism of the 50 most abundant bins identified in ELSC hydrothermal plumes. Heat map on left panel indicates abundance of bins for each sampled hydrothermal plume and is displayed as a percentage of the total community as shown in the legend. Black boxes indicate identified genes encoding carbon metabolism and electron donors and acceptors for energy metabolism.
Functional resolution of metagenomic bins
We analyzed metagenomic bins across the five hydrothermal plumes at ELSC to identify genes encoding for energy and carbon metabolism, including utilization of electron donors, electron acceptors and carbon fixation pathways (Figure 2).
Oxidation of reduced sulfur
The genetic potential for utilization of different forms of reduced S species (H2S, S0 and S2O32-, SO32-) as electron donors was determined by searching genomic bins for the following genes: the sox pathway (soxABCDXYZ) (Supplementary Table S10) and rhodanese sulfurtransferase for oxidation of S2O32-; adenosine phosphosulfate reductase (apr), sulfate adenylyltransferase (sat) and sulfite oxidoreductase for oxidation of SO32-; sulfur oxygenase reductase (sor) and reverse acting-dissimilatory sulfite reductase (rdsrAB) for oxidation of S0; and flavocytochrome sulfide dehydrogenase (fcc) and sulfide:quinone oxidoreductase (sqr) for oxidation of H2S. Collectively, genes for oxidation of sulfur were the most common genes for lithotrophy identified in genomic bins, being present in 20 of the 50 most abundant organisms including the abundant SUP05 and SAR324 (Figure 2).
Hydrogen oxidation
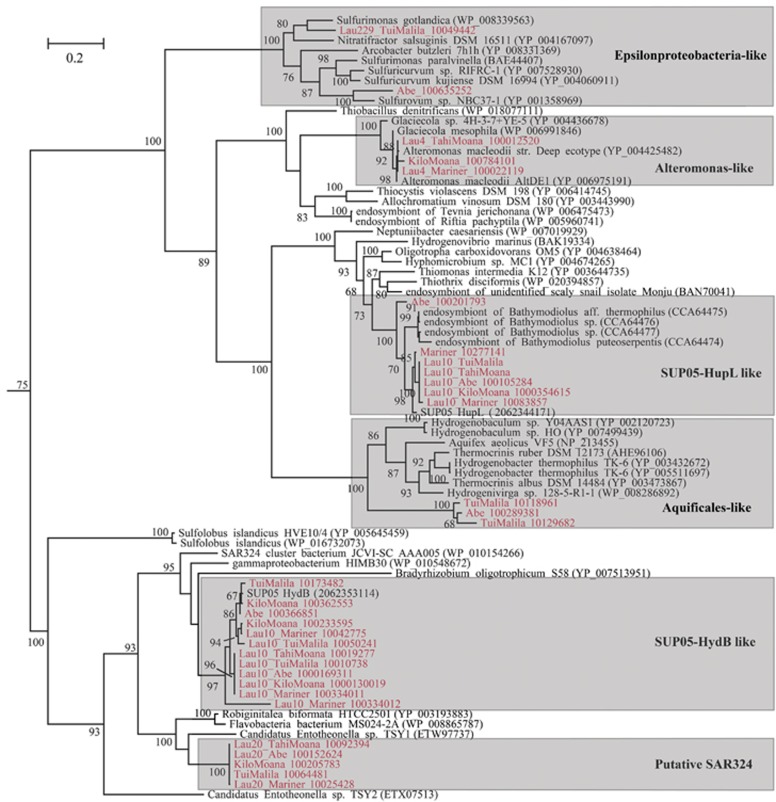
A total of 24 Ni-Fe hydrogenase operons were identified in the ELSC metagenomes, comprising three distinct forms of hydrogenases. Organisms possessing the type I membrane-bound Ni-Fe hydrogenases for oxidation of H2 include Alteromonacadae (Lau4), SUP05 (Lau10), Epsilonproteobacteria (Lau229), Poribacteria (Lau21), SAR324 (Lau20) and Flavobacteria (Lau23) (Figure 3). Amongst these, SUP05 and SAR324 possessed the ‘hyd' type Ni-Fe hydrogenases previously observed to be transcriptionally active outside of hydrothermal plumes in the deep waters of Carmen Basin and Guaymas Basin the Gulf of California (Anantharaman et al., 2013). Only Cyanobacteria (Lau27), Melainabacteria (Lau2) and Flavobacteria (Lau23) possessed the type IIa cyanobacterial uptake type Ni-Fe hydrogenases. Type III NADP-reducing Ni-Fe hydrogenases were identified in Melainabacteria (Lau2), Pseudomonadaceae (Lau 177) and Aquificae (Lau227) (Vignais and Billoud, 2007). Notably, genes for oxidation of reduced sulfur species were observed in each of the hydrogen oxidizing organisms possessing the type I Ni-Fe hydrogenases. No other forms of hydrogenases (Fe-Fe or Ni-Fe-selenium) were identified.
Figure 3.
Phylogeny of group 1 membrane-bound Ni-Fe hydrogenase large subunit inferred with maximum likelihood. Bootstrap values >60 are shown. Sequences in red are from ELSC. Gray boxes indicate phylogenetic clusters of ELSC sequences.
Ammonia oxidation
In contrast to the diversity of organisms and genes for sulfur and H2 oxidation, only two genomic bins contained genes encoding the oxidation of NH3 and CH4. A single genomic bin (Lau19) of the Marine Group I Archaea (Thaumarchaea) containing 10 genomes (including near-complete and partial) had genes for oxidation of ammonia in the form of ammonia monooxygenase (amo) (Konneke et al., 2005). No ammonia oxidizing bacteria were identified in the ELSC plumes.
Nitrite oxidation
We identified two bacterial phyla, Nitrospinae (Lau17 and Lau41) and Nitrospirae (Lau44), that are potentially involved in nitrite oxidation (Lücker et al., 2010; Baker et al., 2013; Luecker et al., 2013). Nitrite oxidoreductase (nxr) genes were identified in the Nitrospinae bin but not in the Nitrospirae bin (Supplementary Table 11). However, we recovered nxr genes on short contigs that share homology with both groups, hence their taxonomic assignment cannot be conclusively determined. Nitrospinae were more abundant than Nitrospirae at ELSC (Figure 2).
Methane oxidation
A genomic bin (Lau113) containing three near-complete genomes from three distinct locations (Abe, Mariner, Kilo Moana) was identified as a member of the OPU-3 group of Methylococcaceae (Gammaproteobacteria). All three recovered genomes possessed identical particulate methane monooxygenases (pmo) for methane oxidation (Supplementary Figure S4; Hanson and Hanson, 1996) as well as soxXYZAB genes for oxidation of thiosulfate (Supplementary Figure S5). Considering the novelty of these findings, we checked these contigs for presence of chimeric regions and homogeneity of read coverage. Contigs containing pmo and sox genes were confidently assigned to Lau113 through tetranucleotide frequency binning, while blastp of flanking genes against NCBI-nr and IMG revealed complete phylogenetic congruity with methane-oxidizing Methylococcaceae bacteria. Phylogenetic analyses of the pmoA genes confirmed that they belonged to the OPU-3 group (Supplementary Figure S4). No other methane or hydrocarbon monooxygenase genes were identified at ELSC.
Autotrophy
Given the prevalence and importance of primary production in the dark oceans, both in hydrothermal plumes and in locations far removed from vents (Aristegui et al., 2009; Reinthaler et al., 2010; Dick et al., 2013), we identified genes involved in carbon fixation. Three of the six carbon fixation pathways currently known (Hügler and Sievert, 2010) were confidently identified, including the Calvin–Benson–Bassham Cycle, the reductive tricarboxylic acid cycle and the 3-hydroxy propionate/4 hydroxy-butyrate Cycle. We identified none of the above carbon fixation pathways in methane-oxidizing OPU-3 (Lau113), which likely utilize the ribulose monophosphate pathway to assimilate carbon. The Calvin–Benson–Bassham cycle was the most abundant carbon fixation pathway in the ELSC plumes (Supplementary Fig S6).
Heterotrophy
Heterotrophic metabolism in the metagenomic bins was inferred based on prior knowledge of specific microbial groups, absence of genes for carbon fixation, and presence of genes for organic carbon transport and degradation. The most abundant heterotrophs in the ELSC metagenome were SAR11 (Lau14), Alteromonas (Lau4), Marine Group A/SAR406 (Lau47 and Lau104) and Marixanthomonas (Lau23) (Figure 2). The most commonly observed genes for uptake of organic carbon compounds were amino acid transporters (annotated as branched chain and polar amino acid transporters) and di and tricarboxylate transporters.
Fermentation
An essentially complete genome sequence measuring 0.92 Mb on just five contigs was recovered from a novel Candidate Division TM7 (Saccharibacteria) bacterium with SSU rRNA identity of only 84% to the recently identified Candidatus Saccharimonas aalborgensis (Albertsen et al., 2013). Similar to previous observations in this lineage (Albertsen et al., 2013; Kantor et al., 2013), we infer a non-respiring fermentative lifestyle for this bacterium based on the lack of genes for the tricarboxylic acid cycle, respiration or an electron transport chain. To our knowledge this represents the first genomic evidence for fermentation in the pelagic ocean, though it was hypothesized long ago (Karl et al., 1984).
Bioenergetic modeling of potential electron donors
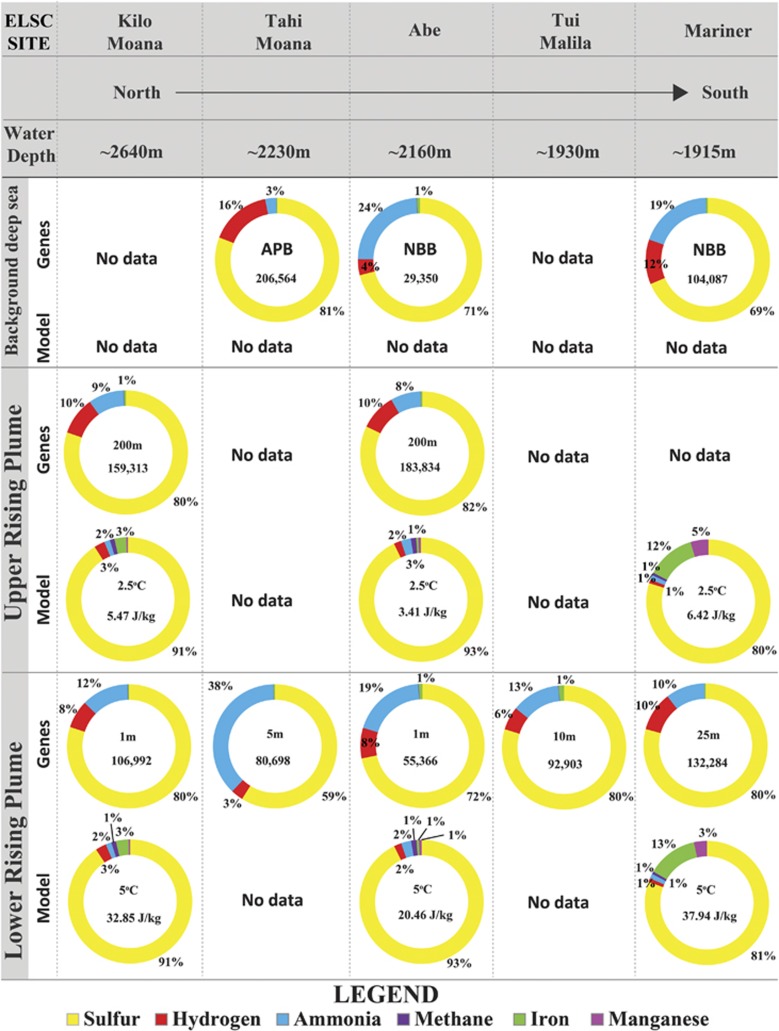
Because data on concentrations of dissolved chemicals in the plume samples studied here are not available, we used equilibrium thermodynamic reaction path modeling to estimate dissolved chemistry in the hydrothermal plumes at Abe, Kilo Moana and Mariner. Results indicate that similar to the hydrothermal plumes of Guaymas Basin, the ELSC plumes are abundant in reduced sulfur species. Although there is uncertainty with regard to the exact speciation of sulfur, our model suggests that S0 may be more prevalent than H2S within ELSC plumes. In contrast to Kilo Moana and Abe, plumes at Mariner had high concentrations of particulate Fe2+ and Mn2+. To assess the relative importance of different lithotrophic metabolisms, we utilized bioenergetic modeling to estimate the free energy yields of various electron donor/acceptor combinations based on modeled plume chemistry. In rising hydrothermal plume fluids with predicted temperatures of 2.5–5 °C, the total free energy available ranged from 3.41 to 37.94 J kg−1 of plume fluid (Figure 4). Oxidation of elemental S with O2 accounted for most of this energy (79.8–92.9%), while free energy yield from the oxidation of H2, CH4 and NH3 with O2 were minor in comparison (Figure 4). The free energy yields of Fe2+ and Mn2+ oxidation ranged from 0.6 to 13.2% and 0.4 to 4.7% respectively, with the higher values predicted for the Mariner vent site. Oxygen was predicted to be the most commonly used electron acceptor with a minor role for NO3− in the fluids of the rising plume (data not shown). Processes involving the use of alternate electron acceptors like NO3−, NO2−, NO, Fe3+, Mn4+ and SO42- were predicted to have a greater metabolic role only at higher temperatures (>50 oC; data not shown), which only occur near vent orifices and the subsurface.
Figure 4.
Comparison of gene abundance and thermodynamic-bioenergetic estimates of available free energy associated with electron donors for lithotrophy in rising ELSC hydrothermal plumes. Inset data indicate the following from top to bottom: (1) Height above vent orifice for genes; temperature used for model. (2) Gene abundance associated with electron donor oxidation (normalized for gene length and dataset size); total available free energy per kilogram of plume fluid for model. CTD denotes sample taken with CTD rather than SUPR. Gene abundances for sulfur oxidation are the sum of all reduced sulfur species. APB, above plume background; NBB, near bottom background.
Overall distribution of lithotrophic metabolisms across the ELSC
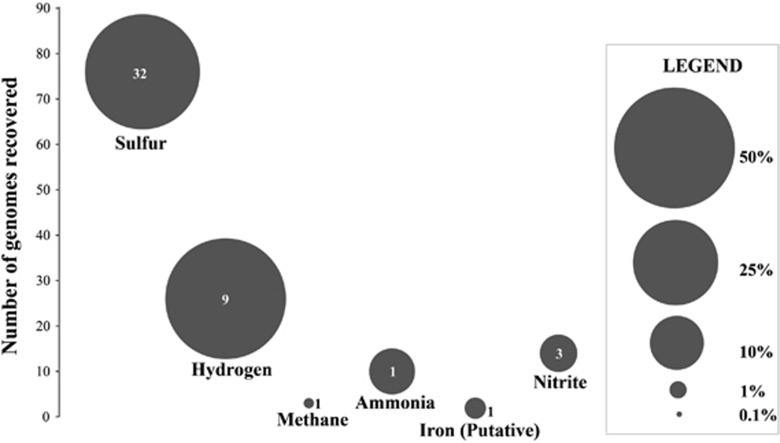
Metagenomic data indicate that genes for oxidation of sulfur (all reduced sulfur species considered) were the most abundant of any inorganic electron donor in ELSC communities, consistent with bioenergetic models of available free energy for chemosynthesis (Figure 4). Sulfur-oxidizing bacteria dominate both in terms of abundance (that is, abundance of reads mapped to genomes with sulfur oxidation genes) and diversity (that is, number of genomes or genomic bins containing sulfur oxidation genes). Genomes with genes for H2 oxidation are present at abundance similar to those for sulfur oxidation but are less diverse, while those with genes for oxidation of nitrogen species and iron account for a relatively small proportion of the genomic abundance and diversity of the ELSC community (Figure 5). Comparison of average abundance of genes for oxidation of each electron donor between sample types showed that relative to background and the lower rising plume, the upper rising plume generally had higher abundance of genes for the oxidation of sulfide, S0, thiosulfate and hydrogen, but not methane, nitrite or sulfite (Supplementary Figure S7). Based on Student's t-tests, many of the differences between upper and lower plume (S0, thiosulfate and hydrogen) were significant (P<0.05), and none of the differences between plume and background were significant (P>0.05) (Supplementary Figure S7). Data from individual samples showed that differences between lower and upper plume were pronounced at some sites, such as Abe, where genes for S0 oxidation increased in abundance by sevenfold from the lower rising plume (1 m) to the upper rising plume (200 m) (Supplementary Figure S8).
Figure 5.
Genomic abundance of organisms utilizing six abundant electron donors in ELSC hydrothermal plumes. Size of the bubble indicates total abundance in the community of identified bins as indicated in the legend. Numbers inside the circles indicate the number of distinct genomic bins associated with the oxidation of the respective electron donors.
Discussion
The ELSC hosts hydrothermal vent fields on a north–south axis along which changes in host rock chemistry shape the geochemistry of hydrothermal vent fluids. We used metagenomics to study the impact of these geochemical gradients on the genetic and metabolic potential of ELSC plume microbial communities. Through a combination of de novo genomic assembly and tetranucleotide binning, distinct genomic bins were identified, resulting in the genomic resolution of taxonomic groups. These results provide insights into the energy metabolism and functional biogeochemical roles of microorganisms in ELSC plumes, many of which are from novel microbial groups that are globally distributed in hydrothermal plumes and the pelagic deep oceans. Our results show that although ELSC plumes are dominated by populations previously reported to be abundant and ubiquitous in the deep water column, such as SUP05, SAR324, Thaumarchaea (Dick and Tebo, 2010; Lesniewski et al., 2012) and SAR11 (Thrash et al., 2014), they also contain populations such as Epsilonproteobacteria, Aquificae, Chloroflexi and Planctomycetes that are likely indigenous to hydrothermal environments such as vent chimneys and extinct sulfides (Flores et al., 2012; Sylvan et al., 2013). These results reinforce links between plumes and seafloor habitats that were hypothesized long ago (Winn et al., 1986) and further supported recently by a 16 S rRNA gene study of our samples (Sheik et al., 2015).
The most abundant members of microbial communities of the five different hydrothermal plumes at ELSC are remarkably similar and dominated by sulfur-oxidizing bacteria. This indicates that the high concentration and bioenergetic potential of sulfur in the water column overprints the geochemical differences in host rocks and end-member hydrothermal fluid chemistry along the ELSC (Mottl et al., 2011), which have been found to influence microbial diversity on the seafloor (Flores et al., 2012; Sylvan et al. 2013). These results are consistent with a gene-centric mathematical model of the Abe vent at ELSC, which shows that sulfur metabolisms dominate and that seafloor populations were rapidly diluted out of the rising plume (Reed et al., 2015) and also with more extensive analysis of ELSC plumes, showing that differences in community composition between vent fields are not significant (Sheik et al., 2015). Observed variation in the abundance of specific genes across specific samples is to be expected given the dynamic nature of plumes, and could reflect variation in the proportions of different water sources (end-member hydrothermal fluid, plume and background), which is a key determinant of plume community structure (Reed et al., 2015). Some consistent differences in the membership of dominant organisms were observed between vent fields, chiefly the increased abundance of Alteromonas and Marinobacter populations at Mariner (Sheik et al., 2015; Figure 2). These microbial groups have been shown to be involved in iron uptake (Li et al., 2014b) and iron and manganese oxidation (Edwards et al., 2004; Singer et al., 2011), respectively, and their increased abundance at Mariner correlates with increased iron and manganese concentrations. However, elucidation of abundance of genes associated with iron and manganese oxidation in ELSC plumes was not possible as these genes are highly divergent and are yet to be identified comprehensively and conclusively.
Although many of the dominant microbial populations found in Guaymas Basin plumes (Dick and Tebo, 2010; Lesniewski et al., 2012) are also abundant at ELSC, methanotrophs that were diverse and abundant at Guaymas Basin (Li et al., 2014a) are present only at low abundance at ELSC (Figure 5). Methane concentrations of 5–57 μM in the hydrothermal fluids of the ELSC are 3 orders of magnitude lower than sediment-hosted hydrothermal vents of Guaymas Basin. Bioenergetic modeling highlights this difference in the two vent systems, indicating that methane oxidation potentially contributes up to 93% of all available energy at Guaymas Basin (Anantharaman et al., 2013), but only about 1% of the total available free energy from catabolic reactions at ELSC (Figure 4).
We observed the presence of multiple pathways for lithotrophy in many genomes (Figure 2), including potential for oxidation of both sulfur and H2 in SUP05, SAR324, Epsilonproteobacteria and Aquificales, and oxidation of methane and sulfur in Methylococcaceae. This suggests that metabolic versatility is a common strategy in deep-sea microorganisms. While the ability to grow on multiple electron donors has been reported many times (Nakagawa et al., 2005; Petersen et al., 2011; Anantharaman et al., 2013; Sheik et al., 2014), to our knowledge this is the first report of potential for lithotrophy in a methanotroph. This suggests that facultative methanotrophy extends beyond utilization of organic acids and ethanol (Semrau et al., 2011) and includes sulfur as an alternative energy source. Given the widespread nature of OPU-3 Methylococcaceae in marine environments (Tavormina et al., 2010; Kessler et al., 2011; Rivers et al., 2013; Li et al., 2014a), further studies are warranted to test whether it is capable of both methane and sulfur oxidation as suggested by our data. An important implication of this metabolic plasticity is that the proliferation of some genes (for example, CH4 or H2 oxidation) could be linked to growth on other substrates (for example, sulfur) (Reed et al., 2015). Because the data presented here only reveals metabolic potential, we are unable to determine whether these metabolisms are simultaneously active in the same sample as observed previously for H2 and S oxidation by SUP05 (Petersen et al., 2011; Anantharaman et al., 2013), or whether they are employed separately in distinct geochemical environments.
Metabolic versatility associated with sulfur oxidation may impart functional redundancy (Allison and Martiny, 2008) for sulfur oxidation in ELSC plumes, similar to other sulfur-dominated systems (Jones et al., 2012). The diversity and versatility of sulfur-oxidizing microorganisms that are abundant both in ELSC plumes and in surrounding waters could make sulfur oxidation resilient in the dynamic setting of deep-sea hydrothermal plumes, allowing it to persist during long periods between sulfur availability. Such extended deficits of sulfur could be brought on by perturbations to ocean currents and hydrothermal activity.
The shotgun sequencing approach employed here yielded sequence information on the whole plume community, including all three domains of life (Bacteria, Archaea and Eukarya) and all ecological roles (primary producers, heterotrophs, grazers and viruses). The prevalence of putative viruses in the ELSC metagenomes indicates that they are abundant in hydrothermal plumes, as has been observed in the broader deep oceans (Hara et al., 1996). Indeed, detailed analysis of these viral genomes shows that they often contain sulfur oxidation genes that likely function as auxiliary metabolic genes and serve as an important reservoir of genetic diversity (Anantharaman et al., 2014). In addition, the finding of a Bathymodiolinae mussel genome, despite no visual signs of animals on the sample filters, suggests that microscopic mussel larvae and/or tissues were present in the plume, supporting the notion that hydrothermal plumes serve as a dispersal vector for animals over large oceanic distances and regimes (Mullineaux et al., 1995; Dick et al., 2013). Our results also indicate that heterotrophy is widespread in hydrothermal plumes; although most hydrothermal plume research findings have focused on primary production via chemosynthesis, clearly the fate of organic carbon as determined by heterotrophs also warrants attention. Chemosynthetic production in plumes can serve as a regionally significant source of organic carbon to the deep ocean (de Angelis et al., 1993; McCollom, 2000; Lam et al., 2008), providing fuel for heterotrophy in addition to that from sinking particles (Karl et al., 1984).
Conclusions
Overall, our study provides insights into the genomic content and metabolic potential of archaea, bacteria, eukarya and viruses that inhabit ELSC hydrothermal plumes. Oxidation of reduced sulfur species constitutes the most abundant chemolithotrophic energy metabolism in all ELSC hydrothermal plumes. Sulfur oxidation genes were observed in hydrogen- and methane-oxidizing organisms, suggesting that dominant primary producers in the ELSC hydrothermal plumes have diverse metabolic strategies. The abundance and diversity of sulfur-oxidizing microorganisms, coupled with their metabolic versatility, indicates functional redundancy of sulfur oxidation that could allow it to persist in the dynamic settings of hydrothermal plumes. Additional research is needed to understand how and why plume microbes use multiple electron donors. Metagenomic analyses and bioenergetic models also point to a comparatively minor metabolic role for hydrogen, methane and ammonia oxidation in fueling primary production at ELSC. Despite prominent differences in fluid geochemistry between sites at the ELSC, including highly enriched concentrations of iron at Mariner, the effect on the microbial community appears to be relatively minor, being masked by the dominance of sulfur metabolism. Finally, the genomic data presented here highlights opportunities for future studies with respect to the two understudied components of deep-ocean microbial communities, eukarya and viruses.
Data archiving
The nucleotide sequences are available from DOE JGI-IMG/MER—Taxon Object IDs (Kilo Moana: 3300001680, Abe: 3300001681, Mariner: 3300001678, Tahi Moana: 3300001679, Tui Malila: 3300001676 and Guaymas: 3300001683) and NCBI (BioProject: PRJNA234377, GenBank: JYMV00000000, Sequence Read Archive: SRP040975). SSU rRNA sequences are available from Genbank: KP005000-66.
Acknowledgments
This project is funded in part by the Gordon and Betty Moore Foundation and the National Science Foundation Ridge2000 (R2K) program (OCE 1038006 and OCE-1038055). We thank AL Reysenbach, M Tivey, C Fisher, P Girguis, G Luther and the Eastern Lau Spreading Center 2009 scientific parties (NSF OCE-0424953, OCE02-040985, OCE-0728391, OCE-0752469 and OCE-0751839), the captain and crew of R/V Thomas G Thompson, and the ROV Jason team. We thank J Sylvan and S White for sampling and logistical support. We also thank the University of Michigan Rackham Graduate School Faculty Research Fellowship Program for their support and Sunit Jain for his assistance with bioinformatics.
The authors declare no conflict of interest.
Footnotes
Supplementary Information accompanies this paper on The ISME Journal website (http://www.nature.com/ismej)
Supplementary Material
References
- Albertsen M, Hugenholtz P, Skarshewski A, Nielsen KL, Tyson GW, Nielsen PH. (2013). Genome sequences of rare, uncultured bacteria obtained by differential coverage binning of multiple metagenomes. Nat Biotechnol 31: 533–538. [DOI] [PubMed] [Google Scholar]
- Allers E, Wright JJ, Konwar KM, Howes CG, Beneze E, Hallam SJ et al. (2013). Diversity and population structure of Marine Group A bacteria in the Northeast subarctic Pacific Ocean. ISME J 7: 256–268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allison SD, Martiny JBH. (2008). Resistance, resilience, and redundancy in microbial communities. Proc Natl Acad Sci USA 105: 11512–11519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ. (1990). Basic local alignment search tool. J Mol Biol 215: 403–410. [DOI] [PubMed] [Google Scholar]
- Anantharaman K, Breier JA, Sheik CS, Dick GJ. (2013). Evidence for hydrogen oxidation and metabolic plasticity in widespread deep-sea sulfur-oxidizing bacteria. Proc Natl Acad Sci USA 110: 330–335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anantharaman K, Duhaime MB, Breier JA, Wendt K, Toner BM, Dick GJ. (2014). Sulfur oxidation genes in diverse deep-sea viruses. Science 344: 757–760. [DOI] [PubMed] [Google Scholar]
- Aristegui J, Gasol JM, Duarte CM, Herndl GJ. (2009). Microbial oceanography of the dark ocean's pelagic realm. Limnol Oceanogr 54: 1501–1529. [Google Scholar]
- Baker BJ, Lesniewski RA, Dick GJ. (2012). Genome-enabled transcriptomics reveals archaeal populations that drive nitrification in a deep-sea hydrothermal plume. ISME J 6: 2269–2279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baker BJ, Sheik CS, Taylor CA, Jain S, Bhasi A, Cavalcoli JD et al. (2013). Community transcriptomic assembly reveals microbes that contribute to deep-sea carbon and nitrogen cycling. ISME J 7: 1962–1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bowers TS, Von Damm KL, Edmond JM. (1985). Chemical evolution of mid-ocean ridge hot springs. Geochim Cosmochim Acta 49: 2239–2252. [Google Scholar]
- Breier JA, Rauch CG, McCartney K, Toner BM, Fakra SC, White SN et al. (2009). A suspended-particle rosette multi-sampler for discrete biogeochemical sampling in low-particle-density waters. Deep Sea Res I 56: 1579–1589. [Google Scholar]
- Breier JA, Toner BM, Fakra SC, Marcus MA, White SN, Thurnherr AM et al. (2012). Sulfur, sulfides, oxides and organic matter aggregated in submarine hydrothermal plumes at 9°50′N East Pacific Rise. Geochim Cosmochim Acta 88: 216–236. [Google Scholar]
- Canfield DE, Stewart FJ, Thamdrup B, De Brabandere L, Dalsgaard T, Delong EF et al. (2010). A cryptic sulfur cycle in oxygen-minimum-zone waters off the Chilean coast. Science 330: 1375–1378. [DOI] [PubMed] [Google Scholar]
- Ciccarelli FD, Doerks T, von Mering C, Creevey CJ, Snel B, Bork P. (2006). Toward automatic reconstruction of a highly resolved tree of life. Science 311: 1283–1287. [DOI] [PubMed] [Google Scholar]
- de Angelis MA, Lilley MD, Baross JA. (1993). Methane oxidation in deep-sea hydrothermal plumes of the endeavour segment of the Juan de Fuca Ridge. Deep Sea Res I 40: 1169–1186. [Google Scholar]
- DeLong EF, Preston CM, Mincer T, Rich V, Hallam SJ, Frigaard NU et al. (2006). Community genomics among stratified microbial assemblages in the ocean's interior. Science 311: 496–503. [DOI] [PubMed] [Google Scholar]
- Di Rienzi SC, Sharon I, Wrighton KC, Koren O, Hug LA, Thomas BC et al. (2013). The human gut and groundwater harbor non-photosynthetic bacteria belonging to a new candidate phylum sibling to Cyanobacteria. ELife 2: e01102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dick GJ, Anantharaman K, Baker BJ, Li M, Reed DC, Sheik CS. (2013). The microbiology of deep-sea hydrothermal vent plumes: ecological and biogeographic linkages to seafloor and water column habitats. Front Microbiol 4: 124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dick GJ, Andersson AF, Baker BJ, Simmons SL, Thomas BC, Yelton AP et al. (2009. a). Community-wide analysis of microbial genome sequence signatures. Genome Biol 10: R85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dick GJ, Clement BG, Webb SM, Fodrie FJ, Bargar JR, Tebo BM. (2009. b). Enzymatic microbial Mn(II) oxidation and Mn biooxide production in the Guaymas Basin deep-sea hydrothermal plume. Geochim Cosmochim Acta 73: 6517–6530. [Google Scholar]
- Dick GJ, Tebo BM. (2010). Microbial diversity and biogeochemistry of the Guaymas Basin deep-sea hydrothermal plume. Environ Microbiol 12: 1334–1347. [DOI] [PubMed] [Google Scholar]
- Distel DL, Lane DJ, Olsen GJ, Giovannoni SJ, Pace B, Pace NR et al. (1988). Sulfur-oxidizing bacterial endosymbionts: analysis of phylogeny and specificity by 16S rRNA sequences. J Bacterioll 170: 2506–2510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edgar RC. (2004). MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 32: 1792–1797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edwards KJ, Bach W, McCollom TM, Rogers DR. (2004). Neutrophilic iron-oxidizing bacteria in the ocean: their habitats, diversity, and roles in mineral deposition, rock alteration, and biomass production in the deep-sea. Geomicrobiol J 21: 393–404. [Google Scholar]
- Ferrini VL, Tivey MK, Carbotte SM, Martinez F, Roman C. (2008). Variable morphologic expression of volcanic, tectonic, and hydrothermal processes at six hydrothermal vent fields in the Lau back-arc basin. Geochem Geophys Geosyst 9: Q07022. [Google Scholar]
- Flores GE, Shakya M, Meneghin J, Yang ZK, Seewald JS, Geoff Wheat C et al. (2012). Inter-field variability in the microbial communities of hydrothermal vent deposits from a back-arc basin. Geobiology 10: 333–346. [DOI] [PubMed] [Google Scholar]
- German CR, Bowen A, Coleman ML, Honig DL, Huber JA, Jakuba MV et al. (2010). Diverse styles of submarine venting on the ultraslow spreading Mid-Cayman Rise. Proc Natl Acad Sci USA 107: 14020–14025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanson RS, Hanson TE. (1996). Methanotrophic bacteria. Microbiol Rev 60: 439–471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hara S, Koike I, Terauchi K, Kamiya H, Tanoue E. (1996). Abundance of viruses in deep oceanic waters. Marine Ecol Prog Ser 145: 269–277. [Google Scholar]
- Hügler M, Sievert SM. (2010). Beyond the Calvin cycle: autotrophic carbon fixation in the ocean. Annu Rev Marine Sci 3: 261–289. [DOI] [PubMed] [Google Scholar]
- Janecky DR, Seyfried WE. (1984). Formation of massive sulfide deposits on oceanic ridge crests—incremental reaction models for mixing between hydrothermal solutions and seawater. Geochim Cosmochim Acta 48: 2723–2738. [Google Scholar]
- Jannasch HW, Mottl MJ. (1985). Geomicrobiology of deep-sea hydrothermal vents. Science 229: 717–725. [DOI] [PubMed] [Google Scholar]
- Jones DS, Albrecht HL, Dawson KS, Schaperdoth I, Freeman KH, Pi Y et al. (2012). Community genomic analysis of an extremely acidophilic sulfur-oxidizing biofilm. ISME J 6: 158–170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones P, Binns D, Chang H-Y, Fraser M, Li W, McAnulla C et al. (2014). InterProScan 5: genome-scale protein function classification. Bioinformatics 30: 1236–1240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones WJ, Won YJ, Maas PAY, Smith PJ, Lutz RA, Vrijenhoek RC. (2006). Evolution of habitat use by deep-sea mussels. Marine Biol 148: 841–851. [Google Scholar]
- Kadko D. (1993). An assessment of the effect of chemical scavenging within submarine hydrothermal plumes upon ocean geochemistry. Earth Planet Sci Lett 120: 361–374. [Google Scholar]
- Kantor RS, Wrighton KC, Handley KM, Sharon I, Hug LA, Castelle CJ et al. (2013). Small genomes and sparse metabolisms of sediment-associated bacteria from four candidate phyla. mBio 4: e00708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karl DM, Knauer GA, Martin JH, Ward BB. (1984). Bacterial chemolithotrophy in the ocean Is associated with sinking particles. Nature 309: 54–56. [Google Scholar]
- Kessler JD, Valentine DL, Redmond MC, Du M, Chan EW, Mendes SD et al. (2011). A persistent oxygen anomaly reveals the fate of spilled methane in the deep Gulf of Mexico. Science 331: 312–315. [DOI] [PubMed] [Google Scholar]
- Konneke M, Bernhard AE, de la Torre JR, Walker CB, Waterbury JB, Stahl DA. (2005). Isolation of an autotrophic ammonia-oxidizing marine archaeon. Nature 437: 543–546. [DOI] [PubMed] [Google Scholar]
- Lam P, Cowen JP, Jones RD. (2004). Autotrophic ammonia oxidation in a deep-sea hydrothermal plume. FEMS Microbiol Ecol 47: 191–206. [DOI] [PubMed] [Google Scholar]
- Lam P, Cowen JP, Popp BN, Jones RD. (2008). Microbial ammonia oxidation and enhanced nitrogen cycling in the Endeavour hydrothermal plume. Geochim Cosmochim Acta 72: 2268–2286. [Google Scholar]
- Lesniewski RA, Jain S, Anantharaman K, Schloss PD, Dick GJ. (2012). The metatranscriptome of a deep-sea hydrothermal plume is dominated by water column methanotrophs and lithotrophs. ISME J 6: 2257–2268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li H, Durbin R. (2009). Fast and accurate short read alignment with Burrows–Wheeler transform. Bioinformatics 25: 1754–1760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li H, Handsaker B, Wysoker A, Fennell T, Ruan J, Homer N et al. (2009). The sequence Alignment/Map format and SAMtools. Bioinformatics 25: 2078–2079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li M, Jain S, Baker BJ, Taylor C, Dick GJ. (2014. a). Novel hydrocarbon monooxygenase genes in the metatranscriptome of a natural deep-sea hydrocarbon plume. Environ Microbiol 16: 60–71. [DOI] [PubMed] [Google Scholar]
- Li M, Toner BM, Baker BJ, Breier JA, Sheik CS, Dick GJ. (2014. b). Microbial iron uptake as a mechanism for dispersing iron from deep-sea hydrothermal vents. Nat Commun 5: 3192. [DOI] [PubMed] [Google Scholar]
- Luecker S, Nowka B, Rattei T, Spieck E, Daims H. (2013). The genome of Nitrospina gracilis illuminates the metabolism and evolution of the major marine nitrite oxidizer. Front Microbiol 4: 27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lücker S, Wagner M, Maixner F, Pelletier E, Koch H, Vacherie B et al. (2010). A Nitrospira metagenome illuminates the physiology and evolution of globally important nitrite-oxidizing bacteria. Proc Natl Acad Sci USA 107: 13479–13484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Markowitz VM, Ivanova NN, Szeto E, Palaniappan K, Chu K, Dalevi D et al. (2008). IMG/M: a data management and analysis system for metagenomes. Nucleic Acids Res 36: D534–D538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinez F, Taylor B, Baker ET, Resing JA, Walker SL. (2006). Opposing trends in crustal thickness and spreading rate along the back-arc Eastern Lau Spreading Center: Implications for controls on ridge morphology, faulting, and hydrothermal activity. Earth Planet Sci Lett 245: 655–672. [Google Scholar]
- McCollom T. (2000). Geochemical constraints on primary productivity in submarine hydrothermal vent plumes. Deep Sea Res II 47: 85–101. [Google Scholar]
- Mottl MJ, Seewald JS, Wheat CG, Tivey MK, Michael PJ, Proskurowski G et al. (2011). Chemistry of hot springs along the Eastern Lau Spreading Center. Geochim Cosmochim Acta 75: 1013–1038. [Google Scholar]
- Mullineaux LS, Wiebe PH, Baker ET. (1995). Larvae of benthic invertebrates in hydrothermal vent plumes over Juan de Fuca Ridge. Marine Biol 122: 585–596. [Google Scholar]
- Nakagawa S, Takai K, Inagaki F, Hirayama H, Nunoura T, Horikoshi K et al. (2005). Distribution, phylogenetic diversity and physiological characteristics of epsilon-Proteobacteria in a deep-sea hydrothermal field. Environ Microbiol 7: 1619–1632. [DOI] [PubMed] [Google Scholar]
- Namiki T, Hachiya T, Tanaka H, Sakakibara Y. (2012). MetaVelvet: an extension of Velvet assembler to de novo metagenome assembly from short sequence reads. Nucleic Acids Res 40: e155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peng Y, Leung HCM, Yiu SM, Chin FYL. (2012). IDBA-UD: a de novo assembler for single-cell and metagenomic sequencing data with highly uneven depth. Bioinformatics 28: 1420–1428. [DOI] [PubMed] [Google Scholar]
- Petersen JM, Zielinski FU, Pape T, Seifert R, Moraru C, Amann R et al. (2011). Hydrogen is an energy source for hydrothermal vent symbioses. Nature 476: 176–180. [DOI] [PubMed] [Google Scholar]
- Pinard R, de Winter A, Sarkis GJ, Gerstein MB, Tartaro KR, Plant RN et al. (2006). Assessment of whole genome amplification-induced bias through high-throughput, massively parallel whole genome sequencing. BMC Genomics 7: 216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pruesse E, Quast C, Knittel K, Fuchs BM, Ludwig W, Peplies J et al. (2007). SILVA: a comprehensive online resource for quality checked and aligned ribosomal RNA sequence data compatible with ARB. Nucleic Acids Res 35: 7188–7196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reed DC, Breier JA, Jiang H, Anantharaman K, Klausmeier CA, Toner BM et al. (2015). Predicting the response of the deep-ocean microbiome to geochemical perturbations by hydrothermal vents. ISME J; e-pub ahead of print 6 February 2015; doi:10.1038/ismej.2015.4.. [DOI] [PMC free article] [PubMed]
- Reinthaler T, van Aken HM, Herndl GJ. (2010). Major contribution of autotrophy to microbial carbon cycling in the deep North Atlantic's interior. Deep Sea Res II 57: 1572–1580. [Google Scholar]
- Rivers AR, Sharma S, Tringe SG, Martin J, Joye SB, Moran MA. (2013). Transcriptional response of bathypelagic marine bacterioplankton to the Deepwater Horizon oil spill. ISME J 7: 2315–2329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmieder R, Lim YW, Edwards R. (2012). Identification and removal of ribosomal RNA sequences from metatranscriptomes. Bioinformatics 28: 433–435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Semrau JD, DiSpirito AA, Vuilleumier S. (2011). Facultative methanotrophy: false leads, true results, and suggestions for future research. FEMS Microbiol Lett 323: 1–12. [DOI] [PubMed] [Google Scholar]
- Sheik CS, Anantharaman K, Breier JA, Sylvan JB, Dick GJ. (2015). Spatially resolved sampling reveals dynamic microbial communities in rising hydrothermal plumes across a back-arc basin. ISME J 9: 1434–1445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sheik CS, Jain S, Dick GJ. (2014). Metabolic flexibility of enigmatic SAR324 revealed through metagenomics and metatranscriptomics. Environ Microbiol 16: 304–317. [DOI] [PubMed] [Google Scholar]
- Singer E, Webb EA, Nelson WC, Heidelberg JF, Ivanova N, Pati A et al. (2011). Genomic potential of marinobacter aquaeolei, a biogeochemical ‘Opportunitroph'. Appl Environ Microbiol 77: 2763–2771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stamatakis A. (2006). RAxML-VI-HPC: maximum likelihood-based phylogenetic analyses with thousands of taxa and mixed models. Bioinformatics 22: 2688–2690. [DOI] [PubMed] [Google Scholar]
- Sunamura M, Higashi Y, Miyako C, Ishibashi J-I, Maruyama A. (2004). Two bacteria phylotypes are predominant in the Suiyo Seamount Hydrothermal Plume. Appl Environ Microbiol 70: 1190–1198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swan BK, Martinez-Garcia M, Preston CM, Sczyrba A, Woyke T, Lamy D et al. (2011). Potential for chemolithoautotrophy among ubiquitous bacteria lineages in the dark ocean. Science 333: 1296–1300. [DOI] [PubMed] [Google Scholar]
- Sylvan JB, Pyenson BC, Rouxel O, German CR, Edwards KJ. (2012). Time-series analysis of two hydrothermal plumes at 9°50′N East Pacific Rise reveals distinct, heterogeneous bacterial populations. Geobiology 10: 178–192. [DOI] [PubMed] [Google Scholar]
- Sylvan JB, Sia TY, Haddad AG, Briscoe LJ, Toner BM, Girguis PR et al. (2013). Low temperature geomicrobiology follows host rock composition along a geochemical gradient in Lau Basin. Front Microbiol 4: 61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tagliabue A, Bopp L, Dutay J-C, Bowie A, Chever F, Jean-Baptiste P et al. (2010). Hydrothermal contribution to the oceanic dissolved iron inventory. Nat Geosci 3: 252–256. [Google Scholar]
- Tavormina PL, Ussler W 3rd, Joye SB, Harrison BK, Orphan VJ. (2010). Distributions of putative aerobic methanotrophs in diverse pelagic marine environments. ISME J 4: 700–710. [DOI] [PubMed] [Google Scholar]
- Teske A, Hinrichs K-U, Edgcomb V, de Vera Gomez A, Kysela D, Sylva SP et al. (2002). Microbial diversity of hydrothermal sediments in the Guaymas Basin: evidence for anaerobic methanotrophic communities. Appl Environ Microbiol 68: 1994–2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thorvaldsdóttir H, Robinson JT, Mesirov JP. (2013). Integrative Genomics Viewer (IGV): high-performance genomics data visualization and exploration. Brief Bioinform 14: 178–192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thrash JC, Ben T, Brandon KS, Zachary CL, Tanja W, Edward FD et al. (2014). Single-cell enabled comparative genomics of a deep ocean SAR11 bathytype. ISME J 8: 1440–1451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toner BM, Fakra SC, Manganini SJ, Santelli CM, Marcus MA, Moffett JW et al. (2009). Preservation of iron(II) by carbon-rich matrices in a hydrothermal plume. Nat Geosci 2: 197–201. [Google Scholar]
- Vignais PM, Billoud B. (2007). Occurrence, classification, and biological function of hydrogenases: an overview. Chem Rev 107: 4206–4272. [DOI] [PubMed] [Google Scholar]
- Walsh DA, Zaikova E, Howes CG, Song YC, Wright JJ, Tringe SG et al. (2009). Metagenome of a versatile chemolithoautotroph from expanding oceanic dead zones. Science 326: 578–582. [DOI] [PubMed] [Google Scholar]
- Winn CD, Karl DM, Massoth GJ. (1986). Microorganisms in deep-sea hydrothermal plumes. Nature 320: 744–746. [Google Scholar]
- Wright JJ, Konwar KM, Hallam SJ. (2012). Microbial ecology of expanding oxygen minimum zones. Nat Rev Microbiol 10: 381–394. [DOI] [PubMed] [Google Scholar]
- Yamamoto M, Takai K. (2011). Sulfur metabolisms in epsilon- and gamma-Proteobacteria in deep-sea hydrothermal fields. Front Microbiol 2: 192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zellmer KE, Taylor B. (2001). A three-plate kinematic model for Lau Basin opening. Geochem Geophys Geosyst 2: 1020. [Google Scholar]
- Zerbino DR, Birney E. (2008). Velvet: algorithms for de novo short read assembly using de Bruijn graphs. Genome Res 18: 821–829. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.