Abstract
Vitamin C deficiency globally affects several hundred million people and has been associated with increased morbidity and mortality in numerous studies. In this study, bioavailability of the oxidized form of vitamin C (l-dehydroascorbic acid or DHA)—commonly found in vitamin C containing food products prone to oxidation—was studied. Our aim was to compare tissue accumulation of vitamin C in guinea pigs receiving different oral doses of either ascorbate or DHA. In all tissues tested (plasma, liver, spleen, lung, adrenal glands, kidney, muscle, heart, and brain), only sporadic differences in vitamin C accumulation from ascorbate or DHA were observed except for the lowest dose of DHA (0.25 mg/ml in the drinking water), where approximately half of the tissues had slightly yet significantly less vitamin C accumulation than from the ascorbate source. As these results contradicted data from rats, we continued to explore the ability to recycle DHA in blood, liver and intestine in guinea pigs, rats and mice. These investigations revealed that guinea pigs have similar recycling capacity in red blood cells as observed in humans, while rats and mice do not have near the same ability to reduce DHA in erythrocytes. In liver and intestinal homogenates, guinea pigs also showed a significantly higher ability to recycle DHA compared to rats and mice. These data demonstrate that DHA in guinea pigs—as in humans—is almost as effective as ascorbate as vitamin C source when it comes to taking up and storing vitamin C and further suggest that the guinea pig is superior to other rodents in modeling human vitamin C homeostasis.
Abbreviations: DHA, L-dehydroascorbic acid; RBC, erythrocyte; ODS, Osteogenic Disorder Shionogi
Keywords: Vitamin C, Bioavailability, Ascorbate recycling, Guinea pig, Rat, Mouse, Human
Graphical abstract
Highlights
-
•
Dehydroascorbic acid is an effective vitamin C source in guinea pigs.
-
•
Like in humans, efficient recycling of vitamin C has evolved in guinea pigs.
-
•
The guinea pig is a better model of human vitamin C homeostasis than rat and mouse.
1. Introduction
Ascorbate—the reduced form of vitamin C—is a powerful antioxidant and plays an important role as cofactor in many biological reactions [1]. Suboptimal levels of vitamin C have been associated with increased morbidity and mortality from conditions such as cancer and cardiovascular disease in numerous epidemiological studies [2]. Physiologically, ascorbate is under tight endogenous regulation ensuring homeostasis in plasma and tissues by a specialized set of transport proteins and in particular during deficiency, efficient reuptake in the kidneys combined with intracellular recycling by enzymatic systems capable of reducing the oxidized form of vitamin C (l-dehydroascorbic acid or DHA) back to ascorbate ensures a minimal loss of the body stores [3], [4]. In recent years, several studies have shown that ascorbate has a variety of physiological functions as co-factor in enzymatic processes such as e.g. neurotransmitter synthesis [5] and vasorelaxation [6]. A widely used approach to investigate the significance of these actions upon health and disease is to use animal models where suboptimal levels of vitamin C can be achieved by adjusting the dietary intake. Guinea pigs have been used for this purpose for decades, but also mice and rats manipulated to be devoid of the intrinsic ability to synthesize ascorbate are available [7], [8], moreover supplementation to e.g. rats and mice capable of synthesizing ascorbate has frequently been used. Yet like humans, guinea pigs have adapted to not being able to synthesize ascorbate through millions of years of evolution [9], and such absolute dependency on a dietary supply of this vital micronutrient may have strengthened the preservation mechanisms. It has been reported that DHA administered to scorbutic Osteogenic Disorder Shionogi (ODS) rats had only 10% vitamin C activity [10], however this species—and concurrent strain—has not underwent a comparable degree of evolutionary adaptation. Our aim with the present experiments was therefore to investigate the bioavailability of vitamin C forms in guinea pigs and if the recycling capacity of guinea pigs differs such that DHA—as in humans—will substitute ascorbate more effectively in this species.
2. Materials and methods
2.1. Animals
The experiment was approved by the Danish Animal Experiments Inspectorate. In the bioavailability study, 40 female Hartley guinea pigs (Charles River Laboratories, Kisslegg, Germany) weighing 550–600 g upon arrival, were marked with a subcutaneous microchip in the neck, stratified by weight and randomized into eight groups upon one week after arrival to the animal facility. All groups were provided with ad libitum access to a standard guinea pig diet devoid of vitamin C (Ssniff Spezialdiäten GmbH, Soest, Germany).
2.2. Stability of ascorbate and DHA in solution
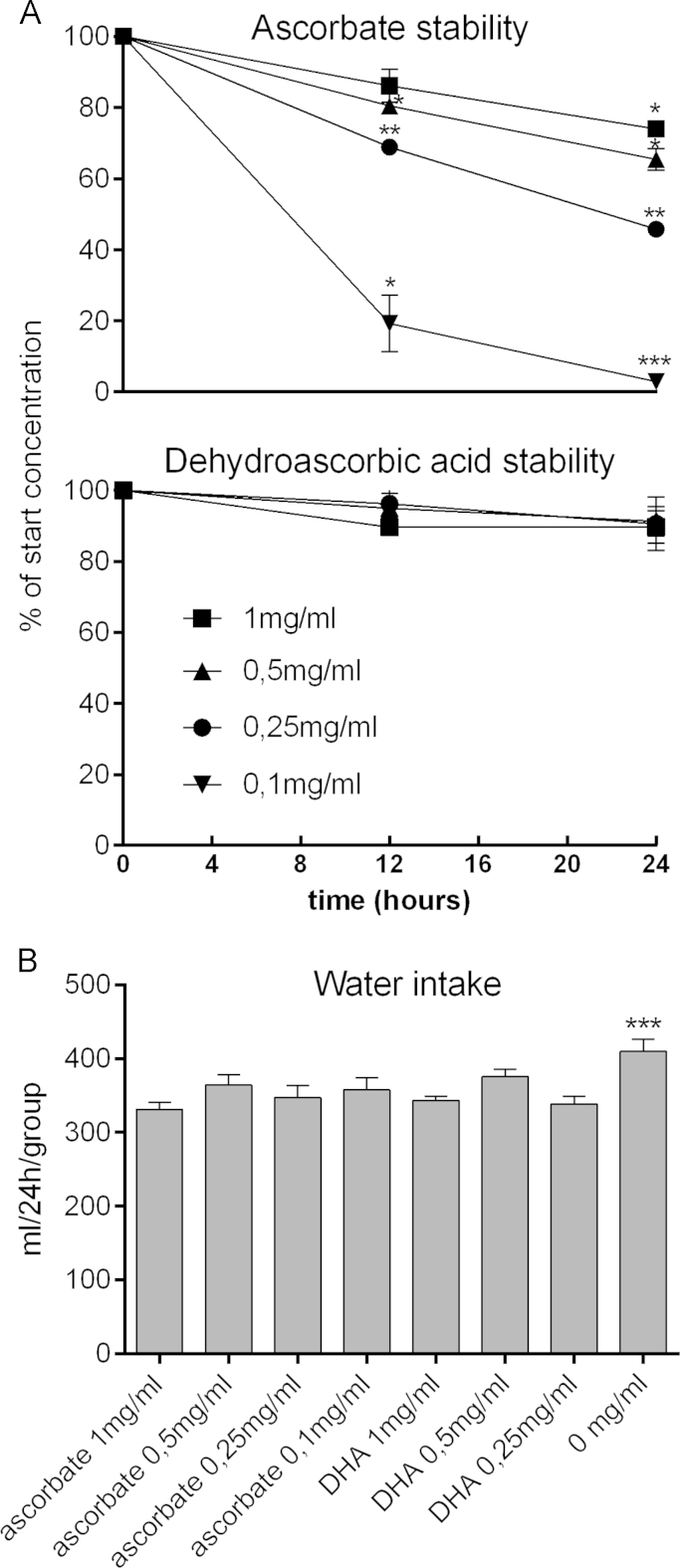
Ascorbate and DHA are both known to degrade rapidly in dilute aqueous solutions [11]. In order to test the stability prior to the in vivo experiments, all doses of ascorbate (0.1, 0.25, 0.5 and 1 mg/ml) and DHA (0.25, 0.5 and 1 mg/ml) were made freshly in MQ water, transferred to drinking bottles, and left in the animal facility for 24 h with sampling every 12 h. From Fig. 1A, it can be seen that 90% of the originally added amount of DHA was still present after 24 h irrespective of the start concentration. With ascorbate, however, there was a concentration dependent degradation with less than 50% of the initial amount still being present after 24 h for the doses of 0.25 and 0.1 mg/ml. For this reason, it was decided that animals belonging to these two groups should receive fresh solutions twice daily.
Fig. 1.
Stability of drinking solutions and water intake. In A, the stability of ascorbate and DHA, respectively, are presented over time. The solutions were freshly made and kept in drinking bottles in the animal facility. Concentrations were normalized to that of the initial preparation. B shows the daily water intake within each group. Data is presented as mean±SEM. *=p≤0.05; **=p≤0.01; ***=p≤0.001 by one-way ANOVA with Bonferroni correction for multiple comparisons.
2.3. Bioavailability study
Animals were group housed in floor pens in an enriched environment at 20±2 °C with a 12:12 h light–dark cycle, free access to hay, inspected daily by trained personnel, and weighed twice weekly. Ascorbate or DHA was supplied in the drinking water for three weeks as outlined above. Weekly blood sampling took place by puncture of Vena saphena lateralis [12] and blood was collected into K3EDTA BD Microtainer tubes [13]. Three weeks after the initiation of the experiment, the guinea pigs were preanaesthetized with Zoletil mix (Zoletil 50 vet® (125 mg Tiletamin and 125 mg zolazepam; Virbac, Carros, France) dispersed in xylazin (20 mg/ml, Rompun®, Bayer, Leverkusen, Germany) and butorphanol (10 mg/ml, Torbugesic®, ScanVet Animal Health, Fredensborg, Denmark)) and subsequently anaesthetized by isofluran inhalation (Isoba vet, MSD Animal Health, Netherlands). Upon absence of reflexes (inter-digital and skin incision) thoracotomy was performed and an intra-cardial blood sample obtained using a syringe with an 18 G, 40 mm needle flushed with 15% K3-EDTA. Immediately after sampling, the animal was euthanized by decapitation. Tissues were immediately dissected and frozen on dry ice prior to storage at −80 °C.
2.4. Recycling study
In a subsequent experiment, four male Hartley guinea pigs (5 weeks old, 400 g bodyweight) (Charles River Laboratories, Kisslegg, Germany), four male Spraque Dawley rats (7 weeks old, 200 g bodyweight (Taconic, Ejby, Denmark)) and four male black6 (B6NTac) mice (7 weeks old, 23 g bodyweight (Taconic, Ejby, Denmark)) were euthanized as described above following one week of equilibration and blood, liver and intestinal samples (sectioned from the middle third of the jejunum) were obtained. Blood samples were immediately centrifuged (5 min at 2000×g, 4 °C) and erythrocytes (RBCs) were isolated and washed three times in 10 volumes of PBS. Subsequently, RBCs were diluted to 25% with PBS and used in the recycling experiments are described below. Tissues were gently rinsed and blotted dry and subsequently homogenized in nine volumes of PBS and used directly in recycling experiments as outlined below.
2.5. Vitamin C measurements
Blood samples were immediately centrifuged for 2 min at 16,000×g (4 °C) and stabilized with ice cold 10% (w/v) MPA containing 2 mmol/L Na2-EDTA as reported previously [14] and stored at −80 °C until analysis. Tissue samples (approximately 50 mg) were taken from −80 °C and homogenized in cold (4 °C) PBS and centrifuged (15.000×g, for 1 min at 4 °C) before stabilization with MPA. The stability of ascorbate and DHA in MPA stabilized plasma has been studied previously and found adequate in preserving the in vivo equilibrium [15]. Ascorbate and total vitamin C (ascorbate+DHA; obtained by reduction with tris[2-carboxyethyl]phosphinehydrochloride as described in [16]) were quantified by high-performance liquid chromatography with coulometric determination and the DHA concentrations were subsequently assessed by subtraction of ascorbate from total vitamin C using uric acid as endogenous internal standard [15].
2.6. Ascorbate recycling measurements
Recycling capacity of DHA to ascorbate in RBCs was essentially assessed as described previously [17]. In brief, RBCs (6.25% hematocrit) were preconditioned for 20 min at 37 °C under gentle rocking, after which they were incubated for 20 min in the presence of 100 µM DHA. Aliquots were taken at t=0 and 20 min and analyzed for ascorbate as described above. For intestinal and liver tissue, 250 µl tissue homogenate was diluted with 710 µl PBS and preincubated for 20 min at 37 °C, after which 40 µl 2.5 mM DHA was added for a final concentration of 100 µM. Following a 20 min incubation period, the accumulated ascorbate was measured.
2.7. Statistics
In the water stability experiment, one-sided t-tests were used to compare the observed level to the initial 100%. To compensate for multiple comparisons, a Bonferroni factor of 4 for the ascorbate solutions and 3 for the DHA solutions was applied to the p-values (Fig. 1). Body and organ weights (Table 1) and the relative oral bioavailability of ascorbate and DHA in tissues (Table 2) were analyzed by one-way ANOVA using Bonferroni correction for multiple comparisons. The data obtained in the recycling experiments were not normally distributed (Fig. 3). Thus, data were compared by Kruskal–Wallis multiple comparison test for non-parametric data. Finally, ascorbate concentrations between species were analyzed by two-way ANOVA using tissue and species as factors (Table 3). A p-value less than 0.05 was considered statistically significant.
Table 1.
Body- and organ weight.
| Dose (mg/ml) | Bodyweight at experiment start (g) | Bodyweight at euthanization (g) | Liver (g) | Spleen (g) | Kidneys (g) | Brain (g) | Lungs (g) | Adrenal glands (g) | Heart (g) |
|---|---|---|---|---|---|---|---|---|---|
| 0 mg/ml (n=5) | 575±41 | 620±42 | 16.9±0.8 | 1.20±0.37 | 4.18±0.59 | 4.20±0.16 | 3.62±0.31 | 0.299±0.051 | 1.56±0.18 |
| Ascorbate 1 mg/ml (n=5) | 562±20 | 647±32 | 19.5±4.0 | 0.68±0.04** | 3.92±0.23 | 3.85±0.07 | 3.48±0.50 | 0.285±0.070 | 1.60±0.16 |
| Ascorbate 0.5 mg/ml (n=5) | 553±44 | 629±59 | 17.0±4.4 | 0.74±0.19** | 4.06±0.75 | 4.06±0.27 | 3.36±0.36 | 0.270±0.051 | 1.58±0.25 |
| Ascorbate 0.25 mg /ml (n=5) | 568±13 | 668±26 | 19.1±2.6 | 0.74±0.05* | 3.82±0.13 | 3.94±0.15 | 3.46±0.26 | 0.278±0.020 | 1.60±0.07 |
| Ascorbate 0.1 mg/ml (n=5) | 566±46 | 636±60 | 16.0±2.2 | 0.72±0.08** | 3.80±0.37 | 4.03±0.27 | 3.34±0.29 | 0.231±0.031 | 1.54±0.18 |
| DHA 1 mg/ml (n=5) | 574±32 | 674±49 | 19.1±2.8 | 0.86±0.15 | 4.02±0.30 | 4.02±0.11 | 3.52±0.31 | 0.256±0.035 | 1.64±0.13 |
| DHA 0.5 mg/ml (n=5) | 563±20 | 666±23 | 18.0±1.0 | 0.90±0.22 | 4.02±0.19 | 4.00±0.10 | 3.48±0.19 | 0.271±0.037 | 1.58±0.23 |
| DHA 0.25 mg/ml (n=5) |
553±25 |
663±31 |
18.9±0.5 |
0.80±0.10 |
4.16±0.21 |
3.91±0.10 |
3.52±0.08 |
0.256±0.047 |
1.64±0.17 |
Bodyweights from the beginning and end of the experiment are provided. The individual organ weights were assessed upon dissection. *=p≤0.05; **p≤0.01 as compared to “0 mg/ml” by one-way ANOVA using Bonferroni correction for multiple comparisons.
Table 2.
Statistics for Fig. 2.
| Statistical analysis | Test parameters | Water supplement | Plasma | Liver | Spleen | Lung | Adrenal glands | Kidney | Muscle | Heart | Cerebellum | Cortex |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| One way ANOVA | Difference between doses on tissue total vitamin C tissue levels | Ascorbate | *** | *** | *** | *** | *** | *** | n.s. | *** | ** | ** |
| DHA | *** | *** | n.s. | * | *** | *** | *** | *** | n.s. | n.s. | ||
| Difference between doses on tissue ASCORBATE levels | Ascorbate | *** | *** | *** | ** | *** | *** | n.s. | *** | ** | ** | |
| DHA | ** | *** | * | n.s. | *** | *** | ** | *** | n.s. | n.s. | ||
| Difference between doses on tissue DHA levels | Ascorbate | n.s. | n.s. | n.s. | * | n.s. | n.s. | n.s. | *** | n.s. | n.s. | |
| DHA | n.s. | n.s. | n.s. | * | n.s. | n.s. | *** | *** | ** | n.s. | ||
| Selected Bonferroni comparisons following one way ANOVA |
ascorbate versus DHA dose upon total vitamin C tissue levels | 1 mg/ml | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. |
| 0.5 mg/ml | *** | n.s. | n.s. | n.s. | n.s. | * | n.s. | n.s. | n.s. | n.s. | ||
| 0.25 mg/ml | n.s. | ** | n.s. | ** | n.s. | *** | n.s. | *** | n.s. | n.s. | ||
| ascorbate versus DHA dose upon ascorbate tissue levels | 1 mg/ml | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. | |
| 0.5 mg/ml | *** | n.s. | n.s. | n.s. | n.s. | * | n.s. | n.s. | n.s. | n.s. | ||
| 0,25 mg/ml | n.s. | ** | n.s. | n.s. | n.s. | *** | n.s. | n.s. | n.s. | n.s. | ||
| ascorbate versus DHA dose upon DHA tissue levels |
1 mg/ml | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. | |
| 0.5 mg/ml | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. | ||
| 0.25 mg/ml |
n.s. |
n.s. |
n.s. |
n.s. |
n.s. |
n.s. |
n.s. |
n.s. |
n.s. |
n.s. |
The data of Fig. 2 was analyzed by one-way ANOVA using dose as factor and subsequently for the comparison of ascorbate to DHA accumulation using Bonferroni correction for multiple comparisons. For both types of tests *=p≤0.05; **=p≤0.01; and ***=p≤0.001, whereas n.s.=non significant.
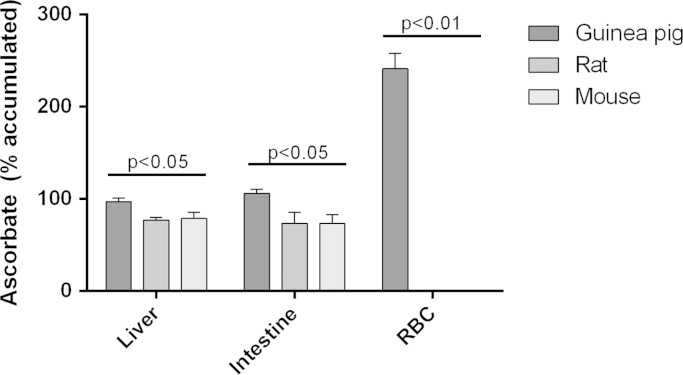
Fig. 3.
Recycling capacity in liver, intestine and erythrocytes of guinea pig, rat and mouse. Liver or intestinal homogenate or isolated erythrocytes were incubated with 100 µmol/L dehydroascorbic acid for 20 min and the accumulation of ascorbate was expressed as % of initial concentration. Erythrocytes from rats and mice did not contain any initial ascorbate, nor did they accumulate any following incubation. Data were compared by Kruskal–Wallis multiple comparison test for non-parametric data.
Table 3.
Vitamin C concentrations in liver and intestine from guinea pig, rat and mouse. No statistically significant differences were observed by two-way ANOVA using tissue and species as factors.
| Guinea pig | Rat | Mouse | |
|---|---|---|---|
| Liver Ascorbate (nmol/g tissue) | 1031±281 | 844±280 | 707±197 |
| Intestinal Ascorbate (nmol/g tissue) |
629±105 |
700±178 |
857±183 |
3. Results
3.1. Water intake
The average daily water intake within each group can be seen in Fig. 1B. The guinea pigs on vitamin C deficient diet drank slightly but significantly more water than the groups receiving ascorbate or DHA supplementation. There was no difference in the water intake between the dosage groups.
3.2. Body and tissue weights
The guinea pigs were randomized and stratified for weight at the beginning of the study and the bodyweight was monitored twice weekly. As can be seen in Table 1, there was no difference between groups neither at start nor at euthanization. Yet the weight of the depleted guinea pigs (0 mg/ml group) started to stagnate a few days prior to euthanization (data not shown). The weights of the organs, given in Table 1, were not affected by the different doses except for the spleens in the depleted group, which were significantly enlarged.
3.3. Tissue vitamin C levels
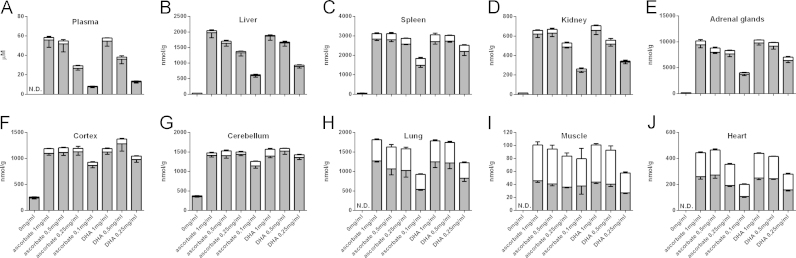
In the tissues shown in Fig. 2 (plasma, liver, spleen, kidney, adrenal glands, cortex and cerebellum from the brain, lung, heart, and skeletal muscle), vitamin C levels were evaluated as ascorbate alone, DHA alone, and as total vitamin C levels (ascorbate+DHA), respectively. Statistics from these data are given in Table 2. In all tissues, we found a significant effect of dose on one or more of the measures of vitamin C content with both ascorbate and DHA supplementation reflecting that a dose response relationship was present. In half of the tissues investigated, we found no significant difference between ascorbate and DHA dosing when given in equivalent concentrations, whereas there were significant differences with only the lower doses in plasma, liver, lung, kidney and heart. This reflects that DHA is almost as effective as ascorbate when it comes to provide vitamin C accumulation within tissues. From Fig. 2, it is also apparent that lungs, heart, and muscle contain more vitamin C in the form of DHA than the remaining tissues. Regarding the plasma levels of vitamin C there was no difference in ascorbate, DHA or total vitamin C levels, respectively, between any of the groups at the start of the experiment (one way ANOVA, data not shown) and the levels of vitamin C adjusted within the first week after which they remained relatively constant (data not shown).
Fig. 2.
Tissue vitamin C levels. Vitamin C levels in plasma (A), liver (B), spleen (C), kidney (D), adrenal glands (E), cortex (F), cerebellum (G), lung (H), heart (I), and skeletal muscle (J). Vitamin C levels are depicted as ascorbate (gray bar part), oxidized vitamin C (DHA) (white bar part), and total vitamin C (total bar height). N.D.=Not depicted. Statistics are present in Table 2. Data is presented as mean±SEM.
3.4. Recycling capacity in RBC, liver and intestine
The recycling capacity was assessed in RBCs, liver, and intestine in guinea pigs, rats, and mice as described above. The results are shown in Fig. 3. Significant effects of species was observed for both RBC, liver, and intestinal recycling capacity (p<0.05 or less). Only guinea pigs were able to recycle DHA to ascorbate in RBCs (p<0.01). For liver and intestinal tissue, the rats and mice were unable to maintain their vitamin C pool reduced as indicated by the fact that the ascorbate concentration dropped during the 20 min incubation period. In guinea pigs, the vitamin C pool was intact after 20 min suggesting an ability to balance in situ oxidation and recycling. Initial levels of tissue ascorbate are found in Table 3. Washed RBCs did not contain ascorbate and no statistically significant differences were observed by two-way ANOVA using tissue and species as factors.
4. Discussion
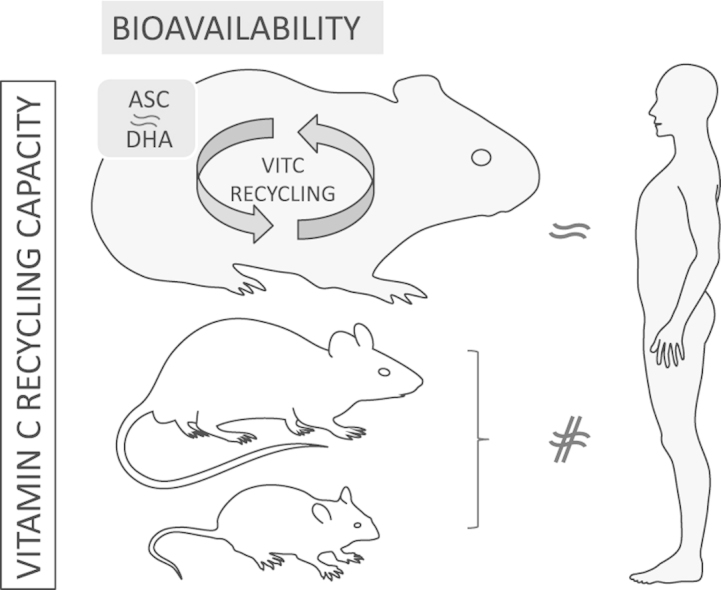
The present study demonstrates that DHA constitutes an equally good vitamin C source as its reduced form ascorbate in guinea pigs. Moreover, our data suggest that the guinea pig is superior to mouse and rat and more similar to humans when it comes to the ability to recycle vitamin C.
The presence of DHA in foods containing vitamin C due to oxidation has been widely acknowledged and several decades ago, it was found that DHA could indeed substitute vitamin C in humans [18], [19], [20]; findings that have been repeated in recent years as well [21]. Even though the uptake mechanisms differ with ascorbate being actively taken up by the sodium dependent vitamin C transporters (SVCTs) and DHA utilizing facilitated diffusion into cells through the glucose transporters (GLUT), the ability of cells to almost instantly reduce DHA back to ascorbate suggests that DHA would indeed be expected to be as effective as ascorbate [22]. To the best of our knowledge only very few studies exist that directly compare DHA to ascorbate administration in animal models whereof the majority is several decades old [10], [23], [24], [25], [26], [27], [28], [29]. In general, it is reported that DHA is as effective as ascorbate in raising vitamin C levels, yet there are a few exceptions: Ogiri et al. showed that DHA can only account for 10% of vitamin C activity in the OSD rat [10], and Hornig et al. found that isotope labeled DHA appeared slower and to a smaller extent in several tissues upon IV injection in guinea pigs. There seems to be a common consensus that the conversion of administered DHA to ascorbate depends on the route of administration with differences in appearance and distribution pattern when using oral ingestion, IP, and IV, respectively [26], [27], [28]. However, we consider oral ingestion as the most relevant route of administration when comparing ascorbate and DHA as this mimics uptake in humans, and we hypothesized that the conflicting results obtained by Ogiri et al. [10] may rather relate to the rat being less efficient in recycling DHA compared to species like humans and guinea pigs that lost the ability to synthesize vitamin C millions of years ago and where a compensatory increase in recycling capacity would be likely to have evolved. One example could be the RBCs, where guinea pigs in this study showed a recycling capacity comparable to that of humans, while rats and mice did not have a significant capacity to recycle DHA. We have previously shown that the RBCs of guinea pigs have the capacity to recycle the entire vitamin C pool of the blood once approximately every three minutes and similar data have been found in humans suggesting that in these species, RBCs constitute a substantial antioxidant reserve in vivo [30], [31].
When investigating the potential impact of vitamin C supplementation and vitamin C deficiency in relation to human disease development, we recommend that great care should be taken in choosing the right animal model. Transgenic technology in mice and rats certainly allows for mechanistic elucidations that would otherwise be hard to obtain (e.g. [8]), yet it might have significant impact on the results when a species with less capacity for recycling vitamin C is used to extrapolate to the human situation. However, a limitation of our study is that we have not assessed if the variety of transgenic rats and mice used in vitamin C research have in fact the same recycling capacity as their respective wild types.
Finally, when comparing the different tissues, it immediately becomes apparent from Fig. 2 that the brain is far more effective in retaining vitamin C as it firstly does not become depleted in the unsupplemented group and, secondly, that the tissue concentration remains relatively constant even with 10 times lower doses (1 mg/ml versus 0.1 mg/ml). The capacity of the brain to hold on to vitamin C is widely acknowledged [32], [33], [34] and our confirmatory finding supports the physiological relevance of the data. Moreover, we recently reported that saturation of the brain occurs faster than for most other tissues [35]. It can also be extracted from Fig. 2 that lung, muscle, and heart are less efficient in keeping the DHA/total vitamin C ratio low presumably due to a higher turnover driven by the high oxygen tension of these tissues.
In conclusion, we find that DHA can substitute ascorbate as vitamin C source with respect to tissue uptake and storage in guinea pigs as in humans and that they appear to have similar capacity to recycle vitamin C as opposed to mice and rats. This may render the guinea pig as a more appropriate animal model when studying in vivo effects of vitamin C recycling.
Conflicts of interest
The authors declare no conflicts of interest that could affect the present work.
Acknowledgments
We would like thank Annie Bjergby Kristensen and Joan Frandsen for excellent technical assistance in relation to the recycling experiments and Belinda Bringtoft, Maiken Lindblad and Stine Hasselholt for their assistance with the animals. This study was supported by the Danish National Research Councils (271-08-0763) and the Lifepharm Centre for in vivo pharmacology.
References
- 1.Lykkesfeldt J., Michels A.J., Frei B. Vitamin C. Adv. Nutr. 2014;5:16–18. doi: 10.3945/an.113.005157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Frei B., Birlouez-Aragon I., Lykkesfeldt J. Authors' perspective: what is the optimum intake of vitamin C in humans? Crit. Rev. Food Sci. Nutr. 2012;52:815–829. doi: 10.1080/10408398.2011.649149. [DOI] [PubMed] [Google Scholar]
- 3.May J.M., Cobb C.E., Mendiratta S., Hill K.E., Burk R.F. Reduction of the ascorbyl free radical to ascorbate by thioredoxin reductase. J. Biol. Chem. 1998;273:23039–23045. doi: 10.1074/jbc.273.36.23039. [DOI] [PubMed] [Google Scholar]
- 4.Wells W.W., Xu D.P., Washburn M.P. Glutathione: dehydroascorbate oxidoreductases. Methods Enzymol. 1995;252:30–38. doi: 10.1016/0076-6879(95)52006-6. [DOI] [PubMed] [Google Scholar]
- 5.Menniti F.S., Knoth J., Diliberto E.J., Jr. Role of ascorbic acid in dopamine beta-hydroxylation. The endogenous enzyme cofactor and putative electron donor for cofactor regeneration. J. Biol. Chem. 1986;261:16901–16908. [PubMed] [Google Scholar]
- 6.Tveden-Nyborg P., Lykkesfeldt J. Does vitamin C deficiency increase lifestyle-associated vascular disease progression? Evidence based on experimental and clinical studies. Antioxidants Redox Signal. 2013;19:2084–2104. doi: 10.1089/ars.2013.5382. [DOI] [PubMed] [Google Scholar]
- 7.Kawai T., Nishikimi M., Ozawa T., Yagi K. A missense mutation of l-gulono-gamma-lactone oxidase causes the inability of scurvy-prone osteogenic disorder rats to synthesize l-ascorbic acid. J. Biol. Chem. 1992;267:21973–21976. [PubMed] [Google Scholar]
- 8.Maeda N., Hagihara H., Nakata Y., Hiller S., Wilder J., Reddick R. Aortic wall damage in mice unable to synthesize ascorbic acid. Proc. Natl. Acad. Sci. USA. 2000;97:841–846. doi: 10.1073/pnas.97.2.841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Yang H. Conserved or lost: molecular evolution of the key gene GULO in vertebrate vitamin C biosynthesis. Biochem. Genet. 2013;51:413–425. doi: 10.1007/s10528-013-9574-0. [DOI] [PubMed] [Google Scholar]
- 10.Ogiri Y., Sun F., Hayami S., Fujimura A., Yamamoto K., Yaita M., Kojo S. Very low vitamin C activity of orally administered l-dehydroascorbic acid. J. Agric. Food Chem. 2002;50:227–229. doi: 10.1021/jf010910f. [DOI] [PubMed] [Google Scholar]
- 11.Lykkesfeldt J., Loft S., Poulsen H.E. Determination of ascorbic acid and dehydroascorbic acid in plasma by high-performance liquid chromatography with coulometric detection – are they reliable biomarkers of oxidative stress? Anal. Biochem. 1995;229:329–335. doi: 10.1006/abio.1995.1421. [DOI] [PubMed] [Google Scholar]
- 12.Birck M.M., Tveden-Nyborg P., Lindblad M.M., Lykkesfeldt J. Non-terminal blood sampling techniques in guinea pigs. J. Vis. Exp. 2014:e51982. doi: 10.3791/51982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lykkesfeldt J. Ascorbate and dehydroascorbic acid as biomarkers of oxidative stress: validity of clinical data depends on vacutainer system used. Nutr. Res. 2012;32:66–69. doi: 10.1016/j.nutres.2011.11.005. [DOI] [PubMed] [Google Scholar]
- 14.Lykkesfeldt J., Determination of ascorbic acid and dehydroascorbic acid in biological samples by high-performance liquid chromatography using subtraction methods: reliable reduction with Tris[2-carboxyethyl]phosphine hydrochloride [published erratum appears in Anal. Biochem. 2001; 292, 173]. Analytical Biochemistry, 2000, 282. pp. 89–93. [DOI] [PubMed]
- 15.Lykkesfeldt J. Ascorbate and dehydroascorbic acid as reliable biomarkers of oxidative stress: analytical reproducibility and long-term stability of plasma samples subjected to acidic deproteinization. Cancer Epidemiol. Biomark. Prev. 2007;16:2513–2516. doi: 10.1158/1055-9965.EPI-07-0639. [DOI] [PubMed] [Google Scholar]
- 16.Lykkesfeldt J. Determination of ascorbic acid and dehydroascorbic acid in biological samples by high-performance liquid chromatography using subtraction methods: reliable reduction with tris[2-carboxyethyl]phosphine hydrochloride. Anal. Biochem. 2000;282:89–93. doi: 10.1006/abio.2000.4592. [DOI] [PubMed] [Google Scholar]
- 17.Lykkesfeldt J., Viscovich M., Poulsen H.E. Ascorbic acid recycling in human erythrocytes is induced by smoking in vivo. Free Radic. Biol. Med. 2003;35:1439–1447. doi: 10.1016/j.freeradbiomed.2003.08.006. [DOI] [PubMed] [Google Scholar]
- 18.Linkswiler H. The effect of the Ingestion of ascorbic acid and dehydroascorbic acid upon the blood levels of these two components in human subjects. J. Nutr. 1958;64:43–54. doi: 10.1093/jn/64.1.43. [DOI] [PubMed] [Google Scholar]
- 19.Sabry J.H., Fisher K.H., Dodds M.L. Human utilization of dehydroascorbic acid. J. Nutr. 1958;64:457–466. doi: 10.1093/jn/64.3.457. [DOI] [PubMed] [Google Scholar]
- 20.Todhunter E.N., Mc M.T., Ehmke D.A. Utilization of dehydroascorbic acid by human subjects. J. Nutr. 1950;42:297–308. doi: 10.1093/jn/42.2.297. [DOI] [PubMed] [Google Scholar]
- 21.Tsujimura M., Higasa S., Nakayama K., Yanagisawa Y., Iwamoto S., Kagawa Y. Vitamin C activity of dehydroascorbic acid in humans--association between changes in the blood vitamin C concentration or urinary excretion after oral loading. J. Nutr. Sci. Vitaminol. 2008;54:315–320. doi: 10.3177/jnsv.54.315. [DOI] [PubMed] [Google Scholar]
- 22.Lindblad M., Tveden-Nyborg P., Lykkesfeldt J. Regulation of vitamin C homeostasis during deficiency. Nutrients. 2013;5:2860–2879. doi: 10.3390/nu5082860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Langham M. The transfer of l-ascorbic acid and dehydro-l-ascorbic acid into the aqueous humour of the rabbit and cat. J. Physiol. 1950;111:388–393. doi: 10.1113/jphysiol.1950.sp004489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Cui Y., Otsuka M., Fujiwara Y. Reduction of dehydroerythorbic acid in vitamin C-deficient guinea pigs. J. Nutr. Sci. Vitaminol. 2001;47:316–320. doi: 10.3177/jnsv.47.316. [DOI] [PubMed] [Google Scholar]
- 25.Hirst E.L. Ascorbic acid as the antiscorbutic factor. Biochem. J. 1933;27:1271–1278. doi: 10.1042/bj0271271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hornig D., Weber F., Wiss O. Uptake and release of [I–14C]ascorbic acid and [I-14C]dehydroascorbic acid by erythrocytes of guinea pigs. Clin. Chim. Acta. 1971;31:25–35. doi: 10.1016/0009-8981(71)90358-5. [DOI] [PubMed] [Google Scholar]
- 27.Hornig D., Weber F., Wiss O. Tissue distribution of labelled material in vitamin C-deficient guinea pigs after intravenous injection of 1–14C) ascorbic acid or (1–14C) dehydroascorbic acid. Int. J. Vitam. Nutr. Res. 1972;42:511–523. [PubMed] [Google Scholar]
- 28.Hornig D., Weber F., Wiss O. Studies on the distribution of (1–14C) ascorbic acid and (1–14C) dehydroascorbic acid in guinea pigs after oral application. Int. J. Vitam. Nutr. Res. 1974;44:217–229. [PubMed] [Google Scholar]
- 29.Dayton P.G., Snell M.M., Perel J.M. Ascorbic and dehydroascorbic acids in guinea pigs and rats. J. Nutr. 1966;88:338–344. doi: 10.1093/jn/88.3.338. [DOI] [PubMed] [Google Scholar]
- 30.May J.M., Qu Z.C., Whitesell R.R. Ascorbic acid recycling enhances the antioxidant reserve of human erythrocytes. Biochemistry. 1995;34:12721–12728. doi: 10.1021/bi00039a031. [DOI] [PubMed] [Google Scholar]
- 31.Lykkesfeldt J. Increased oxidative damage in vitamin C deficiency is accompanied by induction of ascorbic acid recycling capacity in young but not mature guinea pigs. Free. Radic. Res. 2002;36:567–574. doi: 10.1080/1071576022411256. [DOI] [PubMed] [Google Scholar]
- 32.Harrison F.E., May J.M. Vitamin C function in the brain: vital role of the ascorbate transporter SVCT2. Free Radic. Biol. Med. 2009;46:719–730. doi: 10.1016/j.freeradbiomed.2008.12.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Tveden-Nyborg P., Johansen L.K., Raida Z., Villumsen C.K., Larsen J.O., Lykkesfeldt J. Vitamin C deficiency in early postnatal life impairs spatial memory and reduces the number of hippocampal neurons in guinea pigs. Am. J. Clin. Nutr. 2009;90:540–546. doi: 10.3945/ajcn.2009.27954. [DOI] [PubMed] [Google Scholar]
- 34.Tveden-Nyborg P., Vogt L., Schjoldager J.G., Jeannet N., Hasselholt S., Paidi M.D., Christen S., Lykkesfeldt J. Maternal vitamin C deficiency during pregnancy persistently impairs hippocampal neurogenesis in offspring of guinea pigs. PLos One. 2012;7:e48488. doi: 10.1371/journal.pone.0048488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Hasselholt S., Tveden-Nyborg P., Lykkesfeldt J. Distribution of vitamin C is tissue specific with early saturation of the brain and adrenal glands following differential oral dose regimens in guinea pigs. Br. J. Nutr. 2015;113:1539–1549. doi: 10.1017/S0007114515000690. [DOI] [PubMed] [Google Scholar]