Abstract
Purpose
Prostate cancer is a common cancer in men in the world. It is rapidly increasing. This study investigated the incidence and mortality of prostate cancer and the relationship with the Human Development Index (HDI) and its dimensions in Asia in 2012.
Methods
The study was conducted based on data from the world data of cancer and the World Bank (including the HDI and its components). The standardized incidence and mortality rates of prostate cancer were calculated for Asian countries. The correlation between incidence, mortality rates, and the HDI and its components were assessed with the use of the correlation test, using SPSS software.
Results
There was a total of 191,054 incidences and 81,229 death were recorded in Asian countries in 2012. Among the Asian countries, the five countries with the highest standardized incidence rates of prostate cancer were Israel, Turkey, Lebanon, Singapore, and Japan, and the five countries with the highest standardized mortality rates were Turkey, Lebanon, Timor-Leste, Armenia, and the Philippines. The correlation between standardized incidence rate of prostate cancer and the HDI was 0.604 (P ≤ 0.001), with life expectancy at birth 0.529 (P = 0.002), with mean years of schooling 0.427 (P = 0.001), and with level of income per each person of the population 0.349 (P = 0.013). Also, between the standardized mortality rate and the HDI, it was 0.228 (P = 0.127).
Conclusions
A significant and positive correlation was observed between the standardized incidence rate of prostate cancer, and the HDI and its dimensions, such as life expectancy at birth, mean years of schooling, and income level of the population per each person of population. However, there was no significant correlation between the standardized mortality rate, and the HDI and its dimensions.
Keywords: Asia, Correlation, Human Development Index, Prostate cancer
1. Introduction
Prostate cancer is the sixth most common cancer in the world, the second most common cancer in men, and the most common cancer in men in Europe, North America, and parts of Africa.1, 2 The number of new cases estimated was 513,000 patients in 2000, while the number of new cases estimated was 1.1 million people in 2012. This suggests an increased incidence of prostate cancer in the past decade.3, 4 It is expected that by 2030, 1.7 million new cases and 499,000 deaths will occur in the entire world.5 The cancer will be known as the most common cancer in men in the future.4 This cancer includes 15% of all new cancer cases in men. Approximately 70% of all new cases of the cancer occur in developed countries.3 Fortunately, many people survive for many years after diagnosis. This means that cancer registration systems encompass only the two ends of the spectrum of the disease and are inadequate to determine the true burden of the disease.6 The incidence rate of prostate cancer differs about 50 times in various populations. The lowest incidence of the disease is seen in Asian countries, and included 14% of all cases in 2008, especially in Tianjin, China (1.9/100,000 person-years). The highest incidence occurred in North America and Scandinavia, especially in African-American people (137/100,000 person-years).1, 7 This might be due to the availability of screening tests used for diagnosis such as prostate specific antigen (PSA), and other factors, including nutrition, genetics, lifestyle, environmental factors, physical activity, smoking, race, and cancer registry systems.8, 9, 10 The incidence of prostate cancer is directly correlated with age. Almost 75% of new cancer cases occur in people older than 85 years. In other words, incidence of this cancer increases with the increase in life expectancy.1, 11 However, the cause of this cancer is unknown.9 One of the earliest methods of detection of this cancer is PSA,7 which has been used to screen since the late 1980s. After using the test, a significant increase was observed in the incidence of prostate cancer in 1990; then, the steep of increasing the cancer was less.12 The level of development is one of the factors that greatly affects the distribution of prostate cancer. In developed countries, prostate cancer is the most common cancer in men, while in less developed countries, the incidence is lower than that of other cancers.6 This relationship can also be considered in the context of socioeconomics in communities. People with a higher socioeconomic level have a higher incidence of the cancer.13 Several studies suggest an impact of awareness and level of income on the distribution of mortality from this type of cancer. People with a low awareness and low incomes are more likely to die from prostate cancer. These factors are barriers to treatment of cancer in these individuals.14, 15, 16
The role of the HDI, and the incidence and mortality of prostate cancer have been considered in a number of studies.17 Information on the incidence and mortality of prostate cancer can be useful for medical planning and research activities. According to the possible role of the HDI, the aim of this study was to investigate the incidence and mortality of prostate cancer and the relationship with the HDI and its dimensions in Asia in 2012.
2. Materials and methods
This study was an ecologic study in Asia for assessment of the correlation between the age-specific incidence and mortality rate (ASR) and the HDI and its details that include: life expectancy at birth, mean years of schooling, and gross national income (GNI) per capita. Data about the ASR for every Asian country for the year 2012 is obtained from the global cancer project available at http://globocan.iarc.fr/Default.aspx and data about the HDI is obtained from the Human Development Report 201318 that includes information about the HDI and its details for every country in the world for the year 2012.
2.1. Method of estimate of the ASRs in the global cancer project by the international agency for research on cancer
2.1.1. Age-specific incidence rate estimate
The methods of estimation are country-specific and the quality of the estimation depends upon the quality and on the amount of information available for each country. In theory, there are as many methods as countries, and because of the variety and the complexity of these methods, an overall quality score for the incidence and mortality estimates combined is almost impossible to establish. However an alphanumeric scoring system which independently describes the availability of incidence and mortality data has been established at the country level. The combined score is presented together with the estimates for each country, with an aim of providing a broad indication of the robustness of the estimation.
The methods to estimate the sex- and age-specific incidence rates of cancer for a specific country fall into one of the following broad categories, in the following order of priority: (1) rates projected to 2012 (38 countries); (2) most recent rates applied to the 2012 population (20 countries); (3) estimated from national mortality by modelling, using incidence mortality ratios derived from recorded data in country-specific cancer registries (13 countries); (4) estimated from national mortality estimates by modelling, using incidence mortality ratios derived from recorded data in local cancer registries in neighboring countries (9 European countries); (5) estimated from national mortality estimates using modelled survival (32 countries); (6) estimated as the weighted average of the local rates (16 countries); (7) one cancer registry covering part of a country is used as representative of the country profile (11 countries); (8) age/sex-specific rates for “all cancers” were partitioned using data on relative frequency of different cancers (by age and sex) (12 countries); and (9) the rates are those of neighboring countries or registries in the same area (33 countries).19, 20, 21
2.1.2. Age-specific mortality rate estimate
Depending on the degree of detail and accuracy of the national mortality data, six methods have been utilized in the following order of priority: (1) rates projected to 2012 (69 countries); (2) most recent rates applied to the 2012 population (26 countries); (3) estimated as the weighted average of regional rates (1 country); (4) estimated from national incidence estimates by modelling, using country-specific survival (2 countries); (5) estimated from national incidence estimates using modelled survival (83 countries); and (6) the rates are those of neighboring countries or registries in the same area (3 countries).19, 20, 21
2.2. HDI
The HDI is a composite measure of indicators along three dimensions: life expectancy, educational attainment, and command over the resources needed for a decent living. All groups and regions have seen notable improvement in all HDI components, with faster progress in low and medium HDI countries. On this basis, the world is becoming less unequal. Nevertheless, national averages hide large variations in human experience. Wide disparities remain within countries of both the North and the South; income inequality within and between many countries has been rising.17
2.3. Statistical analysis
In this study, we used to the correlation bivariate method for assessment of the correlation between the ASR and the HDI and its details that include: life expectancy at birth, mean years of schooling, and GNI per capita. Statistical significance was assumed if P < 0.05. All reported P values are two-sided. Statistical analyses were performed using SPSS (version 15.0, SPSS Inc., Chicago, IL, USA).
3. Results
A total of 191,054 prostate cancer cases were recorded in Asian countries in 2012. The five countries with the highest number of patients were Japan (55,970 cases), China (46,745 cases), India (19,095 cases), Indonesia (13,663 cases), and Turkey (12,650 cases), respectively. The five countries include a total of 148,123 cases, 77.52 percentage of all cases in Asia.
Among Asian countries, the five countries with the highest standardized incidence rates of prostate cancer were: Israel with 84.3/100,000, Turkey with 40.6/100,000, Lebanon with 37.2/100,000, Singapore with 33.1/100,000, and Japan with 30.4/100,000. The five countries with the lowest standardized incidence rates of the cancer were Bhutan with 1.2/100,000, Nepal with 1.5/100,000, Bangladesh with 1.7/100,000, Uzbekistan with 2/100,000, and Turkmenistan with 2.1/100,000 (Table 1 and Fig. 1).
Table 1.
Number, crude, and standardized incidence and mortality rates of prostate cancer in Asian countries in 2012 (sorted by age standardized rates from highest to lowest).
| Prostate – estimated incidence, all ages |
Prostate – estimated mortality, all ages |
||||||
|---|---|---|---|---|---|---|---|
| Population | N | Crude rate | ASR (W) | Population | N | Crude rate | ASR (W) |
| Israel | 4,134 | 108.8 | 84.3 | Turkey | 7,231 | 19.5 | 22.8 |
| Turkey | 12,650 | 34.0 | 40.6 | Lebanon | 411 | 19.6 | 17.1 |
| Lebanon | 807 | 38.5 | 37.2 | Timor-Leste | 33 | 5.5 | 14.2 |
| Singapore | 1,212 | 45.8 | 33.1 | Armenia | 269 | 18.6 | 13.1 |
| Japan | 55,970 | 90.9 | 30.4 | The Philippines | 2,580 | 5.3 | 11.3 |
| Republic of Korea | 10,351 | 42.7 | 30.3 | State of Palestine | 83 | 3.8 | 10.3 |
| Armenia | 476 | 32.9 | 27.4 | Indonesia | 9,191 | 7.5 | 9.8 |
| Brunei | 29 | 13.9 | 21.8 | Kazakhstan | 494 | 6.3 | 8.6 |
| Georgia | 570 | 28.1 | 18.6 | Jordan | 164 | 4.9 | 8.3 |
| The Philippines | 4,858 | 10.0 | 18.0 | Syrian Arab Republic | 484 | 4.5 | 7.7 |
| Timor-Leste | 42 | 6.9 | 16.5 | Georgia | 278 | 13.7 | 7.6 |
| Jordan | 285 | 8.6 | 15.3 | Brunei | 9 | 4.3 | 7.0 |
| State of Palestine | 130 | 6.0 | 15.2 | Bahrain | 15 | 1.8 | 6.5 |
| Kazakhstan | 1,021 | 13.0 | 14.9 | Iraq | 398 | 2.4 | 6.4 |
| Indonesia | 13,663 | 11.2 | 14.8 | Israel | 395 | 10.4 | 6.3 |
| Kuwait | 112 | 6.5 | 14.5 | Oman | 43 | 2.5 | 6.3 |
| Bahrain | 39 | 4.6 | 13.5 | Islamic Republic of Iran | 2,297 | 6.0 | 6.2 |
| Qatar | 40 | 2.7 | 13.2 | Qatar | 13 | 0.9 | 6.0 |
| Islamic Republic of Iran | 4,111 | 10.7 | 12.6 | United Arab Emirates | 26 | 0.5 | 5.6 |
| Syrian Arab Republic | 738 | 6.9 | 11.9 | Cambodia | 144 | 2.0 | 5.0 |
| Malaysia | 1,186 | 8.0 | 10.8 | Japan | 11,644 | 18.9 | 5.0 |
| Oman | 77 | 4.5 | 10.2 | Kyrgyzstan | 71 | 2.6 | 4.8 |
| United Arab Emirates | 89 | 1.6 | 10.0 | Saudi Arabia | 321 | 2.0 | 4.8 |
| Saudi Arabia | 703 | 4.4 | 9.5 | Malaysia | 501 | 3.4 | 4.6 |
| Iraq | 556 | 3.3 | 8.7 | Republic of Korea | 1,696 | 7.0 | 4.6 |
| Azerbaijan | 301 | 6.4 | 8.5 | Singapore | 169 | 6.4 | 4.5 |
| Kyrgyzstan | 126 | 4.7 | 7.3 | Azerbaijan | 170 | 3.6 | 4.5 |
| Thailand | 3,182 | 9.3 | 7.2 | Pakistan | 2,356 | 2.6 | 3.9 |
| Maldives | 8 | 4.9 | 6.4 | Afghanistan | 194 | 1.1 | 3.7 |
| Cambodia | 189 | 2.7 | 5.6 | Thailand | 1,700 | 5.0 | 3.7 |
| China | 46,745 | 6.6 | 5.3 | Myanmar | 597 | 2.5 | 3.4 |
| Pakistan | 3,041 | 3.3 | 5.3 | Kuwait | 24 | 1.4 | 3.0 |
| Myanmar | 755 | 3.1 | 4.3 | Maldives | 5 | 3.1 | 3.0 |
| India | 19,095 | 2.9 | 4.2 | India | 12,231 | 1.9 | 2.7 |
| Afghanistan | 237 | 1.4 | 3.7 | Lao PDR | 45 | 1.4 | 2.7 |
| Lao PDR | 60 | 1.9 | 3.5 | China | 22,603 | 3.2 | 2.5 |
| Vietnam | 1,275 | 2.9 | 3.4 | Vietnam | 872 | 2.0 | 2.5 |
| Mongolia | 26 | 1.9 | 3.4 | Yemen | 97 | 0.8 | 2.3 |
| Republic of Korea | 355 | 2.9 | 3.2 | Mongolia | 17 | 1.2 | 2.1 |
| Sri Lanka | 362 | 3.5 | 3.0 | Tajikistan | 32 | 0.9 | 1.9 |
| Yemen | 122 | 0.9 | 2.7 | Sri Lanka | 207 | 2.0 | 1.6 |
| Tajikistan | 47 | 1.4 | 2.3 | Uzbekistan | 129 | 0.9 | 1.5 |
| Turkmenistan | 37 | 1.5 | 2.1 | Turkmenistan | 22 | 0.9 | 1.5 |
| Uzbekistan | 189 | 1.4 | 2.0 | Republic of Korea | 146 | 1.2 | 1.3 |
| Bangladesh | 923 | 1.2 | 1.7 | Bangladesh | 717 | 0.9 | 1.2 |
| Nepal | 127 | 0.8 | 1.5 | Nepal | 103 | 0.7 | 1.2 |
| Bhutan | 3 | 0.8 | 1.2 | Bhutan | 2 | 0.5 | 0.7 |
ASR, age-specific incidence and mortality rate.
Fig. 1.
Standardized incidence and mortality rate of prostate cancer in Asia in 2012. ASR, age-specific incidence and mortality rate.
However, in 2012 in Asia, the number of deaths due to prostate cancer was 81,229 cases. The five countries with the highest number of deaths were China (22,603 cases), India (12,231 cases), Japan (11,644 cases), Indonesia (9,191 cases), and Turkey (7,231 cases). The countries included a total of 62,900 cases (77.43%) of the total mortality in Asia.
In the Asian countries, the five countries with the highest standardized mortality rates from prostate cancer were Turkey with 22.8/100,000, Lebanon with 17.1/100,000, Timor-Leste with 14.2/100,000, Armenia with 13.1/100,000, and the Philippines with 11.3/100,000. The five countries which had the lowest standardized mortality rates from the cancer were Bhutan with 0.7/100,000, Nepal with 1.2/100,000, Bangladesh with 1.2/100,000, Republic of Korea with 1.3/100,000, and Turkmenistan with 1.5/100,000 (Table 1 and Fig. 1).
In Table 2, amounts related to the HDI and its dimensions for each of the Asian countries (sorted based on the HDI) are shown. Asian countries are classified according to the HDI as follows: three countries in the very high category, four countries in the high category, 35 countries in the middle category, three countries in the low category, and one country in the unknown category.
Table 2.
Human Development Index and its components in Asian countries in 2012.
| Population | Human Development Index (HDI) | Life expectancy at birth | Mean year of schooling | Gross national income (GNI) per capita | |
|---|---|---|---|---|---|
| Very high human development | Japan | 0.912 | 83.6 | 11.6 | 32,545 |
| Republic of Korea | 0.909 | 80.7 | 11.6 | 28,231 | |
| Israel | 0.9 | 81.9 | 11.9 | 26,224 | |
| Singapore | 0.895 | 81.2 | 10.1 | 52,613 | |
| Brunei | 0.855 | 78.1 | 8.6 | 45,690 | |
| Qatar | 0.834 | 78.5 | 7.3 | 87,478 | |
| United Arab Emirates | 0.818 | 76.7 | 8.9 | 42,716 | |
| High human development | Bahrain | 0.796 | 75.2 | 9.4 | 19,154 |
| Kuwait | 0.79 | 74.7 | 6.1 | 52,793 | |
| Saudi Arabia | 0.782 | 74.1 | 7.8 | 22,616 | |
| Malaysia | 0.769 | 74.5 | 9.5 | 13,676 | |
| Kazakhstan | 0.754 | 67.4 | 10.4 | 10,451 | |
| Georgia | 0.745 | 73.9 | 12.1 | 5,005 | |
| Lebanon | 0.745 | 72.8 | 7.9 | 12,364 | |
| Islamic Republic of Iran | 0.742 | 73.2 | 7.8 | 10,695 | |
| Azerbaijan | 0.734 | 70.9 | 11.2 | 8,153 | |
| Oman | 0.731 | 73.2 | 5.5 | 24,092 | |
| Armenia | 0.729 | 74.4 | 10.8 | 5,540 | |
| Turkey | 0.722 | 74.2 | 6.5 | 13,710 | |
| Sri Lanka | 0.715 | 75.1 | 9.3 | 5,170 | |
| Medium human development | Jordan | 0.7 | 73.5 | 8.6 | 5,272 |
| China | 0.699 | 73.7 | 7.5 | 7,945 | |
| Turkmenistan | 0.698 | 65.2 | 9.9 | 7,782 | |
| Thailand | 0.69 | 74.3 | 6.6 | 7,722 | |
| Maldives | 0.688 | 77.1 | 5.8 | 7,478 | |
| Mongolia | 0.675 | 68.8 | 8.3 | 4,245 | |
| State of Palestine | 0.67 | 73 | 8 | 3,359 | |
| Philippines | 0.654 | 69 | 8.9 | 3,752 | |
| Uzbekistan | 0.654 | 68.6 | 10 | 3,201 | |
| Syrian Arab Republic | 0.648 | 76 | 5.7 | 4,674 | |
| Indonesia | 0.629 | 69.8 | 5.8 | 4,154 | |
| Kyrgyzstan | 0.622 | 68 | 9.3 | 2,009 | |
| Tajikistan | 0.622 | 67.8 | 9.8 | 2,119 | |
| Viet Nam | 0.617 | 75.4 | 5.5 | 2,970 | |
| Iraq | 0.59 | 69.6 | 5.6 | 3,557 | |
| Timor-Leste | 0.576 | 62.9 | 4.4 | 5,446 | |
| India | 0.554 | 65.8 | 4.4 | 3,285 | |
| Cambodia | 0.543 | 63.6 | 5.8 | 2,095 | |
| Lao PDR | 0.543 | 67.8 | 4.6 | 2,435 | |
| Bhutan | 0.538 | 67.6 | 2.3 | 5,246 | |
| Low human development | Bangladesh | 0.515 | 69.2 | 4.8 | 1,785 |
| Pakistan | 0.515 | 65.7 | 4.9 | 2,566 | |
| Myanmar | 0.498 | 65.7 | 3.9 | 1,817 | |
| Nepal | 0.463 | 69.1 | 3.2 | 1,137 | |
| Yemen | 0.458 | 65.9 | 5.3 | 928 | |
| Afghanistan | 0.374 | 49.1 | 3.1 | 1,000 | |
| Other countries or territories | Republic of Korea | – | – | – | – |
3.1. Standardized incidence rate and HDI
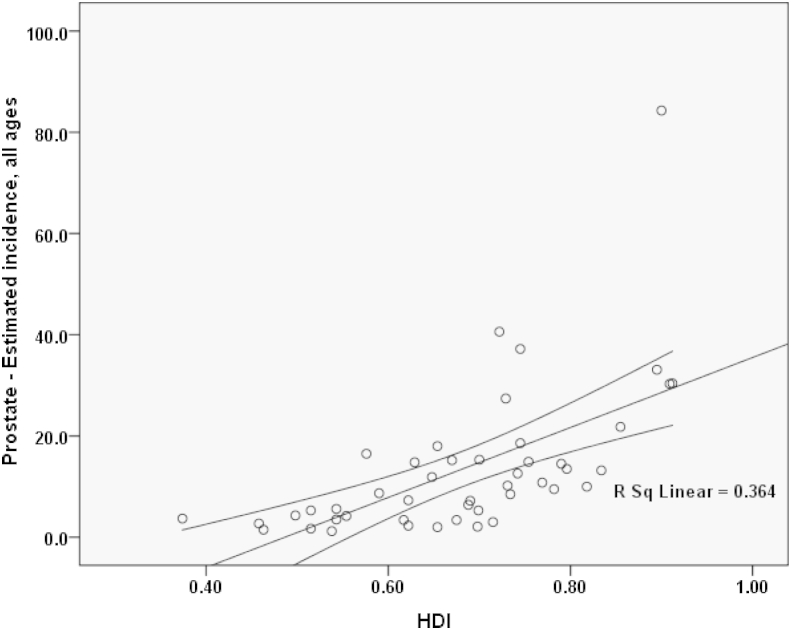
A positive correlation was seen between the standardized incidence rate of prostate cancer and HDI of ∼0.604. This association was statistically significant (P = 0.001). There was also a positive correlation between the standardized incidence rate of the cancer and dimensions of HDI. In other words, there was a positive correlation between the standardized incidence rate and life expectancy at birth of ∼0.529 (P = 0.002), A positive correlation between the standardized incidence rate and mean years of schooling of ∼0.427 (P = 0.001), and a positive correlation between the standardized incidence rate and the level of income per each person of the population equal to 0.349 (P = 0.013; Fig. 2).
Fig. 2.
Correlation between the Human Development Index and standardized incidence of prostate cancer in Asia in 2012. HDI, Human Development Index.
3.2. The standardized mortality rate and HDI
There was a positive correlation between the standardized mortality rate for prostate cancer and HDI of ∼0.228 which was not significant (P = 0.127). There was also a positive correlation between the standardized mortality rate of the cancer and the dimensions of HDI which was not significant. There was a positive correlation of 0.125 between the standardized mortality rate of the cancer and expectancy at birth (P = 0.604), a positive correlation of 0.139 between the standardized mortality rate of the cancer and mean years of schooling (P = 0.357), and a positive correlation of between the standardized mortality rate of the cancer and the level of income per each person of population (R = 0.044 and p = 0.77), which was not significant (Fig. 3).
Fig. 3.
Correlation between the Human Development Index and standardized mortality rates for prostate cancer in Asia in 2012. HDI, Human Development Index.
4. Discussion
Prostate cancer is associated with a large dispersion in incidence and death in Asian countries. The evidence suggests an increase in the burden of disease in this region in the future.6 More than 60% of the world’s population lives in Asia, and most countries in the region are developing. The cancer in the continent is expected to dramatically increase. Therefore, health macro policies to deal with this cancer in the future are an important requirement.22
Our findings showed that the incidence of prostate cancer in Asian countries is related to HDI, which determines the progress of a country in three dimensions; life expectancy, level of education, and the mean income. Countries such as Israel, Turkey, Lebanon, Singapore, Japan, and Republic of Korea had the highest standardized incidences of prostate cancer. According to the HDI, the countries are in the very high and high categories. By contrast, Tajikistan, Turkmenistan, Uzbekistan, Bangladesh, Nepal, and Bhutan had the lowest standardized incidences of prostate cancer. All of these countries in terms of the HDI are in the middle and low categories. Other studies have shown that the incidence of prostate cancer is greater in countries with high HDI.17, 22 This may be due to lifestyle, diet, and most importantly, public access to PSA and an existing cancer registry system.2 Also, differences in average life expectancy, Western foods high in calories and fat, and medical checkup for prostate cancer in Asian countries, may explain the difference in incidence in different countries.23
Life expectancy at birth is one of the dimensions of HDI. It is associated with all types of cancers, especially prostate cancer. Most cases of prostate cancer occur in old age, especially after 85 years.9 Reports revealed that Japan, Republic of Korea, Singapore, and Israel have the highest life expectancy at birth in Asian countries.24 However, in this study, these countries had a high rank in the incidence of prostate cancer among all of the Asian countries. In countries with high life expectancy associated with development, the deaths due to infectious diseases decrease. Hence, the possibility that more people reach old age is enhanced and the risk of cancer, especially prostate cancer, increases.11 Such results were seen in other studies.1, 12 According to other research, the incidence of prostate cancer is higher in urban areas than in rural areas in some Asian countries.25
Another dimension of the HDI is the level of education or awareness. A significant association was seen between the low level of knowledge and the level of PSA.14 Multiple linear regression analysis showed that in men with low levels of education compared with men who had high levels of education, PSA higher than 20 mg/mL was twice.16 People with higher education have a proper diet, more physical activity, better job opportunities, and low exposure to other risk factors for prostate cancer.9
One of the dimensions of the HDI is adequate income, which is specified by gross domestic product. Bray et al17also indicated that with rising income levels and improved levels of development, people have better access to diagnostic services and screening tests, such as PSA. As a result, many subclinical cases are detected and the incidence of the disease increases. Access to diagnostic methods and the screening test using PSA rise with increasing income levels and improved socioeconomic status.13
In our study, no relationship was observed between prostate cancer mortality rates in Asian countries and the HDI and its dimensions. In countries with lower HDI, medical checkup for prostate cancer is low and in countries with higher HDI, PSA screening and medical checkup for prostate cancer is high. So, the diagnostic rate of asymptomatic prostate cancer in early stages would be lower in lower HDI countries. Considering the lower screening rate in the lower HDI countries and the result of no significant difference in the mortality between the developed and the developing countries, the conclusion of this study seems to reflect the meta-analysis results, which is “Although PSA screening leads to over-diagnosis and over-treatment of prostate cancer, it does not contribute decreasing both prostate cancer-specific and overall mortality. Although the diagnostic rate of early stage prostate cancer by PSA screening is higher in the Asian developed countries with higher HDI, it would not improve prostate cancer-specific survival rate so much and would not lead to a significant difference in cancer mortality compared to the developing countries without an adequate screening system for prostate cancer.23
There are various effective factors such as nutrition, occupation environmental risk factors, smoking, lifestyle, and demographic factors, including age, genetic susceptibility, and socioeconomic situation, on prostate cancer.8, 10 However, the main cause of this cancer is unknown.9 Any variation in the incidence of prostate cancer and its relationship with macroeconomic determinants should be cautiously interpreted. The effect of these factors will also be considered.17
To detect early prostate cancer, screening tests should be used. Digital rectal examination (DRE) is one of the tests, which is easy. Given the low sensitivity of the test, prostate specific antigen (PSA) is another test recommended. PSA is commonly applied in the world and increases the incidence of prostate cancer (due to the diagnosis of cancer cases in the latent period). However, it reduces death.7, 26, 27
This was an ecological study. The ecological fallacy will occur if results are inferred and concluded at the individual level. The results of this study are attributable only at the population level. However, it should be stated that the cancer registry system in Asia is still not fully equal in all areas and sometimes differences in the quality and coverage of data are observed.
In conclusion, the incidence of prostate cancer is high in countries with greater development. A significant and positive correlation was observed between the standardized incidence rate of prostate cancer, and the HDI and its dimensions, such as life expectancy at birth, mean years of schooling, and income level of the population per each person of population. However, there was no significant correlation between the standardized mortality rate, and the HDI and its dimensions.
Conflicts of interest
Authors have no conflict of interest.
References
- 1.Grönberg H. Prostate cancer epidemiology. Lancet. 2003;361:859–864. doi: 10.1016/S0140-6736(03)12713-4. [DOI] [PubMed] [Google Scholar]
- 2.Hilal L., Shahait M., Mukherji D., Charafeddine M., Farhat Z., Temraz S. Prostate cancer in the Arab world: A view from the inside. Clin Genitourin Cancer. 2015 doi: 10.1016/j.clgc.2015.05.010. [DOI] [PubMed] [Google Scholar]
- 3.WHO. GLOBOCAN 2012: Estimated cancer incidence, mortality and prevalence worldwide in 2012 [Internet] Lyon: International Agency for Research on Cancer. [cited 2012]. Available from: http://globocan.iarc.fr.
- 4.Parkin D.M., Muir C.S., Whelan S., Gao Y., Ferlay J., Powell J., editors. Cancer incidence in five continents, volume VI. International Agency for Research on Cancer Scientific Publications; Lyon: 1992. [Google Scholar]
- 5.Jain S., Saxena S., Kumar A. Epidemiology of prostate cancer in India. Meta Gene. 2014;2:596–605. doi: 10.1016/j.mgene.2014.07.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Baade P.D., Youlden D.R., Cramb S.M., Dunn J., Gardiner R.A. Epidemiology of prostate cancer in the Asia-Pacific region. Prostate Int. 2013;1:47–58. doi: 10.12954/PI.12014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kitagawa Y., Mizokami A., Namiki M. Trends of clinical symptoms and prognosis of middle-aged prostate cancer patients after instigation of prostate specific antigen-based population screening. Prostate Int. 2013;1:65–68. doi: 10.12954/PI.12017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Baade P.D., Youlden D.R., Krnjacki L.J. International epidemiology of prostate cancer: geographical distribution and secular trends. Mol Nutr Food Res. 2009;53:171–184. doi: 10.1002/mnfr.200700511. [DOI] [PubMed] [Google Scholar]
- 9.Hsing A.W., Chokkalingam A.P. Prostate cancer epidemiology. Front Biosci. 2006;11:1388–1413. doi: 10.2741/1891. [DOI] [PubMed] [Google Scholar]
- 10.Zhang L., Yang B.X., Zhang H.T., Wang J.G., Wang H.L., Zhao X.J. Prostate cancer: an emerging threat to the health of aging men in Asia. Asian J Androl. 2011;13:574–578. doi: 10.1038/aja.2010.126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Pienta K.J., Esper P.S. Risk Factors for Prostate Cancer. Ann Intern Med. 1993;118:793–803. doi: 10.7326/0003-4819-118-10-199305150-00007. [DOI] [PubMed] [Google Scholar]
- 12.Yu X.Q., Luo Q., Smith D.P., Clements M.S., O'Connell D.L. Prostate cancer prevalence in New South Wales Australia: a population-based study. Cancer Epidemiol. 2015;39:29–36. doi: 10.1016/j.canep.2014.11.009. [DOI] [PubMed] [Google Scholar]
- 13.Liu L., Cozen W., Bernstein L., Ross R.K., Deapen D. Changing relationship between socioeconomic status and prostate cancer incidence. J Natl Cancer Inst. 2001;93:705–709. doi: 10.1093/jnci/93.9.705. [DOI] [PubMed] [Google Scholar]
- 14.Bennett C.L., Ferreira M.R., Davis T.C., Kaplan J., Weinberger M., Kuzel T. Relation between literacy, race, and stage of presentation among low-income patients with prostate cancer. J Clin Oncol. 1998;16:3101–3104. doi: 10.1200/JCO.1998.16.9.3101. [DOI] [PubMed] [Google Scholar]
- 15.Kim S.P., Knight S.J., Tomori C., Colella K.M., Schoor R.A., Shih L. Health literacy and shared decision making for prostate cancer patients with low socioeconomic status. Cancer Invest. 2001;19:684–691. doi: 10.1081/cnv-100106143. [DOI] [PubMed] [Google Scholar]
- 16.Wolf M.S., Knight S.J., Lyons E.A., Durazo-Arvizu R., Pickard S.A., Arseven A. Literacy, race, and PSA level among low-income men newly diagnosed with prostate cancer. Urology. 2006;68:89–93. doi: 10.1016/j.urology.2006.01.064. [DOI] [PubMed] [Google Scholar]
- 17.Bray F., Jemal A., Grey N., Ferlay J., Forman D. Global cancer transitions according to the Human Development Index (2008–2030): a population-based study. Lancet Oncol. 2012;13:790–801. doi: 10.1016/S1470-2045(12)70211-5. [DOI] [PubMed] [Google Scholar]
- 18.Malik K. Human Development Report 2013: the rise of the South [Internet]. New York: Human Progress in a Diverse World United Nations Development Programme. [cited 2013, Jul 29]. Available at: http://hdrundporg/en/content/human-development-report-2013.
- 19.Ferlay J., Soerjomataram I., Dikshit R., Eser S., Mathers C., Rebelo M. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136:E359–E386. doi: 10.1002/ijc.29210. [DOI] [PubMed] [Google Scholar]
- 20.Jemal A., Bray F., Center M.M., Ferlay J., Ward E., Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61:69–90. doi: 10.3322/caac.20107. [DOI] [PubMed] [Google Scholar]
- 21.Parkin D.M. The evolution of the population-based cancer registry. Nat Rev Cancer. 2006;6:603–612. doi: 10.1038/nrc1948. [DOI] [PubMed] [Google Scholar]
- 22.Bray F., Ren J.S., Masuyer E., Ferlay J. Global estimates of cancer prevalence for 27 sites in the adult population in 2008. Int J Cancer. 2013;132:1133–1135. doi: 10.1002/ijc.27711. [DOI] [PubMed] [Google Scholar]
- 23.Djulbegovic M., Beyth R.J., Neuberger M.M., Stoffs T.L., Vieweg J., Djulbegovic B. Screening for prostate cancer: systematic review and meta-analysis of randomised controlled trials. BMJ. 2010;341:c4543. doi: 10.1136/bmj.c4543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.WHO . World Health Organization; Geneva: 2010. World Health Statistics 2010. [Google Scholar]
- 25.Hebert J.R., Ghumare S.S., Gupta P.C. Stage at diagnosis and relative differences in breast and prostate cancer incidence in India: comparison with the United States. Asian Pac J Cancer Prev. 2006;7:547. [PubMed] [Google Scholar]
- 26.WHO . World Health Organization; Geneva: 2002. National cancer control programmes: policies and managerial guidelines. [Google Scholar]
- 27.Schröder F.H., Hugosson J., Roobol M.J., Tammela T.L.J., Ciatto S., Nelen V. Screening and prostate-cancer mortality in a randomized European study. N Engl J Med. 2009;360:1320–1328. doi: 10.1056/NEJMoa0810084. [DOI] [PubMed] [Google Scholar]