Abstract
On August 1, 2013, the German Patent and Trademark Office issued a patent for the “Non-embryo-destructive extraction of pluripotent embryonic stem cells, stem cells obtained by this process and their uses” (DE 10 2004 062 184 B4). The patent document describes a non-embryo-destructive process to harvest embryonic stem cells from the inner cell mass (ICM) during the blastocyst development stage. The patent application was filed with the German Patent Office in Munich on December 23, 2004 and the patent claim was published in 2006. The patent was granted on August 1, 2013. Processing the patent application was a lengthy affair due to the fact that, for a long time, the prevailing opinion in Germany was that genetic screening of embryos (preimplantation genetic diagnosis) was prohibited under the German Embryo Protection Act (ESchG). A ruling by the German Federal Court in 2010 proved this opinion to be false. Animal studies have provided the evidence that the described procedure is technically feasible; healthy offspring were born after stem cells were harvested from the blastocyst and stored. We report here on a technique for the non-embryo-destructive extraction of pluripotent embryonic stem cells together with potential future applications for stem cells harvested in this manner.
Key words: stem cell extraction, embryo-preserving, preimplantation genetic diagnosis (PIGD), preimplantation therapy (PIT)
Abstract
Zusammenfassung
Am 1. August 2013 wurde vom Deutschen Patent- und Markenamt das Patent für die Patentschrift „Embryonenerhaltende Gewinnung pluripotenter embryonaler Stammzellen, derart gewonnene Stammzellen und Verwendung derselben“ (DE 10 2004 062 184 B4) erteilt. Das vorliegende Patent beschreibt ein Verfahren zur embryonenerhaltenden Gewinnung embryonaler Stammzellen aus der inneren Zellmasse (ICM) im Entwicklungsstadium der Blastocyste. Es wurde am 23. 12. 2004 beim Deutschen Patentamt in München angemeldet und 2006 als Patentanspruch offengelegt, also veröffentlicht. Rechtswirksam erteilt wurde das Patent am 1. 8. 2013. Die lange Bearbeitungsfrist ergab sich u. a. dadurch, dass in Deutschland lange Zeit die Meinung vorherrschte, dass genetische Untersuchungen am Embryo (Präimplantationsdiagnostik) nach dem Embryonenschutzgesetz (ESchG) verboten seien, was sich erst nach einem Urteil des Bundesgerichtshof im Jahre 2010 als unzutreffend erwies. Der Nachweis, dass das dargestellte Verfahren auch technisch umsetzbar ist, erfolgte im Tierversuch, nach Stammzellasservation wurde aus den Blastozysten eine gesunde Nachkommenschaft geboren. Im Artikel werden die Techniken der embryonenerhaltenden Gewinnung pluripotenter embryonaler Stammzellen beschrieben und die potenziellen Möglichkeiten einer Anwendung aufgezeigt.
Schlüsselwörter: Stammzellgewinnung, embryonenerhaltend, Präimplantationsdiagnostik (PID), Präimplantationstherapie (PIT)
Introduction
Stem cells differ from normally differentiated cells principally through their capacity for unlimited division and their potential to develop into very different cell types with characteristic phenotypes and specialized functions 1. It is this potential that makes stem cells so interesting for many different applications in pharmaceutics and medicine. The main aim is to replace and/or regenerate damaged tissue or tissue which no longer functions adequately 1.
The “classic” method for harvesting stem cells is based on isolating stem cells from preimplantation embryos. However, all established procedures to date result in destruction of the embryo 1. This makes these techniques extremely controversial from an ethical standpoint. They are often rejected outright; in some countries these techniques are prohibited and considered a criminal offense (cf. the German Embryo Protection Act [Embryonenschutzgesetz, ESchG]).
Given these issues, the question arises whether it might be possible to obtain embryonic stem cells without putting the life of the embryo and its capacity to develop into an individual human being at risk. If this were possible, it would invalidate one of the most serious ethical arguments levelled against reproductive medicine, according to which reproductive medicine should, in principle, be directed toward the promotion of human life but in the case of stem cell harvesting actively “lends itself” to the destruction of early forms of human life. The fact that “orphaned” preimplantation embryos are usually used does not change any of the premises of this ethical discussion as these embryos could also be donated, which is permissible under the German Embryo Protection Act (cf. the website of Netzwerk Embryonenspende, a German charitable association which aims to ensure that supernumerary embryos are made available to childless couples for implantation).
A technique for non-embryo-destructive stem cell extraction would create another, previously unknown dimension, namely that individuals born in future could possess a depot of their own embryonic stem cells. This would, of course, be impossible with destructive embryonic stem cell harvesting. It is currently impossible to estimate to what extent this would become a significant medical option in the future, although it should be noted that already now the use of autologous stem cells offers greater benefits than the use of non-autologous donor stem cells.
A procedure which allows stem cells to be obtained from the inner cell mass (ICM) without affecting the embryo and its potential to develop could already become important now, in the context of preimplantation genetic diagnosis (PIGD). Biopsies of blastocysts are increasingly being used for this type of diagnosis (rather than the original biopsies of cleavage-stage embryos), although it is currently not yet clear which cell entities of the blastocyst offer the most reliable results. Currently there is some evidence that ICM cells would be more useful rather than trophectoderm cells.
Below we present and comment on a German federal patent describing a technique which allows individual stem cells to be harvested for their later use in regenerative medicine or reproductive medicine without destruction of the embryo.
Overview
Technique for the non-embryo-destructive extraction of pluripotent embryonic stem cells
The technique for the non-embryo-destructive extraction of pluripotent embryonic stem cells has been described in detail in the aforementioned patent (DE 10 2004 062 184 B4) 1. In brief, after fixation of the blastocyst using a holding pipette (Fig. 1 a) a channel is opened up through the zona pellucida (the protective external layer of blastocysts) (Fig. 1 a). The opening in the zona pellucida is created using any one of a number of different procedures already in use in assisted reproductive medicine. Potential methods include chemical techniques (e.g. the use of acidic Tyrodeʼs solution), mechanical methods and laser techniques 1. An instrument to mobilize the stem cells is introduced through the opening and guided through the trophectoderm layer until it reaches the inner cell mass (Fig. 1 b). The following instruments are used for mobilization: 1, a special pipette with a diameter of approximately 10 µm with a double-sided blade used to detach individual cells from the inner cell mass through careful turning of the pipette. Aspiration of the stem cells which are detached from the inner cell mass is done using either an aspiration pipette or a double-lumen pipette (Fig. 1 b).
Fig. 1 a.
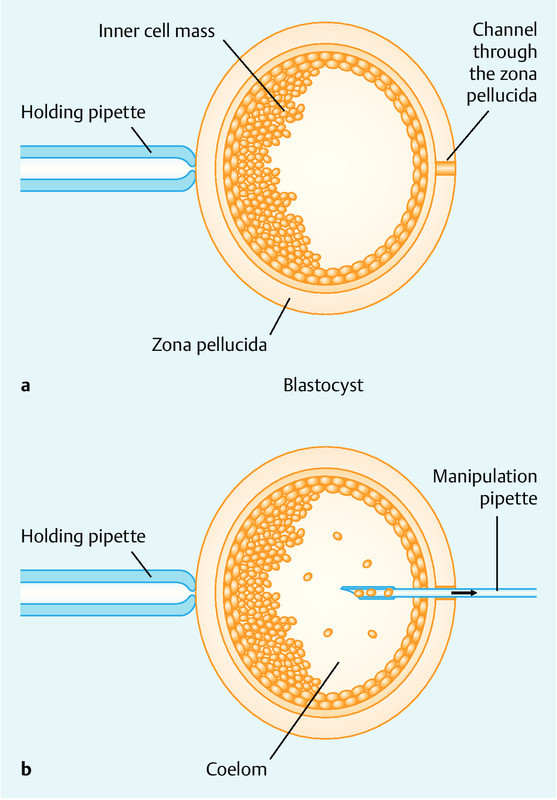
and b a Diagram of a blastocyst showing the channel used to introduce the manipulation pipette (from 1). b Harvesting of mobilized cells from the inner cell mass using a special pipette 1).
After harvesting the cells are placed in a suitable culture medium and expanded in accordance with the standard methods used to cultivate embryonic stem cells 1. The patent lists a number of different cultivation methods to increase the number of stem cells. Cultivation typically consists of culture on mouse fibroblast feeder cell layers. It is also possible to use other feeder cells types, e.g. primate cells, instead of mouse fibroblasts. One method of cultivation consists of plating the cells obtained on a mouse fibroblast layer to create cell accumulations, the removal of these cell accumulations and division of the accumulations into individual groups of cells, followed by repeat plating on fibroblast feeder cell layers. The ES cell medium described in WO 96/22362 consists of 80 % DMEM (Dulbeccoʼs modified Eagleʼs medium), 20 % fetal bovine serum (FBS), and 0.1 mM β-mercapto-ethanol along with small amounts of other additives, e.g. amino acids, antibiotics, etc., and is commonly used to cultivate embryonic stem cells. However, other well-known cell cultivation media can also be used 1.
Non-embryo-destructive stem cell extraction
The standard established methods to harvest human embryonic stem cells (hESC) are based – following the first publication by Evans and Kaufmann 2 – on the destruction of human embryos 3, 4, 5, 6, 7, either during the early cleavage stage or at the blastocyst development stage 3, 8, 9. “Supernumerary” embryos which may accrue during ART treatment are usually used 5, 10, 11, occasionally also embryos with genetic anomalies (after PGS [preimplantation genetic screening]) which are not used for embryo transfer (ET) 11, 12. This approach is legal in many countries – although not in Germany – but remains ethically controversial 13, 14, possibly with the exception of the use of embryos with arrested development 6, 15. However, in several countries there is no need to harvest embryonic stem cells without destroying the embryo.
Between the years 2006 and 2008, the working group around Robert Lanza published several studies describing the non-embryo-destructive extraction of stem cells in the early cleavage stage 16, 17, 18, 19. It is well known that individual blastomeres obtained with this method have the potential to develop into tissue or organs 20, 21, although even at the 2-cell stage cells exhibit a certain preference to develop further into a trophoblast or into the inner cell mass (ICM) 8. The ability to develop stem cell lines from biopsies taken in the early cleavage stage has also been reported by other groups 15, 22, 23, 24.
The use of ICM cells, i.e. blastocysts, is more difficult 17, 24, 25. This is basically due to the fact that blastocysts already exhibit cellular polarization 26 into either polarized trophectoderm or non-polarized ICM. This problem can be solved without difficulty through the addition of laminin, with the result that the efficacy of this method for stem cell harvesting approaches that of other techniques 17, 27, 28.
The aforementioned patent describes a method for the non-embryo-destructive extraction of embryonic stem cells from the inner cell mass (ICM) during the blastocyst development stage 1. The application was filed on December 23, 2004 with the German Patent Office in Munich, and the patent application was published in 2006. The patent was granted on August 1, 2013. The length of time it took to process the patent application was because, for a long time, the prevailing opinion in Germany was that genetic screening of embryos (preimplantation genetic diagnosis [PIGD], PGS) was prohibited under the German Embryo Protection Act (ESchG). It took a ruling by the German Federal Court to disprove this opinion 29. The animal studies we conducted have since provided evidence that the described procedure is technically feasible 30; healthy offspring were born after stem cells were harvested from the blastocyst and preserved.
Preservation of embryonic stem cells for homologous use
From the perspective of reproductive medicine, the above described procedure allows embryonic stem cells to be preserved during ART treatment. The biopsied stem cells can be immediately cultivated further (expanded) and then cryopreserved. Immediate cryopreservation with storage of the blastomeres in suitable storage containers (e.g. suspended in a matrix for vitrification or in an empty zona pellucida) would also be possible. The goal would be to use these stem cells at a later date for the benefit of the child which developed out of the biopsied blastocyst.
Such early stem cells would undoubtedly have the greatest potential for development, greater than that of fetal or adult stem cells 31, 32. Moreover, since they would be HLA-identical, there would be no risk of immunopathological reactions as can occur with heterologous stem cells 33, 34. In the event that after preservation of ICM stem cells the transfer of the blastocyst would not result in a pregnancy and the birth of a child, there would still remain the possibility of allogeneic use.
Biopsy of the inner cell mass (ICM) for PIGD appears to offer more reliable results
Recently a number of studies were published on trophectoderm biopsy (TEB), a technique for PIGD or PGS, which raised several interesting issues 35, 36, 37. The authors of these studies investigated the trophectoderm, the blastocoel fluid, and the inner cell mass from the same blastocyst and found that the respective genetic findings could differ: it is apparently possible that genetic anomalies may be present in trophectoderm cells and in the blastocoel fluid even if the genetics of the ICM are unremarkable. One suggestion, currently being discussed, is that genetically abnormal cells of the ICM are “disposed of” by being transferred to the blastocoel fluid and/or the trophectoderm, and that essentially this constitutes a form of “embryonic self-repair” 38. If this hypothesis can be confirmed, then the consequence would be that unremarkable genetic findings based on TEB would be reliable, while abnormal findings could not preclude the presence of a genetically unremarkable embryoblast. This would raise several questions – at the very least in this context – about the role of TEB as a technique for PIGD/PGS, with preference having potentially to be given to direct biopsies of the ICM (ICM-B). In this case, the technique described in the patent would offer one means of ICM-B.
The last potential area of application looks more to the future.
Preimplantation therapy (PIT)
One of the main criticisms levelled against PIGD or PGS is the fact that these test methods ultimately result in the rejection of genetically abnormal embryos, which amounts to a negative selection. Particularly in the case of monogenic diseases, the diagnostic techniques have no curative intent; their exclusive goal is to detect genetic abnormalities, not to rectify them. The patented technique offers the potential for some form of “preimplantation therapy” (PIT). However, such PIT concepts are still looking ahead to the future as the genetic correction of monogenic changes is not yet a routine procedure. However, a number of techniques have been described and tested in animal experiments which are working toward that end 39, 40, 41. A means of correcting human trisomy 21 using this method has also already been described in principle 42, 43, 44. If the option to perform such corrections should become an established procedure, then it would be useful to undertake such corrections as early as possible 45. According to the current state of knowledge, a curative approach would not even require all cells of the ICM to be rectified; the creation of a mosaic, possibly based on reprogramming 46, should suffice to prevent a later clinical manifestation of the associated disease. The same would probably apply to mitochondriopathies.
Conclusion
The method described in the patent allows pluripotent stem cells to be obtained from the inner cell mass of an embryo without destroying the embryo itself or impairing its potential for development. This does not only have technical implications through the description of an additional and new method for stem cell harvesting but also involves other, primarily ethical, considerations, both for current practices in reproductive medicine and for the future. A non-embryo-destructive method for stem cell harvesting weakens a number of important arguments currently dominating the ethical discussion of genetic procedures which focus on embryo-destructive stem cell extraction. The method could rapidly assume greater significance in current reproductive medicine, particularly if the hypothesis is confirmed that PIGD of trophectoderm cells results in a considerable number of false pathological results which biopsy of the ICM does not. Options such as the preimplantation therapy (PIT) of preimplantation embryos or the concept of people with disease utilizing their own embryonic stem cell depot to repair organ defects with the help of these (or other comparable) techniques may point the way the future applications.
Footnotes
Conflict of Interest R. D. and M. W. B. declare that they have no conflicts of interest. W. W. declares that he is the holder of patent DE 10 2004 062 184 B4.
Supporting Information
References
- 1.DPMAregister DE 10 2004 062 184.5.Online:https://register.dpma.de/DPMAregister/pat/register?AKZ=1020040621845last access: 20.04.2015
- 2.Evans M J, Kaufmann M H. Establishment in culture of pluripotential cells from mouse embryo. Nature. 1981;292:154–156. doi: 10.1038/292154a0. [DOI] [PubMed] [Google Scholar]
- 3.Thomson J A, Itskovitz-Eldor J, Shapiro S S. et al. Embryonic stem cell lines derived from human blastocysts. Science. 1998;282:1145–1147. doi: 10.1126/science.282.5391.1145. [DOI] [PubMed] [Google Scholar]
- 4.Heins N, Englund M C, Sjöblom C. et al. Derivation, characterization, and differentiation of human embryonic stem cells. Stem Cells. 2004;22:367–376. doi: 10.1634/stemcells.22-3-367. [DOI] [PubMed] [Google Scholar]
- 5.Mateizel I, De Temmerman N, Ullmann U. et al. Derivation of human embryonic stem cell lines from embryos obtained after IVF and PGD for monogenic disorders. Mol Hum Reprod. 2006;21:503–511. doi: 10.1093/humrep/dei345. [DOI] [PubMed] [Google Scholar]
- 6.Zhang X, Stoikovic P, Przyborski S. et al. Derivation of human embryonic stem cells from developing and arrested embryos. Stem Cells. 2006;24:2669–2676. doi: 10.1634/stemcells.2006-0377. [DOI] [PubMed] [Google Scholar]
- 7.Amit M, Itskovitz-Eldor J. Sources, derivation and culture of human embryonic stem cells. Semin Reprod Med. 2006;24:298–303. doi: 10.1055/s-2006-954939. [DOI] [PubMed] [Google Scholar]
- 8.Piotrowska K, Wianny F, Pedersen R A. et al. Blastomeres arising from the first cleavage division have distinguishable fates in normal mouse development. Development. 2001;128:3739–3748. doi: 10.1242/dev.128.19.3739. [DOI] [PubMed] [Google Scholar]
- 9.Smith A G. Embryo-derived stem cells: of mice and men. Annu Rev Cell Dev Biol. 2001;17:435–462. doi: 10.1146/annurev.cellbio.17.1.435. [DOI] [PubMed] [Google Scholar]
- 10.Pickering S J, Braude P R, Burns C J. et al. Preimplantation genetic diagnosis as a novel source of embryos for stem cell research. Reprod Biomed Online. 2003;7:353–364. doi: 10.1016/s1472-6483(10)61877-9. [DOI] [PubMed] [Google Scholar]
- 11.Cowan C A, Klimanskaya I, McMahon M S. et al. Derivation of embryonic stem-cell lines from human blastocysts. N Engl J Med. 2004;350:1353–1356. doi: 10.1056/NEJMsr040330. [DOI] [PubMed] [Google Scholar]
- 12.Verlinsky Y, Streichenko N, Kukharenko V. et al. Human embryonic stem cell lines with genetic diorders. Reprod Biomed Online. 2005;10:105–110. doi: 10.1016/s1472-6483(10)60810-3. [DOI] [PubMed] [Google Scholar]
- 13.Hug K. Sources of human embryos for stem cell research: ethical problems and their possible solutions. Medicina (Kaunas) 2005;41:1002–1010. [PubMed] [Google Scholar]
- 14.Evans M. Ethical sourcing of human embryonic stem cells – rational solutions? Nat Rev Mol Cell Biol. 2005;6:663–667. doi: 10.1038/nrm1698. [DOI] [PubMed] [Google Scholar]
- 15.Feki A, Bosman A, Dubuisson J B. et al. Derivation of the first Swiss human embryonic stem cell line from a single blastomere of an arrested four-cell stage embryo. Swiss Med Wkly. 2008;138:540–550. doi: 10.4414/smw.2008.12385. [DOI] [PubMed] [Google Scholar]
- 16.Chung Y, Klimanskaya I, Becker S. et al. Embryonic and extraembryonic stem cell lines derived from single mouse blastomeres. Nature. 2006;439:216–219. doi: 10.1038/nature04277. [DOI] [PubMed] [Google Scholar]
- 17.Klimanskaya I, Chung Y, Becker S. et al. Human embryonic stem cell lines derived from single blastomeres. Nature. 2006;444:481–485. doi: 10.1038/nature05142. [DOI] [PubMed] [Google Scholar]
- 18.Klimanskaya I, Chung Y, Becker S. et al. Derivation of human embryonic stem cells from single blastomeres. Nat Protoc. 2007;2:1963–1972. doi: 10.1038/nprot.2007.274. [DOI] [PubMed] [Google Scholar]
- 19.Chung Y, Klimnaskaya I, Becker S. et al. Human embryonic stem cell lines generated without embryo destruction. Cell Stem Cell. 2008;2:113–117. doi: 10.1016/j.stem.2007.12.013. [DOI] [PubMed] [Google Scholar]
- 20.Kelly S J. Studies of the developmental potential of 4- and 8-cell stage blastomeres. J Exp Zool. 1977;200:365–376. doi: 10.1002/jez.1402000307. [DOI] [PubMed] [Google Scholar]
- 21.Pedersen R A. New York, USA: Cambridge University Press; 1986. Potency, Lineage and Allocation in Preimplantation Mouse Embryos; pp. 3–33. [Google Scholar]
- 22.Geens M, Mateizel I, Sermon K. et al. Human embryonic stem cell lines derived from single blastomeres of two 4-cell stage embryos. Hum Reprod. 2009;24:2709–2717. doi: 10.1093/humrep/dep262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Giritharan G, Ilic D, Gormley M. et al. Human embryonic stem cells derived from embryos at different stages of development share similar transcription profiles. PLoS ONE. 2011;6:e26570. doi: 10.1371/journal.pone.0026570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Cenariu M, Pall E, Cernea C. et al. Evaluation of bovine embryo biopsy techniques according to their ability to preserve embryo viability. J Biomed Biotech. 2012 doi: 10.1155/2012/541384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Fong C Y, Richards M, Bongso A. Unsuccessful derivation of human embryonic stem cell lines from pairs of human blastomeres. Reprod Biomed Online. 2006;13:295–300. doi: 10.1016/s1472-6483(10)60629-3. [DOI] [PubMed] [Google Scholar]
- 26.Johnson M H, McConell J M. Lineage allocation and cell polarity during mouse embryogenesis. Semin Cell Dev Biol. 2004;15:573–581. doi: 10.1016/j.semcdb.2004.04.002. [DOI] [PubMed] [Google Scholar]
- 27.Ilic D, Giritharan G, Zdravkovic T. et al. Derivation of human embryonic stem cell lines from biopsied blastomeres on human feeders with minimal exposure to xenomaterials. Stem Cell Dev. 2009;18:1343–1350. doi: 10.1089/scd.2008.0416. [DOI] [PubMed] [Google Scholar]
- 28.Klimanskaya I. Embryonic stem cells from blastomeres maintaining embryo viability. Semin Reprod Med. 2013;31:49–55. doi: 10.1055/s-0032-1331797. [DOI] [PubMed] [Google Scholar]
- 29.Bundesgerichtshof, 5. Strafsenat . Urteil vom 6. Juli 2010: 5 StR 386/09
- 30.Dittrich R, Lotz L, Würfel W. et al. Offspring after embryo-preserving biopsy of the embryoblast with standard ICSI equipment in mouse blastocysts. In Vivo. 2011;25:935–939. [PubMed] [Google Scholar]
- 31.Bianco P, Robey P G, Simmons P J. Mesenchymal stem cells: revisiting history, concepts, and assays. Cell Stem Cell. 2008;4:313–319. doi: 10.1016/j.stem.2008.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Snykers S, De Kock J, Rogiers V. et al. In vitro differentiation of embryonic and adult stem cells into hepatocytes: state of the art. Stem Cells. 2009;27:577–605. doi: 10.1634/stemcells.2008-0963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Gyurkocza B, Rezvani A, Storb R F. Allogeneic hematopoietic cell transplantation: the state of the art. Exp Rev Hematol. 2010;3:285–299. doi: 10.1586/ehm.10.21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Farge D, Labopin M, Tyndall A. et al. Autologous hematopoietic stem cell transplantation for autoimmune diseases: an observational study on 12 yearsʼ experience from the European Group for Blood and Marrow Transplantation Working Party on Autoimmune Disease. Haematologica. 2010;95:284–292. doi: 10.3324/haematol.2009.013458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Tobler K J Zhao Y Ross R et al. Comparative genomic hybridization microarray (aCGH) analysis of DNA isolated from blastocoels fluid from 26 blastocysts Fertil Steril 2014101(Suppl.)e4 [Google Scholar]
- 36.Tobler K J Zhao Y Ross R et al. Blastocoel (BF) harbours embryonic DNA that may result from the marginalization of aneuploid cells during embryogenesis Fertil Steril 2014102(Suppl.)e205 [Google Scholar]
- 37.Tobler K J Zhao Y Ross R et al. The potential use of blastocoels fluid (BF) from expanded blastocysts as a less invasive form of embryo biopsy for preimplantation genetic testing Fertil Steril 2014102(Suppl.)e183–e184. [Google Scholar]
- 38.Würfel W. Der frühe Embryo – Netzwerke autologer, maternaler und iatrogener Regulation. Gynäkologische Endokrinologie. 2015;13:92–97. [Google Scholar]
- 39.Polzin V J, Anderson D L, Anderson G B. et al. Production of sheep-goat chimeras by inner cell mass transplantation. J Animal Sci. 1987;65:325–330. doi: 10.2527/jas1987.651325x. [DOI] [PubMed] [Google Scholar]
- 40.Zheng Y L, Jiang M X, OuYang Y C. et al. Production of mouse by inter-strain inner cell mass replacement. Zygote. 2005;13:73–77. doi: 10.1017/s0967199405003035. [DOI] [PubMed] [Google Scholar]
- 41.Murakami M, Ferguson C E, Perez O. et al. Transfer of inner cell mass cells derived from bovine nuclear transfer embryos into the trophoblast of bovine in vitro-produced embryos. Cloning Stem Cells. 2006;8:51–60. doi: 10.1089/clo.2006.8.51. [DOI] [PubMed] [Google Scholar]
- 42.Li B L, Chang K H, Wang P R. et al. Trisomy correction in Down syndrome induced pluripotent stem cells. Cell Stem Cell. 2012;5:615–619. doi: 10.1016/j.stem.2012.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Jiang J, Jing Y, Cost G J. et al. Translation dosage compensation to trisomy 21. Nature. 2013;500:296–300. doi: 10.1038/nature12394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Disteche C M. How to correct chromosomal trisomy. Cell Res. 2013;23:1345–1346. doi: 10.1038/cr.2013.135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Kay M A. State-of-the art gene based therapies: the road ahead. Nature Rev Genet. 2011;12:316–328. doi: 10.1038/nrg2971. [DOI] [PubMed] [Google Scholar]
- 46.Mikkers H M, Freund C, Mummery C L. et al. Cell replacement therapies: is it time to reprogram? Hum Gene Ther. 2014;25:866–874. doi: 10.1089/hum.2014.097. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


