Abstract
Airway remodeling is a key characteristic of chronic asthma, particularly in patients with a fixed airflow limitation. The mechanisms underlying airway remodeling are poorly understood, and no therapeutic option is available. The Wnt/β-catenin signaling pathway is involved in various physiological and pathological processes, including fibrosis and smooth muscle hypertrophy. In this study, we investigated the roles of Wnt/β-catenin signaling in airway remodeling in patients with asthma. Wnt7a mRNA expression was prominent in induced sputum from patients with asthma compared with that from healthy controls. Next, we induced a chronic asthma mouse model with airway remodeling features, including subepithelial fibrosis and airway smooth muscle hyperplasia. Higher expression of Wnt family proteins and β-catenin was detected in the lung tissue of mice with chronic asthma compared to control mice. Blocking β-catenin expression with a specific siRNA attenuated airway inflammation and airway remodeling. Decreased subepithelial fibrosis and collagen accumulation in the β-catenin siRNA-treated mice was accompanied by reduced expression of transforming growth factor-β. We further showed that suppressing β-catenin in the chronic asthma model inhibited smooth muscle hyperplasia by downregulating the tenascin C/platelet-derived growth factor receptor pathway. Taken together, these findings demonstrate that the Wnt/β-catenin signaling pathway is highly expressed and regulates the development of airway remodeling in chronic asthma.
Introduction
Asthma is a chronic airway inflammatory disease characterized by respiratory symptoms and expiratory airflow limitations that vary over time.1 Variable airflow limitation is a key characteristic of asthma, but some patients, particularly those with long-standing asthma, have a fixed airflow limitation which does not fully recover with treatment.2 Accumulating data support that asthma is associated with a rapid decline in lung function despite pharmacological treatment, including inhaled corticosteroids.3, 4 The development of a fixed airway obstruction and the rapid decline of lung function in patients with asthma are explained by airway remodeling.5 Airway remodeling refers to structural changes that occur in the airway, including subepithelial fibrosis, smooth muscle hyperplasia and goblet cell hyperplasia. The mechanisms underlying the development of airway remodeling remain poorly understood. Chronic airway inflammation in patients with asthma has been suggested to be the trigger for airway remodeling, through the production of inflammatory mediators and cytokines.6 However, suppressing airway inflammation does not fully prevent the development of airway remodeling.7 Thus, understanding the mechanisms of airway remodeling could shed light on a novel therapeutic approach to controlling airway remodeling in patients with asthma.
The Wnt/β-catenin signaling pathway is a fundamental mechanism involved in various biological activities, such as cell proliferation, morphogenesis and development.8 The Wnt family of proteins is composed of many secreted glycoproteins with highly conserved cysteine residues. Binding of Wnt proteins to cell surface receptors leads to stabilization of the cytosol and translocation of β-catenin to the nucleus. As the key regulator of this signaling pathway, β-catenin stimulates the transcription of target genes in cooperation with T-cell-factor/lymphoid enhancer-binding factor.9 Aberrant Wnt/β-catenin signaling has been suggested to be involved in a variety of human pathologies, including cancers and metabolic, inflammatory and fibrotic diseases.10, 11 Wnt signaling has been suggested to have important roles in lung cancer, pulmonary fibrosis and pulmonary hypertension.12, 13, 14 In particular, Wnt signaling has roles in lung tissue fibrosis, as in other areas of the body.15 Increased β-catenin expression is observed in murine lung tissue and in patients with idiopathic pulmonary fibrosis.16, 17 Previous studies have demonstrated that blocking Wnt/β-catenin signaling diminishes pulmonary fibrosis in a murine model.18, 19 In addition to pulmonary fibrosis, the Wnt/β-catenin signaling pathway has important roles in pulmonary arterial hypertension, in which vascular smooth muscle cell proliferation is a key characteristic.20
Given that subepithelial fibrosis and smooth muscle hyperplasia are the main features of airway remodeling, we hypothesized that Wnt/β-catenin signaling is involved in the development of airway remodeling in patients with asthma. In this study, we compared the expression of Wnt molecules between patients with asthma and healthy controls without asthma. Then, we examined whether the Wnt/β-catenin signaling pathway has a role in airway remodeling in a murine model of asthma. By using a long-term ovalbumin (OVA) challenge, we generated a mouse model of chronic asthma that showed airway remodeling features. We then assessed Wnt/β-catenin signaling expression in the lung tissue and observed the effect of blocking this signaling on airway remodeling.
Materials and Methods
Induced sputum from patients with asthma and the controls
Induced sputum was collected from patients with asthma and control subjects without asthma according to our institutional protocol. All subjects had given written informed consent, and this study was approved by the Institutional Review Board of Hanyang University. The characteristics of the study subjects are shown in Table 1. All of the patients with asthma were using controllers, including inhaled corticosteroids and long-acting β2 agonists and were in stable condition without exacerbation. A hypertonic saline solution (3% NaCl) was nebulized in an ultrasonic nebulizer (Sirius; Technomed, Florence, Italy) with a 2.8 ml min−1 output and inhaled for 5-min periods for up to 15 min. The subjects were asked to rinse their mouth and throat carefully and expectorate the sputum into a container every 5 min after starting the nebulization. After the sputum volume was assessed, the sputum samples were diluted with an equal volume of 0.01 M dithiothreitol (Sigma-Aldrich, St. Louis, MO, USA) in nuclease-free water, incubated on a rocker at room temperature for 30 min, and aspirated in and out of a pipette to further dissolve the mucus plugs. RNA was extracted from the sputum samples with TRI REAGENT (Sigma-Aldrich). The purity of the RNA preparations was checked by measuring the absorbance ratios at 260/280 nm. We used the total RNA (30 ng) to synthesize the complementary DNA with a Maxime RT PreMix kit (Oligo dT Primer; iNtRON Biotechnology, Inc., Seongnam, Korea). PCR was performed with a LightCycler (Roche-Applied Science, Manheim, Germany) and LightCycler FastStart DNA Master SYBR Green I (Roche-Applied Science).
Table 1. Subject characteristics.
| Asthma | Control | |
|---|---|---|
| Sample size (n) | 8 | 3 |
| Male (n (%)) | 2 (25%) | 1 (33%) |
| Age (years) | 64.3±6.3 | 45.7±8.7 |
| Duration of asthma | 9.25±7.3 | Not applicable |
| FEV1 (L) | 0.97±0.45 | 2.77±0.30 |
| FEV1/ FVC (%) | 57.7±13.9 | 75.8±3.5 |
Abbreviations: FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity.
Data are the mean±s.d.
Murine model of chronic asthma
Female BALB/c mice aged 6–8 weeks (Orient Bio Inc., Seongnam, Korea) were used in this experiment. The mice were housed in humidity- and temperature-controlled rooms under a 12 h light: 12 h dark photocycle with ad libitum access to food and water following our animal laboratory protocols. The experimental protocol was approved by the Institutional Animal Care and Use Committee.
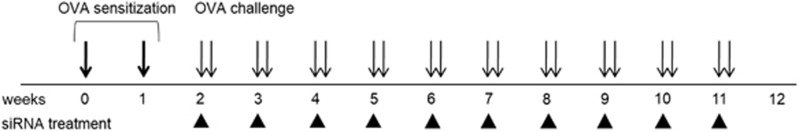
We sensitized and challenged the mice with OVA for a longer period of time than is usually required to induce acute asthma in order to induce chronic asthma with airway remodeling features. Briefly, the mice in the asthma group were sensitized on days 1 and 7 by an intraperitoneal injection of OVA (75 μg per mouse; Sigma-Aldrich) and aluminum hydroxide (2 mg per mouse, Sigma-Aldrich) in 200 μl phosphate-buffered saline. One week after the second injection, the mice in the asthma group were challenged with intranasal administration of 20 μl OVA (50 μg) in 0.9% NaCl solution twice a week for the next 10 weeks (Figure 1). The control mice were given the same volume of normal saline instead of OVA on the same schedule. The mice were killed 24 h after the last intranasal OVA administration. Each experimental group included five or more animals.
Figure 1.
Murine model of chronic asthma. Bold arrows indicate intraperitoneal sensitization to OVA, and the arrows indicate the intranasal OVA challenge in the mice. The black triangles represent the administration of β-catenin siRNA. OVA, ovalbumin.
Analysis of inflammatory cells from bronchoalveolar lavage
Bronchoalveolar lavage was performed with 1 ml of sterile phosphate-buffered saline after a tracheal cannulation to count the inflammatory cells in the bronchoalveolar lavage fluid (BALF). The BALF was centrifuged at 3,000 r.p.m. for 15 min, and the cell pellet was resuspended in 100 μl of phosphate-buffered saline at 4 °C. The total number of cells was determined with a hemocytometer. The slides were prepared by cytocentrifugation (Cytospin 3; Thermo Scientific, Pittsburgh, PA, USA) and stained with a Diff-Quik staining kit (Sysmex Co., Kobe, Japan). We counted 200 cells per slide under a light microscope using morphological criteria to determine the differential count for each inflammatory cell type (macrophages, eosinophils, neutrophils and lymphocytes).
Histological examination
The left lung was fixed in 10% neutral-buffered formalin for 48 h and then embedded in paraffin. Sagittal sections were cut to a thickness of 3 μm and stained with hematoxylin and eosin and Masson's trichrome stain. Histology was evaluated under a light microscope.
β-catenin immunohistochemistry
The lung tissue was examined by immunohistochemical staining for β-catenin and α-smooth muscle actin (SMA) with an anti-rabbit β-catenin antibody (Cell Signaling Technology, Beverly, MA, USA). In brief, 4-μm-thick sections were mounted on poly-L-lysine-coated slides, de-waxed and washed in TBS for 10 min. After three washes in phosphate-buffered saline with 1% saponin, the endogenous peroxidase activity was inhibited using a 3% hydrogen peroxide solution in methanol for 10 min. The sections were incubated with the primary antibody solution (1:50) for 2 h at room temperature, and the negative control slides were incubated with rabbit IgG (ABR; Affinity Bioreagent, Golden, CO, USA). A secondary biotinylated anti-Ig antibody (Dako, Carpentaria, CA, USA) was added, and the mixture was incubated for 30 min at room temperature. After being rinsed, the slides were incubated with streptavidin-conjugated HRP (Dako) and washed with deionized water. The diaminobenzidine substrate solution was added, and the mixture was incubated for 10 min. A brown-colored reaction represented a positive result.
RNA extraction and real-time PCR
The right lung was stored at −80 °C in TRI REAGENT (Sigma-Aldrich) for later RNA analysis. RNA was extracted from the right lung using TRI REAGENT. The purity of the RNA preparations was checked by measuring the absorbance ratios at 260/280 nm. The primers were designed for real-time PCR of the specific target mRNAs, including β-catenin, Wnt1, Wnt3a, Wnt5a, Wnt7a, transforming growth factor (TGF)-β, tenascin C and platelet-derived growth factor receptor (PDGFR). Total RNA (1 μg) was used to synthesize complementary DNA with a Maxime RT PreMix kit (Tech Dragon, Ltd., Hong Kong, China). PCR was performed with a LightCycler (Roche-Applied Science) and LightCycler FastStart DNA Master SYBR Green I (Roche-Applied Science).
Collagen assay
The right lung was ligated at the level of the main bronchus, excised at the hilum and frozen immediately in liquid nitrogen for the collagen assay. The lung collagen content was determined by assaying the total soluble collagen using the Sircol soluble collagen assay (Biocolor; Carrickfergus Co., Antrim, UK) according to the manufacturer's manual.
Statistical analysis
The data are expressed as the mean±the s.e.m. The analyses were performed using SPSS for Windows software (ver. 19.0; SPSS Inc., Chicago, IL, USA). The Mann–Whitney U-test was used to compare the two groups, and a P-value<0.05 was considered significant.
Results
Expression of β-catenin and Wnt family proteins in patients with chronic asthma
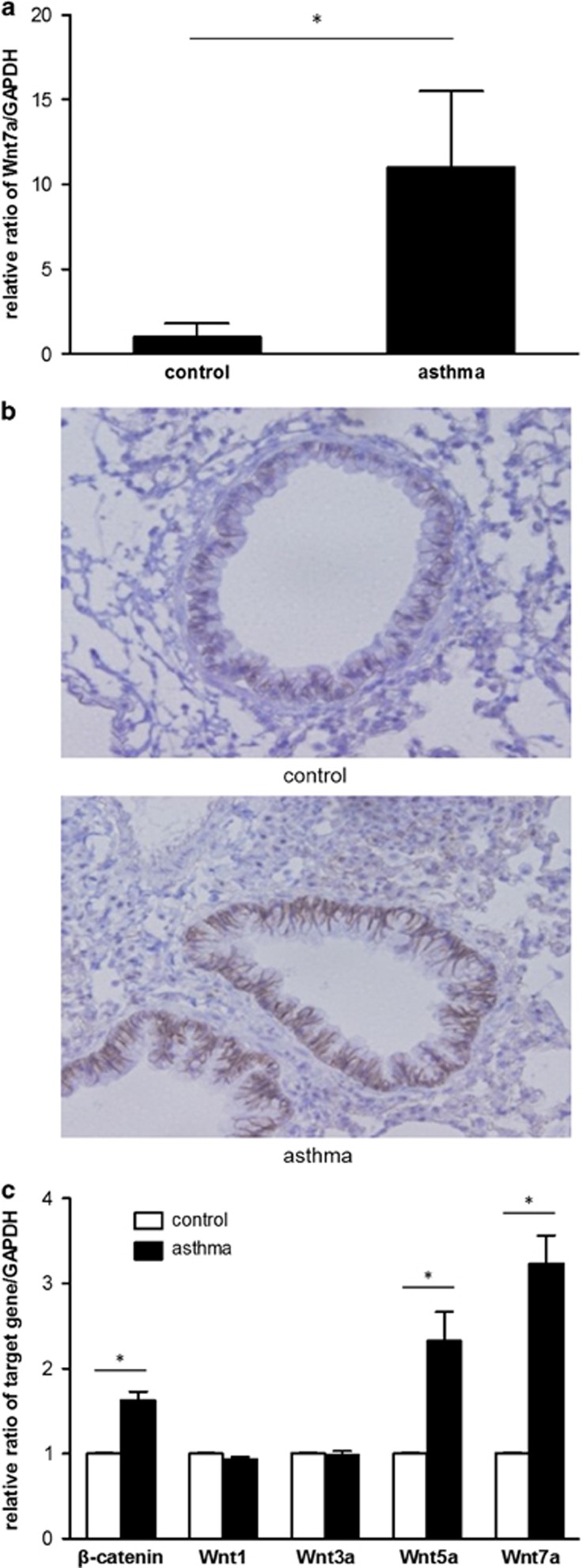
First, we assessed whether Wnt/β-catenin signaling is expressed in the airway of patients with asthma using induced sputum from patients and controls (Table 1). The Wnt7a mRNA expression level was significantly higher in the induced sputum from the patients with asthma compared with the controls (Figure 2a), suggesting an upregulation of the Wnt/β-catenin pathway in the airways of humans with asthma. Furthermore, β-catenin expression was markedly elevated, mainly in the bronchial epithelial cells, in mice with asthma compared with the controls (Figure 2b). In addition, quantitative real-time PCR of the lung tissue revealed significantly higher β-catenin expression in the asthma group compared with the control group (Figure 2c). Of the various Wnt proteins, Wnt5a and Wnt7a showed an elevated expression in the asthma group compared with the controls (Figure 2c). No significant difference between the groups was found for the expression of Wnt1 and Wnt3a.
Figure 2.
Wnt/β-catenin signaling expression in asthma. (a) Wnt7a mRNA expression in induced sputum from the patients with asthma and the control subjects. (b) Immunohistochemical staining of β-catenin in the mouse lung tissue and (c) reverse transcription-PCR for β-catenin, Wnt1, Wnt3a, Wnt5a and Wnt7a mRNA in the mouse lung tissue. *P<0.05 between the two groups.
The effects of β-catenin siRNA on airway remodeling
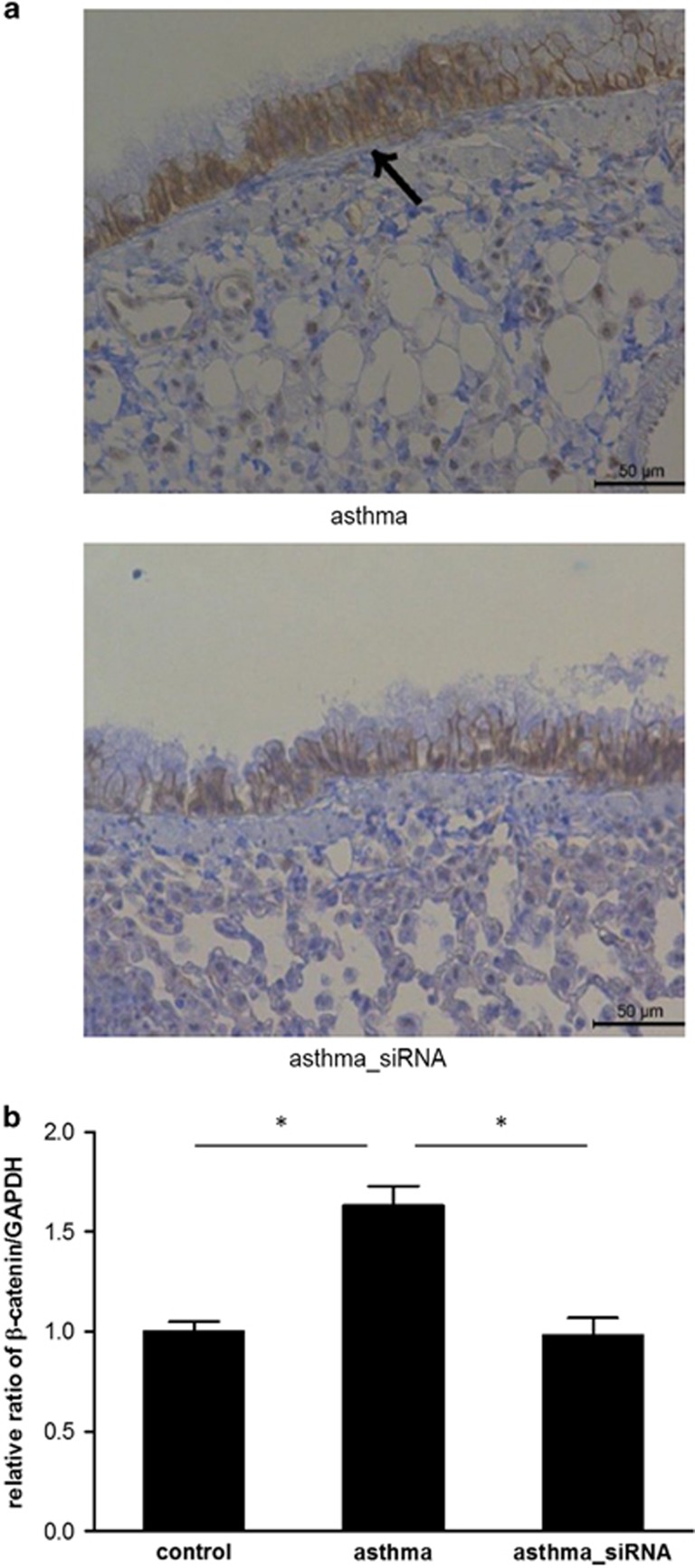
Next, to suppress β-catenin expression during the development of asthma, we delivered a specific siRNA targeting β-catenin into the mice during the OVA challenge, as shown in Figure 1. Similarly to the previous experiment, significantly higher β-catenin expression in the mice with asthma was confirmed (Figure 3a). We observed that β-catenin siRNA reduced β-catenin expression in the airways of mice with asthma (Figure 3a). Furthermore, real-time PCR showed effective suppression of β-catenin mRNA in the lung tissue after the use of β-catenin siRNA (Figure 3b). Collectively, β-catenin siRNA effectively diminished β-catenin expression in the chronic asthma mouse model.
Figure 3.
Inhibition of β-catenin using specific siRNA. (a) Immunohistochemical staining for β-catenin and (b) reverse transcription-PCR for β-catenin mRNA using the lung tissue from the control mice, those with asthma, and those with asthma treated with β-catenin siRNA (asthma_siRNA). *P<0.05 between the two groups.
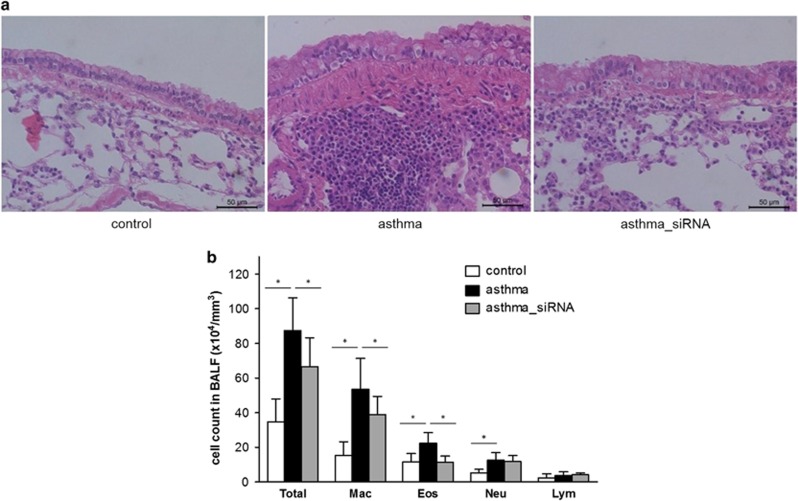
Airway remodeling was evident during the histological evaluation of mice with chronic asthma, as demonstrated by the structural changes in the airway, including smooth muscle hyperplasia, mucous metaplasia and subepithelial fibrosis (Figure 4a). A BAL fluid analysis showed an increase in the number of inflammatory cells in the mice with asthma, as well as in the number of macrophages, eosinophils and neutrophils (Figure 4b). Next, we investigated whether Wnt/β-catenin signaling regulates the development of airway remodeling in the mouse model of chronic asthma by administering β-catenin siRNA. The features of airway remodeling were markedly attenuated in the lung tissue from the β-catenin siRNA-treated mice compared with the mice with chronic asthma (Figure 4a). Consistent with the histology, the total number of inflammatory cells, including macrophages and eosinophils, decreased in the mice with asthma that were administered β-catenin siRNA (Figure 4b). These data suggest that suppressing Wnt/β-catenin signaling prevented airway remodeling in the OVA-sensitized and challenged mice.
Figure 4.
Histology and inflammatory cell counts. (a) Hematoxylin and eosin staining of the lung tissue from the control mice, those with asthma and those with asthma treated with β-catenin siRNA (asthma_siRNA) and (b) the inflammatory cell counts in the bronchoalveolar lavage fluid. *P<0.05 between the two groups.
The effects of the Wnt/β-catenin signaling pathway on airway remodeling
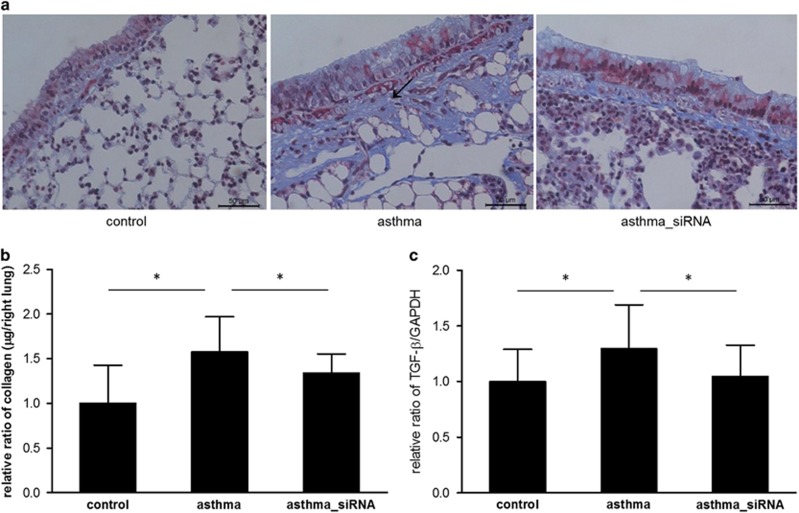
A significant increase in collagen deposition in the subepithelial fibrotic tissues in the chronic asthma group was detected by Masson's trichrome staining (Figure 5a). This result was confirmed quantitatively by the Sircol collagen assay (Figure 5b). Quantitative real-time PCR showed significantly higher TGF-β mRNA expression in the asthma group compared with the control group (Figure 5c). Collagen synthesis and subepithelial fibrosis were ameliorated when we administered β-catenin siRNA to suppress Wnt signaling. We also observed that β-catenin siRNA treatment reduced the expression of TGF-β mRNA in the mice with asthma (Figure 5c). These findings show that blocking Wnt/β-catenin signaling decreases collagen synthesis and subepithelial fibrosis in mice with chronic asthma by reducing TGF-β synthesis.
Figure 5.
The effects of β-catenin siRNA treatment on subepithelial fibrosis. (a) Masson's trichrome stain, (b) Sircol collagen assay of the lung tissue and (c) reverse transcription-PCR for transforming growth factor-β mRNA. *P<0.05 between the two groups.
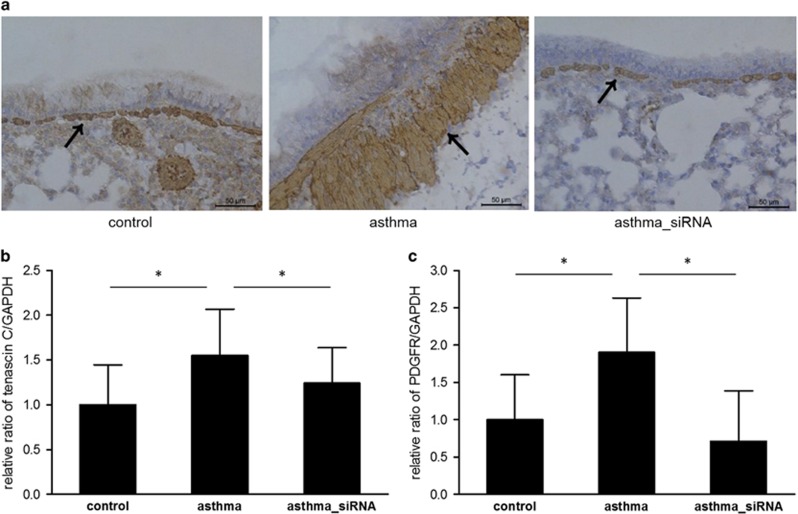
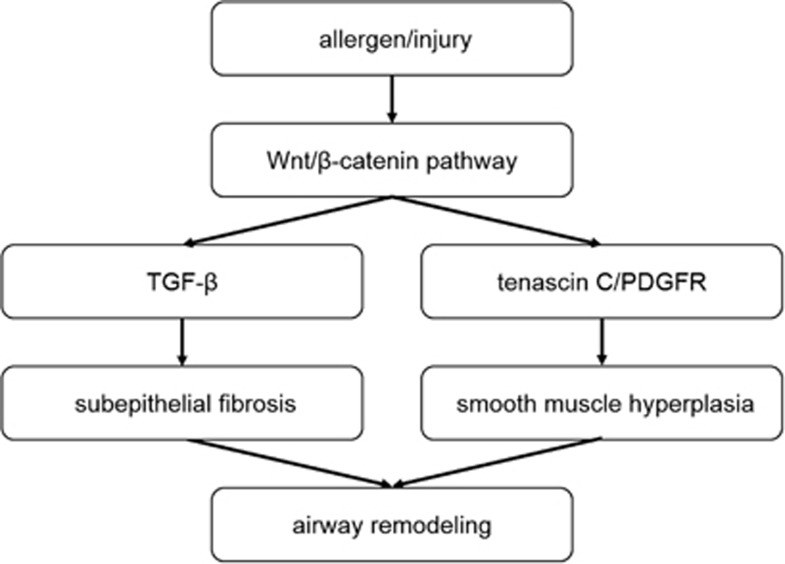
To visualize and compare airway smooth muscle hyperplasia between the two groups, we performed immunohistochemical staining of the lung tissue using an α-SMA antibody. As shown in Figure 6a, the expression of α-SMA was markedly higher in the asthma group compared with the control. To explore the mechanism underlying the increased α-SMA synthesis, we measured tenascin C and PDGFR expression using quantitative real-time PCR. The asthma group showed a significantly higher expression of tenascin C and PDGFR compared with the control group (Figures 6b and c). Next, we showed that treating the mice with asthma with the β-catenin siRNA inhibited tenascin C and PDGFR synthesis, leading to a decrease in α-SMA production. Taken together, these data illustrate that the Wnt/β-catenin signaling pathway is also involved in smooth muscle hyperplasia by upregulating tenascin C and PDGFR in chronic asthma (Figure 7).
Figure 6.
The effects of β-catenin siRNA treatment on smooth muscle hyperplasia. (a) Immunohistochemical staining for α-smooth muscle actin. (b) Reverse transcription-PCR for tenascin-C and (c) platelet-derived growth factor receptor mRNA. *P<0.05 between the two groups.
Figure 7.
The role of Wnt/β-catenin signaling in airway remodeling.
Discussion
In this study, we showed that the Wnt/β-catenin signaling pathway is upregulated in human patients and in a murine model of chronic asthma. We obtained the first evidence of the involvement of Wnt7a in human asthma. We also observed high expression of the Wnt/β-catenin pathway in a mouse model of chronic asthma. In addition, we demonstrated that blocking β-catenin attenuated subepithelial fibrosis and smooth muscle hyperplasia, which are key features of airway remodeling. This is the first experimental study to reveal the roles of Wnt/β-catenin in airway remodeling in asthma.
A possible link between Wnt/β-catenin signaling and asthma was suggested by a few previous studies. Stimulating the adenosine A3 receptor, which is an inflammatory mediator in asthma, downregulated β-catenin in melanoma cells, suggesting a role for Wnt/β-catenin signaling in airway inflammation in asthma.21 Asthma is a heterogeneous disease, and asthma subtypes can be divided based on the degree of Th2 inflammation.22 A microarray analysis of peripheral blood mononuclear cells treated with Th2 cytokines, such as interleukins IL-13 and IL-4, showed that Wnt5a is a major biomarker of the Th2 immune response.23 The positive correlation between Th2 inflammation and Wnt proteins was confirmed by whole-genome microarray and quantitative real-time PCR analyses using bronchial biopsies from subjects with and without asthma.24 In addition to experimental studies, a genetic study in children showed that Wnt signaling genes are associated with impaired lung function in two childhood asthma cohorts.25 Consistent with this study, another genetic study, in an adult population, found a significant association between a single nucleotide polymorphism of the Wnt-1-induced secreted protein 1 gene and lung function in Chinese adults with asthma.26 These findings support that Wnt/β-catenin signaling is involved in asthma and has roles in Th2 inflammation and the decline in lung function. In line with these reports, we provide the first evidence that the Wnt/β-catenin signaling pathway is highly upregulated in the asthmatic airway in vivo by showing high Wnt7a expression in the induced sputum from patients with asthma. In addition to humans, mice with chronic asthma also had a higher expression of Wnt5a and Wnt7a in the lung tissue compared with the controls. Our observation of high Wnt5a expression in asthma agrees with previous observations from experimental studies that used peripheral blood mononuclear cells23 and bronchial tissue from humans.24 As the chronic asthma model used in our experiment was induced by OVA sensitization and challenge, the airway inflammation was characterized by high eosinophilic infiltration and Th2 cytokine expression. Thus, it is speculated that Wnt5a is associated with Th2-induced inflammation and has roles in the development of airway remodeling. In contrast to Wnt5a, no significant association was found between Wnt7a and asthma. Wnt7a has been implicated in lung carcinogenesis27 and pulmonary arterial hypertension.28 More precise roles for Wnt proteins in asthma remain to be discovered in the future.
In addition to airway inflammation, structural changes in the airway develop earlier during the disease course, even in children.29 Thickening of the epithelial basement membrane is a key feature of airway remodeling. Activated fibroblasts in the asthmatic airway differentiate into myofibroblasts, which secrete inflammatory mediators and extracellular matrix proteins such as collagen.30 Persistent activation of fibroblasts leads to subepithelial fibrosis, which is refractory to anti-inflammatory treatment. The Wnt/β-catenin signaling pathway has been suggested to be involved in pulmonary fibrosis, as in other organs, including the liver, skin and kidneys. Now, targeting the Wnt/β-catenin pathway is drawing attention as a new therapeutic approach to control fibrosis and fibrotic diseases.31, 32 We used a β-catenin-specific siRNA to silence β-catenin expression in mice and observed a marked reduction in subepithelial fibrosis and collagen accumulation in the β-catenin-specific siRNA-treated mice. We also showed downregulation of TGF-β, a profibrotic cytokine, in the mice administered β-catenin siRNA, suggesting that β-catenin affects the synthesis of TGF-β. Our finding of an interaction between β-catenin and TGF-β agrees with previous studies.33, 34 Nevertheless, the crosstalk mechanisms between β-catenin and TGF-β remains unexplained. Moreover, it is possible that another mechanism underlies Wnt/β-catenin-induced pulmonary fibrosis in asthma.
In addition to subepithelial fibrosis, increased smooth muscle mass is a major change that occurs during airway remodeling that is frequently associated with decreased lung function.35, 36 In this study, we observed that α-SMA expression increased in the airways of the mice with chronic asthma and that blocking β-catenin attenuated the smooth muscle changes. A number of studies have shown upregulation of proliferative factors and downregulation of counteracting regulatory mechanisms, such as apoptosis, associated with smooth muscle hyperplasia during airway remodeling.37, 38 Of the proliferative factors in the airway smooth muscle cells, PDGF has a mitogenic effect on human smooth muscle cells.39, 40 In addition, Wnt signaling has a proliferative effect on lung smooth muscle by activating tenascin C and promoting PDGFR.41 Consistent with this report, our results showed decreased expression of tenascin C and PDGFR mRNA in the β-catenin siRNA-treated mice. These findings suggest that the Wnt/β-catenin pathway has a proliferative effect on the airway smooth muscle cells via the tenascin C/PDGFR pathway in a murine model of chronic asthma.
In conclusion, the Wnt/β-catenin signaling pathway was highly expressed in human patients and a mouse model of chronic asthma. Blocking this pathway dampened airway remodeling, such as subepithelial fibrosis and smooth muscle hyperplasia, by downregulating TGF-β and tenascin C/PDGFR. These findings suggest that Wnt/β-catenin regulates the development of airway remodeling in chronic asthma.
Acknowledgments
This study was supported by a National Research Foundation of Korea Grant from the Korean Government (NRF-2012S1A2A1A01031801).
The authors declare no conflict of interest.
References
- 1Reddel HK, Bateman ED, Becker A, Boulet LP, Cruz AA, Drazen JM et al. A summary of the new GINA strategy: a roadmap to asthma control. Eur Respir J 2015; 46: 622–639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2Jarjour NN, Erzurum SC, Bleecker ER, Calhoun WJ, Castro M, Comhair SA et al. Severe asthma: lessons learned from the National Heart, Lung, and Blood Institute Severe Asthma Research Program. Am J Respir Crit Care Med 2012; 185: 356–362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3Lange P, Parner J, Vestbo J, Schnohr P, Jensen G. A 15-year follow-up study of ventilatory function in adults with asthma. N Engl J Med 1998; 339: 1194–1200. [DOI] [PubMed] [Google Scholar]
- 4Witt CA, Sheshadri A, Carlstrom L, Tarsi J, Kozlowski J, Wilson B et al. Longitudinal changes in airway remodeling and air trapping in severe asthma. Acad Radiol 2014; 21: 986–993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5Al-Muhsen S, Johnson JR, Hamid Q. Remodeling in asthma. J Allergy Clin Immunol 2011; 128: 451–462. [DOI] [PubMed] [Google Scholar]
- 6ten Brinke A, Zwinderman AH, Sterk PJ, Rabe KF, Bel EH. Factors associated with persistent airflow limitation in severe asthma. Am J Respir Crit Care Med 2001; 164: 744–748. [DOI] [PubMed] [Google Scholar]
- 7Berair R, Brightling CE. Asthma therapy and its effect on airway remodelling. Drugs 2014; 74: 1345–1369. [DOI] [PubMed] [Google Scholar]
- 8MacDonald BT, Tamai K, He X. Wnt/beta-catenin signaling: components, mechanisms, and diseases. Dev Cell 2009; 17: 9–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9Macdonald BT, Semenov MV, He X. SnapShot: Wnt/beta-catenin signaling. Cell 2007; 131: 1204. [DOI] [PubMed] [Google Scholar]
- 10Clevers H, Nusse R. Wnt/beta-catenin signaling and disease. Cell 2012; 149: 1192–1205. [DOI] [PubMed] [Google Scholar]
- 11George SJ. Wnt pathway: a new role in regulation of inflammation. Arterioscler Thromb Vasc Biol 2008; 28: 400–402. [DOI] [PubMed] [Google Scholar]
- 12Pongracz JE, Stockley RA. Wnt signalling in lung development and diseases. Respir Res 2006; 7: 15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13Van Scoyk M, Randall J, Sergew A, Williams LM, Tennis M, Winn RA. Wnt signaling pathway and lung disease. Transl Res 2008; 151: 175–180. [DOI] [PubMed] [Google Scholar]
- 14Konigshoff M, Eickelberg O. WNT. signaling in lung disease: a failure or a regeneration signal? Am J Respir Cell Mol Biol 2010; 42: 21–31. [DOI] [PubMed] [Google Scholar]
- 15Guo Y, Xiao L, Sun L, Liu F. Wnt/beta-catenin signaling: a promising new target for fibrosis diseases. Physiol Res 2012; 61: 337–346. [DOI] [PubMed] [Google Scholar]
- 16Liu L, Carron B, Yee HT, Yie TA, Hajjou M, Rom W. Wnt pathway in pulmonary fibrosis in the bleomycin mouse model. J Environ Pathol Toxicol Oncol 2009; 28: 99–108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17Chilosi M, Poletti V, Zamo A, Lestani M, Montagna L, Piccoli P et al. Aberrant Wnt/beta-catenin pathway activation in idiopathic pulmonary fibrosis. Am J Pathol 2003; 162: 1495–1502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18Kim TH, Kim SH, Seo JY, Chung H, Kwak HJ, Lee SK et al. Blockade of the Wnt/beta-catenin pathway attenuates bleomycin-induced pulmonary fibrosis. Tohoku J Exp Med 2011; 223: 45–54. [DOI] [PubMed] [Google Scholar]
- 19Henderson WR Jr., Chi EY, Ye X, Nguyen C, Tien YT, Zhou B et al. Inhibition of Wnt/beta-catenin/CREB binding protein (CBP) signaling reverses pulmonary fibrosis. Proc Natl Acad Sci USA 2010; 107: 14309–14314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20de Jesus Perez V, Yuan K, Alastalo TP, Spiekerkoetter E, Rabinovitch M. Targeting the Wnt signaling pathways in pulmonary arterial hypertension. Drug Discov Today 2014; 19: 1270–1276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21Fishman P, Madi L, Bar-Yehuda S, Barer F, Del Valle L, Khalili K. Evidence for involvement of Wnt signaling pathway in IB-MECA mediated suppression of melanoma cells. Oncogene 2002; 21: 4060–4064. [DOI] [PubMed] [Google Scholar]
- 22Woodruff PG, Modrek B, Choy DF, Jia G, Abbas AR, Ellwanger A et al. T-helper type 2-driven inflammation defines major subphenotypes of asthma. Am J Respir Crit Care Med 2009; 180: 388–395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23Syed F, Huang CC, Li K, Liu V, Shang T, Amegadzie BY et al. Identification of interleukin-13 related biomarkers using peripheral blood mononuclear cells. Biomarkers 2007; 12: 414–423. [DOI] [PubMed] [Google Scholar]
- 24Choy DF, Modrek B, Abbas AR, Kummerfeld S, Clark HF, Wu LC et al. Gene expression patterns of Th2 inflammation and intercellular communication in asthmatic airways. J Immunol 2011; 186: 1861–1869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25Sharma S, Tantisira K, Carey V, Murphy AJ, Lasky-Su J, Celedon JC et al. A role for Wnt signaling genes in the pathogenesis of impaired lung function in asthma. Am J Respir Crit Care Med 2010; 181: 328–336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26Wang SH, Xu F, Dang HX, Yang L. Genetic variations in the Wnt signaling pathway affect lung function in asthma patients. Genet Mol Res 2013; 12: 1829–1833. [DOI] [PubMed] [Google Scholar]
- 27Kim TH, Moon JY, Kim SH, Paik SS, Yoon HJ, Shin DH et al. Clinical significance of aberrant Wnt7a promoter methylation in human non-small cell lung cancer in Koreans. J Korean Med Sci 2015; 30: 155–161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28Rai PR, Cool CD, King JA, Stevens T, Burns N, Winn RA et al. The cancer paradigm of severe pulmonary arterial hypertension. Am J Respir Crit Care Med 2008; 178: 558–564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29Lezmi G, Gosset P, Deschildre A, Abou-Taam R, Mahut B, Beydon N et al. Airway remodeling in preschool children with severe recurrent wheeze. Am J Respir Crit Care Med 2015; 192: 164–171. [DOI] [PubMed] [Google Scholar]
- 30Roche WR, Beasley R, Williams JH, Holgate ST. Subepithelial fibrosis in the bronchi of asthmatics. Lancet 1989; 1: 520–524. [DOI] [PubMed] [Google Scholar]
- 31Enzo MV, Rastrelli M, Rossi CR, Hladnik U, Segat D. The Wnt/beta-catenin pathway in human fibrotic-like diseases and its eligibility as a therapeutic target. Mol Cell Ther 2015; 3: 1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32Lam AP, Gottardi CJ. beta-catenin signaling: a novel mediator of fibrosis and potential therapeutic target. Curr Opin Rheumatol 2011; 23: 562–567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33Carre AL, James AW, MacLeod L, Kong W, Kawai K, Longaker MT et al. Interaction of wingless protein (Wnt), transforming growth factor-beta1, and hyaluronan production in fetal and postnatal fibroblasts. Plast Reconstr Surg 2010; 125: 74–88. [DOI] [PubMed] [Google Scholar]
- 34Cheon SS, Wei Q, Gurung A, Youn A, Bright T, Poon R et al. Beta-catenin regulates wound size and mediates the effect of TGF-beta in cutaneous healing. Faseb j 2006; 20: 692–701. [DOI] [PubMed] [Google Scholar]
- 35Noble PB, Pascoe CD, Lan B, Ito S, Kistemaker LE, Tatler AL et al. Airway smooth muscle in asthma: linking contraction and mechanotransduction to disease pathogenesis and remodelling. Pulm Pharmacol Ther 2014; 29: 96–107. [DOI] [PubMed] [Google Scholar]
- 36Prakash YS. Airway smooth muscle in airway reactivity and remodeling: what have we learned? Am J Physiol Lung Cell Mol Physiol 2013; 305: L912–L933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37Berair R, Saunders R, Brightling CE. Origins of increased airway smooth muscle mass in asthma. BMC Med 2013; 11: 145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38Bara I, Ozier A, Tunon de Lara JM, Marthan R, Berger P. Pathophysiology of bronchial smooth muscle remodelling in asthma. Eur Respir J 2010; 36: 1174–1184. [DOI] [PubMed] [Google Scholar]
- 39Hirst SJ, Barnes PJ, Twort CH. PDGF isoform-induced proliferation and receptor expression in human cultured airway smooth muscle cells. Am J Physiol 1996; 270: L415–L428. [DOI] [PubMed] [Google Scholar]
- 40Ito I, Fixman ED, Asai K, Yoshida M, Gounni AS, Martin JG et al. Platelet-derived growth factor and transforming growth factor-beta modulate the expression of matrix metalloproteinases and migratory function of human airway smooth muscle cells. Clin Exp Allergy 2009; 39: 1370–1380. [DOI] [PubMed] [Google Scholar]
- 41Cohen ED, Ihida-Stansbury K, Lu MM, Panettieri RA, Jones PL, Morrisey EE. Wnt signaling regulates smooth muscle precursor development in the mouse lung via a tenascin C/PDGFR pathway. J Clin Invest 2009; 119: 2538–2549. [DOI] [PMC free article] [PubMed] [Google Scholar]