INTRODUCTION
From the technical aspect of liver surgery, control of bleeding during hepatic parenchymal resection is one of the most important procedures in hepatectomy. Pringle's maneuver, a temporary cross-clamping of the hepatoduodenal ligament, has often been used for this purpose[1]. This is the simplest and useful technique to reduce intraoperative blood loss. Unfortunately, this method has resulted in normothermic ischemia-reperfusion injury of the liver. Many clinical observations indicate that prolonged intraoperative normothermic ischemia and succeeding reperfusion of the liver are the most significant disadvantages of Pringle's manever, and may cause postoperative functional disorder of the liver[2-6].
Although the process of ischemia-reperfusion injury of the liver is complicated, it consists of three pathologic components: hepatic ischemia, portal congestion, and reinflow of arterial and congested portal blood. Ischemia-reperfusion injury is not merely an ischemic injury but is also related to reperfusion of blood. Moreover, it has been revealed that reactive oxygen radicals and some chemical mediators activated during recirculation play an important role in the development of liver injury after ischemia[7-11]. Because portal congestion is an extremely harmful factor that can induce bacterial translocation, generation of chemical mediators, and reactive oxygen radicals in the portal flow, it is conceivable that reinflow of congested portal blood containing activated pathogens may play a role in the development of ischemia-reperfusion injury of the liver. However, the influence of portal congestion on the development of liver injury during ischemia-reperfusion injury has not been well researched. Only a few authors have described the significance of the reperfusion of congested portal blood[12].
Heat shock protein 72 (HSP72) is produced in the liver after normothermic ischemia and reperfusion[13-15]. HSP72 is now being intensively investigated as one of the most fundamental stress proteins. The protein is produced in response to a variety of stress-induced stimuli and is considered to be an important factor which helps to maintain cellular homeostasis and increase the chance of pateint survival[16-23].
In the present study, we investigated the induction of HSP72 in the liver tissue after 15-min Pringle's maneuver with or without portosystemic shunt in the rat model, to determine the influence of congested portal blood on the development of ischemia-reperfusion injury in liver.
MATERIALS AND METHODS
Male Wistar rats weighing 200 g to 300 g were housed in chip-bedded cages in a climate-controlled room (24 °C ± 1 °C) under a 12-hour light-dark cycle. The rats were provided with ordinary rat chow and water ad libitum-according to the animal protection guidelines of Kyoto University. Rats were premedicated with an intramuscular injection of atropine sulfate (0.05 mg) and anesthetized with an intraperitoneal injection of pentobarbital sodium (40 mg/kg rats weight) before all operative procedures. They were divided into two groups. In the first group (Group P), the abdomen was opened through a median laparotomy and the liver was detached from its ligaments. Total hepatic ischemia with portal congestion was produced by clamping the hepatoduodenal ligament for 15 min with a microvascular clamp. In the second group (Group S), the abdomen was opened in the same way and the liver was also detached from its ligaments. Extracorporeal portosystemic shunt was achieved from the cecal branch of the superior mesenteric vein to the right external jugular vein, using a polyethylene tube (0.58 mm inner diameter × 200 mm long, Dural Plastics Co., Australia) filled with 0.04 mL heparinized saline solution (250 U/mL). The liver was subjected to warm ischemia by cross-clamping the hepatoduodenal ligament for 15 min with a microvascular clamp. However, portal congestion was prevented by the extracorporeal shunt. During these interventions, the abdomen was temporarily closed and an electric heater was placed under the animal to keep the rectal temperature at 36 °C ± 1 °C. Then, declamping allowed reperfusion until sampling after specified periods.
Liver tissue was removed at 48 h after each intervention and Western blot analysis was performed to detect HSP72 (monoclonal SPA-810, StressGen Biotechnologies Corp., Canada)[13]. To measure adenosine nucleotide concentration, liver tissue was sampled by freeze-clamping at 1 (n = 6), 3 (n = 6) and 48 h (n = 6) after reperfusion. Enzymatic assay was performed to measure the concentration of adenine nucleotides in the liver tissue[24,25]. The energy charge potential (EC) was calculated according to the formula proposed by Atkinson: (ATP + 0.5 ADP)/(ATP + ADP + AMP)[26], where ATP, ADP and AMP are adenosine tri-, di-, and monophosphate, respectively. Blood samples were taken from the aorta at 1 (n = 6), 3 (n = 6), and 48 h (n = 6) after the end of each intervention. The serum concentration of aspartate aminotransferase (AST), alanine aminotransferase (ALT) and lactic dehydrogenase (LDH) were measured at a biochemical laboratory. After sampling, rats were killed.
The values were expressed as mean ± standard deviation (SD). For the statistical analysis, Student's t test was performed for the biochemical parameters. A P value < 0.05 was considered to be significant.
RESULTS
Comparing these two groups, HSP72 was markedly overexpressed in group P but its accumulation in group S was very slight (Figure 1).
Figure 1.
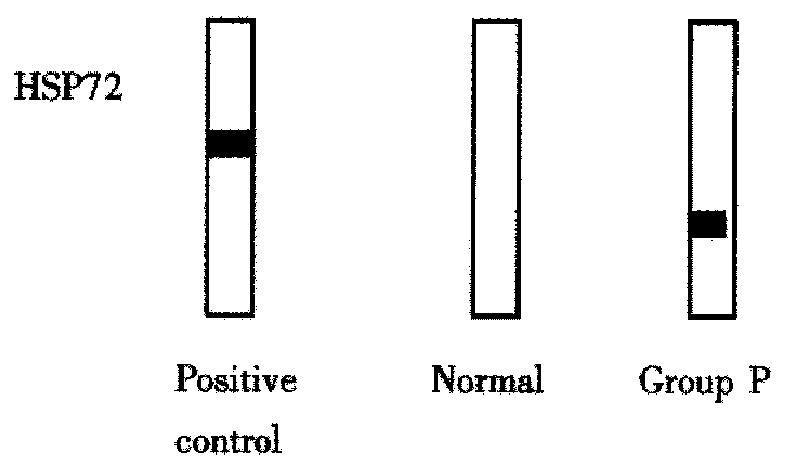
Heat shock protein (HSP) production in the rat liver after 48 h of reperfusion in each group compared with that in normal liver (5 μg protein per lane). HSC70, which contains inducible HSP72 in approximately 10 per cent, was used as a positive control (0.2 μg protein per lane).
In the comparison between AST, ALT and LDH levels in group P and group S (Table 1). There was a trend towards higher transaminase levels in group P, but the difference was not statistically significant (P > 0.05).
Table 1.
Changes in AST, ALT and LDH (IU/L) in serum after reperfusion (n = 6)
| Parameter | 1 h | 3 h | 48 h |
| Group P | |||
| AST | 4832 ± 1589 | 4896 ± 3270 | 199 ± 93 |
| ALT | 4860 ± 2015 | 5218 ± 3230 | 266 ± 155 |
| LDH | 45235 ± 21748 | 44619 ± 29975 | 588 ± 265 |
| Group S | |||
| AST | 4147 ± 1069 | 2731 ± 1282 | 204 ± 72 |
| ALT | 4230 ± 1460 | 2689 ± 1459 | 181 ± 103 |
| LDH | 35752 ± 15954 | 26327 ± 21973 | 476 ± 248 |
Results are expressed as means ± SD. There was no statistically significant difference in AST, ALT and LDH at any time point examined between two groups.
There were no significant differences between the two groups at any time point examined (P > 0.05, Table 2).
Table 2.
Change in ATP levels (mmol/g wet wt tissue) and EC in liver tissue (n = 6)
| Parameter | 1 h | 3 h | 48 h |
| Group P | |||
| ATP | 1.49 ± 0.69 | 1.91 ± 0.42 | 3.08 ± 0.42 |
| EC | 0.67 ± 0.11 | 0.62 ± 0.07 | 0.83 ± 0.03 |
| Group S | |||
| ATP | 1.60 ± 0.55 | 2.22 ± 0.81 | 3.00 ± 0.22 |
| EC | 0.64 ± 0.14 | 0.68 ± 0.12 | 0.80 ± 0.05 |
EC, energy charge. Results are expressed as means ± SD. There were no significant differences in ATP concentration and EC level at 1 h, 3 h, and 48 h between two groups (P > 0.05).
DISCUSSION
Heat shock proteins are intracellular proteins that have been well conserved during evolution. They are present in small amounts in normal cells and larger amounts in cells subjected to a variety of stressful stimuli. Heat shock stress, which induces the synthesis of HSPs, is closely related to the physiologic body temperature of the organism[16,27]. This is the reason by which special attention was paid to control the body temperature of the animals during operations in this study to obviate the possible effects of body temperature changes. The rectal temperature of the animals was strictly controlled in the range of 36 °C ± 1 °C using an electric heating pad.
Schoeniger et al[14] reported that prolonged ischemia induced by Pringle's maneuver did not activate the HSP72 gene in the porcine liver until restoration of blood flow. They concluded that transcription of HSP72 mRNA was highly dependent on the generation of superoxide anions. This participation of superoxide anions in the activation of HSP72 gene partly suggests a contribution by the reinflow of portal blood, but the role of portal blood pooling was not a focus of their report. We observed that the reinflow of congested portal blood into ischemic liver after a short-term Pringle's maneuver contributed to the production of HSP72 in the liver tissue. However, the liver subjected to the same period of ischemia produced less HSP72 when reperfusion was performed with non-congested portal blood. This observation suggests an adverse impact of congested portal blood on liver function after ischemia and reperfusion. In contrast, Tacchini et al[28] reported that inflow occlusion to the partial liver for a longer period (up to 60 min) also induced expression of HSP72 mRNA in the ischemic lobe in rat liver. In their experiment, portal congestion did not occur because of hemi-occlusion of the hepatic inflow. The discrepancy in the influence of congested portal blood in the production of HSP72 between their results and ours may be due to the difference in ischemic periods. When the ischemic stress is sufficient due to a longer insult, even non-congested blood may cause oxidative stress sufficient to provoke the activation of heat shock genes. However, when the ischemic derangement of hepatic parenchyma is not so severe, the nature of the blood used for reperfusion may have a marked impact on the results. These findings imply that portal pooling would act appreciably as a key stress factor to increase the production of HSP72, but it would not be an essential factor when the degree of pre-existing ischemia is already high.
Although the mechanism by which the reflow of congested portal blood induces HSP72 production is still unclear, the process seems to be complex. The mechanism most likely involves factors that operate in the ischemic reperfused liver. Active oxygen species and free radicals are known to activate heat shock genes[29,30]. It is likely that free radicals generated in the small bowel during portal congestion play a role in the development of liver tissue injury during reperfusion[31-38]. Kawamoto et al[7] reported that the temporary occlusion of the portal vein and hepatic artery both increased xanthine oxidase activity in portal plasma and increased the permeability of the intestine during reperfusion. Both of these were prevented when a portosystemic shunt was performed and splanchnic vascular congestion was prevented. Other potential causative factors in the production of HSP72 include endotoxin and various cytokines translocated from the intestines during portal congestion. Recently, participation of both endotoxin and bacteria in the production of HSP72 has been reported[17,27,39-41]. Pringle's maneuver for 15 min was observed to have significantly increased plasma endotoxin levels in the portal blood and enhanced bacterial translocation into the portal blood from the gut[9].
Our results also suggest that HSP72 might prove useful in evaluating the degree of hepatic ischemia-reperfusion injury. Levels of enzymes in serum (including AST, ALT, and LDH), ATP and EC in liver tissue have been commonly used to assess hepatocellular injury. In the present study, no significant difference was observed between the congested and non-congested groups at any time point examined. Thus, even the ATP concentration and EC cannot differentiate these two groups. Animals could fully recover from liver injury as long as the Pringle's maneuver lasted only 15 min, and no animals died as a result of it. However, the fact that the stress brought on by the reperfusion of congested portal blood was clearly greater than that produced by the reperfusion of non-congested blood, as demonstrated by the production of HSP72, implies that the accumulation of stress under a repeated or prolonged Pringle's maneuver would likely result in irreversible liver damage. This concept is also consistent with the report of Nitta et al[12]. They reported that conventional parameters or arterial ketone body ratio could be used to detect the ill-effect of congested portal blood during reperfusion when Pringle's maneuver was repeated four times, although they failed to detect it after a single 30 min Pringle's maneuver in dogs. In this sense, the production of HSP72 seems to be more sensitive than conventional parameters to detect the given stress during ischemia-reperfusion injury.
In conclusion, HSP72 induced in the liver tissue by a short-term Pringle's maneuver was associated with the reinflow of congested portal blood into ischemic tissue. The influence of this congested portal blood upon reperfusion of ischemic tissue was detected by HSP72, but not by conventional biochemical parameters. HSP72 is suggested to be useful for determination of the degree of hepatic ischemia-reperfusion injury.
Footnotes
This work was supported partly by Grant 90089102 from the Scientific Research Fund of the Ministry of Education, Japan
Chao-Liu Dai, MD. graduated from Wenzhou Medical College in 1985, and received doctor degree from China Medical University in 1992. He is an Associated Professor and Vice-Director of the Department of Hepatobiliary Surgery, the Second Clinical College of China Medical University. He has published 40 papers.
Edited by Jason Carr
References
- 1.Pringle JH. V. Notes on the Arrest of Hepatic Hemorrhage Due to Trauma. Ann Surg. 1908;48:541–549. doi: 10.1097/00000658-190810000-00005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Battersby C, Balderson G, Winch J, Burnett W. Acute icclusion of the portal vein in the calf. J Surg Res. 1971;11:95–100. doi: 10.1016/0022-4804(71)90142-9. [DOI] [PubMed] [Google Scholar]
- 3.JOHNSTONE FR. Acute ligation of the portal vein. Surgery. 1957;41:958–971. [PubMed] [Google Scholar]
- 4.Delva E, Camus Y, Nordlinger B, Hannoun L, Parc R, Deriaz H, Lienhart A, Huguet C. Vascular occlusions for liver resections. Operative management and tolerance to hepatic ischemia: 142 cases. Ann Surg. 1989;209:211–218. doi: 10.1097/00000658-198902000-00012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Huguet C, Gavelli A, Chieco PA, Bona S, Harb J, Joseph JM, Jobard J, Gramaglia M, Lasserre M. Liver ischemia for hepatic resection: where is the limit. Surgery. 1992;111:251–259. [PubMed] [Google Scholar]
- 6.Hasselgren PO. Prevention and treatment of ischemia of the liver. Surg Gynecol Obstet. 1987;164:187–196. [PubMed] [Google Scholar]
- 7.Kawamoto S, Tashiro S, Miyauchi Y, Inoue M. Mechanism for enterohepatic injury caused by circulatory disturbance of hepatic vessels in the rat. Proc Soc Exp Biol Med. 1991;198:629–635. doi: 10.3181/00379727-198-43300. [DOI] [PubMed] [Google Scholar]
- 8.Serizawa A, Nakamura S, Suzuki S, Nakano M. Involvement of platelet-activating factor in cytokine production and neutrophil activation after hepatic ischemia-reperfusion. Hepatology. 1996;23:1656–1663. doi: 10.1002/hep.510230649. [DOI] [PubMed] [Google Scholar]
- 9.Liu L, Jeppsson B, Bengmark S. Bacterial translocation into portal blood from the gut during portal triad occlusion. Dig Surg. 1992;9:95–101. [Google Scholar]
- 10.Colletti LM, Remick DG, Burtch GD, Kunkel SL, Strieter RM, Campbell DA. Role of tumor necrosis factor-alpha in the pathophysiologic alterations after hepatic ischemia/reperfusion injury in the rat. J Clin Invest. 1990;85:1936–1943. doi: 10.1172/JCI114656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Chen XH, Bao MS, Li ZZ. Effects of adenosine and its mechanism in ischemic preconditioning in rat liver in vivo. Shijie Huaren Xiaohua Zazhi. 1999;7:298–299. [Google Scholar]
- 12.Nitta N, Yamamoto S, Ozaki N, Morimoto T, Mori K, Yamaoka Y, Ozawa K. Is the deterioration of liver viability due to hepatic warm ischemia or reinflow of pooled-portal blood in intermittent portal triad cross-clamping. Res Exp Med (Berl) 1988;188:341–350. doi: 10.1007/BF01851202. [DOI] [PubMed] [Google Scholar]
- 13.Kume M, Yamamoto Y, Saad S, Gomi T, Kimoto S, Shimabukuro T, Yagi T, Nakagami M, Takada Y, Morimoto T, et al. Ischemic preconditioning of the liver in rats: implications of heat shock protein induction to increase tolerance of ischemia-reperfusion injury. J Lab Clin Med. 1996;128:251–258. doi: 10.1016/s0022-2143(96)90026-8. [DOI] [PubMed] [Google Scholar]
- 14.Schoeniger LO, Andreoni KA, Ott GR, Risby TH, Bulkley GB, Udelsman R, Burdick JF, Buchman TG. Induction of heat-shock gene expression in postischemic pig liver depends on superoxide generation. Gastroenterology. 1994;106:177–184. doi: 10.1016/s0016-5085(94)95209-4. [DOI] [PubMed] [Google Scholar]
- 15.Schiaffonati L, Rappocciolo E, Tacchini L, Cairo G, Bernelli-Zazzera A. Reprogramming of gene expression in postischemic rat liver: induction of proto-oncogenes and hsp 70 gene family. J Cell Physiol. 1990;143:79–87. doi: 10.1002/jcp.1041430110. [DOI] [PubMed] [Google Scholar]
- 16.Lindquist S, Craig EA. The heat-shock proteins. Annu Rev Genet. 1988;22:631–677. doi: 10.1146/annurev.ge.22.120188.003215. [DOI] [PubMed] [Google Scholar]
- 17.Schlesinger MJ. Heat shock proteins. J Biol Chem. 1990;265:12111–12114. [PubMed] [Google Scholar]
- 18.Kimoto S, Yamamoto Y, Yamagami K, Ishikawa Y, Kume M, Yamamoto H, Ozaki N, Yamaoka Y. The augmentative effect of repeated heat shock preconditioning on the production of heat shock protein 72 and on ischemic tolerance in rat liver tissue. Int J Hyperthermia. 2000;16:247–261. doi: 10.1080/026567300285268. [DOI] [PubMed] [Google Scholar]
- 19.Yamagami K, Yamamoto Y, Kume M, Kimoto S, Yamamoto H, Ozaki N, Yamamoto M, Shimahara Y, Toyokuni S, Yamaoka Y. Heat shock preconditioning ameliorates liver injury following normothermic ischemia-reperfusion in steatotic rat livers. J Surg Res. 1998;79:47–53. doi: 10.1006/jsre.1998.5403. [DOI] [PubMed] [Google Scholar]
- 20.Yamamoto Y, Kume M, Yamaoka Y. Implications of heat shock proteins during liver surgery and liver perfusion. Recent Results Cancer Res. 1998;147:157–172. doi: 10.1007/978-3-642-80460-1_15. [DOI] [PubMed] [Google Scholar]
- 21.Shimabukuro T, Yamamoto Y, Kume M, Kimoto S, Okamoto R, Morimoto T, Yamaoka Y. Induction of heat shock response: effect on the rat liver with carbon tetrachloride-induced fibrosis from ischemia-reperfusion injury. World J Surg. 1998;22:464–468; discussion 464-468;. doi: 10.1007/s002689900417. [DOI] [PubMed] [Google Scholar]
- 22.Kume M, Yamamoto Y, Yamagami K, Ishikawa Y, Uchinami H, Yamaoka Y. Pharmacological hepatic preconditioning: involvement of 70-kDa heat shock proteins (HSP72 and HSP73) in ischaemic tolerance after intravenous administration of doxorubicin. Br J Surg. 2000;87:1168–1175. doi: 10.1046/j.1365-2168.2000.01509.x. [DOI] [PubMed] [Google Scholar]
- 23.Yamamoto H, Yamamoto Y, Yamagami K, Kume M, Kimoto S, Toyokuni S, Uchida K, Fukumoto M, Yamaoka Y. Heat-shock preconditioning reduces oxidative protein denaturation and ameliorates liver injury by carbon tetrachloride in rats. Res Exp Med (Berl) 2000;199:309–318. doi: 10.1007/s004339900040. [DOI] [PubMed] [Google Scholar]
- 24.Lamprecht W, Trautschold I. Adenosine-5'-triphosphate. Determination with hexokinase and glucose-6-phosphate dehydrogenase. In: Bergmeyer HU, ed , editors. Methods of Enzymatic Analysis. Vol 4. 2nd English ed, New York: Academic Press; 1974. pp. 2101–2110. [Google Scholar]
- 25.Jaworek D, Gruber W, Bermeyer HU. Adenosine-5'-diphosphate and adenosine5'-monophosphate. In: Bergmeyer HU, ed , editors. Methods of Enzymatic Analysis. Vol 4. 2nd English ed, New York: Academic Press; 1974. pp. 2127–2131. [Google Scholar]
- 26.Atkinson DE. The energy charge of the adenylate pool as a regulatory parameter. Interaction with feedback modifiers. Biochemistry. 1968;7:4030–4034. doi: 10.1021/bi00851a033. [DOI] [PubMed] [Google Scholar]
- 27.Beck SC, Paidas CN, Mooney ML, Deitch EA, De Maio A. Presence of the stress-inducible form of hsp-70 (hsp-72) in normal rat colon. Shock. 1995;3:398–402. [PubMed] [Google Scholar]
- 28.Tacchini L, Schiaffonati L, Pappalardo C, Gatti S, Bernelli-Zazzera A. Expression of HSP 70, immediate-early response and heme oxygenase genes in ischemic-reperfused rat liver. Lab Invest. 1993;68:465–471. [PubMed] [Google Scholar]
- 29.Marubayashi S, Dohi K, Ochi K, Kawasaki T. Role of free radicals in ischemic rat liver cell injury: prevention of damage by alpha-tocopherol administration. Surgery. 1986;99:184–192. [PubMed] [Google Scholar]
- 30.Adkison D, Höllwarth ME, Benoit JN, Parks DA, McCord JM, Granger DN. Role of free radicals in ischemia-reperfusion injury to the liver. Acta Physiol Scand Suppl. 1986;548:101–107. [PubMed] [Google Scholar]
- 31.Wang WT, Lin LN, Xu ZJ, Wang ZM. Role of polymorphonuclear leukocyte in hepatic ischemia and reperfusion injury and the protective effect of Ligustrazini. Huaren Xiaohua Zazhi. 1998;6:774–775. [Google Scholar]
- 32.Osarogiagbon UR, Choong S, Belcher JD, Vercellotti GM, Paller MS, Hebbel RP. Reperfusion injury pathophysiology in sickle transgenic mice. Blood. 2000;96:314–320. [PubMed] [Google Scholar]
- 33.Liang LJ, He Q, Lu MD, Peng BG, Huang JF. The protective effects of panax notoginseng on liver ischemia-reperfusion injury in rats. Xin Xiaohuabingxue Zazhi. 1997;5:419–420. [Google Scholar]
- 34.Suzuki M, Fukuhara K, Unno M, Htwe T, Takeuchi H, Kakita T, Matsuno S. Correlation between plasma and hepatic phosphatidylcholine hydroperoxide, energy charge, and total glutathione content in ischemia reperfusion injury of rat liver. Hepatogastroenterology. 2000;47:1082–1089. [PubMed] [Google Scholar]
- 35.Sakata C, Tanaka H, Takemura S, Hirohashi K, Minamiyama Y, Nakamura A, Inoue M, Kinoshita H. Post-ischemic intraportal adenosine administration protects against reperfusion injury of canine liver. J Hepatobiliary Pancreat Surg. 2000;7:78–85. doi: 10.1007/s005340050158. [DOI] [PubMed] [Google Scholar]
- 36.Chen XH, Li ZZ, Bao MS. Ischemic preconditioning protects liver from ischemia-reperfusion injury in rats. Xin Xiaohuabingxue Zazhi. 1997;5:763–764. [Google Scholar]
- 37.Chen XH, Li ZZ, Bao MS, Zheng HX. Effect of nitric oxide on liver ischemia/reperfusion injury in rats in vivo. Shijie Huaren Xiaohua Zazhi. 1999;7:295–297. [Google Scholar]
- 38.Ito K, Ozasa H, Sanada K, Horikawa S. Doxorubicin preconditioning: a protection against rat hepatic ischemia-reperfusion injury. Hepatology. 2000;31:416–419. doi: 10.1002/hep.510310222. [DOI] [PubMed] [Google Scholar]
- 39.Polla BS. A role for heat shock proteins in inflammation. Immunol Today. 1988;9:134–137. doi: 10.1016/0167-5699(88)91199-1. [DOI] [PubMed] [Google Scholar]
- 40.Seitz CS, Kleindienst R, Xu Q, Wick G. Coexpression of heat-shock protein 60 and intercellular-adhesion molecule-1 is related to increased adhesion of monocytes and T cells to aortic endothelium of rats in response to endotoxin. Lab Invest. 1996;74:241–252. [PubMed] [Google Scholar]
- 41.Hensler T, Köller M, Alouf JE, König W. Bacterial toxins induce heat shock proteins in human neutrophils. Biochem Biophys Res Commun. 1991;179:872–879. doi: 10.1016/0006-291x(91)91899-n. [DOI] [PubMed] [Google Scholar]


