Abstract
The efficacy of mother-to-child placental transfer of antibodies specific to malaria blood stage antigens was investigated in the context of placental malaria infection, taking into account IgG specificity and maternal hypergammaglobulinemia. The impact of the resulting maternal antibody transfer on infections in infants up to the age of 6 months was also explored. This study showed that i) placental malaria was associated with a reduced placental transfer of total and specific IgG, ii) antibody placental transfer varied according to IgG specificity and iii) cord blood malaria IgG levels were similar in infants born to mothers with or without placental malaria. The number of malaria infections was negatively associated with maternal age, whereas it was not associated with the transfer of any malaria-specific IgG from the mother to the fetus. These results suggest that i) malaria-specific IgG may serve as a marker of maternal exposure but not as a useful marker of infant protection from malaria and ii) increasing maternal age contributes to diminishing febrile infections diagnosed in infants, perhaps by means of the transmission of an effective antibody response.
Introduction
During pregnancy, the sequestration of Plasmodium falciparum-infected erythrocytes in the placenta is known to decrease maternofetal antibody exchange. Several studies have demonstrated an association between placental malaria (PM) and decreased maternal antibody transfer to the fetus, taking into account maternal antibody levels at delivery [1–3]. One of these studies, enrolling 213 mother/child pairs in The Gambia, showed that prematurity and low birth weight were associated with a PM-dependent decrease of Haemophilus influenzae- and Streptococcus pneumoniae-specific antibody transfer [2]. The authors speculated that this decrease in maternal antibody transfer weakened the infants, making them more vulnerable to bacterial infections [2]. The authors of the above-cited studies showed that the efficacy of the maternal antibody transfer in patients with PM varied according to antibody specificity [1–3]. Okoko et al. demonstrated that PM was associated with decreased anti-measles but not anti-tetanus toxoid antibody transfer in the same cohort [2]. In contrast, Scott et al. showed that PM was not associated with a lower anti-measles antibody level [4]. Deloron et al. observed no difference in the transfer of antibodies targeting P. falciparum extracts according to placental infection [5]. The effect of PM in specific antimalarial antibody transfer has not been completely clarified.
The transport of IgG across the placenta is an active and selective process specifically mediated by the neonatal Fc receptor (FcRn). One study revealed that high total and anti-measles antibody levels in maternal peripheral blood were associated with reduced efficacy of specific antibody transfer [6]. Saturation of the limited number of FcRn receptors on syncytiotrophoblasts could be generated by high maternal IgG levels [7–9] and may therefore introduce a bias in the analysis of PM’s effect on the efficacy of maternal-specific antibody transfer. Maternal hypergammaglobulinemia is thus an essential factor to consider [1–3], and it was subsequently determined and introduced in the analyses of the present study.
Few studies have investigated the role of transferred maternal antibodies on infants’ clinical protection from malaria. Wilson et al. showed that functional anti-malaria antibodies crossed the placenta, by assessing their functionality using the parasite growth inhibition assay [10]. However, another study showed a positive association between maternal antibody transfer and malaria incidence in the 1st year of life [11]. Obviously, the mechanisms determining maternal antibody transfer and its impact on infants’ malaria infections remain unclear.
In the present study, we investigated serum reactivity against immunogenic antigens from P. falciparum blood stages representing promising vaccine candidates—Apical Membrane Antigen 1 (AMA1), Merozoite Surface Protein (MSP) 1–19, MSP2 (two allelic families: 3D7 and FC27), MSP3 and Glutamate-Rich Protein (GLURP, two regions: R0 and R2)–to highlight the effect of PM in specific antimalarial antibody transfer, considering IgG specificity and maternal hypergammaglobulinemia. The present study also investigated whether lower maternal antibody transfer has consequences on infant health.
Material and Methods
Study design and sample collection
The study took place from June 2007 to January 2010 in Southern Benin (Tori Bossito district). A weekly follow-up of 535 infants from birth to 18 months is detailed in a previous publication [12]. The present work focused on the first 6-month period. Maternal circulating blood (CIRC) and cord blood (CORD) samples were collected in Vacutainer® EDTA (Ethylene diaminetetraacetic acid) tubes at delivery and placental blood smears (from the maternal side of the placenta) were made. Plasma was stored at −80°C. Infections were defined as any febrile event (≥37.5°C) and malaria infection was defined as a febrile event with a positive rapid diagnostic test or a positive thick blood smear. Symptomatic malaria infections were treated with artemether/lumefantrine combination therapy, as per the National Malaria Control Program (Benin) recommendations.
Data collection
The following data were collected: gestational age (estimated using the Ballard method [13], gravidity (primigravid/multigravid), maternal age, maternal weight before delivery, low infant birth weight (defined as a birth weight ≤2500 g, [14]) and presence/absence of PM (defined by the presence of asexual forms of P. falciparum in thick placental smears). Table 1 shows a summary of these variables, according to presence/absence of PM.
Table 1. Characteristics of the population group.
| Variables | n (%) | No placental malaria (n = 481) | With placental malaria (n = 54) | p-value | |
|---|---|---|---|---|---|
| Hypergammaglobulinemia a | < 16 g.L-1 | 366 (68.4) | 329 (89.9) | 37 (10.1) | 0.986 |
| ≥ 16 g.L-1 | 169 (31.6) | 152 (89.9) | 17 (10.1) | ||
| Gravidity a | primigravid | 82 (15.3) | 67 (81.7) | 15 (18.3) | 0.007 |
| multigravid | 453 (84.7) | 414 (91.4) | 39 (8.6) | ||
| Mother's weight (kilograms) b | 535 (100) | 61.8 [47.5;76.1] | 57.9 [51.3;64.5] | 0.053 | |
| Mother's age (years) b | 535 (100) | 27.7 [22.2;33.2] | 25.6 [19.5;31.6] | 0.007 | |
| Low birth weight (grams) a | < 2500 | 48 (9.0) | 40 (83.3) | 8 (16.7) | 0.113 |
| ≥ 2500 | 487 (91.0) | 441 (90.6) | 46 (9.4) | ||
| Gestational age (weeks) b | 535 (100) | 38.5 [36.7;40.3] | 38.3 [36.5;40.1] | 0.437 |
a: Chi2 test;
b: Student unpaired t-test;
percentages are written in parentheses; standard deviations are written in square brackets; in bold: p<0.05.
P. falciparum recombinant antigens
MSP1-19 (Uganda-Palo-Alto strain) and MSP3 (F32 strain) were produced at the Pasteur Institute (Paris, France). MSP2 (3D7 and FC27) were donated by collaborators from La Trobe University (Melbourne, Australia). GLURP-R0 (F32 strain) and GLURP-R2 (F32 strain) were produced by the Infection-Immunity Department of the Statens Serum Institute of Copenhagen (Denmark) and AMA1 was donated by the Biomedical Primate Research Centre (Rijswijk, The Netherlands). All recombinant proteins were expressed in Escherichia coli except AMA1, in Pichia pastoris.
Measurement of antibodies against P. falciparum recombinant antigens
Enzyme-Linked ImmunoSorbent Assay (ELISA) [15,16] was performed to assess malaria antibody concentrations; Afro Immuno Assay (AIA) protocols were followed that are developed to standardize methods for evaluating malaria vaccines and sponsored by the African Malaria Network Trust (AMANET [www.amanet-trust.org]). Briefly, standard curves were established using purified human IgG (Binding Site, France) to determine the concentration of specific antibodies. Each point was tested in duplicate.
Specific and total IgG were measured using recombinant proteins diluted in phosphate-buffered saline (PBS) for specific IgG or an anti-human IgG (Fab-specific, Sigma Aldrich, France) diluted in carbonate buffer for total IgG, both were coated at 0.1-μg/well on MaxiSorp Nunc plates (Thermo Fisher Scientific, Denmark) and blocked with 3% powdered milk, 0.1% Tween, 20 PBS. Maternal and cord blood samples were diluted at 1:200 for all IgG-specific, recombinant proteins except for AMA1 (1:2,000) and total IgG (1:1,000,000). An anti-human IgG coupled with peroxidase (1:3,000, Caltag, UK) was used to reveal the reaction with TMB One (3, 3′, 5, 5′-tetramethylbenzidine, Mast Diagnostic, France). Plates were read at 450 nm.
Management of ELISA data
ELISA optical densities (OD) were analyzed with ADAMSEL FLP b039 software (http://www.emvda.org/portfolio/project-index/optimalvac-project-completed), to determine antibody concentrations (μg/mL). Discordant duplicates (with a variation coefficient >15%) were re-tested.
A stochastic expectation maximization algorithm [17], already applied to ELISA analyses [18], was used to impute data in cases where the OD was below detection thresholds or was oversaturated [referred to as “Low” and “High” concentration values (μg/mL), respectively].
Placental antibody transfer
Prior to analyses, antibody levels were log-transformed (natural logarithm (ln)). Placental antibody transfer was defined as the natural logarithm of the ratio between neonatal and maternal antibody levels expressed in μg/mL (ln (CORD/CIRC)).
Statistical analyses
For quantitative variables, means were compared using a Student unpaired t-test. A chi-squared test was used to compare categorical variables (Table 1). Linear regression was used for both univariate and multivariate analyses of the antibody transfer. Analyses were adjusted according to maternal-specific IgG levels, hypergammaglobulinemia (defined as total IgG ≥16 g.L−1 [1–3]) and the variables listed in the “Data collection” section. A logistic regression was used for both univariate and multivariate analyses of the impact of the maternal antibody transfer on infant infections. A multinomial logistic model investigated the effect of maternal antibody transfer on malaria or non-malaria infections in the infants. Multivariate analysis was performed, including the variables with p≤0.20 in the univariate step. Statistical significance was set at p<0.05 in the linear regression analysis and at p<0.01 in the logistic regression. All statistical analyses were performed using Stata, version 13.0 (StatCorp LP, College Station, TX, USA).
Ethics
This study protocol was approved by the institutional Ethics Committee of the Faculté des Sciences de la Santé from the Université d’Abomey-Calavi in Benin and the Comité Consultatif de Déontologie et d’Ethique from the French Institut de Recherche pour le Développement. The nature of the project was explained in detail to the participants and informed consent was obtained from the women included in the study. All women signed informed consent before enrollment (which also included their children) and could withdraw from the study at any time [12].
Results
Description of the cohort
Ten percent of the pregnant women enrolled had an active placental infection at delivery (n = 54). Maternal hypergammaglobulinemia, maternal weight, infant gestational age and low infant birth weight were not affected by PM. The risk of PM was higher in primigravid mothers and in younger mothers (Table 1).
IgG levels in maternal peripheral blood and cord blood
Maternal-specific IgG levels were higher at delivery in women presenting with PM than those without PM. This difference was significant for AMA1, MSP1-19, MSP2-3D7 and MSP3 (all p<0.049; Table 2). Interestingly, most antimalarial IgG levels in cord blood were similar, whether or not the mother presented with PM at delivery, with the exception of higher IgG levels to MSP2-FC27 (p = 0.035) and lower total IgG levels (p = 0.026) in PM infection (Table 2).
Table 2. Cord blood and maternal specific antibody levels as well as their ratio, according to placental malaria infection.
| Antigen | Bloodcompartment | IgG levels b | p-value | |
|---|---|---|---|---|
| No placental infection (n = 481) | With placental infection (n = 54) | |||
| AMA1 | CIRC a | 577.81 [573.68;581.93] | 897.33 [893.61;901.04] | 0.03 |
| CORD a | 518.62 [514.95;522.28] | 667.16 [663.44;670.87] | 0.178 | |
| (CORD/CIRC) a | 0.89 [-0.69;2.46] | 0.74 [-0.87;2.36] | 0.007 | |
| MSP1-19 | CIRC | 29.91 [22.65;37.17] | 51.50 [46.41;56.58] | 0.053 |
| CORD | 22.53 [16.55;28.50] | 27.37 [22.83;31.90] | 0.442 | |
| (CORD/CIRC) | 0.76 [-1.03;2.55] | 0.53 [-1.35;2.41] | <0.001 | |
| MSP2-3D7 | CIRC | 84.64 [80.32;88.96] | 130.34 [126.52;134.16] | 0.039 |
| CORD | 47.16 [43.35;50.97] | 58.23 [54.91;61.56] | 0.268 | |
| (CORD/CIRC) | 1.79 [-0.12;3.71] | 2.24 [0.34;4.14] | 0.018 | |
| MSP2-FC27 | CIRC | 50.09 [46.81;53.37] | 65.79 [62.90;68.69] | 0.107 |
| CORD | 94.01 [89.70;98.32] | 145.62 [142.07;149.17] | 0.035 | |
| (CORD/CIRC) | 1.88 [-0.08;3.83] | 2.21 [0.32;4.11] | 0.085 | |
| MSP3 | CIRC | 8.17 [2.50;13.83] | 13.99 [8.41;19.59] | 0.031 |
| CORD | 5.50 [0.42;10.59] | 7.33 [2.50;12.15] | 0.22 | |
| (CORD/CIRC) | 0.67 [-1.01;2.35] | 0.52 [-1.11;2.15] | 0.001 | |
| GLURP-R0 | CIRC | 5.49 [0.63;10.36] | 7.41 [2.31;12.51] | 0.189 |
| CORD | 3.36 [-1.07;7.80] | 3.46 [-1.17;8.09] | 0.894 | |
| (CORD/CIRC) | 0.61 [-1.11;2.33] | 0.47 [-1.13;2.06] | 0.001 | |
| GLURP-R2 | CIRC | 36.97 [33.04;40.90] | 54.62 [49.45;59.78] | 0.052 |
| CORD | 21.47 [17.89;25.06] | 22.70 [18.73;26.68] | 0.763 | |
| (CORD/CIRC) | 0.57 [-1.15;2.29] | 0.42 [-1.67;2.51] | <0.001 | |
| Nonspecific total IgG | CIRC | 12096.24 [12093.78;12098.71] | 10943.12 [10940.42;10945.81] | 0.444 |
| CORD | 10703.50 [10701.21;10705.78] | 8177.39 [8174.82;8179.97] | 0.026 | |
| (CORD/CIRC) | 0.88 [-1.21;2.98] | 0.75 [-1.13;2.63] | 0.105 |
a: CIRC: maternal peripheral blood at delivery; CORD: cord blood, (CORD/CIRC): ratio representing the transfer of maternal IgG to the neonate at birth;
b: geometric mean [95% confidence interval] of the IgG concentrations;
Student’s unpaired t-test was performed; n: effective; in bold: p< 0.05.
Placental transfer of malaria-specific IgG
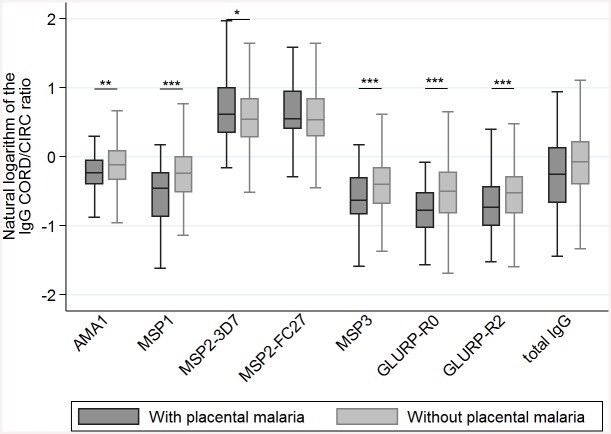
Placental transfer of malaria-specific IgG was significantly reduced in the presence of PM (all p<0.019) for all antigens except MSP2-FC27 and total IgG (Table 2, Fig 1). PM and maternal-specific IgG levels were the two main factors consistently associated with the reduction of placental transfer, except MSP2 (PM) and total IgG (maternal IgG levels) (Table 3). Maternal hypergammaglobulinemia impaired the transfer of some specific IgG (directed to AMA1, MSP2-FC27 and GLURP-R2) and total IgG from the mother to the fetus (Table 3). Other factors were occasionally associated with the CORD/CIRC ratio: the transfer of IgG specific to MSP2-3D7 was positively associated with gestational age, maternal age and maternal-specific IgG levels. The transfer of MSP1 was negatively associated with gestational age.
Fig 1. Cord-to-mother IgG transfer according to placental malaria.
CIRC: maternal peripheral blood at delivery; CORD: cord blood, ln(CORD/CIRC): ratio representing the transfer of maternal IgG to the neonate at birth; * <0.05; ** ≤0.01; *** ≤0.001.
Table 3. Linear regression model showing the factors involved in impaired maternal IgG transfer.
| AMA1 | MSP1 | MSP2-3D7 | ||||
| Variables | coeff a | p-value | coeff | p-value | coeff | p-value |
| Placental malaria | -0.126 [-0.245;-0.006] | 0.039 | -0.293 [-0.44;-0.145] | <0.001 | ||
| Gestational age | -0.029 [-0.054;-0.004] | 0.023 | 0.265 [0.096;0.433] | 0.002 | ||
| Maternal age | 0.016 [0.005;0.027] | 0.004 | ||||
| Maternal-specific IgG | -0.118 [-0.144;-0.092] | <0.001 | -0.135 [-0.158;-0.112] | <0.001 | 0.187 [0.152;0.221] | <0.001 |
| Hypergammaglobulinemia | -0.097 [-0.176;-0.018] | 0.016 | ||||
| MSP2-FC27 | MSP3 | GLURP-R0 | ||||
| Variables | coeff | p-value | coeff | p-value | coeff | p-value |
| Placental malaria | -0.188 [-0.324;-0.051] | 0.007 | -0.232 [-0.375;-0.089] | 0.002 | ||
| Gestational age | ||||||
| Maternal age | ||||||
| Maternal-specific IgG | 0.084 [0.037;0.132] | <0.001 | -0.107 [-0.13;-0.083] | <0.001 | -0.113 [-0.141;-0.086] | <0.001 |
| Hypergammaglobulinemia | 0.2 [0.08;0.319] | 0.001 | ||||
| GLURP-R2 | Total IgG | |||||
| Variables | coeff | p-value | coeff | p-value | ||
| Placental malaria | -0.252 [-0.398;-0.106] | 0.001 | ||||
| Gestational age | ||||||
| Maternal age | ||||||
| Maternal-specific IgG | -0.153 [-0.185;-0.121] | <0.001 | ||||
| Hypergammaglobulinemia | -0.157 [-0.253;-0.061] | 0.001 | -0.581 [-0.705;-0.458] | <0.001 |
a: coeff [95% confidence interval]: coefficient determined by the linear model. A positive (negative) coefficient corresponds to a positive (negative) association between two variables.
Maternal antibody transfer and infant infections
Maternal age was negatively associated with the presence of a febrile infection (malaria or non-malaria) in 6-month-old infants (Table 4). Given the number of tests, these results must be carefully interpreted and therefore differences with p<0.01 were considered significant. In this context, hypergammaglobulinemia and maternal IgG levels to MSP2-FC27 tended to be negatively and positively associated with the occurrence of an infant infection, respectively. Neither the transfer of other malaria-specific IgG nor of total IgG was associated with infection incidence. Placental malaria was not associated with 6-month-old infant infections. When infections were split into non-malaria (mainly gastrointestinal (31.6%) and respiratory (65.2%) febrile syndromes [19]) and malaria, only maternal age was negatively associated with the presence of a malaria infection in infants less than 6 months of age (Table 5).
Table 4. Logistic regression model showing the factors involved in infant infections.
| Factors | coeff a | p-value |
|---|---|---|
| Hypergammaglobulinemia | -0.457 [-0.849;-0.064] | 0.023 |
| Maternal age | -0.049 [-0.082;-0.017] | 0.003 |
| Maternal IgG to MSP2-FC27 | 0.291 [0.011;0.570] | 0.042 |
a: coeff [95% confidence interval]: coefficient determined by the logistic model. A positive (negative) coefficient corresponds to a positive (negative) association between two variables; in bold: p<0.01.
Table 5. Multinomial logistic regression model showing the factors involved in infant malaria/non-malaria infections.
| Infections | Factors | coeff a | p-value |
|---|---|---|---|
| Non-malaria infection | Hypergammaglobulinemia | -0.474 [-0.910;-0.037] | 0.033 |
| Maternal age | -0.041 [-0.076;-0.005] | 0.025 | |
| Maternal IgG to MSP2-FC27 | 0.314 [0.008;0.620] | 0.044 | |
| Malaria infection | Hypergammaglobulinemia | -0.413 [-1.024;-0.198] | 0.185 |
| Maternal age | -0.072 [-0.122;-0.021] | 0.005 | |
| Maternal IgG to MSP2-FC27 | 0.229 [-0.200;0.658] | 0.296 |
a: coeff [95% confidence interval]: coefficient determined by the logistic model. A positive (negative) coefficient corresponds to a positive (negative) association between two variables; in bold: p<0.01.
Discussion
This study aimed to describe the neonatal transfer of maternal antibodies specific to malaria blood-stage antigens. It showed that i) PM was associated with a lower CORD/CIRC ratio of malaria-specific IgG, ii) maternal IgG transfer differed depending on malaria antibody specificity, iii) cord blood antimalarial IgG levels were equivalent after adjustment for hypergammaglobulinemia and PM and iv) older maternal age was associated with fewer malaria infections in infants.
One limitation of this study may be the definition of PM based on light microscopy. As antibody transfer increases at the end of pregnancy, an active placental infection could reinforce the strong association with lower antibody transfer. Using more sensitive methods such as histopathology or molecular biology, which can determine low parasitemia in placental infections, could refine the results. However, a recent study in which placental infection was defined by light microscopy, quantitative PCR or histological examination provided results comparable to those in this study [11].
The present study confirmed that hypergammaglobulinemia or high levels of specific antibodies in maternal blood were associated with a decrease in the CORD/CIRC ratio after adjustment for PM. Regarding the mechanism of placental antibody transfer, it has been suggested that the FcRn expressed by syncytiotrophoblasts in placenta binds IgG with subsequent internalization of the FcRn-IgG complex in endosomal compartments and release of maternal IgG in fetal circulation [20,21]. The amount of IgG transmitted depends on the number of cell-surface receptors because unbound IgG are digested by lysosomal enzymes inside the vesicles, as has been suggested [22]. Some IgG may also be retained in the maternal compartment due to host differences in the functionality and/or density of the FcRn [23] regardless of malaria. We also hypothesize that the parasitized erythrocytes could adhere to syncytiotrophoblast vesicles, where the IgG binding site is assumed to be [21,24], physically impairing IgG capture from the maternal compartment. To our knowledge, no evidence exists to prove that the receptors of parasitized erythrocytes (chondroitin sulfate A) and vesicles are located at different parts of the syncytiotrophoblast. Finally, maternal IgG could be monopolized in the intervillous space to combat a local malaria infection, contributing to decreasing transplacental IgG transfer.
Several studies, including one that uses the same cohort as the current study [25–29], established that infants born to mothers with PM detected at delivery were more susceptible to malaria infections in infancy than others. This occurred independently of similar specific antibody levels measured in the cord blood of infants born to infected and noninfected mothers [14], suggesting that the PM-associated decrease of maternal IgG transfer may not explain the higher risk of malaria infection observed in these infants and may instead be a marker of maternal exposure to parasites [11].
These results show that maternal IgG to MSP2-FC27 tended to be positively related to the number of non-malaria infections in infants under 6 months old, independently of PM. Given the few tests performed, these results should be interpreted with caution. However, a positive association between the transfer of IgG1 to AMA1 as well as IgG to parasite lysate and malaria incidence during the 1st year of life was also observed in another study [11]. Similarly, maternal IgG levels to variant surface antigens expressed by chondroitin sulfate A-adhering parasites were negatively correlated to time before the first parasitemia in Cameroonian infants and positively correlated to mean parasite density, suggesting a nonprotective role [30]. Lastly, a Ghanian study conducted showed that high maternal IgG levels to MSP1-19, MSP2-FC27 and Pf155/RESA were associated with a higher risk of infant infection [31].
Based on these studies and the present work, it seems that maternal IgG specific to malaria is a marker of maternal exposure to P. falciparum. Several studies have already shown that maternal antibodies can inhibit infant responses to different diseases, such as measles or tetanus [32–34]. How maternal antimalarial IgG influences malaria or non-malaria infection incidence in infants remains unclear and needs further investigation.
Interestingly, we found that the older a pregnant woman was, lower the number of malaria infections of her infant was, suggesting that effective maternal immunity acquired with age can be transmitted to offspring. This raises questions on how the immune response evolves in pregnancy and we speculate on the role of one or several maternal molecules. These molecules must i) be produced differently with age and ii) have the property of either crossing the placental barrier or inducing, by their presence at the maternal—fetal interface, tolerance/protection against malaria infections during the first 6 months of life. Cytokines and antibodies are two types of molecules with these properties, in agreement with a definition of transplacental immune regulation formulated by Santner et al. as “the concept that during pregnancy, significant cross-talk occurs between the maternal and fetal immune system with potential long-term effects for both the mother and child” [35]. Few clues are available in the literature, particularly IL-10, which is a cytokine with known immunomodulatory properties [36]. Newborns born to young mothers have been shown to present higher fetal plasma IL-10 levels than newborns born to older mothers [37]. Similarly, maternal IL-10 levels have been negatively associated with parity [38], which may reasonably be related to maternal age. Evidence for alignment between maternal and fetal cellular immunity has been provided, implying T regulatory cells and IL-10 concentrations [35]. Lastly, higher IL-10 levels were found in malaria-exposed, non-sensitized newborns compared to sensitized, unexposed newborns, with this phenotype persisting during childhood [28]. Based on these studies, we hypothesize that IL-10 levels in the sera of mother and her fetus may act as a molecular contributor increasing protection of the infant against malaria infection depending on maternal age.
This study did not explore the functionality of maternal antibodies, possibly one of the immune parameters evolving with maternal age (in that the total IgG quantity was not associated with maternal age resulting from a linear regression coefficient = −0.0038; p = 0.598). Transfer of functional malaria antibodies to the fetus has been demonstrated by measuring their ability to inhibit parasite growth in cord blood and infant blood, but the experiment did not consider maternal blood or maternal age [10]. In fact, the sole antibodies that evolve in a parity- (and therefore age-) dependent manner in malaria-endemic areas are those directed to the variant surface antigen responsible for placental adhesion of infected erythrocytes, represented by the VAR2CSA variant of the P. falciparum erythrocyte membrane protein 1 (PfEMP1) antigen [39–41]. Maternal transfer of functional antibodies or anti-VAR2CSA antibodies to the fetus and the cytokine levels in fetal plasma depending on maternal age should be further investigated to provide new clues on whether or not maternal age plays a protective role in infant health.
In conclusion, This study confirmed lower maternal transfer of IgG directed to several asexual stages of P. falciparum in placental malaria. Why the degree of maternal antimalarial IgG transfer was not related to the incidence of febrile infections in infants’ first 6 months of life, but instead maternal age, needs further investigation.
Acknowledgments
We thank all the women and infants of Tori Bossito who agreed to participate in the study as well as the CERPAGE (Centre d'Étude et de Recherche sur le Paludisme Associé à la Grossesse et l'Enfance) team at Cotonou and the Tori Bossito medical health center staff (F. Loumedjinon and M Daoudou) for performing fieldwork. We are grateful to R. Anders (La Trobe University, Melbourne, Australia), S. Longacre (Pasteur Institute, Paris, France), E. Remarque (Biomedical Primate Research Centre, Rijswijk, The Netherlands) and M. Theisen (Statens Serum Institute, Copenhagen, Denmark) for their gift of P. falciparum recombinant proteins and G. Tavera and I. Malhotra for critical reading of the manuscript. We thank the Faculté des Sciences de la Santé, the Institut des Sciences Biomédicales Appliquées de Cotonou (ISBA) and the Programme National de Lutte contre le Paludisme (PNLP) for their institutional support. C. Dechavanne benefited from the financial support of the Treilles Foundation. This study was funded by the Agence Nationale de la Recherche (ANR) Santé Environnement Santé Travail (SEST 2006; 040 01).
Data Availability
All relevant data are within the paper.
Funding Statement
The funding for this study was received from Agence Nationale de la Recherche (ANR) Santé Environnement Santé Travail (SEST 2006; 040 01). Celia Dechavanne benefited from the financial support of the Treilles Foundation.
References
- 1. de Moraes-Pinto MI, Verhoeff F, Chimsuku L, Milligan PJ, Wesumperuma L, Broadhead RL, et al. Placental antibody transfer: influence of maternal HIV infection and placental malaria. Arch Dis Child Fetal Neonatal Ed. 1998;79: F202–205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Okoko BJ, Wesuperuma LH, Ota MO, Banya WA, Pinder M, Gomez FS, et al. Influence of placental malaria infection and maternal hypergammaglobulinaemia on materno-foetal transfer of measles and tetanus antibodies in a rural west African population. J Health Popul Nutr. 2001;19: 59–65. [PubMed] [Google Scholar]
- 3. Brair ME, Brabin BJ, Milligan P, Maxwell S, Hart CA. Reduced transfer of tetanus antibodies with placental malaria. Lancet. 1994;343: 208–209. [DOI] [PubMed] [Google Scholar]
- 4. Scott S, Cumberland P, Shulman CE, Cousens S, Cohen BJ, Brown DWG, et al. Neonatal Measles Immunity in Rural Kenya: The Influence of HIV and Placental Malaria Infections on Placental Transfer of Antibodies and Levels of Antibody in Maternal and Cord Serum Samples. J Infect Dis. 2005;191: 1854–1860. [DOI] [PubMed] [Google Scholar]
- 5. Deloron P, Dubois B, Le Hesran JY, Riche D, Fievet N, Cornet M, et al. Isotypic analysis of maternally transmitted Plasmodium falciparum-specific antibodies in Cameroon, and relationship with risk of P. falciparum infection. Clin Exp Immunol. 1997;110: 212–218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Hartter HK, Oyedele OI, Dietz K, Kreis S, Hoffman JP, Muller CP. Placental transfer and decay of maternally acquired antimeasles antibodies in Nigerian children. Pediatr Infect Dis J. 2000;19: 635–641. [DOI] [PubMed] [Google Scholar]
- 7. Gitlin D. The Transmission of Passive Immunity from Mother to Young (North-Holland Research Monographs, Frontier of Biology Vol. 18), by F. W. Rogers Brambell. New York: American Elsevier Publishing Company, Inc., 1970, 385 pp, Pediatrics. 1971;48: 172–172. [Google Scholar]
- 8. Jones EA, Waldmann TA. The mechanism of intestinal uptake and transcellular transport of IgG in the neonatal rat. J Clin Invest. 1972;51: 2916–2927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Waldmann TA, Strober W. Metabolism of immunoglobulins. Prog Allergy. 1969;13: 1–110. [DOI] [PubMed] [Google Scholar]
- 10. Wilson PT, Malhotra I, Mungai P, King CL, Dent AE. Transplacentally transferred functional antibodies against Plasmodium falciparum decrease with age. Acta Trop. 2013;128: 149–153. 10.1016/j.actatropica.2013.07.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Moro L, Bardají A, Nhampossa T, Mandomando I, Serra-Casas E, Sigaúque B, et al. Malaria and HIV Infection in Mozambican Pregnant Women Are Associated With Reduced Transfer of Antimalarial Antibodies to Their Newborns. J Infect Dis. 2014. [DOI] [PubMed] [Google Scholar]
- 12. Le Port A, Cottrell G, Martin-Prevel Y, Migot-Nabias F, Cot M, Garcia A. First malaria infections in a cohort of infants in Benin: biological, environmental and genetic determinants. Description of the study site, population methods and preliminary results. BMJ Open. 2012;2: e000342 10.1136/bmjopen-2011-000342 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Ballard JL, Khoury JC, Wedig K, Wang L, Eilers-Walsman BL, Lipp R. New Ballard Score, expanded to include extremely premature infants. J Pediatr. 1991;119: 417–423. [DOI] [PubMed] [Google Scholar]
- 14. Logie DE, McGregor IA, Rowe DS, Billewicz WZ. Plasma immunoglobulin concentrations in mothers and newborn children with special reference to placental malaria: Studies in the Gambia, Nigeria, and Switzerland. Bull World Health Organ. 1973;49: 547–554. [PMC free article] [PubMed] [Google Scholar]
- 15. Nebie I, Diarra A, Ouedraogo A, Soulama I, Bougouma EC, Tiono AB, et al. Humoral responses to Plasmodium falciparum blood-stage antigens and association with incidence of clinical malaria in children living in an area of seasonal malaria transmission in Burkina Faso, West Africa. Infect Immun. 2008;76: 759–766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Lusingu JPA, Vestergaard LS, Alifrangis M, Mmbando BP, Theisen M, Kitua AY, et al. Cytophilic antibodies to Plasmodium falciparum glutamate rich protein are associated with malaria protection in an area of holoendemic transmission. Malar J. 2005;4: 48 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Celeux G, Diebolt J. SEM algorithm: a probabilistic learning algorithm for recognition of density mixtures. Rev Stat Appliquée. 1986;34: 35–52. [Google Scholar]
- 18. Moulton LH, Halsey NA. A mixture model with detection limits for regression analyses of antibody response to vaccine. Biometrics. 1995;51: 1570–1578. [PubMed] [Google Scholar]
- 19. Rachas A, Le Port A, Cottrell G, Guerra J, Choudat I, Bouscaillou J, et al. Placental malaria is associated with increased risk of nonmalaria infection during the first 18 months of life in a Beninese population. Clin Infect Dis Off Publ Infect Dis Soc Am. 2012;55: 672–678. [DOI] [PubMed] [Google Scholar]
- 20. Roopenian DC, Akilesh S. FcRn: the neonatal Fc receptor comes of age. Nat Rev Immunol. 2007;7: 715–725. [DOI] [PubMed] [Google Scholar]
- 21. Palmeira P, Quinello C, Silveira-Lessa AL, Zago CA, Carneiro-Sampaio M. IgG placental transfer in healthy and pathological pregnancies. Clin Dev Immunol. 2012;2012: 985646 10.1155/2012/985646 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Saji F, Koyama M, Matsuzaki N. Current topic: human placental Fc receptors. Placenta. 1994;15: 453–466. [DOI] [PubMed] [Google Scholar]
- 23. Sachs UJH, Socher I, Braeunlich CG, Kroll H, Bein G, Santoso S. A variable number of tandem repeats polymorphism influences the transcriptional activity of the neonatal Fc receptor alpha-chain promoter. Immunology. 2006;119: 83–89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Ghetie V, Ward ES. Transcytosis and catabolism of antibody. Immunol Res. 2002;25: 97–113. [DOI] [PubMed] [Google Scholar]
- 25. Le Hesran JY, Cot M, Personne P, Fievet N, Dubois B, Beyemé M, et al. Maternal placental infection with Plasmodium falciparum and malaria morbidity during the first 2 years of life. Am J Epidemiol. 1997;146: 826–831. [DOI] [PubMed] [Google Scholar]
- 26. Mutabingwa TK, Bolla MC, Li J-L, Domingo GJ, Li X, Fried M, et al. Maternal malaria and gravidity interact to modify infant susceptibility to malaria. PLoS Med. 2005;2: e407 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Schwarz NG, Adegnika AA, Breitling LP, Gabor J, Agnandji ST, Newman RD, et al. Placental malaria increases malaria risk in the first 30 months of life. Clin Infect Dis Off Publ Infect Dis Soc Am. 2008;47: 1017–1025. [DOI] [PubMed] [Google Scholar]
- 28. Malhotra I, Dent A, Mungai P, Wamachi A, Ouma JH, Narum DL, et al. Can prenatal malaria exposure produce an immune tolerant phenotype? A prospective birth cohort study in Kenya. PLoS Med. 2009;6: e1000116 10.1371/journal.pmed.1000116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Le Port A, Watier L, Cottrell G, Ouédraogo S, Dechavanne C, Pierrat C, et al. Infections in infants during the first 12 months of life: role of placental malaria and environmental factors. PloS One. 2011;6: e27516 10.1371/journal.pone.0027516 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Cot M, Le Hesran JY, Staalsoe T, Fievet N, Hviid L, Deloron P. Maternally transmitted antibodies to pregnancy-associated variant antigens on the surface of erythrocytes infected with Plasmodium falciparum: relation to child susceptibility to malaria. Am J Epidemiol. 2003;157: 203–209. [DOI] [PubMed] [Google Scholar]
- 31. Riley EM, Wagner GE, Ofori MF, Wheeler JG, Akanmori BD, Tetteh K, et al. Lack of association between maternal antibody and protection of African infants from malaria infection. Infect Immun. 2000;68: 5856–5863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Siegrist C-A. Mechanisms by which maternal antibodies influence infant vaccine responses: review of hypotheses and definition of main determinants. Vaccine. 2003;21: 3406–3412. [DOI] [PubMed] [Google Scholar]
- 33. Gans H, Yasukawa L, Rinki M, DeHovitz R, Forghani B, Beeler J, et al. Immune responses to measles and mumps vaccination of infants at 6, 9, and 12 months. J Infect Dis. 2001;184: 817–826. [DOI] [PubMed] [Google Scholar]
- 34. Gans H, DeHovitz R, Forghani B, Beeler J, Maldonado Y, Arvin AM. Measles and mumps vaccination as a model to investigate the developing immune system: passive and active immunity during the first year of life. Vaccine. 2003;21: 3398–3405. [DOI] [PubMed] [Google Scholar]
- 35. Santner-Nanan B, Straubinger K, Hsu P, Parnell G, Tang B, Xu B, et al. Fetal-maternal alignment of regulatory T cells correlates with IL-10 and Bcl-2 upregulation in pregnancy. J Immunol Baltim Md 1950. 2013;191: 145–153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Mosser DM, Zhang X. Interleukin-10: new perspectives on an old cytokine. Immunol Rev. 2008;226: 205–218. 10.1111/j.1600-065X.2008.00706.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Fievet N, Varani S, Ibitokou S, Briand V, Louis S, Perrin RX, et al. Plasmodium falciparum exposure in utero, maternal age and parity influence the innate activation of foetal antigen presenting cells. Malar J. 2009;8: 251 10.1186/1475-2875-8-251 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Megnekou R, Lissom A, Bigoga JD, Djontu JC. Effects of Pregnancy-associated Malaria on T Cell Cytokines in Cameroonian Women. Scand J Immunol. 2015;81: 508–514. 10.1111/sji.12286 [DOI] [PubMed] [Google Scholar]
- 39. Fowkes FJI, McGready R, Cross NJ, Hommel M, Simpson JA, Elliott SR, et al. New insights into acquisition, boosting, and longevity of immunity to malaria in pregnant women. J Infect Dis. 2012;206: 1612–1621. 10.1093/infdis/jis566 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Dechavanne S, Srivastava A, Gangnard S, Nunes-Silva S, Dechavanne C, Fievet N, et al. Parity-dependent recognition of DBL1X-3X suggests an important role of the VAR2CSA high-affinity CSA-binding region in the development of the humoral response against placental malaria. Infect Immun. 2015;83: 2466–2474. 10.1128/IAI.03116-14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Salanti A, Dahlbäck M, Turner L, Nielsen MA, Barfod L, Magistrado P, et al. Evidence for the Involvement of VAR2CSA in Pregnancy-associated Malaria. J Exp Med. 2004;200: 1197–1203. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All relevant data are within the paper.