Abstract
AIM: To evaluate the methodology, feasibility, safety and efficacy of a novel method called cap-assisted endoscopic sclerotherapy (CAES) for internal hemorrhoids.
METHODS: A pilot study on CAES for grade I to III internal hemorrhoids was performed. Colon and terminal ileum examination by colonoscopy was performed for all patients before starting CAES. Polypectomy and excision of anal papilla fibroma were performed if polyps or anal papilla fibroma were found and assessed to be suitable for resection under endoscopy. CAES was performed based on the requirement of the cap, endoscope, disposable endoscopic long injection needle, enough insufflated air and sclerosing agent.
RESULTS: A total of 30 patients with grade I to III internal hemorrhoids was included. The follow-up was more than four weeks. No bleeding was observed after CAES. One (3.33%) patient claimed mild tenesmus within four days after CAES in that an endoscopist performed this procedure for the first time. One hundred percent of patients were satisfied with this novel procedure, especially for those patients who underwent CAES in conjunction with polypectomy or excision of anal papilla fibroma.
CONCLUSION: CAES as a novel endoscopic sclerotherapy should be a convenient, safe and effective flexible endoscopic therapy for internal hemorrhoids.
Keywords: Sclerotherapy, Hemorrhoids, Cap-assisted endoscopic sclerotherapy, Colonoscopy, Colon, Papilla fibroma, Hemorrhoidal disease
Core tip: Sclerotherapy is the most effective therapy for grade I or II internal hemorrhoids. Traditional sclerotherapy may cause iatrogenic risk due to misplaced injections. We designed a novel technique called cap-assisted endoscopic sclerotherapy (CAES) for hemorrhoids by flexible endoscopy. Our study demonstrated that CAES is a safe, effective and convenient endoscopic therapeutic strategy for grade I, grade II and partial grade III internal hemorrhoids. The colon preparation and colonoscopy before CAES brought more benefits for patients, including possible polypectomy and excision of anal papilla fibroma under colonoscopy. This study implies the future contribution of endoscopists on hemorrhoidal disease.
INTRODUCTION
Hemorrhoidal disease is one of the most common anorectal disorders that affects mainly adults of any age and sex[1-4]. The etiology of hemorrhoids remains controversial. Nowadays, the most widely accepted theory is that hemorrhoidal disease is the abnormal dilatation and distortion of the vascular channel, together with destructive changes in the supporting connective tissue of the anal cushion[5]. The submucosal vascular cushions are a normal anatomical structure of the anal canal and their existence with symptoms such as bleeding, prolapse, pain, thrombosis, mucus discharge and pruritus indicates hemorrhoidal disease[6]. The true incidence of hemorrhoids is difficult to estimate as many patients are reluctant to seek medical suggestions for various personal, cultural and socioeconomic reasons[7]. Approximately 50% of individuals require treatment for hemorrhoids in their 50s or older, and 10%-20% of patients need surgical therapies[8].
Hemorrhoids have been well described for thousands of years. However, the treatment of hemorrhoids has only substantially evolved during the past few decades[1]. The current therapies for hemorrhoids can be grouped into conservative management, office-based procedures and surgical treatment[8-10]. Increased fiber intake, medical therapies and lifestyle changes are included in the conservative treatment options for non-thrombosed hemorrhoids[10]. If conservative management is unsuccessful, several office-based modalities could be options, including rubber-band ligation, injection sclerotherapy, laser photocoagulation, bipolar diathermy, cryotherapy, Dopplerguided hemorrhoidal artery ligation and infrared coagulation[8,9,11]. When an office-based therapy is still ineffective, patients may consider further intervention, such as hemorrhoidectomy, thrombectomy of external hemorrhoids and stapled hemorrhoidectomy[7,9].
As an crucial component of many non-surgical practices, sclerotherapy is most effective for grade I and II internal hemorrhoids, especially for patients who have an increased risk of bleeding[2]. However, traditional sclerotherapy is performed by physicians through an anoscope. This method may cause iatrogenic risk and complications due to misplaced injections[2]. Therefore, there is scope for improvement in the field of sclerotherapy for hemorrhoids.
With the development of interventional flexible endoscopy and in order to solve the problems above, we designed a novel method called cap-assisted endoscopic sclerotherapy (CAES) for internal hemorrhoids. This article presents our pilot study on the methodology, feasibility, safety and clinical findings using CAES for internal hemorrhoids.
MATERIALS AND METHODS
Patient inclusion and exclusion criteria
This observational study was carried out in the Second Affiliated Hospital of Nanjing Medical University. All eligible patients with symptoms and signs of grade I, grade II or grade III internal hemorrhoids requiring further interventional procedures after failure of conservative treatment were included in the study. Internal hemorrhoids are graded based on protrusion and reducibility (grade I, hemorrhoids characterized by prominent vasculature with engorgement but no prolapse; grade II, hemorrhoids prolapse only with straining but spontaneously reduce; grade III, hemorrhoids prolapse beyond the dentate line with straining and require manual reduction; grade IV, hemorrhoids prolapse beyond the dentate line with straining but cannot be reduced manually)[12,13]. All included cases for analysis were followed up for at least four weeks.
Acute thrombosed hemorrhoids with anal pain, stricture, fissure, fistula, fecal incontinence, ulcerative colitis, Crohn’s disease and any bleeding risk condition were excluded. Patients with acute diarrhea in the last 12 h, severe complications, cancer, stroke, pregnancy, puerperium, mental disorders and portal hypertension were also excluded. Colon and terminal ileum examination by colonoscopy was performed for all patients before starting CAES. Polypectomy was performed if polyps were found and assessed to be suitable to be resected under endoscope. Informed consent was obtained from all participants.
Concept and methods of CAES
As shown in Figure 1, the regular cap used in endoscopic submucosal dissection was fixed on the top of the colonoscope. This cap is used to maximize visibility of the endoscopic view with enough insufflated air through the channel within the endoscope. A disposable endoscopic long injection needle (e.g., the specially designed long needle: DT-EN-W322, 10/15/20 mm length, 22 g, Detian Medical, Changzhou, China) through the operating channel is used for the injection of the sclerosing agent. The needle is advanced into the submucosa of the targeted area of the hemorrhoids. The injecting points are above the dentate line. The sclerosing agent (Lauromacrogol injection, Tianyu Pharmaceutical, Xi’an, China), 1-2 mL for each injecting point, is injected while retracting the needle slowly. During the procedure, enough air is given for exposure of the endoscopic view. Before the complete retrieval of the needle from the tissue, as a suggestion, it is found to be helpful if you do not withdraw and stop the needle from moving for 5 s to prevent bleeding. The same procedure is performed for each targeted site under endoscopic view. Before taking out the endoscope, enough suction of air in the colon and visible rectal contents should be carried out to avoid or relieve abdominal distention and the feeling of defecation after the procedure.
Figure 1.
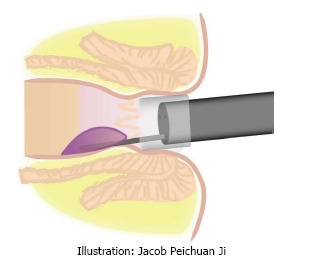
Illustration of cap-assisted endoscopic sclerotherapy.
Preparation and education
Although antibiotic prophylaxis was suggested for predisposing valvular heart disease because of the possibility of bacteremia after sclerotherapy[14], antibiotics were not used before and after CAES in this study. Besides, for safety considerations and observational requirements in this study, patients were required to rest in bed on the first night after the procedure. All individuals were required to follow medical instructions for avoiding constipation and diarrhea. Medicines were prescribed to soften the stools after the procedure if the patient had constipation.
Safety and satisfaction survey
Complications were recorded during and four weeks after CAES. The intensity of CAES and the relationship between the complications and CAES were described using the Common Terminology Criteria for Adverse Events (version 3.0). Intensity of complications was classified as mild, moderate, severe and disabling. The relationship between the complications and CAES was categorized as unrelated, possible, probable and definitely related to CAES. All patients were required to have a face to face communication at the doctor’s office for the assessment of safety, efficacy and degree of satisfaction of the CAES. The level of satisfaction was classified into two degrees: positive, satisfied and pleased to introduce the CAES to other patients; negative, not satisfied and did not like this procedure. The second colonoscopy would be performed if the patient had bleeding or any other anorectal symptoms.
RESULTS
Patient characteristics
Table 1 shows the characteristics of the patients, including gender, age, classification of the internal hemorrhoids, grade of prolapse, previous hemorrhoidectomy history and other related information. A total of 30 patients with grade I, grade II or grade III internal hemorrhoids was included for analysis in this study.
Table 1.
Patient demographics and clinical results
| Patient demographics | n (%) |
| Total included cases | 30 |
| Classification of internal hemorrhoids | |
| Grade I | 7 (23.33) |
| Grade II | 21 (70.0) |
| Grade III | 2 (6.67) |
| Grade IV | 0 |
| Male | 22 (73.33) |
| Age (mean ± SD) | 45.5 ± 4.2 |
| With hemorrhoidectomy history | 6 (20.0) |
| With rectal mucosal prolapse | 4 (13.33) |
| With polyps on hemorrhoid lesions | 1 (3.33) |
| With external hemorrhoids | 0 |
| Colon and terminal ileum examination before CAES | 30 (100) |
| Polypectomy during colonoscopy before CAES | 7 (23.33) |
| Excision of anal papilla fibroma after CAES | 1 (3.33) |
| Biopsy for the polyps on hemorrhoids before CAES | 1 (3.33) |
| Complications during and post-CAES | 0 |
| Post-CAES rectal bleeding | 0 |
| Post-CAES rectal mild pain or tenesmus | 1 (3.33) |
| Positive satisfaction on CAES | 30 (100) |
Data are frequency counts (percentage of total) or the mean ± SD. CAES: Cap-assisted endoscopic sclerotherapy.
Clinical findings
Colon and terminal ileum examination by colonoscopy before CAES was performed in all patients for differential diagnosis of other possible diseases related to intestinal bleeding. No complications were observed during the procedure. However, we have to highlight that the needle could not be retrieved immediately when the injection was finished. It is suggested to keep the needle stable within the tissue for 5 s. If the needle was taken out from the tissue too quickly, bleeding would occur and the endoscopic view was affected by the blood. Figure 2 shows the procedures of CAES for internal hemorrhoids and the excision for anal papilla fibroma.
Figure 2.
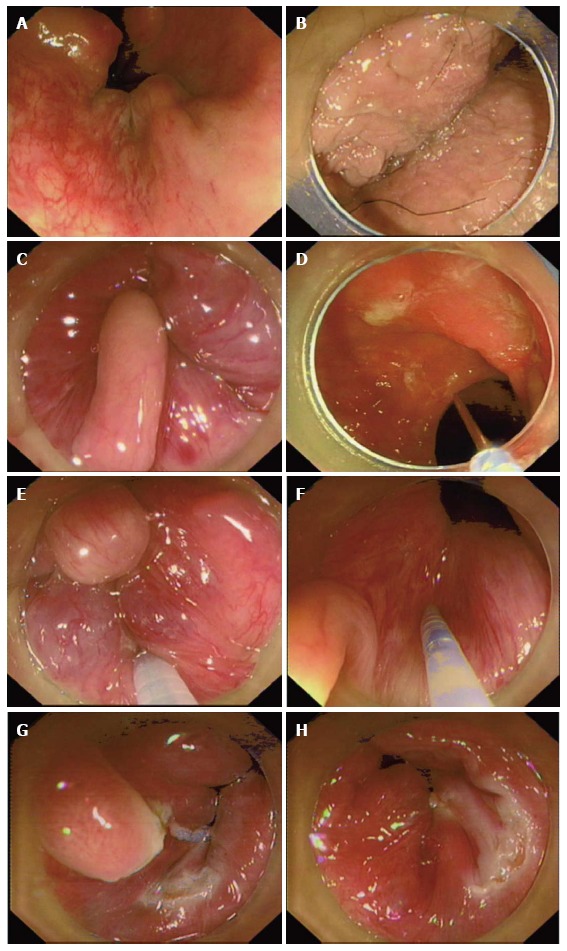
Procedures of cap-assisted endoscopic sclerotherapy for internal hemorrhoids and the excision for anal papilla fibroma. A: Internal hemorrhoids with retroflection of the endoscope; B: The anal region under cap-assisted endoscopic view; C: Internal hemorrhoids and anal papilla fibroma under cap-assisted endoscopic view with enough insufflated air; D: The disposable endoscopic long injection needle through operating channel; E: Injection of lauromacrogol into submucosa of internal hemorrhoids with the cap-assisted endoscopic view; F: Injecting of lauromacrogol into submucosa of internal hemorrhoids close to papilla fibroma before dissection; G: Dissection of anal papilla fibroma (confirmed by the followed pathology) after cap-assisted endoscopic sclerotherapy (CAES); H: No bleeding after CAES and dissection of anal papilla lesion before ending all procedures.
The patients were required to stay in hospital for 12 h after the procedure for safety considerations in this pilot observational study. No complications were observed during and after the procedure of polypectomy, CAES, excision of anal papilla fibroma and biopsy of polyps on the hemorrhoid lesion. All patients could return to normal activities after they were discharged from hospital. One (3.33%) patient claimed mild tenesmus within four days after CAES. This adverse event was finally confirmed as the result of one injection site that was chosen below the dentate line by an endoscopist who performed this procedure for the first time. One hundred percent of patients were satisfied with this novel procedure. Those patients who underwent CAES in junction with polypectomy or excision of anal papilla fibroma expressed strong feeling of satisfaction for the therapeutic strategy.
DISCUSSION
Sclerotherapy dates back at least one century[15] and has been regarded as traditional therapy for grade I and II internal hemorrhoids. A variety of sclerosing agents, including ethanolamine, quinine, hypertonic saline solution, 5% phenol in oil, aluminum potassium sulfate and tannic acid, have been used in injection sclerotherapy for treating hemorrhoids[16-19]. Traditionally doctors had to use an anoscope during the sclerotherapy procedure. Misplacement of the sclerosing injection may result in potential complications, including pain, impotence, prostatitis, mucosal ulceration or necrosis and prostatic abscess[10]. These complications emphasize the importance of precise placement of the injection with the sclerosing agent. In order to avoid the above complications, this prospective study was designed to evaluate the feasibility and efficacy of CAES for internal hemorrhoids under colonoscopy.
The preliminary results based on 30 cases demonstrated that CAES should be an effective interventional flexible endoscopic therapy for selected candidates with grade I to grade III internal hemorrhoids. After CAES, 100% of patients achieved the expected clinical response. The follow-up within 4 wk showed sustained clinical efficacy. No severe or obvious complications were observed and none of the suffered complications were definitely related to CAES in the study. These results indicated that CAES was safe and helpful to prevent iatrogenic risk from misplaced injections. The length of a common commercial endoscopic injection needle was not suggested in CAES because of its short length (e.g., 4 or 5 mm), which seems to require more sites for injection and induce more mucosal injury and potential inflammation. Our specially designed needle, with a 15-20 mm length, is an important tool for enough submucosal injection with the sclerosing agent. Based on our experience, this CAES technique with the transparent cap is able to treat all hemorrhoids in a forward view fashion. There might be no need to have retroflection for the CAES procedure. Importantly, it is impossible for endoscopist to have retroflection in all cases.
In the present study, a high level of patient satisfaction (100%) and the convenience from adequate medical health or psychophysical protection for doctors also provide evidence to support CAES to be promising for the future. Actually, CAES brought additional benefits for patients, such as colonoscopy, possible polypectomy, excision of anal papilla fibroma and biopsy of polyps on hemorrhoid lesions under endoscopy.
Another advantage of doing an endoscopic procedure before CAES to that of using a plain disposable anoscope is that bleeding and other anorectal symptoms related to different colorectal diseases could be better differentiated[20]. A population-based study in the United States[21] reported in the hematochezia cohort showed significantly higher rates of diverticulosis, polyp or multiple polyps, mucosal abnormality/colitis, tumor and solitary ulcers on colonoscopy findings. Anorectal diseases, including hemorrhoids, are frequent in patients with intestinal disease. Hemorrhoids have been reported to occur in 20% of patients with UC[22] and approximately 7% of patients with CD[23]. In these selected cases, lesions in the colon and terminal ileum were observed during the examination by colonoscopy before CAES, which should be an effective way to have an early diagnosis of CD and UC with hemorrhoids. Therefore, colon preparation and colonoscopy is important when dealing with hemorrhoids as it would save the related medical cost and colon preparation for patients.
The cap, endoscope, air, long needle, sclerosing agent and endoscopic view should be the key points for the endoscopist to perform the CAES. This CAES technique is simple but the possible risk should be considered for physicians. One patient claimed mild tenesmus within four days after CAES. This complication was finally confirmed as the result of one injection site chosen below the dentate line by an endoscopist who performed this procedure for the first time. This lesson highlighted the importance of training for CAES. With the necessary training, the angle, direction and depth of injection under endoscopic view could be controlled very well and it would be easy to avoid the risk of injuring deeper tissues or injecting outside of the hemorrhoid.
All cases were required to be hospitalized for bed rest on the first night after the procedure, according to the design of this observational study. However, this hospitalization would not be required if the patient has no other condition except hemorrhoids. For prevention of recurrence of hemorrhoids, medicines and health education are important to maintain soft defecation within the first week after CAES if necessary.
There are some limitations in the present study. The sample size of this pilot study was small but a larger prospective study based on these preliminary results is ongoing. This was not a controlled study with the comparison of other traditional interventional therapies. Therefore, a rigorous randomized clinical trial should be designed to provide more evidence for the practice of CAES. Although CAES and the required preparation of colon and colonoscopy showed advantages and low medical costs for the diagnosis and therapy of anorectal diseases related to hemorrhoidal disease, a cost-effective analysis is needed for further study.
In conclusion, CAES is an innovation of endoscopic sclerotherapy. It should be a convenient, safe and effective flexible endoscopic therapy for internal hemorrhoids. Traditionally, hemorrhoids are commonly treated by surgeons. However, the present study implies the future contribution of endoscopists for hemorrhoidal disease.
ACKNOWLEDGMENTS
We would like to acknowledge Dr. Kashish Dhawan (Dayanand Medical College and Hospital, Ludhiana, India) for kindly editing the manuscript and giving professional suggestions.
COMMENTS
Background
The current therapies for hemorrhoids can be grouped into conservative management, office-based procedures and surgical treatment. As a crucial component of many non-surgical practices, sclerotherapy is most effective for grade I and II internal hemorrhoids. However, traditional sclerotherapy is performed by physicians using an anoscope. This method may cause iatrogenic risk and complications due to misplaced injections. Additionally, an anoscope has the limitation of only being used within the anus. Therefore, there is scope for improvement in the field of sclerotherapy for hemorrhoids.
Research frontiers
With the development of interventional flexible endoscopy, the authors designed a novel method called cap-assisted endoscopic sclerotherapy (CAES) for internal hemorrhoids. This article presents the authors’ pilot study with the methodology, feasibility, safety, clinical findings and their experience using CAES for internal hemorrhoids.
Innovations and breakthroughs
This study demonstrated CAES is a safe, effective and convenient endoscopic therapeutic strategy for grade I, grade II and partial grade III internal hemorrhoids. The colon preparation and colonoscopy are the steps before the final sclerotherapy. The colon preparation and colonoscopy before CAES brought more benefits for patients, including possible polypectomy and excision of anal papilla fibroma under endoscopy. Besides, in these selected cases, lesions in the colon and terminal ileum were observed before CAES during the colonoscopy itself, which should be an effective way to have an early diagnosis of Crohn's disease and Ulcerative colitis with hemorrhoids. Therefore, it would save the related medical cost and colon preparation for patients. This study implies the future contribution of endoscopists on hemorrhoidal disease.
Applications
This pilot study based on 30 cases demonstrated that CAES should be an effective interventional flexible endoscopic therapy for selected candidates with grade I to grade III internal hemorrhoids. After CAES, 100% of patients achieved the expected clinical response. The follow-up within 4 wk further showed the sustained clinical efficacy. No severe or obvious complications were observed and none of the suffered complications were definitely related to CAES in the study. These results indicated that CAES was safe and helpful in preventing iatrogenic risk from misplaced injections. The authors’ specially designed needle, 10/15/20 mm in length, is an important tool to ensure enough submucosal injection with the sclerosing agent. Based on the authors’ experience, this CAES technique with the transparent cap is able to treat all hemorrhoids in a forward view fashion. There may be no need for retroflection for the CAES procedure. Importantly, it is impossible for endoscopist to have retroflection in all cases. In the present study, a high level of patient satisfaction (100%) and the convenience of adequate medical health or psychophysical protection for doctors also provide evidence for supporting CAES to be promising for the future. Actually, CAES brought additional benefits for patients, such as colonoscopy, possible polypectomy, excision of anal papilla fibroma and biopsy of polyps on hemorrhoid lesions under endoscopy.
Terminology
The concept and methods of CAES: The regular cap used in endoscopic submucosal dissection was fixed on the top of the colonoscope. This cap is used to maximize visibility of the endoscopic view with enough insufflated air through the channel within the endoscope. A disposable endoscopic long injection needle through the operating channel is advanced into the submucosa of the targeted area of the hemorrhoids. The injecting points are above the dentate line. The sclerosing agent for each injecting point is injected slowly while retracting the needle slowly. During the procedure, enough air was given for exposure of the endoscopic view. Before the complete retrieval of the needle from the tissue, as a suggestion, it is helpful if you do not withdraw and stop the needle from moving for 5 s to prevent bleeding. The same procedure is performed for each targeted site under endoscopic view.
Peer-review
The authors describe a modification of the band ligation technique of hemorrhoid therapy by using ESD caps with sclerotherapy. The use of a cap mounted on the tip of an endoscope was useful to stabilize its position for precise injection of a sclerosing agent through a long needle. Overall, the model is elegant and the results seem to be promising. In addition to a novel technique, their analysis is rigorous, including the use of a post-procedure questionnaire. The images and diagram are also excellent.
Footnotes
P- Reviewer: Fusaroli P, Schneider E S- Editor: Qi Y L- Editor: Roemmele A E- Editor: Lu YJ
Supported by The Intestine Initiative Foundation, Clinical Science and Technology Foundation of Jiangsu Province, No. BL2014097; the National Science and Technology Major Project, No. 2012BAI06B03; and the National Gastroenterology Research Project, No. 2015BAI13B07.
Institutional review board statement: The study was reviewed and approved by the Second Affiliated Hospital of Nanjing Medical University Institutional Review Board.
Informed consent statement: All study participants, or their legal guardian, provided informed written consent prior to study enrollment.
Conflict-of-interest statement: We declare that there are no conflicts of interest to disclose.
Data sharing statement: No additional data are available.
Open-Access: This article is an open-access article which was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/
Peer-review started: September 1, 2015
First decision: October 14, 2015
Article in press: December 4, 2015
References
- 1.Altomare DF, Giuratrabocchetta S. Conservative and surgical treatment of haemorrhoids. Nat Rev Gastroenterol Hepatol. 2013;10:513–521. doi: 10.1038/nrgastro.2013.91. [DOI] [PubMed] [Google Scholar]
- 2.Jacobs D. Clinical practice. Hemorrhoids. N Engl J Med. 2014;371:944–951. doi: 10.1056/NEJMcp1204188. [DOI] [PubMed] [Google Scholar]
- 3.MacKay D. Hemorrhoids and varicose veins: a review of treatment options. Altern Med Rev. 2001;6:126–140. [PubMed] [Google Scholar]
- 4.Riss S, Weiser FA, Schwameis K, Riss T, Mittlböck M, Steiner G, Stift A. The prevalence of hemorrhoids in adults. Int J Colorectal Dis. 2012;27:215–220. doi: 10.1007/s00384-011-1316-3. [DOI] [PubMed] [Google Scholar]
- 5.Thomson WH. The nature of haemorrhoids. Br J Surg. 1975;62:542–552. doi: 10.1002/bjs.1800620710. [DOI] [PubMed] [Google Scholar]
- 6.Elshazly WG, Gazal AE, Madbouly K, Hussen A. Ligation anopexy versus hemorrhoidectomy in the treatment of second- and third-degree hemorrhoids. Tech Coloproctol. 2015;19:29–34. doi: 10.1007/s10151-014-1235-8. [DOI] [PubMed] [Google Scholar]
- 7.Acheson AG, Scholefield JH. Management of haemorrhoids. BMJ. 2008;336:380–383. doi: 10.1136/bmj.39465.674745.80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Johanson JF, Sonnenberg A. The prevalence of hemorrhoids and chronic constipation. An epidemiologic study. Gastroenterology. 1990;98:380–386. doi: 10.1016/0016-5085(90)90828-o. [DOI] [PubMed] [Google Scholar]
- 9.Nisar PJ, Scholefield JH. Managing haemorrhoids. BMJ. 2003;327:847–851. doi: 10.1136/bmj.327.7419.847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lohsiriwat V. Hemorrhoids: from basic pathophysiology to clinical management. World J Gastroenterol. 2012;18:2009–2017. doi: 10.3748/wjg.v18.i17.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Siddiqui UD, Barth BA, Banerjee S, Bhat YM, Chauhan SS, Gottlieb KT, Konda V, Maple JT, Murad FM, Pfau P, et al. Devices for the endoscopic treatment of hemorrhoids. Gastrointest Endosc. 2014;79:8–14. doi: 10.1016/j.gie.2013.07.021. [DOI] [PubMed] [Google Scholar]
- 12.Cataldo P, Ellis CN, Gregorcyk S, Hyman N, Buie WD, Church J, Cohen J, Fleshner P, Kilkenny J, Ko C, et al. Practice parameters for the management of hemorrhoids (revised) Dis Colon Rectum. 2005;48:189–194. doi: 10.1007/s10350-004-0921-4. [DOI] [PubMed] [Google Scholar]
- 13.Haas PA, Haas GP, Schmaltz S, Fox TA. The prevalence of hemorrhoids. Dis Colon Rectum. 1983;26:435–439. doi: 10.1007/BF02556521. [DOI] [PubMed] [Google Scholar]
- 14.Adami B, Eckardt VF, Suermann RB, Karbach U, Ewe K. Bacteremia after proctoscopy and hemorrhoidal injection sclerotherapy. Dis Colon Rectum. 1981;24:373–374. doi: 10.1007/BF02603422. [DOI] [PubMed] [Google Scholar]
- 15.Khoury GA, Lake SP, Lewis MC, Lewis AA. A randomized trial to compare single with multiple phenol injection treatment for haemorrhoids. Br J Surg. 1985;72:741–742. doi: 10.1002/bjs.1800720924. [DOI] [PubMed] [Google Scholar]
- 16.Hachiro Y, Kunimoto M, Abe T, Kitada M, Ebisawa Y. Aluminum potassium sulfate and tannic acid (ALTA) injection as the mainstay of treatment for internal hemorrhoids. Surg Today. 2011;41:806–809. doi: 10.1007/s00595-010-4386-x. [DOI] [PubMed] [Google Scholar]
- 17.Kaidar-Person O, Person B, Wexner SD. Hemorrhoidal disease: A comprehensive review. J Am Coll Surg. 2007;204:102–117. doi: 10.1016/j.jamcollsurg.2006.08.022. [DOI] [PubMed] [Google Scholar]
- 18.Graham-Stewart CW. Injection treatment of haemorrhoids. Br Med J. 1962;1:213–216. doi: 10.1136/bmj.1.5273.213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Santos G, Novell JR, Khoury G, Winslet MC, Lewis AA. Long-term results of large-dose, single-session phenol injection sclerotherapy for hemorrhoids. Dis Colon Rectum. 1993;36:958–961. doi: 10.1007/BF02050633. [DOI] [PubMed] [Google Scholar]
- 20.Tarrant KM, Barclay ML, Frampton CM, Gearry RB. Perianal disease predicts changes in Crohn’s disease phenotype-results of a population-based study of inflammatory bowel disease phenotype. Am J Gastroenterol. 2008;103:3082–3093. doi: 10.1111/j.1572-0241.2008.02212.x. [DOI] [PubMed] [Google Scholar]
- 21.Gralnek IM, Ron-Tal Fisher O, Holub JL, Eisen GM. The role of colonoscopy in evaluating hematochezia: a population-based study in a large consortium of endoscopy practices. Gastrointest Endosc. 2013;77:410–418. doi: 10.1016/j.gie.2012.10.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Selvaggi F, Sciaudone G, Guadagni I, Pellino G. Ileal pouch-anal anastomosis after stapled haemorrhoidopexy for unrecognized ulcerative colitis. Colorectal Dis. 2010;12:e172. doi: 10.1111/j.1463-1318.2009.02003.x. [DOI] [PubMed] [Google Scholar]
- 23.Keighley MR, Allan RN. Current status and influence of operation on perianal Crohn’s disease. Int J Colorectal Dis. 1986;1:104–107. doi: 10.1007/BF01648416. [DOI] [PubMed] [Google Scholar]


