Abstract
Asbestos related pleuropulmonary disease has been emerging health problem for recent years. It can cause variable clinical symptoms and radiological abnormalities. However, there has been no report for their characteristics in subjects who were environmentally exposed to asbestos. We reviewed the CT images of 35 people who were environmentally exposed to asbestos in Chungnam province, Korea. The study result showed high incidence of pleural plaque and pulmonary fibrosis on chest CT (94% and 77%, respectively). The common CT findings of lung parenchymal lesions were as follows: centrilobular opacities (94%), subpleural dot-like or branching opacities (80%), interlobular septal thickening (57%), intralobular interstitial thickening (46%), parenchymal bands (43%) and subpleural curvilinear line (29%). There were no significant differences in the prevalence of pulmonary fibrosis and pleural plaques according to sex, age and duration of exposure. In conclusion, pleural plaque and pulmonary fibrosis are common asbestos-related CT finding in the exposed people. Asbestos related lung parenchymal CT findings in the participants with environmental exposure show similar to those observed in the occupational exposure.
Keywords: Asbestos, Environmental Exposure, Pulmonary Fibrosis, Asbestosis, Plaque, Multidetector Computed Tomography
Graphical Abstract
INTRODUCTION
Asbestosis is defined as diffuse pulmonary interstitial fibrosis caused by the inhalation of excessive amounts of asbestos fibers. The pathogenesis of asbestos-related pleuropulmonary disease is complex and not yet completely understood. Fiber dose, dimension, and chemical composition may influence fibrogenicity and carcinogenicity. Factors related to the host, including pulmonary clearance and immunologic status and exposure to other noxious substances such as cigarette smoking are also important in determining the nature and severity of the reaction to inhaled fibers (1,2,3,4). There have been a number of epidemiological reports about pleural mesothelioma as a result of environmental exposure to asbestos (5,6,7,8). However, there had been only a few reports about asbestosis by environmental exposure because it needs relatively high concentration of asbestos and prolonged exposure, usually over 10 to 20 yr (9).
The midwestern part of the Korean Peninsula (Chungnam province) has many historic asbestos mines. Approximately 71% of asbestos mines in Korea were located in this rural area. Unlike other countries, mines and villages intermingle with each other in this area. These mines had been exploited as asbestos mines for several years since the 1930s. They contain mainly three types of asbestos forms, chrysotile, tremolite and actinolite (10). Most mines have been in disuse since 1945, but mining and processing of asbestos were restarted with increased use of slate roofs by the government 5 Year's Planning of Economic Development (1972-1977). Over 100,000 tons of asbestos were produced annually from 1978 to 1983. Since then, the mines have been in a state of neglect for decades (11). Recently, there had been a report about the increasing mortality and incidence of asbestos-related disease among people residing near the asbestos textile factory (12), so more concern and research should be needed in environmentally asbestos exposure groups. Since 2008 the Ministry of Environment of the Republic of Korea carried out a health impact assessment by asbestos exposure for residents who lived near asbestos mines.
To our knowledge, radiographic and computed tomography (CT) findings of asbestosis were characteristic and have been described in the previous reports. However, there has been no report for their characteristics in a subjects environmentally exposed to asbestos. In this study we evaluate the 35 CT findings of the lung and pleural lesions in people who were environmentally exposed to asbestos. Also we assess the frequency of specific CT findings and the correlation of asbestos-related pleuropulmonary disease with demographic characteristics.
MATERIALS AND METHODS
Participants
From August 2008 to February 2009, the Ministry of Environment carried out epidemiological studies, and health impact assessments for residents from 5 villages (Deokjeong, Yadong, Gwangcheon-eup, Cheongso-myeon and Ocheon-myeon), who lived within 2 km from the Kwangcheon, Joongang, and Daeboseoksan mines. Each participant received a letter describing the study, provided informed consent and completed in-person interviews. Among villagers, posteroanterior chest radiographs were performed for 215 residents (about 50% of all residents in 5 villages) who were over 40 yr old, had more than 1 yr residence history during the mining operation period, and lived within 2 km from 3 historic asbestos mines. CT scans were recommended to 110 (51.2%) participants with an abnormal chest radiograph and/or clinical symptoms (cough, dyspnea, shortness of breath and wheezing), and 95 participants underwent a chest CT scan. We excluded 10 participants due to the following conditions: 1) inadequate CT images (n=6); and 2) an incomplete questionnaire (n=4). Among 85 participants, 50 (58.8%) had occupation history in asbestos mining, and the remaining 35 (41.2%) were only residents. Finally, a total of 35 participants (17 men, 18 women; mean age, 68.5 yr; age range, 48-82 yr) with environmental exposure to asbestos were included in our study (Fig. 1).
Fig. 1. Flow chart of participant selection.
Questionnaire
Questions included 1) residence: one's past and present address, the length of residence around a mine and in a slate house, 2) occupation history: any work in asbestos mines and industries, or other occupational asbestos exposure, 3) past medical history: especially related to respiratory diseases, and 4) smoking history.
CT scanning protocol
CT images were acquired using a 64-channel multidetector CT scanner (Sensation 64; Siemens Medical Solutions, Erlangen Germany). Participants were examined in the supine position with full inspiration without administration of intravenous contrast. To remove the gravity-dependent attenuation, a prone position scan with full inspiration was also performed. For the evaluation of lung parenchyma, thin sections (1 mm thickness) were reconstructed at 10 mm intervals from the apex to the diaphragm. And for the evaluation of pleural and other lung parenchymal lesions, scanning and image reconstruction were performed with a section thickness of 5 mm and a reconstruction interval of 5 mm. All images were obtained at window levels appropriate for lung parenchyma (window width, 1,500 HU; window level, -700 HU) and mediastinum (window width, 440 HU; window level, 40-50 HU).
Evaluation of chest radiographs and CT scan
Chest radiographs were evaluated by two board-certified radiologists (with over 10 yr of experience in thoracic radiology), who were blind to the participant information. Reports were classified by the 2000 International Labor Office (ILO) International Classification of Radiographs of Pneumoconioses. Parenchymal abnormalities, pleural abnormalities were evaluated.
Chest CT scans were taken in people with an abnormal chest radiograph and/or clinical symptoms. Serial chest CT scans of participants were interpreted by three board-certified radiologists (with over 10 yr of experience in thoracic radiology) with consensus. Pleural abnormalities were described separately as noncalcified and calcified pleural plaques, and the other pleural abnormalities, such as blunting of the costophrenic angle, pleural effusion and pleural mass were included. Parenchymal abnormalities were reviewed according to the following CT findings: subpleural dot-like or branching opacities, subpleural curvilinear line (SCL), parenchymal bands (PB), intralobular interstitial thickening (IIT), interlobular septal thickening (IST), honeycombing (HC), ground-glass opacities (GGO), emphysema, dilatation of intralobular bronchiole, bronchiolectasis within consolidation, traction bronchiectasis (TB), consolidation and centrilobular opacities (CLOs) and a lung nodule or mass. According to the report of Akira et al. (13,14) subpleural dot-like or branching opacities, SCL, PB, IIT, TB and HC correlated with fibrosis and were significantly more common in patients with asbestosis. Thus we considered it to be lung fibrosis when more than two aforementioned findings were found for more than two sections of chest CT scans. The axial anatomic distribution was analyzed into three zones (upper, middle and lower): the upper lung zone was above the level of the aortic arch, the middle lung zone was between the level of the aortic arch and the level of the right inferior pulmonary vein, and the lower lung zone was below the right inferior pulmonary vein.
Statistical analysis
All statistical analyses were performed using SPSS software (IBM SPSS Statistics ver. 19.0; IBM, Inc., Amonk, NY, USA). We compared the frequencies of pleural plaque and lung fibrosis in the different demographic characteristics using chi-square test. Linear-by-linear association was used to test whether there was a linear association in advanced age and duration of exposure with the prevalence of pleural plaque and lung fibrosis. In all statistical analyses, values of P<0.05 were considered statistically significant.
Ethics statement
This study was approved by the institutional review board of Dongguk University Ilsan Hospital (IRB No. 2011-102) and informed consent was obtained from all participants.
RESULTS
The prevalence of lung fibrosis and pleural plaque depending on demographic characteristics is shown in Table 1. On CT scans, pleural plaque was observed in 94.3% (33/35) of participants. There was no significant difference in prevalence of pleural plaque between male (n=17, 100%) and female (n=16, 88.9%) participants (P=0.163). The prevalence of pleural plaque showed no difference depending on the age groups (P=0.623) nor duration of exposure (P=0.251). Pleural plaque was detected in 15 (100%) of the 15 smokers and in 18 (90%) of the 20 nonsmokers (P=0.214). One patient showed pleural mass which is suspicious of mesothelioma, however pathologic confirmation cannot be accessed. Blunting of the costophrenic angle was observed in 5 participants and none of the participants showed pleural effusion.
Table 1. The prevalence of pleural plaque and lung fibrosis according to sex, smoking history, age groups and duration of exposure.
| Parameters | Lung fibrosis (%) | P value* | Pleural plaque (%) | P value* |
|---|---|---|---|---|
| Total (n = 35) | 27 (77.1) | 33 (94.3) | ||
| Sex | ||||
| Male (n = 17) | 14 (82.4) | 17 (100) | ||
| Female (n = 18) | 13 (72.2) | 0.482 | 16 (88.9) | 0.163 |
| Smoking | ||||
| Yes (n = 15) | 14 (93.3) | 15 (100) | ||
| No (n = 20) | 13 (65) | 0.052 | 18 (90) | 0.214 |
| Age (yr) | ||||
| 40-49 (n = 2) | 0 (0) | 2 (100) | ||
| 50-59 (n = 4) | 3 (75) | 4 (100) | ||
| 60-69 (n = 11) | 10 (90.9) | 9 (81.8) | ||
| 70-79 (n = 17) | 13 (76.5) | 17 (100) | ||
| > 80 (n = 1) | 1 (100) | 0.129† | 1 (100) | 0.623† |
| Duration of exposure (yr) | ||||
| 10-19 (n = 1) | 1 (100) | 1 (100) | ||
| 20-29 (n = 3) | 2 (66.7) | 3 (100) | ||
| 30-39 (n = 8) | 3 (37.5) | 6 (75) | ||
| 40-49 (n = 7) | 6 (85.7) | 7 (100) | ||
| 50-59 (n = 8) | 8 (100) | 8 (100) | ||
| 60-69 (n = 8) | 7 (87.5) | 0.084† | 8 (100) | 0.251† |
Data are presented as number (%). *Comparison was done by chi-square test; †By Linear-by-linear association test.
On chest CT scans, 77.1% (27/35) participants showed lung fibrosis. There were no significant differences in the prevalence of lung fibrosis in male (n=14, 82.4%) and female participants (n=13, 72.2%) (P=0.482). The prevalence of lung fibrosis showed no difference depending on the age groups (P=0.129) nor duration of exposure (P=0.084). Lung fibrosis was more frequently seen in the smokers (n=14, 93.3%) than in the nonsmokers (n=13, 65%) (P=0.052), however, it was not statistically significant. Lung nodule was observed in 8 participants and none of the participants showed lung mass.
Seven participants showed pleural plaque without lung fibrosis while only one participant showed lung fibrosis without pleural plaque. Both pleural plaque and lung fibrosis were found in 26 participants. A female participant showed no evidence of pleural plaque or pulmonary fibrosis.
Parenchymal abnormalities were as follows: CLOs (94.3%), subpleural dot-like or branching opacities (80%), IST (57.1%), IIT (45.7%), PB (42.9%), SCL (28.6%), emphysema (17.1%), GGO (14.3%), TB (11.4%), dilatation of intralobular bronchiole (8.6%) and HC (2.9%) (Table 2). CLOs were evenly distributed in the upper, middle and lower lung zones, and emphysema was more common in upper lung zones (Fig. 2 and 3). The other findings were more frequently seen in the lower lung zones. PB and TB were more common findings in older paticipants (P value, 0.021 and 0.033, respectively) and dilatation of intralobular bronchiole were more common findings in longer exposures (P=0.039) (Table 3).
Table 2. Comparison of prevalence of CT findings.
| Findings | No. (%) of cases | ||
|---|---|---|---|
| Total n=35 | Akira et al. (13), n=7 | Aberle et al. (15), n=29 | |
| Centrilobular opacities | 33 (94.3) | ||
| Subpleural dot-like or branching opacities | 28 (80) | 7 (100) | 59%/14%* |
| Interlobular septal thickening | 20 (57.1) | 7 (100) | 97%/86%* |
| Intralobular interstitial thickening | 16 (45.7) | 7 (100) | |
| Parenchymal bands | 15 (42.9) | 5 (71) | 66% |
| Subpleural curvilinear line | 10 (28.6) | 4 (57) | 28%/10%* |
| Emphysema | 6 (17.1) | ||
| GGO | 5 (14.3) | 4 (57) | |
| Traction bronchiectasis | 4 (11.4) | 4 (57) | |
| Dilatation of intralobular bronchiole | 3 (8.6) | ||
| Honeycombing | 1 (2.9) | 2 (29) | 17% |
| Bronchiolectasis within consolidation | 0 (0) | ||
| Consolidation | 0 (0) | ||
Data are presented as number (%).*Supine/prone, respectively. GGO, ground-glass opacities.
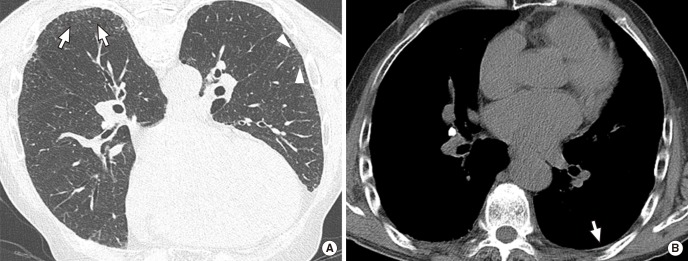
Fig. 2. CT images of a 72-yr-old man with 72 yr residence history. (A) Prone positioned HRCT scan shows subpleural dot-like or branching opacities (arrows), and intralobular interstitial thickening (arrowheads). (B) Noncalcified thin pleural plaque is also noted in left lower posterior portion (arrow).
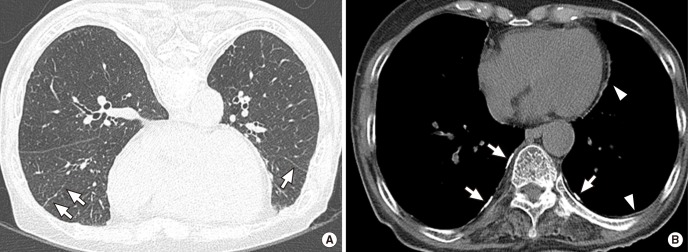
Fig. 3. CT images of a 77-yr-old woman with 69 yr residence history. (A) Prone positioned HRCT scan shows parenchymal band in right mid lobe and left lower lobe (arrows). (B) Multifocal bilateral discontinuous calcified (arrows) and noncalcified (arrowheads) pleural plaques are also noted in mediastinal and bilateral lower posterior areas.
Table 3. P values of CT findings according to sex, smoking history, age groups and duration of exposure.
| Findings | Sex* | Smoking* | Age† | Duration of exposure† |
|---|---|---|---|---|
| Centrilobular opacities | 1.0 | 1.0 | 0.284 | 0.470 |
| Subpleural dot-like or branching opacities | 0.691 | 0.672 | 0.147 | 0.328 |
| Interlobular septal thickening | 0.625 | 0.767 | 0.530 | 0.158 |
| Intralobular interstitial thickening | 0.404 | 0.142 | 0.148 | 0.466 |
| Parenchymal bands | 0.625 | 0.278 | 0.021 | 0.060 |
| Subpleural curvilinear line | 1.0 | 0.266 | 0.456 | 0.514 |
| Emphysema | 0.088 | 0.064 | 0.956 | 0.182 |
| GGO | 1.0 | 1.0 | 0.208 | 0.965 |
| Traction bronchiectasis | 0.338 | 0.292 | 0.033 | 0.082 |
| Dilatation of intralobular bronchiole | 0.603 | 0.565 | 0.183 | 0.039 |
| Honeycombing | 0.486 | 0.429 | 0.455 | 0.248 |
| Bronchiolectasis within consolidation | NA | NA | NA | NA |
| Consolidation | NA | NA | NA | NA |
Data are presented as number (%).*Comparison was done by chi-square test; †By linear-by-linear association test. GGO, ground-glass opacities; NA, not applicable because of negative CT findings.
DISCUSSION
Our study show high prevalence of lung fibrosis and plaque among village people with environmental asbestos exposure. Unlike mesothelioma or lung cancer, asbestosis from environmental exposure is rare because asbestosis requires relatively high concentrations and long latency of asbestos exposure. The high prevalence of asbestos related pleural and pulmonary abnormality in our study may be explained by three reasons. First, most villages are located close to mines (within 2 km from the mines) and mines intermingled with villages. So people had been exposed to an atmosphere contaminated with asbestos every day. Second, participants had more than 1 yr residence history near a mine during its operating period, and considering that most mines were in disuse in late 1980s, the mean residence time during operation period was about 30 yr. It means they had been exposed to high concentration of asbestos during their daily activities. Last, over 90% of participants in our study had over 50 yr residence history, which implies they had been exposed to asbestos for a long time.
On 35 chest CT scans, pleural plaques were found in 94% (n=33) and lung fibrosis were found in 77% (n=27). A high incidence prevalence of lung fibrosis on CT scans in our study may be explained by some reasons. First, a CT has the superior ability to detect lung fibrosis. Many studies showed that computed tomography is more sensitive in detecting parenchymal abnormalities in an asbestos exposed subject (15,16,17,18,19,20,21,22,23). Aberle et al. (16) reported that for 45 subjects with a clinical diagnosis of asbestosis, 35 (78%) showed abnormal chest radiographs while 45 (100%) CT findings suggestive of asbestosis. Staples et al. (18) demonstrated that 57 (34%) of 169 asbestos-exposed subjects for whom asbestosis was highly suspected by CT had normal chest radiographs. Second, lung fibrosis is underestimated on chest radiograph due to following reasons. There had been no large scale research of asbestosis in Korea before, so radiologists were not familiar with findings of asbestosis. Furthermore Korea is still an endemic area with pulmonary tuberculosis which can hide lung fibrosis.
Our study showed insignificant correlation of lung fibrosis according to smoking history. Cigarette smoking has been suggested as an additional factor in the pathogenesis of asbestosis. Several studies revealed that smoking increases the prevalence of small parenchymal opacities and/or the progression rate of asbestosis among asbestos-exposed workers (24,25,26). On the other hand, Aberle et al. (16) showed no relationship between HRCT probability scores for lung fibrosis with a smoking history. However quantification of pack-year smoking history is more relevant than simple smoking status. Also, started smoking during mining operation and after closure might have different effect to asbestosis occurrence, which was omitted in our study. Considering only a small number of participants were included in our study, a future study based on a larger population with qualified information could show significant correlation between lung fibrosis with a smoking.
Subpleural dot-like or branching opacities, IST, IIT, PB, SCL were common findings in our study, which was similar to Akira et al. (13). Histologically, thickened intralobular lines, pleural-based opacities, PBs, SCLs, TB and HC correlated with fibrosis. However, the IST and GGO could be due to fibrosis or edema (13). On the other hand, IST and PB were the more frequent CT findings in Aberle et al. (15) compared with our study (86% vs. 57.1% and 66% vs. 42.9%, respectively). Although the subjects of these studies have been occupationally exposed to asbestos, our study shows similar CT findings in the participants with environmentally exposed to asbestos. CLOs were the most common CT findings (94.3%) in our study which we presumed related to nonspecific dust exposure.
Only one participant was suspicious for malignant mesothelioma in our study. But pathologic confirmation was not performed due to a patient's refusal. According to the Korea Central Cancer Registry report, there are 9 reports of malignant mesothelioma in village and city of these area (Hongseong-gun, 5; Boryeong-si, 4). Incidence of malignant mesothelioma is also higher as compared to other regions of Korea (27). Our study included relatively small numbers of environmental asbestos exposures, so a large population based retrospective cohort study would be needed to evaluate the malignant mesothelioma morbidity and mortality in these areas.
Our study had some limitations. First, we did not evaluate the chest radiographs and CT of all residents in the village, instead we included participants with relatively long and heavy exposure which could lead to selection bias. Second, histologic confirmation and a functional assessment were not done in the subjects. But according to a modified asbestosis guidelines published by the American Thoracic Society (ATS) in 2004 (28), our study population can be classified as having asbestos-related disease. Third, some report showed household contamination with asbestos from industrial sources may result in radiologic changes (29). However, we could not exclude the individuals lived with asbestos workers due to lack of history.
In conclusion, our study shows high frequency of pleural plaque and pulmonary fibrosis in subjects with environmentally exposed to asbestos. CT findings of lung parenchymal lesions were more common in the following order: CLO, subpleural dot-like or branching opacities, IST, IIT, PB and SCL. Asbestos related lung parenchymal CT findings in the participants with environmental exposure shows similar to those observed in the occupational exposure.
Footnotes
Funding: This research was supported by the Korea Ministry of Environment under "The Environmental Health Action Program".
DISCLOSURE: The authors have no potential conflicts of interest to disclose.
AUTHOR CONTRIBUTION: Study design: Kim JS. Data collection: Lee EK, Kim JS. Data review and analysis: Lee EK, Kim JS, Kim YK, Park JS. Manuscript writing: Lee EK, Kim JS. Approval of manuscript: all authors.
References
- 1.Silva CI, Müller NL. Asbestos-related disease. In: Müller NL, Silva CI, editors. Imaging of the chest. Philadelphia: Elsevier Saunders; 2008. pp. 1140–1166. [Google Scholar]
- 2.Fraser RS, Colman N, Müller NL, Paré PD. Pulmonary disease caused by inhaled inorganic dust. In: Fraser RS, Colman N, Müller NL, Paré PD, editors. Synopsis of diseases of the chest. 3rd ed. Philadelphia: Elsevier Saunders; 2005. pp. 714–743. [Google Scholar]
- 3.Donaldson K, Brown RC, Brown GM. New perspectives on basic mechanisms in lung disease. 5. Respirable industrial fibres: mechanisms of pathogenicity. Thorax. 1993;48:390–395. doi: 10.1136/thx.48.4.390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lippmann M. Effects of fiber characteristics on lung deposition, retention, and disease. Environ Health Perspect. 1990;88:311–317. doi: 10.1289/ehp.9088311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Goldberg P, Goldberg M, Marne MJ, Hirsch A, Tredaniel J. Incidence of pleural mesothelioma in New Caledonia: a 10-year survey (1978-1987) Arch Environ Health. 1991;46:306–309. doi: 10.1080/00039896.1991.9934393. [DOI] [PubMed] [Google Scholar]
- 6.Metintas S, Metintas M, Ucgun I, Oner U. Malignant mesothelioma due to environmental exposure to asbestos: follow-up of a Turkish cohort living in a rural area. Chest. 2002;122:2224–2229. doi: 10.1378/chest.122.6.2224. [DOI] [PubMed] [Google Scholar]
- 7.Senyiğit A, Bayram H, Babayiğit C, Topçu F, Nazaroğlu H, Bilici A, Leblebici IH. Malignant pleural mesothelioma caused by environmental exposure to asbestos in the Southeast of Turkey: CT findings in 117 patients. Respiration. 2000;67:615–622. doi: 10.1159/000056290. [DOI] [PubMed] [Google Scholar]
- 8.Ökten F, Köksal D, Önal M, Özcan A, Şimşek C, Ertürk H. Computed tomography findings in 66 patients with malignant pleural mesothelioma due to environmental exposure to asbestos. Clin Imaging. 2006;30:177–180. doi: 10.1016/j.clinimag.2005.12.027. [DOI] [PubMed] [Google Scholar]
- 9.Asbestos, asbestosis, and cancer: the Helsinki criteria for diagnosis and attribution. Scand J Work Environ Health. 1997;23:311–316. [PubMed] [Google Scholar]
- 10.Song SH, Hwang JH, Hwang BG, Kim HW. Occurrence types and mineralogical characteristics of asbestos for the Kwangcheon area, Chungnam. J Korean Soc Occup Environ Hyg. 2008;18:271–281. [Google Scholar]
- 11.Ahn YS, Kim HR. Asbestosis epidemics caused by non-occupational neighborhood exposure. J Korean Med Assoc. 2009;52:472–481. [Google Scholar]
- 12.Kang DM, Gu DC, Kim KH. Asbestos-related diseases among asbestos textile factory workers and residents around the factory. J Korean Med Assoc. 2009;52:482–488. [Google Scholar]
- 13.Akira M, Yamamoto S, Yokoyama K, Kita N, Morinaga K, Higashihara T, Kozuka T. Asbestosis: high-resolution CT-pathologic correlation. Radiology. 1990;176:389–394. doi: 10.1148/radiology.176.2.2367652. [DOI] [PubMed] [Google Scholar]
- 14.Akira M, Yamamoto S, Inoue Y, Sakatani M. High-resolution CT of asbestosis and idiopathic pulmonary fibrosis. AJR Am J Roentgenol. 2003;181:163–169. doi: 10.2214/ajr.181.1.1810163. [DOI] [PubMed] [Google Scholar]
- 15.Aberle DR, Gamsu G, Ray CS, Feuerstein IM. Asbestos-related pleural and parenchymal fibrosis: detection with high-resolution CT. Radiology. 1988;166:729–734. doi: 10.1148/radiology.166.3.3340770. [DOI] [PubMed] [Google Scholar]
- 16.Aberle DR, Gamsu G, Ray CS. High-resolution CT of benign asbestos-related diseases: clinical and radiographic correlation. AJR Am J Roentgenol. 1988;151:883–891. doi: 10.2214/ajr.151.5.883. [DOI] [PubMed] [Google Scholar]
- 17.Friedman AC, Fiel SB, Fisher MS, Radecki PD, Lev-Toaff AS, Caroline DF. Asbestos-related pleural disease and asbestosis: a comparison of CT and chest radiography. AJR Am J Roentgenol. 1988;150:269–275. doi: 10.2214/ajr.150.2.269. [DOI] [PubMed] [Google Scholar]
- 18.Staples CA, Gamsu G, Ray CS, Webb WR. High resolution computed tomography and lung function in asbestos-exposed workers with normal chest radiographs. Am Rev Respir Dis. 1989;139:1502–1508. doi: 10.1164/ajrccm/139.6.1502. [DOI] [PubMed] [Google Scholar]
- 19.Akira M, Yokoyama K, Yamamoto S, Higashihara T, Morinaga K, Kita N, Morimoto S, Ikezoe J, Kozuka T. Early asbestosis: evaluation with high-resolution CT. Radiology. 1991;178:409–416. doi: 10.1148/radiology.178.2.1987601. [DOI] [PubMed] [Google Scholar]
- 20.Oksa P, Suoranta H, Koskinen H, Zitting A, Nordman H. High-resolution computed tomography in the early detection of asbestosis. Int Arch Occup Environ Health. 1994;65:299–304. doi: 10.1007/BF00405693. [DOI] [PubMed] [Google Scholar]
- 21.Lynch DA. CT for asbestosis: value and limitations. AJR Am J Roentgenol. 1995;164:69–71. doi: 10.2214/ajr.164.1.7998571. [DOI] [PubMed] [Google Scholar]
- 22.Harkin TJ, McGuinness G, Goldring R, Cohen H, Parker JE, Crane M, Naidich DP, Rom WN. Differentiation of the ILO boundary chest roentgenograph (0/1 to 1/0) in asbestosis by high-resolution computed tomography scan, alveolitis, and respiratory impairment. J Occup Environ Med. 1996;38:46–52. doi: 10.1097/00043764-199601000-00016. [DOI] [PubMed] [Google Scholar]
- 23.Soulat JM, Lauque D, Esquirol Y, Déprés M, Giron J, Claudel R, Carles P. High-resolution computed tomography abnormalities in ex-insulators annually exposed to asbestos dust. Am J Ind Med. 1999;36:593–601. doi: 10.1002/(sici)1097-0274(199912)36:6<593::aid-ajim1>3.0.co;2-z. [DOI] [PubMed] [Google Scholar]
- 24.Weiss W. Cigarette smoke, asbestos, and small irregular opacities. Am Rev Respir Dis. 1984;130:293–301. doi: 10.1164/arrd.1984.130.2.293. [DOI] [PubMed] [Google Scholar]
- 25.Blanc PD, Golden JA, Gamsu G, Aberle DR, Gold WM. Asbestos exposure-cigarette smoking interactions among shipyard workers. JAMA. 1988;259:370–373. doi: 10.1001/jama.259.3.370. [DOI] [PubMed] [Google Scholar]
- 26.Barnhart S, Thornquist M, Omenn GS, Goodman G, Feigl P, Rosenstock L. The degree of roentgenographic parenchymal opacities attributable to smoking among asbestos-exposed subjects. Am Rev Respir Dis. 1990;141:1102–1106. doi: 10.1164/ajrccm/141.5_Pt_1.1102. [DOI] [PubMed] [Google Scholar]
- 27.Kim I, Kim EA, Kim JY. Compensation for occupational cancer. J Korean Med Sci. 2014;29:S40–S46. doi: 10.3346/jkms.2014.29.S.S40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.American Thoracic Society. Diagnosis and initial management of nonmalignant diseases related to asbestos. Am J Respir Crit Care Med. 2004;170:691–715. doi: 10.1164/rccm.200310-1436ST. [DOI] [PubMed] [Google Scholar]
- 29.Anderson HA, Lilis R, Daum SM, Fischbein AS, Selikoff IJ. Household-contact asbestos neoplastic risk. Ann N Y Acad Sci. 1976;271:311–323. doi: 10.1111/j.1749-6632.1976.tb23127.x. [DOI] [PubMed] [Google Scholar]