Abstract
OBJECTIVE
This study aims to present an updated clinical picture of the infected cardiac myxoma. Revankar & Clark made a systematic review of infected cardiac myxoma based on the literature before 1998. Since then, there has not been any updated information describing its recent changing trends.
METHODS
A comprehensive literature search of infected cardiac myxoma was conducted on MEDLINE, Highwire Press and Google between 1998 and 2014.
RESULTS
In comparison with Revankar & Clark's series, the present series disclosed a significantly decreased overall mortality. It is believed that refinement of the prompt diagnosis and timely management (use of sensitive antibiotics and surgical resection of the infected myxoma) have resulted in better outcomes of such patients.
CONCLUSION
The present series of infected cardiac myxoma illustrated some aggravated clinical manifestations (relative more occasions of high-grade fever, multiple embolic events and the presence of refractory microorganisms), which should draw enough attention to careful diagnosis and treatment. In general, the prognosis of infected cardiac myxoma is relatively benign and the long-term survival is always promising.
Keywords: Embolism, Infection, Myxoma
INTRODUCTION
Cardiac myxomas are rare. The classical clinical manifestations of cardiac myxomas can be a triad with constitutional, obstructive and embolic symptoms[1]. The infected cardiac myxoma is a very rare condition with only sporadic cases reported in the literature. In 1998, Revankar & Clark[2] presented a complete literature collection of infected cardiac myxoma with a total of 40 cases, with detailed descriptions of the clinical features of this rare condition. They found no clear distinction between infected and uninfected cardiac myxomas, however, the infected myxomas are associated with more febrile symptoms and a higher risk of embolic events. In order to highlight the recent trends of infected cardiac myxoma, an updated review is made based on a renewed literature collection.
METHODS
A comprehensive literature search was conducted on MEDLINE, Highwire Press and Google between 1998 and 2014. The search terms included "myxoma", "heart", "heart valve", "endocarditis", "infected", "bacteremia", "blood culture" and "sepsis". Data were extracted from the text, figures and tables, with details of the study population, demographics, onset symptom, duration of disease, risk factor, complication, microorganism, antibiotic use and surgical treatment, timing of surgical operation, follow-up duration and main outcomes including survival, postoperative complication, requirement of further surgical procedure and mortality.
Reference values of white blood cell count, hemoglobin, erythrocyte sedimentation rate and C-reaction protein were 4.0-10.0×109/L, >12 g/dL (>11 g/dL for females), <20 mm/h and 0-1 mg/dL, respectively.
Quantitative data were presented as mean±standard deviation with range and median values, and intergroup differences were compared using the unpaired t test. Frequencies were compared using Fisher's exact test. Results with P<0.05 were considered statistically significant.
Patients' information
A total of 39 reports[3-41] in terms of infected cardiac myxoma were collected and these involved 39 patients. Their information was listed in Table 1. According to the diagnostic criteria of infected cardiac myxoma, 34 (87.2%) were definite[3-36], 4 (10.3%) were probable[37-40] and 1 (2.6%) was possible[41], with a compatible distribution of the three-level infected cardiac myxomas reported by Revankar & Clark[2]. The locations of the infected cardiac myxomas were left atrium 27 (69.2%), right atrium 5 (12.8%), mitral valve 5 (12.8%), right ventricle 1 (2.6%) and unstated 1 (2.6%), also similar to the percentages of the locations reported by Revankar & Clark[2]. The only difference was a lack of biatrial myxomas in the present series but with multiple left atrial myxomas instead. There were 23 (62.2%) males and 14 (37.8%) females (x2=4.4, P=0.062) with a male-to-female ratio of 1.64, while gender of 2 patients was not given. Their ages were 49.2±15.7 (range, 12-74; median,52) years (n=37). No age difference was found between male and female patients (50.7±11.4 years vs. 46.9±21.3 years, P=0.4786).
Table 1.
Summary of 39 patients with infected cardiac myxoma
| Serial No | Year | Author | Sex | Age | Microorganism | Diagnostic means | Location of myxoma | Operation | Outcome |
|---|---|---|---|---|---|---|---|---|---|
| Definite | |||||||||
| 1 | 2011 | Adamira | F | 58 | Staphylococcus & Enterococcus faecalis | TTE,TEE | LAx2 | Yes | Survived |
| 2 | 2006 | Awab | M | 58 | Histoplasma capsulatum | TTE | LA | Yes | Survived |
| 3 | 2011 | Belgi Yildirim | M | 47 | Streptococcus viridans | TTE | LA | Yes | Survived |
| 4 | 2010 | Bhanot | M | 54 | Staphylococcus lugdunensis | CT, TEE | LA | Yes | Survived |
| 5 | 2001 | Brazill | M | 52 | Streptococci viridans | TTE, TEE | LA | Yes | Survived |
| 6 | 2007 | Chan | M | 44 | Streptococcus viridans | TTE | RV | Yes | Survived |
| 7 | 2011 | Chang | M | 49 | Streptococcus bovis | TTE, TEE | LA | Yes | Survived |
| 8 | 2001 | Dekkers | M | 40 | Streptococcus mutans | TTE, TEE | LA | Yes | Survived |
| 9 | 2008 | Falasca | F | 63 | Staphylococcus spp | TTE | LA | Yes | Survived |
| 10 | 2013 | Furukawa | M | 62 | Streptococcus agalactiae | TTE, TEE | LA | Yes | Survived |
| 11 | 2005 | García-Quintana | F | 58 | Streptococcus oralis | TTE | LA | Yes | Survived |
| 12 | 2004 | Gregory | M | 43 | MSSA | TTE (-), TEE (+) | LA | Yes | Survived |
| 13 | 2007 | Guler | F | 12 | MSSA | TTE | MV (AML) | Yes | Survived |
| 14 | 2008 | Janion | M | 67 | MRSA | TTE | LA | Yes | Survived |
| 15 | 2005 | Juang | M | 42 | G-positive cocci | TEE | LA | Yes | Survived |
| 16 | 2004 | Karachalios | M | 55 | Streptococcus viridans | TTE | LA | Yes | Survived |
| 17 | 2007 | Leone | F | 74 | Enterococcus faecalis | TTE | LA | Yes | Survived |
| 18 | 2013 | Nagata | M | 66 | Streptococcus mitis | TEE | LA | Yes | Survived |
| 19 | 2001 | Oyama | M | 57 | MRSA | TTE | LA | Yes | Survived |
| 20 | 2004 | Parks | M | 36 | Staphylococcus aureus (oxacillin-resistant) (ORSA) | TTE | RA | Yes | Survived |
| 21 | 2002 | Prince | F | 12 | Streptococcus viridans | TTE | LA | Yes | Survived |
| 22 | 2001 | Puvaneswary | M | 50 | Streptococcus viridans & Streptococcus salivarius | TTE (-), TEE (+) | RA | Yes | Survived |
| 23 | 2006 | Quigley | M | 55 | Streptococcus mutans | TEE | LA | Yes | Survived |
| 24 | 2005 | Riad | M | 36 | MRSA | TTE | RA | Yes | Survived |
| 25 | 2010 | Sasaki | M | 69 | Klebsiella pneumoniae | TEE | LA | Yes | Survived |
| 26 | 2010 | Surovcík | F | 59 | Enterococcus faecalis | TTE, TEE, CT | LA | Yes | Survived |
| 27 | 2002 | Tanaka | F | 70 | Enterococcus faecalis | TEE | LA | Yes | Survived |
| 28 | 1999 | Toda | M | 20 | Bacterial colonies | TTE | MV (PML, posterior commissure) | Yes | Survived |
| 29 | 2009 | Trimeche | F | 46 | MSSA | TTE, TEE | LA | NG | Died |
| 30 | 2002 | Uchino | F | 47 | Streptococcus bovis | TTE | LA | NG | Survived |
| 31 | 2006 | Veitch | F | 35 | Staphylococcus aureus | TTE, TEE | MV (AML) | Yes | Survived |
| 32 | 2011 | Yoshioka | M | 52 | MSSA | TTE | LA | Yes | Survived |
| 33 | 2006 | Zechini | M | 55 | Catalase-negative microorganisms (G-positive cocci by staining) | TTE, TEE | LA | NG | Survived |
| 34 | 2013 | Zwinkels | ? | Streptococcus (Group C) | TTE, cardiac CT angiogram | RA | Yes | Survived | |
| Probable | |||||||||
| 35 | 2007 | Bernstein | F | 60 | Streptococcus mitis | TEE | MV (PML) | Yes | Survived |
| 36 | 2005 | Liu | F | 12 | Neisseria lactamica | TTE | MV (AML) | Yes | Survived |
| 37 | 1998 | Marshall | F | 50 | Actinobacillus actinomycetemcomitans | TTE | LA | Yes | Survived |
| 38 | 2001 | Pinede | ? | Streptococcus viridans | ? | ? | Yes | Survived | |
| Possible | |||||||||
| 39 | 2005 | Despott | M | 57 | Klebsiella pneumoniae | TTE | RA | No | Died of right lung abscess |
AML=anterior mitral leaflet; CT=computed tomography; F=female; LA=left atrium; M=male; MRSA=methicillin-resistant Staphylococcus aureus; MSSA=methicillin-sensitive Staphylococcus aureus; MV=mitral valve; NG=not given; PML= posterior mitral leaflet; RA=right atrium; TEE=transesophageal echocardiography; TTE= transthoracic echocardiography
Risk factors
Recent dental procedures, recent infections, previous invasive procedures and immunocompromised conditions were the risk factors that led to a cardiac myxoma infected. No predominance was noted between the above risk factors. No significant difference was found in each risk factor between the present series and the series of Revankar & Clark[2] (Table 2).
Table 2.
Risk factors.
| Risk factor | Present | Revankar & Clark's | x2 | P value |
|---|---|---|---|---|
| Recent dental problem | 3 (7.7) | 9 (22) | 3.4 | 0.115 |
| Dental surgery | 1 | |||
| Reconstructive dental procedures; coronary catheterization and angioplasty 2 years earlier | 1 | |||
| Dental decay | 1 | |||
| Recent infection | 4 (10.3) | 4 (10) | 0.0 | 1.000 |
| Achilles tendon infection | 2 | |||
| Urinary tract infection | 1 | |||
| Cellulitis, web-space abscess of hand | 1 | |||
| Invasive procedure | 4 (10.3) | 2 (5) | 0.8 | 0.432 |
| Umbilical hernia repair | 1 | |||
| Amputation above knee for intractable osteomyelitis | 1 | |||
| Multiple surgery, closed trauma of the left knee | 1 | |||
| Acupuncture for weight reduction | 1 | |||
| Immunocompromised condition | 5 (12.8) | 3 (7.5) | 0.6 | 0.481 |
| Intravenous drug use, hepatitis C infection | 2 | |||
| Diabetes mellitus | 1 | |||
| Breast cancer (surgery, radiotherapy, chemotherapy), pharyngitis | 1 | |||
| Traveled to Mexico 3 months before, patent fossa ovalis | 1 | |||
| Total | 16 (41.0) | 18 (45) | 0.1 | 0.821 |
Clinical features
The onset symptoms and signs of the 37 patients (2 patients' symptoms were not reported) with infected cardiac myxoma were listed in Table 3. Fever was the most common symptom accounting for 97.3% of all cases. The fever nature was persistent in 2 (5.6%), recurrent in 1 (2.8%), intermittent in 1 (2.8%) and with a fever of unknown origin in 3 (8.3%) patients. In general, constitutional symptoms were more frequent than obstructive or neurological symptoms. The duration of symptoms before admission was 1.6±1.7 (range, 0.1-6; median, 1) months (n=21).
Table 3.
Onset symptoms and signs of the patients with infected cardiac myxoma
| Feature | Present | Revankar & Clark's | x2 | P value |
|---|---|---|---|---|
| Symptom | ||||
| Fever | 36 (97.3) | 37 (92) | 0.9 | 0.616 |
| Embolic events | 10 (27.0) | 5 (12) | 2.6 | 0.151 |
| Weight loss | 9 (24.3) | 13 (32) | 0.6 | 0.460 |
| Dyspnea | 8 (21.6) | 3 (8) | 3.1 | 0.106 |
| Fatigue | 8 (21.6) | 11 (28) | 0.4 | 0.605 |
| Neurologic symptoms | 7 (18.9) | 8 (20) | 0.0 | 1.000 |
| Malaise | 6 (16.2) | 11 (28) | 1.4 | 0.279 |
| Rigor/shivers/chills | 6 (16.2) | 11 (28) | 1.4 | 0.279 |
| Night sweat | 5 (13.5) | 11 (28) | 2.3 | 0.165 |
| Weakness | 3 (8.1) | 5 (12) | 0.4 | 0.713 |
| Abdominal pain | 3 (8.1) | 3 (8) | 0.0 | 1.000 |
| Anorexia | 3 (8.1) | 3.4 | 0.106 | |
| Edemas | 3 (8.1) | 3.4 | 0.106 | |
| Chest pain/distress | 2 (5.4) | 2.2 | 0.228 | |
| Cough | 2 (5.4) | 4 (10) | 0.6 | 0.676 |
| Nausea/vomiting | 2 (5.4) | 1 (2) | 0.4 | 0.605 |
| Headache | 1 (2.7) | 4 (10) | 1.7 | 0.360 |
| Arm pain | 1 (2.7) | 1.1 | 0.481 | |
| Arthralgia | 1 (2.7) | 6 (15) | 3.5 | 0.110 |
| Diarrhea | 1 (2.7) | 3 (8) | 0.9 | 0.616 |
| Hemoptysis | 1 (2.7) | 1.1 | 0.481 | |
| Jaundice | 1 (2.7) | 1.1 | 0.481 | |
| Lethargy | 1 (2.7) | 1.1 | 0.481 | |
| Back pain | 1 (2.7) | 1.1 | 0.481 | |
| Myalgias | 1 (2.7) | 1.1 | 0.481 | |
| Palpitation | 1 (2.7) | 1.1 | 0.481 | |
| Septic shock | 1 (2.7) | 1.1 | 0.481 | |
| Sign | ||||
| Temperature (°C) | ||||
| <37.8 | 5/28 (17.9) | 3/32 (9) | 0.9 | 0.454 |
| 37.8-38.9 | 9/28 (32.1) | 19/32 (59) | 4.5 | 0.042 |
| >38.9 | 14/28 (50) | 10/32 (31) | 2.2 | 0.189 |
| Heart murmur | 18 (48.6) | 26 (65) | 2.1 | 0.172 |
| Loud S, | 3 (8.1) | 14 (35) | 8.1 | 0.006 |
| Extra heart sound | 9 (22) | 9.4 | 0.002 | |
| "Tumor plop" | 2 (5) | 1.9 | 0.494 | |
| Splenomegaly | 3 (8) | 2.9 | 0.241 | |
| Skin lesions | 3 (8.1) | 6 (15) | 0.9 | 0.484 |
| Neurological deficits | 2 (5.4) | 3 (8) | 0.1 | 1.000 |
Laboratory findings showed that leukocyte count was elevated in 20 patients (76.9%) and normal in 6 (23.1%) patients (x2=15.1, P=0.000) with a quantitative result of 15.7±7.3 (range, 4.9-33.6; median, 15.2) ×109/L (n=26). Hemoglobin was reduced indicating a mild anemia in 13 (92.9%) patients and normal in 1 (7.1%) patient (x2=20.6, P=0.000) with a quantitative result of 10.4±1.3 (range, 9-13.7; median, 10.8) g/dL (n=14). Erythrocyte sedimentation rate and C-reactive protein were elevated in all studied cases: erythrocyte sedimentation rate was 83.3±28.4 (range, 37-124; median, 83) mm/h (n=15) and C-reaction protein was 15.8±10.5 (range, 3-39.9; median, 13.2) mg/dL (n=17). Microscopic hematuria was present in 2 (5.4%) patients.
Microorganisms
All 39 patients had microbiological evidence confirmed by one or more investigations including blood culture, culture of resected myxoma, histopathological inspection for bacteria or confirmation by polymerase chain reaction. Blood culture for microbiological isolation was positive in 37, negative in 1 and unstated in 1. Cultures of resected myxoma tissues were done in 15 patients: 9 (60%) were positive and 6 (40%) were negative. Histopathological studies of the resected myxomas were performed in 30 patients. They were histopathologically inspected for bacteria in 14 patients: 13 (92.9%) were positive and 1 (7.1%) was negative. The 41 bacteria of 39 patients were summarized in Table 4.
Table 4.
Pathogens of infective cardiac myxoma
| Pathogens | Present | Revankar & Clark's | x2 | P value |
|---|---|---|---|---|
| Streptococci | 17 (41.5) | 20 (50) | 0.6 | 0.507 |
| Streptococcus viridans | 7 | |||
| Streptococcus mutans | 2 | |||
| Streptococcus bovis | 2 | |||
| Streptococcus mitis | 2 | |||
| Streptococcus (Group C) | 1 | |||
| Streptococcus agalactiae | 1 | |||
| Streptococcus oralis | 1 | |||
| Streptococcus salivarius | 1 | |||
| Staphylococci | 12 (29.3) | 7 (18) | 1.6 | 0.295 |
| MSSA | 4 | |||
| MRSA | 3 | |||
| Staphylococcus aureus | 2 (1 was oxacillin-resistant (ORSA)) | |||
| Staphylococcus lugdunensis | 1 | |||
| Staphylococcus spp | 1 | |||
| Staphylococcus (species not given) | 1 | |||
| Enterococcus faecalis | 4 (9.8) | 2 (5) | 0.7 | 0.675 |
| Gram-negative bacilli | 2 (4.9) | 3 (8) | 0.2 | 0.675 |
| Klebsiella pneumoniae | 2 | |||
| Gram-negative cocci | 1 (2.4) | 1.0 | 1.000 | |
| Neisseria lactamica | 1 | |||
| Fungus | 1 (2.4) | 3 (8) | 1.1 | 0.359 |
| Histoplasma capsulatum | 1 | |||
| Actinomyce | 1 (2.4) | 1.0 | 1.000 | |
| Actinobacillus actinomycetemcomitans | 1 | |||
| Unknown | 3 (7.3) | 3 (8) | 0.0 | 1.000 |
| Bacterial colonies | 1 | |||
| Gram-positive cocci | 2 |
Histopathology showed inflammatory cell infiltrate in 11 (36.7%), necrosis in 4 (13.3%) and micro-/focal abscesses in 2 (6.7%) patients, respectively.
Complications
There were 12 patients in whom complications developed. Of these, embolic events in 10 (8 were multiple sites or multiple organs), sepsis in 4 (one was septic shock), disseminated intravascular coagulation in 3 and lung abscess in 1. The embolic events were shown in Table 5. In comparison, 18 patients of Revankar & Clark's[2] series developed embolic events and only one of them were multisites (80% (8/10) vs. 5.6% (1/18), x2=16.3, P=0.000).
Table 5.
Embolic events.
| Location | n (%) |
|---|---|
| Cerebral | 1 (10) |
| Cerebral, peripheral (extremities) | 1 (10) |
| Cerebral, splenic | 1 (10) |
| Cerebral, splenic, renal | 1 (10) |
| Coronary (left anterior descending coronary artery) | 1 (10) |
| Multiple (location not indicated) | 1 (10) |
| Multiple peripheral (left common + external iliac arteries + right deep femoral artery) | 1 (10) |
| Pulmonary | 1 (10) |
| Splenic | 1 (10) |
| Splenic, renal | 1 (10) |
Treatment
Thirty-eight of 39 patients received a surgical treatment of infected cardiac myxoma and the reason that only patient did not undergo any surgical procedure was due to rapid deterioration leading to death. Of the 38 surgical operations, 30 were isolated cardiac myxoma resections, 5 were cardiac myxoma resection with concurrent cardiac surgical procedures (mitral valve replacement in 2, and mitral valve replacement + coronary artery bypass grafting, mitral valve replacement + embolectomy of the left anterior descending coronary artery + coronary artery bypass grafting, and pulmonary valve replacement in 1 patient each) and 3 with staged peripheral operations (3rd toe of right foot amputation & right femoropoplitereal bypass; right common iliac artery thrombectomy and embolectomy of the left common and external iliac and right deep femoral arteries in 1 patient each).
The timing for surgical operation was reported in 26 patients, 4 of them were operated on an urgent basis and 22 had a delay of 18±13.5 (range, 3-42; median, 14) days (n=21) after admission for the purpose of sufficient preoperative antibiotic treatment and stabilization of patients' conditions. Preoperative antibiotic treatment was described in 26 patients. The frequently used antibiotics included vancomycin (7 patients), penicillin (4 patients) and ampicillin (3 patients). Sometimes, they were used along with gentamicin (80 mg every 8 h). Other antibiotics were imipenem, meropenem, peperacillin, trimethoprim/sulfamethoxazole, oxicillin, ampicillin, nafcillin, teicoplanin, azitromicine, ceftriaxone, clindamycin and amoxicillin-clavulanic acid.
The duration of preoperative antibiotic use was 22.3±13.4 (range, 7-42; median, 18) days (n=12). Postoperative antibiotic regimens included vancomycin (500 mg every 6 h), ceftazidime (2 g every 8 h) and netilmicin (150 mg every 12 h) followed by cefepime (1 g every 12 h) plus teicoplanin (400 mg daily), nafcillin (2 g intravenously every 6 h) and gentamicin and antifungal drugs with a therapeutic course of 31.2±5.1 (range, 27-42; median, 28) days (n=9).
Outcomes
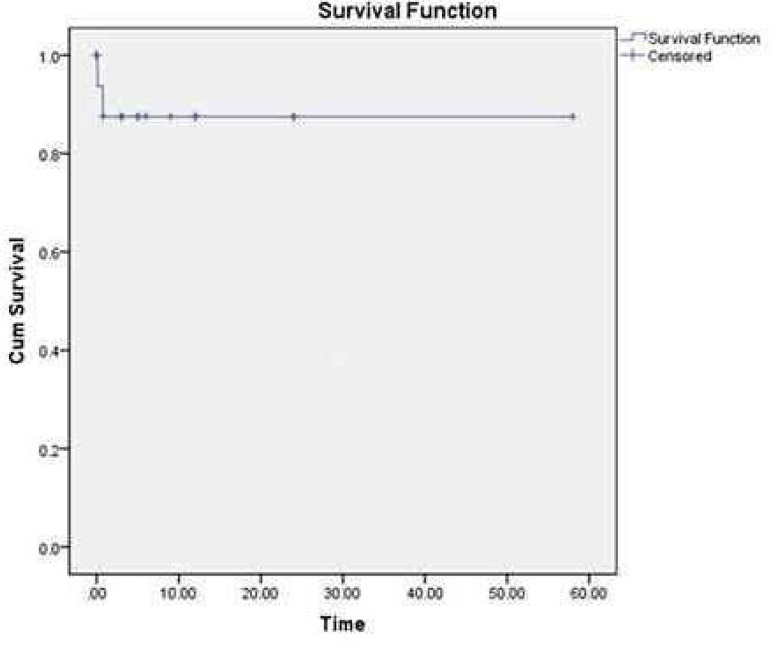
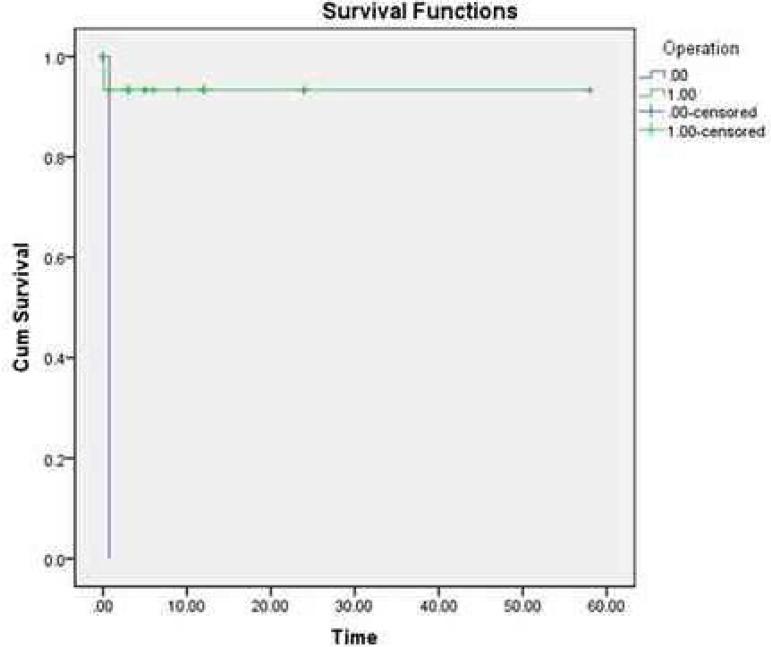
During a follow-up period of 11.1±14.5 (0.1-58; median, 8.5) months (n=16), 37 (92.6%) patients survived and 2 (7.4%) died. Of the 37 survived patients, 35 (94.6%) were event-free and 2 (5.4%) were complicated with ruptured saccular abdominal aortic aneurysm with renal infarct and septic emboli requiring aortoiliac bypass in one patient, and increased cerebral hematoma requiring craniotomy in another. One patient died of rupture of a right lung abscess 3 weeks after admission and the other died of disseminated intravascular coagulation on postoperative day 10. The overall mortality was 5.1% and the operative mortality was 2.6%, with a significantly reduced overall mortality in comparison with that of the patient series of Revankar & Clark (overall mortality: 5.1% vs. 21%, x2=4.0, P=0.047; operative mortality: 3% vs. 2.6%, x2=1.4, P=0.239). The overall survival rate was 92.6% (Figure 1). The survival rates of the surgical and non-surgical patients were 96.2% and 0%, respectively (Figure 2).
Fig. 1.
The overall survival of the present series was 92.6%.
Fig. 2.
The survival rates of the surgical and non-surgical patients were 96.2% and 0%, respectively.
DISCUSSION
In 1998, Revankar & Clark[2] defined infected cardiac myxoma in three levels based on clinical and pathological findings of the myxoma:
Definite infected cardiac myxoma
1. Documented myxoma by pathology and
2a. Microorganisms seen on pathology or
2b. Positive blood cultures and inflammation on pathology.
Probable infected cardiac myxoma
Documented myxoma by pathology and
Positive blood cultures or inflammation on pathology.
Possible infected cardiac myxoma
Characteristic appearance by transthoracic or transesophageal echocardiography and
Positive blood cultures.
Using these criteria, the three-level infected cardiac myxoma accounted for 85%, 12.5% and 2.5% respectively as reported by Revankar & Clark[2], and 87.2%, 10.3% and 2.6% in the present series.
The diagnosis of infected cardiac myxoma can be a challenge. The time interval from symptom onset to establishment of the diagnosis varied from 0 to 126 (median, 4) months[40]. Pathogens can be evidenced by blood culture, culture or staining of resected myxoma, and occasionally confirmation by polymerase chain reaction is helpful. The transthoracic or transesophageal echocardiography is a reliable means for the detection of a cardiac myxoma. Only in patients with rapid deterioration leading to sudden death was the diagnosis of a cardiac myxoma established by autopsy.
Comparisons between the two series revealed that the present series were characterized by few occasions of moderate-grade fever (while more occasions of high-grade fever in spite of lack of a statistical significance) and abnormal heart sound, but more uncommon microorganisms and more severe complications -- multiple embolic events. Other clinical features of the two patient series did not differ from each other. The somehow aggravated situations may contribute to the presence of refractory microorganisms, such as Gram-negative bacteria, fungus and actinomyce.
In comparison with Revankar & Clark's[2] series, the present series disclosed a significantly decreased overall mortality. It is believed that refinement of the prompt diagnosis and timely management (use of sensitive antibiotics and surgical resection of the infected myxoma) have resulted in better outcomes of such patients.
CONCLUSION
In conclusion, the present series of infected cardiac myxoma illustrated somehow aggravated clinical manifestations (relative more occasions of high-grade fever, multiple embolic events and the presence of refractory microorganisms), which should draw sufficient attention to be careful in the diagnosis and treatment. In general, the prognosis of infected cardiac myxoma is relative benign and now the long term survival is always promising.
| Authors' roles & responsibilities | |
|---|---|
| SMY | Study conception and design; analysis and/or interpretation of data; manuscript writing, final approval of the manuscript. |
Footnotes
Work carried out at The First Hospital of Putian, Teaching Hospital, Fujian Medical University, Putian, Fujian Province, People's Republic of China.
No financial support.
REFERENCES
- 1.Nath MP, Singh B, Chakrabarty A. Left atrial myxoma presenting as stroke: case report & review of literature; Indian Anaesth Forum; [March 23, 2014]. Available from: http://www.theiaforum.org. [Google Scholar]
- 2.Revankar SG, Clark RA. Infected cardiac myxoma. Case report and literature review. Medicine (Baltimore) 1998;77(5):337–344. doi: 10.1097/00005792-199809000-00003. [DOI] [PubMed] [Google Scholar]
- 3.Adamira M, Justik P, Ulman J, Brezina A, Mirejovsky T, Trnkova M. Two sporadic infected cardiac myxomas in 1 patient. Tex Heart Inst J. 2011;38(2):191–193. [PMC free article] [PubMed] [Google Scholar]
- 4.Awab A, Hamadani M, Sud B, Voskuhl GW. Infected atrial myxoma. Med J Aust. 2006;185(6):332–332. doi: 10.5694/j.1326-5377.2006.tb00590.x. [DOI] [PubMed] [Google Scholar]
- 5.Belgi Yildirim A, Er A, Küçük M, Ozbilim G. Infected giant left atrial myxoma: an unusual phenomenon. Anadolu Kardiyol Derg. 2011;11(1):83–85. doi: 10.5152/akd.2011.015. [DOI] [PubMed] [Google Scholar]
- 6.Bhanot N, Sahud AG, Bhat S, Lane S, Manyam H, Chan-Tompkins NH. Fever of unknown origin: a case of cardiac myxoma infected with Staphylococcus lugdunensis. South Med J. 2010;103(7):697–700. doi: 10.1097/SMJ.0b013e3181e1e45f. [DOI] [PubMed] [Google Scholar]
- 7.Brazill PL. Infected cardiac myxoma: an unusual phenomenon. Can Oper Room Nurs J. 2001;19(3):20–21. [PubMed] [Google Scholar]
- 8.Chan V, Veinot JP, Hynes M, Lapierre H, Ruel M. Infected right ventricular myxoma and pulmonary valve endocarditis. J Thorac Cardiovasc Surg. 2007;134(1):248–249. doi: 10.1016/j.jtcvs.2007.03.026. [DOI] [PubMed] [Google Scholar]
- 9.Chang JH, Kim JY, Yoon JW, Seol MD, Won DJ, Cho WH, et al. A case of infected left atrial myxoma with concomitant mitral valve endocarditis. Korean Circ J. 2011;41(10):618–621. doi: 10.4070/kcj.2011.41.10.618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Dekkers P, Elbers HR, Morshuis WJ, Jaarsma W. Infected left atrial myxoma. J Am Soc Echocardiogr. 2001;14(6):644–645. doi: 10.1067/mje.2001.111476. [DOI] [PubMed] [Google Scholar]
- 11.Falasca K, Ucciferri C, Mancino P, Di Girolamo A, Vecchiet J. Infected atrial myxoma: a rare cause of fever. Infez Med. 2008;16(1):40–42. [PubMed] [Google Scholar]
- 12.Furukawa A, Kishi S, Aoki J. Large infected atrial myxoma with vegetations. Rev Esp Cardiol. 2013;66(4):310–310. doi: 10.1016/j.rec.2011.03.031. [DOI] [PubMed] [Google Scholar]
- 13.García-Quintana A, Martín-Lorenzo P, Suárez de Lezo J, Díaz-Escofet M, Llorens R, Medina A. Infected left atrial myxoma. Rev Esp Cardiol. 2005;58(11):1358–1360. [PubMed] [Google Scholar]
- 14.Gregory SA, O'Byrne WT, 3rd, Fan P. Infected cardiac myxoma. Echocardiography. 2004;21(1):65–67. doi: 10.1111/j.0742-2822.2004.02177.x. [DOI] [PubMed] [Google Scholar]
- 15.Guler N, Ozkara C, Kaya Y, Saglam E. Ruptured abdominal aortic aneurysm after resection of an infected cardiac myxoma. Tex Heart Inst J. 2007;34(2):233–235. [PMC free article] [PubMed] [Google Scholar]
- 16.Janion M, Sielski J, Ciuraszkiewicz K. Sepsis complicating giant cardiac myxoma. Am J Emerg Med. 2008;26(3):387–387. 3–4. doi: 10.1016/j.ajem.2007.07.030. [DOI] [PubMed] [Google Scholar]
- 17.Juang SE, Lai HC, Lan YC, Liu TJ, Lai HC. Left atrial infective endocarditis with giant vegetation without involvement of the mitral valve: case report of transesophageal echocardiography in diagnosis. Acta Anaesthesiol Taiwan. 2005;43(3):165–167. [PubMed] [Google Scholar]
- 18.Karachalios G, Bablekos G, Karachaliou I, Zoganas L, Charalabopoulos A, Charalabopoulos K. Left atrial myxoma prolapsing into the left ventricle. Case report and review of the literature. Chemotherapy. 2004;50(6):297–301. doi: 10.1159/000082629. [DOI] [PubMed] [Google Scholar]
- 19.Leone S, dell'aquila G, Giglio S, Magliocca M, Maio P, Nigro FS, et al. Infected atrial myxomacase report and literature review. Infez Med. 2007;15(4):256–261. [PubMed] [Google Scholar]
- 20.Nagata T, Totsugawa T, Katayama K, Kuinose M, Yoshitaka H, Uesugi T. Infected cardiac myxoma. J Card Surg. 2013;28(6):682–684. doi: 10.1111/jocs.12184. [DOI] [PubMed] [Google Scholar]
- 21.Oyama H, Nakayama M, Ikeda A, Maeda M, Miyahara T, Inoue S, et al. A case of cardiac myxoma with multiple brain hemorrhage. No Shinkei Geka. 2001;29(6):533–537. [PubMed] [Google Scholar]
- 22.Parks JD, Thangathurai D, Farhoomand L, Riad MG. Infected atrial myxoma presenting with septic shock. Mar 30, 2014. http://www.usc.edu/schools/medicine//departments/anesthesiology/assets/WARC2004/parks1.pdf [DOI] [PubMed] [Google Scholar]
- 23.Prince JM, Larsen R, Janner D. Infected cardiac myxoma. Pediatr Infect Dis J. 2002;21(2):177–178. doi: 10.1097/00006454-200202000-00023. [DOI] [PubMed] [Google Scholar]
- 24.Puvaneswary M, Thomson D. Magnetic resonance imaging features of an infected right atrial myxoma. Australas Radiol. 2001;45(4):501–503. doi: 10.1046/j.1440-1673.2001.00963.x. [DOI] [PubMed] [Google Scholar]
- 25.Quigley RL, Meursing DF, Rossman MI. Left atrial myxoma and mitral valve endocarditis--a cause and effect: a case report. Heart Surg Forum. 2006;9(1):E486–E487. doi: 10.1532/HSF98.20051152. [DOI] [PubMed] [Google Scholar]
- 26.Riad MG, Parks JD, Murphy PB, Thangathurai D. Infected atrial myxoma presenting with septic shock. J Cardiothorac Vasc Anesth. 2005;19(4):508–511. doi: 10.1053/j.jvca.2005.05.010. [DOI] [PubMed] [Google Scholar]
- 27.Sasaki A, Baba T. Infected left atrial myxoma: report of a case. Kyobu Geka. 2010;63(11):1009–1011. [PubMed] [Google Scholar]
- 28.Surovcík R, Jebavý P, Feuereisl R, Frídl P, Pavlovic J, Sterba D. Infected myxoma as a cause of acute infective endocarditis. Vnitr Lek. 2010;56(2):154–156. [PubMed] [Google Scholar]
- 29.Tanaka M, Kawahito K, Adachi H, Yamaguchi A, Ino T. Infected left atrial myxoma with mitral valve endocarditis. Jpn J Thorac Cardiovasc Surg. 2002;50(3):137–139. doi: 10.1007/BF02913478. [DOI] [PubMed] [Google Scholar]
- 30.Toda R, Moriyama Y, Shiota K, Toyohira H, Taira A. Myxoma of mitral valve associated with infective endocarditis. Jpn J Thorac Cardiovasc Surg. 1999;47(6):285–287. doi: 10.1007/BF03218012. [DOI] [PubMed] [Google Scholar]
- 31.Trimeche B, Bouraoui H, Garbaa R, Mahdhaoui A, Ben Rhomdane M, Ernez-Hajri S, et al. Systemic embolism and septic shock complicated left atrial myxoma: case report. [March 30, 2014];Case Rep Med. 2009 doi: 10.1155/2009/306375. Available from: http://www.hindawi.com/journals/crim/2009/306375/abs/ [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Uchino K, Mochida Y, Ebina T, Tobe M, Kobayashi S, Yano Y, et al. Infected left atrial myxoma. Intern Med. 2002;41(11):957–960. doi: 10.2169/internalmedicine.41.957. [DOI] [PubMed] [Google Scholar]
- 33.Veitch AM, Manghat NE, Kakani NK, Lewis CT, Ring NJ. Systemic septic embolisation secondary to an atrial myxoma in a young woman. Emerg Radiol. 2006;12(3):137–139. doi: 10.1007/s10140-005-0444-x. [DOI] [PubMed] [Google Scholar]
- 34.Yoshioka D, Takahashi T, Ishizaka T, Higuchi T. Successful surgical resection of infected left atrial myxoma in a case complicated with disseminated intravascular coagulation and multiple cerebral infarctions: case report. J Cardiothorac Surg. 2011;6:68–68. doi: 10.1186/1749-8090-6-68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zechini B, Cipriani P, Papadopoulou S, Di Nucci G, Petrucca A, Teggi A. Endocarditis caused by Lactococcus lactis subsp. lactis in a patient with atrial myxoma: a case report. Diagn Microbiol Infect Dis. 2006;56(3):325–328. doi: 10.1016/j.diagmicrobio.2006.04.011. [DOI] [PubMed] [Google Scholar]
- 36.Zwinkels RL, van der Sar-van der Brugge S, Sleeswijk Visser SJ. An unexpected cause of sepsis in a patient with dental decay. Rare example of an infected right atrial calcified myxoma, with extensive calcified pulmonary emboli. Acute Med. 2013;12(1):34, 59-61. [PubMed] [Google Scholar]
- 37.Bernstein JM, Leasure W, Buel A. Getting to the heart of the matter. Skinmed. 2007;6(6):290–292. doi: 10.1111/j.1540-9740.2007.06187.x. [DOI] [PubMed] [Google Scholar]
- 38.Liu YL, Liu XH, Cai KH. Cardiac valve myxomas with infected endocarditis in the youngerly. Zhonghua Bing Li Xue Za Zhi. 2005;34(10):695–696. [PubMed] [Google Scholar]
- 39.Marshall C, McDonald M. Recurrent subacute bacterial endocarditis as a presentation of left atrial myxoma. Aust N Z J Med. 1998;28(3):350–350. doi: 10.1111/j.1445-5994.1998.tb01965.x. [DOI] [PubMed] [Google Scholar]
- 40.Pinede L, Duhaut P, Loire R. Clinical presentation of left atrial cardiac myxoma. A series of 112 consecutive cases. Medicine (Baltimore) 2001;80(3):159–172. doi: 10.1097/00005792-200105000-00002. [DOI] [PubMed] [Google Scholar]
- 41.Despott E, Cachia MJ. An unusual cause of shortness of breath and fever. Malta Med J. 2005;17(1):36–38. [Google Scholar]