Abstract
Background
We have recently shown that inhibition of peptidylarginine deiminase (PAD) improves survival in a rodent model of lethal cecal ligation and puncture (CLP). The roles of PAD inhibitors in hemorrhagic shock (HS), however, are largely unknown. The goal of this study was to investigate the effects of YW3-56, a novel PAD inhibitor, on survival following severe HS.
Methods
Mouse macrophages were exposed to hypoxic conditions followed by reoxygenation in the presence or absence of YW3-56. Enzyme-linked immunosorbent assay (ELISA) was performed to measure levels of secreted tumor necrosis factor α (TNF-α) and interleukin (IL)-6 in the culture medium. Cell viability was determined by methyl thiazolyl tetrazolium assay. In the survival experiment, anesthetized male Wistar-Kyoto rats (n=10/group) were subjected to 55% blood loss, treated with or without YW3-56 (10 mg/kg, intraperitoneally). Survival was monitored for 12 h. In the non-survival experiment, morphorlogic changes of the lungs were examined. Levels of circulating cytokine-induced neutrophil chemoattractant 1 (CINC-1) and myeloperoxidase (MPO) in the lungs were measured by ELISA. Expression of lung intercellular adhesion molecules-1 (ICAM-1) was also determined by Western blotting.
Results
Hypoxia/reoxygenation (H/R) insult induced TNF-α and IL-6 secretion from macrophages, which was significantly attenuated by YW3-56 treatment. YW3-56 treatment also increased cell viability when macrophages were exposed to H/R up to 6/15h, and improved survival rate from 20% to 60% in lethal HS rat model. Compared to the sham groups, pulmonary MPO activity and ICAM-1 expression in the HS group were significantly increased, and acute lung injury was associated with a higher degree of CINC-1 levels in serum. Intraperitoneal delivery of YW3-56 significantly reduced pulmonary MPO and ICAM-1 expression and attenuated acute lung injury (ALI).
Conclusions
Our results demonstrate for the first time that administration of YW3-56, a novel PAD inhibitor, can improve survival in a rat model of hemorrhagic shock as well as in a cell culture model of H/R. The survival advantage is associated with an attenuation of local and systemic pro-inflammatory cytokines and the protection against ALI following hemorrhage. Thus, PAD inhibition may represent a novel and promising therapeutic strategy for severe HS.
Keywords: Peptidylarginine deiminase inhibitor, hemorrhagic shock, survival, inflammation, rat
1. Introduction
Severe hemorrhage is a common cause of morbidity and mortality in civilian and combat trauma patients by recruiting neutrophils and promoting the release of inflammatory cytokines. Excessive activation of neutrophils may lead to the development of multiple organ dysfunction syndromes [1], which primarily targets the lungs. Acute lung injury (ALI) is an early deleterious consequence of hemorrhage and is characterized by the accumulation of neutrophils in the lung as well as the production of inflammatory mediators, including complement activation products, cytokines and chemokines, proteases, and oxidants. Interaction of activated neutrophils with resident pulmonary cells, such as endothelial cells and macrophages, appears to play a central role in the pathogenesis of ALI after hemorrhagic shock (HS). Ischemia and reperfusion (I/R) injury with excessive activation of the innate immune system are considered to be key underlying mechanisms responsible for cellular damage. Pro-inflammatory cytokines produced during HS and trauma can cause organ failure and death [2]. Current therapies for massive hemorrhage largely focus on restoring the lost intravascular volume but have failed to address the specific cellular dysfunction caused by shock and resuscitation. Our team has addressed this gap by identifying pharmacological treatments that are capable of activating specific pro-survival and anti-inflammatory pathways in various models of lethal insults [3].
Peptidylarginine deiminases (PADs) constitute a family of calcium dependent enzymes that post-translationally convert positively charged arginine residues to neutrally charged citrulline. PAD- mediated citrullination can modify the tertiary structure of target substrates and/or alter protein-protein interactions, thereby affecting various cellular processes [4]. Recently, protein citrullination has garnered increased attention due to its implications in various human and animal cancers as well as its role in the pathogenesis of various inflammatory conditions such as rheumatoid arthritis (RA), multiple sclerosis, chronic obstructive pulmonary disease (COPD), and neurodegenerative diseases [5]. It has been reported that PAD4 activation can exacerbate the inflammatory response after renal ischemia/reperfusion (I/R) [6] and also plays a crucial role in the pathogenesis of myocardial I/R injury [7].
We have recently shown that inhibition of PAD could improve survival in a lethal model of cecal ligation and puncture (CLP) [8]. However, the role of PAD inhibitors in HS remains largely unknown. As hemorrhage and resuscitation are well known to create a systemic inflammatory response, we reasoned that blockage of PAD would be beneficial. Thus, the goal of this study was to investigate whether treatment with YW3-56, a novel PAD inhibitor, could improve survival following lethal HS in a rat model by attenuating systematic and local inflammation. .
2. Materials and methods
2.1. Animals
Experiments were designed and performed in accordance with the statutes from The Guide for the Care and Use of Laboratory Animals (National Research Council, 1996 edition). This study complied with the Animal Welfare Act and other Federal regulations and was approved by our Institutional Animal Care and Use Committee.
2.2. Cell cultures and in vitro experimental procedures
RAW 264.7 macrophage cells were cultured in Dulbecco's Modified Eagle's Medium, supplemented with 10% fetal bovine serum, 100 U/ml of penicillin, and 100 U/ml of streptomycin (Invitrogen, Grand Island, NY) at 37°C and 5% CO2. RAW 264.7 cells were seeded at a density of 1.5×104 cells/well into 96-well plates with fresh DMEM for 24 hours before hypoxia/reoxygenation (H/R). Before induction of hypoxia, the culture medium was replaced with low serum media (0.5% FBS). Cells were treated with or without YW3-56 (5.0μM) and then exposed to hypoxia for 6h by transferring the culture plates to a humidified incubation chamber thermos-regulated at 37 °C and flushed with a gas mixture consisting of 95% N2/5% CO2 described previously [9]. After incubation in hypoxic conditions, the cells were provided with fresh medium and then moved to 95% air/5% CO2 for reoxygenation. The control plates were kept in normoxic condition.
2.3. Detection of TNF-α and IL-6 by enzyme-linked immunosorbent assay (ELISA)
Cell culture supernatants were collected 4 h post-reoxygenation and saved for TNF-α (R&D, MTA00B) and IL-6 (R&D, M6000B) quantification by ELISA according to the manufacturer's instructions. Concentrations were calculated from the standard curve made with recombinant rat TNF-α and IL- 6.
2.4. MTT assay
MTT (3-[3, 5-dimethylthiazol-2-ul]-2, 5-diphenyltetrazolium bromide; M2128, Sigma Aldrich, St. Louis, MO) assay was used to assess cell viability. Cells were treated with or without YW3-56 (5.0μM) and placed in a hypoxia chamber for 6 hours followed by reoxygenation in a normoxic incubator for 15 hours. Twenty microliters of MTT solution (0.5 mg/ml) was added to each well and was incubated at 37 °C for 4 h. After discarding the supernatant carefully, 150 μl dimethyl sulfoxide (DMSO) was added to each well and shaken for 30 min; absorbance was subsequently determined at 590 nm by a microplate reader.
2.5. Surgical procedure
Rats had access to food and water ad libitum before and after surgery. Male Sprague–Dawley rats (220–290 g; Charles River Laboratories, Wilmington, MA) were anesthetized with 5% isoflurane, and a veterinary multichannel anesthesia delivery system (Kent Scientific Corporation, Torrington, CT) was used to administer isoflurane at 0.8%–2.5% inspiratory fraction. Left femoral vessels were then isolated and aseptically cannulated with PE50 polyethylene catheters (Clay Adams, Sparks, MD) primed with heparinized saline (100 USP U/ml). The venous line was used for sampling and hemorrhage. In the survival experiment, rats were subjected to a lethal hemorrhage of 55% total blood volume over 40 min. The hemorrhage volume was calculated using the formula: total blood volume (ml) = rat's weight (g) × 0.06 (ml/g) + 0.77 [10]. Thirty five percent of total blood volume was withdrawn over 10 min from the arterial catheter and the remaining 20% was removed from the venous catheter over the next 30 min. Removal of 55% total blood volume results in severe hemorrhagic shock with very high lethality[10, 11]. The rats were given YW3-56 (10mg/kg, gifted by Dr. Yanming Wang, Penn State University) dissolved in DMSO or vehicle DMSO intraperitoneally 30 minutes after HS (n = 10 per group). After treatment, cannulas were removed, vessels were ligated, skin was closed, and the animals were returned to their cages for monitoring (12 hours) . In the non-survival experiment, animals were randomly assigned to the following three groups (n = 4 per group): (a) sham-operated (instrumentation and no hemorrhage) animals, (b) vehicle-treatment after HS and (c) YW3-56-treatment after HS. Rats were subjected to a sublethal hemorrhage of 40% total blood volume over 10 min. Thirty minutes later, they were randomly assigned to receive either DMSO or YW3-56 (10mg/kg, intraperitoneally). Animals returned to their cages for 6h, and then killed for serum and tissue harvest. Right upper lobes of the lungs were fixed in 4% buffered formalin for histologic analysis, and the rest of the lungs were immediately snap-frozen in liquid nitrogen, and stored at −80°C until further processing.
2.6. Histological analysis
For histological examinations, lung tissues fixed in 10% neutral buffered formalin, were embedded in paraffin, and sectioned at a thickness of 4–6 μm. After deparaffinization and dehydration, the sections were stained with hematoxylin and eosin and were examined using light microscopy by an experienced observer blinded to the group allocation of the samples. Histopathological scoring analysis was performed according to previously described methods. The severity of each category was graded from 0 (minimal) to 4 (maximal) for each of the following four parameters: intravascular congestion, pulmonary edema, inflammatory cellular infiltration, and intra-alveolar hemorrhage. The total score was calculated by adding the scores in each of these categories. In each animal, three separate lung sections were graded to generate the mean score.
2.7. Myeloperoxidase (MPO) activity
Pulmonary MPO levels were quantified using the rat MPO Enzyme-linked immunosorbent assay(ELISA) kit (Cell Sciences, Canton, MA). Rat lung tissue (50 mg/sample) was homogenized using a glass handheld homogenizer in 1 mL of a lysis buffer (200 mM NaCl, 5 mM EDTA, 10 mM Tris, 10% glycine, 1 mM phenylmethylsulfonyl fluoride, 1 μg/mL leupeptide, and 28 μg/mL aprotinin). The samples were centrifuged twice at 1,500 g at 4°C for 15 minutes, and supernatants were analyzed for MPO levels by following the manufacturer's instructions.
2.8. Cytokine-induced neutrophil chemoattractant 1 (CINC-1) assessed by ELISA
CINC-1 levels in the serum were quantified using the Quantikine rat CINC-1 ELISA kit (R&D, RCN100). Serum samples were prepared as described above. CINC-1 concentrations were quantified in 50 μL of serum according to the manufacturer's instructions by measuring optical densitometry values at 450 nm using a SpectraMax® Plus 384 Microplate Reader (Molecular Devices, Sunnyvale, CA).
2.9. Western blot analysis
Cell lysates were prepared from the lung samples using a whole cell lysis buffer kit (BD Biosciences, Bedford, MA) according to the manufacturer's instructions. Protein concentration was determined by bicinchoninic acid method, and equal amounts of samples were loaded per lane. Proteins were detected using specific antibodies against intercellular adhesion molecules-1(ICAM-1, R&D, MAB583), and were incubated with corresponding IRDye 680RD conjugated second antibodies (LI-COR Bioscience, Lincoln, NE). The subsequent signals were visualized using Odyssey CLx Infrared Imaging System (LI-COR), and the results were quantified using ImageJ software (National Institutes of Health, Bethesda, MD).
2.10. Statistical analyses
The Kaplan-Meier method was used to compare the survival, and significance in survival curves between the groups was measured using the non-parametric log-rank test. One-way analysis of variance (ANOVA) models were conducted to examine the effects of YW3-56 on MTT, TNF-α, IL-6, ALI score, MPO, CINC-1, and ICAM-1/β-actin following severe HS. F-test was performed to show the overall significance of differences between the three groups, followed by Bonferroni for multiple inter-group comparisons. P value <0.05 was considered significant. All of the analyses in the present study were performed on Windows 8 using SAS statistical software (SAS version 9.2, SAS Institute Inc., Cary, NC, USA).
3. RESULTS
3.1. YW3-56 administration protected the macrophages from death induced by H/R
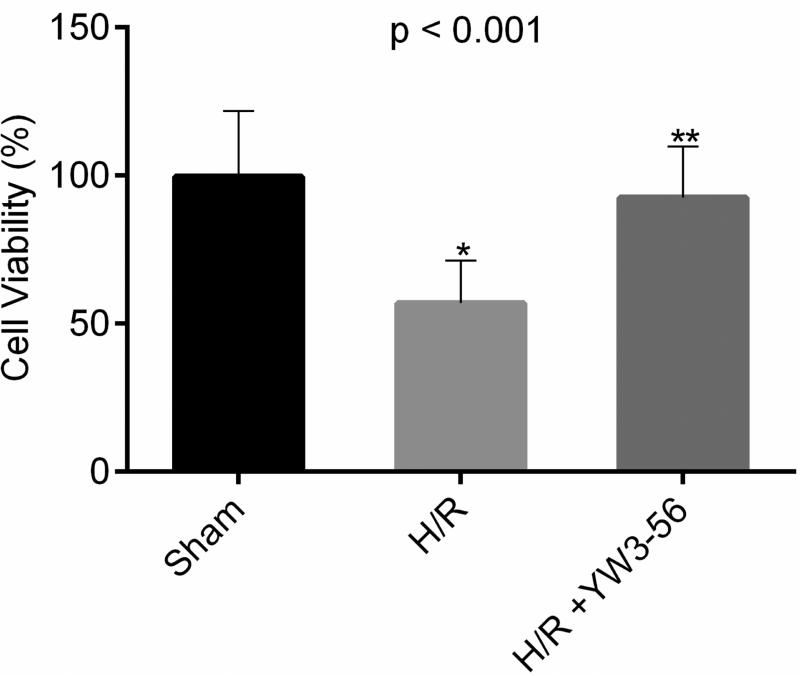
We first analyzed the effects of YW3-56 on cell viability. MTT assays showed that cell viability decreased to approximately 56.6% compared to the controls after the cells were challenged with H/R (6/15h) (Fig. 1, reduction = 42.8%, p < 0.001), whereas treatment with YW3-56 (5 μmol/L) significantly protected the cells from death (improvement = 35.7%, p < 0.001). There was no difference in cell viability between the YW3-56 treatment and the sham groups (p = 0.62).
Fig. 1. Cell viability measured by methyl thiazolyl tetrazolium (MTT) assay after 6h hypoxia followed by 15 h reoxygenation.
Data presented as % change compared with normoxic controls. p < 0.001 shows the overall significant difference among three groups. H/R caused a reduction in viability to 56.8% in the untreated cells (H/R vs. sham: *p < 0.001). Treatment with YW3-56 significantly improved the cell survival to 92.5% (YW3-56+H/R vs. H/R: **p < 0.001).
3.2. YW3-56 administration improved survival in a rat model of lethal HS
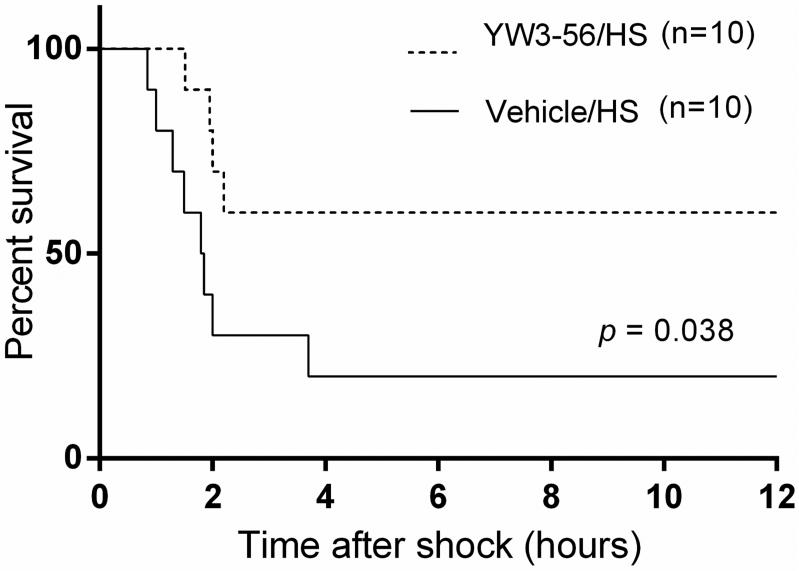
First, we investigated whether YW3-56 could prolong survival in this model of lethal hemorrhagic shock. As shown in Fig. 2, only 20% of rats in the control group survived for 12 hours, with most of the deaths occurring within the first 3hours. However, animals treated with YW3-56 displayed a significantly higher survival rate (60% of rats survived for 12 hours, p = 0.038).
Fig. 2. YW3-56 improved survival in a rat model of severe hemorrhagic shock.
Rats were administered 10 mg/kg of YW3-56 or vehicle dimethyl sulfoxide (DMSO), intraperitoneally, 30 minutes after hemorrhagic shock (HS) (n = 10/group). Survival was monitored for up to 12 h after procedure. Treatment with YW3-56 significantly improved survival compared with DMSO vehicle group (60% vs 20% survival; p = 0.038).
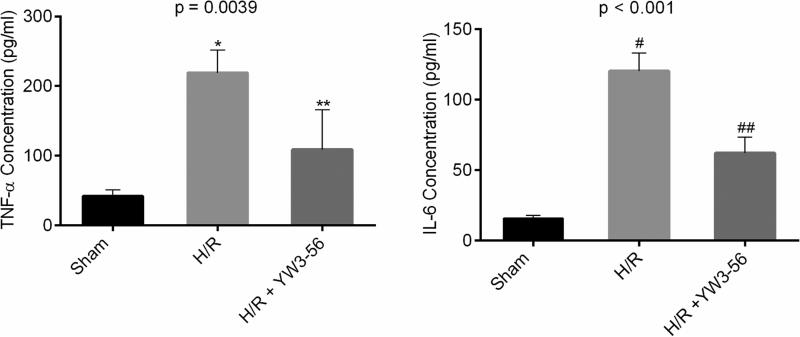
3.3. YW3-56 decreased production of TNF-α and IL-6 in macrophages challenged by H/R
We then investigated whether cells (e.g., macrophages) protected by YW3-56 could have an alteration in the expression levels of pro-inflammatory cytokines. H/R increased the production of TNF-α and IL-6 by the macrophages, as measured in the supernatant of the cell culture, whereas YW3-56 treatment significantly decreased these levels (Fig. 3).
Fig. 3. YW3-56 treatment decreased secretion of TNF-α and IL-6 in macrophages insulted by hypoxia/reoxygenation (H/R).
TNF-α and IL-6 levels were determined by Enzyme-linked immunosorbent assay (ELISA) and expressed as mean values. p = 0.0039 and p < 0.001 show the overall significant differences among three groups in TNF-α and IL-6, respectively. H/R significantly increased (H/R vs. sham: *p = 0.0041 for TNF-α; #p < 0.001 for IL-6) and YW3-56 decreased the expression of TNF-α and IL-6 (H/R+YW3-56 vs. H/R: **p = 0.0387 for TNF-α; ##p = 0.0011 for IL-6).
3.4. YW3-56 administration attenuated ALI in the sub-lethal model of hemorrhagic shock
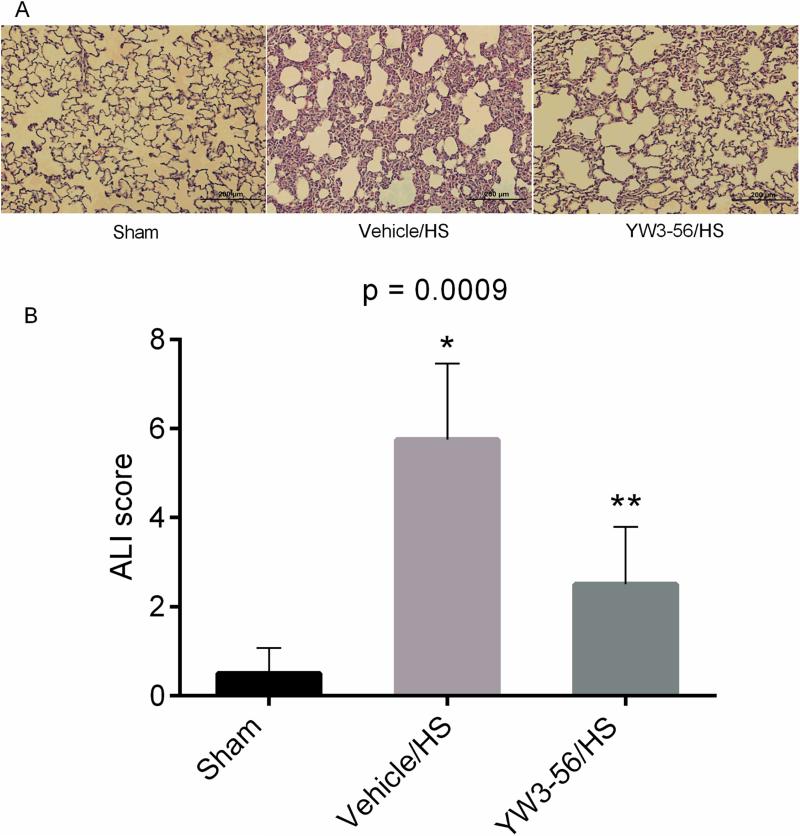
We used a sub-lethal model (40% blood loss) for this experiment to eliminate survival bias, and to ensure the availability matching samples for tissue analysis. The sham group showed normal lung histology. In contrast, the lungs of the vehicle control group were significantly damaged, with interstitial edema, hemorrhage, thickening of the alveolar wall, and infiltration of inflammatory cells. These histological changes were attenuated following YW3-56 treatment (Fig. 4). ALI scoring, performed by a pathologist blinded to the experiment, revealed that HS increased the ALI score, while YW3-56 treatment significantly reduced it (p = 0.0175).
Fig. 4. YW3-56 administration decreased the sub-lethal hemorrhage induced ALI scores.
(A) Representative histological hematoxylin and eosin (H&E)-stained sections of lungs from the sham, vehicle control, and YW3-56 treatment groups (n = 4/group). The pathologic changes were examined by light microscopy in the lung tissues 6 h after hemorrhagic shock (HS). The YW3-56 group had less alveolar congestion, neutrophil infiltration, and alveolar wall thickness. (B) Quantification of histological analysis. Acute lung injury (ALI) was scored as described in the MATERIALS AND METHODS and expressed as mean values. Original magnification × 200. P = 0.0009 shows the overall significant difference among three groups. HS resulted in a significant increase in ALI score at 6 hours (Vehicle/HS vs. sham: *p < 0.01), whereas YW3-56 attenuated this effect (YW3-56/HS vs. vehicle/HS: **p = 0.0175).
3.5. YW3-56 treatment attenuated MPO activity in lung
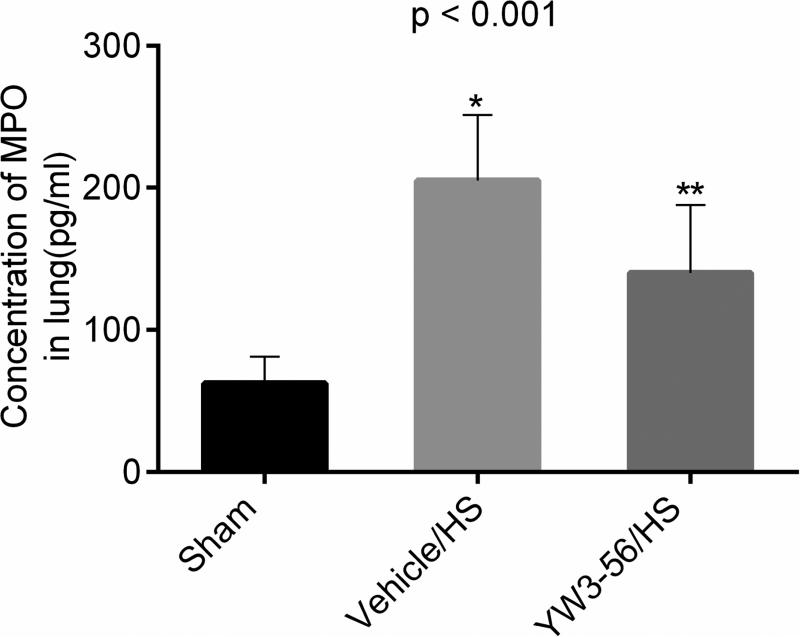
MPO levels were measured in the whole lung tissue lysate as a marker of neutrophil infiltration. Sham animals expressed low levels of MPO in the lungs, and while HS insult increased the MPO levels and YW3-56 treatment significantly attenuated it (Fig. 5, YW3-56/HS vs. vehicle/HS, p = 0.011).
Fig. 5. YW3-56 treatment attenuated myeloperoxidase (MPO) activity in lung.
Rats were hemorrhaged 40% of their blood volume, and were treated with YW3-56 (10 mg/kg, intraperitoneally) or vehicle dimethyl sulfoxide (DMSO). Lung tissue was collected for analysis at the time of end point (6 hours post shock). Data are shown as mean MPO concentration. P < 0.001 signifies significant difference among the three groups (n=8/group). Hemorrhage shock (HS) resulted in a significant increase in lung MPO levels at 6 hours (Vehicle/HS vs. sham: *p < 0.001), whereas YW3-56 attenuated this effect (YW3-56/HS vs. vehicle/HS: **p = 0.011).
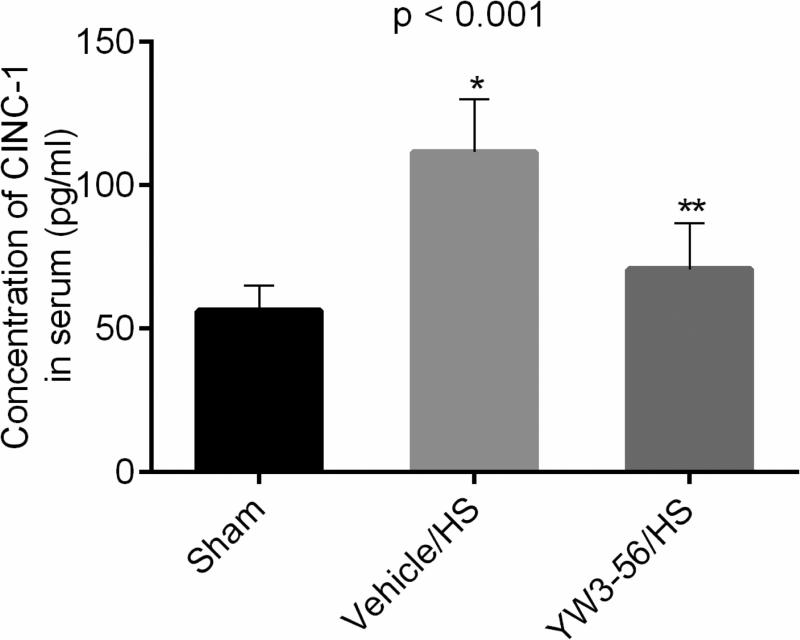
3.6. YW3-56 decreased the levels of CINC-1 in the circulation
Circulating CINC-1 levels were used as an early marker for the subsequent development of organ inflammation and injury. CINC-1 levels rose rapidly (6 h) after hemorrhage (Fig. 6), and this was significantly attenuated by treatment with YW3-56 (YW3-56/HS vs. vehicle/HS: p < 0.001).
Fig. 6. YW3-56 administration attenuated cytokine-induced neutrophil chemoattractant-1 (CINC-1) levels in serum.
Enzyme-linked immunosorbent assay was used to assess the levels of circulating CINC-1 6 hours after shock. Data shown as mean CINC-1 concentration. P < 0.001 shows the overall significant difference among three groups (n = 8/group). Hemorrhage shock (HS) resulted in a significant increase in serum CINC-1 levels at 6 hours (Vehicle/HS vs. sham: *p < 0.001), whereas YW3-56 treatment significantly attenuated this effect (YW3-56/HS vs. vehicle/HS: **p < 0.001).
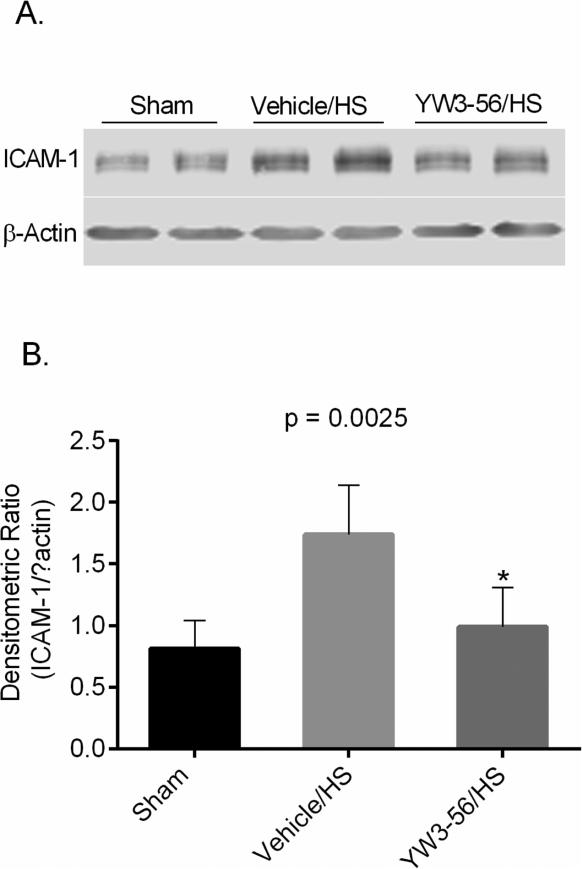
3.7. YW3-56 treatment decreased ICAM-1 expression in the lung tissue
The potential of YW3-56 to reduce pulmonary inflammation in hemorrhaged rats was assessed based on the protein levels of ICAM-1 (an inflammatory marker) 6 h after HS by Western blot analysis (Fig. 7). HS resulted in an approximately 2-fold elevation in the expression of pulmonary ICAM-1, whereas treatment with YW3-56 significantly decreased the ICAM-1 expression in the lungs (YW3-56/HS vs. vehicle/HS: p = 0.0077).
Fig. 7. YW3-56 attenuated the expression of ICAM-1 induced by hemorrhage shock (HS) in rat lung tissue.
Representative western blots (A) and optical densitometry ratio (B) show ICAM-1 and β-actin obtained from lung tissue. Whole tissue lysate of rat lung was subjected to Western blotting with anti-ICAM-1 and anti-β-actin antibodies. P < 0.0025 shows the overall significant difference among three groups. YW3-56 dramatically attenuated the expression of ICAM-1 in the lung (YW3-56/HS vs. vehicle/HS: *p = 0.0077). Specific bands were quantified by densitometric ratio and expressed as mean values. ICAM-1: intercellular adhesion molecules-1.
4. Discussion
In the current study, we investigated the therapeutic effect of YW3-56, a novel PAD inhibitor, in both a cell culture model and a rat HS model. We found that YW3-56 greatly improves survival in vitro and in vivo. Moreover, YW3-56 attenuates ALI as well as the production of pro-inflammatory cytokines and chemokines.
The first purpose of the current study was to determine whether YW3-56 could play a protective role in RAW264.1 cells subjected to an H/R insult. Our data indicate that YW3-56 treatment in this model can cause a significant reduction in cell death compared to the control. Moreover, YW3-56 decreased the secretion of cytokines (TNF-α, IL-6) by the cultured macrophages when exposed to hypoxia/reoxygenation. It has been shown that TNF-α stimulation can induce PAD2 and PAD4 expression within the nuclei of RAW264.7 cells and brain macrophages [12]. Exogenous recombinant PAD4 protein directly increases cytokine mRNA expression and cytokine secretion in cultured mouse kidney proximal tubules [6], supporting the hypothesis that PAD inhibition could attenuate the pro-inflammatory cytokines secretion. The exact mechanisms by which YW3-56 decreases cytokine (TNF-α, IL-6) secretion from the cultured macrophage remains unclear, although it is possible that PAD-mediated citrullination may increase the potency of certain proteins to induce inflammatory cytokine expression. For example, vimentin citrullination stimulates TNF-α and IL-1 production in fibroblast-like synoviocytes [13]. Our findings presented here suggest that PADs are also intimately involved in cell death under H/R conditions. Emerging evidence suggests that PADs may be specifically activated in response to apoptotic stressors.
For example, it has been shown in a mouse model that vimentin is selectively deiminated when peritoneal macrophages undergo apoptosis upon exposure to calcium ionophore [14]. PADs have been shown to be upstream regulators of Ca2+-induced cell death in human neural cells, and PAD inhibition protects cells from apoptosis by hindering calcium-induced cytoskeleton disassembly [15]. The present in vitro findings are in accordance with a previous study in a murine model of ischemic insult where pan-PAD inhibition resulted in significantly reduced apoptotic cell death [16]. It has also been reported that PAD4 inhibition protects renal tubular cells, while recombinant human PAD4 (rPAD4) treatment exacerbates their apoptosis in murine kidneys after renal I/R [6]. Taken together these findings strongly suggest that protein deimination plays an important role in cell damage.
Since YW3-56 could create a pro-survival and anti-inflammatory phenotype in vitro, the other purpose of this study was to determine whether the novel PADs inhibitor would be protective in vivo. We discovered that that YW3-56 treatment significantly improved survivalafter lethal HS. This beneficial effect can be partially attributed to the ability of YW3-56 to attenuate systemic inflammatory response and ALI that was noted in the sub-lethal HS model.
We have previously performed a multiplex assay, based on ELISA and electrochemiluminescence detection principles, which revealed elevated levels of circulating CINC-1 levels as early as at 1h after HS [16]. CINC-1 belongs to the interleukin-8 family that promotes neutrophil chemotaxis, and our study the circulating CINC-1 levels correlated with the lung MPO levels, serving as an early marker for the subsequent development of organ inflammation and injury [17]. Our present study confirms previous findings that severe HS can induce CINC-1 secretion into serum at the early stage. YW3-56 treatment attenuated hemorrhage-induced elevation of circulating CINC-1 and lung MPO levels. Histopathologic results of lung tissue and ALI scores indicate clearly that YW3-56 significantly decreases infiltration and accumulation of inflammatory cells in the lungs. One of the important steps of polymorphonuclear neutrophil (PMN) recruitment to the inflamed tissue is the attachment of circulating PMNs to vascular endothelial cells, which is mediated by cell adhesion molecules expressed on endothelial cells, such as ICAM-1. During ALI, PMNs adhere to vascular endothelium, cross the endothelial barrier, and migrate to the region of inflammation. Therefore, interactions between inflammatory cells and adhesion molecules on the surface of vascular endothelium are an important early mechanism in the development of ALI [18]. HS-induced ICAM-1 expression by endothelial cells has been studied in our lab and others, and expression of ICAM-1 in response to HS was found to be dependent on TLR4 signaling and surface expression of TLR2 [19, 20]. In the current study, we discovered that YW3-56 treatment decreases expression of ICAM-1 and attenuates the infiltration of PMNs in the lung, which contributes to the protection against ALI induced by HS. Therefore, YM3-56 treatment appears to inhibit systemic and local inflammatory changes associated with HS, which seems related to the improvement of survival following lethal blood loss by pharmacologic resuscitation without any other fluid.
The mechanisms by which PAD inhibition attenuates ALI and system inflammation remain to be determined. PADs are calcium-dependent enzymes that catalyze the conversion of peptidylarginine to peptidylcitrulline. Protein citrullination leads to drastic changes in protein charge, structure and function. Of all the PADs, PAD4 is the best-characterized isozyme, which is localized primarily in the nucleus and contains a nuclear localization signal sequence at its N-terminus. PAD4 citrullinates a range of nuclear proteins, such as histones H2A, H3 and H4, ING4, p300/CBP, nucleophosmin, and nuclear lamin C, thereby playing an important role in nuclear functions and the regulation of gene expression [21]. PAD4 has been shown to play a putative role in a variety of pathogenic states, including autoimmune diseases and inflammatory conditions, such as rheumatoid arthritis (RA), multiple sclerosis (MS), ulcerative colitis (UC), systemic lupus erythematosus (SLE), sepsis, and thrombosis [22]. In the present study, we have not investigated changes in PAD activity or protein citrullination in severe HS condition, but evidence from several studies has demonstrated that PADs, especially PAD4 activity and protein citrullination, is influenced by ischemia or ischemia/reperfusion(I/R) conditions. For example, Alexander et al [7] used a mouse model of myocardial ischemia/reperfusion (MI/R) injury and reported that MI/R caused an increase in citrullinated histone H3 at the site of injury. PAD4 deficiency reduced leukocyte recruitment to the infarcted myocardium and prevented nuclear histone citrullination during MI/R injury. Importantly, lack of PAD4 had a cardioprotective effect in the mouse model of acute myocardial infarction (AMI). Pharmacological inhibition of PADs by pan-PAD inhibitor Cl-amidine dramatically reduces neuronal loss following hypoxia and injury volume, indicating that protein citrullination plays an important role in neuronal damage after ischemia insult [16]. A recent study has also reported a robust increase in PAD4 activity following renal I/R, and PAD4 inhibition protects against ischemic acute kidney injury by reducing the inflammatory response[6]. The mechanisms underlying the pathogenesis of severe HS appear to be associated with global ischemia and reperfusion. Thus, it is reasonable to assume that PADs activity and protein citrullination would be exaggerated following HS, which could contribute to systemic inflammation and organ failure. Also PAD4 is known to be activated by ROS, TNF-α, chemokines , H2O2 and lipopolysaccharides (LPS) [22]. We have recently demonstrated that citrullination of histone H3 is only detected in endotoxic shock but not in HS, suggesting that different stimuli inducing the activation of PAD4 may cause different protein citrullination patterns [23].
The lead compound YW3-56 is a novel PADs inhibitor. When compared with Cl-amidine, YW3-56 demonstrated a 5-fold increase in PAD4 inhibition (IC50 about 1–5μM) [24]. Our data indicate that PAD inhibition by YW3-56 may be useful as a therapeutic strategy to attenuate the complication and severity of HS.
This study has certain limitations that must be acknowledged. For logistic reasons, we only measured cytokine expression at selected time points. Similarly, we only studied lung injury in the present project. To further confirm the protective effects of YW3-56, the more clinically relevant “post- treatment” cell culture model and a larger animal model of severe HS should be engaged. The fundamental molecular and cellular signaling events involved in the protective role of PAD inhibition still require further investigation. YW3-56, as a novel PAD inhibitor, can prevent protein citrullination. However, the protein citrullination status, of histones especially, in trauma and severe HS remains unknown. The exact histone posttranslational modification patterns in trauma and other forms of severe shock condition are currently under investigation in our lab.
5. Conclusions
In conclusion, we have demonstrated that YW3-56, a novel PADs inhibitor, can improve survival in a rat model of hemorrhagic shock as well as in a cell culture model of H/R. The survival advantage is associated with an attenuation of local and systemic proinflammatory cytokines and the protection against ALI following severe hemorrhage. Thus, PAD inhibition may represent a novel and promising therapeutic strategy for severe HS.
Acknowledgement
Dr. Alam would like to acknowledge research support provided by National Institutes of Health (R01GM084127).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
This article presented orally at the 10th Annual Academic Surgical Congress on 2/5/2015, Las Vegas, NV Authors’ contributions: Study design (W.H., Y.L., and H.B.A.), conducting experiments (W.H., Z.C., P.Z., and B.L.), data collection/analysis (W.H., Z.C., and P.Z.), writing the article (W.H. and Y.L.), statistical analysis (W.H. and X.L.), data review (Y.M.), critical revision of the article (Y.L. and H.B.A.).
Disclosure
The authors report no proprietary or commercial interest in any product mentioned or concept discussed in this article.
REFERENCES
- 1.Cai B, Deitch EA, Ulloa L. Novel insights for systemic inflammation in sepsis and hemorrhage. Mediators of inflammation. 2010;2010:642462. doi: 10.1155/2010/642462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Yao YM, Redl H, Bahrami S, Schlag G. The inflammatory basis of trauma/shock-associated multiple organ failure. Inflammation research : official journal of the European Histamine Research Society ... [et al.] 1998;47:201–210. doi: 10.1007/s000110050318. [DOI] [PubMed] [Google Scholar]
- 3.Li Y, Alam HB. Modulation of acetylation: creating a pro-survival and anti-inflammatory phenotype in lethal hemorrhagic and septic shock. Journal of biomedicine & biotechnology. 2011;2011:523481. doi: 10.1155/2011/523481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Vossenaar ER, Zendman AJ, van Venrooij WJ, Pruijn GJ. PAD, a growing family of citrullinating enzymes: genes, features and involvement in disease. BioEssays : news and reviews in molecular, cellular and developmental biology. 2003;25:1106–1118. doi: 10.1002/bies.10357. [DOI] [PubMed] [Google Scholar]
- 5.Anzilotti C, Pratesi F, Tommasi C, Migliorini P. Peptidylarginine deiminase 4 and citrullination in health and disease. Autoimmunity reviews. 2010;9:158–160. doi: 10.1016/j.autrev.2009.06.002. [DOI] [PubMed] [Google Scholar]
- 6.Ham A, Rabadi MM, Kim M, Brown KM, Ma Z, et al. Peptidyl arginine deiminase-4 activation exacerbates kidney ischemia reperfusion injury. American journal of physiology. Renal physiology. 2014 doi: 10.1152/ajprenal.00243.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Savchenko AS, Borissoff JI, Martinod K, De Meyer SF, Gallant M, et al. VWF-mediated leukocyte recruitment with chromatin decondensation by PAD4 increases myocardial ischemia/reperfusion injury in mice. Blood. 2014;123:141–148. doi: 10.1182/blood-2013-07-514992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Li Y, Liu Z, Liu B, Zhao T, Chong W, et al. Citrullinated histone H3: a novel target for the treatment of sepsis. Surgery. 2014;156:229–234. doi: 10.1016/j.surg.2014.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zampetaki A, Mitsialis SA, Pfeilschifter J, Kourembanas S. Hypoxia induces macrophage inflammatory protein-2 (MIP-2) gene expression in murine macrophages via NF-kappaB: the prominent role of p42/ p44 and PI3 kinase pathways. FASEB J. 2004;18:1090–1092. doi: 10.1096/fj.03-0991fje. [DOI] [PubMed] [Google Scholar]
- 10.Hwabejire JO, Lu J, Liu B, Li Y, Halaweish I, et al. Valproic acid for the treatment of hemorrhagic shock: a dose-optimization study. The Journal of surgical research. 2014;186(1):363–370. doi: 10.1016/j.jss.2013.09.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Fukudome EY, 1, Kochanek AR, Li Y, Smith EJ, Liu B, Kheirbek T, Lu J, Kim K, Hamwi K, Velmahos GC, Alam HB. Pharmacologic resuscitation promotes survival and attenuates hemorrhage-induced activation of extracellular signal-regulated kinase 1/2. J Surg Res. 2010;163(1):118–126. doi: 10.1016/j.jss.2010.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Mohanan S, Horibata S, McElwee JL, Dannenberg AJ, Coonrod SA. Identification of macrophage extracellular trap-like structures in mammary gland adipose tissue: a preliminary study. Frontiers in immunology. 2013;4:67. doi: 10.3389/fimmu.2013.00067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Fan LY, He DY, Wang Q, Zong M, Zhang H, et al. Citrullinated vimentin stimulates proliferation, pro-inflammatory cytokine secretion, and PADI4 and RANKL expression of fibroblast-like synoviocytes in rheumatoid arthritis. Scandinavian journal of rheumatology. 2012;41:354–358. doi: 10.3109/03009742.2012.670263. [DOI] [PubMed] [Google Scholar]
- 14.Asaga H, Yamada M, Senshu T. Selective deimination of vimentin in calcium ionophore-induced apoptosis of mouse peritoneal macrophages. Biochemical and biophysical research communications. 1998;243:641–646. doi: 10.1006/bbrc.1998.8148. [DOI] [PubMed] [Google Scholar]
- 15.U KP, Subramanian V, Nicholas AP, Thompson PR, Ferretti P. Modulation of calcium-induced cell death in human neural stem cells by the novel peptidylarginine deiminase-AIF pathway. Biochimica et biophysica acta. 2014;1843:1162–1171. doi: 10.1016/j.bbamcr.2014.02.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lange S, Rocha-Ferreira E, Thei L, Mawjee P, Bennett K, et al. Peptidylarginine deiminases: novel drug targets for prevention of neuronal damage following hypoxic ischemic insult (HI) in neonates. Journal of neurochemistry. 2014;130:555–562. doi: 10.1111/jnc.12744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Fukudome EY, Li Y, Kochanek AR, Lu J, Smith EJ, et al. Pharmacologic resuscitation decreases circulating cytokine-induced neutrophil chemoattractant-1 levels and attenuates hemorrhage-induced acute lung injury. Surgery. 2012;152:254–261. doi: 10.1016/j.surg.2012.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Everhart MB, Han W, Sherrill TP, Arutiunov M, Polosukhin VV, et al. Duration and intensity of NF-kappaB activity determine the severity of endotoxin-induced acute lung injury. Journal of immunology (Baltimore, Md. : 1950) 2006;176:4995–5005. doi: 10.4049/jimmunol.176.8.4995. [DOI] [PubMed] [Google Scholar]
- 19.Dekker SE, Sillesen M, Bambakidis T, Jin G, Liu B, et al. Normal saline influences coagulation and endothelial function after traumatic brain injury and hemorrhagic shock in pigs. Surgery. 2014;156:556–563. doi: 10.1016/j.surg.2014.04.016. [DOI] [PubMed] [Google Scholar]
- 20.Li Y, Xiang M, Yuan Y, Xiao G, Zhang J, et al. Hemorrhagic shock augments lung endothelial cell activation: role of temporal alterations of TLR4 and TLR2. American journal of physiology. Regulatory, integrative and comparative physiology. 2009;297:R1670–1680. doi: 10.1152/ajpregu.00445.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bicker KL, Thompson PR. The protein arginine deiminases: Structure, function, inhibition, and disease. Biopolymers. 2013;99:155–163. doi: 10.1002/bip.22127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Rohrbach AS, Slade DJ, Thompson PR, Mowen KA. Activation of PAD4 in NET formation. Frontiers in immunology. 2012;3:360. doi: 10.3389/fimmu.2012.00360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Li Y, Liu B, Fukudome EY, Lu J, Chong W, et al. Identification of citrullinated histone H3 as a potential serum protein biomarker in a lethal model of lipopolysaccharide-induced shock. Surgery. 2011;150:442–451. doi: 10.1016/j.surg.2011.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wang Y, Li P, Wang S, Hu J, Chen XA, et al. Anticancer peptidylarginine deiminase (PAD) inhibitors regulate the autophagy flux and the mammalian target of rapamycin complex 1 activity. The Journal of biological chemistry. 2012;287:25941–25953. doi: 10.1074/jbc.M112.375725. [DOI] [PMC free article] [PubMed] [Google Scholar]