Abstract
Trophoblast stem cells (TSCs) represent the multipotent progenitors that give rise to the different cells of the embryonic portion of the placenta. Here, we analysed the expression of key TSC transcription factors Cdx2, Eomes, and Elf5 in the early developing placenta of mouse embryos and in cultured TSCs and reveal surprising heterogeneity in protein levels. We analysed persistence of TSCs in the early placenta and find that TSCs remain in the chorionic hinge until E9.5 and are lost shortly afterwards. To define the transcriptional signature of bona fide TSCs, we used inducible gain- and loss-of-function alleles of Eomes or Cdx2, and Eomes GFP, to manipulate and monitor the core maintenance factors of TSCs, followed by genome-wide expression profiling. Combinatorial analysis of resulting expression profiles allowed for defining novel TSC marker genes that might functionally contribute to the maintenance of the TSC state. Analyses by qRT-PCR and in situ hybridisation validated novel TSC- and chorion-specific marker genes, such as Bok/Mtd, Cldn26, Duox2, Duoxa2, Nr0b1, and Sox21. Thus, these expression data provide a valuable resource for the transcriptional signature of bona fide and early differentiating TSCs and may contribute to an increased understanding of the transcriptional circuitries that maintain and/or establish stemness of TSCs.
1. Introduction
Trophectoderm (TE) and inner cell mass (ICM) are the first cell lineages that are specified at the 16- to 32-cell (morula) stage around embryonic day 3 (E3.0) of mouse development (reviewed in [1, 2]). Mutual negative feedback inhibition of the two key transcription factors Cdx2 and Oct4 establishes the lineages of Cdx2-expressing TE and Oct4-positive ICM cells [3–6]. Multiple signals, including cell polarity, cell-cell contacts, positional information, and Hippo signalling, converge on the phosphorylation state of Yes-associated protein (YAP). YAP forms a transcriptional complex with TEAD4 to activate Cdx2 transcription in predominantly outer, peripheral cells of the early embryo, thus priming the TE programme [4–13]. Additional layers of regulation ensure lineage restricted Cdx2 and Oct4 expression, such as Notch-signalling, which cooperates with TEAD4 to directly regulate Cdx2 expression [14]. Similarly, the transcription factor AP2γ promotes Cdx2 expression and downregulates Hippo signalling [15]. GATA3 acts in parallel with CDX2 and downstream of TEAD4 to induce an overlapping set of target genes in the trophoblast lineage [16]. Within the polar TE, which is defined by its vicinity to the ICM at the blastocyst stage (E3.5), a population of trophoblast stem cells (TSCs) is established and maintained by ICM-derived Fgf- and Nodal-signals, to generate the main cellular source for formation of the embryonic part of the placenta [13, 17–20]. Following initial specification of TE fate by Cdx2-functions, additional transcriptional regulators, including Eomes [4] and Elf5 [21], initiate expression in the TE lineage or become specifically restricted to TE cells such as AP2γ [22, 23]. Following implantation around E4.5, the polar TE gives rise to the extraembryonic ectoderm (ExE), which contains TSCs, and the ectoplacental cone (EPC), which mediates the embryonic invasion into the decidual wall then connects the embryo to the maternal uterus. TSCs retain their stem cell characteristics, namely, self-renewal capacity and multipotency, only in the polar TE and the ExE while cells of the mural TE lack supportive signalling from the ICM and thus differentiate to primary trophoblast giant cells (TGCs) [17]. TSC maintenance is controlled by transcriptional circuitries involving Elf5 [24, 25], Ets2 [26], and AP2γ [23, 27], which act in positive feed forward loops to maintain Cdx2 and/or Eomes expression. After E7.5, the core set of TSC marker genes, including Cdx2, Eomes, Elf5, and Esrrβ, remain expressed in the chorion in addition to other embryonic and extraembryonic expression domains [24, 28–30].
TSCs can be routinely isolated and cultured from E3.5 blastocysts and E6.5 ExE explants under defined conditions [17]. Assessing the potential to isolate cells with TSC character beyond E6.5, Uy and colleagues have shown that the frequency to obtain TSC clones increases until the 4-somite pair stage (~E8.0 to E8.25) but then rapidly drops and no TSCs can be isolated following the 11-somite pair stage around E8.5 to E8.75 [31]. The T-box transcription factor Eomes was previously used to mark TSCs through different developmental stages and expression of an Eomes::GFP BAC transgene was detected until E14.5 in the outer periphery of the murine placenta [32]. To date it was not clearly shown until which embryonic stage cells with TSC character are maintained in the TSC niche, which is functionally defined by high levels of Fgf and Tgfβ signals [17, 33]. In addition to Eomes [34] and Cdx2 [35], also other transcription factors share essential functions for TSC self-renewal and multipotency including Elf5 [24], Esrrβ [29], Ets2 [26], AP2γ [23, 27, 36], and Sox2 [37]. A recent report demonstrated that the functional loss of the histone demethylase Lsd1 in TSCs results in premature migration of TSCs from their niche, demonstrating additional requirements of proper epigenetic regulation for the propagation of TSCs [38, 39].
In addition to their functional importance during TSC maintenance, Cdx2 [3, 11], Eomes [3], Elf5 [25], Tead4 [11], AP2γ [23], and Gata3 [16] also evoke lineage conversion from mouse embryonic stem (mES) cells to the TE lineage when overexpressed (reviewed in [40]). While the transcriptional programme involving Cdx2, Eomes, Elf5, AP2γ, and Ets2 is key for the maintenance of the trophoblast stemness state, genetic deletions of each of these transcription factors generate remarkable different embryonic phenotypes. Cdx2-deficiency results in implantation defects [41], Eomes-deficient embryos show early postimplantation [34], and the deletions of Elf5 [24], Ets2 [26, 42, 43], or AP2γ [36] lead to postgastrulation lethality. This diversity in loss-of-function phenotypes might indicate differential requirements of these factors for the stemness maintaining regulatory circuitry. Alternatively, different states of TSCs might exist with differential developmental potential and distinct requirements of transcriptional regulation, in analogy to different states of pluripotency, such as that found in naïve or primed pluripotent stem cells [39, 44].
In the current study, we have analysed the endogenous expression of key stemness-maintaining factors, Cdx2, Eomes, and Elf5, in the chorion of gastrulation stage embryos to characterise TSCs by overlapping marker gene expression. We traced Eomes expression through later gastrulation stages and demonstrate that it is lost around E9.5 in the region emanating from the chorionic hinge. Surprisingly, TSC markers consistently show heterogenous, partially nonoverlapping expression in different areas of the late TSC niche, potentially indicating different states of TSCs during placental development. The analysis of TSCs cultured under stemness-maintaining conditions additionally revealed heterogeneous TSC marker expression in vitro, underscoring our findings from embryonic analyses in the chorion. To determine the transcriptional state that describes undifferentiated, bona fide TSCs and to monitor transcriptional changes during early TSC differentiation, we used genetic tools to manipulate Eomes and Cdx2 expression in TSC cultures followed by genome wide transcriptional profiling. First, we generated TSCs that harbour an Eomes GFP reporter allele, thereby marking bona fide TSCs that were enriched by fluorescence activated cell sorting (FACS) and forced towards differentiation by removal of stemness maintaining conditions. Second, we employed TSCs that allow for the inducible deletion of Eomes gene function, and third we used inducible expression of key TSC transcriptional regulators Cdx2 and Eomes in mouse ES (mES) cells for the identification of downstream target genes. Resulting differential expression profiles allow for a detailed description of the TSC signature and the changes during early differentiation, which might be directly or indirectly regulated by a combination of Fgf- and Tgfβ-signalling and the key regulatory factors of TSCs, Cdx2, and Eomes. We used resulting expression profiles to identify novel candidate TSC marker genes at stemness state. To validate this approach, a handful of differentially expressed genes were selected and tested by qRT-PCRs of bona fide TSCs and during early differentiation. Additionally, expression of novel candidates was analysed in the TSC compartment of postimplantation embryos by in situ hybridisation analysis. In summary, this study characterises and describes the expression signature of TSCs within the embryo and in cultured TSCs.
2. Materials and Methods
2.1. Cell Culture
Genetically modified TSCs were isolated from E3.5 blastocysts of animals carrying alleles for Eomes GFP [45], Eomes CA [47], and R26CreER [58] according to standard protocols [17]. TSCs cultured in stemness maintaining conditions (SMC) were kept in 70% mouse embryonic fibroblast- (MEF-) conditioned TSC-medium (MCM) supplemented with hrFGF4 and heparin (F4H) on MEFs [17]. For differentiation conditions (DC), TSCs were cultured in TSC-medium without MCM and F4H on gelatinized plates without feeder cells [17]. Cre-mediated excision of the conditional Eomes CA allele was induced by administration of 1 μg/mL 4-hydroxytamoxifen (Sigma Aldrich, H7904) to the culture medium for up to 3 days.
To generate mES cells that inducibly express Cdx2 or Eomes in response to doxycycline administration, we performed inducible cassette exchange (ICE) using A2lox.cre mES cells [59] and the p2lox-V5 vector system using gateway cloning (Invitrogen) [48]. Expression was induced by administration of 1 μg/mL doxycycline (Fargon, 137087) to the ES cell culture medium composed of DMEM (Gibco, 11960), 15% ES cell-qualified FBS (Gibco, 16141), 2 mM L-glutamine (Gibco, 25030), 50 U/mL penicillin/50 μg/mL streptomycin (Gibco, 15070), 0.1 mM nonessential amino acids (Gibco, 11140), 1 mM sodium pyruvate (Gibco, 11360), 0.1 mM β-mercaptoethanol (Sigma Aldrich, M7522), and 1,000 U/mL leukemia inhibitory factor (Merck Millipore, ESG1107).
2.2. Flow Cytometry
Single cell suspensions of Eomes GFP TSCs cultured in SMC were FACS-purified for GFPhigh cells using a MoFlo Legacy cell sorter (Beckman Coulter). Selected cells were reseeded in SMC without MEF feeder cells for 24 h before preparation of the RNA.
2.3. Immunofluorescence Staining of E7.5–E14.5 Tissue Sections
Whole decidua (E6.5–E8.5) or placenta (E10.5–E14.5) was isolated from Eomes GFP pregnant females on ice, washed in PBS, and fixed in 4% PFA at 4°C for 1 h (E6.5, E7.5) or 3 h (E8.5–E14.5). Fixed tissues were rinsed in PBS and successively transferred to 15% and 30% sucrose dissolved in PBS until samples no longer floated. Tissues were subsequently incubated in embedding medium (7.5% fish skin gelatin, 15% sucrose) for 1 h at 37°C and snap frozen in liquid nitrogen. Frozen blocks were cut at 8 μm sections on a cryostat (CM3050s, Leica), and sections mounted on SuperFrost Plus slides (R. Langenbrinck, 03-0060). Washing steps, blocking, incubation with primary and secondary antibodies, and DAPI staining were performed as described for immunofluorescence on TSCs.
2.4. Immunofluorescence Staining on TSCs
TSC cultures were grown on coverslips coated with 0.1% gelatin, briefly rinsed in PBS, and fixed in 4% PFA for 30 min on ice. Fixed cells were washed with PBS containing 0.1% Tween-20 (PBS-T), blocked for 1 h in PBS-T with 5% bovine serum albumin (BSA-T), and incubated with primary antibodies (Supplementary Table S1 available online at http://dx.doi.org/10.1155/2015/218518) in 5% BSA-T over night at 4°C. After washing off the primary antibodies, samples were incubated for 45 min at RT with conjugated secondary antibodies (Supplementary Table S1) diluted in 5% BSA-T. Cells were counterstained for 10 min at RT with DAPI diluted 1 : 1000 in PBS and mounted with Fluoromount-G. Fluorescent images were captured on a Zeiss Axiovert 200 M, equipped with a 20x Plan-Apochromat objective. Displayed images were eventually merged and brightness adjusted using Adobe Photoshop CS5 software.
2.5. RNA Preparation for Microarray and Quantitative RT-PCR
Total RNA was purified from TSC and ES cell culture dishes using the RNeasy Mini Kit (QIAGEN, 74104) using 350 μL RLT lysis buffer per 6 cm culture dish. On-column DNaseI digestion was applied and RNA eluted in 50 μL RNase free water, yielding concentrations of 179–1.686 ng/μL RNA.
2.6. Microarray and Analysis of Microarray Data
To identify genes overrepresented in TSCs, gene expression datasets obtained from the microarray were sorted for highest negative (Eomes GFP TSC differentiation and Eomes CA) or positive (induced Eomes or Cdx2 expression) fold changes. Resulting lists from induced Eomes deletion or induced Cdx2 or Eomes expression were additionally filtered for genes that were regulated at least 1.5-fold when compared to respective controls. Only genes with p values ≤ 0.05 were considered for further analyses. Finally, median expression values of mES cell- and TSC-based experimental subsets were calculated for each gene. Gene lists were compared by Venn diagrams using the Manteia data mining system (http://manteia.igbmc.fr/).
2.7. Validation by Quantitative RT-PCR and Statistical Analyses
For reverse transcription, the QIAGEN QuantiTect Reverse Transcription Kit (205311) was used according to the protocol with an input of 1.0 μg RNA per sample. Quantitative PCR was carried out in triplicate from three independent experiments, using the Roche Light Cycler 480 (SN: 1126) detection system and the Light Cycler 480 DNA SYBR Green I Master kit (Roche, 04707516001) and the primers listed in Supplementary Table S2. 36B4 and βcat served as reference genes. mRNA expression levels over time were calculated relative to day 0 which was set to 1. Statistical analyses were performed using a two-sided Student's t-test and the standard error of the mean was calculated for each dataset consisting of biological triplicates in technical triplicates each, individually.
2.8. Validation by In Situ Hybridization
Deciduae containing embryos were dissected and fixed in 4% PFAin PBS, dehydrated through ethanol series, and embedded in paraffin before 8 μM sections were prepared on a Leica RM2165 microtome. In situ hybridization on paraffin sections was performed according to standard protocols [60] using Bok/Mtd-, Cldn26-, Cyp26a1-, Duox2-, Duoxa2-, Eomes-, Nr0b1-, and Sox21-specific probes. Eosin counterstaining was performed according to standard protocols [60].
2.9. Animals
All mice were housed in the pathogen-free barrier facility of the University Medical Centre Freiburg in accordance with the institutional guidelines and approval by the regional board.
3. Results
3.1. Bona Fide TSCs Are Marked by Coexpression of Eomes, Cdx2, and Elf5 and Are Lost from the Chorionic Hinge around E9.5
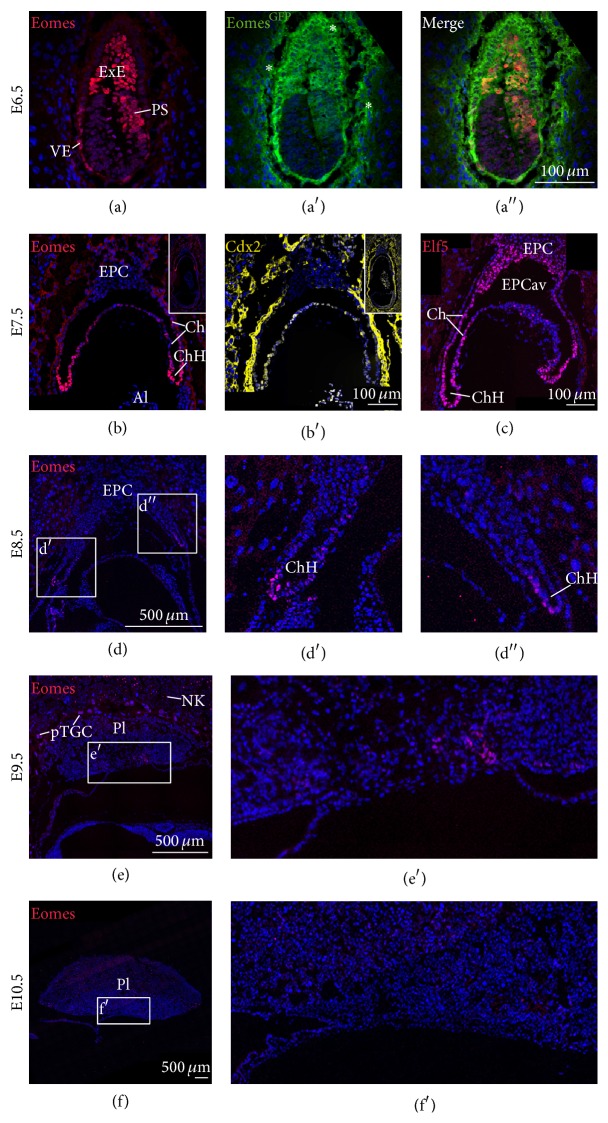
To investigate the spatial and temporal distribution of TSCs during placentogenesis, we performed detailed immunofluorescence (IF) staining using antibodies specific for EOMES, CDX2, and ELF5, in combination with a previously described Eomes GFP reporter allele [45]. EOMES protein and the GFP-reporter can be detected in the TSC compartment of the extraembryonic ectoderm (ExE), in the primitive streak (PS), and in the visceral endoderm (VE) during early gastrulation at E6.5 (Figures 1(a)–1(a′′)) [45]. One day later, at E7.5 Eomes expression is refined to a subregion of the chorion, which we refer to as the chorionic hinge (ChH), and expression is gradually reduced in the remainder of the chorion (Figure 1(b)). CDX2 colocalizes with EOMES within the chorion at E7.5 but, unlike EOMES, the staining intensity shows no gradual changes and is uniform throughout the entire chorion (Figure 1(b′)). CDX2 is additionally found in the extraembryonic mesoderm of the allantois (Figure 1(b′)). Finally, we stained for expression of Elf5, which acts in the transcriptional circuitry that maintains TSC stemness by positive feed-forward regulation of Eomes and Cdx2 expression [25]. Strongest ELF5 staining was detected in the ChH and the distal portion of the ectoplacental cone (EPC) facing the ectoplacental cavity while reduced levels are found throughout the remainder of the chorion and the proximal EPC (Figure 1(c)). At E8.5, EOMES expressing cells are remaining within the tip of the ChH, which is detached from the surrounding tissues, while the central part of the chorion has started to fuse with the allantois and the EPC (Figures 1(d)–1(d′′)). EOMES-positive cells are almost entirely lost around E9.5 from the forming placenta (Figures 1(e) and 1(e′)) with only little EOMES staining in a region emanating from the former ChH (Figure 1(e′)). Following E10.5 placental EOMES staining could not be detected, apart from single EOMES positive cells that most likely represent placental natural killer cells (Figures 1(e) and 1(f)) [46]. In summary, these detailed marker analyses suggest that EOMES-positive TSCs remain in the chorionic hinge as the functional stem cell niche of TSCs until E9.5 and are not found at later time points.
Figure 1.
EOMES colocalizes with TSC markers CDX2 and ELF5 in the E7.5 chorionic hinge and expression is lost around E9.5. Immunofluorescence staining shows nuclear EOMES and Eomes GFP reporter expression in (a–a′′) the visceral endoderm (VE), the primitive streak (PS), and the extraembryonic ectoderm (ExE) at E6.5, (b) within the chorion (Ch) and the chorionic hinge at E7.5, and (d–d′′) in the chorion at E8.5. Asterisks in (a′) indicates unspecific signals in extraembryonic tissues. (b and b′) TSC markers EOMES and CDX2 colocalize in cells of the chorion at E7.5. EOMES staining is most prominent in the chorionic hinge and shows a gradient along the chorion, while CDX2 does not show graded reduction along the chorion. (c) ELF5 similarly localizes to the chorion with strongest staining in the chorionic hinge and additionally expands into the ectoplacental cone (EPC) adjacent to the ectoplacental cavity (EPCav). (e, e′) Faint EOMES staining can be detected at E9.5 in the region emanating from the chorionic hinge, and (f, f′) staining is entirely lost at E10.5. (e) EOMES can be additionally found in parietal trophoblast giant cells (pTGC) and natural killer cells (NK) at E9.5. Al, allantois; EPC, ectoplacental cone; EPCav, ectoplacental cavity ExE, extraembryonic ectoderm; Ch, chorion; ChH, chorionic hinge; Pl, placenta; PS, primitive streak; pTGC, parietal trophoblast giant cells VE, visceral endoderm; NK, NK cells.
3.2. TSCs in Culture Show Heterogeneous Staining for Stemness Factors and Can Be Identified by High Levels of the Eomes GFP Reporter Expression
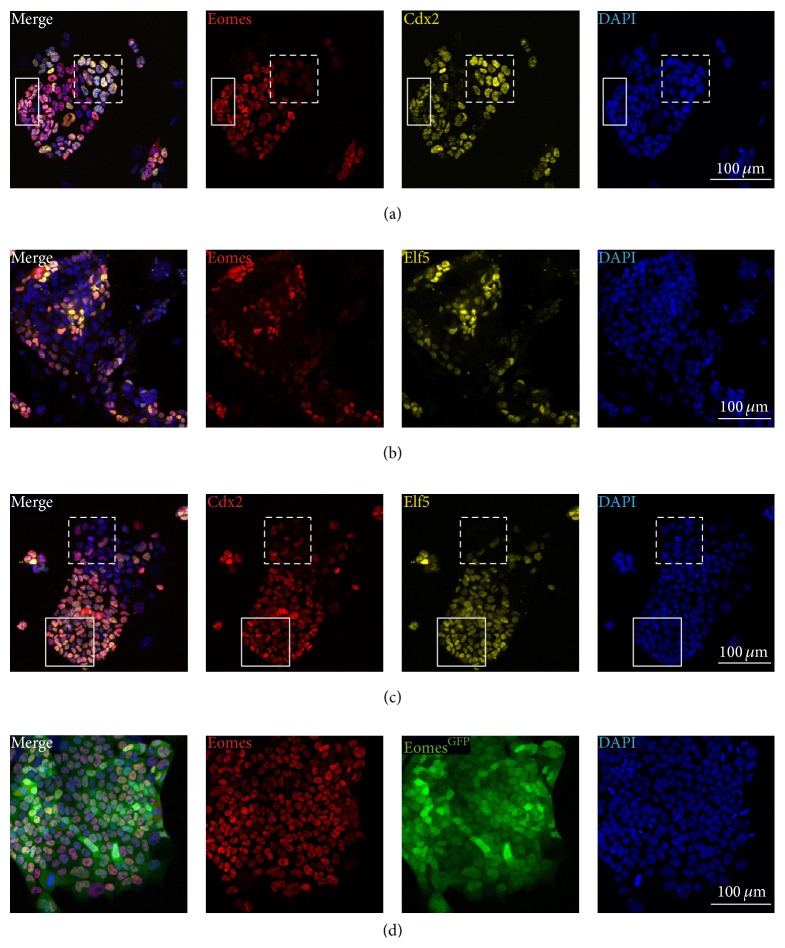
TSCs can be cultured under conditions of continuous stimulation with Fgf- and Tgfβ- growth factors for extended periods of time and multiple passages [17, 33]. However, TSCs in vitro exhibit an eminent tendency towards spontaneous differentiation and in stemness-maintaining conditions about 5–10% of TSCs differentiate towards the trophoblast giant cell fate, indicated early by an increase in nuclear and total cell size. TSC cultures thus intrinsically contain heterogeneous cell types to various degrees. To study the degree of heterogeneity in cultured TSCs, we first investigated the coexpression of TSC markers using antibodies against CDX2, EOMES, and ELF5 in TSCs cultured on feeder layers of mouse embryonic fibroblasts (MEF) in the presence of MEF-conditioned TSC-medium (MCM), human recombinant FGF4 (hrFGF4), and heparin (F4H) (Figures 2(a)–2(c)). Double-IF staining for EOMES and CDX2 showed a surprising degree of mutual staining. Predominantly, cells at the periphery of colonies showed strong CDX2 staining, while only very weak EOMES signal could be observed. These CDX2 positive cells exhibited a markedly enlarged nucleus in comparison to CDX2 and EOMES double positive cells, indicating that they may have differentiated from TSCs that normally present as cells with small nuclei and cell bodies. Colabelling for EOMES and CDX2 (Figure 1(a)), as well as EOMES and ELF5 (Figure 1(b)), and CDX2 and ELF5 (Figure 1(c)) was very consistently found in small cells, likely representing bona fide TSCs. In larger, more differentiated cells, the CDX2 signal is only partially overlapping with EOMES and ELF5 (Figures 1(a) and 1(c)), while ELF5- and EOMES-staining are predominantly overlapping in all cell types (Figure 1(b)).
Figure 2.
TSCs in culture exhibit heterogeneous patterns of EOMES, CDX2, and ELF5 staining which is recapitulated by Eomes GFP reporter expression. Cultured TSCs are colabelled with antibodies against EOMES, CDX2, and ELF5 or the Eomes GFP knock-in reporter allele. (a) EOMES staining and CDX2 staining largely overlap in TSCs of small size, which morphologically match undifferentiated TSCs (solid boxed region), but staining diverges in enlarged cells (dashed boxed region). (b) ELF5 and EOMES immunostaining widely labels the same cells, and accordingly (c) ELF5 and CDX2 do not colocalize in enlarged cells ((c), dashed boxed region). (d) EOMES protein levels and Eomes GFP reporter expression closely correlate and highest fluorescence intensities are found in cells with strongest EOMES staining.
To allow for the analysis of TSCs that harbour a stemness-indicating reporter gene, we generated TSCs from E3.5 blastocysts carrying the Eomes GFP knock-in allele [45]. To characterize resulting Eomes GFP TSCs, we compared GFP-reporter expression with α-EOMES antibody staining. Eomes GFP reporter and EOMES protein expression widely overlap and highest levels of EOMES protein staining were reflected by strongest reporter activity (Figure 2(d)). As an exception, cells undergoing mitosis loose nuclear EOMES staining, while cytoplasmic GFP expression remains clearly detectable. Highest levels of GFP-reporter expression were found in populations of small cells that morphologically qualify for bona fide TSCs. In summary, our marker analysis in cultured TSCs demonstrates that only the combination of multiple stemness markers allows for unambiguous identification of bona fide TSCs. However, the generation of TSCs carrying the Eomes GFP allele is a suitable tool for the isolation of pure populations of undifferentiated TSCs (GFPhigh) that are characterised by high-level expression of Eomes.
3.3. Variable Experimental Setups Delineate the Transcriptional Characteristics of Bona Fide TSCs
Eomes GFP TSCs allow for the enrichment of GFPhigh cells representing fully undifferentiated and thus bona fide TSCs. These should serve as a suitable source to delineate the transcriptional signature of TSC stemness. Eomes maintains TSCs in an undifferentiated state and the genetic deletion of Eomes prohibits TSC maintenance and induces differentiation [34, 41]. In reverse, the expression of Eomes or Cdx2 (and other factors) in mES cells initiates the transcriptional programme that induces lineage conversion towards the TSC lineage [3]. To define the transcriptional signature of TSCs in their bona fide stemness state downstream of Eomes and Cdx2, we thus employed three experimental settings followed by genomewide expression analyses (Figure 3). First, we enriched Eomes GFP TSCs for GFPhigh cells and compared cells under stemness maintaining culture conditions with cells that were differentiated by withdrawal of MCM, F4H, and MEFs by gene expression profiling at daily intervals over a period of three days (Figure 3(a)). In the second approach, we induced Cre-mediated gene deletion in TSCs that homozygously carry an Eomes conditional allele in combination with a tamoxifen- (Tx-) inducible Cre-estrogen receptor fusion (Eomes CA/CA; R26CreERT) [47] and monitored gene expression at 24 h intervals for three days (Figure 3(b)). The recombination efficiency was monitored by PCR (Figure 3(b′)) and EOMES levels by Western blot, showing that EOMES protein was almost absent for 24 h after Tx-administration (Figure 3(b′′)). Third, to identify genes that are positively regulated by Eomes or Cdx2, we generated mES cells with inducible overexpression of both transcription factors. mES cells with inducible expression were generated by site-specific introduction of cDNAs including an N-terminal fusion to a V5-tag [48] into the engineered doxycycline-inducible Hprt locus of p2lox mES cells using inducible cassette exchange (ICE) [49]. Resulting mES cells allow for temporally regulated and moderate overexpression levels of Eomes or Cdx2 in mES cells as monitored by Western blot (Figures 3(c) and 3(c′)). Expression profiles of mES cells were performed before doxycycline induction and at 24 and 48 h following induced expression to identify the early induced genes downstream of Eomes or Cdx2 that initiate lineage conversion of mES cells towards the TE lineage. All three experimental settings were subsequently used for gene array based transcriptional profiling (see Supplementary Table S3).
Figure 3.
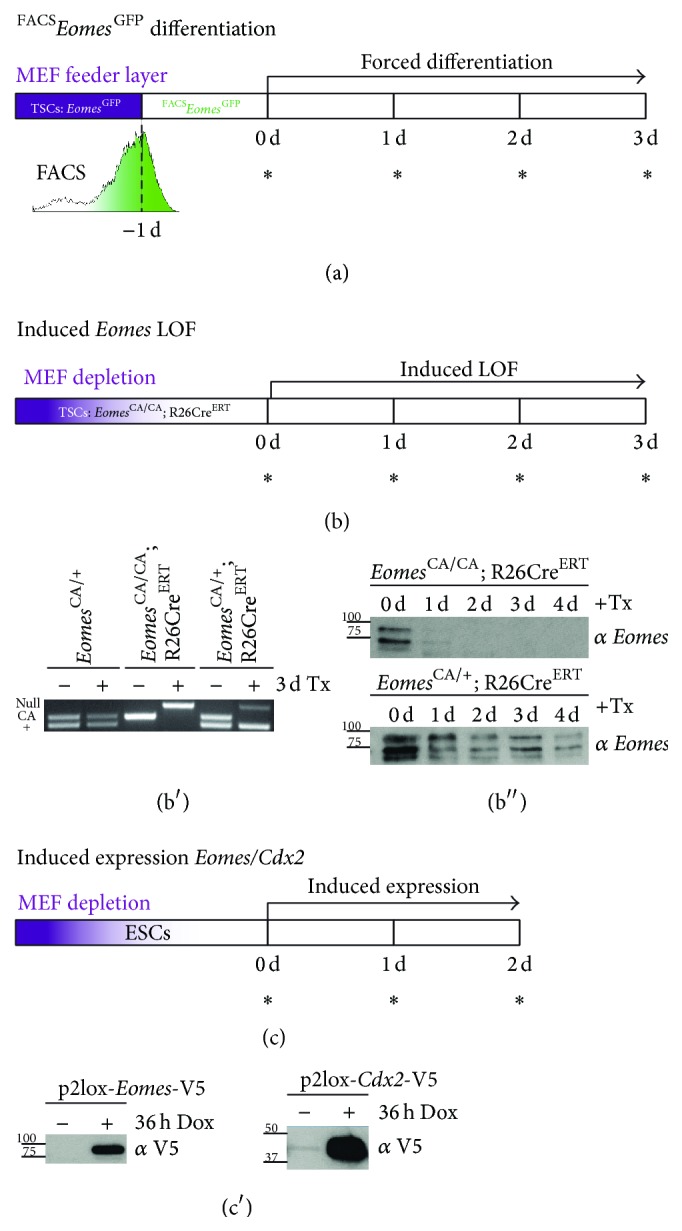
Experimental approaches for the detection and manipulation of the core TSC maintenance factors Eomes and Cdx2, followed by gene expression profiling. Three complementary experimental approaches were chosen for expression profiling of (a, b) TSCs and (c) mES cells with inducible expression of Eomes or Cdx2. (a) For the expression profiling of bona fide TSCs and early stages of differentiation, Eomes GFP TSCs were FACS-enriched for GFPhigh cells and forced towards differentiation by withdrawal of stemness maintaining conditions for 3d. (b) For the identification of Eomes-regulated target genes, TSCs harbouring Eomes conditional alleles (Eomes CA) in combination with a tamoxifen-inducible Cre-estrogen receptor allele (R26CreER) were used for conditional inactivation in vitro. Cells were tamoxifen-treated for 24 h and (b′) Cre-mediated excision was monitored by genotyping PCR, and (b′′) presence of EOMES protein was analysed by Western blot. (c) To transcriptionally profile the initiation of Eomes- and Cdx2-induced target genes during ES cell to TSC conversion, mES cells with doxycycline-regulated expression were induced for 48 h. (c′) The expression of V5-tagged EOMES or CDX2 protein was monitored by Western blot. MEF, mouse embryonic fibroblast; TSC, trophoblast stem cell; asterisks (∗) indicate the different time points of gene expression profiling by gene arrays.
3.4. Comparative Expression Profiling Reveals the Transcriptional Signature of TSCs
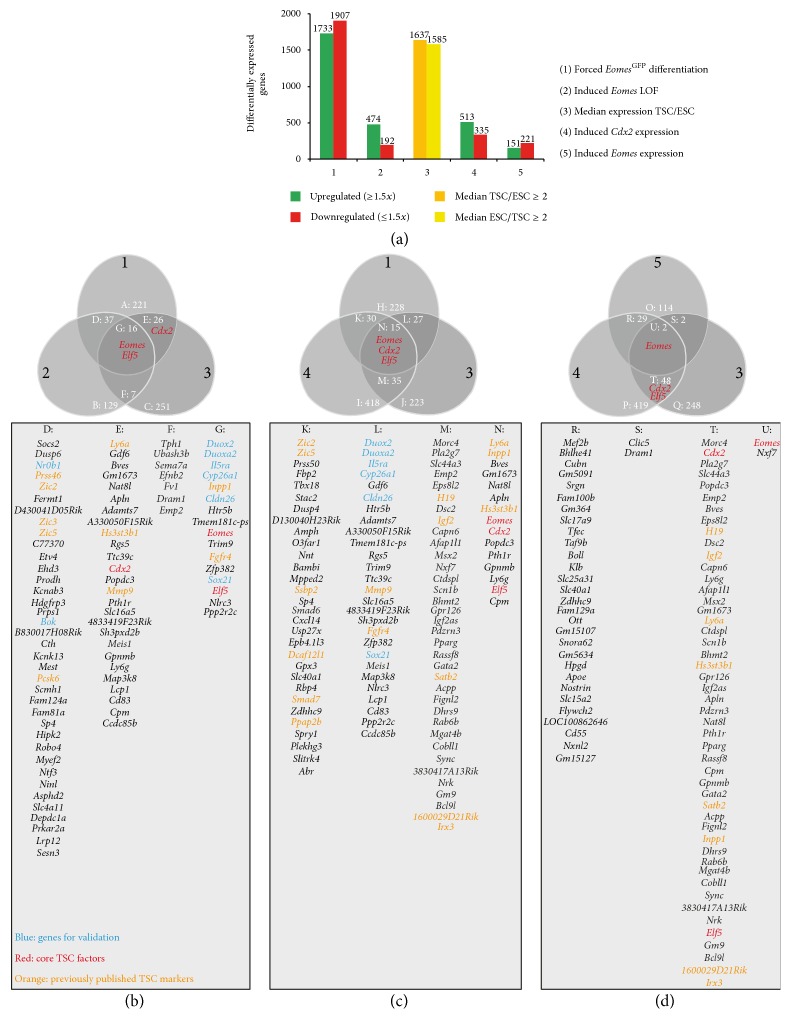
To reveal the transcriptional signature that constitutes and/or maintains a stable TSC state downstream of the key transcription factors Eomes and Cdx2, we performed a combined analysis of our independent gene array datasets and identified those genes that showed differential expression in multiple experiments (Figure 4 and Supplementary Table S4). All experimental interventions resulted in gross changes of differential gene expression that were defined by changes in expression above 1.5-fold with a p value below 0.05 in three biological replicates and a mean level of gene expression above a set background threshold. To identify genes that showed differential expression in multiple experiments, we analysed the intersections of genes that were among (1) the 300 most downregulated genes during differentiation of FACS-enriched EomesGFP-high TSCs, (2) the 189 genes that were downregulated following induced Eomes deletion, (3) the 300 genes that showed the highest relative expression in TSCs compared to mES cells as assessed by the ratio of median expression values in TSCs to the median expression values in mES cells, (4) the 498 genes that were upregulated in response to induced Cdx2 expression, and (5) the 147 genes that were upregulated in response to induced Eomes expression in mES cells (Figure 4 and Supplementary Table S4). The intersections of differentially expressed genes are represented in Venn diagrams and corresponding groups of genes are listed in tables (Figures 4(b)–4(d) and Supplementary Table S4).
Figure 4.
Comparative gene expression analysis reveals candidate TSC marker genes. (a) Genome-wide expression analysis generated five data sets of differentially expressed genes resulting from different experimental settings as shown in Figure 3. (1) The first group contains regulated genes after 3 days of differentiation of Eomes GFP-high FACS-enriched TSCs when compared to undifferentiated Eomes GFP-high TSCs. (2) The second group represents differentially regulated genes 3 days following Eomes gene deletion in TSCs (LOF, loss-of-function). (3) The third group comprises genes which show significantly increased or decreased median expression in TSCs versus mES cells, (4, 5) and the fourth and fifth group are differentially expressed genes 2 days after induced expression of Cdx2 or Eomes, respectively. The bar chart indicates numbers of differentially expressed genes for each gene group. (b–d) Venn diagrams indicate groups of intersecting, differentially expressed genes. The genes of resulting groups are listed in the table below each Venn diagram. (b) Comparison between the 300 most strongly downregulated genes in differentiating Eomes GFP FACS-enriched TSCs, the 189 genes downregulated following Eomes deletion, and the 300 genes with highest expression ratio between TSCs and mES cells. Of note, Cdx2, Eomes, and Elf5 expression levels decrease during TSC differentiation. The loss of Eomes function does not significantly reduce Cdx2 expression after 3d. In (c) the set of differentially regulated genes after Eomes gene deletion was exchanged with those 498 genes that were upregulated following Cdx2-induction in mES cells. In (d) genes that were induced by Eomes or Cdx2 expression in mES cells were compared to 300 genes with the highest expression ratio in TSCs versus mES cells. Note that Eomes is neither significantly upregulating Elf5 nor Cdx2 expression. Differences in number of genes listed in (a) and in (b–d) result from genes with multiple transcripts but identical gene symbols. The selected cut-off criteria for genes to be included in datasets were a positive or negative fold change ≥ 1.5 in response to treatment and a p value ≤ 0.05. Genes selected for further analyses are highlighted in blue, core genes of the TSC positive feedback loop in red, and genes that were previously identified as TSC markers in orange.
In a first comparative analysis, we investigated the intersection of those genes that were (1) downregulated in differentiating Eomes GFP-high TSCs, (2) downregulated in TSCs following induced deletion of Eomes, and (3) presented with a high relative expression in TSCs in comparison to mES cells (Figure 4(b)). 16 genes (group G) matched these criteria and thus were considered candidate Eomes target genes, which are lost during differentiation of TSCs. Within this group were the known specific TSC markers, Elf5, and Eomes itself. Additionally, this group contained novel genes that were not previously described in TSCs, such as Duox2 and Duoxa2 or Cldn26. Cdx2 was not included in the intersection, since the deletion of Eomes caused only a mild reduction of Cdx2 within the short time interval of the experiment.
Next, we compared genes that were (1) downregulated during differentiation of Eomes GFP-high TSCs, that were (4) positively regulated by Cdx2 in mES cells, and that were (3) TSC-specific (Figure 4(c)). The resulting group of 15 genes included Eomes, Elf5, and Cdx2 itself.
To compare the early transcriptional changes induced by Cdx2 or Eomes that initiate the conversion of mES cells to TSC-like cells, we analysed the intersecting TSC-specific genes (3) that were upregulated following two days of Cdx2 (4) or Eomes (5) induction (Figure 4(d)). Surprisingly, except for Eomes, none of the known TSC factors was significantly upregulated by both Cdx2 and Eomes after two days of induced expression. However, this might reflect the previous observations that Eomes is not as effective during the conversion of mES cells to TSCs in comparison to Cdx2. While Cdx2 expression initiates TSC-specific gene expression within 48 hours, including upregulation of Eomes and Elf5, Eomes expression fails to effectively induce TSC-specific genes within the 2-day interval, despite positive regulation of published Eomes targets, such as Fgf5, Mesp2, and Mixl1 [50, 51].
To validate the gene array data of differentially expressed genes, we performed quantitative RT-PCRs for established TSC marker genes and selected genes that were at intersections of differentially regulated genes in more than one experiment. To validate specific expression in bona fide TSCs, we compared expression levels in FACS-enriched Eomes GFP-high TSCs with levels during forced differentiation by removal of stemness maintaining conditions (Figure 5). All tested known and novel TSC marker genes showed highest expression in undifferentiated TSCs and significantly reduced levels following induction of differentiation with changes in expression over a minimum of two magnitudes (Figure 5).
Figure 5.
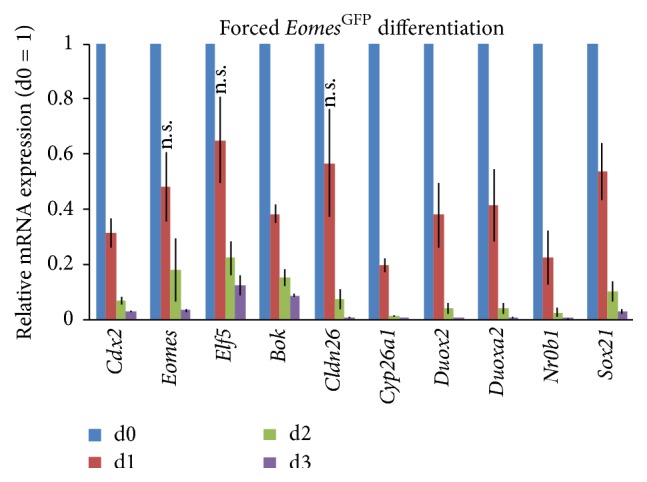
Genes identified by expression arrays and comparative analyses are downregulated during TSC differentiation when validated by qPCR. mRNA expression levels for genes selected by dataset comparisons were quantified at 24 h intervals by qRT-PCR of undifferentiated FACS-enriched Eomes GFP-high TSCs at day 0 (d0) and after forced differentiation for three days (d1–d3). Expression levels are depicted as means of biological triplicates relative to d0, which was set to 1. All genes were significantly (p value ≤ 0.05) downregulated at different time points unless indicated otherwise (n.s., not significant). Error bars indicate the standard error of the mean. p values were calculated according to two-sided Student's t-test. All marker genes show grossly reduced expression during the course of 3 days of differentiation.
3.5. Novel Marker Genes Label TSCs during Development of the Early Placenta
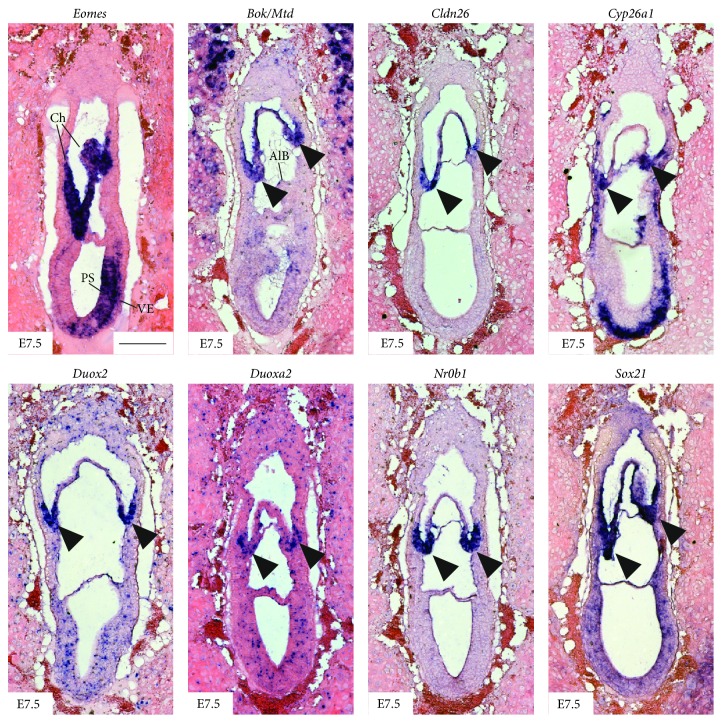
To generally validate if the approach revealed novel markers for TSCs during embryonic development, candidate genes were further analysed by in situ hybridisation using histological sections of E7.5 embryos. We focused on genes with previously unknown functions in the murine TE, such as Bok/Mtd, Duox2, Duoxa2, Nr0b1, and Sox21. To our knowledge, specific surface markers for TSCs that would allow for antibody-mediated FACS-sorting have not been identified so far. Thus, we additionally analysed expression of the transmembrane protein-coding gene Cldn26 (Tmem114). For Sox21, it was shown that it is involved in the regulation of intestinal and pluripotent stem cells and is induced by Sox2, an important stem cell factor in both mES cells and TSCs [52, 53]. Accordingly, Sox21 was also included into the following analysis. Expression of Cyp26a1 in the trophectoderm was previously described and together with Eomes served as a positive control for expression in the chorion. Of the seven tested novel marker genes, six showed expression in entire chorion (Bok, Cldn26, and Sox21) or were more specifically restricted to the chorionic hinge (Nr0b1, Duox2, and Duoxa2) (Figure 6).
Figure 6.
Differentially expressed genes show regional specific expression in the chorion of E7.5 embryos. In situ hybridisation analysis on sagittal sections of E7.5 mouse embryos reveals specific expression of Eomes as positive control and novel TSC marker genes Bok/Mtd, Cldn26, Cyp26a1, Duox2, Duoxa2, Nr0b1, and Sox21, in the entire chorion, or limited expression in the chorionic hinge (indicated by arrowheads). AlB, allantoic bud; Ch, chorion; PS, primitive streak; VE, visceral endoderm. Scale bar: 200 μM.
In conclusion, the in situ hybridisation analysis revealed that the multimodal expression profiling of TSCs in culture serves as a valuable resource for the identification of novel TSC marker genes in vitro, but also for TSCs of the developing placenta.
4. Discussion
In the present report we describe a resource for the transcriptional signature of TSCs. We used genetically modified TSCs and mES cells to experimentally interfere with stemness maintaining regulation of TSCs downstream of Eomes and Cdx2, followed by the assessment of transcriptional changes. The combined analysis of expression data resulted in a comprehensive list of candidate TSC marker genes, of which a handful were tested and positively validated for their specific expression in cultured undifferentiated TSCs and in the chorion of the gastrulating embryo. Thus, the presented expression data will serve as a valuable resource for further studies of stemness maintaining regulatory circuitries of TSCs.
Using TSCs that harbour the Eomes GFP reporter allele allowed for the purification of GFPhigh TSCs that by morphology and in their transcriptional signature likely resemble bona fide TSCs without contaminating early differentiating TSCs that are normally found in TSC cultures. Corresponding expression data of GFPhigh TSCs thus resemble the actual signature of bona fide TSCs, underscored by the rapid, gross, and early changes in gene expression that we found during their differentiation. Expression profiles of TSCs in stemness maintaining conditions and during induced differentiation, either by removal of MCM and F4H or by genetic interference with the TSC regulatory circuitry, were previously reported and had revealed partially overlapping gene lists [53–55]. However, the validation of candidates from our comparative expression analysis identified several additional and novel TSC marker genes. Among those is the transcription factor Sox21 that is strongly expressed in the chorion of the E7.5 embryo and is downregulated during differentiation of TSCs in culture. Interestingly, it was previously demonstrated that Sox21 is induced by Sox2, and, unlike Sox2 [53], Sox21 negatively regulates transcription of Cdx2 in mES and colon cancer cells [52]. This obvious difference in expression in mES cells and TSCs makes Sox21 an interesting candidate that, like Sox2 [53], might act differently in the circuitry of TSC and mES stemness factors. Functions of Sox21 and other novel candidates in TSC biology and TE development will be addressed in ongoing studies.
The importance of Cdx2 as lineage determining transcription factor was apparently demonstrated by the ability to convert mES cells to TSCs when overexpressed in mES cells [3], even though more recent studies suggest that this lineage conversion might not be complete at the phenotypic, transcriptional, and epigenetic level [56]. Eomes similarly has the capacity to induce TSC fate; however, Eomes seems less potent and possibly induces even less complete lineage conversion [3]. This notion is underscored by our datasets, which display more profound transcriptional responses following Cdx2 in comparison to Eomes expression, when induced from the identical doxycycline-inducible locus. This difference might, at least partially, arise from the reduced induction of Elf5 in Eomes-expressing mES cells which is known as a central component of a positive feed-forward regulation of the core TSC circuitry [25].
While both CDX2 and EOMES were previously used as markers for TSCs, coimmunofluorescence staining of cultured TSCs revealed a remarkable heterogeneity of labelling. Only a subset of TSCs that can be morphologically distinguished by a small cell and nucleus size consistently showed colabelling of CDX2 and EOMES, together with ELF5. We suspect that these cells resemble bona fide TSCs. It will require further fate analysis to reveal if the presence of individual key TSC factors primes future fate decisions, similar to lineage specifying roles of key pluripotency factors during embryonic development and in differentiating mES cells [50, 57]. It is noteworthy that EOMES is also detected in parietal trophoblast giant cells (pTGC) (Figure 1(e)) and thus it is tempting to speculate that differentiation towards the trophoblast giant cell lineage might be promoted by EOMES.
The immune-detection of EOMES, CDX2, and ELF5 in the chorion at late gastrulation stages revealed the presence of TSCs until E9.0–E9.5 in the remaining chorion, and the absence of EOMES-positive cells at later stages. These findings are in line with studies by Uy et al. that demonstrated the presence of TSCs that can be isolated and cultured until the 11-somite stage but not afterwards [31]. Another report that used an Eomes::GFP BAC-transgenic reporter [32] detected GFP positive cells in the margins of the E14.5 placenta. However, the nature of these cells was not defined in detail. We were unable to detect similar cells using EOMES-antibody staining. Thus, we conclude that TSCs are indeed only maintained until E9.5 in the remaining portions of the chorion from which they can be isolated as previously reported [31].
In summary, this study contributes to the characterisation of TSCs during early phases of placentogenesis and of TSCs in culture. We demonstrate that TSCs exhibit a remarkable degree of heterogeneity with respect to protein levels of key TSC transcription factors. TSCs are lost around E9.5 from their stem cell niche in the chorion. We used complementary genetic tools of cultured TSCs and ES cells to determine the transcriptional profile of TSCs in their fully undifferentiated and early differentiating state. Comparative analysis revealed several new TSC marker genes. Presented data can be used as a valuable resource for future studies of TSCs and the corresponding transcriptional regulatory network.
Supplementary Material
Supplementary Table S1: List of antibodies used for immunofluorescence and western blotting at indicated dilutions.
Supplementary Table S2: List of primers used for qPCR and to generate in situ riboprobes. Restriction sites attached to the primers to ensure directed cloning of the PCR product are underlined.
Supplementary Table S3: Gene expression array data sets.
Supplementary Table S4: Gene groups of Venn diagram intersections.
Acknowledgments
The authors thank Klaus Geiger for FACS of Eomes GFP cells, Marie Follo for supporting the qRT-PCR analysis, Michael Kyba for A2lox ES cells, Hynek Wichterle for the P2lox gateway cloning vector, and Gerd Walz for thoughtful discussion and generous support. This work was supported by an Exploration Grant from the Boehringer Ingelheim Foundation to Sebastian J. Arnold, a project grant (A03) of the Collaborative Research Centre 850 (SFB 850), and the Emmy Noether Programme (AR732/1-1) of the German Research Foundation (DFG) to Sebastian J. Arnold.
Conflict of Interests
The authors declare that there is no conflict of commercial interests.
Authors' Contribution
Sebastian J. Arnold conceived the study, and Georg Kuales and Sebastian J. Arnold performed experiments and analysed the data. Matthias Weiss and Oliver Sedelmeier assisted in and supervised some experiments. Dietmar Pfeifer performed microarray experiments and data analysis. Georg Kuales assembled the figures. Georg Kuales and Sebastian J. Arnold wrote and edited the paper.
References
- 1.Arnold S. J., Robertson E. J. Making a commitment: cell lineage allocation and axis patterning in the early mouse embryo. Nature Reviews Molecular Cell Biology. 2009;10(2):91–103. doi: 10.1038/nrm2618. [DOI] [PubMed] [Google Scholar]
- 2.Zernicka-Goetz M., Morris S. A., Bruce A. W. Making a firm decision: multifaceted regulation of cell fate in the early mouse embryo. Nature Reviews Genetics. 2009;10(7):467–477. doi: 10.1038/nrg2564. [DOI] [PubMed] [Google Scholar]
- 3.Niwa H., Toyooka Y., Shimosato D., et al. Interaction between Oct3/4 and Cdx2 determines trophectoderm differentiation. Cell. 2005;123(5):917–929. doi: 10.1016/j.cell.2005.08.040. [DOI] [PubMed] [Google Scholar]
- 4.Ralston A., Rossant J. Cdx2 acts downstream of cell polarization to cell-autonomously promote trophectoderm fate in the early mouse embryo. Developmental Biology. 2008;313(2):614–629. doi: 10.1016/j.ydbio.2007.10.054. [DOI] [PubMed] [Google Scholar]
- 5.Cockburn K., Rossant J. Making the blastocyst: lessons from the mouse. The Journal of Clinical Investigation. 2010;120(4):995–1003. doi: 10.1172/jci41229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gasperowicz M., Natale D. R. C. Establishing three blastocyst lineages-then what? Biology of Reproduction. 2011;84(4):621–630. doi: 10.1095/biolreprod.110.085209. [DOI] [PubMed] [Google Scholar]
- 7.Yagi R., Kohn M. J., Karavanova I., et al. Transcription factor TEAD4 specifies the trophectoderm lineage at the beginning of mammalian development. Development. 2007;134(21):3827–3836. doi: 10.1242/dev.010223. [DOI] [PubMed] [Google Scholar]
- 8.Zhao B., Wei X., Li W., et al. Inactivation of YAP oncoprotein by the Hippo pathway is involved in cell contact inhibition and tissue growth control. Genes and Development. 2007;21(21):2747–2761. doi: 10.1101/gad.1602907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ota M., Sasaki H. Mammalian Tead proteins regulate cell proliferation and contact inhibition as transcriptional mediators of Hippo signaling. Development. 2008;135(24):4059–4069. doi: 10.1242/dev.027151. [DOI] [PubMed] [Google Scholar]
- 10.Nishioka N., Yamamoto S., Kiyonari H., et al. Tead4 is required for specification of trophectoderm in pre-implantation mouse embryos. Mechanisms of Development. 2008;125(3-4):270–283. doi: 10.1016/j.mod.2007.11.002. [DOI] [PubMed] [Google Scholar]
- 11.Nishioka N., Inoue K.-I., Adachi K., et al. The Hippo signaling pathway components Lats and Yap pattern Tead4 activity to distinguish mouse trophectoderm from inner cell mass. Developmental Cell. 2009;16(3):398–410. doi: 10.1016/j.devcel.2009.02.003. [DOI] [PubMed] [Google Scholar]
- 12.Sasaki H. Mechanisms of trophectoderm fate specification in preimplantation mouse development. Development Growth and Differentiation. 2010;52(3):263–273. doi: 10.1111/j.1440-169x.2009.01158.x. [DOI] [PubMed] [Google Scholar]
- 13.Latos P. A., Hemberger M. Review: the transcriptional and signalling networks of mouse trophoblast stem cells. Placenta. 2014;35:S81–S85. doi: 10.1016/j.placenta.2013.10.013. [DOI] [PubMed] [Google Scholar]
- 14.Rayon T., Menchero S., Nieto A., et al. Notch and hippo converge on Cdx2 to specify the trophectoderm lineage in the mouse blastocyst. Developmental Cell. 2014;30(4):410–422. doi: 10.1016/j.devcel.2014.06.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Cao Z., Carey T. S., Ganguly A., Wilson C. A., Paul S., Knott J. G. Transcription factor AP-2 induces early Cdx2 expression and represses HIPPO signaling to specify the trophectoderm lineage. Development. 2015;142(9):1606–1615. doi: 10.1242/dev.120238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ralston A., Cox B. J., Nishioka N., et al. Gata3 regulates trophoblast development downstream of Tead4 and in parallel to Cdx2. Development. 2010;137(3):395–403. doi: 10.1242/dev.038828. [DOI] [PubMed] [Google Scholar]
- 17.Tanaka S., Kunath T., Hadjantonakis A.-K., Nagy A., Rossant J. Promotion to trophoblast stem cell proliferation by FGF4. Science. 1998;282(5396):2072–2075. doi: 10.1126/science.282.5396.2072. [DOI] [PubMed] [Google Scholar]
- 18.Guzman-Ayala M., Ben-Haim N., Beck S., Constam D. B. Nodal protein processing and fibroblast growth factor 4 synergize to maintain a trophoblast stem cell microenvironment. Proceedings of the National Academy of Sciences of the United States of America. 2004;101(44):15656–15660. doi: 10.1073/pnas.0405429101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Simmons D. G., Cross J. C. Determinants of trophoblast lineage and cell subtype specification in the mouse placenta. Developmental Biology. 2005;284(1):12–24. doi: 10.1016/j.ydbio.2005.05.010. [DOI] [PubMed] [Google Scholar]
- 20.Roberts R. M., Fisher S. J. Trophoblast stem cells. Biology of Reproduction. 2011;84(3):412–421. doi: 10.1095/biolreprod.110.088724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Pearton D. J., Broadhurst R., Donnison M., Pfeffer P. L. Elf5 regulation in the trophectoderm. Developmental Biology. 2011;360(2):343–350. doi: 10.1016/j.ydbio.2011.10.007. [DOI] [PubMed] [Google Scholar]
- 22.Winger Q., Huang J., Auman H. J., Lewandoski M., Williams T. Analysis of transcription factor AP-2 expression and function during mouse preimplantation development. Biology of Reproduction. 2006;75(3):324–333. doi: 10.1095/biolreprod.106.052407. [DOI] [PubMed] [Google Scholar]
- 23.Kuckenberg P., Buhl S., Woynecki T., et al. The transcription factor TCFAP2C/AP-2γ cooperates with CDX2 to maintain trophectoderm formation. Molecular and Cellular Biology. 2010;30(13):3310–3320. doi: 10.1128/mcb.01215-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Donnison M., Beaton A., Davey H. W., Broadhurst R., L'Huillier P., Pfeffer P. L. Loss of the extraembryonic ectoderm in Elf5 mutants leads to defects in embryonic patterning. Development. 2005;132(10):2299–2308. doi: 10.1242/dev.01819. [DOI] [PubMed] [Google Scholar]
- 25.Ng R. K., Dean W., Dawson C., et al. Epigenetic restriction of embryonic cell lineage fate by methylation of Elf5. Nature Cell Biology. 2008;10(11):1280–1290. doi: 10.1038/ncb1786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wen F., Tynan J. A., Cecena G., et al. Ets2 is required for trophoblast stem cell self-renewal. Developmental Biology. 2007;312(1):284–299. doi: 10.1016/j.ydbio.2007.09.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kuckenberg P., Kubaczka C., Schorle H. The role of transcription factor Tcfap2c/TFAP2C in trophectoderm development. Reproductive BioMedicine Online. 2012;25(1):12–20. doi: 10.1016/j.rbmo.2012.02.015. [DOI] [PubMed] [Google Scholar]
- 28.Beck F., Erler T., Russell A., James R. Expression of Cdx-2 in the mouse embryo and placenta: possible role in patterning of the extra-embryonic membranes. Developmental Dynamics. 1995;204(3):219–227. doi: 10.1002/aja.1002040302. [DOI] [PubMed] [Google Scholar]
- 29.Luo J., Sladek R., Bader J.-A., Matthyssen A., Rossant J., Giguère V. Placental abnormalities in mouse embryos lacking the orphan nuclear receptor ERR-β . Nature. 1997;388(6644):778–782. doi: 10.1038/42022. [DOI] [PubMed] [Google Scholar]
- 30.Ciruna B. G., Rossant J. Expression of the T-box gene Eomesodermin during early mouse development. Mechanisms of Development. 1999;81(1-2):199–203. doi: 10.1016/S0925-4773(98)00243-3. [DOI] [PubMed] [Google Scholar]
- 31.Uy G. D., Downs K. M., Gardner R. L. Inhibition of trophoblast stem cell potential in chorionic ectoderm coincides with occlusion of the ectoplacental cavity in the mouse. Development. 2002;129(16):3913–3924. doi: 10.1242/dev.129.16.3913. [DOI] [PubMed] [Google Scholar]
- 32.Kwon G. S., Hadjantonakis A.-K. Eomes:GFP—a tool for live imaging cells of the trophoblast, primitive streak, and telencephalon in the mouse embryo. Genesis. 2007;45(4):208–217. doi: 10.1002/dvg.20293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Erlebacher A., Price K. A., Glimcher L. H. Maintenance of mouse trophoblast stem cell proliferation by TGF-β/activin. Developmental Biology. 2004;275(1):158–169. doi: 10.1016/j.ydbio.2004.07.032. [DOI] [PubMed] [Google Scholar]
- 34.Russ A. P., Wattler S., Colledge W. H., et al. Eomesodermin is required for mouse trophoblast development and mesoderm formation. Nature. 2000;404(6773):95–99. doi: 10.1038/35003601. [DOI] [PubMed] [Google Scholar]
- 35.Chawiengsaksophak K., James R., Hammond V. E., Köntgen F., Beck F. Homeosis and intestinal tumours in Cdx2 mutant mice. Nature. 1997;386(6620):84–87. doi: 10.1038/386084a0. [DOI] [PubMed] [Google Scholar]
- 36.Auman H. J., Nottoli T., Lakiza O., Winger Q., Donaldson S., Williams T. Transcription factor AP-2gamma is essential in the extra-embryonic lineages for early postimplantation development. Development. 2002;129(11):2733–2747. doi: 10.1242/dev.129.11.2733. [DOI] [PubMed] [Google Scholar]
- 37.Avilion A. A., Nicolis S. K., Pevny L. H., Perez L., Vivian N., Lovell-Badge R. Multipotent cell lineages in early mouse development depend on SOX2 function. Genes and Development. 2003;17(1):126–140. doi: 10.1101/gad.224503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Zhu D., Hölz S., Metzger E., et al. Lysine-specific demethylase 1 regulates differentiation onset and migration of trophoblast stem cells. Nature Communications. 2014;5, article 3174 doi: 10.1038/ncomms4174. [DOI] [PubMed] [Google Scholar]
- 39.Hemberger M., Dean W., Reik W. Epigenetic dynamics of stem cells and cell lineage commitment: digging waddington's canal. Nature Reviews Molecular Cell Biology. 2009;10(8):526–537. doi: 10.1038/nrm2727. [DOI] [PubMed] [Google Scholar]
- 40.Roper S., Hemberger M. Defining pathways that enforce cell lineage specification in early development and stem cells. Cell Cycle. 2009;8(10):1515–1525. doi: 10.4161/cc.8.10.8381. [DOI] [PubMed] [Google Scholar]
- 41.Strumpf D., Mao C.-A., Yamanaka Y., et al. Cdx2 is required for correct cell fate specification and differentiation of trophectoderm in the mouse blastocyst. Development. 2005;132(9):2093–2102. doi: 10.1242/dev.01801. [DOI] [PubMed] [Google Scholar]
- 42.Yamamoto H., Flannery M. L., Kupriyanov S., et al. Defective trophoblast function in mice with a targeted mutation of Ets2. Genes and Development. 1998;12(9):1315–1326. doi: 10.1101/gad.12.9.1315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Georgiades P., Rossant J. Ets2 is necessary in trophoblast for normal embryonic anteroposterior axis development. Development. 2006;133(6):1059–1068. doi: 10.1242/dev.02277. [DOI] [PubMed] [Google Scholar]
- 44.Boroviak T., Nichols J. The birth of embryonic pluripotency. Philosophical Transactions of the Royal Society B: Biological Sciences. 2014;369(1657) doi: 10.1098/rstb.2013.0541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Arnold S. J., Sugnaseelan J., Groszer M., Srinivas S., Robertson E. J. Generation and analysis of a mouse line harboring GFP in the Eomes/Tbr2 locus. Genesis. 2009;47(11):775–781. doi: 10.1002/dvg.20562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Gordon S. M., Chaix J., Rupp L. J., et al. The transcription factors T-bet and Eomes control key checkpoints of natural killer cell maturation. Immunity. 2012;36(1):55–67. doi: 10.1016/j.immuni.2011.11.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Arnold S. J., Hofmann U. K., Bikoff E. K., Robertson E. J. Pivotal roles for eomesodermin during axis formation, epithelium-to-mesenchyme transition and endoderm specification in the mouse. Development. 2008;135(3):501–511. doi: 10.1242/dev.014357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Mazzoni E. O., Mahony S., Iacovino M., et al. Embryonic stem cell-based mapping of developmental transcriptional programs. Nature Methods. 2011;8(12):1056–1058. doi: 10.1038/nmeth.1775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kyba M., Perlingeiro R. C. R., Daley G. Q. HoxB4 confers definitive lymphoid-myeloid engraftment potential on embryonic stem cell and yolk sac hematopoietic progenitors. Cell. 2002;109(1):29–37. doi: 10.1016/S0092-8674(02)00680-3. [DOI] [PubMed] [Google Scholar]
- 50.Teo A. K. K., Arnold S. J., Trotter M. W. B., et al. Pluripotency factors regulate definitive endoderm specification through eomesodermin. Genes & Development. 2011;25(3):238–250. doi: 10.1101/gad.607311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Costello I., Pimeisl I.-M., Dräger S., Bikoff E. K., Robertson E. J., Arnold S. J. The T-box transcription factor Eomesodermin acts upstream of Mesp1 to specify cardiac mesoderm during mouse gastrulation. Nature Cell Biology. 2011;13(9):1084–1092. doi: 10.1038/ncb2304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Kuzmichev A. N., Kim S.-K., D'Alessio A. C., et al. Sox2 acts through Sox21 to regulate transcription in pluripotent and differentiated cells. Current Biology. 2012;22(18):1705–1710. doi: 10.1016/j.cub.2012.07.013. [DOI] [PubMed] [Google Scholar]
- 53.Adachi K., Nikaido I., Ohta H., et al. Context-dependent wiring of Sox2 regulatory networks for self-renewal of embryonic and trophoblast stem cells. Molecular Cell. 2013;52(3):380–392. doi: 10.1016/j.molcel.2013.09.002. [DOI] [PubMed] [Google Scholar]
- 54.Kidder B. L., Palmer S. Examination of transcriptional networks reveals an important role for TCFAP2C, SMARCA4, and EOMES in trophoblast stem cell maintenance. Genome Research. 2010;20(4):458–472. doi: 10.1101/gr.101469.109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Pearton D. J., Smith C. S., Redgate E., van Leeuwen J., Donnison M., Pfeffer P. L. Elf5 counteracts precocious trophoblast differentiation by maintaining Sox2 and 3 and inhibiting Hand1 expression. Developmental Biology. 2014;392(2):344–357. doi: 10.1016/j.ydbio.2014.05.012. [DOI] [PubMed] [Google Scholar]
- 56.Cambuli F., Murray A., Dean W., et al. Epigenetic memory of the first cell fate decision prevents complete ES cell reprogramming into trophoblast. Nature Communications. 2014;5:p. 5538. doi: 10.1038/ncomms6538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Thomson M., Liu S. J., Zou L.-N., Smith Z., Meissner A., Ramanathan S. Pluripotency factors in embryonic stem cells regulate differentiation into germ layers. Cell. 2011;145(6):875–889. doi: 10.1016/j.cell.2011.05.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Vooijs M., Jonkers J., Berns A. A highly efficient ligand-regulated Cre recombinase mouse line shows that LoxP recombination is position dependent. EMBO Reports. 2001;2(4):292–297. doi: 10.1093/embo-reports/kve064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Iacovino M., Bosnakovski D., Fey H., et al. Inducible cassette exchange: a rapid and efficient system enabling conditional gene expression in embryonic stem and primary cells. Stem Cells. 2011;29(10):1580–1588. doi: 10.1002/stem.715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Nagy A., Gertsenstein M., Vintersten K., Behringer R. Manipulating the Mouse Embryo: A Laboratory Manual. 3rd. Cold Spring Harbor Laboratory Press; 2003. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Table S1: List of antibodies used for immunofluorescence and western blotting at indicated dilutions.
Supplementary Table S2: List of primers used for qPCR and to generate in situ riboprobes. Restriction sites attached to the primers to ensure directed cloning of the PCR product are underlined.
Supplementary Table S3: Gene expression array data sets.
Supplementary Table S4: Gene groups of Venn diagram intersections.