Abstract
A growing body of evidence suggests yogic practices may benefit adults with type 2 diabetes (DM2). In this systematic review, we evaluate available evidence from prospective controlled trials regarding the effects of yoga-based programs on specific health outcomes pertinent to DM2 management. To identify qualifying studies, we searched nine databases and scanned bibliographies of relevant review papers and all identified articles. Controlled trials that did not target adults with diabetes, included only adults with type 1 diabetes, were under two-week duration, or did not include quantitative outcome data were excluded. Study quality was evaluated using the PEDro scale. Thirty-three papers reporting findings from 25 controlled trials (13 nonrandomized, 12 randomized) met our inclusion criteria (N = 2170 participants). Collectively, findings suggest that yogic practices may promote significant improvements in several indices of importance in DM2 management, including glycemic control, lipid levels, and body composition. More limited data suggest that yoga may also lower oxidative stress and blood pressure; enhance pulmonary and autonomic function, mood, sleep, and quality of life; and reduce medication use in adults with DM2. However, given the methodological limitations of existing studies, additional high-quality investigations are required to confirm and further elucidate the potential benefits of yoga programs in populations with DM2.
1. Introduction
Type 2 diabetes (DM2) has become a leading public health issue globally, with estimated 366 million people affected in 2011 [1]. This figure represents a more than twofold rise in the last three decades and parallels the growing pandemic of obesity and the increasingly widespread adoption of Western lifestyles [1, 2]. Worldwide prevalence is expected to continue rising in both industrialized and developing countries [2, 3], with numbers projected to reach 552 million adults by 2030 [1, 4]. DM2 is now a leading cause of death and disability and significantly increases risk for both macrovascular complications, such as atherosclerosis, and microvascular complications, such as retinitis, diabetic neuropathy, and renal disease [5]. DM2 is also strongly associated with elevated risk for other serious chronic conditions, including depression and dementia [3, 6]. Cardiovascular disease (CVD) is the primary cause of morbidity and mortality in those with DM2 [7], accounting for at least 65% of deaths in this population [8]. In addition, the social and economic burden of DM2 is substantial and growing rapidly. For example, DM2 accounts for at least 10 percent of all healthcare expenses in the United States, making DM2 the single most costly chronic disease [9].
DM2 is typified by hyperglycemia in the presence of insulin resistance [6]. Other key related hemodynamic and metabolic abnormalities characterizing DM2 include elevated blood pressure, dyslipidemia, and chronic inflammation, as well as hypercoagulation and increased oxidative stress [5, 10–12]. Risk for DM2 rises with increasing age and is further elevated in certain racial and ethnic groups, including non-Hispanic blacks, Asians, Native Americans, and Pacific Islanders [13, 14]. However, while race, age, genetic predisposition, and other nonmodifiable factors are important in the pathogenesis of DM2, lifestyle factors, particularly, physical inactivity, overnutrition, and related obesity, are thought to be primarily responsible for the current global diabetes epidemic [1, 15]. Other contributing lifestyle-related factors include chronic stress, impaired sleep, and smoking [2, 10, 16, 17]. In fact, lifestyle factors may account for 90% of incident diabetes cases [2] and are significant predictors of DM2-related complications and mortality [18]. Thus, a central element of DM2 care is lifestyle management, which is considered critical to the prevention of acute complications and the reduction of risk for long term complications [6].
A central goal in DM2 management is the reduction of blood glucose levels, which has been shown to reduce risk of microvascular and possibly certain other complications [3, 5, 6]. However, while improving glycemic control remains a priority, the critical importance of multifactorial DM2 management has been increasingly emphasized in recent years, reflecting the complex constellation of factors that underlie the development of DM2 and its sequelae [3, 6]. In particular, reducing CVD risk factors is crucial to effective DM management [6]. Additional goals include reducing DM-related distress, alleviating depression, and enhancing emotional well-being and quality of life, factors that are important predictors of glycemic control, complication rates, treatment adherence, and other outcomes [6, 19–24].
In light of the above, identifying sustainable lifestyle interventions with the potential to improve multiple factors of relevance to the management of this complex illness is of clear importance. Mind-body practices such as yoga, which capitalize on the ability of the mind to enhance physical health (and vice versa), appear particularly suited for addressing multifactorial conditions. Yoga is a traditional mind-body system originating in India over 4000 years ago [25]. In recent decades, the practice of yoga has been rising in both developed and developing countries worldwide [26–29], and the field of yoga therapy is now growing rapidly [30]. The goals of yoga, a Sanskrit term meaning “yoke or union,” do not center primarily on physical fitness, but rather on integration of mind, body, and spirit, cultivation of balance, calm, harmony, and awareness, and, in classic yoga traditions, the attainment of selflessness and spiritual enlightenment [25, 26]. Of the several major branches of yoga, the most widely practiced forms include Raja (royal or classical) yoga and the closely related Hatha yoga, sometimes known as the yoga of activity [25, 26]. Mantra yoga, emphasizing the use of specific sounds or chants to achieve mental and spiritual transformation, was popularized in the West by Maharishi Mahesh Yogi, the founder of Transcendental Meditation (TM). Hatha and Raja yoga emphasize specific postures (asanas), including both active and relaxation or restorative poses, as well as breath control (pranayama), concentration (dharana), and meditation (dhyana), with some schools focusing primarily on restorative/meditative practices. Hatha yoga, the branch of yoga most widely practiced in the US and other Western countries, also incorporates cleansing exercises, mantras or chants, and specific hand gestures (mudras). Originally developed to prepare the body for meditation, Hatha yoga itself encompasses many different styles, including Iyengar, Ashtanga, Integral, Kundalini, Viniyoga, Vinyasa, Kripalu, and Bikram yoga [25, 26].
A growing body of evidence suggests yoga practice may reduce risk for CVD and lead to improvements in physical health and well-being in a range of populations [31–37], potentially including those with DM2. In this systematic review, we critically evaluate available evidence from controlled trials regarding the effects of yoga-based programs on health-related outcomes in adults with DM2. We also outline major limitations in the current literature, briefly discuss possible mechanisms that may underlie observed benefits, and suggest directions for future research.
2. Methods
We searched eight databases via EbscoHost (Academic Search Complete, Alt HealthWatch, CINAHL with Full Text, Health Source: Nursing/Academic Edition, MEDLINE, PsycARTICLES, PsycINFO, and SPORTDiscus with Full Text) from their inceptions through March 2015 for original, scholarly articles reporting on controlled trials of yoga in the management of diabetes. The basic search strategy, (yoga OR yogic) AND diabetes, yielded 468 results; the same search conducted in the Cochrane Central Register of Controlled Trials resulted in 37 trials. A secondary search of the Physiotherapy Evidence Database (PEDro) for the terms yog∗ AND diabetes yielded 34 articles. Titles and abstracts of the citations were scanned to identify potential articles for inclusion in this review. Potentially eligible papers were retrieved for more detailed review. In addition, we manually searched our own files, the citation sections of all identified articles, and the reference sections of recent review articles regarding diabetes and yoga.
Original, controlled studies were included if they evaluated the effects of yoga and yoga-based interventions on measures of glycemic control and insulin resistance, lipid profiles, body weight or composition, blood pressure, or other relevant outcomes, such as oxidative stress, nervous system function, cardiopulmonary function, mood and sleep impairment, or medication use. We excluded uncontrolled trials, cross-sectional studies, case series, and case studies, as well as trials that were published only in dissertation or abstract form. We also excluded articles that did not specifically target adults with diabetes, involve an intervention focused on yoga as the major component, study a yoga program of at least two week's duration, or report quantitative outcome data. Studies targeting populations with type 1 diabetes were also excluded, as the etiology and management for type 1 diabetes differ from those for DM2.
We categorized clinical measures and outcomes evaluated into several domains. For each domain, we summarized findings from relevant studies. Study quality was evaluated using the PEDro scale, which scores papers based on the following 10 criteria: (1) subjects were randomly allocated to groups, (2) allocation was concealed, (3) the groups were similar at baseline regarding the most important prognostic indicators, (4) there was blinding of all subjects, (5) there was blinding of all therapists who administered the therapy, (6) there was blinding of all assessors who measured at least one key outcome, (7) measures of at least one key outcome were obtained from more than 85% of the subjects initially allocated to groups, (8) all subjects for whom outcome measures were available received the treatment or control condition as allocated or, where this was not the case, data for at least one key outcome was analyzed by “intention to treat,” (9) the results of between group statistical comparisons are reported for at least one key outcome, and (10) the study provides both point measures and measures of variability for at least one key outcome. One point is awarded for each criterion reported in the paper. In addition, PEDro assesses whether study eligibility criteria were specified; however, as this does not address statistical or internal validity, no points are awarded for this criterion. Whenever available, we reported the score given in the PEDro database; when the study was not in PEDro, we assigned a score based on the PEDro instructional materials (http://www.pedro.org.au/wp-content/uploads/PEDro_scale.pdf). In our discussion of findings, we also considered recent meta-analyses of RCTs targeting populations with diabetes.
Each eligible study identified was classified into one of two design categories: randomized controlled trials (RCTs) or nonrandomized controlled trials (NRCTs). In RCTs, each participant is randomly assigned to one of at least two groups: an active intervention (i.e., yoga) group and one or more comparator groups. In NRCTs, the experimental design and analytic strategy are similar to that of RCT's, but the allocation to a given treatment is not performed randomly. Articles were selected and data extracted and evaluated by both authors; conflicts were resolved by discussion and consensus.
3. Results
Of 539 potentially relevant abstracts and citation indices scanned, 159 potentially eligible papers were identified for closer review. A total of 33 papers reporting findings from 25 original studies, including 13 nonrandomized controlled trials (NRCTs) [38–50] and 12 randomized controlled trials (RCTs) [51–70], representing a total of 2170 enrolled participants met our eligibility criteria and were included in this review.
Of the 25 studies, 24 (96%) specified adults with DM2; in the one remaining investigation, participants were recruited from a diabetes clinic, but authors did not specify type of diabetes (Table 1). Age range was wide in most studies, with some specifying no upper age limit [57, 60–62, 65]. Of the 23 studies that specified age cut-offs (12 NRCT, 11 RCTs), 8 (34.8%) excluded patients under 30/35 years of age; an additional 10 (43.5%) excluded those under 40/45 years of age. While several studies included adults over the age of 65 [61, 62, 66–70], only one study specifically targeted elder adults [61, 62]. All but two studies [51, 52, 60] included participants of both genders; likewise, only two studies clearly specified exclusion of patients on anti-DM medications [38, 43]. Patients ranged from newly or recently diagnosed DM2 to those diagnosed with DM2 for 10 years or more, often within the same study, with stated exclusion and inclusion criteria varying from study to study. Of the 17 studies with sufficient detail to allow determination, all but 2 [45, 65] excluded patients with significant complications (Table 1), although, again, exclusion conditions were not uniform across studies.
Table 1.
Characteristics of eligible studies investigating the effects of yoga-based programs in adults with diabetes (25 controlled trials, including 12 randomized (RCTs) and 13 nonrandomized (NRCTs)).
| NRCTs (N) | RCTs (N) | Total | ||
|---|---|---|---|---|
| N | % | |||
| Participant characteristics | ||||
| Target population: adults with | ||||
| Type 2 diabetes only | 12 | 12 | 24 | 96.0% |
| Unspecified diabetes | 1 | 0 | 1 | 4.0% |
| One gender only specified | 0 | 2 | 2 | 8.0% |
| Excluding those on DM meds | ||||
| Yes | 2 | 0 | 2 | 8.0% |
| Not specified | 0 | 1 | 1 | 4.0% |
| Excluding those with DM complications | ||||
| Yes | 9 | 10 | 19 | 76.0% |
| No | 1 | 1 | 2 | 8.0% |
| Not specified | 3 | 1 | 4 | 16.0% |
| Age range in years | ||||
| ≥18–26 | 0 | 3 | 3 | 12.0% |
| 30/35–55/60/65 | 6 | 2 | 8 | 32.0% |
| 40/45–55/60 | 3 | 2 | 5 | 20.0% |
| 40/45–65/70/75 | 3 | 2 | 5 | 20.0% |
| 50–70/>60 y | 0 | 2 | 2 | 8.0% |
| Not specified | 1 | 1 | 2 | 8.0% |
| Years since DM diagnosis | ||||
| ≥0-1 year | 0 | 2 | 2 | 8.0% |
| 1/2 years | 1 | 0 | 1 | 4.0% |
| 2–5 years | 0 | 1 | 1 | 4.0% |
| 5–10 years | 0 | 1 | 1 | 4.0% |
| 0/1–10 years | 7 | 1 | 8 | 32.0% |
| >15 years | 0 | 2 | 2 | 8.0% |
| Not specified | 5 | 5 | 10 | 40.0% |
| Sample size | ||||
| <25 | 0 | 2 | 2 | 8.0% |
| 25–40 | 2 | 2 | 4 | 16.0% |
| 41–60 | 4 | 4 | 8 | 32.0% |
| >60 | 7 | 4 | 11 | 44.0% |
| Location | ||||
| India | 13 | 7 | 20 | 80.0% |
| UK | 0 | 2 | 2 | 8.0% |
| Cuba | 0 | 2 | 2 | 8.0% |
| Iran | 0 | 1 | 1 | 4.0% |
| Year published | ||||
| 2010–2014 | 7 | 7 | 14 | 56.0% |
| 2005–2009 | 2 | 3 | 5 | 20.0% |
| 2000–2004 | 3 | 1 | 4 | 16.0% |
| Prior to 2000 | 1 | 1 | 2 | 8.0% |
| Yoga intervention∗ | ||||
| Yoga-based program alone | ||||
| Including asanas | 11 | 10 | 21 | 84.0% |
| Not including asanas | 0 | 2 | 2 | 8.0% |
| Yoga combined with other interventions | ||||
| Including asanas | 3 | 0 | 3 | 12.0% |
| Not including asanas | 0 | 0 | 0 | 0.0% |
| Duration | ||||
| <8 weeks | 6 | 1 | 7 | 28.0% |
| 12 weeks/3 months | 5 | 7 | 12 | 48.0% |
| 4–6 months | 2 | 2 | 4 | 16.0% |
| >6 months | 0 | 2 | 2 | 8.0% |
| Frequency of practice¥ | ||||
| <3x/week | 0 | 1 | 1 | 4.3% |
| 3x/week | 1 | 3 | 4 | 17.4% |
| 4-5x/week | 2 | 1 | 3 | 13.0% |
| 6-7x/week | 8 | 7 | 15 | 65.2% |
| Program structure¥¥ | ||||
| Classes only | 9 | 7 | 16 | 69.6% |
| Classes combined with home practice | 1 | 5 | 6 | 26.1% |
| Training session combined with home practice | 1 | 0 | 1 | 4.3% |
| Comparison condition∗∗ | ||||
| Usual care/no treatment | 11 | 8 | 19 | 76.0% |
| Attention control | 0 | 1 | 1 | 4.0% |
| Active comparator | 3 | 5 | 8 | 32.0% |
| >1 control | 1 | 2 | 3 | 12.0% |
All but 2 were published in 2000 or later, with the majority (56%) being published in the last 5 years. Most (80%) of the studies were conducted in India. The vast majority, 76%, were moderate-sized studies that included over 40 participants, with 44% including 61 or more participants and 8 (32%) including at least 100 participants. Yoga interventions ranged from 15 days [60] to 12 months [69] in duration, with 72% including at least 12 weeks of practice (Table 1). Programs varied substantially in practice frequency, intensity, and content, including, for example, a 3-month Hatha yoga program in which participants attended 1-2 classes/week [65], a 90-day program of daily deep yoga relaxation practice (yoga nidra) [64], a 6-month Sudarshan Kriya (SKY) rhythmic breathing program, with classes once/week and daily home practice [56], and 3- to 12-month comprehensive yoga program with practice 6 to 7 days/week [57]. Ninety-two percent of the yoga programs incorporated active asanas or yoga poses. Comparator conditions also varied widely, from wait list/no treatment [43, 65], standard care [39–42, 45–51, 54–56, 58, 63–66, 70], and/or group education [61] to a comprehensive conventional exercise program [57, 60, 66, 69]. Three studies included more than one comparator group [38, 63, 66]. For some studies, standard care included only medications [39, 40, 48, 49, 58, 63, 64], whereas in others, standard care also included a special diet [38, 41, 42, 47, 50, 54, 56, 70] and/or walking or other exercises [47, 48, 50, 54–56].
3.1. Effects of Yoga on Metabolic, Anthropometric, and Hemodynamic Indices
Twenty-five eligible studies assessed the potential impact of yoga-based programs on one or more anthropometric or physiologic markers of importance to diabetes management and prognosis, including measures of glucose tolerance and insulin resistance, lipid profiles, body weight and composition, and blood pressure. Findings of these studies are summarized in Table 2 and are discussed briefly below.
Table 2.
Summary of study characteristics and major findings.
| First author, year, and location | Tx duration | Study population | N enrolled/completed (per group) [% retention] | Yoga intervention | Comparison condition | Assessment times | Major findings | PEDro scale score (range 0–10) |
|---|---|---|---|---|---|---|---|---|
| Nonrandomized controlled trials (NRCTs) | ||||||||
|
| ||||||||
| Dash, 2014, India [42] | 40 days | Adults 40–60 yo w/DM2 for 0–10 y; Excl: BMI > 25; DM1; DM2 w/nephropathy, CAD, retinopathy, alcoholism; practicing any yoga; 37% F | 60 (30Y, 30C) [100%]# | Yoga (AS, PR, SH, and M): 30–40 min/d; + prescribed diet, oral meds | Prescribed diet, oral meds | Before and after: FBG, PPBG, A1c, TC, Tg, LDL, HDL, and VLDL |
Yoga: Sig ↓ in FBG, PPBG, A1c, TC, Tg, LDL, VLDL, Sig ↑ HDL; C: Sig ↓ in FBG, PPBG, VLDL; Y vs. C: Sig ↓ in FBG, PPBG, A1c, TC. TG, LDL, Sig ↑ HDL |
4 |
|
| ||||||||
| Popli, 2014, India [43] | 6 mos | Adults 30–60 yo w/DM2, no previous yoga experience; Excl: Pts already on oral hypoglycemic meds or insulin, w/alcoholism, BG > 300 mg/dL, or end-organ damage (e.g., retinal detachment, nephropathy, and peripheral neuropathy) | 130 (80Y, 50C) | Yoga (AS, PR): 1 hr with yoga trainer, 5 d/wk for 1st month, and then continuing practice at home for remaining 5 mos | No treatment | Monthly: FBG; 3x/mo: PPBG, A1c |
Yoga: Sig ↓ in FBG, PPBG, A1c; C: NS |
1 |
|
| ||||||||
| Bindra, 2013, India [39] | up to 90 days | Adults 35–65 yo w/DM2 for not >10 y and no complications; Excl: those w/RA, cancer, TB, and MI and not willing to do yoga | 100 (50Y, 50C) [100%]# | Yoga (not described) + oral DM meds | Oral DM meds only | Before and after: FBG, A1c, TC, LDL, HDL, and Tg |
Baseline yoga versus C: NS differences between the groups; After 90 days, Yoga versus C: Signif. differences between the groups in FBG, A1c, TC, LDL, HDL |
5 |
|
| ||||||||
| Balaji, 2011, India [44] | 3 mos | Uncomplicated DM2 Pts 40–55 yo w/DM duration 1–10 y | 44 (22Y, 22C) [100%]# | Yoga (AS, PR, SH): daily for 1 hr; subgroup T1 (n = 16): + oral drugs; subgroup T2 (n = 6): + oral drugs and insulin | 22 controls | Before and after: FBG, PPBG, A1c, TC, Tg, LDL, HDL, and insulin requirement/day (subgroup T2), wt, BMI, W : H |
Yoga T1: Sig ↓ in FBG, A1c, Tg, LDL, wt, BMI, W : H; Yoga T2: Sig ↓ in FBG, PPBG, A1c, Tg, LDL, wt, BMI, W : H, insulin/d; C: NS |
2 |
|
| ||||||||
| Hegde, 2011, India [45] | 3 mos | DM clinic Pts w/DM2; age: 40–75 y; Excl: smoking, alcoholism | 123/120 (60Y, 63C); strat'd by complications [98%]∗∗∗ | Yoga (AS, PR, and SH): at least 3 d/wk; + standard care | Standard care (given general oral and written information about diet and exercise) | Before and after: FBG, PPBG, A1c, oxidative stress (MDA, glutathione, SOD, Vit. C, and Vit. E), BMI, waist circumference, W : H, and BP | Y versus C: Sig ↓ in FBG, PPBG, A1c, BMI, MDA, Sig ↑ glutathione, Vit. C | 4 |
|
| ||||||||
| Kyizom, 2010, India [46] | 45 days; groups matched for age and sex | DM clinic Pts, 35–60 yo, w/uncomplicated DM2 from 2–10 y | 60 (30Y, 30C) [100%]# | Yoga (AS, PR, and SH): daily for 45 min/d; + standard care | Standard care | Before and after: FBG, PPBG, event-related evoked potentials at frontal (Fz), vertex (Cz), and parietal (Pz) areas, peak latencies, and baseline to peak amplitudes of N2 and P3 |
Yoga: Sig ↓ in FBG, PPBG; Sig ↓ in latency of N2 at Cz and P300 at all the montages (Fz, Cz, and Pz); Sig improvement in amplitude of wave N2 at Cz and P300 at Pz montage C: NS |
2 |
|
| ||||||||
| Malhotra, 2010, India [47] | 40 days; controls matched on age and DM severity | Pts from endocrine metabolic clinic w/DM2 for 0–10 y, 30–60 yo, on recommended diet and oral DM meds; Excl: CAD, nephropathy, and proliferative retinopathy | 106 (56Y, 50C)/64 (26Y, 38C) [60%] | Yoga (AS, PR, and SH): 40–60 min/d; + diet and DM meds | Standard care: meds, diet, and light exercises (walking) | Before and after: FBG, PPBG, TC, Tg, HDL, LDL, VLDL, MDA, and insulin; Wt, W : H, BMI, LBM, and BSA; SBP, DBP, HR, and QT interval; SVC, FEV1, FVC, FEV1 : FVC ratio, PEFR, and MVV; median nerve conduction velocity (m/sec), amplitude (mV), and latency (msec) measured proximally (at elbow) and distally (at wrist) |
Yoga: Sig ↓ in FBG, TC, Tg, MDA, SBP, DBP, heart rate, W : H; Sig improvement in pulmonary, CV fxn, Sig ↑ in left hand nerve conduction velocity and amplitude (distal) C: Sig ↓ in LBM (worsening), nerve conduction amplitude (right proximal), Sig ↑ in Tg (worsening), insulin (in 4 pts), HR, PEFR; Y versus C: not analyzed |
2 |
|
| ||||||||
| Mahapure, 2008, India [41] | 6 wks | Pts 40–58 yo, w/DM2 on regular diet and antidiabetic drug regimen | 40 (30Y, 10C) [100%]# | Yoga (AS, PR, SH); 1 hr/d, every d except Sundays; + standard care | Standard care: diet and meds | Before and after: FBG, A1c, and SOD |
Yoga: Sig ↓ in FBG, A1c, Sig ↑ SOD; C: NS; Y versus C: Sig ↓ in FBG, A1c, Sig ↑ SOD |
3 |
|
| ||||||||
| Singh, 2008, India [48] | 45 days | Pts 35–60 yo, w/uncomplicated DM2 for 1–10 y | 60 (30Y, 30C) [100%]# | Yoga (AS, PR, and SH): daily for 45 min/d; + standard care | Standard care (conventional meds) | Before and after: FBG, PPBG, A1c, TC, Tg, LDL, VLDL, HDL, insulin, wt, and BMI |
Yoga: Sig ↓ in Wt, FBG, PPBG, insulin, TC, Tg, VLDL, LDL; Sig ↑ in HDL; C: Sig ↑ Wt; Y versus C: not given |
2 |
|
| ||||||||
| Agte, 2004, India [40] | 4 mos | Previously diagnosed DM2 Pts 45–65 yo; having stabilized glucose levels, and taking conventional prescribed medications; BMI: 25.4 (Y), 25.6 (C); 53% F | 87 (57Y, 30C)/65 (35Y, 30C) [75%] | SKY + PR, AS, and M; interactive discussions on stress-free living; nutritional counseling w/emphasis on eating fresh fruits and vegetables; 6 d course | Without SKY training | Before and after: FBG, PPBG, A1c, TC, Tg, HDL, and MDA |
Yoga: Sig ↓ in FBG, PPBG, TC, Tg, MDA; C: NS; Y versus C: not presented |
1 |
|
| ||||||||
| Agrawal, 2003, India [49] | 3 mos | Pts randomly selected from diabetes clinic (type of DM not specified, but background implies that study targeted DM2); Excl: Pts w/liver disease, alcoholism, malnutrition, thyrotoxicosis, TB, or who were noncooperative | 200/154 (82Y, 72C) [77%] | Yogic lifestyle program: health rejuvenation exercises (5 min), AS (18 min), abdominal exercises (7 min), and R/M/PR (30 min); at least 5 d/wk + diet | Standard medical tx or standard home exercise program | Before and after: FBG, A1c, TC, HDL, LDL, VLDL, BP, BMI, W : H, QOL (satisfaction, impact, and worry), psychological assessment, self-evaluation, doses of metformin, glipizide, and insulin, BP, and renal function | Y versus C: Sig ↓ FBG, A1c, BMI, W : H, TC, LDL, VLDL, SBP, DBP; metformin, glipizide, and insulin doses; QOL satisfaction, impact, and worry scores, psychological assessment, and self-evaluation; Sig ↑ HDL | 3 |
|
| ||||||||
| Malhotra, 2002, India [50] | 40 days; controls matched on age, sex, SES, and DM severity | DM2 for 0–10 y; 30–60 yo; Excl: cardiac, renal, and proliferative retinal complications | 40 (20Y, 20C) [100%] | Supervised yoga (AS, PR, and SH): daily for 30–40 min/d; + medication, diet | Medication, diet, and light exercise (walking) | Before and after 40-day treatment: median nerve conduction velocity (m/sec), amplitude (mV), and latency (msec) measured proximally (at elbow) and distally (at wrist) |
Yoga: Sig ↑ conduction velocity in left hand; C: Sig ↓ in amplitude at right elbow (proximal) |
4 |
|
| ||||||||
| Khare, 1999, India [38] | 3 mos, non-DM controls matched for age, sex | 60 DM2 Pts (DM for 1-2 y) from DM clinics (in yoga/diet groups), 20 non-DM matched on age, sex; 40–70 y on vegetarian diet | 80 (20, 20, 20, 20) [100%]# | (1) Yoga: AS (3 poses, 2–5 min each) and SH (20 min) only (2) Yoga + strict diet Yoga: 2x/d |
(1) No intervention (non-DM adults) (2) Strict diet alone (20 DM adults) |
Baseline and 1, 2, and 3 months: BG, serum fructosamine, TC, and Wt |
Yoga: after 3 mos, sig ↓ BG, Fruc, TC, and Wt Yoga + diet: after 3 mos, sig ↓ BG, Fruc, TC, and Wt Diet: after 3 mos, sig ↓ TC and Wt non-DM, no tx: after 3 mos, NS |
1 |
|
| ||||||||
| Randomized controlled trials (RCTs) | ||||||||
|
| ||||||||
| Habibi, 2013, Iran [51, 52] | 12 wks | Women w/DM2, 45–60 yo, not taking insulin; without DM complications or hx of CVD | 26 (16Y, 10C) [100%]# | Yoga: 75 min, 3 d/wk | Standard care | Before and after: BP, FBG, F insulin, TC, HDL, LDL, leptin, Wt, and BMI | Y versus C: Sig ↓ in FBG, insulin, Tg | 4† [52] |
|
| ||||||||
| Jyotsna, 2012, India [53–56] | 6 mos | Adults w/DM2 plus: lifestyle modification, A1c of 6–9%, and oral hypoglycemic agents for past 6 months; Excl: Pts w/retinopathy, glaucoma, uncontrolled HTN, CAD, overt complications of diabetes, and nephropathy; mean age (y): 49.92 ± 11.46 (yoga), 47.25 ± 10.80 (control) | 120 (64Y, 56C) [100%]# | Sudarshan Kriya yoga (SKY: a rhythmic cyclical breathing, preceded by PR); 3 d group training program followed by classes 1x/wk (long kriya) and daily home practice (short kriya) + standard care | Standard care (oral antidiabetic drugs and diet and exercise advice) | Before and 6 mos after tx: CAFTs, FBG, PPBG, and A1c; Before and 3 and 6 mos after tx: QOL (domains: (1) physical health, (2) psychological well-being, (3) social relationships, and (4) environment) |
Yoga: Sig ↑ Symp fxn; C: NS; Y versus C: Sig ↑ overall cardiac autonomic fxn; Sig ↓ PPBG; Sig ↑ QOL: 3 mos after: domains 1 and 2 and total QOL; 6 mos after: domains 2 and 4 and total QOL |
4† [53, 54]; 4 [55]; 5† [56] |
|
| ||||||||
| Nagarathna, 2012, India [57] | 9 mos | Pts >25 yo with DM2 >1 y (FBG >120 mg% when dx), stable dose of oral hypoglycemic agents or insulin for at least 3 wks, no prior yoga practice, no major complications; mean age: 52.4 y; 31% F | 277 (141Y, 136C)/173 (88Y, 85C) [62%] | Yoga (AS, PR, M, devotional sessions, and lectures): 1 hr/d, 5 d/wk for 12 weeks [cleansing techniques (kriyas) performed 1x/wk]; one 2 hr class/wk and 1 hr daily home practice for 9 mos | Exercises and walking designed to achieve a comparable intensity of physical exertion, nonyogic breathing exercises, supine rest, and lectures: 1 hr/d, 5 d/wk for 12 wks; one 2-hour class/wk and 1-hour daily home practice for 9 mos | Before and after: TC, Tg, HDL, LDL, VLDL, FBG, PPBG, A1c, and oral hypoglycemic medication requirement |
Yoga: Sig ↓ meds, FBG, PPBG, A1c, Tg, TC, LDL, VLDL; Sig ↑ HDL; C: Sig ↓ PPBG, A1c, Tg, TC, VLDL; Y versus C: Sig ↓ meds, LDL; Sig ↑ HDL |
6 |
|
| ||||||||
| Shantakumari, 2012, India [58, 59] | 3 mos | Outputs with DM2 + HTN and dyslipidemia, 35–55 yo; Excl those w/known retinopathy, nephropathy, CAD, and cerebrovascular diseases; avg DM duration: 5–10 y; 48% F | 100 (50Y, 50C) [100%]# | Yoga: 1 hr daily; AS (30–35 min), PR (10 min), and M (15 min); + standard care | Standard care (oral hypoglycemic drugs, no yoga) | Before and after: FBG, PPBG, TC, HDL, LDL, Tg, Wt, W : H, BMI, SBP, and DBP |
Yoga: Sig ↓ Wt, W : H, Tg, TC, LDL; C: Sig ↑ Wt |
6† [59]; 5† [58] |
|
| ||||||||
| Subramaniyan, 2012, India [60] | 15 days | Males ≥18 yo w/DM2 on standard care and medically eligible for walking/yoga per physician; age: 55% were 31–40 yo | 20 [100%]# | Yoga: 60 min (6-7 a.m.) daily; AS (~30 min), sun salutation (~6 min), SH (~25 min) + routine meds | Brisk walking: 60 min (6-7 a.m.) daily + routine meds | Before and after: FBG |
Yoga: Sig ↓ in FBG Walking: Sig ↓ in FBG |
4 |
|
| ||||||||
| Vaishali, 2011, India [61, 62] | 12 wks | Diabetic clinic Pts, >60 yo, w/DM2 >15 y and ≥1 metabolic risk factor (high FBG, pre-HTN, overwt/obese, high TC), on antidiabetic meds >10 y; Excl: uncontrolled HTN, severe neuropathy, hx of foot lesions, unstable proliferative retinopathy, and nephropathy | 60/57 [95%] | Yoga (individualized AS, PR, and SH): 45–60 min under supervision, 6 d/wk | Educational group (general healthy lifestyle and exercise): 1x/mo | Before and after: FBG, A1c, TC, Tg, LDL, and HDL |
Yoga: Sig ↓ in FBG, A1c, TC, Tg, LDL; Sig ↑ HDL
C: NS Y versus C: Sig ↓ in A1c, FBG, TC, Tg, LDL; Sig ↑ HDL |
6† [61]; 6 [62] |
|
| ||||||||
| Pardasany, 2010, India [63] | 12 wks | Adults w/DM2, 40–60 yo; Excl: hx of renal disease, arthritis, HBP, intermittent claudication, foot injury or ulcers, breathlessness, and cardiac disease; 38% F | 45 (15, 15, 15) [100%]# | Hatha yoga (12 AS and 6/7 PR): 3x/wk; oral hypoglycemic meds | (1) Yang-style tai chi (TC, 24 forms): 3x/wk, oral hypoglycemic meds; (2) Oral hypoglycemic meds, no exercise (C) |
Before and after: FBG, PPBG, A1c, TC, and LDL |
Yoga: Sig ↓ FBG, PPBG, A1c, TC, LDL TC: Sig ↓ FBG, PPBG, A1c, TC, LDL C: NS Y versus C: Sig ↓ FBG, PPBG, TC, LDL; TC versus C: Sig ↓ FBG, PPBG, A1c, TC; Tai chi versus Y: NS |
4† |
|
| ||||||||
| Amita, 2009, India [64] | Up to 90 days | Middle aged (35–65 yo), DM2 Pts on oral hypoglycemics; Excl: >200 mg/dL FBG, >300 mg/dL PPBG; hx of DM complications or other systemic conditions; 29% F | 41 (20Y, 21C) [100%]# | Yoga nidra (deep relaxation): 45 min, daily | Standard care (no yoga) | Every 30 days: FBG, PPBG, and symptoms related to DM (i.e., insomnia, palpitations, sweating, distress, headache, and anxiety) |
Yoga: Sig ↓ FBG (60 and 90 d), PPBG (90 d), total symptoms (insomnia, distress, etc. after 3 mos); C: not given |
1† |
|
| ||||||||
| Skoro-Kondza, 2009, UK (London) [65] | 12 wks | Adults (>18 yo) w/DM2, not on insulin; most well-controlled (A1c x = 6.9); mean duration of DM2 (y): 30 ± 5; mean age (y): 60 ± 10; nonwhite: 55% at one site, 40% at the other site; 61% F | 59 [100%]#∗ | Yoga (PR, AS, and SH): 90 min class, 3x/wk | Wait list (both groups given leaflets on healthy lifestyle and encouraged to exercise) | Before and after and six months later: A1c, wt, waist circumference, W : H, BMI, TC, LDL, Tg, HDL, SBP, and DBP (used to calculate cardiovascular risk score); diabetes-related QOL, and self-efficacy. |
Yoga: NS; C: NS: Y versus C: NS Note:Quantitative data provided only for A1c |
4† |
|
| ||||||||
| Gordon, 2008, Cuba [66–68] | 24 wks, age and sex matched controls | Pts 40–70 yo w/DM2 from 1–10 y, w/at least 3 mos of training in DM education, exercise, diet, and medication according to IDF recommendations; no severe complications, nonsmoker, nonalcoholic | 231 (77 Pts: 62 females, 15 males per group) [100%]#∗∗∗ | Yoga: PR (20 min), warm-up exercises (25 min), AS (60 min), and SH (15 min); 1 class/wk for 24 wks plus home yoga exercise (followed IDF criteria, with yoga as exercise) | (1) PT: warm-up exercises (15 min), aerobic walking (30 min), flexibility exercises (20 min), aerobic dance (20 min), games (25 min), warm-down (10 min); 1 class/wk for 24 wks plus home exercise 3-4x/wk (followed IDF criteria); (2) Control: tx plan as per their doctors, no active exercise tx |
Baseline and 3 and 6 mos: FBG, A1c, insulin, TC, Tg, HDL, LDL, VLDL, BMI, MDA, PLA2 activity, POX, and SOD and catalase activity; microalbuminuria, creatinine; cortisol, TSH, T3, and T4 |
Yoga: 3 mos: Sig ↓ FBG, TC; 6 mos: Sig ↓ FBG, A1c, TC, VLDL, microalbuminuria, MDA; Sig ↑ SOD; PT: 3 mos: Sig ↓ FBG, TC; 6 mos: Sig ↓ FBG, TC, VLDL, microalbuminuria, MDA; Sig ↑ SOD; C: 3 mos: NS; 6 mos: NS; Y versus C: 3 mos: Sig ↓ FBG, A1c, TC; 6 mos: Sig ↓ FBG, A1c, TC, MDA, BMI, microalbuminuria, Sig ↑ % insulin receptor binding PT versus C: 3 mos: Sig ↓ FBG, A1c, TC; 6 mos: Sig ↓ FBG, A1c, TC, MDA, BMI, microalbuminuria, Sig ↑ % insulin receptor binding |
6† [66, 67]; 4† [68] |
|
| ||||||||
| Céspedes, 2002, Cuba [69] | 12 mos | DM2 patients, 50–70 y, DM duration 2–5 y, without malnutrition or severe complications, in good mental health | 40 (22 Y, 18 C) [100%]# | Moderate intensity yoga (PR, AS): 60 min class, 3x/wk + lifestyle advice, soybean-rich diet | Moderate intensity aerobic exercise: 60 min class, 3x/wk + lifestyle advice, soybean-rich diet | Before and after: BG, TC, HDL, LDL, Tg, and creatinine |
Yoga: Sig ↓ TC, LDL, Tg, creatinine; Sig ↑ HDL; PT: Sig ↓ TC, LDL, Tg, creatinine; Sig ↑ HDL; Y versus C: Sig ↓ TC, LDL, Tg, creatinine; Sig ↑ HDL |
5 |
|
| ||||||||
| Monro, 1992, UK [70] | 12 wks | DM2 controlled with meds (N = 13; yoga group = 8) or diet (N = 8); Excl: end stage liver or kidney disease or congestive cardiac failure; age (y): 45–67 | 21 (11Y, 10C) [100%]∗∗ | Yoga (PR, AS, and R) + standard care: 90 min, classes offered 5x/wk (most attended classes 1-2x/wk and practiced at home 1-2x/wk) | Standard care (continuing on medication, diet) | Before and after: FBG and A1c | Y versus C: Sig ↓ in A1c, FBG | 5† |
#Assumed (retention not specifically reported), ∗adherence very low, ∗∗adherence moderate, ∗∗∗adherence excellent, and †score from PEDro database.
A1c: glycosylated hemoglobin A1c, AHA: American Heart Association, AS: yoga asanas or postures, avg: average, BF: biofeedback, BP: blood pressure, BSA: body surface area, C: control, CAD: coronary artery disease, CAFT: cardiac autonomic function tests, cal: calorie, Clin: clinical, comp: composition, CVD: cardiovascular disease, d: day, DM: diabetes mellitus, Excl: excluded, F: female, FBG: fasting blood glucose, FEV1: forced expiratory value in first second, FVC: forced vital capacity, h: hour, HDL: high density lipoprotein, HR: heart rate, HTN: hypertension, hx: history, IFNγ: interferon gamma, a cytokine critical for innate and adaptive immunity against viral and intracellular bacterial infections and for tumor control, IR: insulin resistance (markers of), KR: kriyas or cleansing exercises, LBM: lean body mass, LDL: low density lipoprotein, M: meditation, MDA: malondialdehyde, MI: myocardial infarction, mo: month, MVV: maximal voluntary ventilation, PEFR: peak expiratory flow rate, PLA2: phospholipase A2, PMR: progressive muscle relaxation, POX: protein oxidation, PPBG: postprandial blood glucose, PR: pranayama or yogic breathing exercises, QOL: quality of life, R: relaxation poses (nonspecified), RA: rheumatoid arthritis, Resid: residential, SH: shavasana or corpse pose, a traditional yoga relaxation pose, Sig: significant, SNS/PNS: markers of sympathetic/parasympathetic activation, including heart rate and catecholamine levels, SOD: superoxide dismutase, SVC: slow vital capacity, TB: tuberculosis, TC: total cholesterol, Tg: triglycerides, veg: vegetarian, VLDL: very low density lipoprotein, W : H: waist-hip ratio, wk: week, wt: weight, y: years, and yo: years old.
3.1.1. Measures of Glycemia and Insulin Resistance
Twenty-four studies investigated the effects of yoga on markers of glycemia and insulin resistance, with all but two documenting significant, postintervention improvement in one or more measures following the practice of yoga either alone or in combination with other therapies. All but two programs [55, 64] incorporated active yoga asanas or poses; interventions ranged in duration from 15 days [60] to 12 months [69]. Of the 12 NRCTs (total N = 1090 participants), all reported significant improvement in one or more indices of glycemia/insulin resistance; reductions in postprandial blood glucose (PPBG) [40, 42–46, 48], fasting blood glucose (FBG) [38–49], fasting insulin [48], fructosamine [38], and glycosylated hemoglobin (HbA1c) [39, 41–45, 49] were observed among participants completing a yoga-based intervention compared to controls receiving no treatment [43], standard care [39–42, 44–46, 48, 49], standard care with light exercise [47], or a low fat vegetarian diet alone [38].
Of the 12 RCTs evaluating the effects of yogic practices on indices of glycemia and insulin resistance (N = 1040 participants), 10 reported significant reductions in at least one measure (Tables 2 and 3). Reported improvements again included significant declines in PPBG [53, 55, 63, 64], FBG [51, 59–61, 63, 64, 66, 70], insulin [51], and HbA1c [61, 66, 70] in those assigned to a yoga-based program versus standard care [51, 55, 58, 63, 64, 66, 67, 70], group education [61], or brisk walking [60]. Two additional trials observed significant beneficial changes in PPBG, FBG, and HbA1c following a comprehensive yoga program [57, 66] that were similar to those detected in participants assigned to a relatively intensive exercise intervention [57, 66].
Table 3.
Observed percent change with yoga in metabolic indices, body weight, and blood pressure among adults with type 2 diabetes (N = 23 controlled trials), stratified by study design and analytic comparisons presented (before/after, intergroup). Only studies reporting significant changes included.
| Findings, by clinical measure | Study design | |||
|---|---|---|---|---|
| Nonrandomized controlled trials | Randomized controlled trials | |||
| Yoga versus baseline | Yoga versus controls | Yoga versus baseline | Yoga versus controls | |
| Measures of insulin resistance | ||||
| Fasting glucose | 14.33–33.23% [38, 40–44, 46–48] | 9.87–29.75% [39, 41, 42, 45, 49] | 7.21–27.53% [57, 60, 63, 64, 66] | 13.46–36.2% [51, 61, 63, 66, 70] |
| Postprandial glucose | 5.92–38.65% [40, 42–44, 46, 48] | 11.61–35.5% [42, 45] | 7.03–18.89% [57, 63, 64] | 11.17–14.22% [55, 63] |
| Fasting glycosylated hemoglobin (HbA1c) | 1.55–21.23% [41–44] | 1.03–7.44% [39, 41, 42, 45, 49] | 0.31–14.17% [57, 63, 67] | 7.77–17.78% [61, 67, 70] |
| Blood lipid profiles | ||||
| Total cholesterol | 5.97–14.61% [38, 40, 42, 47, 48] | 4.11–8.63% [39, 42, 49] | 1.37–37.01% [57, 58, 63, 66, 69] | 4.74–18.3% [61, 63, 66, 69] |
| Triglycerides | 3.97–16.35% [40, 42, 44, 47, 48] | 3.53% [42] | 14.33–38.0% [57, 58, 69] | 1.06–10.96% [51, 61, 69] |
| Low-density lipoprotein (LDL) | 2.18–14.88% [42, 44, 48] | 1.38–15.05% [39, 42, 49] | 8.33–48.45% [57, 58, 63, 69] | 3.39–11.42% [57, 61, 63, 69] |
| High-density lipoprotein (HDL) | 8.28–15.21% [42, 48] | 5.01–24.96% [39, 42, 49] | 6.99–101.59% [57, 69] | 9.1–54.92% [57, 61, 69] |
| Very low-density lipoprotein (VLDL) | 15.17–16.06% [42, 48] | 13.28% [49] | 7.23–22.66% [57, 66] | |
| Anthropometric measures | ||||
| BMI | 7.52–10.34% [44] | 2.72–3.56% [45, 49] | 4.32% [67] | |
| Body weight | 3.48–6.95% [38, 44, 48] | 4.18% [58] | ||
| Waist-hip circumference ratio | 5.38–8.99% [44, 47] | 3.17% [49] | 5.32% [58] | |
| Blood pressure | ||||
| Systolic blood pressure | 11.27% [47] | 5.03% [49] | ||
| Diastolic blood pressure | 12.92% [47] | 3.46% [49] | ||
In contrast, two RCTs documented no significant changes in these parameters. In a small 12-month Cuban trial of 40 adults with uncomplicated DM2, yoga group participants showed modest declines in FBG (7%) that did not differ significantly from those of the exercise group (1.7%) [69]. Similarly, a British study of 59 participants with DM2 showed no significant improvements either in glucose control [65], findings that may be in part attributable to low rates of participant compliance (50% class attendance and 0% performance of home practice).
Collectively, 92% of the controlled trials reviewed, including 12/12 NRCTs and 10/12 RCTs, reported improvements in glucose control with yoga-based programs that were statistically and clinically significant, suggesting that yogic practices may improve glucose control in adults with DM2. However, between group comparisons were not reported in ten studies (7 NRCTs [38, 40, 43, 44, 46–48], 3 RCTs [60, 63, 64]), and, as discussed below, additional methodologic or other limitations plagued most trials, hindering interpretation of findings. Magnitude of effects varied substantially among studies, from relatively modest to very large improvements depending on the study design, population, and comparator group (Table 3).
3.1.2. Lipid Profiles
Of the 25 studies included in this review, 16 (N = 1575 total participants) examined the effects of yoga programs on lipid profiles in those with DM2, with all but one trial [65] reporting significant improvement in one or more lipid indices (Tables 2–4). Studies all incorporated active yoga asanas and varied in length from 40 days [42] to 12 months [69]; in all except one trial [65], participants assigned to the yoga intervention practiced at least 3 days/week. As indicated in Table 2, all 8 NRCTs (N = 737 participants) reported significant improvements in lipid profiles, including reductions in levels of total cholesterol (TC), low-density lipoprotein cholesterol (LDL), very low-density lipoprotein cholesterol (VLDL), and triglycerides (Tg), and increases in high-density lipoprotein cholesterol (HDL) relative to standard care [38–40, 42, 44, 48, 49], or standard care with light exercise [47].
Table 4.
Recent published meta-analyses regarding effects of yoga on risk indices relevant to T2DM, summarized findings.
| Measure | First author, year | Population | Included studies | Comparator | Mean difference (95% CI) | P | ||
|---|---|---|---|---|---|---|---|---|
| Design | Number | Total Pts | ||||||
| Glucose tolerance | ||||||||
| Fasting blood glucose (mg/dL) | Cramer, 2014 [34] | DM2 | RCTs | 7 | 525 | Usual care | −25.56 (−39.60, −11.53) | <0.01 |
| Chu, 2014 [71] | DM or MS | RCTs | 6 | 315 | All | −8.77 (−26.77, 9.23) | 0.34 | |
| HbA1c | Cramer, 2014 [34] | DM2 | RCTs | 7 | 550 | Usual care | −0.49 (−1.03, 0.05) | 0.07 |
| Lipid profiles | ||||||||
| Total cholesterol | Cramer, 2014 [34] | DM2 | RCTs | 5 | 516 | Usual care | −13.09 (−19.60, −6.59) | <0.01 |
| Cramer, 2014 [34] | DM2 | RCTs | 2 | 317 | Exercise | −8.08 (−19.20, 3.03) | 0.21 | |
| Chu, 2014 [71] | DM or MS | RCTs | 5 | 365 | All | −20.98 (−33.23, −8.72) | 0.0008 | |
| Chu, 2014 [71] | CVD risk factors | RCTs | 2 | 109 | All | −18.85 (−37.24, −0.46) | 0.04 | |
| HDL | Cramer, 2014 [34] | DM2 | RCTs | 5 | 516 | Usual care | 5.51 (3.56, 7.47) | <0.01 |
| Cramer, 2014 [34] | DM2 | RCTs | 2 | 317 | Exercise | 4.24 (1.75, 6.72) | 0.01 | |
| Chu, 2014 [71] | DM or MS | RCTs | 5 | 365 | All | 2.45 (−0.33, 5.24) | 0.08 | |
| Chu, 2014 [71] | CVD risk factors | RCTs | 2 | 109 | All | 3.95 (1.69, 6.21) | 0.0006 | |
| LDL | Cramer, 2014 [34] | DM2 | RCTs | 5 | 515 | Usual care | −10.26 (−20.85, 0.32) | 0.06 |
| Cramer, 2014 [34] | DM2 | RCTs | 2 | 317 | Exercise | −9.24 (−16.55, −1.93) | 0.01 | |
| Chu, 2014 [71] | DM or MS | RCTs | 5 | 365 | All | −21.27 (−32.93, −9.62) | <0.00001 | |
| Chu, 2014 [71] | CVD risk factors | RCTs | 2 | 109 | All | −14.42 (−20.47, −8.36) | 0.0003 | |
| VLDL | Cramer, 2014 [34] | DM2 | RCTs | 2 | 308 | Usual care | −4.81 (−6.67, −2.96) | <0.01 |
| Triglycerides | Cramer, 2014 [34] | DM2 | RCTs | 5 | 509 | Usual care | −23.60 (−36.78, −10.43) | <0.01 |
| Cramer, 2014 [34] | DM2 | RCTs | 2 | 317 | Exercise | −22.98 (−60.97, 15.00) | 0.24 | |
| Chu, 2014 [71] | DM or MS | RCTs | 5 | 365 | All | −18.61 (−32.61, −4.60) | 0.009 | |
| Chu, 2014 [71] | CVD risk factors | RCTs | 2 | 109 | All | −27.47 (−60.09, 5.15) | 0.10 | |
| Body composition | ||||||||
| Waist-hip ratio | Cramer, 2014 [34] | DM2 | RCTs | 3 | 311 | Usual care | −0.02 (−0.03, −0.00) | <0.01 |
| BMI | Chu, 2014 [71] | DM or MS | RCTs | 3 | 155 | All | −1.63 (−2.25, −1.01) | <0.00001 |
| Chu, 2014 [71] | CVD risk factors | RCTs | 2 | 77 | All | −0.99 (−2.38, 0.40) | 0.16 | |
| Weight (kg) | Chu, 2014 [71] | DM or MS | RCTs | 3 | 151 | All | −3.27 (−4.99, −1.54) | 0.0002 |
| Chu, 2014 [71] | CVD risk factors | RCTs | 6 | 273 | All | −1.95 (−5.25, 1.35) | 0.25 | |
| Blood pressure (BP) (mmHg) | ||||||||
| Systolic BP | Cramer, 2014 [34] | DM2 | RCTs | 3 | 237 | Usual care | −6.87 (−14.68, 0.94) | 0.08 |
| Cramer, 2014 [34] | Non-DM high risk | RCTs | 8 | 347 | Usual care | −10.00 (−16.42, −3.59) | <0.01 | |
| Cramer, 2014 [72] | HT | RCTs | 6 | 278 | Usual care | −9.65 (−17.66, −2.06) | 0.01 | |
| Hagins, 2013 [73] | HT (all) | RCTs, NRCTs | 11 | 431 | Usual care | −7.96 (−10.65, −5.27) | 0.0002 | |
| Hagins, 2013 [73] | HT (Y incl M, PR, AS) | RCTs, NRCTs | 13 | 656 | All | −8.17 (−12.45, −3.89) | NR | |
| Hagins, 2013 [73] | HT (Y > 58.9 h) | RCTs, NRCTs | 6 | 215 | All | −9.73 (−17.66, −1.79) | NR | |
| Chu, 2014 [71] | DM or MS | RCTs | 3 | 80 | All | −9.39 (−15.14, −3.63) | 0.001 | |
| Chu, 2014 [71] | CVD risk factors | RCTs | 10 | 461 | All | −7.36 (−13.39, −1.33) | 0.001 | |
| Diastolic BP | Cramer, 2014 [34] | DM2 | RCTs | 2 | 210 | Usual care | −0.79 (−5.22, 3.65) | 0.73 |
| Cramer, 2014 [34] | Non-DM high risk | RCTs | 8 | 347 | Usual care | −7.45 (−12.70, −2.21) | <0.01 | |
| Cramer, 2014 [72] | HT | RCTs | 6 | 278 | Usual care | −7.22 (−12.83, −1.62) | 0.01 | |
| Hagins, 2013 [73] | HT (all) | RCTs, NRCTs | 11 | 431 | Usual care | −6.14 (−9.39, −2.89) | NR | |
| Hagins, 2013 [73] | HT (Y incl M, PR, AS) | RCTs, NRCTs | 13 | 656 | All | −5.52 (−7.92, −3.11) | NR | |
| Chu, 2014 [71] | DM or MS | RCTs | 3 | 80 | All | −5.41 (−8.82, −2.01) | 0.002 | |
| Chu, 2014 [71] | CVD risk factors | RCTs | 10 | 461 | All | −6.97 (−11.80, −2.13) | 0.005 | |
| Autonomic fxn | ||||||||
| Heart rate | Cramer, 2014 [34] | Non-DM high risk | RCTs | 3 | 133 | Usual care | −10.89 (−22.83, 1.04) | 0.07 |
AS: asana, CAD: coronary artery disease, CI: confidence interval, CVD: cardiovascular disease, fxn: function, HT: hypertension, M: meditation, MS: metabolic syndrome, NR: not reported, NRCT: nonrandomized controlled trial, PR: pranayama, Pts: participants, and Y: yoga.
Likewise, 7 of the 8 RCTs (totaling 838 participants) reported significant beneficial changes in serum lipids following completion of a yoga-based program. These included significant declines in TC [58, 61, 63, 66, 69], LDL [57, 58, 61, 63, 69], and Tg [51, 58, 61, 69] and significant increases in HDL [57, 58, 61, 63, 66, 69] relative to standard care [51, 58, 63, 66], group education [61], or a moderate intensity exercise program [57, 69]. Only one RCT, a 12-week UK study of 59 adults, reported no significant improvement in any of the serum lipids following the yoga program [65], findings, again, likely in part attributable to poor participant compliance.
Overall, 94% of the studies reviewed reported improvements in lipid levels following completion of yogic programs of varying intensity and duration. Again, reported changes were clinically as well as statistically significant, with magnitude of effects varying substantially with the study and target population (Table 3). However, again, interpretation of findings is hampered by the lack of between group comparisons in several studies, as well as other design, methodologic, and/or reporting limitations characterizing many of the studies, discussed in more detail below.
3.1.3. Body Weight and Composition
Nine controlled trials have evaluated the potential influence of yoga on body weight and composition in adults with DM2, including 6 NRCTs and 3 RCTs; again, all but one study [65] (89%) documented improvements in those assigned to a yoga intervention. All of the yoga programs included active asanas. Trials ranged from 40 days [47] to 6 months [66] in duration, and, in all but the one trial reporting no significant benefits [65], participants practiced at least 3 times per week. Of the 6 NRCTs (N = 613 total participants), all reported significant beneficial changes in at least one measure of body composition, including reductions in weight [38, 44, 48], body mass index (BMI) [44, 45, 48, 49], and waist-hip ratio [44, 47, 49] relative to standard care [38, 44, 45, 48, 49] or standard care and light exercise [47]. Likewise, 2 of the 3 RCTs (N = 390 total participants), including a 6-month Cuban study in 231 adults [66] and a 3-month Indian trial of 100 adults [58], reported significant decreases in weight [58], BMI [66], and waist-hip ratio [58] in participants assigned to a yoga program versus standard care; yoga group improvements were again comparable to those documented following a moderate intensity physical activity program [66]. In contrast, a UK study of 59 adults showed no significant reduction in weight or BMI relative to standard care [65], although poor participant adherence renders interpretation of findings problematic.
3.1.4. Blood Pressure
Only 5 of the 25 controlled trials targeting adults with DM2 evaluated the potential effects of yogic practices on blood pressure, including 3 NRCTs and 2 RCTs (N = total of 588 participants). Trials varied in length from 40 days to 3 months, and all included active asanas. Three of the five studies, two NRCTs and one RCT [59], in Indian adults showed significant drops in systolic and diastolic blood pressure relative to standard care [49, 59] or standard care combined with walking [47]. Another Indian study of 123 adults [45] showed similar, but not statistically significant, declines in blood pressure in participants assigned to the yoga versus the standard care group. One exploratory RCT of British adults [65] did not present any quantitative data on blood pressure but reported no significant changes following a 12-week versus standard care program.
3.2. Observed Effects of Yoga on Other Pertinent Indices
There is mounting evidence, reviewed briefly below, that yoga may improve other risk indices pertinent to the DM2 management as well, including oxidative stress, impairments in mood and sleep, nervous system and pulmonary function, and medication usage. Oxidative stress, an imbalance between free radicals and antioxidants, has been strongly implicated in the development of DM2 and diabetes-related complications [74, 75]. For example, the oxidative imbalance that often characterizes DM2 can lead to prothrombotic changes, endothelial dysfunction, and chronic vascular inflammation, exacerbate insulin resistance and associated hyperglycemia, and ultimately promote organ damage; these reciprocally related changes are thought to mediate many of the atherosclerotic and thrombotic alterations associated with the metabolic syndrome and to play a central role in the pathogenesis and progression of diabetes and CVD [10, 75–78]. Thus, interventions that reduce oxidative stress in those with diabetes may aid in improving risk profiles and decreasing diabetes-related complications. To date, at least five controlled trials have examined the effects of yoga on measures of oxidative stress in adults with DM2. Yoga interventions all incorporated active asanas and ranged from 40 days to 6 months in duration; all indicated beneficial changes with yoga (Table 2). For example, four NRCTs in Indian adults documented improvements in indices of oxidative balance; these included significant declines in MDA [40, 45, 47] among participants completing a 40-day [47] to 4-month yoga program [40] versus those assigned to standard care [40, 45] or standard care with light exercise [47]; in their 3-month study of 123 adults, Hegde et al. also reported significantly greater increases in serum levels of the antioxidants glutathione and Vitamin C in the yoga versus standard care group [45]; likewise, Mahapure et al. noted significantly greater increases in superoxide dismutase (SOD) levels in the yoga group relative to the standard care control in their 6-week study [41]. Additionally, in an RCT of 231 Cuban adults, those assigned to a 24-week yoga program showed increases in activity of the antioxidant, SOD, and reductions in malondialdehyde (MDA), an estimate of lipid oxidative damage, that were comparable to those completing a 24-week conventional exercise program, and greater than participants assigned to standard care [66].
Mood and sleep impairment, common comorbid conditions in those with DM2, are reciprocally related to the development and progression of DM2 [16, 79]. For example, depression and sleep disturbance, both characterized by dysregulation of the hypothalamic-pituitary-adrenal (HPA) axis, sympathetic overactivity, and reduced cardiovagal tone [80–88], have been shown to increase subsequent risk for prediabetes [89] and incident diabetes [16, 90–98], impair metabolic control in persons with diabetes [99–104], and lead to excess morbidity and mortality [10, 22, 76, 93–95, 105–109]. However, although there is substantial evidence to suggest that yogic practices are helpful in reducing symptoms of depression and anxiety [35, 37, 80, 110, 111], alleviating stress [10, 80, 112, 113], improving sleep [80, 110, 111, 114–119], and enhancing psychological well-being [80, 112] in both healthy and clinical populations, few controlled trials have examined the effects of yoga on these endpoints in populations with DM2. Our search identified only four that specifically assessed indices of psychological status, including one NRCT [49] and three RCTs [53, 55, 64, 65]; significant improvements were noted with yogic practices in all but one study (Table 2). These included beneficial changes in multiple domains of quality of life [49, 53, 55], measures of psychological well-being [49], and symptoms of distress [64]. Studies regarding the effects of yoga on sleep in those with DM2 are even fewer. We identified only one trial of DM2 patients, an RCT conducted in India, that examined the effects of yoga on any measure of impaired sleep; in this study of 41 adults, participants completing a program of daily yoga nidra (a deep relaxation yoga practice) experienced a reduction in the prevalence of insomnia, from 43 to 5% [64].
3.2.1. Nervous System Function
Autonomic nervous system dysfunction, a correlate of obesity and poor cardiorespiratory fitness, has been strongly and bidirectionally related to insulin resistance and hypertension, implicated in the development of diabetes [120, 121] and CVD [122], and associated with increased risk for morbidity and mortality in individuals with DM2 [123, 124]. While more than 30 controlled studies, including 17 RCTs, have evaluated the effect of yoga on markers of sympathetic/parasympathetic activation and cardiovagal function [125, 126], our search identified only three that targeted adults with DM2, all conducted in India. These include two NRCTs [47, 127] assessing the effects of 3-month program of simple pranayama exercises [127] and a 40-day comprehensive yoga intervention [47] and an RCT of SKY yoga (a cyclical breathing practice) in adults enrolled in a diabetes lifestyle modification program that included daily brisk walking [54, 55] (Table 2). Participants who completed a pranayama program showed significant improvements in multiple indices of cardiac autonomic function [47, 55] and significant reductions in heart [47] and respiratory rate [127], relative to controls receiving standard care [55, 127] or standard care combined with exercise [47]. These findings suggest that yogic practices may promote a reduction in sympathetic activation, enhancement of cardiovagal function, and a shift in autonomic nervous system balance from primarily sympathetic to parasympathetic in adults with DM2. In addition, findings of three recent NRCTs suggest that yoga may also help mitigate the central and peripheral nervous system damage associated with DM2. For example, following completion of a 45-day daily yoga program, participants showed significant declines in the latency of event-related potentials (ERP), a marker of higher brain, including cognitive and memory function, and increases in ERP amplitude compared to those assigned to standard care [46]. Likewise, adults with DM2 who completed a 40-day yoga intervention showed significant improvements in certain measures of median nerve conduction velocity and amplitude relative to controls [47, 50], although incomplete information and lack of between group comparisons limit interpretation of findings.
3.2.2. Pulmonary Function
Compromised lung function is both an important complication [128, 129] and a significant predictor [130] of DM2 and has been inversely associated with insulin resistance [128] and glycemic exposure [129, 131]. While studies in adults with DM2 are limited, available data from two NRCTs support a possible beneficial influence of yogic practices on pulmonary function in this population [47, 127]. Reported improvements included significant increases in forced expiratory volume, forced vital capacity, peak expiratory flow rate, and maximum voluntary ventilation following completion of a 3-month program of simple yogic breathing exercises [127] or a 40-day comprehensive program [47].
3.2.3. Medication Use
Three controlled trials to date (two NRCTs [44, 49], one RCT [57]) have shown significant reductions in diabetes medication use in patients completing a three- [44, 49] to nine-month [57] yoga program relative to those assigned to standard care [44, 49] or a comprehensive exercise program [57]. Some of these declines were quite substantial. For example, in their trial of 154 adults with diabetes, Agrawal et al. reported 26 to 40% reductions in medication use in the yoga group at the 3-month follow-up [49].
4. Discussion
Overall, findings of these studies suggest that yoga-based practices may have significant beneficial effects on multiple factors important in DM2 management and prevention, including glycemic control, insulin resistance, lipid profiles, body composition, and blood pressure. These findings are further supported by recently published meta-analyses regarding the effects of yoga on specific CVD risk factors of relevance to DM2, detailed in Table 4 [34, 71–73]. For example, in subanalyses restricted to RCTs in adults with DM2, Cramer et al. reported greater average declines of 26 mg/dL in FBG and 0.5% in HbA1c, greater mean reductions of 13 mg/dL in TC, 10 mg/dL in LDL, 5 mg/dL in VLDL, and 24 mg/dL in Tg levels, and a higher average increase of 6 mg/dL in HDL in participants assigned to a yoga versus standard care group [34]. Notably, the authors also found significantly greater reductions in LDL (9 mg/dL) and higher increases in HDL levels (4 mg/dL) in the yoga versus conventional exercise group. Likewise, Cramer et al. also reported greater mean reductions in waist-hip ratio in participants with DM2 assigned to a yoga versus a standard care group [34]. Similarly, in a subanalysis of RCTs in adults with diabetes or metabolic syndrome, Chu et al. reported a mean BMI reduction of over 1.6 kg/m2 in those in the yoga versus control group [71]. While studies regarding the effects of yoga on blood pressure in adults with DM2 remain few, recent meta-analyses regarding the effects of yoga on other populations at risk for CVD suggest that yoga may be beneficial for regulating blood pressure in patients with diabetes as well. For example, in subanalyses of RCTs limited to 8 trials of adults without frank diabetes but at high risk of DM2 and CVD (e.g., those with obesity, impaired glucose tolerance, or metabolic syndrome), the authors noted significant mean declines of 10 mmHg in systolic blood pressure and 7.5 mmHg in diastolic blood pressure in participants receiving a yoga intervention versus standard care [34]. Meta-analyses of controlled trials in adults with hypertension have yielded similar findings [72, 73], indicating significant declines of 8–10 mmHg in systolic blood pressure and 6-7 mmHg in diastolic blood pressure in the yoga versus standard care groups (Table 4).
More limited data suggest that yoga may also lower oxidative stress, decrease sympathetic activation and improve nervous system function, enhance pulmonary performance, mood, sleep, and quality of life, and reduce medication use in those with DM2. Relatively few studies have examined the effects of yogic practices on psychological status and sleep, which are frequently compromised in DM2 and significant contributors to diabetes progression. Likewise, studies evaluating the effects of yoga on clinical outcomes and certain emerging risk factors in DM2, including proinflammatory markers, remain sparse, and studies to determine optimal yoga dosing and program structure for different DM2 populations are lacking. As indicated in Table 1, the vast majority of controlled studies published to date regarding the effects of yoga for DM2 have been conducted in India, underscoring the need for rigorous trials in other racial/ethnic groups and in other developed and developing countries. Finally, as discussed in more detail below and reflected in the study quality scores (Table 2), the methodological limitations characterizing existing studies highlight the need for additional high-quality RCTs that address these concerns.
5. Yoga and DM2: Possible Underlying Mechanisms
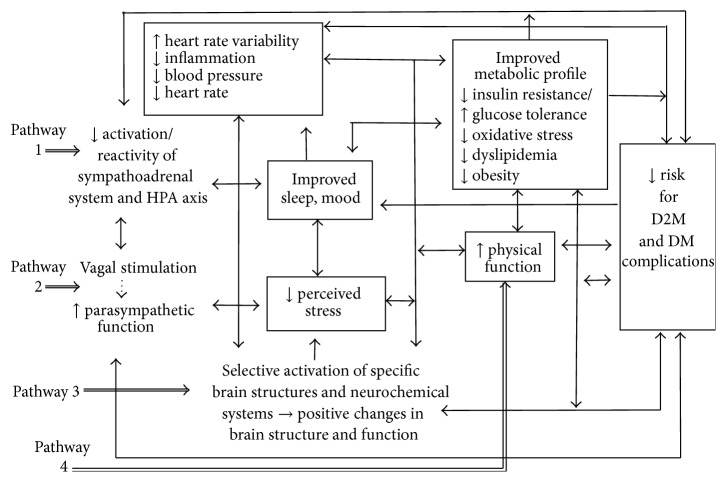
Although the mechanisms underlying the apparent beneficial effects of yoga therapy on diabetes risk profiles are not yet well understood, mechanistic pathways are likely complex and interacting. The observed changes may occur through at least four pathways, as depicted in Figure 1 [10, 76, 113, 117, 125, 132].
Figure 1.
Some pathways by which yoga practices may influence outcomes in those with and at risk for type 2 diabetes (DM2). Figure adapted with permission from Hansen and Innes [132].
First, yoga may lessen the negative impact of stress and promote multiple positive downstream effects on metabolic function, neuroendocrine status, and related inflammatory responses and, ultimately, reduce risk for CVD and other vascular complications, by enhancing well-being and reducing reactivity and activation of the HPA axis and the sympathoadrenal system [10, 76, 113, 132]. For instance, recent controlled trials suggest even short term yoga training programs can reduce perceived stress, improve mood, and lower catecholamine and cortisol levels, cardiovascular response to stress, blood pressure, and other indices of sympathetic activation in both healthy and clinical populations, including adults with diabetes [10, 35, 80, 113, 132–134]. Sympathetic activation, HPA dysregulation, and psychological distress have also been linked to the development and exacerbation of several risk factors for DM2 [10, 76, 80, 83, 88, 132, 133, 135, 136], including insulin resistance, impaired glucose tolerance, hypertension, dyslipidemia, central obesity, and proinflammatory and prothrombotic states. These conditions have been shown to have adverse effects on metabolic control and neuroendocrine function and have also been implicated in elevated risk for DM2, CVD, and other vascular disorders [10, 76, 80, 83, 88, 132, 133, 135].
Second, yogic practices may shift the autonomic nervous system balance from primarily sympathetic to parasympathetic, by directly enhancing parasympathetic output, possibly via vagal stimulation [76, 122, 137], resulting in positive changes in cardiovagal function and associated neuroendocrine, hemodynamic, and inflammatory profiles, in sleep and affect, and in related downstream metabolic parameters (Figure 1, pathway 2) [10, 80, 113, 133]. For example, recent controlled studies in adults with DM2, CVD, hypertension, and other chronic conditions, as well as in healthy populations, have shown yogic exercises to reduce resting heart rate, enhance baroreflex sensitivity, and increase heart rate variability, both immediately and following short term (6–12 weeks) yoga programs [10, 113, 133, 138, 139]. Heart rate variability, resting heart rate, and baroreflex sensitivity are widely used markers of cardiovagal autonomic function, parasympathetic activation, and autonomic balance [76, 120, 140, 141] and are thought to in part reflect stimulation of the vagus nerve [76, 122, 137]. These factors have been strongly linked to increased risk for DM2, as well as CVD [10, 76, 80, 142].
Third, yoga may also promote favorable changes in autonomic balance, memory and mood, neurological structure and function, and related metabolic and inflammatory responses by selectively activating specific brain structures and neurochemical systems related to attention and positive affect (Figure 1, pathway 3), as suggested by recent neurophysiological and neuroimaging research findings [143–146]. And finally, yogic practices may improve both metabolic and psychological risk profiles, support increased physical activity, enhance neuroendocrine function, improve body composition, and promote weight loss by increasing strength, overall fitness, and physical function (Figure 1, pathway 4). Yoga may also reduce CVD risk in other ways. For instance, by reducing stress and leading to improved sleep and mood, yoga may indirectly improve CVD risk profiles by leading to healthier lifestyle choices and enhanced self-care [147]. Yoga may also increase resilience to stress, a factor that has been linked to improved outcomes in DM2 [148, 149], although study findings to date have been inconsistent [150–152].
In addition, yoga may benefit those with DM2 indirectly by encouraging improvements in health-related attitudes and lifestyle choices [147, 153, 154] and by providing a source of social support, a factor linked to improved diabetes self-care and clinical outcomes [155, 156]. As suggested by recent studies of patients with heart failure, chronic obstructive pulmonary disease, and DM1 [157–159], yogic breathing practices may also, by increasing arterial and tissue oxygenation, alleviate underlying hypoxia and thereby enhance autonomic cardiac and respiratory function and related endpoints in adults with diabetes [157–160]. Finally, several recent genomic investigations in dementia caregivers [161, 162] and healthy adults [163, 164] suggest that yogic meditative practices can slow cellular aging and induce beneficial epigenetic changes in pathways regulating inflammation, oxidative stress, energy metabolism, insulin secretion, mitochondrial function, and other related factors; these changes may, in turn, help buffer the deleterious effects of stress, improve glucose control, enhance mood, sleep, and autonomic function, reduce blood pressure, and promote improvements in other related risk factors of relevance to DM2 management [76, 165, 166].
6. Study Limitations and Directions for Future Research
This systematic review was limited in that it did not include unpublished studies, dissertations, or abstracts. In addition, some controlled trials of yogic practices may have been missed if the article did not make reference to the word yoga or yogic, although every effort was made to locate all relevant studies meeting our inclusion criteria. Many of the studies reviewed here suffer from methodological problems, poor reporting, and other limitations that render interpretation of findings challenging and the formation of definitive conclusions difficult. PEDro scores ranged from a low of 1 [38, 40, 43, 64] to a high of 6 [57, 59, 61, 62, 66, 67] points, out of 10 (Table 2). Importantly, participants were not randomly assigned to treatment in more than half of the studies, increasing risk for selection bias, and concealment of treatment allocation was reported in only 3 [57, 61, 65] of the 25 trials. Among the 12 RCTs, only 2 [57, 65] specified how randomization was performed, and only 7 [52–56, 58, 59, 61–63, 66–68, 70] demonstrated baseline comparability between the groups regarding the most important prognostic indicators. Interestingly, 8 [39, 44–50] of the 13 NRCTs also exhibited groups that were similar at baseline.
Given the nature of the intervention, blinding of the therapists and participants was likely not feasible; understandably, none of the studies satisfied these two PEDro criteria. This deficiency in blinding potential places even greater importance on ensuring that outcome assessors are blinded to group assignment. Unfortunately, blinded outcome assessment was specifically addressed in only two trials [57, 66, 67], raising the possibility of information bias. Retention was not always reported and varied widely (from 60% to 100%), with less than half of the studies (n = 11 [39, 42, 45, 50, 56, 58–62, 66, 67, 69, 70]) reporting outcome data from 85% or more of those initially assigned to a group. Reasons for dropout were rarely specified. Only five studies included specific data on participant adherence [45, 56, 65, 67, 70], and even in these studies, adherence was highly variable, ranging from excellent [45, 67] to very poor [65]; moreover, for one study [53], adherence data were reported for only a subset of participants [56].
In some articles, details regarding analytic methods were sparse, and/or analyses were inadequate. For example, while all studies in this review included a comparator group, between group comparisons were not presented in 10 of the 25 studies, including 2 [60, 64] RCTs. In those studies documenting attrition, only eight [39, 42, 50, 57, 59, 60, 65, 69] indicated all participants completed the study as designed or that intention to treat analyses had been conducted. Additionally, although all but two [64, 65] studies provided point measures and measures of variability, other data were often incompletely or confusingly presented. Furthermore, in many papers, the study population, participant recruitment, yoga-based intervention, and/or comparator condition were poorly described, rendering replication of these studies challenging and limiting conclusions. Most of the trials were relatively small, with 56% of them including 60 or fewer participants and 24% including 40 or less (Table 1), although, overall, sample size has been increasing in the last decade, especially the last 5 years.
While the investigations published to date have yielded overall positive results, the considerable heterogeneity in study design, intervention, comparator condition, and duration characterizing the 25 trials render direct comparisons across studies challenging. Participant characteristics also varied substantially, both within and between studies. Nonetheless, while this heterogeneity precludes specific recommendations for particular yoga programs or subpopulations, the consistently positive findings reported in multiple and varied samples of adults with DM2 suggest that yoga may be helpful for a broad range of patient groups. Of the 25 eligible controlled studies identified in this review, only five were conducted in countries other than India, including two in the UK, two in Cuba, and one in Iran. Rigorous trials in Western and many other countries, including many developing nations, where DM2 is now reaching epidemic proportions, remain few. It thus remains unclear if the positive findings reported in existing studies can be generalized to other populations, cultures, and/or regions. As indicated earlier, the mechanisms underlying the observed benefits of yoga are still poorly understood. Finally, rigorous dosing, cost-effectiveness, and long-term follow-up studies are lacking, as are trials assessing the effects of yoga on clinical endpoints such as diabetes-related morbidity and mortality. All of these areas warrant future research.
7. Conclusion
In conclusion, the findings of controlled trials published to date suggest that yogic practices may promote significant improvements in several indices of major importance in the management of DM2, including glycemic control, lipid levels, and body composition. More limited data suggest that yoga may also lower oxidative stress and blood pressure, enhance pulmonary and nervous system function, improve mood, sleep, and quality of life, and reduce medication use in adults with DM2. However, given the methodological limitations and heterogeneity of existing studies, findings must be interpreted with caution. Additional high-quality investigations are required to confirm and further elucidate the potential therapeutic benefits of standardized yoga programs in populations with DM2 [10, 132].
Acknowledgments
This work was performed at West Virginia University and was supported by the National Center for Complementary and Alternative Medicine [Grant no. 1-K01-AT004108 to KEI] and West Virginia University (Faculty Incentive Award).
Disclaimer
The contents are solely the responsibility of the authors and do not represent the official views of West Virginia University or the National Institutes of Health.
Conflict of Interests
The authors declare that there is no conflict of interests regarding the publication of this paper.
References
- 1.Lam D. W., LeRoith D. The worldwide diabetes epidemic. Current Opinion in Endocrinology, Diabetes and Obesity. 2012;19(2):93–96. doi: 10.1097/med.0b013e328350583a. [DOI] [PubMed] [Google Scholar]
- 2.Chen L., Magliano D. J., Zimmet P. Z. The worldwide epidemiology of type 2 diabetes mellitus—present and future perspectives. Nature Reviews Endocrinology. 2012;8(4):228–236. doi: 10.1038/nrendo.2011.183. [DOI] [PubMed] [Google Scholar]
- 3.Inzucchi S. E., Bergenstal R. M., Buse J. B., et al. Management of hyperglycemia in type 2 diabetes: a patient-centered approach: position statement of the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD) Diabetes care. 2012;35(6):1364–1379. doi: 10.2337/dc12-0413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.International Diabetes Federation. IDF Diabetes Atlas. Brussels, Belgium: International Diabetes Federation; 2011. [Google Scholar]
- 5.Fowler M. J. Microvascular and macrovascular complications of diabetes. Clinical Diabetes. 2011;29(3):116–122. doi: 10.2337/diaclin.29.3.116. [DOI] [Google Scholar]
- 6.American Diabetes Association. Standards of medical care in diabetes—2014. Diabetes Care. 2014;37(supplement 1):S14–S80. doi: 10.2337/dc14-s014. [DOI] [PubMed] [Google Scholar]
- 7.Joseph J. J., Golden S. H. Type 2 diabetes and cardiovascular disease: what next? Current Opinion in Endocrinology, Diabetes and Obesity. 2014;21(2):109–120. doi: 10.1097/med.0000000000000044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.American Heart Association. Cardiovascular Disease & Diabetes, 2012, http://www.heart.org/HEARTORG/Conditions/Diabetes/WhyDiabetesMatters/Cardiovascular-Disease-Diabetes_UCM_313865_Article.jsp/
- 9.Petersen M. Economic costs of diabetes in the US in 2012. Diabetes Care. 2012;36(4):1033–1046. doi: 10.2337/dc12-2625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Innes K. E., Vincent H. K. The influence of yoga-based programs on risk profiles in adults with type 2 diabetes mellitus: a systematic review. Evidence-based Complementary and Alternative Medicine. 2007;4(4):469–486. doi: 10.1093/ecam/nel103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wellen K. E., Hotamisligil G. S. Inflammation, stress, and diabetes. Journal of Clinical Investigation. 2005;115(5):1111–1119. doi: 10.1172/JCI200525102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Rana J. S., Nieuwdorp M., Jukema J. W., Kastelein J. J. P. Cardiovascular metabolic syndrome—an interplay of, obesity, inflammation, diabetes and coronary heart disease. Diabetes, Obesity & Metabolism. 2007;9(3):218–232. doi: 10.1111/j.1463-1326.2006.00594.x. [DOI] [PubMed] [Google Scholar]
- 13.Yoon K.-H., Lee J.-H., Kim J.-W., et al. Epidemic obesity and type 2 diabetes in Asia. The Lancet. 2006;368(9548):1681–1688. doi: 10.1016/s0140-6736(06)69703-1. [DOI] [PubMed] [Google Scholar]
- 14.Zimmet P., Shaw J., Murray S., Sicree R. Diabetes: an epidemic in full flight: forecasting the future. Bulletin of the International Diabetes Federation. 2003;48:12–16. [Google Scholar]
- 15.Hu F. B. Globalization of diabetes. The role of diet, lifestyle, and genes. Diabetes Care. 2011;34(6):1249–1257. doi: 10.2337/dc11-0442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cappuccio F. P., D'Elia L., Strazzullo P., Miller M. A. Quantity and quality of sleep and incidence of type 2 diabetes: a systematic review and meta-analysis. Diabetes Care. 2010;33(2):414–420. doi: 10.2337/dc09-1124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Barone M. T. U., Menna-Barreto L. Diabetes and sleep: a complex cause-and-effect relationship. Diabetes Research and Clinical Practice. 2011;91(2):129–137. doi: 10.1016/j.diabres.2010.07.011. [DOI] [PubMed] [Google Scholar]
- 18.Lin C.-C., Li C.-I., Liu C.-S., et al. Impact of lifestyle-related factors on all-cause and cause-specific mortality in patients with type 2 diabetes: the Taichung diabetes study. Diabetes Care. 2012;35(1):105–112. doi: 10.2337/dc11-0930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Black S. A., Markides K. S., Ray L. A. Depression predicts increased incidence of adverse health outcomes in older Mexican Americans with type 2 diabetes. Diabetes Care. 2003;26(10):2822–2828. doi: 10.2337/diacare.26.10.2822. [DOI] [PubMed] [Google Scholar]
- 20.Atlantis E., Fahey P., Foster J. Collaborative care for comorbid depression and diabetes: a systematic review and meta-analysis. BMJ Open. 2014;4(4) doi: 10.1136/bmjopen-2013-004706.e004706 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Beverly E. A. Stressing the importance of diabetes distress: a comment on Baek et al. Annals of Behavioral Medicine. 2014;48(2):137–139. doi: 10.1007/s12160-014-9602-2. [DOI] [PubMed] [Google Scholar]
- 22.Bruce D. G., Davis W. A., Starkstein S. E., Davis T. M. E. A prospective study of depression and mortality in patients with type 2 diabetes: the Fremantle Diabetes Study. Diabetologia. 2005;48(12):2532–2539. doi: 10.1007/s00125-005-0024-3. [DOI] [PubMed] [Google Scholar]
- 23.Hessler D., Fisher L., Glasgow R. E., et al. Reductions in regimen distress are associated with improved management and glycemic control over time. Diabetes Care. 2014;37(3):617–624. doi: 10.2337/dc13-0762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Safren S. A., Gonzalez J. S., Wexler D. J., et al. A randomized controlled trial of cognitive behavioral therapy for adherence and depression (CBT-AD) in patients with uncontrolled type 2 diabetes. Diabetes Care. 2014;37(3):625–633. doi: 10.2337/dc13-0816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Fishman L., Saltonstall E. Integrative Pain Medicine: The Science and Practice of Complementary and Alternative Medicine in Pain Management. Humana Press; 2008. Yoga in pain management; pp. 259–284. [DOI] [Google Scholar]
- 26.Garfinkel M., Schumacher H. R. Yoga. Rheumatic Disease Clinics of North America. 2000;26(1):125–132. doi: 10.1016/s0889-857x(05)70126-5. [DOI] [PubMed] [Google Scholar]
- 27.Chandler K. The emerging field of yoga therapy. Hawaii Medical Journal. 2001;60(11):286–287. [PubMed] [Google Scholar]
- 28.Raub J. A. Psychophysiologic effects of Hatha Yoga on musculoskeletal and cardiopulmonary function: a literature review. Journal of Alternative and Complementary Medicine. 2002;8(6):797–812. doi: 10.1089/10755530260511810. [DOI] [PubMed] [Google Scholar]
- 29.Chappie C. K. Modern yoga. Religious Studies Review. 2008;34(2):71–76. doi: 10.1111/j.1748-0922.2008.00256.x. [DOI] [Google Scholar]
- 30.Jeter P. E., Slutsky J., Singh N., Khalsa S. B. Yoga as a therapeutic intervention: a bibliometric analysis of published research studies from 1967 to 2013. The Journal of Alternative and Complementary Medicine. 2015 doi: 10.1089/acm.2015.0057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ross A., Thomas S. The health benefits of yoga and exercise: a review of comparison studies. Journal of Alternative and Complementary Medicine. 2010;16(1):3–12. doi: 10.1089/acm.2009.0044. [DOI] [PubMed] [Google Scholar]
- 32.Büssing A., Michalsen A., Khalsa S. B. S., Telles S., Sherman K. J. Effects of yoga on mental and physical health: a short summary of reviews. Evidence-Based Complementary and Alternative Medicine. 2012;2012:7. doi: 10.1155/2012/165410.165410 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Patel N. K., Newstead A. H., Ferrer R. L. The effects of yoga on physical functioning and health related quality of life in older adults: a systematic review and meta-analysis. Journal of Alternative and Complementary Medicine. 2012;18(10):902–917. doi: 10.1089/acm.2011.0473. [DOI] [PubMed] [Google Scholar]
- 34.Cramer H., Lauche R., Haller H., Steckhan N., Michalsen A., Dobos G. Effects of yoga on cardiovascular disease risk factors: a systematic review and meta-analysis. International Journal of Cardiology. 2014;173(2):170–183. doi: 10.1016/j.ijcard.2014.02.017. [DOI] [PubMed] [Google Scholar]
- 35.Uebelacker L. A., Epstein-Lubow G., Gaudiano B. A., Tremont G., Battle C. L., Miller I. W. Hatha yoga for depression: Critical review of the evidence for efficacy, plausible mechanisms of action, and directions for future research. Journal of Psychiatric Practice. 2010;16(1):22–33. doi: 10.1097/01.pra.0000367775.88388.96. [DOI] [PubMed] [Google Scholar]
- 36.Balasubramaniam M., Telles S., Doraiswamy P. M. Yoga on our minds: a systematic review of yoga for neuropsychiatric disorders. Frontiers in Psychiatry. 2013;3, article 117 doi: 10.3389/fpsyt.2012.00117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Cramer H., Lauche R., Langhorst J., Dobos G. Yoga for depression: a systematic review and meta-analysis. Depression and Anxiety. 2013;30(11):1068–1083. doi: 10.1002/da.22166. [DOI] [PubMed] [Google Scholar]
- 38.Khare K., Jain D. Effect of yoga on plasma glucose and serum fructosamine level in NIDDM. Yoga Mimamsa. 1999;33:1–9. [Google Scholar]
- 39.Bindra M., Nair S., Darotiya S. Influence of pranayamas and yoga-asanas on blood glucose, lipid profile and HbA1c in type 2 diabetes. International Journal of Pharma and Bio Sciences. 2013;4(1):169–172. [Google Scholar]
- 40.Agte V. V., Tarwadi K. Sudarshan kriya yoga for treating type 2 diabetes: a preliminary study. Alternative and Complementary Therapies. 2004;10(4):220–222. doi: 10.1089/1076280041580323. [DOI] [Google Scholar]
- 41.Mahapure H. H., Shete S. U., Bera T. K. Effect of yogic exercise on super oxide dismutase levels in diabetics. International Journal of Yoga. 2008;1(1):21–26. doi: 10.4103/0973-6131.36792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Dash S., Thakur A. K. Effect of yoga in patient's with type-II diabetes mellitus. Journal of Evolution of Medical and Dental Sciences. 2014;3(7):1642–1655. doi: 10.14260/jemds/2014/2038. [DOI] [Google Scholar]
- 43.Popli U., Subbe C. P., Sunil K. Research letter-the role of yoga as a lifestyle modification in treatment of diabetes mellitus: results of a pilot study. Alternative Therapies in Health and Medicine. 2014;20(6):24–26. [PubMed] [Google Scholar]
- 44.Balaji P. A., Varne S. R., Ali S. S. Effects of yoga—pranayama practices on metabolic parameters and anthropometry in type 2 diabetes. International Multidisciplinary Research Journal. 2011;1(10):1–4. [Google Scholar]
- 45.Hegde S. V., Adhikari P., Kotian S., Pinto V. J., D'Souza S., D'Souza V. Effect of 3-month yoga on oxidative stress in type 2 diabetes with or without complications: a controlled clinical trial. Diabetes Care. 2011;34(10):2208–2210. doi: 10.2337/dc10-2430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kyizom T., Singh S., Singh K. P., Tandon O. P., Kumar R. Effect of pranayama & yoga-asana on cognitive brain functions in type 2 diabetes-P3 event related evoked potential (ERP) Indian Journal of Medical Research. 2010;131(5):636–640. [PubMed] [Google Scholar]
- 47.Malhotra V., Singh S., Sharma S. B., et al. The status of NIDDM patients after yoga asanas: assessment of important parameters. Journal of Clinical and Diagnostic Research. 2010;4(3):2652–2667. [Google Scholar]
- 48.Singh S., Kyizom T., Singh K. P., Tandon O. P., Madhu S. V. Influence of pranayamas and yoga-asanas on serum insulin, blood glucose and lipid profile in type 2 diabetes. Indian Journal of Clinical Biochemistry. 2008;23(4):365–368. doi: 10.1007/s12291-008-0080-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Agrawal R., Aradhana R., Hussain S., Beniwal R., Sabir M., Kochar D. Influence of yogic treatment on quality of life outcomes, glycaemic control and risk factors in diabetes mellitus. International Journal of Diabetes in Developing Countries. 2003;23:130–134. [Google Scholar]
- 50.Malhotra V., Singh S., Tandon O. P., Madhu S. V., Prasad A., Sharma S. B. Effect of Yoga asanas on nerve conduction in type 2 diabetes. Indian Journal of Physiology & Pharmacology. 2002;46(3):298–306. [PubMed] [Google Scholar]
- 51.Habibi N., Marandi S. Effect of 12 weeks of yoga practice on glucose, insulin and triglycerides serum level in women with diabetes type II. Journal of Gorgan University of Medical Sciences. 2014;15(4):1–7. [Google Scholar]
- 52.Habibi N., Farsani Z., Yazdani B., Arianshakib R., Noruozi P. The influence of yoga on risk profiles programs in women with diabetes type II. Advances in Environmental Biology. 2013;7(4):550–555. [Google Scholar]
- 53.Jyotsna V. P., Dhawan A., Sreenivas V., Deepak K. K., Singla R. Completion report: effect of Comprehensive Yogic Breathing program on type 2 diabetes: a randomized control trial. Indian Journal of Endocrinology and Metabolism. 2014;18(4):582–584. doi: 10.4103/2230-8210.137499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Jyotsna V. P., Ambekar S., Singla R., et al. Cardiac autonomic function in patients with diabetes improves with practice of comprehensive yogic breathing program. Indian Journal of Endocrinology and Metabolism. 2013;17(3):480–485. doi: 10.4103/2230-8210.111645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Jyotsna V. P., Ambekar S., Joshi A., et al. Prospective randomized controlled intervention trial: comprehensive yogic breathing program improves cardiac autonomic functions and quality of life in diabetes. Indian Journal of Endocrinology and Metabolism. 2012;16(supplement 2):S489–S491. doi: 10.4103/2230-8210.95692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Jyotsna V. P., Joshi A., Ambekar S., Kumar N., Dhawan A., Sreenivas V. Comprehensive yogic breathing program improves quality of life in patients with diabetes. Indian Journal of Endocrinology and Metabolism. 2012;16(3):423–428. doi: 10.4103/2230-8210.95692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Nagarathna R., Usharani M. R., Rao A. R., Chaku R., Kulkarni R., Nagendra H. R. Efficacy of yoga based life style modification program on medication score and lipid profile in type 2 diabetes-a randomized control study. International Journal of Diabetes in Developing Countries. 2012;32(3):122–130. doi: 10.1007/s13410-012-0078-y. [DOI] [Google Scholar]
- 58.Shantakumari N., Sequeira S., El Deeb R. Effects of a yoga intervention on lipid profiles of diabetes patients with dyslipidemia. Indian Heart Journal. 2013;65(2):127–131. doi: 10.1016/j.ihj.2013.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Shantakumari N., Sequeira S., Eldeeb R. Effect of a yoga intervention on hypertensive diabetic patients. Journal of Advances in Internal Medicine. 2012;1(2):60–63. doi: 10.3126/jaim.v1i2.6526. [DOI] [Google Scholar]
- 60.Subramaniyan T. G., Subramaniyan N., Chidambaram M. Brisk walking and yoga as adjuvant therapy in management of type 2 diabetes mellitus. International Journal of Students Research. 2012;2(1):43–46. doi: 10.4103/2230-7095.113492. [DOI] [Google Scholar]
- 61.Vaishali K., Kumar K. V., Adhikari P., UnniKrishnan B. Effects of yoga-based program on glycosylated hemoglobin level serum lipid profile in community dwelling elderly subjects with chronic type 2 diabetes mellitus—a randomized controlled trial. Physical & Occupational Therapy in Geriatrics. 2012;30(1):22–30. doi: 10.3109/02703181.2012.656835. [DOI] [Google Scholar]
- 62.Vaishali K., Kumar K. V., Adhikari P., Unni Krishnan B. Effects of yoga-based program on glycosylated hemoglobin level and serum lipid profile in community dwelling elderly subjects with chronic type 2 diabetes mellitus—a randomized controlled trial. Indian Journal of Ancient Medicine and Yoga. 2011;4(2):69–76. [Google Scholar]
- 63.Pardasany A., Shenoy S., Sandhu J. S. Comparing the efficacy of tai chi chuan and hatha yoga in type 2 diabetes mellitus patients on parameters of blood glucose control and lipid metabolism. Indian Journal of Physiotherapy and Occupational Therapy. 2010;4(3):11–16. [Google Scholar]
- 64.Amita S., Prabhakar S., Manoj I., Harminder S., Pavan T. Effect of Yoga-Nidra on blood glucose level in diabetic patients. Indian Journal of Physiology and Pharmacology. 2009;53(1):97–101. [PubMed] [Google Scholar]
- 65.Skoro-Kondza L., Tai S. S., Gadelrab R., Drincevic D., Greenhalgh T. Community based yoga classes for type 2 diabetes: an exploratory randomised controlled trial. BMC Health Services Research. 2009;9, article 33 doi: 10.1186/1472-6963-9-33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Gordon L. A., Morrison E. Y., McGrowder D. A., et al. Effect of exercise therapy on lipid profile and oxidative stress indicators in patients with type 2 diabetes. BMC Complementary and Alternative Medicine. 2008;8, article 21 doi: 10.1186/1472-6882-8-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Gordon L., Morrison E. Y., McGrowder D. A., et al. Changes in clinical and metabolic parameters after exercise therapy in patents with type 2 diabetes. Archives of Medical Science. 2008;4(4):427–437. [Google Scholar]
- 68.Gordon L., Morrison E. Y., McGrowder D., et al. Effect of yoga and traditional physical exercise on hormones and percentage insulin binding receptor in patients with type 2 diabetes. American Journal of Biochemistry and Biotechnology. 2008;4(1):35–42. doi: 10.3844/ajbbsp.2008.35.42. [DOI] [Google Scholar]
- 69.Céspedes E. M., Riverón G., Alonso C. A., Gordon L. Evolución metabólica de pacientes diabéticos tipo 2 sometidos a un tratamiento combinado de dieta y ejercicios yoga. Revista Cubana de Investigaciones Biomedicas. 2002;21(2):1–5. [Google Scholar]
- 70.Monro R., Power J., Coumar A., Nagarathna R., Dandona P. Yoga therapy for NIDDM: a controlled trial. Complementary Medical Research. 1992;6(2):66–68. [Google Scholar]
- 71.Chu P., Gotink R. A., Yeh G. Y., Goldie S. J., Hunink M. G. M. The effectiveness of yoga in modifying risk factors for cardiovascular disease and metabolic syndrome: a systematic review and meta-analysis of randomized controlled trials. European Journal of Preventive Cardiology. 2014 doi: 10.1177/2047487314562741. [DOI] [PubMed] [Google Scholar]
- 72.Cramer H., Haller H., Lauche R., Steckhan N., Michalsen A., Dobos G. A systematic review and meta-analysis of yoga for hypertension. American Journal of Hypertension. 2014;27(9):1146–1151. doi: 10.1093/ajh/hpu078. [DOI] [PubMed] [Google Scholar]
- 73.Hagins M., States R., Selfe T., Innes K. Effectiveness of yoga for hypertension: systematic review and meta-analysis. Evidence-Based Complementary and Alternative Medicine. 2013;2013:13. doi: 10.1155/2013/649836.649836 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Ceriello A. Oxidative stress and diabetes-associated complications. Endocrine Practice. 2006;12(supplement 1):60–62. doi: 10.4158/ep.12.s1.60. [DOI] [PubMed] [Google Scholar]
- 75.Vincent H. K., Innes K. E., Vincent K. R. Oxidative stress and potential interventions to reduce oxidative stress in overweight and obesity. Diabetes, Obesity and Metabolism. 2007;9(6):813–839. doi: 10.1111/j.1463-1326.2007.00692.x. [DOI] [PubMed] [Google Scholar]
- 76.Innes K. E., Vincent H. K., Taylor A. G. Chronic stress and insulin-resistance-related indices of cardiovascular disease risk. Part I. Neurophysiological responses and pathological sequelae. Alternative Therapies in Health and Medicine. 2007;13(4):46–52. [PubMed] [Google Scholar]
- 77.Maritim A. C., Sanders R. A., Watkins J. B., III Diabetes, oxidative stress, and antioxidants: a review. Journal of Biochemical & Molecular Toxicology. 2003;17(1):24–38. doi: 10.1002/jbt.10058. [DOI] [PubMed] [Google Scholar]
- 78.Stadler K. Oxidative stress in diabetes. In: Ahmad S. I., editor. Diabetes Advances in Experimental Medicine and Biology. New York, NY, USA: Landes Bioscience and Springer Science; 2013. pp. 272–287. [DOI] [PubMed] [Google Scholar]
- 79.Nouwen A., Winkley K., Twisk J., et al. Type 2 diabetes mellitus as a risk factor for the onset of depression: a systematic review and meta-analysis. Diabetologia. 2010;53(12):2480–2486. doi: 10.1007/s00125-010-1874-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Innes K. E., Selfe T. K., Taylor A. G. Menopause, the metabolic syndrome, and mind-body therapies. Menopause. 2008;15(5):1005–1013. doi: 10.1097/01.gme.0b013e318166904e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Innes K. E., Vincent H. K., Taylor A. G. Chronic stress and insulin resistance-related indices of cardiovascular disease risk, part I: neurophysiological responses and pathological sequelae. Alternative Therapies in Health and Medicine. 2007;13(4):46–52. [PubMed] [Google Scholar]
- 82.Weinstein A. A., Deuster P. A., Kop W. J. Heart rate variability as a predictor of negative mood symptoms induced by exercise withdrawal. Medicine & Science in Sports & Exercise. 2007;39(4):735–741. doi: 10.1249/mss.0b013e31802f590c. [DOI] [PubMed] [Google Scholar]
- 83.Golden S. H. A review of the evidence for a neuroendocrine link between stress, depression and diabetes mellitus. Current Diabetes Reviews. 2007;3(4):252–259. doi: 10.2174/157339907782330021. [DOI] [PubMed] [Google Scholar]
- 84.Kyrou I., Chrousos G. P., Tsigos C. Stress, visceral obesity, and metabolic complications. Annals of the New York Academy of Sciences. 2006;1083:77–110. doi: 10.1196/annals.1367.008. [DOI] [PubMed] [Google Scholar]
- 85.Reagan L. P., Grillo C. A., Piroli G. G. The As and Ds of stress: metabolic, morphological and behavioral consequences. European Journal of Pharmacology. 2008;585(1):64–75. doi: 10.1016/j.ejphar.2008.02.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Mausbach B. T., Ancoli-Israel S., von Känel R., et al. Sleep disturbance, norepinephrine, and D-dimer are all related in elderly caregivers of people with Alzheimer disease. Sleep. 2006;29(10):1347–1352. doi: 10.1093/sleep/29.10.1347. [DOI] [PubMed] [Google Scholar]
- 87.McEwen B. S. Sleep deprivation as a neurobiologic and physiologic stressor: allostasis and allostatic load. Metabolism. 2006;55(10, supplement 2):S20–S23. doi: 10.1016/j.metabol.2006.02.003. [DOI] [PubMed] [Google Scholar]
- 88.McEwen B. S. Central effects of stress hormones in health and disease: understanding the protective and damaging effects of stress and stress mediators. European Journal of Pharmacology. 2008;583(2-3):174–185. doi: 10.1016/j.ejphar.2007.11.071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Whincup P. H., Kaye S. J., Owen C. G., et al. Birth weight and risk of type 2 diabetes: a systematic review. The Journal of the American Medical Association. 2008;300(24):2886–2897. doi: 10.1001/jama.2008.886. [DOI] [PubMed] [Google Scholar]
- 90.Knol M. J., Twisk J. W., Beekman A. T., Heine R. J., Snoek F. J., Pouwer F. Depression as a risk factor for the onset of type 2 diabetes mellitus. A meta-analysis. Diabetologia. 2006;49(5):837–845. doi: 10.1007/s00125-006-0159-x. [DOI] [PubMed] [Google Scholar]
- 91.Eriksson A.-K., Ekbom A., Granath F., Hilding A., Efendic S., Östenson C.-G. Psychological distress and risk of pre-diabetes and Type 2 diabetes in a prospective study of Swedish middle-aged men and women. Diabetic Medicine. 2008;25(7):834–842. doi: 10.1111/j.1464-5491.2008.02463.x. [DOI] [PubMed] [Google Scholar]
- 92.Engum A. The role of depression and anxiety in onset of diabetes in a large population-based study. Journal of Psychosomatic Research. 2007;62(1):31–38. doi: 10.1016/j.jpsychores.2006.07.009. [DOI] [PubMed] [Google Scholar]
- 93.Suarez E. C. Self-reported symptoms of sleep disturbance and inflammation, coagulation, insulin resistance and psychosocial distress: evidence for gender disparity. Brain, Behavior, and Immunity. 2008;22(6):960–968. doi: 10.1016/j.bbi.2008.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Trenell M. I., Marshall N. S., Rogers N. L. Sleep and metabolic control: waking to a problem? Clinical & Experimental Pharmacology & Physiology. 2007;34(1-2):1–9. doi: 10.1111/j.1440-1681.2007.04541.x. [DOI] [PubMed] [Google Scholar]
- 95.Irwin M. R., Wang M., Ribeiro D., et al. Sleep loss activates cellular inflammatory signaling. Biological Psychiatry. 2008;64(6):538–540. doi: 10.1016/j.biopsych.2008.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Meisinger C., Heier M., Loewel H. Sleep disturbance as a predictor of type 2 diabetes mellitus in men and women from the general population. Diabetologia. 2005;48(2):235–241. doi: 10.1007/s00125-004-1634-x. [DOI] [PubMed] [Google Scholar]
- 97.Nilsson P. M., Rööst M., Engström G., Hedblad B., Berglund G. Incidence of diabetes in middle-aged men is related to sleep disturbances. Diabetes Care. 2004;27(10):2464–2469. doi: 10.2337/diacare.27.10.2464. [DOI] [PubMed] [Google Scholar]
- 98.Knol M. J., Twisk J. W. R., Beekman A. T. F., Heine R. J., Snoek F. J., Pouwer F. Depression as a risk factor for the onset of type 2 diabetes mellitus. A meta-analysis. Diabetologia. 2006;49(5):837–845. doi: 10.1007/s00125-006-0159-x. [DOI] [PubMed] [Google Scholar]
- 99.Chan O., Inouye K., Riddell M. C., Vranic M., Matthews S. G. Diabetes and the hypothalamo-pituitary-adrenal (HPA) axis. Minerva Endocrinologica. 2003;28(2):87–102. [PubMed] [Google Scholar]
- 100.Surwit R. S., van Tilburg M. A. L., Zucker N., et al. Stress management improves long-term glycemic control in type 2 diabetes. Diabetes Care. 2002;25(1):30–34. doi: 10.2337/diacare.25.1.30. [DOI] [PubMed] [Google Scholar]
- 101.Fisher E. B., Thorpe C. T., Devellis B. M., Devellis R. F. Healthy coping, negative emotions, and diabetes management: a systematic review and appraisal. Diabetes Educator. 2007;33(6):1080–1103. doi: 10.1177/0145721707309808. [DOI] [PubMed] [Google Scholar]
- 102.Pirraglia P. A., Gupta S. The interaction of depression and diabetes: a review. Current Diabetes Reviews. 2007;3(4):249–251. doi: 10.2174/1573399810703040249. [DOI] [PubMed] [Google Scholar]
- 103.Konen J. C., Summerson J. H., Dignan M. B. Family function, stress, and locus of control. Relationships to glycemia in adults with diabetes mellitus. Archives of Family Medicine. 1993;2(4):393–402. doi: 10.1001/archfami.2.4.393. [DOI] [PubMed] [Google Scholar]
- 104.Knutson K. L., Van Cauter E., Zee P., Liu K., Lauderdale D. S. Cross-sectional associations between measures of sleep and markers of glucose metabolism among subjects with and without diabetes. The coronary artery risk development in young adults (CARDIA) sleep study. Diabetes Care. 2011;34(5):1171–1176. doi: 10.2337/dc10-1962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Cappuccio F. P., D'Elia L., Strazzullo P., Miller M. A. Sleep duration and all-cause mortality: a systematic review and meta-analysis of prospective studies. Sleep. 2010;33(5):585–592. doi: 10.1093/sleep/33.5.585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Zhang X., Norris S. L., Gregg E. W., Cheng Y. J., Beckles G., Kahn H. S. Depressive symptoms and mortality among persons with and without diabetes. American Journal of Epidemiology. 2005;161(7):652–660. doi: 10.1093/aje/kwi089. [DOI] [PubMed] [Google Scholar]
- 107.Katon W. J., Rutter C., Simon G., et al. The association of comorbid depression with mortality in patients with type 2 diabetes. Diabetes Care. 2005;28(11):2668–2672. doi: 10.2337/diacare.28.11.2668. [DOI] [PubMed] [Google Scholar]
- 108.Lin E. H. B., Heckbert S. R., Rutter C. M., et al. Depression and increased mortality in diabetes: unexpected causes of death. Annals of Family Medicine. 2009;7(5):414–421. doi: 10.1370/afm.998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Katon W., Fan M.-Y., Unützer J., Taylor J., Pincus H., Schoenbaum M. Depression and diabetes: a potentially lethal combination. Journal of General Internal Medicine. 2008;23(10):1571–1575. doi: 10.1007/s11606-008-0731-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Chen K.-M., Chen M.-H., Chao H.-C., Hung H.-M., Lin H.-S., Li C.-H. Sleep quality, depression state, and health status of older adults after silver yoga exercises: cluster randomized trial. International Journal of Nursing Studies. 2009;46(2):154–163. doi: 10.1016/j.ijnurstu.2008.09.005. [DOI] [PubMed] [Google Scholar]
- 111.Chen K.-M., Chen M.-H., Lin M.-H., Fan J.-T., Lin H.-S., Li C.-H. Effects of yoga on sleep quality and depression in elders in assisted living facilities. The Journal of Nursing Research. 2010;18(1):53–61. doi: 10.1097/jnr.0b013e3181ce5189. [DOI] [PubMed] [Google Scholar]
- 112.Lin K.-Y., Hu Y.-T., Chang K.-J., Lin H.-F., Tsauo J.-Y. Effects of yoga on psychological health, quality of life, and physical health of patients with cancer: a meta-analysis. Evidence-Based Complementary and Alternative Medicine. 2011;2011:12. doi: 10.1155/2011/659876.659876 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Innes K. E., Vincent H. K., Taylor A. G. Chronic stress and insulin resistance-related indices of cardiovascular disease risk, part 2: a potential role for mind-body therapies. Alternative Therapies in Health and Medicine. 2007;13(5):44–51. Erratum in: Alternative Therapies in Health and Medicine, vol. 13, no. 6, p. 15, 2007. [PubMed] [Google Scholar]
- 114.Streeter C. C., Theodore H. Whitfield, Owen L., et al. Effects of yoga versus walking on mood, anxiety, and brain GABA levels: a randomized controlled MRS study. Journal of Alternative and Complementary Medicine. 2010;16(11):1145–1152. doi: 10.1089/acm.2010.0007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Kirkwood G., Rampes H., Tuffrey V., Richardson J., Pilkington K. Yoga for anxiety: a systematic review of the research evidence. British Journal of Sports Medicine. 2005;39(12):884–891. doi: 10.1136/bjsm.2005.018069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Innes K. E., Selfe T. K., Agarwal P., Williams K., Flack K. L. Efficacy of an eight-week yoga intervention on symptoms of restless legs syndrome (RLS): a pilot study. Journal of Alternative and Complementary Medicine. 2013;19(6):527–535. doi: 10.1089/acm.2012.0330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Innes K. E., Selfe T. K. The effects of a gentle yoga program on sleep, mood, and blood pressure in older women with restless legs syndrome (RLS): a preliminary randomized controlled trial. Evidence-Based Complementary and Alternative Medicine. 2012;2012:14. doi: 10.1155/2012/294058.294058 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Innes K. E., Vincent H. K., Taylor A. G. Chronic stress and insulin resistance-related indices of cardiovascular disease risk. Part 2. A potential role for mind-body therapies. Alternative Therapies in Health and Medicine. 2007;13(5):44–51. [PubMed] [Google Scholar]
- 119.Innes K. E., Vincent H. K. The influence of yoga-based programs on risk profiles in adults with type 2 diabetes mellitus: a systematic review. Evidence-Based Complementary and Alternative Medicine. 2007;4(4):469–486. doi: 10.1093/ecam/nel103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Carnethon M. R., Prineas R. J., Temprosa M., Zhang Z.-M., Uwaifo G., Molitch M. E. The association among autonomic nervous system function, incident diabetes, and intervention arm in the diabetes prevention program. Diabetes Care. 2006;29(4):914–919. doi: 10.2337/diacare.29.04.06.dc05-1729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Carnethon M. R., Golden S. H., Folsom A. R., Haskell W., Liao D. Prospective investigation of autonomic nervous system function and the development of type 2 diabetes: the atherosclerosis risk in communities study, 1987–1998. Circulation. 2003;107(17):2190–2195. doi: 10.1161/01.cir.0000066324.74807.95. [DOI] [PubMed] [Google Scholar]
- 122.Singh R. B., Kartik C., Otsuka K., Pella D., Pella J. Brain-heart connection and the risk of heart attack. Biomedicine and Pharmacotherapy. 2002;56(supplement 2):257s–265s. doi: 10.1016/s0753-3322(02)00300-1. [DOI] [PubMed] [Google Scholar]
- 123.Maser R. E., Mitchell B. D., Vinik A. I., Freeman R. The association between cardiovascular autonomic neuropathy and mortality in individuals with diabetes: a meta-analysis. Diabetes Care. 2003;26(6):1895–1901. doi: 10.2337/diacare.26.6.1895. [DOI] [PubMed] [Google Scholar]
- 124.Manzella D., Paolisso G. Cardiac autonomic activity and type II diabetes mellitus. Clinical Science. 2005;108(2):93–99. doi: 10.1042/cs20040223. [DOI] [PubMed] [Google Scholar]
- 125.Innes K. E., Bourguignon C., Taylor A. G. Risk indices associated with the insulin resistance syndrome, cardiovascular disease, and possible protection with yoga: a systematic review. Journal of the American Board of Family Practice. 2005;18(6):491–519. doi: 10.3122/jabfm.18.6.491. [DOI] [PubMed] [Google Scholar]
- 126.Innes K. Yoga-based programs for cardiovascular disease, diabetes, and related chronic conditions. Time for a new health care paradigm?. Proceedings of the Yoga and Health: Research and Practice—An International Conference; April 2014; London, UK. [Google Scholar]
- 127.Tiwari S., Tiwari S. K., Gehlot S., Singh G. Outcome of breathing exercise (pranayam) on spirometric parameters in type 2 diabetic individuals: a clinical study. Journal of Stress Physiology & Biochemistry. 2012;8(4):218–225. [Google Scholar]
- 128.Lawlor D. A., Ebrahim S., Smith G. D. Associations of measures of lung function with insulin resistance and Type 2 diabetes: findings from the British Women's Heart and Health study. Diabetologia. 2004;47(2):195–203. doi: 10.1007/s00125-003-1310-6. [DOI] [PubMed] [Google Scholar]
- 129.Davis W. A., Knuiman M., Kendall P., Grange V., Davis T. M. E. Glycemic exposure is associated with reduced pulmonary function in type 2 diabetes: the Fremantle Diabetes Study. Diabetes Care. 2004;27(3):752–757. doi: 10.2337/diacare.27.3.752. [DOI] [PubMed] [Google Scholar]
- 130.Ford E. S., Mannino D. M., National Health and Nutrition Examination Survey Epidemiologic Follow-Up Study Prospective association between lung function and the incidence of diabetes: findings from the National Health and Nutrition Examination Survey Epidemiologic Follow-up Study. Diabetes Care. 2004;27(12):2966–2970. doi: 10.2337/diacare.27.12.2966. [DOI] [PubMed] [Google Scholar]
- 131.McKeever T. M., Weston P. J., Hubbard R., Fogarty A. Lung function and glucose metabolism: an analysis of data from the Third National Health and Nutrition Examination Survey. American Journal of Epidemiology. 2005;161(6):546–556. doi: 10.1093/aje/kwi076. [DOI] [PubMed] [Google Scholar]
- 132.de G R Hansen E., Innes K. E. The benefits of yoga for adults with type 2 diabetes: a review of the evidence and call for a collaborative, integrated research initiative. International Journal of Yoga Therapy. 2013;23(2):71–83. [PubMed] [Google Scholar]
- 133.Innes K. E., Bourguignon C., Taylor A. G. Risk indices associated with the insulin resistance syndrome, cardiovascular disease, and possible protection with yoga: a systematic review. The Journal of the American Board of Family Medicine. 2005;18(6):491–519. doi: 10.3122/jabfm.18.6.491. [DOI] [PubMed] [Google Scholar]
- 134.Khatri D., Mathur K. C., Gahlot S., Jain S., Agrawal R. P. Effects of yoga and meditation on clinical and biochemical parameters of metabolic syndrome. Diabetes Research and Clinical Practice. 2007;78(3):e9–e10. doi: 10.1016/j.diabres.2007.05.002. [DOI] [PubMed] [Google Scholar]
- 135.Kyrou I., Tsigos C. Stress mechanisms and metabolic complications. Hormone & Metabolic Research. 2007;39(6):430–438. doi: 10.1055/s-2007-981462. [DOI] [PubMed] [Google Scholar]
- 136.Kyrou I., Chrousos G. P., Tsigos C. Stress, visceral obesity, and metabolic complications. Stress, Obesity, and Metabolic Syndrome. 2006;1083:77–110. doi: 10.1196/annals.1367.008. [DOI] [PubMed] [Google Scholar]
- 137.Brown R. P., Gerbarg P. L. Sudarshan Kriya yogic breathing in the treatment of stress, anxiety, and depression: part I—neurophysiologic model. The Journal of Alternative and Complementary Medicine. 2005;11(1):189–201. doi: 10.1089/acm.2005.11.189. Erratum in: The Journal of Alternative and Complementary Medicine, vol. 11, no. 2, pp. 383-384, 2005. [DOI] [PubMed] [Google Scholar]
- 138.McCaffrey R., Ruknui P., Hatthakit U., Kasetsomboon P. The effects of yoga on hypertensive persons in Thailand. Holistic Nursing Practice. 2005;19(4):173–180. doi: 10.1097/00004650-200507000-00009. [DOI] [PubMed] [Google Scholar]
- 139.Khattab K., Khattab A. A., Ortak J., Richardt G., Bonnemeier H. Iyengar yoga increases cardiac parasympathetic nervous modulation among healthy yoga practitioners. Evidence-Based Complementary and Alternative Medicine. 2007;4(4):511–517. doi: 10.1093/ecam/nem087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.De Meersman R. E., Stein P. K. Vagal modulation and aging. Biological Psychology. 2007;74(2):165–173. doi: 10.1016/j.biopsycho.2006.04.008. [DOI] [PubMed] [Google Scholar]
- 141.Thayer J. F., Sternberg E. Beyond heart rate variability. Annals of the New York Academy of Sciences. 2006;1088:361–372. doi: 10.1196/annals.1366.014. [DOI] [PubMed] [Google Scholar]
- 142.Thayer J. F., Lane R. D. The role of vagal function in the risk for cardiovascular disease and mortality. Biological Psychology. 2007;74(2):224–242. doi: 10.1016/j.biopsycho.2005.11.013. [DOI] [PubMed] [Google Scholar]
- 143.Wang D. J. J., Rao H., Korczykowski M., et al. Cerebral blood flow changes associated with different meditation practices and perceived depth of meditation. Psychiatry Research: Neuroimaging. 2011;191(1):60–67. doi: 10.1016/j.pscychresns.2010.09.011. [DOI] [PubMed] [Google Scholar]
- 144.Newberg A. B., Wintering N., Waldman M. R., Amen D., Khalsa D. S., Alavi A. Cerebral blood flow differences between long-term meditators and non-meditators. Consciousness and Cognition. 2010;19(4):899–905. doi: 10.1016/j.concog.2010.05.003. [DOI] [PubMed] [Google Scholar]
- 145.Rubia K. The neurobiology of meditation and its clinical effectiveness in psychiatric disorders. Biological Psychology. 2009;82(1):1–11. doi: 10.1016/j.biopsycho.2009.04.003. [DOI] [PubMed] [Google Scholar]
- 146.Kjaer T. W., Bertelsen C., Piccini P., Brooks D., Alving J., Lou H. C. Increased dopamine tone during meditation-induced change of consciousness. Cognitive Brain Research. 2002;13(2):255–259. doi: 10.1016/S0926-6410(01)00106-9. [DOI] [PubMed] [Google Scholar]
- 147.Alexander G. K., Innes K. E., Selfe T. K., Brown C. J. ‘More than I expected’: perceived benefits of yoga practice among older adults at risk for cardiovascular disease. Complementary Therapies in Medicine. 2013;21(1):14–28. doi: 10.1016/j.ctim.2012.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Steinhardt M. A., Mamerow M. M., Brown S. A., Jolly C. A. A resilience intervention in African American adults with type 2 diabetes: a pilot study of efficacy. Diabetes Educator. 2009;35(2):274–284. doi: 10.1177/0145721708329698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Yi J. P., Vitaliano P. P., Smith R. E., Yi J. C., Weinger K. The role of resilience on psychological adjustment and physical health in patients with diabetes. British Journal of Health Psychology. 2008;13(2):311–325. doi: 10.1348/135910707X186994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Hartfiel N., Havenhand J., Khalsa S. B., Clarke G., Krayer A. The effectiveness of yoga for the improvement of well-being and resilience to stress in the workplace. Scandinavian Journal of Work, Environment & Health. 2011;37(1):70–76. doi: 10.5271/sjweh.2916. [DOI] [PubMed] [Google Scholar]
- 151.Johnston J. Paper. 29. Boston, Mass, USA: Northeastern University; 2011. The impact of yoga on military personnel with post traumatic stress disorder. [Google Scholar]
- 152.Noggle J. J., Steiner N. J., Minami T., Khalsa S. B. S. Benefits of yoga for psychosocial well-being in a us high school curriculum: a preliminary randomized controlled trial. Journal of Developmental and Behavioral Pediatrics. 2012;33(3):193–201. doi: 10.1097/dbp.0b013e31824afdc4. [DOI] [PubMed] [Google Scholar]
- 153.Dalen J., Smith B. W., Shelley B. M., Sloan A. L., Leahigh L., Begay D. Pilot study: mindful Eating and Living (MEAL): weight, eating behavior, and psychological outcomes associated with a mindfulness-based intervention for people with obesity. Complementary Therapies in Medicine. 2010;18(6):260–264. doi: 10.1016/j.ctim.2010.09.008. [DOI] [PubMed] [Google Scholar]
- 154.Dittmann K. A., Freedman M. R. Body awareness, eating attitudes, and spiritual beliefs of women practicing Yoga. Eating Disorders. 2009;17(4):273–292. doi: 10.1080/10640260902991111. [DOI] [PubMed] [Google Scholar]
- 155.van Dam H. A., van der Horst F. G., Knoops L., Ryckman R. M., Crebolder H. F. J. M., van den Borne B. H. W. Social support in diabetes: a systematic review of controlled intervention studies. Patient Education and Counseling. 2005;59(1):1–12. doi: 10.1016/j.pec.2004.11.001. [DOI] [PubMed] [Google Scholar]
- 156.Tang T. S., Brown M. B., Funnell M. M., Anderson R. M. Social support, quality of life, and self-care behaviors among african americans with type 2 diabetes. Diabetes Educator. 2008;34(2):266–276. doi: 10.1177/0145721708315680. [DOI] [PubMed] [Google Scholar]
- 157.Bernardi L., Rosengård-Bärlund M., Groop P.-H. Cardio-respiratory interactions in diabetes. Proceedings of the 8th Conference of the European Study Group on Cardiovascular Oscillations (ESGCO '14); May 2014; Trento, Italy. IEEE; pp. 227–228. [DOI] [Google Scholar]
- 158.Bernardi L., Spadacini G., Bellwon J., Hajric R., Roskamm H., Frey A. W. Effect of breathing rate on oxygen saturation and exercise performance in chronic heart failure. The Lancet. 1998;351(9112):1308–1311. doi: 10.1016/s0140-6736(97)10341-5. [DOI] [PubMed] [Google Scholar]
- 159.Pomidori L., Campigotto F., Amatya T. M., Bernardi L., Cogo A. Efficacy and tolerability of yoga breathing in patients with chronic obstructive pulmonary disease: a pilot study. Journal of Cardiopulmonary Rehabilitation and Prevention. 2009;29(2):133–137. doi: 10.1097/hcr.0b013e31819a0227. [DOI] [PubMed] [Google Scholar]
- 160.Cazzato S., Bernardi F., Salardi S., et al. Lung function in children with diabetes mellitus. Pediatric Pulmonology. 2004;37(1):17–23. doi: 10.1002/ppul.10399. [DOI] [PubMed] [Google Scholar]
- 161.Black D. S., Cole S. W., Irwin M. R., et al. Yogic meditation reverses NF-κB and IRF-related transcriptome dynamics in leukocytes of family dementia caregivers in a randomized controlled trial. Psychoneuroendocrinology. 2013;38(3):348–355. doi: 10.1016/j.psyneuen.2012.06.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Lavretsky H., Epel E. S., Siddarth P., et al. A pilot study of yogic meditation for family dementia caregivers with depressive symptoms: effects on mental health, cognition, and telomerase activity. International Journal of Geriatric Psychiatry. 2013;28(1):57–65. doi: 10.1002/gps.3790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Dusek J. A., Otu H. H., Wohlhueter A. L., et al. Genomic counter-stress changes induced by the relaxation response. PLoS ONE. 2008;3(7) doi: 10.1371/journal.pone.0002576.e2576 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Kaliman P., Álvarez-López M. J., Cosín-Tomás M., Rosenkranz M. A., Lutz A., Davidson R. J. Rapid changes in histone deacetylases and inflammatory gene expression in expert meditators. Psychoneuroendocrinology. 2014;40(1):96–107. doi: 10.1016/j.psyneuen.2013.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Lowell B. B., Shulman G. I. Mitochondrial dysfunction and type 2 diabetes. Science. 2005;307(5708):384–387. doi: 10.1126/science.1104343. [DOI] [PubMed] [Google Scholar]
- 166.Wellen K. E., Hotamisligil G. S. Inflammation, stress, and diabetes. The Journal of Clinical Investigation. 2005;115(5):1111–1119. doi: 10.1172/jci200525102. [DOI] [PMC free article] [PubMed] [Google Scholar]