Abstract
Objective
Impairments in cognition and everyday functioning are common in schizophrenia and bipolar disorder. Based on two studies of schizophrenia (SCZ) and bipolar I disorder (BPI) with similar methods, this paper presents factor analyses of cognitive and functional capacity (FC) measures. The overall goal of these analyses was to determine whether performance-based assessments should be examined individually, or aggregated on the basis of the correlational structure of the tests and as well as to evaluate the similarity of factor structures in SCZ and BPI.
Method
Veterans Affairs (VA) Cooperative Studies Program study #572, evaluated cognitive and FC measures among 5,414 BPI and 3,942 SZ patients. A second study evaluated similar neuropsychological (NP) and FC measures among 368 BPI and 436 SZ patients. Principal components analysis, as well as exploratory and confirmatory factor analyses, were used to examine the data.
Results
Analyses in both datasets suggested that NP and FC measures were explained by of a single underlying factor in BPI and SCZ patients, both when analyzed separately or as in a combined sample. The factor structure in both studies was similar, with or without inclusion of FC measures; homogeneous loadings were observed for that single factor across cognitive and FC domains across the samples.
Conclusions
The empirically derived factor model suggests that NP performance and FC are best explained as a single latent trait applicable to people with schizophrenia and bipolar illness. This single measure may enhance the robustness of the analyses relating genomic data to performance-based phenotypes.
Schizophrenia and bipolar disorder are chronic psychiatric diseases that have long been known to have a substantial genetic component to their etiology (Sullivan et al., 2003; Kieseppä et al., 2004), with possible overlap in genetic contributions (Lichtenstein et al., 2009). Recent studies of the genetics of severe mental illness have focused on identifying endophenotypes (Braff et al., 2007), commonly defined as traits that are stable over time, simpler than the end state disease, present in relatives, and amenable to direct treatment. For example, cognitive impairments are prominent in both schizophrenia and bipolar disorder and have been suggested as potential endophenotypes. It is known that cognitive impairments tend to have a heritable component (Gur et al., 2007; Greenwood et al., 2007), and that many of the more functionally relevant aspects of cognitive impairment are known to be consistently heritable—including episodic memory (heritability range = 0.3 to 0.6), attention/vigilance (mean = 0.54), working memory (range = 0.3 to 0.6), and executive functioning (range = 0.3 to 0.6). Although neuropsychological (NP) functioning in bipolar disorder has been somewhat less extensively studied, a considerable increase in interest has occurred recently. Specifically, euthymic patients have considerable impairments, and these impairments while euthymic are related to disability (Wingo et al., 2009).
Measurement of disability and its determinants has also advanced in recent years, including the development of highly reliable performance-based measures of functional skills (referred to as “functional capacity”) that can be applied with high precision and fidelity in research settings (Keefe et al., 2011; Harvey et al., 2014). These measures have been shown to be related to both cognitive deficits and to real-world disability in both schizophrenia and bipolar disorder (Bowie et al., 2010). It was recently argued (Harvey et al., 2012) that these functional capacity (FC) measures are themselves potential endophenotypes, reflecting skills that are quite similar across different cultures (McIntosh et al., 2011), present in individuals with spectrum conditions (McClure et al., 2014), and not influenced by wide variations in environmental and social support (Harvey et al., 2009).
When relating cognition and functional capacity to genomics, one important concern is how to approach the analysis of individual performance-based measures. Multiple studies have suggested that the factor structure of cognition in schizophrenia may actually be a simple one, with results of several large-scale studies suggesting that single factor solutions fit the data, particularly with abbreviated assessment batteries. For instance, the results of the large-scale (n=1,332) CATIE study baseline neurocognitive assessment (Keefe et al., 2006) found that a single-factor structure fit the data better than more complex models. In addition, and consistent with earlier results (Dickinson et al., 2004), a few of the measures accounted for the majority of the variance in the composite scores. In a smaller and separate sample from this study, assessing only people with schizophrenia (Harvey et al.2013), it was also found that neuropsychological testing and functional capacity performance constituted a single factor, which met all criteria for multivariate longitudinal stability of all indicators (factor scores, factor loadings, and error co-variances) over a follow-up period of between 6 weeks to 6 months.
Factor analyses of cognitive performance in bipolar disorder are not as common. Two studies (Czobor et al., 2007; Schretlen, et al., 2013) examined patients with schizophrenia and bipolar disorder using cognitive testing and factor analysis. Both papers suggested a 6-factor model that fit similarly in the two patient groups. The sample sizes were smaller than the two current studies (Schretlen et al.:126 BP and 110 SCZ; Czobor et al.:155 BPD and 250 SCZ) and functional capacity was not examined. Nonetheless, these results suggested a similar factor structure in people with schizophrenia and bipolar disorder, a topic to be examined in the present study.
The current paper presents the results of analyses of the factor structure of performance-based measures of cognition and functional capacity from two different genomically-focused studies. One study is Cooperative Studies Program (CSP) #572, a large-scale genomic study of 9,356 Veterans with BPI or schizophrenia. Another study is the FUNCAP study, which performed a reassessment of samples of BPI and schizophrenia patients (n=804) previously ascertained on the basis of being from an Ashkenazi Jewish background. In contrast to some previous genetic studies of severe mental illness, all participants in both studies were seen in person, diagnosed with a structured clinical interview, examined for the presence of co-morbidities, and tested in person with performance-based assessments of cognition and functional capacity. The results of the performance-based assessments in both of these studies have been reported previously (e.g., Bowie et al., 2010; Harvey et al., 2014; Mausbach et al., 2010).
As the FUNCAP study provided the preliminary results for the CSP-572 performance-based strategies, the cognitive and functional capacity measures were nearly identical. In parallel analyses conducted separately in each study, principal component analyses (PCA) were used as a data reduction technique. Exploratory factor analyses were then used and followed by confirmatory factor analyses (CFA), testing the hypothesis of a unique underlying factor in both BPI and SCZ. Patients with these diagnoses were analyzed separately, as well as in a combined sample, with and without the inclusion of functional capacity measures in the factor models. For these two study populations each two diagnostic groups, our goal was to determine whether the factor structure of cognition and functional capacity was multidimensional or not, and whether cognition and functional capacity measures were statistically related to a single underlying latent trait indexing global performance.
Methods
Participants
Both studies had similar diagnostic criteria for entry. Patients were required to meet lifetime (DSM-IV) criteria for schizophrenia, any subtype, or bipolar I disorder, any current state. Patients with major neurologic illnesses, or systemic medical illnesses that could interfere with central nervous system function and test performance were excluded. Patients with diagnoses of substance abuse were not excluded, given the co-occurrence in the population and issues of representativeness. Participants were not enrolled if they appeared to be intoxicated at a study visit, but could be reassessed at a later date. Schizoaffective disorder was an exclusion criterion, in that we did not anticipate being able to recruit samples with confirmed schizoaffective disorder that was comparable in size to the other two diagnostic groups for the planned genomic analyses. Patients with Bipolar I disorder (only) were selected for participation, because of concerns at the outset of the study that the diagnosis of bipolar II disorder might have less reliability. Potential VA participants were identified with medical record information or referred from their clinicians. A HIPPA waiver was obtained which allowed a targeted mailing. Identified patients, either through medical records or clinicians, were then sent an invitational mailing from the local site investigator to assess their interest in participating in a VA research study. A total of 27 different VA sites participated in this study at different times (with an effective steady-state of 25) and contributed research participants during the enrollment period. These sites were selected on the basis of several criteria, including previous successful participation in VA research on severe mental illness, and the availability of a sample of Veterans adequate to recruit; full details on this study were presented previously (Harvey et al., 2014).
All FUNCAP study participants were of full or mixed Ashkenazi Jewish (AJ) background, which was determined from ancestry of four grandparents. The restriction to AJ ancestry was made to take potential advantage of founder effects in this population (Bray et al., 2010). Participants were recruited nationally through advertisements in newspapers and Jewish publications, talks given at community centers and synagogues, and through the Epidemiology-Genetics (EPIGEN) Program website. Details of recruitment, assessment and consensus diagnostic procedures for the FUNCAP/EPIGEN studies are available in several publications (Chen et al., 2009; Fallin et al., 2003; Fallin et al., 2004,Fallin et al., 2005).
The VA study was approved by the VA Central IRB, and all patients provided written informed consent. The FUNCAP study was approved by the Johns Hopkins University IRB. No patients who required the permission of a guardian to participate were enrolled. In addition to the study visit, information from medical charts, the patients' clinicians, or other informants were used, if needed, to confirm diagnoses—with all VA participants receiving the Structured Clinical Interview for the DSM (SCID; First et al., 2005) and all FUNCAP patients assessed with the Diagnostic interview for genetic studies (DIGS).
Measures
Patients were assessed with a number of assessments that varied across the studies, including clinical assessments and other evaluations of everyday functioning.
Functional capacity
Two different performance-based functional capacity measures were administered, both of which had previous evidence of high psychometric quality (Keefe et al., 2011; Harvey et al., 2011; 2013). These measures are related to both cognitive test performance and everyday functional disability in patients with schizophrenia and bipolar disorder. They were selected because they are highly correlated with performance on longer versions of the assessment, even given their abbreviated nature (Heaton et al., 2004; Mausbach et al., 2007).
One of the tests of functional abilities was the UPSA-B (Mausbach et al., 2007), a measure of functional capacity in which patients are asked to perform everyday tasks related to communication and finances. During the Communication subtest, participants role-play exercises using an unplugged telephone, such as making an emergency call, dialing a number from memory, and calling to reschedule a doctor's appointment. For the Finance subtest, participants count change, read a utility bill, and write a check for the bill. The UPSA-B requires approximately 10-15 minutes, and raw scores range from 0-20, with higher scores indicating better functional capacity. The Advanced Finances subscale of the Everyday Functioning Battery (EFB; Heaton et al., 2004), designed to examine financial management in higher functioning individuals was also administered. The Advanced Finances test requires individuals to prepare bank deposits and write checks to pay bills, maintain a checkbook balance, and organize payments such that a pre-specified amount of money is left available at the end of the task. This instrument was selected because at the time the study was planned, we were concerned that younger individuals with schizophrenia might demonstrate ceiling effects on the UPSA-B, requiring a more difficult test. Total scores on the Advanced Finances subtest range from 0-13. Due to incomplete data on the EFB in the FUNCAP study resulting in a small sample available for analysis, we examined the EFB only in the VA patient sample.
Neuropsychological (NP) assessment
Among people with serious mental illness, several recent studies have suggested that performance on a very limited set of NP measures adequately captures the overall levels of impairment in cognitive functioning (Keefe et al., 2006). Evidence also suggests that abbreviated cognitive assessments are equivalently associated with impairments in everyday outcomes and functional capacity measures, compared to longer assessments (Keefe et al., 2004). The FUNCAP study was initiated prior the completion of the MATRICS initiative, and the assessments in that study therefore included some tests that are not in the MATRICS Consensus Cognitive Battery (MCCB; Nuechterlein et al., 2008), and some tests that measure similar constructs (verbal working memory and verbal episodic memory, reasoning and problem solving, processing speed) are not absolutely identical to those in the MCCB. All CSP 572 tests were selected from the MCCB, using only paper and pencil tasks, given that more missing data has been found in large scale studies with computerized tests than with paper and pencil assessments (Keefe et al., 2006). This difference is due possibly to problems with tester training in computer administration. More saliently, VA IT requirements would have made data transfer from laptop computers (required at field sites) to the data management center challenging. Table 1 presents the constructs and tests across the two different studies.
Table 1. Neuropsychological and Functional Capacity Performance Constructs and Tests across Studies.
| CSP572 | FUNCAP | |
|---|---|---|
| Adaptive Function | ||
| Communication & finance: | UPSA Brief Advanced Finances- EFB | UPSA Brief |
| Cognitive Function | ||
| Processing Speed: | Animal Naming | Animal Naming |
| Trail Making Part A | Trail Making Part A Trail Making Part B |
|
| MCCB Symbol Digit | WAIS-IV Digit Symbol | |
| Verbal Working Memory: | Maryland Letter-Number Span | WAIS-IV Letter-Number Sequencing |
| Verbal Learning Test (VLT): | Hopkins | Rey-Auditory |
| Reasoning and Problem Solving: | NAB Mazes | Wisconsin Card Sorting Test CPT-IP |
UPSA = UCSD Performance-Based Skills Assessment Battery; EFB = Everyday Functioning Battery; MCCB = MATRICS Consensus Cognitive Battery; WAIS-IV = Wechsler Adult Intelligence Scale; NAB = Neuropsychological Assessment Battery; CPT-IP = Continuous Performance Test, Identical Pair version
Procedures
Tester training, test administration, and data monitoring was performed separately across the two studies. In CSP 572, all testers were trained all in-person (by Philip D. Harvey), with replacement testers trained in webinars or at the annual meeting. All participant testing was performed at VA field sites by study coordinators with various educational backgrounds. Every case record form was electronically transferred to a central data management facility, where an algorithm designed to detect testing errors was applied to every case record form. Any questionable cases were referred to a national study coordinator who examined the case record form and referred any questions to the expert trainer. In addition, all testers had an in person examination of their case record forms for their first 5 assessments, and then randomly selected case record forms were examined by the expert trainer. In the FUNCAP study, two PhD level psychologists made home visits and performed the cognitive and functional capacity assessments. Data were transferred to a central data management facility at Johns Hopkins University. Testers were expert trained (also by Philip D. Harvey) in-person prior to the initiation of the study, and their testing was continuously expert monitored during the study.
Data Analyses
Statistical analyses began with principal components analysis and exploratory factor analysis, computed in each diagnostic group, in each study, based on standardized assessment scores (z-scores). These analyses were performed with TIBCO Spotfire S+ version 8.2 (TIBCO Software Inc., Palo Alto, CA, USA) and SAS version 9.2 (SAS Institute Inc., Cary, NC, USA) software packages. After evaluation of the likely number of factors present in the data, confirmatory factor analysis (CFA) used Mplus version 7.2 (Muthen & Muthen, Los Angeles, CA, USA) to examine the data and quantify the fit of the models. These models were fit in both SZ and BP patient samples separately in each study, and then combined. The analyses were performed with the adaptive and cognitive domains combined, as well as within cognitive function assessments alone. Missing values for specific assessment measures were present in less than 1% in both CSP#572 schizophrenia and bipolar samples, and less than 4% in the FUNCAP study. Cases were deleted list-wise in principal components and confirmatory factor analyses when values were missing.
Choosing the first principal components that explain most of the variation in the original assessment measures were based on three widely used criteria: a) Cattell's criterion of plotted ordered eigenvalues of the orthogonal transformation (screeplots); b) the “90% criterion” of including the first few components that explain a threshold amount of the variance; and c) Kaiser's criterion to exclude components with eigenvalues below the average (Mardia et al., 1979).
Exploratory factor analyses were also performed to uncover the underlying relational structure between measured assessments, and to inform confirmatory factor analyses on the number of factors to be used in testing hypotheses. Confirmatory factor models were compared on several chi-square based goodness-of-fit statistics, such as the Root Mean Square Error of Approximation (RMSEA), the Comparative Fit Index (CFI), and the Non-Normed Fit Index (NNFI), aka Tucker-Lewis Index (TLI).
Results
Sample Characteristics
The VA CSP#572 sample was older (mean age ∼ 54 years, 11 years SD, 18-90 years range) when compared to the FUNCAP sample (mean age ∼ 49 years, 12 years SD, 16-83 years range), and had lower female representation (13.8% vs. 42.7%; see Table 2). In both studies, the schizophrenia sample was slightly younger (CSP#572 mean age ∼ 55 years, range 18-90; FUNCAP mean age ∼ 50 years, range 16-78) than the bipolar sample (CSP#572 mean age ∼ 53 years, range 19-90; FUNCAP mean age ∼ 48 years, range 18-83), and had fewer women (CSP#572 7.3% SZ vs. 18.7%, BP; FUNCAP 35.3% SZ vs. 51.4% BP). In addition, the schizophrenia sample was less educated (CSP#572 56.5% SZ vs. 75.3% BP with higher than high school degree, FUNCAP SZ mean ∼ 14 years of education, range 6-20, vs. BP mean ∼ 16 years of education, range 10-20), and was also less likely to be married, cohabitating, or in a civil commitment relationship (CSP#572 19.6% SZ vs. 33.2% BP ever married, FUNCAP 21.1% SZ vs. 52.7% BP ever married). Racial variation only occurred in the CSP#572 study, with the schizophrenia sample having a higher minority representation (52% African American and 11.6% Other Non-White Race) than the bipolar sample (23.5% African American and 12.3% Other Non-White Race).
Table 2.
Demographic Characteristics of participants in CSP572 and FUNCAP studies.
| CSP572 | FUNCAP | |||
|---|---|---|---|---|
| Schizophrenia N=3942 | Bipolar Disorder N=5414 | Schizophrenia N=436 | Bipolar Disorder N=368 | |
| Age (mean years ± SD) | 55.1 ± 10.1 | 52.6 ± 11.5 | 50.1 ± 9.8 | 48.1 ± 13.2 |
| Male | 92.7% | 81.4% | 64.7% | 48.4% |
| Marital Status | ||||
| Ever Married | 60.0% | 81.8% | 21.1% | 52.7% |
| Never married | 40.0% | 18.2% | 78.7% | 47.0% |
| Education (mean years ± SD) | 14.4 ± 2.5 | 16.1 ± 2.1 | ||
| Less than High Schoo l | 7.3% | 2.5% | ||
| High School | 36.3% | 22.2% | ||
| More than High School | 56.5% | 75.3% | ||
| Race | ||||
| Caucasian | 36.4% | 64.3% | 100% | 100% |
| African-American | 52.0% | 23.5% | ||
| Other | 11.6% | 12.3% | ||
| Ethnicity | ||||
| Latino | 9.8% | 8.8% | ||
Rates of missing data were quite low in both studies, with the most missing data for a neuropsychological test being 0.8% in CSP#572 (symbol coding for schizophrenia patients), and 3.9% in FUNCAP (WCST for schizophrenia patients). In CSP#572, for the functional capacity measures, 2.6% of the schizophrenia patients were missing data on advanced finances, largely because they insisted that they had never written a check and refused the procedure; less than 0.1% of the cases were missing data on the UPSA-B. In FUNCAP, 2.8% of the cases were missing data on the UPSA-B. The raw scores and standard scores for the two Veteran samples in CSP#572 are also shown in Table 3, along with scores from the FUNCAP samples described above.
Table 3.
Performance and Levels of Impairment on Cognitive and Functional measures in CSP572 and FUNCAP studies (see text for details).
| CSP572 | FUNCAP | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Schizophrenia | Bipolar | Schizophrenia | Bipolar | |||||||||
| N | Mean (SD) | IMP | N | Mean (SD) | IMP | N | Mean (SD) | IMP | N | Mean (SD) | IMP | |
| Adaptive Function (UPSA) | 3940 | 14.8 (3.2) | 38.9 | 5410 | 16.6 (2.4) | 15.5 | 424 | 14.6 (4.8) | 32.8 | 366 | 17.6 (2.5) | 7.9 |
| Everyday Functioning Battery (EFB) | 3840 | 8.1 (4.0) | 38.3 | 5334 | 10.4 (2.9) | 14.8 | ||||||
| Trail Making Part A | 3929 | 52.0 (29.9) | 66.9 | 5404 | 39.9 (20.4) | 48.5 | 432 | 60.4 (33.5) | 84.4 | 363 | 44.9 (20.7) | 62.0 |
| Trail Making Part B | 421 | 104.7 (73.8) | 81.0 | 362 | 75.9 (53.4) | 66.9 | ||||||
| Symbol Digit | 3912 | 35.1 (12.7) | 73.2 | 5399 | 43.1 (12.7) | 50.8 | 433 | 54.2 (17.4) | 84.0 | 366 | 66.5 (18.0) | 64.1 |
| Verbal Learning Test | 3920 | 17.9 (5.8) | 81.9 | 5401 | 21.0 (6.0) | 65.6 | 431 | 36.1 (11.7) | 63.1 | 367 | 45.3 (10.7) | 28.5 |
| Letter Number Sequencing | 3931 | 10.2 (4.3) | 71.7 | 5405 | 12.7 (4.0) | 52.7 | 430 | 8.5 (3.3) | 33.5 | 367 | 10.2 (3.0) | 16.0 |
| Animal Fluency | 3932 | 17.5 (5.6) | 57.5 | 5410 | 20.6 (5.7) | 39.0 | 435 | 17.6 (5.8) | 56.7 | 367 | 20.7 (6.1) | 42.1 |
| NAB Mazes/WCST | 3930 | 10.4 (6.9) | 63.4 | 5408 | 13.9 (7.3) | 47.7 | 419 | 27.7 (12.5) | 29.4 | 359 | 20.8 (10.9) | 11.4 |
| Any NP test | 3810 | 96.7 | 4750 | 87.7 | 427 | 97.9 | 315 | 85.6 | ||||
Note. IMP= Impairment as defined by previous results
Detailed analyses of performance differences between the BPI and SCZ samples have been published both for CSP 572 (Harvey et al., 2014) and FUNCAP (Bowie et al., 2010). Impairments in performance on the cognitive tests were defined as performance that was more than 1.0 SD worse than normative standards, using the MCCB norms for CSP 572 and previously published norms for FUNCAP. For UPSA-B performance, we used the UPSA-B score from Mausbach et al. (2007) that separated those patients who were living independently in the community from those who required residential support to identify impaired performance. In both studies, the schizophrenia sample had higher impairments for both cognitive and adaptive function performance-based measures (Table 3), compared to the bipolar sample. The proportion of cases within each diagnostic group who met these criteria for impairment was similar, although not identical, across the two studies. The proportion of patients who were impaired on at least one cognitive measure, however, was essentially identical across the two studies for each of the two diagnostic groups.
Principal Components and Factor Analyses
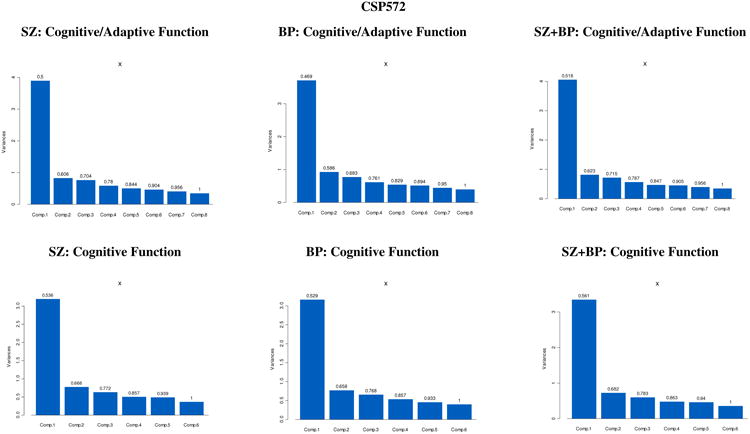
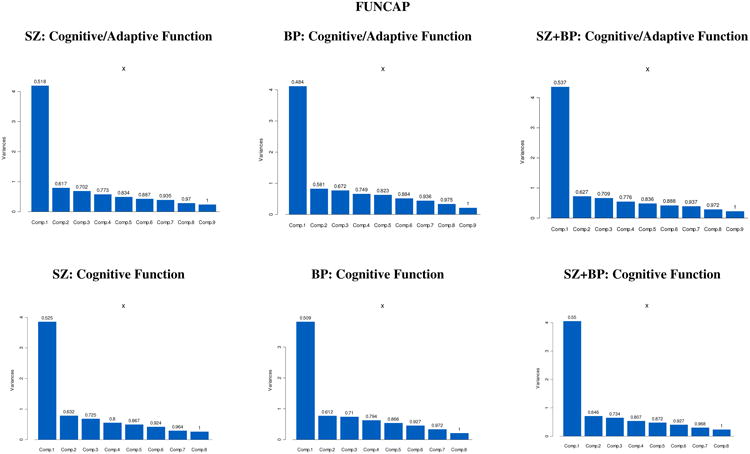
Principal Components analyses in both studies suggest that both cognitive performance and functional capacity measures combined, as well as cognitive performance measures considered alone, can summarily be reduced to one principal component that explains most of the variation in the original variables—in both SZ and BP diagnoses analyzed separately, as well as in the combined sample (see Appendix 1 for eigenvalues, proportion of variance, and cumulative proportion for the first three principal components, and see Appendix 2 for the corresponding individual scree plots). In each of the analyses performed, and for both studies, the first principal component accounted for at least 47% of the variance in the data, and displayed a weighted average of the test scores with all loadings of moderate size.
Maximum likelihood exploratory factor analyses were conducted, testing that estimated and observed covariance matrices were not statistically different. Chi-square test statistics for various factor structure models were compared in conjunction with the factor scree plots, eigenvalues, and factor loadings. In all cases we decided against higher-order factor models as they were not supported by either the inspection of the scree plots, or all but one eigenvalues were less than 1, or due to unclear separation of loading distribution and several item cross-loadings on factors. Exploratory factor analyses in both datasets suggested that cognitive performance and functional capacity measures could be explained in terms of a unique underlying factor that accounts for most common variance among test scores, in both BP and SCZ diagnoses analyzed separately, as well as in a combined sample.
To ensure generalizability of the exploratory factor analyses findings, we additionally fit confirmatory factor analyses, comparing models with specified one and two factors suggested by the previous exploratory factor analyses. The single-factor model showed best goodness-of-fit statistics: Root Mean Square Error of Approximation (RMSEA) was smallest range 0.04-0.08); Comparative Fit Index (CFI) and Non-normed Fit Index (NNFI)/Tucker-Lewis Index (TLI) were largest (range 0.93-0.99; Table 4). Of note, the factor structure was very similar with and without the inclusion of functional capacity measures. Homogeneous loadings ranged from 0.52-0.78 in absolute value for the single factor across cognitive domains, similar to loadings for functional capacity (Table 5) in both studies, and pure cognitive performance-based measures factor scores were very similar to the factor scores for cognitive/adaptive measures combined. In all cases, given that the one and two-factor models were not nested, we used the Akaike Information Criteria (AIC), the Bayesian Information Criteria (BIC), and the sample size adjusted BIC to compare model fits; throughout, all these values were smaller for the one-factor model, suggesting a better fit to the data than the two-factor model.
Table 4.
Goodness-of-Fit Statistics from Confirmatory Factor Analyses.
| CSP572 | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Model | GOF χ2, df, p-value | RMSEA (90% CI) | CFI | NNFI | Model | GOF χ2, df, p-value | RMSEA (90% CI) | CFI | NNFI |
| SZ: CFA with Cognition + Adaptive Function | SZ: CFA with Cognition but w/o Adaptive Function | ||||||||
| Single Factor | 368, 17, < 0.0001 | 0.07, (0.06, 0.08) | 0.96 | 0.93 | Single Factor | 55, 6, < 0.0001 | 0.05, (0.04, 0.06) | 0.99 | 0.98 |
| Two Factor | 641, 19, < 0.0001 | 0.09, (0.08, 0.10) | 0.93 | 0.89 | Two Factor | 258, 8, < 0.0001 | 0.09, (0.08, 0.10) | 0.96 | 0.93 |
| BP: CFA with Cognition + Adaptive Function | BP: CFA with Cognition but w/o Adaptive Function | ||||||||
| Single Factor | 453, 17, < 0.0001 | 0.07, (0.06, 0.08) | 0.96 | 0.93 | Single Factor | 55, 6, < 0.0001 | 0.04, (0.03, 0.05) | 0.99 | 0.99 |
| Two Factor | 797, 19, < 0.0001 | 0.09, (0.08, 0.10) | 0.92 | 0.89 | Two Factor | 317, 8, < 0.0001 | 0.09, (0.08, 0.10) | 0.96 | 0.93 |
| SZ+BP: CFA with Cognition + Adaptive Function | SZ+BP: CFA with Cognition but w/o Adaptive Function | ||||||||
| Single Factor | 897, 17, < 0.0001 | 0.08, (0.07, 0.09) | 0.96 | 0.93 | Single Factor | 87, 6, < 0.0001 | 0.04, (0.03, 0.05) | 0.99 | 0.99 |
| Two Factor | 1550, 19, < 0.0001 | 0.09, (0.08, 0.10) | 0.93 | 0.90 | Two Factor | 556, 8, < 0.0001 | 0.09, (0.08, 0.10) | 0.97 | 0.94 |
| FUNCAP | |||||||||
| Model | GOF χ2, df, p-value | RMSEA (90% CI) | CFI | NNFI | Model | GOF χ2, df, p-value | RMSEA (90% CI) | CFI | NNFI |
| SZ: CFA with Cognition + Adaptive Function | SZ: CFA with Cognition but w/o Adaptive Function | ||||||||
| Single Factor | 57, 21, <0.0001 | 0.07, (0.05, 0.09) | 0.97 | 0.94 | Single Factor | 41, 14, 0.0002 | 0.07 (0.05, 0.10) | 0.97 | 0.94 |
| Two Factor | 109, 26, <0.0001 | 0.09 (0.07, 0.11) | 0.93 | 0.90 | Two Factor | 91, 19, <0.0001 | 0.10 (0.08, 0.12) | 0.93 | 0.89 |
| BP: CFA with Cognition + Adaptive Function | BP: CFA with Cognition but w/o Adaptive Function | ||||||||
| Single Factor | 45, 21, 0.0019 | 0.06 (0.03, 0.08) | 0.96 | 0.94 | Single Factor | 39, 14, 0.0004 | 0.07 (0.05, 0.10) | 0.97 | 0.93 |
| Two Factor | 97, 26, <0.0001 | 0.09 (0.07, 0.11) | 0.94 | 0.92 | Two Factor | 80, 19, <0.0001 | 0.10 (0.08, 0.12) | 0.94 | 0.92 |
| SZ+BP: CFA with Cognition + Adaptive Function | SZ+BP: CFA with Cognition but w/o Adaptive Function | ||||||||
| Single Factor | 112, 21, <0.0001 | 0.08 (0.06, 0.09) | 0.96 | 0.92 | Single Factor | 82, 14, <0.0001 | 0.08 (0.06, 0.10) | 0.96 | 0.93 |
| Two Factor | 200, 26, <0.0001 | 0.10 (0.08, 0.11) | 0.92 | 0.88 | Two Factor | 172, 19, <0.0001 | 0.10 (0.09, 0.12) | 0.94 | 0.92 |
GOF: Goodness of Fit; χ2: chi-square; df: degrees of freedom
RMSEA: Root Mean Square Error of Approximation (smaller -> better)
CFI: Comparative Fit Index (larger -> better)
NNFI: Non-normed Fit Index/Tucker-Lewis Index (larger -> better)
Table 5.
Factor loadings for the different models in the two samples.
| CSP 572 | |||||
|---|---|---|---|---|---|
| SZ: Cognitive/Adaptive Function | BP: Cognitive/Adaptive Function | SZ+BP: Cognitive/Adaptive Function | |||
| UPSA | 0.68 | UPSA | 0.55 | UPSA | 0.66 |
| EFB | 0.70 | EFB | 0.59 | EFB | 0.69 |
| TMTA | −0.54 | TMTA | −0.59 | TMTA | −0.59 |
| MCCB | 0.72 | MCCB | 0.70 | MCCB | 0.74 |
| HVLT | 0.61 | HVLT | 0.60 | HVLT | 0.64 |
| LNS | 0.74 | LNS | 0.71 | LNS | 0.75 |
| Animal | 0.53 | Animal | 0.53 | Animal | 0.57 |
| NAB | 0.58 | NAB | 0.60 | NAB | 0.61 |
| SZ: Cognitive Function | BP: Cognitive Function | SZ+BP: Cognitive Function | |||
| TMTA | −0.54 | TMTA | −0.58 | TMTA | −0.59 |
| MCCB | 0.74 | MCCB | 0.71 | MCCB | 0.75 |
| HVLT | 0.64 | HVLT | 0.62 | HVLT | 0.66 |
| LNS | 0.73 | LNS | 0.70 | LNS | 0.74 |
| Animal | 0.59 | Animal | 0.58 | Animal | 0.62 |
| NAB | 0.59 | NAB | 0.62 | NAB | 0.63 |
| FUNCAP | |||||
| SZ: Cognitive/Adaptive Function | BP: Cognitive/Adaptive Function | SZ+BP: Cognitive/Adaptive Function | |||
| UPSA-B | 0.74 | UPSA-B | 0.55 | UPSA-B | 0.71 |
| TMTA | −0.61 | TMTA | −0.66 | TMTA | −0.66 |
| TMTB | −0.76 | TMTB | −0.72 | TMTB | −0.76 |
| Animal | 0.52 | Animal | 0.53 | Animal | 0.56 |
| Dygsym | 0.63 | Dygsym | 0.72 | Dygsym | 0.70 |
| LNS | 0.77 | LNS | 0.66 | LNS | 0.74 |
| RAVLT | 0.67 | RAVLT | 0.60 | RAVLT | 0.69 |
| CPT | 0.72 | CPT | 0.69 | CPT | 0.73 |
| WCST | −0.58 | WCST | −0.59 | WCST | −0.62 |
| SZ: Cognitive Function | BP: Cognitive Function | SZ+BP: Cognitive Function | |||
| TMTA | −0.62 | TMTA | −0.65 | TMTA | −0.66 |
| TMTB | −0.75 | TMTB | −0.69 | TMTB | −0.74 |
| Animal | 0.55 | Animal | 0.54 | Animal | 0.58 |
| Dygsym | 0.65 | Dygsym | 0.73 | Dygsym | 0.72 |
| LNS | 0.78 | LNS | 0.65 | LNS | 0.74 |
| RAVLT | 0.67 | RAVLT | 0.61 | RAVLT | 0.69 |
| CPT | 0.74 | CPT | 0.71 | CPT | 0.75 |
| WCST | −0.57 | WCST | −0.59 | WCST | −0.62 |
Discussion
The results of this study found that cognitive performance and performance-based indices of functional capacity were defined by a single statistical dimension. A single factor solution fit the data in two separate study samples assessed with slightly different cognitive assessments, and the solution was unifactorial across: 1) diagnostic groups, 2) the inclusion or exclusion of functional capacity measures, and 3) across samples with very different demographic characteristics. The similarity of the solutions across wide variations in educational, racial, and ethnic backgrounds, and their attendant correlates in life experience, underscores the commonalities of these performance-based impairments in these two neuropsychiatric conditions. In every comparison, the single factor model fit better than a model that posited that functional capacity was a separate factor.
Although years of research has focused on symptomatic and treatment differences between schizophrenia and bipolar illness, recent data cited above suggests that there may be common influences on the illness and common influences on cognitive impairments. Symptomatic treatments in schizophrenia and bipolar disorder, although different (e.g., lithium for bipolar disorder) are equivalently effective at symptomatic reduction within each condition and equivalently inadequate at improvement of cognitive functioning and everyday disability. Previous analyses of the FUNCAP data have suggested that the everyday outcomes in both schizophrenia and bipolar disorder have similar predictors in the domains of cognition and functional capacity (Bowie et al., 2010; Mausbach et al., 2010). These findings of similar correlations with community functioning, combined with our findings of highly similar factor structures for cognition, and a unifactorial solution for cognition and functional capacity, will provide guidance for future genomic analyses which will examine commonalities in the relationships of genomic factors to cognition and functional capacity across the two diagnostic groups.
The COGS study examined genomic correlates of individual performance-based tests, finding different maximal LOD scores at different loci for spatial processing (2p25 and 16q23), sensorimotor dexterity (2q24 and 2q32), the California Verbal Learning Test (8q24), the degraded-stimulus Continuous Performance Test (10q26), face memory (10q26 and 12p12), and the Letter-Number Span (14q23 (Greenwood et al., 2011). These results suggest that it may be possible to identify genomic association to individual performance-based domains. A recent paper (Seidman et al., 2015) presented a factor analysis of the COGS-II data, including schizophrenia patients (n=83), their nonpsychotic siblings (n=151), and community comparison subjects (n=209) with complete data on a battery of 12 neurocognitive and social cognitive tests. Their results suggested a multi-factorial solution with 5 distinct factors. The variation in impairment levels across this very diverse subject sample may have led to a more heterogenous factor structure, and only 20% of the cases had a schizophrenia diagnosis. Of interest is the fact that the working memory and episodic verbal memory factors demonstrated significant heritability, consistent with the findings in previous COGS analyses. Our large sample size, and soon to be available genomic data, will allow us to attempt to replicate the findings of genomic association with multi-trial verbal learning and letter-number sequencing generated in the COGS study, which seem supported by the latest heritability analyses.
Several limitations apply to the analyses of the data from these two initiatives, some inherent to the study of Veterans, and other highly selected samples, and others arising from the practical decisions necessary to conduct an in-person studies of over 10,000 people with severe mental illness. Veterans typically do not have a particularly early age of onset, given the need for being “healthy” at entry into military service; in addition, most Veterans (including those in this sample) are male. AJ samples are more educated that the US population as a whole, and the sample had relatively better performance on various measures than earlier samples of patients. Accordingly, and as expected, this sample does not include participants with illness for less than 5 years, and is older as well as predominately male. In addition, performance-based assessment of intelligence was not possible within the scope of time per case that was allocated to assessment. Everyday outcomes were not assessed in the CSP 572, because of the logistical challenges of informant-based assessments in the large sample and the knowledge that self-reports of everyday functioning may have limited validity in these populations. Finally, it may be possible in the future to collect these data with greater efficiency by using either computer or smart-phone technology to remotely deliver neuropsychological and functional capacity assessments and possibly to measure everyday outcomes with ecological momentary assessment strategies. Selection of potential endophenotypes is also affected by the research design. Several of the psychophysiological measures from the COGS study would also not be feasible in a study like CSP 572, and those results were not yet available at the time the FUNCAP study was launched.
The factor structure of cognition in severe mental illness can also be affected by the assessment strategies. Larger and more detailed cognitive assessment batteries and smaller samples have sometimes led to more complex solutions (Gladsjo, et al., 2004). In that study, a six-factor solution was found and functional capacity was found to correlate with all of the 6 factors at levels that ranged from r=.46 to r=.64. The same number of factors were found in the two comparative studies of bipolar disorder and schizophrenia cited above (Czobor et al., 2007; Schretlen, et al., 2013), with similarly large batteries and smaller samples. Although these are not small-scale studies by most standards, the largest in-person study of people with severe mental illness with performance-based assessments prior to the current one, Keefe et al. (2006), with an analyzable sample of 1332 schizophrenia patients and a cognitive assessment battery with 9 different tests, three of them computerized, also found a single-factor solution as the best fitting model. The large sample size in the CSP 572 sample, and the extreme similarity of the fit of the factor models in the two different studies, argues against the assertion that the current results are not the best model that could be derived from this set of tests. Further, consistent findings in multiple studies (Leifker et al., 2011) suggest high levels of correlation between UPSA-B (and UPSA) scores and NP test performance.
In summary, the results of the current analyses suggest that examination of performance based measures of cognition and functional capacity as a single ability variable may have usefulness for identification of genomic contributions to cognitive and functional deficits. The evidence from the COGS studies of association to specific genomic variants and high levels of heritability of at two of the components of our assessment (episodic verbal memory and verbal working memory) will allow for highly powered examination of association to global ability traits versus specific endophenotypes, and will also allow us to compare the association of genomics to global versus specific cognitive ability domains within and across large samples of people with diagnoses of schizophrenia and bipolar disorder.
Acknowledgments
Funding source: This research was supported by the Department of Veterans Affairs Cooperative Studies Program.
Dr. Harvey has served as a consultant to Boeheringer Ingelheim, Forum Pharmaceuticals, Genentech, Otsuka America, Roche, Sanofi, Sunovion Pharma, and Takeda Pharma, on phase 2 or 3 drug development; this consulting work is not related to the content of the paper.
Appendix 1. Principal ComponentAnalyses: Eigenvalues, Proportion of Variance Explained, and Cumulative Proportion
| CSP572 | |||||||
|---|---|---|---|---|---|---|---|
| SZ: CFA with Cognition + Adaptive Function | SZ: CFA with Cognition but w/o Adaptive Function | ||||||
| Component 1 | Component 2 | Component 3 | Component 1 | Component 2 | Component 3 | ||
| Eigen Values | 3.89 | 0.82 | 0.76 | Eigen Values | 3.19 | 0.78 | 0.63 |
| Proportion of Variance | 0.50 | 0.11 | 0.10 | Proportion of Variance | 0.54 | 0.13 | 0.11 |
| Cumulative Proportion | 0.50 | 0.61 | 0.70 | Cumulative Proportion | 0.54 | 0.67 | 0.77 |
| BP: CFA with Cognition + Adaptive Function | BP: CFA with Cognition but w/o Adaptive Function | ||||||
| Component 1 | Component 2 | Component 3 | Component 1 | Component 2 | Component 3 | ||
| Eigen Values | 3.71 | 0.93 | 0.77 | Eigen Values | 3.16 | 0.77 | 0.66 |
| Proportion of Variance | 0.47 | 0.12 | 0.11 | Proportion of Variance | 0.53 | 0.13 | 0.11 |
| Cumulative Proportion | 0.47 | 0.59 | 0.68 | Cumulative Proportion | 0.53 | 0.66 | 0.77 |
| SZ+BP: CFA with Cognition + Adaptive Function | SZ+BP: CFA with Cognition but w/o Adaptive Function | ||||||
| Component 1 | Component 2 | Component 3 | Component 1 | Component 2 | Component 3 | ||
| Eigen Values | 4.05 | 0.82 | 0.72 | Eigen Values | 3.35 | 0.73 | 0.60 |
| Proportion of Variance | 0.52 | 0.10 | 0.10 | Proportion of Variance | 0.56 | 0.12 | 0.10 |
| Cumulative Proportion | 0.52 | 0.62 | 0.78 | Cumulative Proportion | 0.56 | 0.68 | 0.78 |
| FUNCAP | |||||||
| SZ: CFA with Cognition + Adaptive Function | SZ: CFA with Cognition but w/o Adaptive Function | ||||||
| Component 1 | Component 2 | Component 3 | Component 1 | Component 2 | Component 3 | ||
| Eigen Values | 4.20 | 0.80 | 0.69 | Eigen Values | 3.85 | 0.79 | 0.68 |
| Proportion of Variance | 0.52 | 0.10 | 0.09 | Proportion of Variance | 0.52 | 0.11 | 0.09 |
| Cumulative Proportion | 0.52 | 0.62 | 0.70 | Cumulative Proportion | 0.52 | 0.63 | 0.72 |
| BP: CFA with Cognition + Adaptive Function | BP: CFA with Cognition but w/o Adaptive Function | ||||||
| Component 1 | Component 2 | Component 3 | Component 1 | Component 2 | Component 3 | ||
| Eigen Values | 4.12 | 0.83 | 0.77 | Eigen Values | 3.82 | 0.77 | 0.74 |
| Proportion of Variance | 0.48 | 0.10 | 0.09 | Proportion of Variance | 0.51 | 0.10 | 0.10 |
| Cumulative Proportion | 0.48 | 0.58 | 0.67 | Cumulative Proportion | 0.51 | 0.61 | 0.71 |
| SZ+BP: CFA with Cognition + Adaptive Function | SZ+BP: CFA with Cognition but w/o Adaptive Function | ||||||
| Component 1 | Component 2 | Component 3 | Component 1 | Component 2 | Component 3 | ||
| Eigen Values | 4.36 | 0.72 | 0.67 | Eigen Values | 4.06 | 0.71 | 0.65 |
| Proportion of Variance | 0.54 | 0.09 | 0.08 | Proportion of Variance | 0.55 | 0.10 | 0.09 |
| Cumulative Proportion | 0.54 | 0.63 | 0.71 | Cumulative Proportion | 0.55 | 0.65 | 0.73 |
Appendix 2. Individual scree plots for different measure groupings in the two samples
|
|
Footnotes
Conflicts of interest: No other authors report any conflicts of interest.
References
- Bowie CR, Depp C, McGrath JA, Wolyniec P, Mausbach BT, Thornquist MH, Luke J, Pulver AE. Prediction of real-world functional disability in chronic mental disorders: a comparison of schizophrenia and bipolar disorder. American Journal of Psychiatry. 2010;167:1116–1124. doi: 10.1176/appi.ajp.2010.09101406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Braff DL, Freedman R, Schork NJ, Gottesman II. Deconstructing schizophrenia: an overview of the use of endophenotypes in order to understand a complex disorder. Schizophrenia Bulletin. 2007;33:21–32. doi: 10.1093/schbul/sbl049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bray SM, Mulle JG, Dodd AF, Pulver AE, Wooding S, Warren ST. Signatures of founder effects, admixture, and selection in the Ashkenazi Jewish population. Proceedings of the National Academy of Sciences. 2010;107:16222–16227. doi: 10.1073/pnas.1004381107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen PL, Avramopoulos D, Lasseter VK, McGrath JA, Fallin MD, Liang KY, et al. Valle D. Fine mapping on chromosome 10q22-q23 implicates Neuregulin 3 in schizophrenia. The American Journal of Human Genetics. 2009;84:21–34. doi: 10.1016/j.ajhg.2008.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Czobor P, Jaeger J, Berns SM, Gonzalez C, Loftus S. Neuropsychological symptoms dimensions in biloar disorder and schizophrenia. Bipolar Disorders. 2007;9:71–92. doi: 10.1111/j.1399-5618.2007.00428.x. [DOI] [PubMed] [Google Scholar]
- Dickinson D, Iannone VN, Wilk CM, Gold JM. General and specific cognitive deficits in schizophrenia. Biological Psychiatry. 2004;55:826–33. doi: 10.1016/j.biopsych.2003.12.010. [DOI] [PubMed] [Google Scholar]
- Fallin MD, Lasseter VK, Wolyniec PS, McGrath JA, Nestadt G, Valle D, Liang KY, Pulver AE. Genomewide linkage scan for schizophrenia susceptibility loci among Ashkenazi Jewish families shows evidence of linkage on chromosome 10q22. American Journal of Human Genetics. 2003;73:601–611. doi: 10.1086/378158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fallin MD, Lasseter VK, Wolyniec PS, McGrath JA, Nestadt G, Valle D, Liang KY, Pulver AE. Genomewide linkage scan for bipolar-disorder susceptibility loci among Ashkenazi Jewish families. American Journal of Human Genetics. 2004;75:204–219. doi: 10.1086/422474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fallin MD, Lasseter VK, Avramopoulos D, Nicodemus KK, Wolyniec PS, McGrath JA, et al. Pulver AE. Bipolar I disorder and schizophrenia: a 440-single-nucleotide polymorphism screen of 64 candidate genes among Ashkenazi Jewish case-parent trios. American Journal of Human Genetics. 2005;77:918–936. doi: 10.1086/497703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- First MB, Spitzer RL, Gibbon M, Williams JBW. User's Guide for the Structured Clinical Interview for DSM-IV Axis I (SCID-I) Washington, DC: American Psychiatric Press; 1997. [Google Scholar]
- Gladsjo JA, McAdams LA, Palmer BW, Moore DJ, Jeste DV, Heaton RK. A six-factor model of cognition in schizophrenia and related psychotic disorders: relationships with clinical symptoms and functional capacity. Schizophrenia Bulletin. 2004;30:739–54. doi: 10.1093/oxfordjournals.schbul.a007127. [DOI] [PubMed] [Google Scholar]
- Greenwood TA, Braff DL, Light GA, Cadenhead KS, Calkins ME, Dobie DJ, et al. Schork NJ. Initial heritability analyses of endophenotypic measures for schizophrenia: the consortium on the genetics of schizophrenia. Archives of General Psychiatry. 2007;64:1242–1250. doi: 10.1001/archpsyc.64.11.1242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greenwood TA, Lazzeroni LC, Murray SS, Cadenhead KS, Calkins ME, Dobie DJ, et al. Braff DL. Analysis of 94 candidate genes and twelve endophenotypes for schizophrenia from the Consortium on the Genetics of Schizophrenia. American Journal of Psychiatry. 2011;168:930–946. doi: 10.1176/appi.ajp.2011.10050723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gur RE, Calkins ME, Gur RC, Horan WP, Nuechterlein KH, Seidman LJ, Stone WS. The consortium on the genetics of schizophrenia: neurocognitive endophenotypes. Schizophrenia Bulletin. 2007;33:49–68. doi: 10.1093/schbul/sbl055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harvey PD, Heaton RK, Carpenter WT, Green MF, Gold JM, Schoenbaum M. Diagnosis of schizophrenia: consistency across information sources and stability of the condition. Schizophrenia Research. 2012;140:9–14. doi: 10.1016/j.schres.2012.03.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harvey PD, Helldin L, Bowie CR, Heaton RK, Olsson AK, Patterson TL. Performance-based measurement of functional disability in schizophrenia: a cross-national Study in the United States and Sweden. American Journal of Psychiatry. 2009;166:821–827. doi: 10.1176/appi.ajp.2009.09010106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harvey PD, Keefe RSE. Technology, society, and mental illness: challenges and opportunities for assessment and intervention. Innovations in Clinical Neuroscience. 2012;9:47–50. [PMC free article] [PubMed] [Google Scholar]
- Harvey PD, McClure MM, Patterson TL, McGrath JA, Pulver AE, Bowie CR, Siever LJ. Impairment in functional capacity as an endophenotype candidate in severe mental illness. Schizophrenia Bulletin. 2012;38:1318–1326. doi: 10.1093/schbul/sbr046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harvey PD, Raykov T, Twamley EW, Vella L, Heaton RK, Patterson TL. Validating the measurement of real-world functional outcome: phase I results of the VALERO study. American Journal of Psychiatry. 2011;168:1195–201. doi: 10.1176/appi.ajp.2011.10121723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harvey PD, Raykov T, Twamley E, Vella L, Heaton RK, Patterson TL. Factor structure of neurocognition and functional capacity in schizophrenia: A multidimensional examination of temporal stability. Journal of the International Neuropsychological Society. 2013;19:656–663. doi: 10.1017/S1355617713000179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harvey PD, Siever LJ, Huang GD, Muralidhar S, Zhao H, Miller P, et al. Concato J. The genetics of functional disability in schizophrenia and bipolar illness: methods and initial results for VA Cooperative Study #572. Neuropsychiatric Genetics. 2014;165:381–389. doi: 10.1002/ajmg.b.32242. [DOI] [PubMed] [Google Scholar]
- Heaton RK, Marcotte TD, Mindt MR, Sadek J, Moore DJ, Bentley H, Grant I HNRC Group. The impact of HIV-associated neuropsychological impairment on everyday functioning. Journal of the International Neuropsychological Society. 2004;10:317–331. doi: 10.1017/S1355617704102130. [DOI] [PubMed] [Google Scholar]
- Keefe RS, Bilder RM, Harvey PD, Davis SM, Palmer BW, Gold JM, et al. Lieberman JA. Baseline neurocognitive deficits in the CATIE Schizophrenia Trial. Neuropsychopharmacology. 2006;31:2033–2046. doi: 10.1038/sj.npp.1301072. [DOI] [PubMed] [Google Scholar]
- Keefe RSE, Fox KH, Harvey PD, Cucchiaro J, Siu C, Loebel A. Characteristics of the MATRICS Consensus Cognitive Battery in a 29-site antipsychotic schizophrenia clinical trial. Schizophrenia Research. 2011;125:161–168. doi: 10.1016/j.schres.2010.09.015. [DOI] [PubMed] [Google Scholar]
- Keefe RS, Goldberg TE, Harvey PD, Gold JM, Poe MP, Coughenour L. The brief assessment of cognition in schizophrenia: reliability, sensitivity, and comparison with a standard neurocognitive battery. Schizophrenia Research. 2004;68:283–297. doi: 10.1016/j.schres.2003.09.011. [DOI] [PubMed] [Google Scholar]
- Kieseppä T, Partonen T, Haukka J, Kaprio J, Lönnqvist J. High concordance of bipolar I disorder in a nationwide sample of twins. American Journal of Psychiatry. 2004;10:1814–1821. doi: 10.1176/ajp.161.10.1814. [DOI] [PubMed] [Google Scholar]
- Leifker FR, Patterson TL, Heaton RK, Harvey PD. Validating measures of real-world outcome: The results of the VALERO Expert Survey and RAND Appropriateness Panel. Schizophrenia Bulletin. 2011;37:334–343. doi: 10.1093/schbul/sbp044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lichtenstein P, Yip BH, Björk C, Pawitan Y, Cannon TD, Sullivan PF, Hultman CM. Common genetic determinants of schizophrenia and bipolar disorder in Swedish families: a population-based study. Lancet. 2009;373:234–239. doi: 10.1016/S0140-6736(09)60072-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mardia KV, Kent JT, Bibby JM. Multivariate Analysis. London: Academic Press; 1979. [Google Scholar]
- Mausbach BT, Harvey PD, Goldman SR, Jeste DV, Patterson TL. Development of a brief scale of everyday functioning in persons with serious mental illness. Schizophrenia Bulletin. 2007;33:1364–1372. doi: 10.1093/schbul/sbm014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mausbach BT, Harvey PD, Pulver AE, Depp CA, McGrath JA, Bowie CR, Patterson TL. Relationship of the Brief UCSD Performance-based Skills Assessment (UPSA-B) to multiple indicators of functioning in people with schizophrenia and bipolar disorder. Bipolar Disorders. 2010;2:45–55. doi: 10.1111/j.1399-5618.2009.00787.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McIntosh BJ, Zhang XY, Kosten T, Tan SP, Xiu MH, Harvey PD. Performance based assessment of functional skills in severe mental illness: results of a large-scale study in China. Journal of Psychiatric Research. 2011;45:1089–1094. doi: 10.1016/j.jpsychires.2011.01.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neuchterlein KH, Green MF, Kern RS, Baade LE, Barch DM, Cohen JD, et al. Marder SR. The MATRICS Consensus Cognitive Battery: part 1: test selection, reliability, and validity. American Journal of Psychiatry. 2008;165:203–213. doi: 10.1176/appi.ajp.2007.07010042. [DOI] [PubMed] [Google Scholar]
- Schretlen DJ, Peña J, Aretouli E, Orue I, Cascella NG, Pearlson GD, Ojeda N. Confirmatory factor analysis reveals a latent cognitive structure common to bipolar disorder, schizophrenia, and normal controls. Bipolar Disorders. 2013;15:422–433. doi: 10.1111/bdi.12075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seidman LJ, Hellemann G, Nuechterlein KH, Greenwood TA, Braff DL, Cadenhead KS, et al. Green MF. Factor structure and heritability of endophenotypes in schizophrenia: findings from the Consortium on the Genetics of Schizophrenia (COGS-1) Schizophrenia Research. 2015;163:73–79. doi: 10.1016/j.schres.2015.01.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sullivan PF, Kendler KS, Neale MC. Schizophrenia as a complex trait: evidence from a meta-analysis of twin studies. Archives of General Psychiatry. 2003;60:1187–1192. doi: 10.1001/archpsyc.60.12.1187. [DOI] [PubMed] [Google Scholar]
- Wingo AP, Harvey PD, Baldessarini RJ. Neurocognitive impairment in bipolar disorder patients: functional implications. Bipolar Disorders. 2009;11:113–125. doi: 10.1111/j.1399-5618.2009.00665.x. [DOI] [PubMed] [Google Scholar]


