Abstract
Pluripotent embryonic stem cells (ESCs) are characterized by distinct epigenetic features including a relative enrichment of histone modifications related to active chromatin. Among these is tri‐methylation of lysine 4 on histone H3 (H3K4me3). Several thousands of the H3K4me3‐enriched promoters in pluripotent cells also contain a repressive histone mark, namely H3K27me3, a situation referred to as “bivalency”. While bivalent promoters are not unique to pluripotent cells, they are relatively enriched in these cell types, largely marking developmental and lineage‐specific genes which are silent but poised for immediate action. The H3K4me3 and H3K27me3 modifications are catalyzed by lysine methyltransferases which are usually found within, although not entirely limited to, the Trithorax group (TrxG) and Polycomb group (PcG) protein complexes, respectively, but these do not provide selective bivalent specificity. Recent studies highlight the family of ATP‐dependent chromatin remodeling proteins as regulators of bivalent domains. Here, we discuss bivalency in general, describe the machineries that catalyze bivalent chromatin domains, and portray the emerging connection between bivalency and the action of different families of chromatin remodelers, namely INO80, esBAF, and NuRD, in pluripotent cells. We posit that chromatin remodeling proteins may enable “bivalent specificity”, often selectively acting on, or selectively depleted from, bivalent domains.
Keywords: chromatin, chromatin remodeling, embryonic stem cells, epigenetics, histone modifications
Subject Categories: Chromatin, Epigenetics, Genomics & Functional Genomics; Stem Cells
Glossary
- ASH2L
Ash2 (absent, small, or homeotic)‐like (Drosophila) protein
- BAF250a
AT‐rich interactive domain 1A (Swi1 like) protein
- Bmi1
B lymphoma Mo‐MLV insertion region 1 protein
- BRG1
SWI/SNF‐related, matrix‐associated, actin‐dependent regulator of chromatin, subfamily a, member 4
- Cbx7
chromobox homolog 7 protein
- CFP1
CXXC finger 1 (PHD domain) protein
- CHARGE
coloboma, heart defect, atresia choanae (also known as choanal atresia), retarded growth and development, genital abnormality, and ear abnormality
- CHD
chromodomain‐helicase‐DNA‐binding protein
- ChIP
chromatin immunoprecipitation
- EED
embryonic ectoderm development protein
- esBAF
brahma‐associated factor complex associated with embryonic stem cells
- ESC
embryonic stem cell
- EZH
enhancer of zeste homolog protein
- H3K27me3
trimethylated lysine‐27 on histone H3
- H3K4me3
trimethylated lysine‐4 on histone H3
- HCNE
highly conserved non‐coding elements
- INO80
inositol‐requiring 80
- iPSC
induced pluripotent stem cell
- ISWI
imitation switch
- LSD
lysine‐specific demethylase 1
- MLL
mixed lineage leukemia protein
- MNase
micrococcal nuclease
- MYC
v‐myc avian myelocytomatosis viral oncogene homolog
- NuRD
nucleosome remodeling deacetylase
- OCT4
POU domain, class 5, transcription factor 1 protein
- PcG
polycomb group
- Phc
polyhomeotic‐like 1 (Drosophila) protein
- PRC
polycomb repressive complex
- PTMs
posttranslational modifications
- RBBP5
retinoblastoma‐binding protein 5
- RING1B
ring finger protein 2
- rRNA
ribosomal RNA
- SET
SET domain containing protein
- SMARCD1
SWI/SNF‐related, matrix‐associated, actin‐dependent regulator of chromatin, subfamily d, member 1 protein
- SUZ12
suppressor of zeste 12 homolog (Drosophila) protein
- SWI/SNF
SWItch/Sucrose non‐fermentable
- Tip60
K(lysine) acetyltransferase 5
- TrxG
trithorax group
- TSS
transcription start site
- WDR5
WD repeat domain 5 protein
Introduction
The genetic information of a living cell is stored within the DNA. However, additional layers of regulation provide the epigenetic information, which, in concert with transcription factors, enables the same primary DNA sequence to confer different identities to different cell types, developmental stages, disease states, etc. In eukaryotes, the DNA is wrapped around a histone octamer comprised of a pair of each of the core histones H2A, H2B, H3, and H4, which together form the nucleosome. Nucleosomes are the basic repeating units of chromatin, and they are arranged in a higher order chromatin structure through the binding of linker histones, H1 proteins, between adjacent nucleosomes. Thus, despite having the same genetic makeup, different transcriptional outcomes of different cell types of the same organism are achieved through a variety of epigenetic modifications including DNA methylation, histone posttranslational modifications (PTMs), chromatin organization, etc. So far, several histone modifications with physiological importance have been identified, such as acetylation, methylation, phosphorylation, ubiquitylation, sumoylation, ADP ribosylation, deimination, proline isomerization, biotinylation, citrullination, and more 1, 2, 3. Apart from influencing local chromatin structure, these modifications are also recognized by specific adaptor proteins which in turn recruit protein complexes and thereby affect gene regulation. Some of these histone marks such as H3K4me3, H3K9ac, and H3K14ac are associated with actively transcribed genes and some other modifications, for example, H3K27me3 and H3K9me3, are enriched within repressed regions. Activation and repression are believed to occur, at least partly, through charge‐mediated chromatin decompaction and chromodomain‐containing protein binding, respectively 3, 4. Interestingly, a subset of promoters associated with both activating (H3K4me3) and repressive (H3K27me3) marks, also known as “bivalent” modifications, has been discovered in mouse embryonic stem cells (ESCs) 5, 6. Several recent reviews thoroughly covered the field of bivalency, especially in pluripotent ESCs 7, 8, 9, 10. Here, we focus on the emerging link between bivalent histone modifications and ATP‐dependent chromatin remodeling in ESCs. The term “chromatin remodeling” is often used to describe any change or modification to chromatin including histone modification. Here, by “chromatin remodeling”, we specifically refer to the action of the family of ATP‐dependent chromatin remodeling factors, described below. When other forms of chromatin structure alterations are referred to, we describe the specific mode of alteration or modification. We will briefly summarize initial and recent experiments establishing the existence and role of bivalent domains in undifferentiated ESCs and during differentiation, and will argue that both H3K4me3 and H3K27me3, and especially the cross talk between them, are intimately linked with chromatin remodeling in pluripotent ESCs.
Bivalent modifications
The first evidence for the existence of bivalent modifications came from studies in pluripotent mouse ESCs 5, 6. Using sequential chromatin immunoprecipitation (ChIP) and tiling arrays, highly conserved non‐coding elements (HCNEs) were found to be enriched with bivalent histone modifications H3K4me3 and H3K27me3, marking lowly expressed developmental regulators 6. Shortly thereafter, early replicating genes were similarly shown to possess bivalent domains 5. Depletion of the EED subunit of the Polycomb repressive complex 2 (PRC2) led to an almost complete loss of H3K27me3 resulting in an upregulation of the bivalent genes analyzed. Despite the presence of the activating histone marks, the expression of the bivalent genes in both studies varied from very low to no expression, suggesting that these genes are poised for immediate activation. Supporting this notion, upon differentiation, some of these bivalent modifications were resolved, either losing the H3K27me3 mark permitting their expression, or losing the H3K4me3, rendering them stably silent (Fig 1).
Figure 1. The bivalency concept.
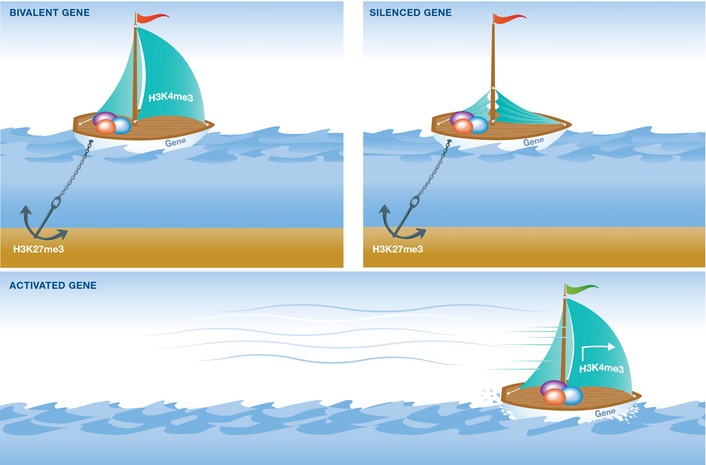
A bivalent gene, depicted as a boat (top left), is ready to go (sail up: H3K4me3) but is held in check (anchor: H3K27me3). Once the sail is down (top right), the gene is stably silenced (only H3K27me3), but if instead the anchor is lifted (bottom), the gene is promptly activated (only H3K4me3). [Correction added on 2 December 2015 after first online publication: “H4K4me3” has been corrected to “H3K4me3”.]
Bivalent histone modifications were also identified in human ESCs 11, 12 and induced pluripotent stem cells (iPSCs) 13, 14, 15, marking developmentally regulated genes, similar to the situation found in mESCs. Other stem cell types, such as hematopoietic stem cells, were also shown to possess a similar bivalent chromatin architecture, containing thousands of bivalently marked, developmentally regulated promoters 16. However, although many of these bivalent domains are resolved during differentiation, a subset of promoters retains its bivalent state following even terminal differentiation 17. Therefore, bivalency may not merely reflect a transient, flexible chromatin state during differentiation, but rather a condition present in most or even all cell types. Supporting this idea, bivalent domains were found in a number of different non‐stem‐cell lines 14, 18, including differentiated human T cells, where weakly expressed genes were found to possess additional acetylation marks on H3K9 and H3K14 along with H3K4me3 and H3K27me3 in their promoters 19, 20. Bivalent domains were reported in several cancer cells as well 21, 22, 23, 24, where they were suggested to promote their plasticity and responsiveness, potentially serving as a novel unexplored therapeutic avenue 22. These studies suggest that bivalent modifications are present in both pluripotent and non‐pluripotent cells, where they maintain genes largely in a repressed state, but at the same time, keep them poised for activation until a proper signal is perceived.
Despite the rapidly expanding literature reporting different aspects of bivalent chromatin, whether bivalent domains serve an actual function has recently been questioned 25, 26. In addition, the existence of bivalent domains on the same nucleosome has not been unequivocally demonstrated. Apart from the many ChIP experiments, bivalent chromatin was shown using micrococcal nuclease (MNase) digestion of chromatin followed by liquid chromatography and mass spectrometry 27, suggesting an asymmetric configuration of chromatin on opposite H3 tails. However, all evidence to date relies on population studies, and therefore, the seeming presence of two opposing marks on the same nucleosome may be the outcome of cellular heterogeneity. Single nucleosome resolution, the smoking gun of bivalent chromatin, has yet to be reported.
Establishment and maintenance of bivalent modifications in ESCs
Trithorax group (TrxG) proteins and Polycomb group (PcG) proteins assemble into multimeric protein complexes and are largely, although not exclusively, responsible for the deposition of H3K4me3 and H3K27me3 marks, respectively (Fig 2). The exact mechanism behind the recruitment of these protein complexes to specific sites is not entirely clear; however, initial studies showed that bivalent domains are predominantly associated with CpG islands in ESCs 6. In addition to DNA methylation, multiple studies have demonstrated that selected histone modifications, several transcription factors (TFs), and some non‐coding RNAs (ncRNAs) also play a role in this process 7.
Figure 2. Main protein complexes catalyzing bivalent chromatin marks.
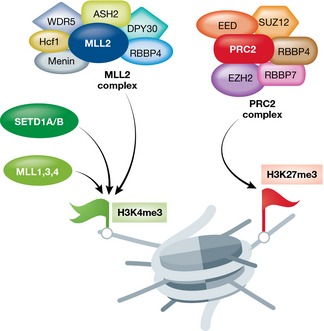
Left: Protein complexes catalyzing H3K4 methylation (green flag). Right: The PRC2 complex catalyzing H3K27 methylation (red flag). Shown are only the main proteins and protein complexes catalyzing H3K4/H3K27 methylation. Less abundant subunits are not depicted. [Correction added on 2 December 2015 after first online publication: “H4K4” has been corrected to “H3K4”.]
H3K4me3
In mammalian systems, SETD1A, SETD1B, and MLL complexes, among others, which share several subunits including WDR5, RBBP5, ASH2L, and DPY‐30, catalyze the deposition of the H3K4me3 mark 28 (Fig 2). SETD1A/B complexes seem to be responsible for global H3K4me3 deposition, whereas MLL1–MLL4 complexes likely serve more specific functions. Among those, MLL2, but not MLL1 or SETD1, was shown to act as the main methyltransferase at bivalent promoters 25, 26. MLL1 and MLL2 contain DNA‐binding domains termed CXXC or zinc finger CXXC (ZF‐CXXC) motifs, which specifically recognize unmethylated CpG islands 29, 30. These motifs act to recruit MLL complexes to chromatin templates by promoting target site recognition. Similarly, SETD1A/B complexes contain a CXXC finger protein 1 (CFP1) subunit, which includes a DNA‐binding domain selectively recognizing unmethylated CpGs 31. Loss of CFP1 most strongly affects H3K4 methylation at promoters of highly expressed genes in ESCs, but not at bivalent gene promoters 32. Other SETD1A/B and MLL components were also shown to be important in ESCs or early ESC differentiation. Knockdown of WDR5 or ASH2L, for instance, results in aberrant expression programs and defective self‐renewal and pluripotency 33, 34, 35, 36, and knockdown of RBBP5 or DPY‐30 has little effect on self‐renewal but leads to improper ESC neuronal differentiation 37. Interestingly, knockdown of DPY‐30 alters H3K4 methylation specifically at bivalent domains in ESCs 38, suggesting a selective developmentally related function of this subunit. MLL2 depletion also results in skewed differentiation, along all three germ layers 39, and Mll2‐null mice die before embryonic day E11.5, showing drastically reduced expression of several Hox genes 40. Taken together, these studies highlight the important role that H3K4 methylation and its maintenance plays in development, pluripotency, ESC biology, and early ESC differentiation.
H3K27me3
The PRC2 complex is responsible for the deposition of H3K27me3 marks at bivalent promoters (Fig 2). The core PRC2 complex is composed of enhancer of zeste (EZH2 or EZH1), embryonic ectoderm development (EED), suppressor of zeste 12 (SUZ12), as well as RBBP4 (RbAp48) and RBBP7 (RbAp46) 41. EZH2 is the catalytic subunit, acting as the methyltransferase of H3K27, which in turn is recognized by chromodomain‐containing proteins such as CBX proteins, as well as by the EED subunit itself 42. In mouse ESCs, CBX7, for instance, the primary CBX protein expressed in ESCs 43, 44, was shown to interact with H3K27me3 thereby recruiting the PRC1 complex 43, 45. RING1B and BMI1 subunits of the PRC1 complex in turn deposit the ubiquitination of H2AK119 46, 47. It was further suggested that the deposition of PRC1 and the H2AK119ub mark (H2A ubiquitylated on lysine 119) are involved in RNA polymerase 2 (RNAPII) pausing in gene bodies, rendering their repression 48, 49. PRC1‐related H2AK119ub1 was also shown to recruit PRC2 to chromatin, demonstrating a functional link between the two complexes 50. Indeed, many developmentally regulated genes are marked with bivalent domains consisting of both PRC1 and PRC2 complexes, but interestingly, a subset of these bivalent domains have been shown to be exclusively bound by PRC2 51. Unlike the PRC1/PRC2 double positive bivalent domains, these PRC2‐specific bivalent domains usually decorate promoters of genes which are not bona fide developmental genes (often encoding for membrane proteins or proteins of unknown functions) and are only weakly conserved. These findings suggest an additional mechanism of silencing. In ESCs, pluripotency factor binding sites often coincide with positioning of core subunits of MLL and PRC2 complexes on bivalent domains 6, 34, 52. In addition, key pluripotency components, such as OCT4 and MYC, have been shown to interact with components of the MLL and PRC protein complexes 53, 54, suggesting a tight co‐regulation between bivalent domains and the pluripotency network. Indeed, depletion of OCT4 in ESCs results in a selective reduction of H3K4me3 levels on selected genes, providing evidence for the tight relationship between the pluripotency network and H3K4me3 levels 34, although whether reduced H3K4me3 levels are a cause or consequence of reduced transcription is still under question 55. While these observations suggest a functional connection between the maintenance of bivalent modifications and pluripotency, the exact role that pluripotency factors play at bivalent domains remains largely unclear.
Depletion of individual subunits EED or SUZ12 results in deregulation of lineage‐specific genes, although with minimal impact on cell viability and self‐renewal 56, 57, 58, 59, suggesting that PcG complexes serve little function in ESCs or that in ESCs, alternative compensatory mechanisms exist. In contrast to the situation, when ESCs are kept in an undifferentiated state, ESCs deficient of PRC2 components exhibit aberrant differentiation potential when differentiation is induced 56, 57, 58, 59. These situations parallel the postimplantation lethality phenotypes observed in PRC2 knockout mouse models 60, 61, 62. Concomitantly, depletion of PRC1 components such as RING1B and BMI1 also impairs proper differentiation 63, 64, 65, 66. Simultaneous depletion of RING1B and EED in ESCs provokes an even stronger inclination toward differentiation, although self‐renewal can still be preserved under careful culture conditions, and prolonged differentiation results in cell death 59. Taken together, these knockout models demonstrate that akin to TrxG proteins, PcG proteins—arguably through the control of bivalent target genes encoding developmental regulators—are vital for proper ESC differentiation.
Chromatin remodeling and bivalency
Accumulating evidence suggests a tight interplay between ATP‐dependent chromatin remodeling proteins and bivalent histone modifications. While this connection likely plays a role in most, if not all, cell types, it is particularly pertinent for pluripotent stem cells, because of the relative abundance of chromatin remodelers in ESCs 67, and because of their special connection with bivalency, as described below.
Chromatin remodeling proteins are ATP‐dependent complexes that usually contain a catalytic ATPase subunit in addition to regulatory factors mediating protein–protein and chromatin–protein interactions. Chromatin remodelers act to alter chromatin structure by several different mechanisms, including incorporation or ejection of histone octamers, sliding nucleosomes along chromatin templates, and, by histone exchange, altering nucleosome composition 68 (Fig 3). Chromatin remodeling proteins are generally divided into four major families: SWI/SNF (switch/sucrose non‐fermentable), CHD (chromodomain‐helicase DNA‐binding), ISWI (imitation switch), and INO80 (inositol‐requiring 80), each involving different protein complexes and often different and even opposing actions. A growing number of chromatin remodeling proteins have been linked to ESC function and pluripotency and were shown to play essential roles in stemness and/or early differentiation and development 69. Interestingly, at least one member of each of these remodeler families was shown to be essential for early mouse development, before or during implantation 69, demonstrating the importance of chromatin remodelers in pluripotency and early fate decisions.
Figure 3. The classical actions of chromatin remodeling proteins.
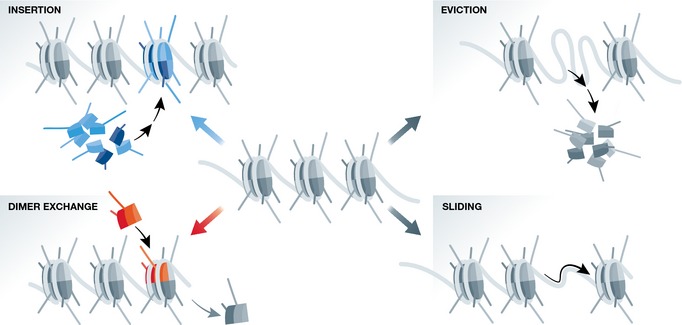
Shown are different functional outcomes mediated by ATP‐dependent chromatin remodeling proteins, including nucleosome insertion (top left), nucleosome eviction (top right), dimer exchange (bottom left), and nucleosome sliding (bottom right). The chromatin remodeling proteins themselves are not depicted.
One of the first clues to the involvement of chromatin remodeling proteins in regulating either H3K4 or H3K27 methylation in developmental genes came from an RNAi screen of chromatin‐related proteins in mouse ESCs 70. Among the dozens of proteins that were identified to have a potential role in maintaining the undifferentiated state in ESCs, the authors specifically identified seven subunits of the Tip60–p400 complex of the INO80 family. Using biochemical and functional assays, the authors found that Tip60–p400 significantly co‐localizes with H3K4me3, especially around the transcription start site (TSS), and that it mostly acts to repress gene expression in ESCs. Knockdown of Tip–p400 resulted in 4% deregulated genes, most of which were upregulated. Interestingly, many of the upregulated genes were found to be classical bivalent early differentiation genes, which are normally silent in ESCs, reminiscent of depletion of PcG components in ESCs 70.
Additionally, PcG proteins were also shown to be functionally linked to chromatin remodeling proteins in ESCs. This relationship was revealed following knockdown (KD) studies of the core component of the SWI/SNF esBAF complex, BRG1, in mouse ESCs 71. Expression analysis following BRG1 KD revealed increased transcription of several PcG subunits of both PRC1 and PRC2 complexes including Bmi1, Cbx7, Ring1, Phc1, and Phc2, promoters of which were directly bound by BRG1, as revealed by ChIP‐seq experiments 71, 72. Moreover, a PRC2 component required for ESC differentiation, Jarid2, interacts with esBAF 73, counteracting PRC2's methyltransferase activity 74, 75. These results suggest a direct association of chromatin remodeling proteins both with promoters of PcG‐related genes and with the PRC2 protein complex itself. However, no co‐localization of PRC2 components with BRG1 was found at chromatin on a genomewide scale 71, hinting that their association is not required for chromatin binding. When BRG1 was knocked out in ESCs, global H3K27me3 levels were not altered, but H3K27me3 displayed a selective elevation in BRG1‐activated genes and a decrease in BRG1‐repressed genes 76. This indicates that BRG1 directly regulates the level and distribution of H3K27me3 at its target genes.
The strong connection between SWI/SNF chromatin remodeling proteins and H3K27me3 in ESCs was recently further supported by our own studies 77. Screening for proteins that are differentially associated with chromatin between undifferentiated and differentiated ESCs, we identified the chromatin remodeling protein SMARCD1 (BAF60a), an additional component of the esBAF complex. ChIP‐seq maps of SMARCD1 in ESCs revealed a distribution not dissimilar from that of H3K27me3 around transcription start sites (TSS) and a significant enrichment in promoters of bivalent genes. Analyzing genomewide maps of H3K27me3 and H3K4me3 before and after SMARCD1 depletion revealed significant redistribution of these marks. In undifferentiated ESCs, both H3K4me3 and H3K27me3 were slightly elevated around TSSs upon SMARCD1 KD. In contrast, in differentiating ESCs, H3K4me3 was still elevated around TSSs, but H3K27me3 was dramatically reduced, by more than 75% around TSSs. Interestingly, bivalent genes were relatively protected from this wave of H3K27me3 elimination, indicating selective regulation of bivalent genes, by an unknown mechanism. Once again, no apparent changes in global levels were observed, as seen in the BRG1‐KO ESCs.
Regulation of bivalent histone modifications by the esBAF complex was further established by an inducible knockout system of the esBAF component BAF250a in mouse ESCs 78. Mapping of nucleosomes, bivalent histone modifications, and the PcG component SUZ12 before and after BAF250a depletion demonstrated that BAF250a mediates nucleosome occupancy and H3K27me3 levels at the upregulated, but not the downregulated genes in ESCs, and that it exerts its function likely by regulating esBAF and PRC2. BAF250a KO led to an increase in nucleosome positioning and a decrease in H3K27me3 levels, especially in bivalent and developmental gene promoters in ESCs. These alterations, accompanied by aberrant expression of developmental and pluripotency genes, resulted in differentiation defects in the BAF250a KO ESCs 78. This study supports a synergic role for esBAF and PRC2 in ESCs.
As indicated above, the catalytic subunit of the esBAF complex is BRG1. In human ESCs, BRG1 depletion resulted in elevated levels of the enhancer‐associated histone modification H3K27ac at BRG1 target gene enhancers 79. This suggests that in addition to its function in mediating H3K27 methylation levels at promoters, BRG1 may also act as a selective repressor at enhancer regions. Support for a dual role for BRG1 in regulating H3K27ac enhancer regions on one hand, and in maintaining PcG‐mediated promoter repression on the other, came from a recent study of in vitro mesodermal differentiation of mouse ESCs 80. BRG1 co‐localization with H3K27ac at distal enhancers is required to maintain their H3K27 acetylation during mesoderm induction, while it is also required to maintain PcG‐mediated repression of non‐mesodermal developmental regulators during differentiation. Taken together, these studies demonstrate a potential role for esBAF chromatin remodeling proteins in regulating bivalent histone modifications, primarily H3K27 methylation, in ESCs and during ESC differentiation, and provide evidence that the interplay between esBAF and PcG acts both to activate and to silence gene expression programs in ESCs.
Another family of chromatin remodeling proteins which was implicated in regulating H3K4/H3K27 methylation is the Chromodomain‐helicase DNA‐binding (CHD) family of proteins. CHD1 was found to regulate open chromatin and pluripotency in mouse ESCs by its association with H3K4me3 and counteracting heterochromatinization in pluripotent cells 81. However, it was later concluded that its association with H3K4me3 is specific to active genes and it is in fact exclusively depleted in the dual H3K4/H3K27 regions 82, suggesting a selective role as an activator, leaving the suppressed bivalent domains intact. Since CHD1 interacts with H3K4me3, it raises the possibility that a mechanism to selectively clear CHD1 from bivalent promoters exists. CHD7 on the other hand was shown, using clustering analysis, to be associated with three distinct protein complexes in ESCs: an enhancer signature cluster, a c‐MYC/n‐MYC‐enriched cluster, and a PcG cluster, containing SUZ12, RING1B, and EZH2 83, suggesting, among other things, a function for CHD7 in PcG‐mediated gene regulation. Supporting this notion, depletion of the CHD7 homolog Kismet in Drosophila resulted in global elevation of H3K27 methylation levels, demonstrating a role for CHD7/Kismet in counteracting PcG activity 84. CHD7 was also shown to associate with the PBAF chromatin remodeling complex during embryogenesis and human ESC differentiation to promote neural crest migration and neural crest gene expression programs 85. CHD7 mutations or other causes of failure to activate neural crest migration have been implicated in the development of CHARGE syndrome, a complex genetic disease affecting the nervous system, heart, vision, ears, and more 86. These studies highlight a link between CHD proteins and H3K27 methylation, thereby affecting bivalency albeit indirectly.
The CHD family of proteins also includes two prominent members of the NuRD chromatin remodeling complex, namely CHD3 and CHD4. The NuRD complex was shown to be essential for proper ESC differentiation 87 and was shown to deacetylate H3K27 in ESCs, enabling the subsequent recruitment of PcG proteins and trimethylation of H3K27. It therefore controls the balance between H3K27 acetylation and methylation, thereby enabling cell differentiation 88, although it likely does not act directly at bivalent promoters since it is repelled by H3K4me3 89. Further supporting the tight connection between NuRD and PcG during development is the association between CHD4 and the H3K27 methyltransferase EZH2 required during astroglial differentiation. CHD4 was found to be essential for EZH2 association with key astroglial gene promoters, suppressing their expression in non‐glial cells, and depletion of CHD4 promotes gliogenesis in vivo 90. A similar role for NuRD, mediated by CTBP2, was also seen in differentiating ESCs during the exit from pluripotency: NuRD facilitates H3K27 deacetylation followed by recruitment of PcG and H3K27 methylation 91. Finally, NuRD was also shown to play a role in regulating H3K4me3‐ and H3K27me3‐marked bivalent rRNA genes 92. Although the latter study was not performed in ESCs, given the importance of the NuRD complex and rRNA transcriptional regulation in pluripotency, it is fair to speculate that it likely plays a similarly important role there too. Consistent with NuRD's role in regulating bivalency, the complex was shown to interact not only with PcG proteins, as discussed above, but also with the H3K4 methyltransferase MLL1 93. But perhaps even more importantly, it was also shown to interact with the H3K4 demethylase LSD1, which occupies the large majority of active genes, as well as approximately two‐thirds of bivalent genes (2003 out of 3,094), in ESCs. Thus, through its association with the NuRD chromatin remodeling complex, LSD1 acts to silence pluripotency genes during early differentiation 94.
Taken together, these studies demonstrate that chromatin remodeling and bivalency, and most significantly PcG‐mediated H3K27 methylation, are oftentimes functionally linked in ESCs and during differentiation, acting to resolve bivalent domains into stably activated or stably repressed states (Table 1; Fig 4). Because this connection between chromatin remodeling and bivalency is only beginning to emerge, the examples provided above are sometimes sketchy or indirect, with only H3K4 or H3K27 being affected. However, as more convincing data are gradually accumulating, it is becoming increasingly clear that remodelers, among other chromatin factors, act to shape and regulate bivalent chromatin domains. For example, non‐canonical, replication‐independent histone variants including H2A.Z and H3.3 have been recently reported to be associated with bivalent chromatin 95, 96, 97, 98. In ESCs, H2A.Z is found in active and bivalent promoters, both of which are enriched with H3K4me3, but absent from repressed H3K4me3‐negative promoters 96. H2A.Z depletion caused derepression of poised genes with concomitant loss of PcG components from bivalent promoters 95, suggesting a potential co‐regulation of H2A.Z and PcG. However, a direct interaction between PcG and H2A.Z has not been reported so far. Along the same lines, H3.3 was shown to be required for the deposition of H3K27 methylation at bivalent promoters in a PRC2‐dependent manner 98. When H3.3 or its chaperone, HIRA, is depleted, H3K27me3 mark is reduced at bivalent promoters, along with reduced PRC2 occupancy and reduced nucleosome turnover 98, albeit with minimal transcriptional changes. This implies that other compensatory mechanisms regulating developmental genes in ESCs are in place, but together portrays a picture which suggests that histone variants and their remodelers might also be a part of the bivalency apparatus.
Table 1.
Chromatin remodelers involved in regulating bivalency
Figure 4. Chromatin remodeling complexes regulating bivalent nucleosomes.
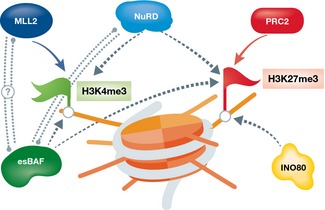
A single schematic bivalent nucleosome is shown (orange) marked with both H3K4me3 (green flag, left) and H3K27me3 (red flag, right). Chromatin remodeling complexes which were shown to regulate either or both marks are shown in green (esBAF), blue (NuRD), and mustard (INO80). Dotted arrows represent suggested regulation; dotted double lines represent potential interaction.
Concluding remarks
Almost a decade has passed since the original discovery of bivalent nucleosomes in ESCs 5, 6. While it was tempting to speculate that bivalent promoters are restricted to developmentally regulated genes, enabling a quick transition to an active or a stable silent state, it is clear today that bivalency is more complicated, extending to different gene families in multiple different cell types. Furthermore, it is increasingly recognized that regulation of the bivalent state is highly complex involving a variety of different proteins and regulators. Here, we highlighted the family of ATP‐dependent chromatin remodeling proteins, which is emerging as an important player in regulating bivalent domains, especially in the context of pluripotent stem cells. While TrxG and PcG proteins provide the mechanisms of action for H3K4/H3K27 methylation, we speculate that chromatin remodeling proteins may provide the required bivalent specificity. It is important to note that bivalency is not restricted to the H3K4me3/H3K27me3 pair; several, albeit more haphazard, examples were documented. For example, trophoblast and extraembryonic endodermal stem cells were shown to contain a large fraction of H3K4me3/H3K9me3 bivalent modifications but little H3K4me3/H3K27me3 bivalency 99, and a unique H3K4me1/H3K27ac/H3K9me3 trivalent signature was observed during the transition from fibroblasts to induced neurons 100. In both these cases, the “non‐canonical” bivalent/trivalent signature enables a quick transition from a closed to an open chromatin state akin to the situation proposed for the “canonical” K4/K27 marks. The next challenge would be to identify meaningful patterns and combinations of histone modifications, decipher their potential roles, and understand whether such chromatin signatures are the cause or the consequence of their suggested function. In addition, programmable nucleases (ZFN, TALEN, and especially CRISPR/Cas9, and mutants thereof) are already emerging as powerful tools that enable the catalysis or removal of specific modifications at bivalent loci, making it possible to study downstream effects on the corresponding genes 101, 102, 103, 104. More specialized systems such as photoactivatable CRISPR switches could take this idea further even to the single cell level 105, 106. The combination of epigenetic reprogramming assays, single cell technologies, and multilevel epigenomic landscape analyses will help decipher the specific roles that multivalent domains and their connection with chromatin remodeling play in pluripotency, ESCs, and development.
Conflict of interest
The authors declare that they have no conflict of interest.
Sidebar A: In need of answers.
Is bivalency a single‐cell phenomenon, or a result of population heterogeneity?
If bivalency is within a single cell, are bivalent histone marks present within the same allele?
If so, do bivalent histone marks appear within the same nucleosome?
If they reside within same nucleosome, are the bivalent marks distributed symmetrically or asymmetrically within the histone pair of the nucleosome?
To answer these questions unequivocally, bivalent histone marks must be monitored at a single‐nucleosome resolution. Single nucleosomes can be reconstituted in vitro or digested using micrococcal nuclease (MNase) and immobilized for visualization.
Does bivalency play a functional role in ESCs or in any other cell type?
What is the role of bivalent promoters in terminally differentiated cells?
Complete selective depletion of bivalent domains may be difficult to achieve, although a recent study 26 demonstrated little effect on early differentiation following depletion of MLL2 which conferred selective depletion of H3K4me3 on bivalent promoters. Additional studies of selective depletion of bivalent marks through the use of genetics or CRISPR/Cas9 approaches will determine the role, if any, that bivalent domains play in pluripotency and embryonic development.
Which of the histone variants are associated with bivalent domains?
Pull‐down of variant modified nucleosomes followed by MS approaches, or single nucleosome assays once single‐nucleosome resolution is achieved, will determine the exact composition/modifications of histone variant‐containing nucleosomes.
How do chromatin remodeling proteins regulate bivalent histone marks?
Do pluripotency factors, in concert with chromatin remodeling complexes, regulate bivalent domains?
Interaction analyses, mutational studies, rescue experiments, and functional assays will help achieve mechanistic insights into the regulation of bivalent domains by chromatin remodeling proteins and/or pluripotency factors. Understanding the mechanism by which chromatin remodelers regulate the level and distribution of bivalent chromatin domains will be key to establish a direct functional connection between chromatin remodeling proteins and bivalency.
Acknowledgements
We thank Bradley Bernstein, Cem Sievers, and Sharon Schlesinger for critical comments, and Alva Biran for help with statistics. AH was supported by the Nucleosome4D network. We thank the Israel Science Foundation (ISF 1252/12 and 657/12 to E.M.) and the European Research Council (ERC‐281781 to E.M.) for financial support.
EMBO Reports (2015) 16: 1609–1619
See the Glossary for abbreviations used in this article.
References
- 1. Kouzarides T (2007) Chromatin modifications and their function. Cell 128: 693–705 [DOI] [PubMed] [Google Scholar]
- 2. Bannister AJ, Kouzarides T (2011) Regulation of chromatin by histone modifications. Cell Res 21: 381–395 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Tessarz P, Kouzarides T (2014) Histone core modifications regulating nucleosome structure and dynamics. Nat Rev Mol Cell Biol 15: 703–708 [DOI] [PubMed] [Google Scholar]
- 4. Azzaz AM, Vitalini MW, Thomas AS, Price JP, Blacketer MJ, Cryderman DE, Zirbel LN, Woodcock CL, Elcock AH, Wallrath LL et al (2014) Human heterochromatin protein 1alpha promotes nucleosome associations that drive chromatin condensation. J Biol Chem 289: 6850–6861 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Azuara V, Perry P, Sauer S, Spivakov M, Jorgensen HF, John RM, Gouti M, Casanova M, Warnes G, Merkenschlager M et al (2006) Chromatin signatures of pluripotent cell lines. Nat Cell Biol 8: 532–538 [DOI] [PubMed] [Google Scholar]
- 6. Bernstein BE, Mikkelsen TS, Xie X, Kamal M, Huebert DJ, Cuff J, Fry B, Meissner A, Wernig M, Plath K et al (2006) A bivalent chromatin structure marks key developmental genes in embryonic stem cells. Cell 125: 315–326 [DOI] [PubMed] [Google Scholar]
- 7. Voigt P, Tee WW, Reinberg D (2013) A double take on bivalent promoters. Genes Dev 27: 1318–1338 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Vastenhouw NL, Schier AF (2012) Bivalent histone modifications in early embryogenesis. Curr Opin Cell Biol 24: 374–386 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Dillon N (2012) Factor mediated gene priming in pluripotent stem cells sets the stage for lineage specification. BioEssays 34: 194–204 [DOI] [PubMed] [Google Scholar]
- 10. Fisher CL, Fisher AG (2011) Chromatin states in pluripotent, differentiated, and reprogrammed cells. Curr Opin Genet Dev 21: 140–146 [DOI] [PubMed] [Google Scholar]
- 11. Pan G, Tian S, Nie J, Yang C, Ruotti V, Wei H, Jonsdottir GA, Stewart R, Thomson JA (2007) Whole‐genome analysis of histone H3 lysine 4 and lysine 27 methylation in human embryonic stem cells. Cell Stem Cell 1: 299–312 [DOI] [PubMed] [Google Scholar]
- 12. Zhao XD, Han X, Chew JL, Liu J, Chiu KP, Choo A, Orlov YL, Sung WK, Shahab A, Kuznetsov VA et al (2007) Whole‐genome mapping of histone H3 Lys4 and 27 trimethylations reveals distinct genomic compartments in human embryonic stem cells. Cell Stem Cell 1: 286–298 [DOI] [PubMed] [Google Scholar]
- 13. Maherali N, Sridharan R, Xie W, Utikal J, Eminli S, Arnold K, Stadtfeld M, Yachechko R, Tchieu J, Jaenisch R et al (2007) Directly reprogrammed fibroblasts show global epigenetic remodeling and widespread tissue contribution. Cell Stem Cell 1: 55–70 [DOI] [PubMed] [Google Scholar]
- 14. Mikkelsen TS, Ku M, Jaffe DB, Issac B, Lieberman E, Giannoukos G, Alvarez P, Brockman W, Kim TK, Koche RP et al (2007) Genome‐wide maps of chromatin state in pluripotent and lineage‐committed cells. Nature 448: 553–560 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Guenther MG, Frampton GM, Soldner F, Hockemeyer D, Mitalipova M, Jaenisch R, Young RA (2010) Chromatin structure and gene expression programs of human embryonic and induced pluripotent stem cells. Cell Stem Cell 7: 249–257 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Cui K, Zang C, Roh TY, Schones DE, Childs RW, Peng W, Zhao K (2009) Chromatin signatures in multipotent human hematopoietic stem cells indicate the fate of bivalent genes during differentiation. Cell Stem Cell 4: 80–93 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Abraham BJ, Cui K, Tang Q, Zhao K (2013) Dynamic regulation of epigenomic landscapes during hematopoiesis. BMC Genom 14: 193 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Mohn F, Weber M, Rebhan M, Roloff TC, Richter J, Stadler MB, Bibel M, Schubeler D (2008) Lineage‐specific polycomb targets and de novo DNA methylation define restriction and potential of neuronal progenitors. Mol Cell 30: 755–766 [DOI] [PubMed] [Google Scholar]
- 19. Roh TY, Cuddapah S, Cui K, Zhao K (2006) The genomic landscape of histone modifications in human T cells. Proc Natl Acad Sci USA 103: 15782–15787 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Barski A, Cuddapah S, Cui K, Roh TY, Schones DE, Wang Z, Wei G, Chepelev I, Zhao K (2007) High‐resolution profiling of histone methylations in the human genome. Cell 129: 823–837 [DOI] [PubMed] [Google Scholar]
- 21. Rodriguez J, Munoz M, Vives L, Frangou CG, Groudine M, Peinado MA (2008) Bivalent domains enforce transcriptional memory of DNA methylated genes in cancer cells. Proc Natl Acad Sci USA 105: 19809–19814 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Bapat SA, Jin V, Berry N, Balch C, Sharma N, Kurrey N, Zhang S, Fang F, Lan X, Li M et al (2010) Multivalent epigenetic marks confer microenvironment‐responsive epigenetic plasticity to ovarian cancer cells. Epigenetics 5: 716–729 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. McGarvey KM, Van Neste L, Cope L, Ohm JE, Herman JG, Van Criekinge W, Schuebel KE, Baylin SB (2008) Defining a chromatin pattern that characterizes DNA‐hypermethylated genes in colon cancer cells. Cancer Res 68: 5753–5759 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Lin B, Lee H, Yoon JG, Madan A, Wayner E, Tonning S, Hothi P, Schroeder B, Ulasov I, Foltz G et al (2015) Global analysis of H3K4me3 and H3K27me3 profiles in glioblastoma stem cells and identification of SLC17A7 as a bivalent tumor suppressor gene. Oncotarget 6: 5369–5381 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Denissov S, Hofemeister H, Marks H, Kranz A, Ciotta G, Singh S, Anastassiadis K, Stunnenberg HG, Stewart AF (2014) Mll2 is required for H3K4 trimethylation on bivalent promoters in embryonic stem cells, whereas Mll1 is redundant. Development 141: 526–537 [DOI] [PubMed] [Google Scholar]
- 26. Hu D, Garruss AS, Gao X, Morgan MA, Cook M, Smith ER, Shilatifard A (2013) The Mll2 branch of the COMPASS family regulates bivalent promoters in mouse embryonic stem cells. Nat Struct Mol Biol 20: 1093–1097 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Voigt P, LeRoy G, Drury WJ III, Zee BM, Son J, Beck DB, Young NL, Garcia BA, Reinberg D (2012) Asymmetrically modified nucleosomes. Cell 151: 181–193 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Shilatifard A (2012) The COMPASS family of histone H3K4 methylases: mechanisms of regulation in development and disease pathogenesis. Annu Rev Biochem 81: 65–95 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Birke M, Schreiner S, Garcia‐Cuellar MP, Mahr K, Titgemeyer F, Slany RK (2002) The MT domain of the proto‐oncoprotein MLL binds to CpG‐containing DNA and discriminates against methylation. Nucleic Acids Res 30: 958–965 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Bach C, Mueller D, Buhl S, Garcia‐Cuellar MP, Slany RK (2009) Alterations of the CxxC domain preclude oncogenic activation of mixed‐lineage leukemia 2. Oncogene 28: 815–823 [DOI] [PubMed] [Google Scholar]
- 31. Lee JH, Voo KS, Skalnik DG (2001) Identification and characterization of the DNA binding domain of CpG‐binding protein. J Biol Chem 276: 44669–44676 [DOI] [PubMed] [Google Scholar]
- 32. Clouaire T, Webb S, Skene P, Illingworth R, Kerr A, Andrews R, Lee JH, Skalnik D, Bird A (2012) Cfp1 integrates both CpG content and gene activity for accurate H3K4me3 deposition in embryonic stem cells. Genes Dev 26: 1714–1728 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Wysocka J, Swigut T, Milne TA, Dou Y, Zhang X, Burlingame AL, Roeder RG, Brivanlou AH, Allis CD (2005) WDR5 associates with histone H3 methylated at K4 and is essential for H3K4 methylation and vertebrate development. Cell 121: 859–872 [DOI] [PubMed] [Google Scholar]
- 34. Ang YS, Tsai SY, Lee DF, Monk J, Su J, Ratnakumar K, Ding J, Ge Y, Darr H, Chang B et al (2011) Wdr5 mediates self‐renewal and reprogramming via the embryonic stem cell core transcriptional network. Cell 145: 183–197 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Stoller JZ, Huang L, Tan CC, Huang F, Zhou DD, Yang J, Gelb BD, Epstein JA (2010) Ash2l interacts with Tbx1 and is required during early embryogenesis. Exp Biol Med 235: 569–576 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Wan M, Liang J, Xiong Y, Shi F, Zhang Y, Lu W, He Q, Yang D, Chen R, Liu D et al (2012) The trithorax group protein Ash2l is essential for pluripotency and maintaining open chromatin in embryonic stem cells. J Biol Chem 288: 5039–5048 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Biondi CA, Gartside MG, Waring P, Loffler KA, Stark MS, Magnuson MA, Kay GF, Hayward NK (2004) Conditional inactivation of the MEN1 gene leads to pancreatic and pituitary tumorigenesis but does not affect normal development of these tissues. Mol Cell Biol 24: 3125–3131 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Jiang H, Shukla A, Wang X, Chen WY, Bernstein BE, Roeder RG (2011) Role for Dpy‐30 in ES cell‐fate specification by regulation of H3K4 methylation within bivalent domains. Cell 144: 513–525 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Lubitz S, Glaser S, Schaft J, Stewart AF, Anastassiadis K (2007) Increased apoptosis and skewed differentiation in mouse embryonic stem cells lacking the histone methyltransferase Mll2. Mol Biol Cell 18: 2356–2366 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Glaser S, Schaft J, Lubitz S, Vintersten K, van der Hoeven F, Tufteland KR, Aasland R, Anastassiadis K, Ang SL, Stewart AF (2006) Multiple epigenetic maintenance factors implicated by the loss of Mll2 in mouse development. Development 133: 1423–1432 [DOI] [PubMed] [Google Scholar]
- 41. Cao R, Wang L, Wang H, Xia L, Erdjument‐Bromage H, Tempst P, Jones RS, Zhang Y (2002) Role of histone H3 lysine 27 methylation in Polycomb‐group silencing. Science 298: 1039–1043 [DOI] [PubMed] [Google Scholar]
- 42. Margueron R, Justin N, Ohno K, Sharpe ML, Son J, Drury WJ III, Voigt P, Martin SR, Taylor WR, De Marco V et al (2009) Role of the polycomb protein EED in the propagation of repressive histone marks. Nature 461: 762–767 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Morey L, Pascual G, Cozzuto L, Roma G, Wutz A, Benitah SA, Di Croce L (2012) Nonoverlapping functions of the Polycomb group Cbx family of proteins in embryonic stem cells. Cell Stem Cell 10: 47–62 [DOI] [PubMed] [Google Scholar]
- 44. O'Loghlen A, Munoz‐Cabello AM, Gaspar‐Maia A, Wu HA, Banito A, Kunowska N, Racek T, Pemberton HN, Beolchi P, Lavial F et al (2012) MicroRNA regulation of Cbx7 mediates a switch of Polycomb orthologs during ESC differentiation. Cell Stem Cell 10: 33–46 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Morey L, Aloia L, Cozzuto L, Benitah SA, Di Croce L (2013) RYBP and Cbx7 define specific biological functions of polycomb complexes in mouse embryonic stem cells. Cell Rep 3: 60–69 [DOI] [PubMed] [Google Scholar]
- 46. Wang H, Wang L, Erdjument‐Bromage H, Vidal M, Tempst P, Jones RS, Zhang Y (2004) Role of histone H2A ubiquitination in Polycomb silencing. Nature 431: 873–878 [DOI] [PubMed] [Google Scholar]
- 47. Cao R, Zhang Y (2004) The functions of E(Z)/EZH2‐mediated methylation of lysine 27 in histone H3. Curr Opin Genet Dev 14: 155–164 [DOI] [PubMed] [Google Scholar]
- 48. Brookes E, de Santiago I, Hebenstreit D, Morris KJ, Carroll T, Xie SQ, Stock JK, Heidemann M, Eick D, Nozaki N et al (2012) Polycomb associates genome‐wide with a specific RNA polymerase II variant, and regulates metabolic genes in ESCs. Cell Stem Cell 10: 157–170 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Min IM, Waterfall JJ, Core LJ, Munroe RJ, Schimenti J, Lis JT (2011) Regulating RNA polymerase pausing and transcription elongation in embryonic stem cells. Genes Dev 25: 742–754 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Cooper S, Dienstbier M, Hassan R, Schermelleh L, Sharif J, Blackledge NP, De Marco V, Elderkin S, Koseki H, Klose R et al (2014) Targeting polycomb to pericentric heterochromatin in embryonic stem cells reveals a role for H2AK119u1 in PRC2 recruitment. Cell Rep 7: 1456–1470 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Ku M, Koche RP, Rheinbay E, Mendenhall EM, Endoh M, Mikkelsen TS, Presser A, Nusbaum C, Xie X, Chi AS et al (2008) Genomewide analysis of PRC1 and PRC2 occupancy identifies two classes of bivalent domains. PLoS Genet 4: e1000242 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Lee TI, Jenner RG, Boyer LA, Guenther MG, Levine SS, Kumar RM, Chevalier B, Johnstone SE, Cole MF, Isono K et al (2006) Control of developmental regulators by Polycomb in human embryonic stem cells. Cell 125: 301–313 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Thomas LR, Wang Q, Grieb BC, Phan J, Foshage AM, Sun Q, Olejniczak ET, Clark T, Dey S, Lorey S et al (2015) Interaction with WDR5 promotes target gene recognition and tumorigenesis by MYC. Mol Cell 58: 440–452 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Ding J, Xu H, Faiola F, Ma'ayan A, Wang J (2011) Oct4 links multiple epigenetic pathways to the pluripotency network. Cell Res 22: 155–167 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Henikoff S, Shilatifard A (2011) Histone modification: cause or cog? Trends Genet 27: 389–396 [DOI] [PubMed] [Google Scholar]
- 56. Pasini D, Bracken AP, Hansen JB, Capillo M, Helin K (2007) The polycomb group protein Suz12 is required for embryonic stem cell differentiation. Mol Cell Biol 27: 3769–3779 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Chamberlain SJ, Yee D, Magnuson T (2008) Polycomb repressive complex 2 is dispensable for maintenance of embryonic stem cell pluripotency. Stem Cells 26: 1496–1505 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Shen X, Liu Y, Hsu YJ, Fujiwara Y, Kim J, Mao X, Yuan GC, Orkin SH (2008) EZH1 mediates methylation on histone H3 lysine 27 and complements EZH2 in maintaining stem cell identity and executing pluripotency. Mol Cell 32: 491–502 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Leeb M, Pasini D, Novatchkova M, Jaritz M, Helin K, Wutz A (2010) Polycomb complexes act redundantly to repress genomic repeats and genes. Genes Dev 24: 265–276 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Faust C, Schumacher A, Holdener B, Magnuson T (1995) The eed mutation disrupts anterior mesoderm production in mice. Development 121: 273–285 [DOI] [PubMed] [Google Scholar]
- 61. O'Carroll D, Erhardt S, Pagani M, Barton SC, Surani MA, Jenuwein T (2001) The polycomb‐group gene Ezh2 is required for early mouse development. Mol Cell Biol 21: 4330–4336 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Pasini D, Bracken AP, Jensen MR, Lazzerini Denchi E, Helin K (2004) Suz12 is essential for mouse development and for EZH2 histone methyltransferase activity. EMBO J 23: 4061–4071 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Leeb M, Wutz A (2007) Ring1B is crucial for the regulation of developmental control genes and PRC1 proteins but not X inactivation in embryonic cells. J Cell Biol 178: 219–229 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Alkema MJ, van der Lugt NM, Bobeldijk RC, Berns A, van Lohuizen M (1995) Transformation of axial skeleton due to overexpression of bmi‐1 in transgenic mice. Nature 374: 724–727 [DOI] [PubMed] [Google Scholar]
- 65. van der Lugt NM, Domen J, Linders K, van Roon M, Robanus‐Maandag E, te Riele H, van der Valk M, Deschamps J, Sofroniew M, van Lohuizen M et al (1994) Posterior transformation, neurological abnormalities, and severe hematopoietic defects in mice with a targeted deletion of the bmi‐1 proto‐oncogene. Genes Dev 8: 757–769 [DOI] [PubMed] [Google Scholar]
- 66. Aloia L, Di Stefano B, Di Croce L (2013) Polycomb complexes in stem cells and embryonic development. Development 140: 2525–2534 [DOI] [PubMed] [Google Scholar]
- 67. Efroni S, Duttagupta R, Cheng J, Dehghani H, Hoeppner DJ, Dash C, Bazett‐Jones DP, Le Grice S, McKay RD, Buetow KH et al (2008) Global transcription in pluripotent embryonic stem cells. Cell Stem Cell 2: 437–447 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Clapier CR, Cairns BR (2009) The biology of chromatin remodeling complexes. Annu Rev Biochem 78: 273–304 [DOI] [PubMed] [Google Scholar]
- 69. Gaspar‐Maia A, Alajem A, Meshorer E, Ramalho‐Santos M (2010) Open chromatin in pluripotency and reprogramming. Nat Rev Mol Cell Biol 12: 36–47 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Fazzio TG, Huff JT, Panning B (2008) An RNAi screen of chromatin proteins identifies Tip60‐p400 as a regulator of embryonic stem cell identity. Cell 134: 162–174 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Ho L, Jothi R, Ronan JL, Cui K, Zhao K, Crabtree GR (2009) An embryonic stem cell chromatin remodeling complex, esBAF, is an essential component of the core pluripotency transcriptional network. Proc Natl Acad Sci USA 106: 5187–5191 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Kidder BL, Palmer S, Knott JG (2009) SWI/SNF‐Brg1 regulates self‐renewal and occupies core pluripotency‐related genes in embryonic stem cells. Stem Cells 27: 317–328 [DOI] [PubMed] [Google Scholar]
- 73. Ho L, Ronan JL, Wu J, Staahl BT, Chen L, Kuo A, Lessard J, Nesvizhskii AI, Ranish J, Crabtree GR (2009) An embryonic stem cell chromatin remodeling complex, esBAF, is essential for embryonic stem cell self‐renewal and pluripotency. Proc Natl Acad Sci USA 106: 5181–5186 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Shen X, Kim W, Fujiwara Y, Simon MD, Liu Y, Mysliwiec MR, Yuan GC, Lee Y, Orkin SH (2009) Jumonji modulates polycomb activity and self‐renewal versus differentiation of stem cells. Cell 139: 1303–1314 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Landeira D, Sauer S, Poot R, Dvorkina M, Mazzarella L, Jorgensen HF, Pereira CF, Leleu M, Piccolo FM, Spivakov M et al (2010) Jarid2 is a PRC2 component in embryonic stem cells required for multi‐lineage differentiation and recruitment of PRC1 and RNA Polymerase II to developmental regulators. Nat Cell Biol 12: 618–624 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Ho L, Miller EL, Ronan JL, Ho WQ, Jothi R, Crabtree GR (2011) esBAF facilitates pluripotency by conditioning the genome for LIF/STAT3 signalling and by regulating polycomb function. Nat Cell Biol 13: 903–913 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Alajem A, Biran A, Harikumar A, Sailaja BS, Aaronson Y, Livyatan I, Nissim‐Rafinia M, Sommer AG, Mostoslavsky G, Gerbasi VR et al (2015) Differential association of chromatin proteins identifies BAF60a/SMARCD1 as a regulator of embryonic stem cell differentiation. Cell Rep 10: 2019–2031 [DOI] [PubMed] [Google Scholar]
- 78. Lei I, West J, Yan Z, Gao X, Fang P, Dennis JH, Gnatovskiy L, Wang W, Kingston RE, Wang Z (2015) BAF250a regulates nucleosome occupancy and histone modifications in priming embryonic stem cell differentiation. J Biol Chem 290: 19343–19352 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Zhang X, Li B, Li W, Ma L, Zheng D, Li L, Yang W, Chu M, Chen W, Mailman RB et al (2014) Transcriptional repression by the BRG1‐SWI/SNF complex affects the pluripotency of human embryonic stem cells. Stem Cell Rep 3: 460–474 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Alexander JM, Hota SK, He D, Thomas S, Ho L, Pennacchio LA, Bruneau BG (2015) Brg1 modulates enhancer activation in mesoderm lineage commitment. Development 142: 1418–1430 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81. Gaspar‐Maia A, Alajem A, Polesso F, Sridharan R, Mason MJ, Heidersbach A, Ramalho‐Santos J, McManus MT, Plath K, Meshorer E et al (2009) Chd1 regulates open chromatin and pluripotency of embryonic stem cells. Nature 460: 863–868 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82. Lin JJ, Lehmann LW, Bonora G, Sridharan R, Vashisht AA, Tran N, Plath K, Wohlschlegel JA, Carey M (2011) Mediator coordinates PIC assembly with recruitment of CHD1. Genes Dev 25: 2198–2209 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. Schnetz MP, Handoko L, Akhtar‐Zaidi B, Bartels CF, Pereira CF, Fisher AG, Adams DJ, Flicek P, Crawford GE, Laframboise T et al (2010) CHD7 targets active gene enhancer elements to modulate ES cell‐specific gene expression. PLoS Genet 6: e1001023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. Srinivasan S, Dorighi KM, Tamkun JW (2008) Drosophila Kismet regulates histone H3 lysine 27 methylation and early elongation by RNA polymerase II. PLoS Genet 4: e1000217 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. Bajpai R, Chen DA, Rada‐Iglesias A, Zhang J, Xiong Y, Helms J, Chang CP, Zhao Y, Swigut T, Wysocka J (2010) CHD7 cooperates with PBAF to control multipotent neural crest formation. Nature 463: 958–962 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Hsu P, Ma A, Wilson M, Williams G, Curotta J, Munns CF, Mehr S (2014) CHARGE syndrome: a review. J Paediatr Child Health 50: 504–511 [DOI] [PubMed] [Google Scholar]
- 87. Kaji K, Caballero IM, MacLeod R, Nichols J, Wilson VA, Hendrich B (2006) The NuRD component Mbd3 is required for pluripotency of embryonic stem cells. Nat Cell Biol 8: 285–292 [DOI] [PubMed] [Google Scholar]
- 88. Reynolds N, Salmon‐Divon M, Dvinge H, Hynes‐Allen A, Balasooriya G, Leaford D, Behrens A, Bertone P, Hendrich B (2011) NuRD‐mediated deacetylation of H3K27 facilitates recruitment of Polycomb Repressive Complex 2 to direct gene repression. EMBO J 31: 593–605 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. Zegerman P, Canas B, Pappin D, Kouzarides T (2002) Histone H3 lysine 4 methylation disrupts binding of nucleosome remodeling and deacetylase (NuRD) repressor complex. J Biol Chem 277: 11621–11624 [DOI] [PubMed] [Google Scholar]
- 90. Sparmann A, Xie Y, Verhoeven E, Vermeulen M, Lancini C, Gargiulo G, Hulsman D, Mann M, Knoblich JA, van Lohuizen M (2013) The chromodomain helicase Chd4 is required for Polycomb‐mediated inhibition of astroglial differentiation. EMBO J 32: 1598–1612 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Kim TW, Kang BH, Jang H, Kwak S, Shin J, Kim H, Lee SE, Lee SM, Lee JH, Kim JH et al (2015) Ctbp2 modulates NuRD‐mediated deacetylation of H3K27 and facilitates PRC2‐mediated H3K27me3 in active embryonic stem cell genes during exit from pluripotency. Stem Cells 33: 2442–2455 [DOI] [PubMed] [Google Scholar]
- 92. Xie W, Ling T, Zhou Y, Feng W, Zhu Q, Stunnenberg HG, Grummt I, Tao W (2012) The chromatin remodeling complex NuRD establishes the poised state of rRNA genes characterized by bivalent histone modifications and altered nucleosome positions. Proc Natl Acad Sci USA 109: 8161–8166 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93. Nakamura T, Mori T, Tada S, Krajewski W, Rozovskaia T, Wassell R, Dubois G, Mazo A, Croce CM, Canaani E (2002) ALL‐1 is a histone methyltransferase that assembles a supercomplex of proteins involved in transcriptional regulation. Mol Cell 10: 1119–1128 [DOI] [PubMed] [Google Scholar]
- 94. Whyte WA, Bilodeau S, Orlando DA, Hoke HA, Frampton GM, Foster CT, Cowley SM, Young RA (2012) Enhancer decommissioning by LSD1 during embryonic stem cell differentiation. Nature 482: 221–225 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Creyghton MP, Markoulaki S, Levine SS, Hanna J, Lodato MA, Sha K, Young RA, Jaenisch R, Boyer LA (2008) H2AZ is enriched at polycomb complex target genes in ES cells and is necessary for lineage commitment. Cell 135: 649–661 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. Ku M, Jaffe JD, Koche RP, Rheinbay E, Endoh M, Koseki H, Carr SA, Bernstein BE (2012) H2A.Z landscapes and dual modifications in pluripotent and multipotent stem cells underlie complex genome regulatory functions. Genome Biol 13: R85 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97. Hu G, Cui K, Northrup D, Liu C, Wang C, Tang Q, Ge K, Levens D, Crane‐Robinson C, Zhao K (2013) H2A.Z facilitates access of active and repressive complexes to chromatin in embryonic stem cell self‐renewal and differentiation. Cell Stem Cell 12: 180–192 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Banaszynski LA, Wen D, Dewell S, Whitcomb SJ, Lin M, Diaz N, Elsasser SJ, Chapgier A, Goldberg AD, Canaani E et al (2013) Hira‐dependent histone H3.3 deposition facilitates PRC2 recruitment at developmental loci in ES cells. Cell 155: 107–120 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99. Rugg‐Gunn PJ, Cox BJ, Ralston A, Rossant J (2010) Distinct histone modifications in stem cell lines and tissue lineages from the early mouse embryo. Proc Natl Acad Sci USA 107: 10783–10790 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100. Wapinski OL, Vierbuchen T, Qu K, Lee QY, Chanda S, Fuentes DR, Giresi PG, Ng YH, Marro S, Neff NF et al (2013) Hierarchical mechanisms for direct reprogramming of fibroblasts to neurons. Cell 155: 621–635 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101. Kleinstiver BP, Prew MS, Tsai SQ, Topkar VV, Nguyen NT, Zheng Z, Gonzales AP, Li Z, Peterson RT, Yeh JJ et al (2015) Engineered CRISPR‐Cas9 nucleases with altered PAM specificities. Nature 523: 481–485 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102. Qi LS, Larson MH, Gilbert LA, Doudna JA, Weissman JS, Arkin AP, Lim WA (2013) Repurposing CRISPR as an RNA‐guided platform for sequence‐specific control of gene expression. Cell 152: 1173–1183 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103. Cheng AW, Wang H, Yang H, Shi L, Katz Y, Theunissen TW, Rangarajan S, Shivalila CS, Dadon DB, Jaenisch R (2013) Multiplexed activation of endogenous genes by CRISPR‐on, an RNA‐guided transcriptional activator system. Cell Res 23: 1163–1171 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104. Hu J, Lei Y, Wong WK, Liu S, Lee KC, He X, You W, Zhou R, Guo JT, Chen X et al (2014) Direct activation of human and mouse Oct4 genes using engineered TALE and Cas9 transcription factors. Nucleic Acids Res 42: 4375–4390 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105. Nihongaki Y, Kawano F, Nakajima T, Sato M (2015) Photoactivatable CRISPR‐Cas9 for optogenetic genome editing. Nat Biotechnol 33: 755–760 [DOI] [PubMed] [Google Scholar]
- 106. Nihongaki Y, Yamamoto S, Kawano F, Suzuki H, Sato M (2015) CRISPR‐Cas9‐based photoactivatable transcription system. Chem Biol 22: 169–174 [DOI] [PubMed] [Google Scholar]


