Abstract
The purpose of the present study was to perform a meta-analysis to evaluate the diagnostic value of Multidetector computed tomography (MDCT) in the pre-operative lymph node (N) staging in gastric cancer (GC) patients. The Medline, Embase and Web of Knowledge were searched for studies assessing the diagnostic value of MDCT in the pre-operative evaluation of TNM staging in GC patients. We pooled the sensitivity, specificity, positive and negative Likelihood ratio (LR+ and LR-), Diagnostic Odds Ratio (DOR) and constructed summary receiver operating characteristic curves (ROC). A total of 30 studies including 6637 GC patients were analyzed. The pooled estimates of sensitivity, specificity, LR+, LR- and DOR of MDCT in the detection of pre-operative N staging in GC patients were 0.67 (95% CI: 0.66-0.69 ), 0.84 (95% CI: 0.83-0.85), 3.25 (95% CI: 2.69-3.93), 0.36 (95% CI: 0.28-0.46) and 10.31 (95% CI: 7.66-13.88), respectively. The results of a summary ROC showed that the AUC and Q* were 0.8338 and 0.7661, respectively. As a control, the AUC and Q* of endoscopic ultrasonography were 0.8063 and 0.7414, respectively. Currently, it is necessary to recommend the routine clinical application of MDCT in the preoperative evaluation of lymph node status in GC patients.
Keywords: Multi-detector computed tomography, MDCT, lymph node staging, gastric cancer, meta-analysis
Introduction
Gastric cancer (GC) is one of the most common malignant tumors in the digestive system and the second most common cause of cancer-related death worldwide [1]. Although the incidence of gastric cancer has been declining in most industrial countries, it remains the most prevalent cancer in East Asian countries [2]. The proportion of early gastric cancer in Korea and Japan has increased owing to improvement in diagnostic method and population screening. However, most of patients with gastric cancer in China and other countries were advanced. The primary treatment of gastric cancer is still surgical resection [3]. However, novel therapeutic approaches have been utilized recently, including endoscopic mucosal resection (EMR), endoscopic submucosal dissection (ESD) or laparoscopic treatment in patients with early GC [4,5] and neoadjuvant chemotherapy in advanced GC [6]. Above new therapeutic approaches were based on accurate pre-surgical TNM staging, especially lymph node (N) staging.
Currently, following the improvement of imaging technique, multi-detector computed tomography (MDCT) has become one of the most common techniques for the pre-surgical TNM staging in GC patients. However, an accurate count of lymph nodes is a tremendous challenge to the radiologists. There has been no universe consensus regarding lymph nodes pathology about measuring method. Criteria for lymph node involvement have been controversial. Recently, most studies considered that the regional lymph nodes were metastases if they were larger than 8 mm in the short-axis diameter [7]. Some researches showed that the accuracy of MDCT concerning pre-operative staging in gastric cancer was similar with endoscopic ultrasonography (EUS). As for MDCT, the accuracy of T staging and N staging were 77.1 to 88.9% and 51 to 71%, respectively while the accuracy of T and N staging concerning EUS were 65 to 92.1% and 63 to 78%, respectively [8,9]. Obviously, the results of pre-operative N staging of MDCT have shown large variation [8-10]. It is difficult to draw a definitive conclusion about the utility of this technique. Therefore, it is urgently necessary to confirm the per-operative diagnostic value of MDCT in order to establish the therapeutic strategy of gastric cancer.
To acknowledge the diagnostic value of pre-operative N staging in gastric cancer, we perform the meta-analysis and systematic review by retrieving relevant literature. Meanwhile, the data of EUS involving in pre-surgical N staging was also analyzed for comparison.
Materials and methods
Literature search
A comprehensive computerized systematic literature search was carried out to retrieve abstracts of publications from studies which assessed MDCT as a diagnostic tool for initial staging before surgery or any treatment in patients with gastric cancer. We retrieved relevant articles with PubMed/Medline, ISI Web of Knowledge and Embase databases (Last updated on 23 Aug 2014). We utilized a search algorithm that was based on a combination of the following text words: (a) Multidetector Computed Tomography or MDCT, (b) gastric cancer or gastric carcinoma or gastric neoplasm or stomach cancer or stomach carcinoma or stomach neoplasm, (c) staging. The searches were restricted to studies done in humans. Two investigators, who were blinded to the author, journal, date of publication and institution, independently retrieved all the articles. Potentially related documents were assessed by reviewing their titles and abstracts and all the studies meeting the eligible criteria were retrieved. For studies utilizing the same samples in different articles, only the most complete information was selected. Information of patients was collected to obtain clinical data with approval of our hospital’s ethics committee (Table 1).
Table 1.
Clinical characteristics of included studies
| Reference and Study ID | Year | Number of patients | Design | Gender (% male) | Mean age (Year) | Gold Standard | Equipment | Blind | Total QUADAS Score |
|---|---|---|---|---|---|---|---|---|---|
| Fujikawa H et al. [17] | 2014 | 525 | P | 69.3 | 63.0 | PSP | MDCT | Yes | 13 |
| Yoshikawa T et al. [18] | 2014 | 75 | P | 70.7 | 66.0 | PSP | MDCT | NR | 10 |
| Hasegawa S et al. [19] | 2013 | 315 | P | NR | NR | PSP | MDCT | NR | 9 |
| Kim SH et al. [7] | 2013 | 171 | R | 61.4 | 62.9 | PSP | MDCT | Yes | 13 |
| Feng XY et al. [20] | 2013 | 610 | P | 79.1 | 57.0 | PSP | MDCT; EUS | Yes | 12 |
| Zilai P et al. [21] | 2013 | 96 | P | 61.5 | 57.0 | PSP | MDCT | Yes | 12 |
| Zhong BY et al. [22] | 2012 | 115 | R | NR | NR | PSP | MDCT | NR | 9 |
| Marrelli D et al. [23] | 2011 | 92 | P | 58.7 | 66.0 | PSP | MDCT | Yes | 13 |
| Ha TK et al. [24] | 2011 | 78 | R | 67.9 | 61.0 | PSP | PET-CT; MDCT | NR | 10 |
| Kim EY et al. [25] | 2011 | 78 | R | 68.0 | 58.0 | PSP | PET-CT; MDCT | Yes | 12 |
| Yan C et al. [26] | 2010 | 61 | P | 65.6 | 59.0 | PSP | MDCT | Yes | 12 |
| Yan C et al. [26] | 2010 | 305 | R | 60.7 | 59.0 | PSP | MDCT | Yes | 13 |
| Lee IJ et al. [27] | 2010 | 148 | R | 63.5 | 59.9 | PSP | MDCT | Yes | 13 |
| Venkataraman et al. [28] | 2010 | 42 | NR | 69.0 | 51.0 | PSP | MDCT; HGS | NR | 10 |
| Park SR et al. [29] | 2010 | 1964 | P | 67.6 | 59.0 | PSP | MDCT | Yes | 13 |
| Hwang SW et al. [30] | 2010 | 247 | P | 61.7 | 53.0 | PSP | MDCT; EUS | NR | 10 |
| Ahn HS et al. [31] | 2009 | 434 | NR | 64.0 | 55.9 | PSP | MDCT; EUS | Yes | 12 |
| Yan C et al. [32] | 2009 | 135 | NR | 59.1 | 59.3 | PSP | MDCT | Yes | 12 |
| Yang QM et al. [33] | 2008 | 78 | NR | NR | NR | PSP | PET-CT; MDCT | NR | 9 |
| Park SR et al. [34] | 2008 | 38 | P | 75.0 | 58.0 | PSP | MDCT; EUS | Yes | 12 |
| Yang DM et al. [35] | 2007 | 44 | R | 77.3 | 57.0 | PSP | MDCT | NR | 10 |
| Chen CY et al. [36] | 2007 | 55 | P | 69.1 | 63.0 | PSP | MDCT | Yes | 12 |
| Ren G et al. [37] | 2007 | 77 | NR | NR | NR | PSP | MDCT | NR | 9 |
| Shinohara T et al. [38] | 2005 | 278 | NR | 69.1 | 60.0 | PSP | MDCT | Yes | 11 |
| Kim HJ et al. [39] | 2005 | 106 | P | 67.9 | 56.0 | PSP | MDCT | Yes | 13 |
| Yun M et al. [40] | 2005 | 81 | R | 65.4 | 56.6 | PSP | PET-CT; MDCT | Yes | 12 |
| Bhandari S et al. [41] | 2004 | 63 | R | 63.5 | 61.0 | PSP | MDCT; EUS | Yes | 12 |
| Lee DH et al. [42] | 2001 | 180 | NR | 62.8 | 56.0 | PSP | MDCT | Yes | 13 |
| D’Elia F et al. [43] | 2000 | 107 | P | 68.2 | 64.0 | PSP | MDCT | No | 11 |
| Hundt et al. [44] | 1999 | 39 | P | 57.5 | 63.0 | PSP | MDCT | Yes | 12 |
P, Prospective; R, Retrospective; PSP, Post-surgery Pathology; MDCT, Multidetector Computed Tomography; NR, Not Reported; PET-CT, Positron Emission Tomography Computed Tomography; EUS, Endoscopic ultrasonography; HGS, Hydrogastric sonography; QUADAS, Quality Assessment of Diagnostic Accuracy Studies.
Study included criteria
Articles were selected if they fulfilled all of the following inclusion criteria: (a) MDCT was used to evaluate gastric cancer patients without surgery or any other treatment; (b) pre-operative lymph node staging of gastric cancer was investigated in the articles and the regional lymph nodes were considered to be involved by metastases if they were larger than 8 mm in the short-axis diameter; (c) sufficient data were obtained to calculate the true-positive (TP), false-positive (FP), true negative (TN) and false-negative (FN) values; (d) post-operation histopathological evaluation was served as a reference standard; (e) articles were published in English and Chinese; (f) 20 or more patients were included; (g) About the quality of the study design, only the study in which the number of the answer “yes” for the 14 items in the Quality Assessment of Diagnostic Accuracy Studies (QUADAS) checklist [11] was more than nine was selected; (h) when data were published in more than one article, the publications with the most details was included. Review articles, letters, case reports, conference records, comments as well as publications that did not provide raw data, were excluded.
Data extraction
The methodological quality of the included studies was evaluated by two investigators independently. The QUADAS checklists were applied to assess the methodological quality of the selected articles. To perform accuracy analysis, we extracted data about the characteristics of patients and studies, including first author, year of publication, sample size, characteristics of study population (gender and age), study design, gold standard, the diagnostic equipment as well as whether the results of MDCT were blinded to the pathological diagnosis.
For each study, we obtained the number of TP, FP, TN and FN cases for MDCT in diagnosing the staging of primary gastric cancer. The data was also recorded for EUS, which was utilized for comparison with MDCT in the eligible articles.
Statistical analysis
Data about the diagnostic performance of MDCT were pooled quantitatively across eligible articles. Data were used to construct 2×2 contingency tables to calculate sensitivity, specificity and diagnostic odds ratio estimators with confidence intervals (CIs), which were plotted graphically in forest plots. A value of 0.5 was added to all cells of studies which contained a count of zero to avoid subsequent problems in odds calculations for articles with sensitivity or specificity of 100%. Likelihood ratios (LR) are also metrics that pool sensitivity and specificity in the calculations. In previous papers, a test was considered clinically useful when positive LR was greater than 5.0 and negative LR was less than 0.2 [12]. Heterogeneity was evaluated by X 2-test and P < 0.05 was considered as existing obvious heterogeneity. If heterogeneity existed, a random-effect model was utilized for the primary meta-analysis to obtain a summary estimate for sensitivity with 95% CI.
Testing of the diagnostic threshold was performed by Spearman’s correlation test. Then we used the derived estimates of sensitivity, specificity and respective variances to construct summary receiver operating characteristic (ROC) curves. The area under the summary ROC curves was used as an alternative general measure of test performance [13,14]. The SROC curve shows the trade-off between sensitivity and specificity across the selected articles [15]. A summary ROC curve located near the upper left corner indicates the better diagnostic modality. All the statistical computations were carried out using Meta-disc (version 1.4, http://www.hrc.es/investigacion/metadisc_en.htm) [16]. Meta-disc is an free software to perform a meta-analysis of researches of assessment of diagnostic tests and screening. P < 0.05 was thought to be statistically significant.
Results
Literature retrieval and inclusion of articles
After the computerized search was performed and reference lists were comprehensively cross-checked, 272 literatures were yielded, of which 223 were excluded according to their titles and abstracts. 49 potentially appropriated articles were included for further retrieval through screening the full text. Among them, 20 articles were excluded because of following reasons: case only reports (n=3), reviews (n=3), only T staging (n=5), essential data missing to construction 2×2 contingency tables (n=7) and overlapping study (n=2). Therefore, 29 eligible articles, meeting all of the inclusion criteria, were included for the analysis [7,17-44]. Yan C et al. reported two independent studies (305 cases prospectively and 61 cases retrospectively) about N staging of gastric cancer patients in an articles [26]. Therefore, 30 studies involved in N staging of gastric cancer patients utilizing MDCT were included for following analysis. The detailed procedure of study inclusion in the meta-analysis was revealed in Figure 1.
Figure 1.

Flow chart of selection processes for eligible studies. MDCT, Multidetector Computed Tomography.
Study characteristics and study quality assessment
The characteristics of the included studies are presented in Table 1. There are a total of 6637 gastric cancer patients in the 30 included studies. Among them, one study reported that 278 GC patients performed MDCT examination and 451 regional lymph nodes detected for evaluating pre-operative N staging [38]. 14 studies enrolled patients prospectively while the other 16 studies were retrospective or not reported. The ratio of male and mean age of every study was revealed in Table 1. In all the 30 studies, post-surgery pathology was served as the gold standard and MDCT was utilized as a diagnostic instrument for pre-operative N staging in gastric cancer patients. Among the 30 included studies, EUS was used in five studies for pre-operative N staging including 1014 gastric cancer patients. There were 20 studies in which the MDCT reviewers were blinded to patients’ clinical data and other test results while the other 10 studies did not report whether they adopted the blinding.
We used the QUADAS tool to assess each selected study. All included studies in the meta-analysis fulfilled nine or more of the fourteen criteria in the QUADAS tool for study quality, which could be found in Table 1. There were no uninterpretable and/or intermediate test results reported (100% for “No” response to question 13). Furthermore, 10 studies were not blinded in the results of the index test results (33.3% for “No” response to questions 10 and 11).
Diagnostic accuracy of pre-surgical N staging using MDCT
All the 30 studies, including 6637 GC patients, involved in pre-operative N staging using MDCT. The data of each study and the results of the statistical pooling are shown in Table 2. The pooled estimates of sensitivity, specificity, positive likelihood ratio (LR+), negative likelihood ratio (LR-) and diagnostic odds ratio (DOR) of MDCT in the detection of pre-operative lymph node staging in GC patients were 0.67 (95% CI: 0.66-0.69), 0.84 (95% CI: 0.83-0.85), 3.25 (95% CI: 2.69-3.93), 0.36 (95% CI: 0.28-0.46) and 10.31 (95% CI: 7.66-13.88), respectively.
Table 2.
Diagnositc value of MDCT in detection of involved lymph node in preoperative GC patients
| Sensitivity | Specificity | Likelihood ratios | Diagnostic Odds Ratio | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
|||||||||||||||
| LR+ | LR- | ||||||||||||||
|
|
|||||||||||||||
| References | No. | TP | FP | FN | TN | V | 95% CI | V | 95% CI | V | 95% CI | V | 95% CI | V | 95% CI |
| Fujikawa H | 525 | 2 | 6 | 45 | 472 | 0.043 | 0.005-0.145 | 0.987 | 0.973-0.995 | 3.390 | 0.704-16.33 | 0.970 | 0.912-1.031 | 3.496 | 0.686-17.83 |
| Yoshikawa T | 75 | 45 | 14 | 8 | 8 | 0.849 | 0.724-0.933 | 0.364 | 0.172-0.593 | 1.334 | 0.954-1.866 | 0.415 | 0.178-0.966 | 3.214 | 1.019-10.14 |
| Hasegawa S | 315 | 50 | 2 | 57 | 206 | 0.467 | 0.370-0.566 | 0.990 | 0.966-0.999 | 48.60 | 12.06-195.9 | 0.538 | 0.450-0.643 | 90.35 | 21.33-382.7 |
| Kim SH | 171 | 39 | 11 | 26 | 95 | 0.600 | 0.471-0.720 | 0.896 | 0.822-0.947 | 5.782 | 3.193-10.47 | 0.446 | 0.329-0.605 | 12.96 | 5.836-28.76 |
| Feng XY | 610 | 361 | 72 | 64 | 113 | 0.849 | 0.812-0.882 | 0.611 | 0.537-0.681 | 2.183 | 1.814-2.626 | 0.247 | 0.191-0.318 | 8.853 | 5.949-13.17 |
| Zilai P | 96 | 62 | 11 | 6 | 17 | 0.912 | 0.818-0.967 | 0.607 | 0.406-0.785 | 2.321 | 1.456-3.700 | 0.145 | 0.064-0.330 | 15.97 | 5.158-49.45 |
| Zhong BY | 115 | 48 | 8 | 12 | 37 | 0.800 | 0.677-0.892 | 0.822 | 0.679-0.920 | 4.500 | 2.371-8.542 | 0.243 | 0.144-0.411 | 18.50 | 6.860-49.89 |
| Marrelli D | 92 | 11 | 4 | 2 | 75 | 0.846 | 0.546-0.981 | 0.949 | 0.875-0.986 | 16.71 | 6.256-44.64 | 0.162 | 0.045-0.580 | 103.1 | 16.85-631.1 |
| Ha TK | 78 | 23 | 14 | 10 | 31 | 0.697 | 0.513-0.844 | 0.689 | 0.534-0.818 | 2.240 | 1.373-3.655 | 0.440 | 0.253-0.765 | 5.093 | 1.922-13.49 |
| Kim EY | 71 | 44 | 1 | 15 | 11 | 0.746 | 0.616-0.850 | 0.917 | 0.615-0.998 | 8.949 | 1.362-58.79 | 0.277 | 0.173-0.443 | 32.27 | 3.837-271.3 |
| Yan C | 61 | 24 | 8 | 7 | 22 | 0.774 | 0.589-0.904 | 0.733 | 0.541-0.877 | 2.903 | 1.557-5.414 | 0.308 | 0.155-0.612 | 9.429 | 2.933-30.31 |
| Yan C | 305 | 140 | 59 | 22 | 84 | 0.864 | 0.802-0.913 | 0.587 | 0.502-0.669 | 2.095 | 1.707-2.571 | 0.231 | 0.153-0.349 | 9.060 | 5.178-15.85 |
| Lee IJ | 148 | 8 | 2 | 22 | 116 | 0.267 | 0.123-0.459 | 0.983 | 0.940-0.998 | 15.73 | 3.522-70.29 | 0.746 | 0.600-0.927 | 21.09 | 4.194-106.1 |
| Venkataraman | 42 | 24 | 1 | 15 | 2 | 0.615 | 0.446-0.766 | 0.667 | 0.094-0.992 | 1.846 | 0.366-9.323 | 0.577 | 0.236-1.409 | 3.200 | 0.266-38.43 |
| Park SR | 1964 | 493 | 221 | 367 | 883 | 0.573 | 0.539-0.607 | 0.800 | 0.775-0.823 | 2.864 | 2.511-3.265 | 0.534 | 0.491-0.580 | 5.367 | 4.394-6.555 |
| Hwang SW | 247 | 37 | 24 | 46 | 140 | 0.446 | 0.337-0.559 | 0.854 | 0.790-0.904 | 3.046 | 1.961-4.733 | 0.649 | 0.530-0.795 | 4.692 | 2.544-8.655 |
| Ahn HS | 434 | 8 | 32 | 39 | 355 | 0.170 | 0.076-0.308 | 0.917 | 0.885-0.943 | 2.059 | 1.009-4.200 | 0.905 | 0.792-1.033 | 2.276 | 0.980-5.284 |
| Yan C | 135 | 16 | 20 | 6 | 93 | 0.727 | 0.498-0.893 | 0.823 | 0.740-0.888 | 4.109 | 2.561-6.593 | 0.331 | 0.167-0.659 | 12.40 | 4.316-35.62 |
| Yang QM | 78 | 26 | 6 | 17 | 29 | 0.605 | 0.444-0.750 | 0.829 | 0.664-0.934 | 3.527 | 1.637-7.598 | 0.477 | 0.320-0.711 | 7.392 | 2.534-21.57 |
| Park SR | 38 | 24 | 4 | 7 | 3 | 0.774 | 0.589-0.904 | 0.429 | 0.099-0.816 | 1.355 | 0.694-2.645 | 0.527 | 0.180-1.544 | 2.571 | 0.462-14.32 |
| Yang DM | 44 | 16 | 4 | 3 | 21 | 0.842 | 0.604-0.966 | 0.840 | 0.639-0.955 | 5.263 | 2.099-13.19 | 0.188 | 0.066-0.538 | 28.00 | 5.474-143.2 |
| Chen CY | 55 | 34 | 5 | 3 | 13 | 0.919 | 0.781-0.983 | 0.722 | 0.465-0.903 | 3.308 | 1.561-7.010 | 0.112 | 0.037-0.345 | 29.48 | 6.145-141.3 |
| Ren G | 77 | 10 | 3 | 22 | 42 | 0.313 | 0.161-0.500 | 0.933 | 0.817-0.986 | 4.688 | 1.401-15.69 | 0.737 | 0.576-0.942 | 6.364 | 1.586-25.54 |
| Shinohara T | 451a | 99 | 28 | 47 | 277 | 0.678 | 0.596-0.753 | 0.908 | 0.870-0.938 | 7.386 | 5.101-10.70 | 0.354 | 0.279-0.450 | 20.84 | 12.37-35.09 |
| Kim HJ | 106 | 36 | 23 | 10 | 37 | 0.783 | 0.636-0.891 | 0.617 | 0.482-0.739 | 2.042 | 1.431-2.912 | 0.353 | 0.197-0.632 | 5.791 | 2.420-13.86 |
| Yun M | 81 | 48 | 11 | 5 | 17 | 0.906 | 0.793-0.969 | 0.607 | 0.406-0.785 | 2.305 | 1.443-3.683 | 0.155 | 0.064-0.377 | 14.84 | 4.500-48.92 |
| Bhandari S | 48b | 16 | 4 | 4 | 24 | 0.800 | 0.563-0.943 | 0.857 | 0.673-0.960 | 5.600 | 2.202-14.24 | 0.233 | 0.096-0.568 | 24.00 | 5.231-110.1 |
| Lee DH | 180 | 59 | 15 | 36 | 70 | 0.621 | 0.516-0.719 | 0.824 | 0.726-0.898 | 3.519 | 2.166-5.718 | 0.460 | 0.349-0.606 | 7.648 | 3.818-15.32 |
| D’Elia F | 107 | 70 | 12 | 2 | 23 | 0.972 | 0.903-0.997 | 0.657 | 0.478-0.809 | 2.836 | 1.790-4.493 | 0.042 | 0.011-0.169 | 67.08 | 13.97-322.2 |
| Hundt W | 39 | 35 | 0 | 2 | 2 | 0.946 | 0.818-0.993 | 1.000 | 0.158-1.000 | 5.605 | 0.446-70.49 | 0.079 | 0.022-0.290 | 71.00 | 2.629 -1918 |
| Pooled data | 6788 | 0.673 | 0.655-0.690 | 0.841 | 0.830-0.853 | 3.247 | 2.686-3.926 | 0.363 | 0.284-0.464 | 10.31 | 7.660-13.88 | ||||
GC, Gastric Cancer; MDCT, Multidetector Computed Tomography; V, Value; CI, Confidence interval; LR, Likelihood ratio; TP, True positive; FP, False Positive; TN, True Negative; FN, False Negative.
There are 451 lymph node in total in 278 gastric cancer patients.
48 cases in 63 gastric cancer patients have complete data of lymph node involvement.
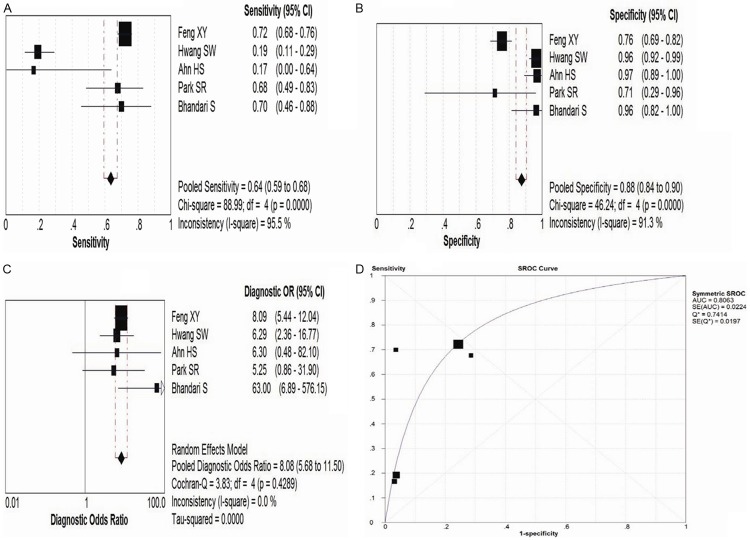
Heterogeneity were found in sensitivity, specificity, LR+, LR- and DOR between 30 included studies after assessment by plotting the above parameters from each study on a forest plot and calculating the heterogeneity x2, which could be seen in Figure 2A-C. The threshold effect was one important extra source of variation in the meta-analysis. To judge whether the threshold effect existed, the spearman correlation test was utilized to verify it. The spearman correlation coefficient was 0.630 and P value was 0.000, which suggested that the threshold effect existed in this meta-analysis. Then we fitted a summary ROC to assess the diagnostic accuracy of MDCT, which could be seen in Figure 2D. The Q* index was calculated as a globe measure of diagnostic accuracy. The AUC and Q* were 0.8338 and 0.7661, respectively (Tables 2 and 3).
Figure 2.
The forest plot and summary ROC curve of MDCT in evaluating pre-operative lymph node staging in gastric cancer patients are illustrated. (A-C) Showed the forest plot of pooled sensitivity (A), specificity (B) and Diagnostic Odds Ratio (C), respectively. (D) Revealed the summary ROC curve of MDCT. MDCT, Multidetector Computed Tomography; ROC, receiver operating characteristic.
Table 3.
Comparison of the accuracy of pre-operative N staging in gastric cancer using MDCT and EUS
| Diagnostic Method | Number of study | Diagnostic Threshold | Pooled Sensitivity | Pooled Specificity | Positive LR | Negative LR | Pooled DOR | AUC | Q* |
|---|---|---|---|---|---|---|---|---|---|
| MDCT | 30 | Yes | 0.67 (0.66-0.69) | 0.84 (0.83-0.85) | 3.25 (2.69-3.93) | 0.36 (0.28-0.46) | 10.31 (7.66-13.88) | 0.8338 | 0.7661 |
| EUS | 5 | No | 0.64 (0.59-0.68) | 0.88 (0.84-0.90) | 3.66 (2.29-5.85) | 0.53 (0.30-0.94) | 8.08 (5.68-11.50) | 0.8063 | 0.7414 |
MDCT, Multidetector Computed Tomography; EUS, Endoscopic ultrasonography; LR, Likelihood Ratio; DOR, Diagnostic Odds Ratio; AUC, area under the curve.
As a control test, EUS has been simultaneously performed in 5 studies [20,30,31,34,41]. The pooled estimates of sensitivity, specificity, LR+, LR- and DOR of CT in the detection of pre-operative lymph node staging in GC patients were 0.64 (95% CI: 0.59-0.68), 0.88 (95% CI: 0.84-0.90), 3.66 (95% CI: 2.29-5.85), 0.53 (95% CI: 0.30-0.94) and 8.08 (95% CI: 5.68-11.50), respectively, which could be seen in Figure 3; Tables 3 and S1. Diagnostic threshold did not exist (Spearman correlation coefficient was 0.500 and P=0.391). The AUC and Q* were 0.8063 and 0.7414, respectively (Table 3 and Figure 3D).
Figure 3.
The forest plot and summary ROC curve of EUS in evaluating pre-operative lymph node staging in gastric cancer patients are presented. (A-C) illustrated the forest plot of pooled sensitivity (A), specificity (B) and Diagnostic Odds Ratio (C), respectively. (D) Showed the summary ROC curve of EUS. ROC, receiver operating characteristic; EUS, endoscopic ultrasonography.
Discussion
Gastric cancer is often diagnosed at an advanced stage in most countries including China. Though the primary management of GC is surgical resection, the treatment principle of GC currently was comprehensive therapy including surgery, chemotherapy, radiotherapy, molecular targeted therapy and other treatment. Accurate preoperative clinical staging, especially N staging, is essential to select proper individualized therapeutic strategy. EUS and MDCT are the most common techniques for the preoperative staging of GC patients. However, the results of pre-operative N staging of MDCT in GC patients were differently reported. To the best of our knowledge, this is the first meta-analysis to evaluate the value of MDCT concerning the pre-operative N staging in gastric cancer patients.
The diagnostic performance of the 30 studies discussed in the systematic review was patient-based. The pooled sensitivity, specificity, positive LR, negative LR and DOR of MDCT in the diagnosis of preoperative lymph node metastasis in GC patients were 0.67, 0.84, 3.25, 0.36 and 10.31, respectively. The global diagnostic accuracy was 0.7661. Though the sensitivity was not satisfactory, the results (a high specificity) suggested that MDCT was a specific diagnostic tool for the evaluation of lymph node metastasis. When compared with EUS, another preoperative diagnostic tool, the global diagnostic accuracy of MDCT was higher when assessing preoperative N staging in GC patients because the Q* of EUS and MDCT was 0.7661 and 0.7414, respectively. Moreover, MDCT was a non-invasive examination and universal in clinical application. Therefore, there is enough evidence to support the routine use of MDCT to evaluate possible lymph node metastasis in GC patients.
Currently, two major classifications are used concerning gastric cancer. The Japanese classification is more elaborate and is based on anatomic involvement, especially the lymph node stations [45]. The other staging system developed by the AJCC and UICC, which was based on the number of involved lymph node and a minimum of 15 examined lymph nodes was recommended for adequate staging [46]. All of the selected studies in this meta-analysis adopted the AJCC/UICC staging system. It is difficult to distinguish the accurate number of involved lymph node when evaluating the pre-operative N staging using MDCT. Therefore, we divided N staging into two groups (N0 and N+ group) to perform the following analysis. As for the assessment standard of metastatic lymph node when using MDCT before operation, there were several different criterias. Most literatures showed that the regional lymph nodes were considered to be involved by metastases if they were larger than 8 mm in the short-axis diameter [7,17,18,47]. Some researches thought that lymph nodes were considered positive for metastasis when the short-axis diameter was larger than 6 mm for perigastric lymph nodes and larger than 8 mm for the extraperigastric lymph nodes, especially rounded nodes with enhancement on contrast-enhanced CT that were sometimes necrotic [20,21]. Kim SH et al. reported that LN metastasis was considered present if the short-axis diameter of any LNs was larger than 8 mm, if there was a cluster of three or more perilesional nodes regardless of size, if the LNs showed strong enhancement (> 100 HU), or if LNs with central necrosis and perinodal infiltration [7].
Lymph node metastasis is the most important prognostic factor of GC and the therapeutic strategy is established based on precise staging. Comprehensive therapy for nodal positive or locally advanced disease was considered to improve survival and reduce the risk of local recurrence, especially in gastric cancer. This meta-analysis showed that the pooled sensitivity was 67%. It is not a satisfactory result due to its inability to detect microscopic nodal invasion, which is common in gastric cancer [48]. However, the pooled sensitivity of MDCT was superior to that of EUS in this study, maybe because EUS was limited by the detection distance. Our meta-analysis results revealed that MDCT, along with EUS, was a specific diagnostic tool for the evaluation of lymph node involvement. Because of existing of the threshold effect, we fit a summary ROC to evaluate the diagnostic accuracy of MDCT. The AUC and Q* were 0.8338 and 0.7661, respectively, suggesting that MDCT was a good diagnostic tool concerning pre-operative N staging in GC patients. As a control, The AUC and Q* of EUS were 0.8063 and 0.7414, respectively, implying that the diagnostic value of MDCT involving in pre-operative N staging in GC patients was superior to that of EUS. Furthermore, some studies reported that MRI, PET-CT and other imaging equipment was also used to evaluate the diagnostic value of pre-operative N staging in GC patients. However, the number of literatures as well as the sample size was so few that there were no reliable results to analyze the diagnostic value.
We should acknowledge some potential limitations in this meta-analysis. Firstly, the presence of clinical heterogeneity in the study design, patient population and quality in these included studies influences the generalization of the results. Secondly, one third studies did not report whether they adopted the blinding. The interpretation of MDCT scans was performed qualitatively in the majority of studies. So there is a risk of subjective interpretation. Thirdly, 14 studies enrolled patients prospectively while the other 16 studies were retrospective. And only 5 studies included conventional imaging (EUS) as a control. Above factors impaired the performance and application of MDCT. To minimize bias in the selection of studies and data extraction, reviewers who blinded to the journal, authors and institution independently retrieved articles according to the inclusion criteria. Moreover, we used the QUADAS tool to guarantee that all the included articles were high quality articles. Finally, the current analysis did not allow region-by-region or node-by-node comparison, which might provide other crucial information.
In conclusion, the present analysis revealed that the diagnostic accuracy of MDCT concerning pre-operative N staging in gastric cancer patients was superior to that of EUS. So currently there is enough evidence to support the routine clinical application of MDCT in the preoperative evaluation of lymph node status in GC patients.
Acknowledgements
This study was supported by Department of Gastrointestinal Surgery and Institute of Clinical Medicine, The First Affiliated Hospital, Zhengzhou University and National Natural Science Foundation of China, Grant No. 81201955. Furthermore, the study was supported by Department of Function, The Fifth Affiliated of Zhengzhou University.
Disclosure of conflict of interest
None.
Supporting Information
References
- 1.Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer. 2010;127:2893–917. doi: 10.1002/ijc.25516. [DOI] [PubMed] [Google Scholar]
- 2.Jung KW, Park S, Kong HJ, Won YJ, Boo YK, Shin HR, Park EC, Lee JS. Cancer statistics in Korea: incidence, mortality and survival in 2006-2007. J Korean Med Sci. 2010;25:1113–21. doi: 10.3346/jkms.2010.25.8.1113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Menges M, Hoehler T. Current strategies in systemic treatment of gastric cancer and cancer of the gastroesophageal junction. J Cancer Res Clin Oncol. 2009;135:29–38. doi: 10.1007/s00432-008-0425-z. [DOI] [PubMed] [Google Scholar]
- 4.Japanese Gastric Cancer Association. Japanese gastric cancer treatment guidelines 2010 (ver. 3) Gastric Cancer. 2011;14:113–23. doi: 10.1007/s10120-011-0042-4. [DOI] [PubMed] [Google Scholar]
- 5.Kim YW, Yoon HM, Eom BW, Park JY. History of minimally invasive surgery for gastric cancer in Korea. J Gastric Cancer. 2012;12:13–7. doi: 10.5230/jgc.2012.12.1.13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ajani JA, Winter K, Okawara GS, Donohue JH, Pisters PW, Crane CH, Greskovich JF, Anne PR, Bradley JD, Willett C, Rich TA. Phase II trial of preoperative chemoradiation in patients with localized gastric adenocarcinoma (RTOG 9904): quality of combined modality therapy and pathologic response. J. Clin. Oncol. 2006;24:3953–8. doi: 10.1200/JCO.2006.06.4840. [DOI] [PubMed] [Google Scholar]
- 7.Kim SH, Kim JJ, Lee JS, Kim SH, Kim BS, Maeng YH, Hyun CL, Kim MJ, Jeong IH. Preoperative N staging of gastric cancer by stomach protocol computed tomography. J Gastric Cancer. 2013;13:149–56. doi: 10.5230/jgc.2013.13.3.149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kwee RM, Kwee TC. Imaging in local staging of gastric cancer: a systematic review. J. Clin. Oncol. 2007;25:2107–16. doi: 10.1200/JCO.2006.09.5224. [DOI] [PubMed] [Google Scholar]
- 9.Kwee RM, Kwee TC. Imaging in assessing lymph node status in gastric cancer. Gastric Cancer. 2009;12:6–22. doi: 10.1007/s10120-008-0492-5. [DOI] [PubMed] [Google Scholar]
- 10.Habermann CR, Weiss F, Riecken R, Honarpisheh H, Bohnacker S, Staedtler C, Dieckmann C, Schoder V, Adam G. Preoperative staging of gastric adenocarcinoma: comparison of helical CT and endoscopic US. Radiology. 2004;230:465–71. doi: 10.1148/radiol.2302020828. [DOI] [PubMed] [Google Scholar]
- 11.Whiting P, Rutjes AW, Reitsma JB, Bossuyt PM, Kleijnen J. The development of QUADAS: a tool for the quality assessment of studies of diagnostic accuracy included in systematic reviews. BMC Med Res Methodol. 2003;3:25–37. doi: 10.1186/1471-2288-3-25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kang S, Kim SK, Chung DC, Seo SS, Kim JY, Nam BH, Park SY. Diagnostic value of 18F-FDG PET for evaluation of para-aortic nodal metastasis in patients with cervical carcinoma: a meta-analysis. J Nucl Med. 2010;51:360–7. doi: 10.2967/jnumed.109.066217. [DOI] [PubMed] [Google Scholar]
- 13.Harbord RM, Deeks JJ, Egger M, Whiting P, Sterne JA. A unification of models for meta-analysis of diagnostic accuracy studies. Biostatistics. 2007;8:239–51. doi: 10.1093/biostatistics/kxl004. [DOI] [PubMed] [Google Scholar]
- 14.Rutter CM, Gatsonis CA. A hierarchical regression approach to meta-analysis of diagnostic test accuracy evaluations. Stat Med. 2001;20:2865–84. doi: 10.1002/sim.942. [DOI] [PubMed] [Google Scholar]
- 15.Moses LE, Shapiro D, Littenberg B. Combining independent studies of a diagnostic test into a summary ROC curve: data-analytic approaches and some additional considerations. Stat Med. 1993;12:1293–316. doi: 10.1002/sim.4780121403. [DOI] [PubMed] [Google Scholar]
- 16.Zamora J, Abraira V, Muriel A, Khan K, Coomarasamy A. Meta-DiSc: a software for meta-analysis of test accuracy data. BMC Med Res Methodol. 2006;6:31. doi: 10.1186/1471-2288-6-31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Fujikawa H, Yoshikawa T, Hasegawa S, Hayashi T, Aoyama T, Ogata T, Cho H, Oshima T, Rino Y, Morita S, Masuda M. Diagnostic value of computed tomography for staging of clinical t1 gastric cancer. Ann Surg Oncol. 2014;21:3002–7. doi: 10.1245/s10434-014-3667-9. [DOI] [PubMed] [Google Scholar]
- 18.Yoshikawa T, Tanabe K, Nishikawa K, Ito Y, Matsui T, Kimura Y, Hasegawa S, Aoyama T, Hayashi T, Morita S, Miyashita Y, Tsuburaya A, Sakamoto J. Accuracy of CT staging of locally advanced gastric cancer after neoadjuvant chemotherapy: cohort evaluation within a randomized phase II study. Ann Surg Oncol. 2014;21(Suppl 3):S385–9. doi: 10.1245/s10434-014-3615-8. [DOI] [PubMed] [Google Scholar]
- 19.Hasegawa S, Yoshikawa T, Shirai J, Fujikawa H, Cho H, Doiuchi T, Yoshida T, Sato T, Oshima T, Yukawa N, Rino Y, Masuda M, Tsuburaya A. A prospective validation study to diagnose serosal invasion and nodal metastases of gastric cancer by multidetector-row CT. Ann Surg Oncol. 2013;20:2016–22. doi: 10.1245/s10434-012-2817-1. [DOI] [PubMed] [Google Scholar]
- 20.Feng XY, Wang W, Luo GY, Wu J, Zhou ZW, Li W, Sun XW, Li YF, Xu DZ, Guan YX, Chen S, Zhan YQ, Zhang XS, Xu GL, Zhang R, Chen YB. Comparison of endoscopic ultrasonography and multislice spiral computed tomography for the preoperative staging of gastric cancer-results of a single institution study of 610 Chinese patients. PLoS One. 2013;8:e78846. doi: 10.1371/journal.pone.0078846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Pan Z, Pang L, Ding B, Yan C, Zhang H, Du L, Wang B, Song Q, Chen K, Yan F. Gastric cancer staging with dual energy spectral CT imaging. PLoS One. 2013;8:e53651. doi: 10.1371/journal.pone.0053651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zhong BY, Liu YX, Huang WF, Liu QQ, Liu SQ, Liu Y. Clinical value of 64-slice spiral 3-phase CT enhanced scanning for preoperative TNM staging assessment of gastric carcinoma. Zhonghua Wei Chang Wai Ke Za Zhi. 2012;15:706–9. [PubMed] [Google Scholar]
- 23.Marrelli D, Mazzei MA, Pedrazzani C, Di Martino M, Vindigni C, Corso G, Morelli E, Volterrani L, Roviello F. High accuracy of multislices computed tomography (MSCT) for para-aortic lymph node metastases from gastric cancer: a prospective single-center study. Ann Surg Oncol. 2011;18:2265–72. doi: 10.1245/s10434-010-1541-y. [DOI] [PubMed] [Google Scholar]
- 24.Ha TK, Choi YY, Song SY, Kwon SJ. F18-fluorodeoxyglucose-positron emission tomography and computed tomography is not accurate in preoperative staging of gastric cancer. J Korean Surg Soc. 2011;81:104–10. doi: 10.4174/jkss.2011.81.2.104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kim EY, Lee WJ, Choi D, Lee SJ, Choi JY, Kim BT, Kim HS. The value of PET/CT for preoperative staging of advanced gastric cancer: Comparison with contrast-enhanced CT. Eur J Radiol. 2011;79:183–8. doi: 10.1016/j.ejrad.2010.02.005. [DOI] [PubMed] [Google Scholar]
- 26.Yan C, Zhu ZG, Yan M, Zhang H, Pan ZL, Chen J, Xiang M, Chen MM, Liu BY, Yin HR, Lin YZ. Size of the largest lymph node visualized on multi-detector-row computed tomography (MDCT) is useful in predicting metastatic lymph node status of gastric cancer. J Int Med Res. 2010;38:22–33. doi: 10.1177/147323001003800103. [DOI] [PubMed] [Google Scholar]
- 27.Lee IJ, Lee JM, Kim SH, Shin CI, Lee JY, Kim SH, Han JK, Choi BI. Diagnostic performance of 64-channel multidetector CT in the evaluation of gastric cancer: differentiation of mucosal cancer (T1a) from submucosal involvement (T1b and T2) Radiology. 2010;255:805–14. doi: 10.1148/radiol.10091313. [DOI] [PubMed] [Google Scholar]
- 28.Venkataraman I, Rao HK, Singh P, Elangovan S, Kate V. Efficacy of hydrogastric sonography and spiral Computed Tomography in staging of gastric carcinoma--a comparative study. J Clin Ultrasound. 2010;38:480–5. doi: 10.1002/jcu.20734. [DOI] [PubMed] [Google Scholar]
- 29.Park SR, Kim MJ, Ryu KW, Lee JH, Lee JS, Nam BH, Choi IJ, Kim YW. Prognostic value of preoperative clinical staging assessed by computed tomography in resectable gastric cancer patients: a viewpoint in the era of preoperative treatment. Ann Surg. 2010;251:428–35. doi: 10.1097/SLA.0b013e3181ca69a7. [DOI] [PubMed] [Google Scholar]
- 30.Hwang SW, Lee DH, Lee SH, Park YS, Hwang JH, Kim JW, Jung SH, Kim NY, Kim YH, Lee KH, Kim HH, Park do J, Lee HS, Jung HC, Song IS. Preoperative staging of gastric cancer by endoscopic ultrasonography and multidetector-row computed tomography. J Gastroenterol Hepatol. 2010;25:512–8. doi: 10.1111/j.1440-1746.2009.06106.x. [DOI] [PubMed] [Google Scholar]
- 31.Ahn HS, Lee HJ, Yoo MW, Kim SG, Im JP, Kim SH, Kim WH, Lee KU, Yang HK. Diagnostic accuracy of T and N stages with endoscopy, stomach protocol CT, and endoscopic ultrasonography in early gastric cancer. J Surg Oncol. 2009;99:20–7. doi: 10.1002/jso.21170. [DOI] [PubMed] [Google Scholar]
- 32.Yan C, Zhu ZG, Yan M, Zhang H, Pan ZL, Chen J, Xiang M, Chen MM, Liu BY, Yin HR, Lin YZ. Value of multidetector-row computed tomography in the preoperative T and N staging of gastric carcinoma: a large-scale Chinese study. J Surg Oncol. 2009;100:205–14. doi: 10.1002/jso.21316. [DOI] [PubMed] [Google Scholar]
- 33.Yang QM, Kawamura T, Itoh H, Bando E, Nemoto M, Akamoto S, Furukawa H, Yonemura Y. Is PET-CT suitable for predicting lymph node status for gastric cancer? Hepatogastroenterology. 2008;55:782–5. [PubMed] [Google Scholar]
- 34.Park SR, Lee JS, Kim CG, Kim HK, Kook MC, Kim YW, Ryu KW, Lee JH, Bae JM, Choi IJ. Endoscopic ultrasound and computed tomography in restaging and predicting prognosis after neoadjuvant chemotherapy in patients with locally advanced gastric cancer. Cancer. 2008;112:2368–76. doi: 10.1002/cncr.23483. [DOI] [PubMed] [Google Scholar]
- 35.Yang DM, Kim HC, Jin W, Ryu CW, Kang JH, Park CH, Kim HS, Jung DH. 64 multidetector-row computed tomography for preoperative evaluation of gastric cancer: histological correlation. J Comput Assist Tomogr. 2007;31:98–103. doi: 10.1097/01.rct.0000234072.16209.ab. [DOI] [PubMed] [Google Scholar]
- 36.Chen CY, Hsu JS, Wu DC, Kang WY, Hsieh JS, Jaw TS, Wu MT, Liu GC. Gastric cancer: preoperative local staging with 3D multi-detector row CT--correlation with surgical and histopathologic results. Radiology. 2007;242:472–82. doi: 10.1148/radiol.2422051557. [DOI] [PubMed] [Google Scholar]
- 37.Ren G, Cai R, Chen KM. Clinical value of multidetector computed tomography in detecting lymph node metastasis of early gastric cancer. Zhonghua Zhong Liu Za Zhi. 2007;29:852–5. [PubMed] [Google Scholar]
- 38.Shinohara T, Ohyama S, Yamaguchi T, Muto T, Kohno A, Kato Y, Urashima M. Clinical value of multidetector row computed tomography in detecting lymph node metastasis of early gastric cancer. Eur J Surg Oncol. 2005;31:743–8. doi: 10.1016/j.ejso.2005.03.005. [DOI] [PubMed] [Google Scholar]
- 39.Kim HJ, Kim AY, Oh ST, Kim JS, Kim KW, Kim PN, Lee MG, Ha HK. Gastric cancer staging at multi-detector row CT gastrography: comparison of transverse and volumetric CT scanning. Radiology. 2005;236:879–85. doi: 10.1148/radiol.2363041101. [DOI] [PubMed] [Google Scholar]
- 40.Yun M, Lim JS, Noh SH, Hyung WJ, Cheong JH, Bong JK, Cho A, Lee JD. Lymph node staging of gastric cancer using (18)F-FDG PET: a comparison study with CT. J Nucl Med. 2005;46:1582–8. [PubMed] [Google Scholar]
- 41.Bhandari S, Shim CS, Kim JH, Jung IS, Cho JY, Lee JS, Lee MS, Kim BS. Usefulness of three-dimensional, multidetector row CT (virtual gastroscopy and multiplanar reconstruction) in the evaluation of gastric cancer: a comparison with conventional endoscopy, EUS, and histopathology. Gastrointest Endosc. 2004;59:619–26. doi: 10.1016/s0016-5107(04)00169-5. [DOI] [PubMed] [Google Scholar]
- 42.Lee DH, Seo TS, Ko YT. Spiral CT of the gastric carcinoma: staging and enhancement pattern. Clin Imaging. 2001;25:32–7. doi: 10.1016/s0899-7071(01)00245-5. [DOI] [PubMed] [Google Scholar]
- 43.D’Elia F, Zingarelli A, Palli D, Grani M. Hydro-dynamic CT preoperative staging of gastric cancer: correlation with pathological findings. A prospective study of 107 cases. Eur Radiol. 2000;10:1877–85. doi: 10.1007/s003300000537. [DOI] [PubMed] [Google Scholar]
- 44.Hundt W, Braunschweig R, Reiser M. Assessment of gastric cancer: value of breathhold technique and two-phase spiral CT. Eur Radiol. 1999;9:68–72. doi: 10.1007/s003300050630. [DOI] [PubMed] [Google Scholar]
- 45.Japanese Research Society for Gastric cancer. The general rules for the gastric cancer study in surgery and pathology. 12th edition. Tokyo: Kanahara Shuppan; 1993. [Google Scholar]
- 46.Roder JD, Bottcher K, Busch R, Wittekind C, Hermanek P, Siewert JR. Classification of regional lymph node metastasis from gastric carcinoma. German Gastric Cancer Study Group. Cancer. 1998;82:621–31. [PubMed] [Google Scholar]
- 47.Habermann CR, Weiss F, Riecken R, Honarpisheh H, Bohnacker S, Staedtler C, Dieckmann C, Schoder V, Adam G. Preoperative staging of gastric adenocarcinoma: comparison of helical CT and endoscopic US. Radiology. 2004;230:465–71. doi: 10.1148/radiol.2302020828. [DOI] [PubMed] [Google Scholar]
- 48.Chen CY, Hsu JS, Wu DC, Kang WY, Hsieh JS, Jaw TS, Wu MT, Liu GC. Gastric cancer: preoperative local staging with 3D multidetector row CT-correlation with surgical and histopathologic results. Radiology. 2007;242:472–82. doi: 10.1148/radiol.2422051557. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.