Abstract
Background
It is not known if the most delayed late potentials are functionally most specific for scar-related ventricular tachycardia (VT) circuits.
Methods and Results
Isochronal late activation maps (ILAM) were constructed to display ventricular activation during sinus rhythm over eight isochrones. Analysis was performed at successful VT termination sites and prospectively tested. 33 patients with 47 scar-related VTs where a critical site was demonstrated by termination of VT during ablation were retrospectively analyzed. In those that underwent mapping of multiple surfaces, 90% of critical sites were on the surface that contained the latest late potential. However, only 11% of critical sites were localized to the latest isochrone (87.5–100%) of ventricular activation. The median percentage of latest activation at critical sites was 78% at a distance from the latest isochrone of 18 mm. Sites critical to reentry were harbored in regions with slow conduction velocity, where 3 isochrones were present within a 1 cm radius. 10 consecutive patients underwent ablation prospectively guided by ILAM, targeting concentric isochrones outside of the latest isochrone. Elimination of the targeted VT was achieved in 90%. Termination of VT was achieved in 6 patients at a mean ventricular activation percentage of 78%, with only 1 requiring ablation in the latest isochrone.
Conclusions
Late potentials identified in the latest isochrone of activation during sinus rhythm are infrequently correlated with successful ablation sites for VT. The targeting of slow conduction regions propagating into the latest zone of activation may be a novel and promising strategy for substrate modification.
Keywords: ablation, ventricular tachycardia, electroanatomic mapping
Introduction
Electroanatomic mapping (EAM) has become an essential tool for substrate-based ablation of scar-related ventricular tachycardia (VT). Structural voltage displays have served to guide ablation lesion sets targeted at transecting scar or encircling border zone tissue.1–4 As the majority of scar-related VT is hemodynamically untolerated, surrogates in sinus rhythm for sites critical to reentry are desirable and late potentials (LP) in sinus rhythm have become a common ablation target as they exhibit a high sensitivity, albeit with variable specificity. 5–7 A commonly employed strategy for VT ablation is the tagging and targeting of abnormal electrograms with local conduction delay on a voltage-based EAM. However, amongst all identified LP in a given substrate, it is not known which zone of late activation most commonly provides the substrate for reentry. In addition, LP may be unrelated to any VT circuits and may represent unnecessary ablation targets.
The purpose of the present study is to systematically evaluate the utility of functional propagation map displays during sinus rhythm by retrospectively creating isochronal late activation maps (ILAM) to correlate the sites critical to reentry with late activation zones. We hypothesized that the most delayed late potentials are not necessarily most the functionally specific for reentry. We prospectively evaluated an ablation strategy targeting regions of slow conduction velocity outside the latest isochrone of ventricular activation during sinus rhythm.
Methods
Patient Population
Retrospective analysis was performed in patients referred for ablation of scar-mediated VT between July 2009-November 2013 at the UCLA Medical Center. Patients with ischemic cardiomyopathy, nonischemic cardiomyopathy, and arrhythmogenic right ventricular cardiomyopathy (ARVC) were included. The diagnosis of ischemic cardiomyopathy (ICM) was established by prior history of infarction with Q waves, focal wall motion abnormality, or fixed perfusion defect correlating with coronary stenosis or prior coronary intervention. All ablation procedures were performed under general anesthesia. Epicardial mapping and ablation was left to the discretion of the operator and usually preferred with a history of previously failed endocardial ablation, or ECG and/or substrate suggestive of epicardial exit was present. The UCLA Medical Center institutional review board approved review of this data.
Electroanatomic Mapping and Ablation
The approach and strategy for ablation of scar-mediated VT at our center has been previously reported.8 Noninvasive programed stimulation was performed under light sedation to assess the morphology of VT and hemodynamic tolerance. Entrainment mapping was performed at the discretion of the operator when VT was hemodynamically tolerated. An isthmus was defined as a site that demonstrated concealed fusion during entrainment with a postpacing interval within 30 ms of the VT cycle length, where the stimulus to QRS interval was equal to EGM-QRS.9 In cases of unmappable VT, LP sites were tagged and pacemapping was performed and sites with multiple exit sites (MES) and pace-mapped induction (PMI) were considered isthmus surrogates.8 Induction of VT was performed with the ablation catheter positioned at these sites where immediate ablation was attempted upon VT initiation, particularly in cases of rapid or hemodynamically unstable VT seen during noninvasive programmed stimulation. High-density electroanatomic maps were created in sinus rhythm using CARTO (Biosense Webster, Diamond Bar, CA) or NAVX (St. Jude Medical, Minneapolis, MN) with standard low voltage settings (0.5–1.5mV). In the cohort of patients mapped using NAVX, field scaling was only applied in cases that resulted in a more spherical ventricular geometry.
RF ablation was performed using an open-irrigated catheter (ThermoCool or ThermCool SF, 3.5 mm, Biosense-Webster, Diamond Bar, CA) at 30–50W, temperature limit 45°C @ 30 ml flow rate or closed-loop irrigated catheter (Chili, Boston Scientific, Natick MA) at 30–50W, temperature limit 45°C. The temperature limit for epicardial RF applications was 50°C. RF energy was applied for 60 seconds for each application.
Isochronal Late Activation Mapping (ILAM)
ILAM were constructed retrospectively by manual annotation of all points for a given chamber and surface mapped acquired during sinus rhythm, with the investigator blinded to identified critical ablation sites. Each electrogram was timed at the offset of the local bipolar electrogram deflection, signifying the completion of local activation. The offset was chosen due to a higher degree of reproducibility and less inter-observer variability than the onset, maximum dV/dT, or amplitude of a LP, which are more arbitrary and subjective at sites with continuous and fractionated activity. (FIGURE 1). Electrogram deflections with an amplitude lower than the baseline noise and/or were not reproducibly seen on preceding beats were disregarded with annotation. The timing of all electrograms comprising an ILAM were confirmed by two additional observers.
Figure 1.

Manual annotation of electrograms during sinus rhythm to the offset of the local electrogram for ILAM construction. In electrograms with continuous and fractionated activity, determination of the onset or maximum slope is subject to more interpretation than return to baseline.
ILAM were displayed with eight equally distributed isochrones of activation (12.5% of ventricular activation comprised each isochrone). This was chosen due to the difficulty in interpreting maps when settings of >8 were assessed, which obscured distinct isochronal contours. With the CARTO system, the entire window was divided by 8 to determine the duration of each isochronal step and using the NAVX system (St. Jude Medical, Minneapolis, MN) the isochronal (LAT) settings were similarly selected with a color contour setting of 1. In maps where combined epicardial and endocardial mapping was performed, or right ventricle was combined with left ventricular activation, the largest window that incorporated the timing of both surfaces or chambers was chosen.
The activation within scar were categorized as either a centripetal (from outside into latest isochrone of activation) or a broad wavefront pattern.10 The number of spatially distinct latest regions of activation was also recorded and compared to the number of VTs induced during the ablation procedure. The offset of the surface QRS complex relative to the ILAM was measured to analyze the proportion of critical sites with late local activation after the QRS complex.
Characterization of critical sites
A site critical for reentry was defined as a site with an electrogram that was mapped within or adjacent to low voltage regions (<1.5mV) that exhibited diastolic activity during VT with termination of VT during ablation. Terminations of VT that occurred with ventricular ectopy were excluded to improve specificity. Additional confirmation of sites critical for reentry included isthmus response to entrainment, pacemap match with stimulus-QRS >30ms, multiple exit site response during pacemapping and/or a pacemapped induction of VT.8, 9, 11, 12
At each critical site, the voltage and electrogram timing was recorded during sinus rhythm. Due to potential map localization discrepancies present between sinus rhythm and VT, these measurements were made from the critical electrogram site during sinus rhythm if the catheter was not moved after induction of VT. In cases where mapping during VT was performed, the termination site was annotated on the map immediately upon termination in sinus rhythm. To account for changes in voltage and timing from the immediate ablation, the critical site was characterized by averaging the three closest neighboring points on the ILAM (consists of mapping points prior to ablation only). Cases of incessant VT (n=5) were excluded due to ablation necessarily performed prior to sinus rhythm mapping.
The ventricular activation percentage (VAP) at each critical site was calculated as the timing of the local electrogram offset in sinus rhythm divided by the entire window of activation mapped. The distance between the critical site from the center of the latest isochrone was calculated using the incorporated measurement tool in the respective EAM system and the number of isochrones present within a 1 cm radius of the critical site was counted to estimate the extent of conduction velocity slowing. Isochronal crowding was defined as local deceleration of propagation in a region within scar with bunching of isochrones (>2 isochrones within 1 cm radius).
Prospective evaluation: Ablation guided by ILAM
10 consecutive patients with scar-related VT underwent ablation prospectively guided by ILAM between April 2014-August 2014. Annotation of all electrograms was performed in real-time and the ILAM was displayed on a split screen with the voltage map. The ablation strategy targeted regions with concentric isochrones with crowding or “bunching” outside of the latest isochrone of activation. The second (75–87.5%) and third latest (62.5–75%) isochrones were preferentially ablated with circumferential lesion sets until LPs and abnormal electrograms were modified or eliminated. In cases of hemodynamically unstable VT as suggested by NIPS during light sedation, induction of VT was performed from the best pacemap site or MES outside the latest isochrone of activation with the immediate ablation upon induction. In patients with termination of VT, characterization of the critical sites was performed in the same manner as retrospective cases. Consolidation lesions were delivered around the critical sites. Repeat induction was performed after substrate modification outside of the latest isochrone by decremental burst pacing starting at 600 ms down to 300 ms and extrastimulus testing at two drive cycles (400, 500ms) from up to three sites using up to quadruple extrastimuli to 200 ms or ventricular refractoriness.
Statistical Analysis
Categorical variable are expressed as numbers and percentages, and were compared using chi-square or Fisher’s exact tests. All continuous data are reported as medians with 25%–75% interquartile percentiles due to the small sample size with non-normal distribution. Wilcoxon rank-sum test (Mann-Whitney) was used to estimate differences between two groups and the Kruskal-Wallis test was used to analyze for differences between three groups. For statistical comparisons, only one critical site for the first VT terminated was compared in patients with multiple critical sites to avoid bias and overrepresentation from any given patient. A p value of <0.05 was statistically significant.
Results
Between July 2009-November 2013, 178 patients underwent ablation for scar-related VT. Amongst these cases, 33 patients with 47 critical sites for VT were identified and met criteria for VT termination seen during the ablation at a critical site that could be retrospectively analyzed.
The etiology of cardiomyopathy was ICM in 64%, NICM in 30%, and ARVC in 6%. Scar was located in the left ventricular anterior wall in 11 (33%), septum in 12 (36%), inferior wall in 20 (61%), lateral wall in 16 (48%), and 5 (15%) had right ventricular scar. The median EF was 30% (IQR 19%–39%) and 48% underwent prior ablation. 97% had ICD previously implanted and 85% were taking antiarrhythmic medications (57% amiodarone). Epicardial mapping was performed in 48% (16/33, 2 with epicardial only) and biventricular mapping was performed in 21%(7/33). Patients with NICM underwent epicardial mapping more frequently (75% vs 33%, p=0.021) and biventricular mapping less frequently than patients with ICM (0% vs 33%, p=0.027). (TABLE 1)
Table 1.
Baseline patient characteristics
| Retrospective (n=33) | Prospective (n=10) | p value | |
|---|---|---|---|
| Age (y) | 68 (57–73) | 73 (62–80) | 0.18 |
| Male | 31 (94%) | 9 (90%) | 0.85 |
| Ejection fraction (%) | 30 (19–39) | 28 (16–39) | 0.49 |
| Antiarrhythmic drug | 28 (85%) | 7 (70%) | 0.49 |
| Amiodarone | 16 (57%) | 6 (60%) | 0.60 |
| ICD | 32 (97%) | 9 (90%) | 0.75 |
| Prior ablation | 16 (48%) | 3 (30%) | 0.39 |
| Mapping system (Carto/Navx) | 17/16 | 7/3 | 0.39 |
| Baseline QRS width (ms) | 150 (110–185) | 183 (154–192) | 0.24 |
| Biventricular mapping | 7 (21%) | 2 (20%) | 0.97 |
| Epicardial mapping | 16 (48%) | 3 (30%) | 0.39 |
Medians (IQR) or percentage are reported.
ILAM maps (median RV endocardial 136 mapping points (86–267), LV endo 325 mapping points (254–468), and epicardial 613 mapping points (420–995) were constructed retrospectively using CARTO in 52% and NAVX in 48% of patients. 67% of ILAM were acquired with intrinsic QRS (133 ms, IQR 108–150ms), 27% were paced from RV (QRS width 191 ms, IQR 185–214ms), and 6% were mapped during biventricular pacing (QRS width 200 ms, IQR 189–211). (TABLE 2) When comparing patients who underwent mapping during intrinsic vs paced QRS, there were no statistically significant differences in the duration of the entire window of ventricular activation that comprised the ILAM (160 ms vs 198 ms, p=0.053) or the post-QRS window of ventricular activation mapped (77 ms vs 67 ms, p=0.418).
Table 2.
Procedural and ILAM characteristics
| Retrospective (n=33) | Prospective (n=6) | p value | |
|---|---|---|---|
| Intrinsic/Paced rhythm | 22/11 | 3/3 | 0.53 |
| Number of induced VTs | 4 (2–4) | 2 (1,4) | 0.53 |
| Number of VT termination sites | 1 (1,2) | 1 (1,1) | 0.21 |
| VT cycle length (ms) | 426 (360–489) | 435 (368–488) | 0.92 |
| Number of EAM points (n=retro/pro) | 512 (312–985) | 554 (341–631) | 0.51 |
| LV (n=28/6) | 325 (254–468) | 261 (203–275) | 0.17 |
| RV (n=10/0) | 136 (86,267) | - | - |
| Epicardium (n=16/3) | 613 (420–995) | 389 (385–415) | 0.11 |
| VAP of critical site (% of window) | 78 (64–83) | 78 (69–86) | 0.86 |
| EGM-QRS/TCL (%) | 16 (9–33) | 44 (24–47) | 0.03 |
| Distance between critical site and latest isochrone (mm) | 18 (7–31) | 13 (10–15) | 0.44 |
| Isochrones with 1cm of critical site | 3 (3,4) | 3.5 (2–5) | 0.99 |
Medians (IQR) or percentage are reported. In patients with multiple critical sites (n=11), only the first site was used for statistical comparison.
A centripetal pattern of activation with concentric isochrones surrounding the latest isochrone was seen in 91% of patients, whereas a broad wavefront was seen in 9%. There was median of 1 (IQR 1–2) distinct latest isochronal region in each patient and this did not correlate with the median number of 4 VTs induced per patient (range 1–7 VTs, p=0.32). 61% of patients had only one latest isochronal region, 27% had two, and 12% had three spatially distinct latest isochronal regions. The median VT cycle length of the first VT terminated was 426 ms (IQR 360–489 ms) and there was no correlation between VT cycle length and the duration of total ventricular activation duration (R2=0.063, p=0.08) or with the post-QRS window duration (R2=0.001, p=0.8).
Location and characteristics of critical sites
Termination of VT was achieved on the endocardial surface in 68% (32/47) and from the epicardium in 32% (15/47). The substrates with epicardial termination were 5 ICM, 9 NICM, and 1 ARVC. Entrainment with concealed fusion was demonstrated in 30% (14/47) of sites. MES was observed at 26% (12/47) and PMI was seen in 36% (17/47).
In 23 patients that underwent mapping of multiple chambers or surfaces, 90% (26/29) of critical sites for VT were harbored in the chamber or surface with the most delayed activation. For combined epi-endo mapping, 87% (20/23) of critical sites for VT were localized to the latest mapped surface (58% epi, 42% endo). In patients that underwent biventricular mapping (RV and LV), the critical site was located in the ventricle that contained the latest isochrone in 100% of cases (6/6). FIGURE 2 shows a biventricular map in a patient with inferior myocardial infarction with extensive right ventricular involvement with two induced VTs. The termination sites are in the second latest isochrone in the right ventricle, which contains the latest isochrone at the basal inferior wall.
Figure 2.
Patient with inferior myocardial infarction with extensive right ventricular involvement with biventricular dense scar. The ILAM demonstrates that the latest isochrone of combined activation is in the basal inferior region in the right ventricle. Two VTs with left bundle branch morphology were terminated in the second latest isochrone of sinus rhythm activation (VAP of 75% and 79%) in the right ventricle.
The median voltage at critical sites was 0.47 mV (IQR 0.17–1.02 ms). There was a trend in critical site location within dense scar in 47% (<0.5mV) compared to 34% within border zone tissue, although this difference was not statistically significant (p=0.4). Nine sites (19%) were in relatively preserved voltage (>1.5mV) and 6 of these were thought be intramural due to equal epicardial and endocardial timing with delayed termination during ablation.
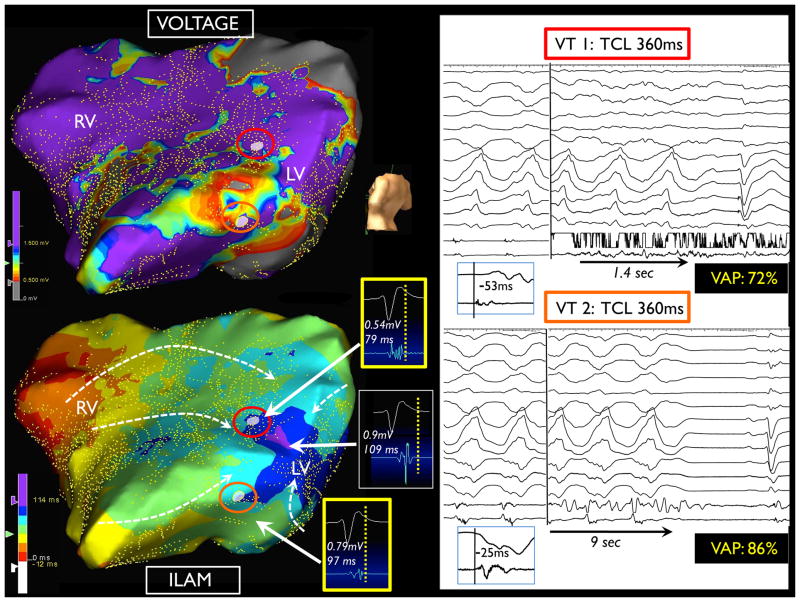
By ILAM analysis, the median VAP at critical sites was 78%. Only 11% (5/47) of critical sites were localized to the latest isochrone (87.5–100%) of activation. The 2nd latest isochrone of activation (75–87.5%) was the most common zone where critical sites were harbored in 36% (17/47) (compared to latest isochrone, p=0.01), followed by the 3rd latest (62.5–75%) isochrones with 28% (13/47) of critical sites (SEE FIGURE 3). 19% (9/47) of critical sites were located within the QRS window while 81% (38/47) had an offset after the QRS. Six presumed intramural sites had a median VAP of 50%. FIGURE 4 shows a voltage map and ILAM in a patient with NICM and ejection fraction of 15% who presented with VT storm. Due to the presence of an LV thrombus, only epicardial mapping and ablation was performed. Two VTs were terminated in the lateral wall of the left ventricle in the second and third latest isochrone of sinus rhythm activation.
Figure 3.
Distribution of sites critical to reentry by ventricular activation percentage in ILAM. The latest isochrone of activation was least frequently associated with a successful ablation site.
Figure 4.
ILAM and voltage map in a patient with NICM and ejection fraction of 15% with VT storm. A patchy lateral wall scar is seen in the left ventricle and the termination sites for the two induced VTs were localized to the second and third latest isochrone (VAP 72% and 86%).
The median distance between critical sites and the center of the latest isochrone was 18 mm (IQR 7–31 mms). Only 15 sites (32%) were within 10mm. 24 sites (51%) were >2 cm and 10 (21%) >5 cm away from the latest zone of activation on the ILAM. At critical sites, there was a median of 3 isochrones (range 2–6) isochrones seen within a 1 cm radius (~38 % of the total ventricular activation). FIGURE 5 shows an example of a patient with NICM with later activation during sinus rhythm in the endocardium than epicardium, despite less extensive and less dense scar. The successful termination site occurs at a region with isochronal crowding where 3 isochrones are seen with a 1cm radius of the critical site.
Figure 5.
ILAM and voltage map in a patient with NICM and ejection fraction of 30% with VT storm that demonstrates for extensive scarring on the epicardium compared to the endocardium. The latest zone of activation is seen in the endocardium and the critical site is located in the thinnest portion of second latest isochrone where 3 isochrones are seen within a 1 cm radius.
Potential influential factors on the ventricular activation percentage
There was no significant difference in the median VAP of critical sites between patients that were mapped during intrinsic rhythm vs paced (78% vs 79%, p=0.59). There were no differences in the VAP of critical sites when analyzed by anatomic location comparing apical to basal sites and septal to lateral sites. (FIGURE 6). When comparing slow VT (TCL>400ms) with faster VT (TCL<400ms), no differences in the VAP of the critical site was observed (79% vs 78%, p=0.9).
Figure 6.
Potential factors that may influence ventricular activation percentage of critical sites include A) intrinsic vs paced complex B) diastolic activation time during VT C) anatomic location in septal to lateral dimension D) anatomic location in apical to basal dimension. Sites in the mid-isthmus had a later VAP compared to sites in the distal isthmus closer to the exit. The anatomic location of the critical site did not significantly influence the VAP.
The median EGM-QRS during VT at critical sites was 76ms (IQR 45–115ms), which represents 16% of the TCL. When analyzing sites located in the mid-isthmus (30–70%) vs distal isthmus (<30% TCL), the VAP was later at mid-isthmus compared to distal sites (81% vs 69%, p=0.06). Among 8 critical sites located in the mid isthmus, 7 (88%) were found within the 62.5–87.5% isochrones. (FIGURE 6)
Prospective cases
10 cases (ICM n=5) were performed prospectively using ILAM to guide ablation. Patient characteristics are shown in TABLE 1. The scar location was LV anterior in 2 (20%), LV septum in 3 (30%), LV inferior in 6 (60%), LV lateral in 4 (40%), and 1 (10%) in the right ventricle. Termination of VT during ablation (median 4.5 seconds) was achieved in 6 of these cases, where 4 were hemodynamically unstable.
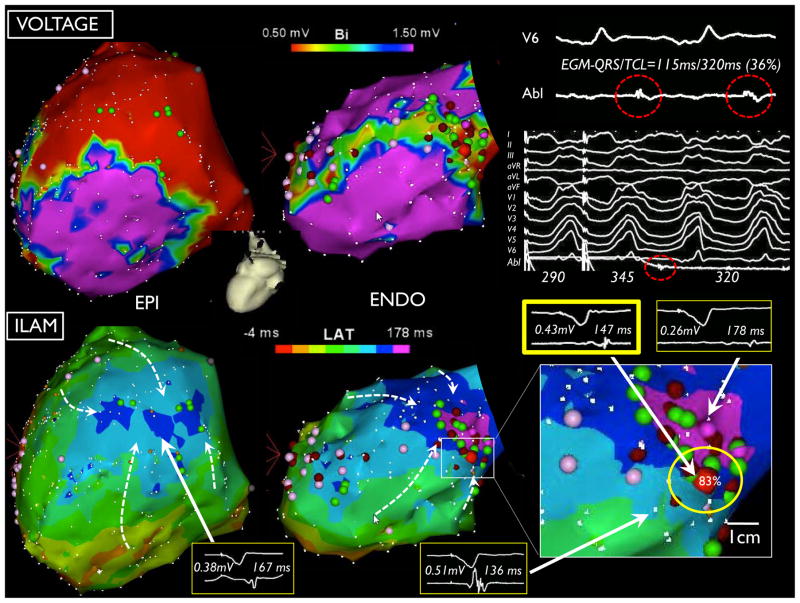
Amongst these 6 VT terminations, 5 achieved termination within the first two radiofrequency applications. 3 patients (NICM=2, Chagas=1) underwent combined epicardial-endocardial mapping. The critical sites in these patients were located on the endocardium and the endocardial ILAM window was more delayed than the epicardial window in all three. In 3 cases, ILAM were created using multiple wavefronts of activation (RV and LV) and none of the critical sites were in the latest isochrones of activation. (FIGURE 7)
Figure 7.
Prospective case of inferoseptal patchy infarct with rapid, untolerated VT localized by pacemapping with induction outside of the latest isochrone of activation. Termination of VT was achieved at this exact site, which was not in the latest region of activation in both sinus rhythm and a lateral pacing wavefront. Ablation lesions are shown around the termination site and circumferentially outside the latest isochrone of activation.
The median VAP at critical sites was 78%. Procedural characteristics of patients with VT termination are shown in TABLE 2. Only one of these termination sites occurred within the latest isochrone of activation (VAP 89%) and this had the latest termination of 42 seconds. A passive bystander was demonstrated in the latest isochrones in a patient with inferior myocardial infarction (FIGURE 8). There were 3.5 isochrones within 1 cm of the successful site and the distance from the critical site from the latest isochrone was 13 mm (IQR 10–16 mm).
Figure 8.
Prospective case of extensive dense scar from inferior infarction. ILAM demonstrates centripetal activation into the latest region of activation (purple isochrones). The latest LP was a bystander with 2:1 passive activation during VT 1 with long stimulus-QRS interval during sinus rhythm and VT2 was successfully entrained and terminated outside the latest isochrone (green dot).
Homogenization targeting the regions outside the latest isochrone in areas of isochronal crowding was performed in all cases. Complete noninducibility was achieved in 40%, elimination of the clinical VT in 50%, and one patient was not tested postablation. At 6±1 months of follow-up, 80% of the patients remained free of VT recurrence, with 50% off antiarrhythmic therapy.
Discussion
The major findings of the present study are:
Sites critical for reentry are frequently identified outside the latest isochrone of ventricular activation during sinus rhythm but are commonly localized to the surface or chamber mapped with the most delayed activation.
Regions of conduction slowing with isochronal crowding that propagate into the latest zone of activation can be identified and targeted with functional mapping displays to eliminate VT.
As most LPs mapped are merely “tagged” on scar voltage maps in clinical practice, this is the first analysis, to our knowledge, to correlate sites critical for scar-related VT with isochronal activation patterns during sinus rhythm in humans.13 It has been previously demonstrated that the targeting of earlier late potentials frequently results in elimination of delayed activation downstream in a channel 14, 15, but the functional significance of these upstream regions during VT has not been systematically evaluated. An understanding of whether a specific late activation zone identified during sinus rhythm has a higher yield for VT elimination may have an immediate impact on substrate ablation strategies.
Since the advent of EAM systems, ablation of VT has evolved from anatomically-based strategies to targeting functional regions of local conduction slowing within scar. Guided by pacemapping, linear lesions sets across dense scar and at border zones have been shown to reduce VT recurrence.1–4, 12 The relationship of critical sites to voltage-based scar anatomy has been previously reported. While Hsia et al16 found that 84% of entrance and mid-isthmus sites were localized to dense scar, Verma et al17 reported that 68% of critical sites were localized to border zone in ICM patients.
Arenal et al described that adjustment of voltage thresholds to more dense settings exposed potential isthmus regions with relatively preserved conducting channels.18 However, Mountantonakis et al demonstrated that channels exposed by threshold adjustments were poorly predictive of isthmuses, suggesting that the macroscopic identification of a channel by structure alone is not always feasible.19 Jamil-Copley et al more recently showed the potential utility of ripple mapping which demonstrates both voltage and propagation channels into scar in a single dynamic display.20 This technology has the advantage of displaying complex or continuous fractionated local electrograms as time-dependent propagation, rather than choosing a single annotation for timing.
As reentrant VT requires fixed and/or functional localized conduction slowing, Brunckhorst et al demonstrated with multiple wavefront mapping that regions with fixed and functional conduction delay may be equally critical for VT maintenance.21 Jais et al further advanced the concept that abnormal multicomponent electrograms within the QRS during sinus rhythm can be functionally delayed with extrastimuli due to local uncoupling.22 In the present study, we demonstrate that the majority of sites critical to reentry exhibit a certain extent of fixed conduction slowing during sinus rhythm with isochronal crowding and electrogram components outside of the QRS, although “intra-QRS” LPs comprised 19% of the cohort. For this reason, ILAM analysis incorporates the entire ventricular window of activation, rather than the post-QRS window alone.
The present findings suggest that an adjunctive EAM display can complement current ablation strategies based on a functional prioritization of activation within scar. Mechanistically, it appears that the latest LPs may not be the most specific and therefore may not require targeted ablation. Zones with the most delayed activation during sinus rhythm may represent regions that are extraneous or proximal to the circuit during VT (entrance, inner loop sites) or bystander sites. Within a given isochrone, the thinnest portion represents regions with the slowest conduction velocity and therefore localized areas of isochronal crowding or “bunching” signify the greatest extent of fixed conduction slowing during sinus rhythm.
Assadi et al first examined the relationship of reentrant isthmus sites with isochronal sinus activation mapping in a postinfarct animal model.13 In >50% of cases, the critical sites were at least 24 mm away from the latest isochrone of activation. These observations were limited to epicardial mapping and were not extended to humans. The present analysis is consistent with these findings, where critical sites are frequently remote from the latest LPs. Importantly, the relationship between regions of slow conduction during sinus rhythm are not expected to mirror regions of slow conduction during VT as functional lines of block develop during a different reentrant wavefront. The pattern and direction of the reentrant wavefront is critically dependent upon local refractory periods, which is not incorporated in the current ILAM analysis.
The centripetal pattern of activation of scar seen with ILAM, from the periphery to the center, has mechanistic and clinical implications. Haqqani et al described a similar propagation in ARVC patients with epicardial scar and hypothesized independent layered activation in the endocardium and epicardial.10 The present findings suggest that this pattern of scar activation is observed in the majority of patients. The centripetal activation of channels also supports that a circumferential lesion set has the potential to electrically isolate the center of late activation within concentric isochrones.
In the present study, the number of regions within scar with the most delayed activation did not correlate with the number of VTs induced. This is consistent with shared isthmuses that may support different VT morphologies and that the targeting of one zone of late activation may simultaneously eliminate non-targeted VTs.6 Additionally, there was no relationship between the VT cycle length and indices of conduction slowing derived from ILAM (i.e. total ventricular activation time, post-QRS windown, or isochrone percentage). This is consistent with regions with less evident slow conduction during sinus rhythm developing a greater degree of functional slowing along the arc of unidirectional block during VT.
Limitations
The generalizability of this study is limited by the relatively small sample size of patients. Importantly, ILAM displays do not actually represent activation into each electrogram site, but rather the completion of activation, as we specifically chose the offset of the last deflection back to the baseline to optimize reproducibility and minimize subjectivity. This is largely due to the lack of standardized criteria for distinguishing near-field potentials from far-field activity. Inter-observer variability is likely to increase when assigning the onset, maximum, or highest frequency deflection of a continuous or fractionated signal.
Studies evaluating LP in sinus rhythm all represent fixed conduction slowing with one directional wavefront of activation. Additionally, the anatomic location of scar influences the relative timing of the near and far field components of a local electrogram. The wide distribution of VAP at critical sites across septal and lateral regions and apical and basal regions observed in this cohort suggests that in the presence of dense scar, the latest isochrones activated through centripetal propagation into scar with concentric isochrones may not significantly vary with the wavefront of activation.
The appearance of ILAM is sensitive to the latest electrogram identified. The value and relative timing of a site during map creation cannot be assessed until the entire ventricle is mapped. The failure to map a later activation site for a given ILAM does not necessarily mean that a later site of activation is not present. Therefore, in the cases where the critical sites were found in the latest isochrone, later sites of activation not identified on the same surface or present transmurally are possible. Isochronal thickness was not measured due to inaccuracies that are present from interpolation.
Termination of VT can theoretically be achieved at different components of the reentrant circuit. It is not conclusive from the present data that termination cannot be achieved in the latest isochrone, but rather that termination is not commonly observed in the regions of most delayed activation in this study. Entrainment mapping was not performed in all cases, but in the real-world, “clean” termination of VT remains an accepted proof of a site critical to the maintenance of reentry when diastolic activity is recorded. In patients with multiple VTs, the exact localization of the critical sites for all morphologies could not be ascertained due to the fact that the majority of VTs are untolerated and variably inducible during ablation.
Conclusions
Late potentials in the latest isochrone of activation during sinus rhythm are infrequently correlated with successful ablation sites for VT. The latest late potentials may be bystander sites during reentry. Slow conduction regions with isochronal crowding propagating into the latest zone of activation may be a promising ablation target for substrate modification. ILAM during sinus rhythm may provide complementary and targeted functional information beyond structural voltage displays for scar-related VT and represents a novel strategy for substrate-based VT ablation.
Acknowledgments
We would like to thank Rich Nagel, Steve Kim, and Heather Macken for their assistance with analysis of EAM data. We appreciate the statistical guidance and review of Chi-Hong Tseng PhD. We would also like to thank William Sauer MD and Srijoy Mahapatra MD for their careful review of the manuscript.
Funding Sources: Supported by the NHLBI (R01HL084261 to KS)
Footnotes
Conflict of Interest Disclosures: None
References
- 1.Soejima K, Suzuki M, Maisel WH, Brunckhorst CB, Delacretaz E, Blier L, Tung S, Khan H, Stevenson WG. Catheter ablation in patients with multiple and unstable ventricular tachycardias after myocardial infarction: short ablation lines guided by reentry circuit isthmuses and sinus rhythm mapping. Circulation. 2001;104:664–669. doi: 10.1161/hc3101.093764. [DOI] [PubMed] [Google Scholar]
- 2.Marchlinski FE, Callans DJ, Gottlieb CD, Zado E. Linear ablation lesions for control of unmappable ventricular tachycardia in patients with ischemic and nonischemic cardiomyopathy. Circulation. 2000;101:1288–1296. doi: 10.1161/01.cir.101.11.1288. [DOI] [PubMed] [Google Scholar]
- 3.Reddy VY, Reynolds MR, Neuzil P, Richardson AW, Taborsky M, Jongnarangsin K, Kralovec S, Sediva L, Ruskin JN, Josephson ME. Prophylactic catheter ablation for the prevention of defibrillator therapy. N Engl J Med. 2007;357:2657–2665. doi: 10.1056/NEJMoa065457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Tanner H, Hindricks G, Volkmer M, Furniss S, Kuhlkamp V, Lacroix D, CDEC, Almendral J, Caponi D, Kuck KH, Kottkamp H. Catheter ablation of recurrent scar-related ventricular tachycardia using electroanatomical mapping and irrigated ablation technology: results of the prospective multicenter Euro-VT-study. J Cardiovasc Electrophysiol. 2010;21:47–53. doi: 10.1111/j.1540-8167.2009.01563.x. [DOI] [PubMed] [Google Scholar]
- 5.Arenal A, Glez-Torrecilla E, Ortiz M, Villacastin J, Fdez-Portales J, Sousa E, del Castillo S, Perez de Isla L, Jimenez J, Almendral J. Ablation of electrograms with an isolated, delayed component as treatment of unmappable monomorphic ventricular tachycardias in patients with structural heart disease. J Am Coll Cardiol. 2003;41:81–92. doi: 10.1016/s0735-1097(02)02623-2. [DOI] [PubMed] [Google Scholar]
- 6.Bogun F, Good E, Reich S, Elmouchi D, Igic P, Lemola K, Tschopp D, Jongnarangsin K, Oral H, Chugh A, Pelosi F, Morady F. Isolated potentials during sinus rhythm and pace-mapping within scars as guides for ablation of post-infarction ventricular tachycardia. J Am Coll Cardiol. 2006;47:2013–2019. doi: 10.1016/j.jacc.2005.12.062. [DOI] [PubMed] [Google Scholar]
- 7.Bogun F, Bender B, Li YG, Groenefeld G, Hohnloser SH, Pelosi F, Knight B, Strickberger SA, Morady F. Analysis during sinus rhythm of critical sites in reentry circuits of postinfarction ventricular tachycardia. J Interv Card Electrophysiol. 2002;7:95–103. doi: 10.1023/a:1020832502838. [DOI] [PubMed] [Google Scholar]
- 8.Tung R, Mathuria N, Michowitz Y, Yu R, Buch E, Bradfield J, Mandapati R, Wiener I, Boyle N, Shivkumar K. Functional pace-mapping responses for identification of targets for catheter ablation of scar-mediated ventricular tachycardia. Circ Arrhythm Electrophysiol. 2012;5:264–272. doi: 10.1161/CIRCEP.111.967976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Stevenson WG, Friedman PL, Sager PT, Saxon LA, Kocovic D, Harada T, Wiener I, Khan H. Exploring postinfarction reentrant ventricular tachycardia with entrainment mapping. J Am Coll Cardiol. 1997;29:1180–1189. doi: 10.1016/s0735-1097(97)00065-x. [DOI] [PubMed] [Google Scholar]
- 10.Haqqani HM, Tschabrunn CM, Betensky BP, Lavi N, Tzou WS, Zado ES, Marchlinski FE. Layered activation of epicardial scar in arrhythmogenic right ventricular dysplasia: possible substrate for confined epicardial circuits. Circ Arrhythm Electrophysiol. 2012;5:796–803. doi: 10.1161/CIRCEP.111.967935. [DOI] [PubMed] [Google Scholar]
- 11.Stevenson WG, Khan H, Sager P, Saxon LA, Middlekauff HR, Natterson PD, Wiener I. Identification of reentry circuit sites during catheter mapping and radiofrequency ablation of ventricular tachycardia late after myocardial infarction. Circulation. 1993;88:1647–1670. doi: 10.1161/01.cir.88.4.1647. [DOI] [PubMed] [Google Scholar]
- 12.Stevenson WG, Sager PT, Natterson PD, Saxon LA, Middlekauff HR, Wiener I. Relation of pace mapping QRS configuration and conduction delay to ventricular tachycardia reentry circuits in human infarct scars. J Am Coll Cardiol. 1995;26:481–488. doi: 10.1016/0735-1097(95)80026-d. [DOI] [PubMed] [Google Scholar]
- 13.Assadi M, Restivo M, Gough WB, el-Sherif N. Reentrant ventricular arrhythmias in the late myocardial infarction period: 17. Correlation of activation patterns of sinus and reentrant ventricular tachycardia. Am Heart J. 1990;119:1014–1024. doi: 10.1016/s0002-8703(05)80230-3. [DOI] [PubMed] [Google Scholar]
- 14.Berruezo A, Fernandez-Armenta J, Mont L, Zeljko H, Andreu D, Herczku C, Boussy T, Tolosana JM, Arbelo E, Brugada J. Combined endocardial and epicardial catheter ablation in arrhythmogenic right ventricular dysplasia incorporating scar dechanneling technique. Circ Arrhythm Electrophysiol. 2012;5:111–121. doi: 10.1161/CIRCEP.110.960740. [DOI] [PubMed] [Google Scholar]
- 15.Tung R, Mathuria NS, Nagel R, Mandapati R, Buch EF, Bradfield JS, Vaseghi M, Boyle NG, Shivkumar K. Impact of local ablation on interconnected channels within ventricular scar: mechanistic implications for substrate modification. Circ Arrhythm Electrophysiol. 2013;6:1131–1138. doi: 10.1161/CIRCEP.113.000867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hsia HH, Lin D, Sauer WH, Callans DJ, Marchlinski FE. Anatomic characterization of endocardial substrate for hemodynamically stable reentrant ventricular tachycardia: identification of endocardial conducting channels. Heart Rhythm. 2006;3:503–512. doi: 10.1016/j.hrthm.2006.01.015. [DOI] [PubMed] [Google Scholar]
- 17.Verma A, Marrouche NF, Schweikert RA, Saliba W, Wazni O, Cummings J, Abdul-Karim A, Bhargava M, Burkhardt JD, Kilicaslan F, Martin DO, Natale A. Relationship between successful ablation sites and the scar border zone defined by substrate mapping for ventricular tachycardia post-myocardial infarction. J Cardiovasc Electrophysiol. 2005;16:465–471. doi: 10.1046/j.1540-8167.2005.40443.x. [DOI] [PubMed] [Google Scholar]
- 18.Arya A, Eitel C, Bollmann A, Wetzel U, Sommer P, Gaspar T, Husser D, Piorkowski C, Hindricks G. Catheter ablation of scar-related ventricular tachycardia in patients with electrical storm using remote magnetic catheter navigation. Pacing Clin Electrophysiol. 2010;33:1312–1318. doi: 10.1111/j.1540-8159.2010.02818.x. [DOI] [PubMed] [Google Scholar]
- 19.Mountantonakis SE, Park RE, Frankel DS, Hutchinson MD, Dixit S, Cooper J, Callans D, Marchlinski FE, Gerstenfeld EP. Relationship between voltage map “channels” and the location of critical isthmus sites in patients with post-infarction cardiomyopathy and ventricular tachycardia. J Am Coll Cardiol. 2013;61:2088–2095. doi: 10.1016/j.jacc.2013.02.031. [DOI] [PubMed] [Google Scholar]
- 20.Jamil-Copley S, Vergara P, Carbucicchio C, Linton N, Koa-Wing M, Luther V, Francis DP, Peters NS, Davies DW, Tondo C, Della Bella P, Kanagaratnam P. Application of Ripple Mapping to Visualise Slow Conduction Channels Within the Infarct-Related Left Ventricular Scar. Circ Arrhythm Electrophysiol. 2015;8:76–86. doi: 10.1161/CIRCEP.114.001827. [DOI] [PubMed] [Google Scholar]
- 21.Brunckhorst CB, Stevenson WG, Jackman WM, Kuck KH, Soejima K, Nakagawa H, Cappato R, Ben-Haim SA. Ventricular mapping during atrial and ventricular pacing. Relationship of multipotential electrograms to ventricular tachycardia reentry circuits after myocardial infarction. Eur Heart J. 2002;23:1131–1138. doi: 10.1053/euhj.2001.3110. [DOI] [PubMed] [Google Scholar]
- 22.Jais P, Maury P, Khairy P, Sacher F, Nault I, Komatsu Y, Hocini M, Forclaz A, Jadidi AS, Weerasooryia R, Shah A, Derval N, Cochet H, Knecht S, Miyazaki S, Linton N, Rivard L, Wright M, Wilton SB, Scherr D, Pascale P, Roten L, Pederson M, Bordachar P, Laurent F, Kim SJ, Ritter P, Clementy J, Haissaguerre M. Elimination of local abnormal ventricular activities: a new end point for substrate modification in patients with scar-related ventricular tachycardia. Circulation. 2012;125:2184–2196. doi: 10.1161/CIRCULATIONAHA.111.043216. [DOI] [PubMed] [Google Scholar]