INTRODUCTION
The main component of a traditional Chinese drug “Pishuang”, arsenic trioxide (As2O3), has obviously selective anti-tumor effect on human hepatocellular carcinoma (HCC) in both in vitro and in vivo studies[1-5]. Due to limited effectiveness when any anti-carcinogen is used alone and obviously increased toxicity when the dose is raised, there is no exception for As2O3. Furthermore, combined chemotherapy contributes to improve therapeutic effectiveness, disperse toxicity and surmount drug-resistance, in which the combination of traditional Chinese and modern medicine has more advantages and characteristics. As a result, we made an experimental study on anti-tumor effect of As2O3 in combination with cisplantin (PDD) or doxorubicin (ADM) on HCC, to investigate the possibility of As2O3 in combination with PDD or ADM and nature of interaction between them, and to provide experimental basis for clinical application.
MATERIALS AND METHODS
Materials
Drugs and reagents As2O3 for injection (5 mg per ampoul, Lot No. 998068, provided by Professor Ma Jun of Harbin Hamaeotoloy and Oncolony Institute), PDD for injection (20 mg per vial, Lot No. 990618, Shandong Qilu Pharmaceutical Factory) or ADM hydrochloride for injection (10 mg per vial, Lot 990406, Shanxi Mlarmaceutical Co. LTD).
Cell lines Human hepatoma Bel-7402 cells were obtained from the Shanghai Cell Bank of Chinese Academy of Sciences and maintained in our laboratory. Bel-7402 cells were routinely cultured in RPMI1640 medium (Gibco) containing 100 mL•L¯¹ fetal calf (FCS) serum at 37 °C in humidified incubator with 50 mL•L¯¹ CO2/95 mL•L¯¹ air.
Animals Mice with hepatoma HepA were obtained from the Shanghai Institute of Materia Medica of Chinese Academy of Sciences and Kunming mice (famale and male weighing, 18 g-22 g) from the Experimental Animal Center of Southerneast University Medical College.
METHODS
Measurement of anticancer activity in vitro The exponent growing Bel-7402 cells in culture flasks were harvested by 2.5 g·L¯¹ EDTA, suspended in RPMI1640 medium with 100 mL•L¯¹ FCS, adjusted to the concentration of 3 × 104 cells•L¯¹, plated into 40-well plates (100 μL cells•well-1) and incubated at 37 °C in 50 mL•L¯¹ CO2/95 mL•L¯¹ air until the cells were stuck with the plates. The cells were then exposed to 100 μL of various concentrations of a drug alone or combination for 48 h, and the controls to 100 μL of RPMI1640 medium with no FCS. After that, the absorption was detected by adding 20 μL tetrazolium (MTT) to each well, incubating for 4 h, sucking out the media, adding 150 μL dimethylsul foxide (DMSO) to dissolve the violet-crystal and measuring at 570 nm. Double wells were used for each drug concentration. Experiments were triplicated. The inhibitory rate was calculated as follows:
IR (%) = [1 - (mean absorption in experiments/mean absorption in controls)] × 100%
Measurement of anticancer activity in vivo The mice with hepatocarcinoma HepA were killed and their ascites Abstracted, adjusted to 2 × 107•mL¯¹ and implanted by subcutaneous injection 200 μL to each mouse. Sixty mice with implanted HepA tumor were randomly divided into control group (saline), and groups of As2O3 alone (2 mg•kg¯¹•d¯¹), PDD alone (1 mg•kg¯¹•d¯¹), As2O3 combined with PDD (As2O3 2 mg•kg¯¹•d¯¹ + PDD 1 mg•kg¯¹•d¯¹), ADM alone (1 mg•kg¯¹•d¯¹), and As2O3 combined with ADM (As2O3 2 mg•kg¯¹•d¯¹ + ADM 1 mg•kg¯¹•d¯¹). Each group was injected intraveously 24 h after transplatation once a day for 7 d continuously. The mice were killed on the 8th day after the treatment and the tumors isolated and weighed. The inhibitory rate of tumor was calculated as follows:
Inhibitory rate of tumor (%) = [1-(mean tumor weight in experiments/mean tumor weight in controls)] × 100%
Statistical method Analysis of variance of two-factor factorial experiment was applied to evaluate anti-cancer activity in vitro and analysis of variance of random experiment was used to evaluate anti-cancer activity in vivo.
Evaluation of interaction of drug combination In vitro experiment: the interaction between As2O3 and PDD or ADM was evaluted by coefficient of drug in interaction (CDI), which was calculated as follows: CDI = AB/(A × B). AB is the absorption ratio between a drug combination group and controls and A or B is that between a drug alone and controls. When CDI value was equal to 1.0, or more than 1.0 or less than 1.0, the nature of the interaction between A and B was considered to be additive or antagonistic or synergistic[6]. In vivo experiment: the interaction between As2O3 and PDD or ADM was evaluated by Q value, which was calculated as follows: Q = E (AB)/[EA+ (1-EA) × EB]. E (AB) is the inhibiting tumor rate and EA or EB is that of a drug alone. When Q value was equal to 0.85-1.15, or less than 0.85 or more than 1.15, additive or antagonistic or synergistic interaction was thought to occur[7].
RESULTS
The effect of As2O3 and/or PDD on HCC
The inhibition rates of As2O3 in vitro, in combination with PDD at various concentrations were more than that of As2O3 or PDD alone (aP < 0.01, F = 58.96), in which the inhibition rates increased more evidently at low concentrations (Figure 1). CDI values of As2O3 and PDD in combination at low concentration were less than 1.0 (Table 1).
Figure 1.
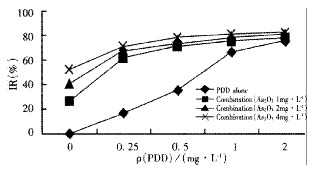
The effect of As2O3 and/or PDD on the growth of Bel-7402 cells in vitro.
Table 1.
CDI value of As2O3 in combination with PDD against Bel-7402 cells
| Cell line | As2O3 (mg•L¯¹) |
PDD (mg•L¯¹) |
|||
| 0.25 | 0.5 | 1 | 2 | ||
| Bel-7 | 1 | 0.60 | 0.60 | 1.00 | 1.22 |
| 2 | 0.62 | 0.70 | 1.03 | 1.38 | |
| 4 | 0.60 | 0.65 | 1.13 | 1.50 | |
The effect of As2O3 and/or ADM on HCC
In vitro the inhibition rates of As2O3 in combination with ADM in vario us concentrations were more than those of As2O3 or ADM alone (aP < 0.01, F = 64.77), in which the inhibition rates increased more evidently in low concentrations (Figure 2). CDI values of As2O3 and ADM in combination in low concentrations were almost equal to 1.0 (Table 2).
Figure 2.
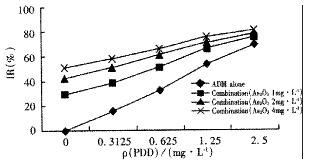
The effect of As2O3 and/or ADM on the growth of Bel-7402 cells in vitro.
Table 2.
CDI value of As2O3 in combination with ADM against Bel-7402 cells
| Cell line | As2O3 (mg•L¯¹) |
ADM (mg•L¯¹) |
|||
| 0.3125 | 0.625 | 1.25 | 2.5 | ||
| Bel-7 | 1 | 1.03 | 1.02 | 1.05 | 1.12 |
| 2 | 1.03 | 0.98 | 1.06 | 1.31 | |
| 4 | 1.00 | 1.05 | 1.08 | 1.28 | |
The effect of As2O3 and/or PDD on HepA implanted tumor
The inhibiting tumor rate of As2O3 in combination with PDD was more than that of As2O3 or PDD alone and Q value was more than 1.15 (Table 3).
Table 3.
The effect of As2O3 and/or PDD on HepA implanted tumor in mice (n = 10)
| Groups | Dose (mg•kg¯¹•d¯¹) | Tumor mass (¯x ± s, g) | Inhibition (%) | Q value |
| Control | NS | 1.53 ± 0.35 | ||
| As2O3 | 2 | 1.07 ± 0.21 | 30.1 | |
| PDD | 1 | 0.82 ± 0.11 | 46.2 | |
| As2O3+PDD | 2+1 | 0.40 ± 0.05 | 73.9a | 1.18 |
P < 0.01, F = 54.05, vs As2O3 or PDD alone.
The effect of As2O3 and/or ADM on HepA implanted tumor
The inhibiting tumor rate of As2O3 in combination with ADM was higher than that of As2O3 or ADM alone and Q value was less than 1.15 but more than 0.85 (Table 4).
Table 4.
The effect of As2O3 and/or ADM on HepA implanted tumor in mice (n = 10)
| Groups | Dose (mg•kg¯¹•d¯¹) | Tumor mass (¯x ± s, g) | Inhibition (%) | Q value |
| Control | NS | 1.53 ± 0.35 | ||
| As2O3 | 2 | 1.07 ± 0.21 | ||
| ADM | 1 | 0.91 ± 0.12 | 40.5 | |
| As2O3+ADM | 2+1 | 0.61 ± 0.11 | 60.1a | 1.03 |
P < 0.05, F = 24.40, vs As2O3 or ADM alone.
DISCUSSION
As2O3, the main component of traditional Chinese drug “Pishuang”, has been applied to treat acute promyelocytic leukemia and yielded notable results. Complete remission rate and long-term survival rate are high and the relapse rate is low in APL patients treated with As2O3[8-10]. The main mechanism of As2O3 is to induce apoptosis of leukemia cells, which is different from all-trans retinoic acid (ATRA)[11-23]. Based on the achievements, the experimental studies on anti-tumor effect of As2O3 in such hematopathy as malignant lymphoma[24] and myeloma[25,26] and solide tumors such as cancers of lung[27], esophagus[28], stomach[29-32], colone[33-35] pancreas[36], mamma[37], cervix [38] and neuroblastoma[39] are in the ascendant.
The morbidity and mortality of hepatocarcinoma is high in China, which is the first cause of death among all kinds of cancers in Jiangshu Province. Due to the hidden onset, low rates of early diagnosis and rapid progression, most patients with hepatocarcinoma cannot be operated on and have to depend on chemotherapy, but the therapeutic effect of the present agents is unsatisfactory. So it is urgent and necessary to go on seeking new drugs and the improving therapeutic METHODS. Our group has taken the lead in conducting the study of As2O3 against liver cancer and found that As2O3 had obviously selective anti-tumor effect on hepatocarcinoma both in vitro and in vivo [1-5]: in vitro As2O3 inhibited the proliferation of several hepatocarcinoma cell lines but not normal human liver cells and in vivo inhibited implanted hepatocarcinoma in mice and prolonged the survival phase of mice with hepatocarcinoma but produced no obvious toxicity. The main mechanism is to induce apoptosis of hepatocarcinoma cells, which also has been proved by other reports[40-46].
To further investigate the best therapeutic way of As2O3 and raise the effect on hepatocarcinoma, we studied As2O3 and PDD or ADM in combination. The experiments in vitro showed that As2O3 in combination with PDD or ADM can increase the effect on HCC Bel-7402 and the increase extent varies at different concentrations, which was greater at lower concentrations. The possible reason is that the anti-tumor activity of an individual drug is saturated at high concentrations and difficult to increase after combination or there was antagonistic action to some extent between two drugs in combination and counteracted part of anti-tumor activity of a drug. CDI values showed that in vitro the nature of interaction is markedly synergistic between As2O3 and PDD and addictive between As2O3 and ADM in low concentrations. On the basis of the experiments in vitro, low-dose PDD or ADM combined with As2O3 was applied to treat HepA tumor implanted in mice, and inhibitory rate of tumor evidently increased as compared with that of a drug alone. Q value showed that in vivo synergistic interaction between As2O3 and PDD and addictive between As2O3 and ADM were thought to occur, which agreed with the results in vitro. These results suggested that low-dose PDD or ADM and As2O3 in combination could increase evidently anti-hepatocarcinoma effect. PDD and ADM are the main anti-hepatocarcinoma agents, but their toxicities in kidney, liver or heart restrict their clinical application, as a result patients cannot tolerate the high-dose agents whereas low dose is difficult to achieve satisfactory results. Considering selectively inhibitory effect of As2O3 on HCC in vitro and unobvious toxicity in vivo, the effect may be improved evidently without increased toxicities or keep satisfactory in poorly-tolerated patients with low dose of PDD or ADM when As2O3 and PDD or ADM in combination are applied to treat hepatocarcinoma.
Footnotes
Edited by Ma JY
Supported by the Youth Science Grant of Jiangshu Province, No.BQ98048.
References
- 1.Chen H, Qin SK, Chen HY, Pan QS, Ma J, Liu WH. A priliminary study on arsenic trioxide as an inducer of apoptosis in human hepatocellular carcinoma cell line. Zhongliu Fangzhi Yanjiu. 1998;25:336–338. [Google Scholar]
- 2.Chen HY, Liu WH, Qin SK. Induction of arsenic trioxide on apoptosis of hepa tocarcinoma cell lines. Shijie Huaren Xiaohua Zazhi. 2000;8:532–535. [Google Scholar]
- 3.Chen H, Qin S, Pan Q. [Antitumor effect of arsenic trioxide on mice experimental liver cancer] Zhonghua Gan Zang Bing Za Zhi. 2000;8:27–29. [PubMed] [Google Scholar]
- 4.Liu L, Qin S, Chen H, Wang J, Chen H, Ma J, Liu W. [An experimental study on arsenic trioxide-selectively induced human hepatocarcinoma cell lines apoptosis and its related genes] Zhonghua Gan Zang Bing Za Zhi. 2000;8:367–369. [PubMed] [Google Scholar]
- 5.Chen H, Pan QS, Qin SK, Chen HY. An experimental study on antitumor effect of arsenic trioxide on liver cancer. Jiangsu Zhongyi. 2000;21:42–43. [Google Scholar]
- 6.Su J, Zhen YS, Qi CQ, Chen WJ. [A fungus-derived novel nucleoside transport inhibitor potentiates the activity of antitumor drugs] Yao Xue Xue Bao. 1994;29:656–661. [PubMed] [Google Scholar]
- 7.Yang XP, Pan QC, Xie BF, Li CJ. Antitumor effect and synergistic action of total alkaloid of common peganum. Zhongcaoyao. 1998;29:609–611. [Google Scholar]
- 8.Zhang P, Wang SY, Hu LH, Shi FD, Qiu FQ, Hong LJ, Han XY, Yang HF, Song YZ, Liu YP, et al. Treatment of acute promyelocytic leukemia with intrav enous arsenic trioxide. Zhonghua Xueyexue Zazhi. 1996;17:58–60. [Google Scholar]
- 9.Zhang P, Wang S, Hu L, Qiu F, Yang H, Xiao Y, Li X, Han X, Zhou J, Liu P. [Seven years' summary report on the treatment of acute promyelocytic leukemia with arsenic trioxide--an analysis of 242 cases] Zhonghua Xue Ye Xue Za Zhi. 2000;21:67–70. [PubMed] [Google Scholar]
- 10.Soignet SL, Maslak P, Wang ZG, Jhanwar S, Calleja E, Dardashti LJ, Corso D, DeBlasio A, Gabrilove J, Scheinberg DA, et al. Complete remission after treatment of acute promyelocytic leukemia with arsenic trioxide. N Engl J Med. 1998;339:1341–1348. doi: 10.1056/NEJM199811053391901. [DOI] [PubMed] [Google Scholar]
- 11.Chen GQ, Zhu J, Shi XG, Ni JH, Zhong HJ, Si GY, Jin XL, Tang W, Li XS, Xong SM, et al. In vitro studies on cellular and molecular mechanisms of arsenic trioxide (As2O3) in the treatment of acute promyelocytic leukemia: As2O3 induces NB4 cell apoptosis with downregulation of Bcl-2 expression and modulation of PML-RAR alpha/PML proteins. Blood. 1996;88:1052–1061. [PubMed] [Google Scholar]
- 12.Chen G, Zhu J, Shi X, Zhong H, Liu W, Jin X, Tang W, Li X, Ni J, Xiong S, et al. [Preliminary study on the arsenic trioxide-induced NB4 cell apoptosis and its molecular mechanisms] Zhonghua Xue Ye Xue Za Zhi. 1997;18:25–28. [PubMed] [Google Scholar]
- 13.Ni J, Chen G, Zhu J, Zhong H, Tang W, Li X, Xiong S, Shen Z, Chen S, Wang Z, et al. [Effects of arsenic trioxide on the subcellular localization of PML/PML-RARalpha protein in leukemic cells] Zhonghua Xue Ye Xue Za Zhi. 1997;18:32–34. [PubMed] [Google Scholar]
- 14.Tang W, Chen G, Shi G. [Double effects of arsenic trioxide (As2O3) on acute promyelocytic leukemic cell line] Zhonghua Yi Xue Za Zhi. 1997;77:509–512. [PubMed] [Google Scholar]
- 15.Chen GQ, Shi XG, Tang W, Xiong SM, Zhu J, Cai X, Han ZG, Ni JH, Shi GY, Jia PM, et al. Use of arsenic trioxide (As2O3) in the treatment of acute promyelocytic leukemia (APL): I. As2O3 exerts dose-dependent dual effects on APL cells. Blood. 1997;89:3345–3353. [PubMed] [Google Scholar]
- 16.Shen ZX, Chen GQ, Ni JH, Li XS, Xiong SM, Qiu QY, Zhu J, Tang W, Sun GL, Yang KQ, et al. Use of arsenic trioxide (As2O3) in the treatment of acute promyelocytic leukemia (APL): II. Clinical efficacy and pharmacokinetics in relapsed patients. Blood. 1997;89:3354–3360. [PubMed] [Google Scholar]
- 17.Look AT. Arsenic and apoptosis in the treatment of acute promyelocytic leukemia. J Natl Cancer Inst. 1998;90:86–88. doi: 10.1093/jnci/90.2.86. [DOI] [PubMed] [Google Scholar]
- 18.Cai X, Jia P, Shi X. [In vitro study on arsenic trioxide-induced apoptosis of retinoic acid resistant acute promyelocytic leukemia cell line (MR-2)] Zhonghua Xue Ye Xue Za Zhi. 1998;19:339–341. [PubMed] [Google Scholar]
- 19.Li J, Sun GL, Su H, Chen HR. In vitro study on effect of two different arsenicals on NB 4 cells proliferation and programmed cell death. Baixuebing. 1998;7:146–149. [Google Scholar]
- 20.Shao W, Fanelli M, Ferrara FF, Riccioni R, Rosenauer A, Davison K, Lamph WW, Waxman S, Pelicci PG, Lo Coco F, et al. Arsenic trioxide as an inducer of apoptosis and loss of PML/RAR alpha protein in acute promyelocytic leukemia cells. J Natl Cancer Inst. 1998;90:124–133. doi: 10.1093/jnci/90.2.124. [DOI] [PubMed] [Google Scholar]
- 21.Sun YP, Chen GQ, Cai X, Huang Y, Shen SY, Jia PM, Shen YL, Yu Y, Chen SJ, Wang ZY, et al. Phenylarsine oxide induces apoptosis in acute promyelocytic leukemia cell line NB4. Aizheng. 1999;18:1–4. [Google Scholar]
- 22.Huang X. [Regulation of arsenic trioxide-inducing apoptosis] Zhonghua Xue Ye Xue Za Zhi. 1999;20:258–260. [PubMed] [Google Scholar]
- 23.Wang ZX, Liao JX, Xu YG, Wang HP, Ma R, Zhou AX. Influence of antioxi dants on apoptosis induced by arsenic trioxide. Baixuebing. 2000;9:153–155. [Google Scholar]
- 24.Zhu XH, Chen GQ, Cai X, Jia PM, Shen YL, Shi XG, Shi GY, Chen SJ, Wang ZY, Chen Z. Influence of arsenic trioxide on malignant lymphocyte. Zhonghua Xueyexue Zazhi. 1999;20:313–314. [Google Scholar]
- 25.Rousselot P, Labaume S, Marolleau JP, Larghero J, Noguera MH, Brouet JC, Fermand JP. Arsenic trioxide and melarsoprol induce apoptosis in plasma cell lines and in plasma cells from myeloma patients. Cancer Res. 1999;59:1041–1048. [PubMed] [Google Scholar]
- 26.Huang XJ, Zhou YH, Cai X, Shen YL, Jia PM, Yang J, Yuan MM, Zhang XG, Chen GQ. Effects of arsenic trioxide on multiple myeloma cells. Zhongguo Aizheng Zazhi. 2000;10:499–509. [Google Scholar]
- 27.Deng YP, Lin C, Zhang XY, Chen JP, Xiao PG, Wu H. Studies on arsenic trioxide induced human pulmonary adenocarcinoma GLC-82 cell apoptosis and its molecular mechanisms. Aizheng. 1999;18:545–549. [Google Scholar]
- 28.Tan LJ, Shi GY, Shi XG, Tang XM. Induction of apoptosis of human esoph ageal cancer cell lines treated with arsenic trioxide. Zhongguo Aizheng Zazhi. 1999;9:85–87. [Google Scholar]
- 29.Gu QL, Shen BH, Li NL, Zhu ZG, Yin HR, Lin YJ. A preliminary study on arsenic trioxide induced apoptosis of gastric cancer lines. Zhonghua Xiaohua Zazhi. 1998;18:69–71. [Google Scholar]
- 30.Deng YP, Lin C, Zhang XY, Chen DQ, Xiao PG, Wu H. Arsenic trioxide induces programmed cell death of human gastric adenocarcinoma SGC7901 cells and decreases c-myc gene expression. Yaoxue Xuebao. 1999;34:333–337. [Google Scholar]
- 31.Gu QL, Li NL, Zhu ZG, Yin HR, Lin YZ. A study on arsenic trioxide inducing in vitro apoptosis of gastric cancer cell lines. World J Gastroenterol. 2000;6:435–437. doi: 10.3748/wjg.v6.i3.435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Tu SP, Zhong J, Tan JH, Jiang XH, Qiao MM, Wu YX, Jiang SH. Induction of apoptosis by arsenic trioxide and hydroxy camptothecin in gastriccancer cells in vitro. World J Gastroenterol. 2000;6:532–539. doi: 10.3748/wjg.v6.i4.532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Yu BM, Cui W, Lu AG, Li DH, Zhen MH. Experimental studies on inhibito ry effect of arsenic trioxide on colon cancer. Zhongliu. 1999;19:289–290. [Google Scholar]
- 34.Yu BM, Cui W, Lu AG, Zheng MH, Li DH. Apoptosis of colon cancer cell induced by arsenic trioxide and its relation to cell cycle. Zhongguo Aizheng Zazhi. 2000;10:159–162. [Google Scholar]
- 35.Zhan J, Xie DR, Yao HR, Lin XG, Liang XW, Xiang YQ. Apoptosis of colon cancer cell line SW620 induced by arsenic trioxide. Shijie Huaren Xiaohua Zazhi. 2001;9:228–229. [Google Scholar]
- 36.Chen QK, Yuan SZ, Huang ZQ. Apoptosis of pancreatic cancer cells and blocking of its cell cycles induced by arsenic trioxide. Zhonghua Yixue Zazhi. 1998;78:578–579. [Google Scholar]
- 37.Yang H, Yu LL, Wang XP, Ma WX, Zheng S. Apoptosis of mammary cancer cells induced by arsenic trioxide. Zhongguo Zhongliu Shengwu Zhiliao Zazhi. 1997;4:245. [Google Scholar]
- 38.Deng YP, Lin C, Liang X, Chen JP, Fu M, Xiao PG, Wu H. Studies on indu ction of apoptosis of human cervicalcarcinoma cells Hela and mechanism of protec tive effect of bcl-2. Zhongguo Kexue. 1999;29:426–433. [Google Scholar]
- 39.Wu G, Zhou YF, Ying DM, Chen TX, Wang YP, Lin Z. Preliminary studies of arsenic trioxide on the proliferation of neuroblastoma cells. Aizheng. 1999;18:263–265. [Google Scholar]
- 40.Zhang T, Wang SS, Qi QH. The morphological and cellular dynamic changes in arsenic trioxide-treated rat liver cancer. Zhongliu. 2001;21:101–105. [Google Scholar]
- 41.Li JT, Qu QJ, Wei Q. Studies on arsenic trioxide induces apoptosis in hepatoma cell lines Bel-7402. Aizheng. 2000;19:1087–1091. [Google Scholar]
- 42.Yang YL, Xu HY, Gao YY, Wu QL, Gao GQ. Influence of arsenic trioxide on human hepatoma cell line. Shijie Huaren Xiaohua Zazhi. 2000;8:1422–1423. [Google Scholar]
- 43.Xu HY, Gao YY, Wu QL, Gao GQ, Yang YL, Chen SX, Liu TF. Proliferation inhibition and apoptosis induction by Arsenic Trioxide on human hepatoma cell line in vitro. Shijie Huaren Xiaohua Zazhi. 2000;8:1233–1237. [Google Scholar]
- 44.Deng ZH, Cai HP, Li S, Zhang XR, Shen JW. Effects of arsenic trioxide on normal and hepatoma cell lines. Zhonghua Xiaohua Zazhi. 1999;19:227–229. [Google Scholar]
- 45.Yang YL, Xu HY, Gao YY, Wu QL, Gao GQ. The effect of arsenic trioxide on human hepatoma cell line BEL-7402 cultured in vitro. World J Gastroenterol. 2000;6(Suppl 3):14. doi: 10.3748/wjg.v6.i5.681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Xu HY, Yang YL, Gao YY, Wu QL, Gao GQ. Effect of arsenic trioxide on human hepatoma cell line BEL-7402 cultured in vitro. World J Gastroenterol. 2000;6:681–687. doi: 10.3748/wjg.v6.i5.681. [DOI] [PMC free article] [PubMed] [Google Scholar]


