Abstract
Background
Treatment of hepatitis C virus (HCV) infection based on peginterferon-α (pegIFNα) and ribavirin induces important changes in cytokine release and T cell activation.
Objective
Immune response to pegIFNα-ribavirin therapy was explored in patients coinfected by HCV and HIV.
Methods
Concentrations of 25 cytokines and CD8+ T cell activation were monitored in HCV/HIV coinfected patients classified as sustained virological responders (SVR, n=19) and non-responders (NR, n=11).
Results
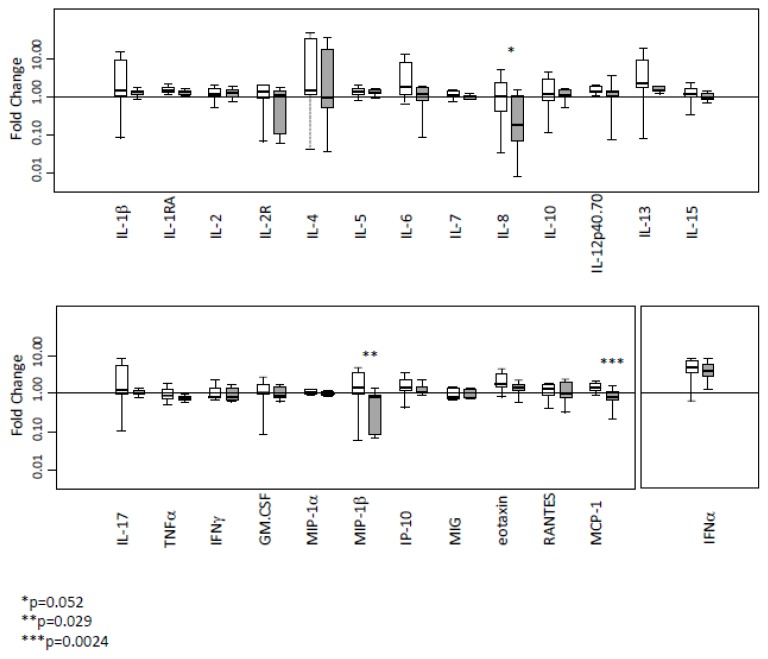
High pretreatment concentrations of IP-10 (CXCL-10) and MCP-1 (CCL-2) were associated with a poor anti-HCV response. PegIFNα-ribavirin therapy increased CD8+ T cell activation and induced significant changes in levels of eleven cytokines related to both Th1 and Th2 responses in SVR (IL-1β, IL-1RA, IL-4, IL-5, IL-6, IL-7, IL-12p40/70, IL-13, IP-10, eotaxin, MCP-1) but of only six cytokines in NR (IL-1β, IL-2, IL-5, IL-12p40/70, IL-13, eotaxin). The highest rise in MIP-1β and MCP-1 levels was observed four weeks after anti-HCV treatment initiation in SVR compared to NR (p=0.002 and p=0.03, respectively), whereas a decrease in IL-8 concentration was associated with treatment failure (p= 0.052).
Conclusions
Higher and broader cytokine responses to pegIFNα-ribavirin therapy were observed in SVR patients compared to NR. Changes in IL-8, MIP-1β, and MCP-1 serum concentrations may be associated with efficacy of pegIFNα- and ribavirin-based therapies in patients coinfected by HCV and HIV.
Introduction
Infection by hepatitis C virus (HCV) and HIV shared common routes of transmission and thus confections with the two viruses are frequent. HCV infection is seen in 15–30% of HIV-infected patients in Western Europe, with almost 20 000 HIV-infected individuals chronically infected with HCV in France.1,2 Since the introduction of antiretroviral therapy (ART), HCV-related liver diseases have become leading causes of morbidity and mortality in HIV-infected individuals.3,4
Faster progression to cirrhosis has been observed in patients coinfected with HCV and HIV by comparison with subjects infected with HCV alone.5 HIV/HCV coinfection is also associated with higher HCV viral levels in serum.6 Finally, ART-related adverse events are more frequent in HCV/HIV coinfected individuals, with increased difficulties in optimally treating HIV infection.7,8
Until recently, a combination of pegIFN and ribavirin has been the standard-of-care for treatment of chronic HCV patients. In coinfected individuals -ribavirin therapy is less efficacious than in single HCV infection.9,10 Approximately 30% of patients receiving a combination of ribavirin with pegIFN have a sustained virological response when infected with HCV genotype 1 and 4.9 This rate increases to 60% when genotype 2 and 3 are involved.10 Most of the current anti-HCV therapeutic options remained based on pegIFN and/or ribavirin in addition to protease inhibitors or NS5A or NS5B inhibitors.11
PegIFN constitutes an active immunotherapy triggering innate immune response and T cell activation. The cytokine response to pegIFNα-based regimens plays probably an important role in treatment outcome. IFNα stimulates Th1 cells and limits secretion of Th2 related cytokines such as IL-4 or IL-10.12 IFNα signals through Janus kinase signal transducer, an activator of transcription pathway, and induction of IFN-stimulated genes.13 Ribavirin synergies this effect by reducing IL-10 production, enhancing autocrine IFN-β and IL-8 secretion.14–16 Several immune factors are predictive of virological response on pegIFNα-ribavirin-based therapy: pretreatment concentration of IFNα-inducible protein 10 (IP-10, also called CXCL-10), polymorphisms near the interleukin-28B gene coding for the IFNα-3, and CD4+ T cell count in HIV/HCV coinfected individuals.18,19 Progressive impairment of immune functions and T cell exhaustion induced by prolonged exposure to virus antigens are fundamental features of HIV infection. Inhibitory networks, such as the programmed cell death protein-1 (PD-1) and IL-10 play a key role in this process.20,21 PD-1 is upregulated on CD8+ and CD4+ T cells and mediates a dysfunction partially reversible on ART.22–24 PD-1 signalling impacts the expression of both Th1 and Th2 cytokines but limits particularly the capacity of Th1 and IFNγ secretion by CD4+ and CD8+ T cells.21
Although HCV clearance depends on immune response induced by pegIFNα-ribavirin therapy, the dynamics of cytokine response and T cell activation in individuals coinfected with HIV and HCV remain poorly characterized.25 In this study, we monitor serum concentration of 25 cytokines alongside with the level of CD8+ T cell activation in HIV/HCV coinfected patients initiating anti-HCV therapy.
Methods
Patient samples
Patients coinfected by HIV/HCV followed at Montpellier University Hospital and initiating anti-HCV therapy were included consecutively between January 2007 and January 2010 after providing written informed consent. The study was approved by the local institution ethical committee (DC-2011-1405). Chronic hepatitis was proved by the presence of serum HCV antibodies and detectable viral RNA. HCV genotype, HCV and HIV-1 viral loads, CD4+ T cell count, and liver enzyme levels were all determined using standard procedures. All patients received pegIFNα2a 180 microg/week plus ribavirin 800 to 1,200 mg daily; patients infected with HCV genotype 2 and 3 received 800 mg daily and genotype 1 and 4 received a weight based dose: 1,000 mg/day for persons less than 75 kg and 1200 mg/day over 75 kg.
Subjects were ranged into two distinct groups based on HCV response to therapy:
Sustained virological responders (SVR) when serum HCV RNA was undetectable 24 weeks after completing therapy, and
non responders (NR) when HCV RNA decrease was < 2 log copies/mL at week 12 or when HCV RNA was detectable at the end of treatment.
Patient relapsing in the 24 weeks period after therapeutic cessation were excluded from the study.
Routine laboratory testing
Serum HCV RNA was quantified using COBAS AmpliPrep/COBAS TaqMan HCV assay (Roche Diagnostic Systems). HCV genotypes were determined using INNO-LiPA HCV II test (Innogenetics). Alanine aminotransferase (ALT) dosages were executed using standard methods.
Quantitation of serum cytokine concentrations
Cytokines were quantified in serum samples obtained before initiation of treatment, and four weeks after the start of PegIFNα-based therapy. A multiplexed microbead assay was used according to manufacturer’s instructions (cytokine twenty-five-plex kit, Life Technologies Ltd, Paisley, UK) and an FIDIS apparatus (BMD). Twenty five cytokines were quantified in duplicate: IL-1β, IL-1RA, IL-2, IL-2R, IL-4, IL-5, IL-6, IL-7, IL-8, IL-10, IL-12p40/70, IL-13, IL-15, IL-17, eotaxin, GM-CSF, IFN-α, IFN-γ, IP-10, MCP-1, MIG, MIP-1α, MIP-1β, RANTES, and TNF-α. Data were analyzed using the MLX-Booster program (BMD). Mean concentrations (pg/ml) of cytokines were all superior to the detection limits, defined as the mean background value plus 2 SD.
Analysis of CD8+/CD38bright T cells
Activation of CD8+ T cells was assessed by flow cytometry analysis on EDTA-treated fresh whole blood using FC 500 apparatus (Beckman Coulter, Miami, Florida). T cell activation was explored at day 0, week 4 and week 12 after initiation of pegIFNα-ribavirin therapy. The expression of CD38bright on CD8+ T cells was analyzed as previously described using a two-colour staining with anti-CD8 and anti-CD38 conjugated to fluorescein isothiocyanate (FITC) and phycoerythrin (PE), respectively (Beckman Coulter).26 The positive threshold for CD38bright analysis was established using the CellQuant CD38/CD8 kit for quantitation of CD38 cell surface expression (BioCytex, Marseille, France) and was defined as 8,500 CD38 binding sites/cells. The CD8+/CD38bright values were expressed as the percentage of CD38bright cells from the CD8+ T cell populations.
Statistical Analysis
The Mann–Whitney U test and Kruskal–Wallis test were used to analyze continuous variables where appropriate. The Friedman test was used to evaluate changes in serum cytokine levels over time. A mixed model was used to analyze the association between T cell activation during anti-HCV therapy and therapeutic response. Statistical analyses were done using SPSS software version 18.0J.
Results
Clinical characteristics at baseline
Thirty HCV/HIV coinfected patients receiving pegIFNα-ribavirin therapy were included in the study. Nineteen (63%) were classified as SVR and eleven (37%) as NR. Patient characteristics are shown in Table 1.
Table 1.
Patient characteristics
| All (n=30) | SVR (n=19) | NR (n=11)* | |
|---|---|---|---|
| Age | 45 | 44 | 47 |
| CD4+ T cells (/μl) | 350 (50–1076) | 378(128–1076) | 328 (50–715) |
| Nadir CD4 T cells (/μl) | 122.5 (32–598) | 124 (33–598) | 119.5 (32–324) |
| ALAT (IU/L) | 48.5 (13–172) | 40.5 (13–172) | 56.5 (24–133) |
| VL HCV S0 (Log10 IU/ml) | 5.96 (5.43–6.68) | 5.49 (4.12–5.81) | 6.68 (5.98–6.84) |
| VL HIV S0 (IU/l) | 0 (0–290) | 0 (0–290) | 0 (0–118) |
| HCV Genotypes 1/2/3/4 | 21/2/7/1 | 10/1/8/0 | 10/0/0/1 |
| Liver fibrosis (F≥2) | 22 | 13 | 9 |
Seven subjects had a reduction of less than 2 log10 in HCV RNA after 12 weeks of therapy (null responders), and four subjects had a reduction of more than 2 log10 (partial responders).
Pretreatment levels of soluble and cellular inflammatory markers
IP-10 levels were higher in the NR group than in the SVR group, P = 0.021 (Table 2). Individuals from the NR group also had higher pretreatment concentrations of MCP-1, P = 0.0009. A trend for a higher serum concentration of IL-8 was also observed in patients of the NR group compared to the SVR group, P = 0.106.
Table 2.
Pre-treatment Cytokine concentrations in serum from SVR and NR patients
| Cytokines | SVR pg/ml mean [CV] | NR pg/ml mean [CV] | p-value (Wilcoxon) |
|---|---|---|---|
| IL-1β | 12.9 [ 34.2 ] | 10.84 [ 4.9 ] | 0.093 |
| IL-1RA | 1556.0 [ 198.0 ] | 1506.0 [ 123.0 ] | 0.779 |
| IL-2 | 6.2 [ 2.4 ] | 5.8 [ 2.6 ] | 0.425 |
| IL-2R | 837.0 [ 812.2 ] | 884.1 [ 155 ] | 0.101 |
| IL-4 | 8.0 [ 177.9 ] | 8.0 [ 173.5 ] | 0.338 |
| IL-5 | 2.3 [ 0.5 ] | 2.3 [ 0 ] | 0.616 |
| IL-6 | 4.6 [ 2.2 ] | 5.0 [ 23.0 ] | 0.353 |
| IL-7 | 18.9 [ 2.3 ] | 20.8 [ 4.3 ] | 0.142 |
| IL-8 | 22.5 [ 73.8 ] | 128.1 [ 707.7 ] | 0.106 |
| IL-10 | 3.6 [ 1.1 ] | 3.1 [ 2.0 ] | 0.777 |
| IL-12p40/70 | 321.5 [ 55.8 ] | 318.3 [ 56.5 ] | 0.948 |
| IL-13 | 11.0 [ 4.1 ] | 11.0 [ 2.7 ] | 0.702 |
| IL-15 | 33.2 [ 9.4 ] | 33.2 [ 3.1 ] | 0.863 |
| IL-17 | 24.8 [ 9.3 ] | 24.0 [ 8 ] | 0.73 |
| TNFα | 8.8 [ 2.6 ] | 8.8 [ 1.1 ] | 0.762 |
| IFNγ | 5.2 [ 1.3 ] | 6.1 [ 0.8 ] | 0.621 |
| GM.CSF | 18.4 [ 9.2 ] | 15.3 [ 13.8 ] | 0.482 |
| MIP-1α | 119.4 [ 1.8 ] | 120.6 [ 6.8 ] | 0.262 |
| MIP-1β | 150.4 [ 24.3 ] | 150.4 [ 23.6 ] | 0.88 |
| IP-10 | 64.3 [ 32.9 ] | 132.0 [ 58.8 ] | 0.021 |
| MIG | 458.6 [ 5.9 ] | 458.6 [ 4.0 ] | 0.497 |
| Eotaxin | 75.5 [ 30.7 ] | 72.9 [ 29.4 ] | 0.813 |
| RANTES | 10540.0 [ 6413.0 ] | 9350.0 [ 10922.0 ] | 0.683 |
| MCP-1 | 347.9 [ 91.2 ] | 442.6 [ 145.7 ] | 0.009 |
| IFNα | 143.0 [ 21.3 ] | 143.0 [ 1.4 ] | 0.477 |
Cytokines change after therapeutic initiation of HCV treatment
Initiation of anti-HCV treatment elicited IFNα rise in both SVR and NR patients (Figure 1 and Supplemental Table 1). This enhancement of IFNα in serum was similar in the two groups. Besides IFNα, the anti-HCV therapy induced a significant increase in the serum levels of eleven cytokines in SVR patients, including cytokines related to Th1-dominant immune responses (IL-12p40/70, IP-10), Th2-type cytokines (IL-4, IL-5, and IL-13), and pro-inflammatory cytokines (IL-1β, IL-1RA, IL-6, IL-7). By comparison, levels of only six cytokines rose in the NR group (Supplemental Table 1). Finally, when results were analyzed without consideration of the HCV therapeutic response (combining SVR and NR patients), we also observed an increase of IL-2, IL-10, and RANTES concentrations four weeks after initiation of anti-HCV treatment (Additional file 1).
Figure 1.
Impact of anti-HCV therapy on cytokine concentrations in serum of NR and SVR. Fold-changes in cytokine levels following pegIFNα-ribavirin therapy initiation in SVR (white bars) and NR (gray bars). Results are expressed as fold changes in cytokine concentrations between week 0 (pretreatment) and week 4 (four weeks of pegIFNα-ribavirin therapy). *p=0.052, **p=0.029, ***p=0.0024.
Comparison of the cytokine level changes induced by pegIFNα-ribavirin between SVR and NR group
Changes in cytokines were compared among SVR and NR using the ratio between baseline and four weeks of treatment (Figure 1). A sharp increase of MCP-1 and MIP-1β concentration was observed after initiation of anti-HCV therapy in the SVR group whereas the value decreased or remained stable in the NR group (p=0.0024 and p=0.029, respectively). Furthermore, the IL-8 concentration remains stable after a one-month period of pegIFNα-ribavirin therapy in most of the patients responding to anti-HCV treatment, whereas this concentration declined in NR patients (p=0.052).
Changes in T cell activation over the three-months period of therapy initiation
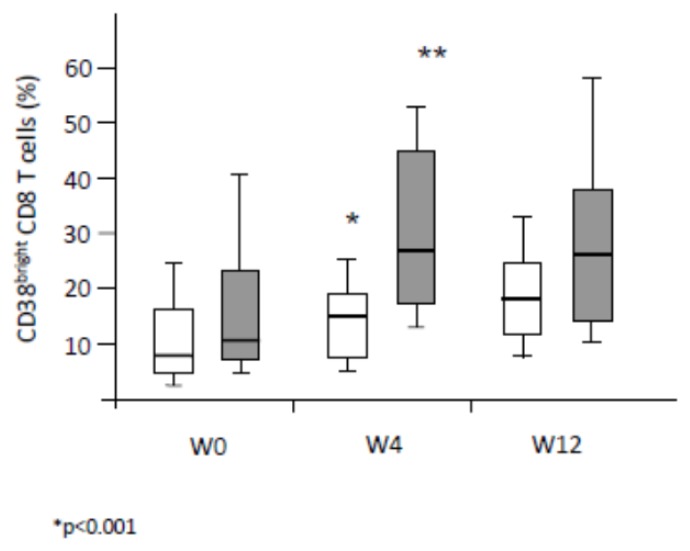
The impact of pegIFNα-ribavirin on T cell activation was explored at day 0, week 4 and week 12 (Figure 2). CD8+ T cell activation before the treatment tends to be higher in NR patients compared to the SVR patients, (p=0.066, Figure 2). Anti-HCV therapy induced a major increase in CD38bright expression on CD8 T+ cells in the two groups of patients (p<0.001). No significant differences were observed between the two groups in the slope of CD38 cell surface expression of CD8+ T cells suggesting that the over time changes of this activation marker not be associated with a better immune response to therapy.
Figure 2.
Changes in T cell activation over a 3-month period of pegIFNα-ribavirin therapy. Fold-changes in CD38bright expression on CD8 T cells following pegIFNα-ribavirin therapy initiation in SVR (white bars) and NR (gray bars). A significant increase of CD38bright expression between two consecutive time periods is indicated: *p<0.001.
Discussion
Although the virological response to pegIFNα/ribavirin-based therapy is intimately associated with the host immunity, the monitoring of HCV treatment is based on the serum HCV-RNA level decay regardless of the immune response. In this study, CD8+ T cell activation and levels of 25 cytokines were analyzed in HIV/HCV coinfected patients with favourable versus unfavourable therapeutic outcome.
PegIFNα/ribavirin administration induced a dramatic change in serum cytokine concentrations and CD8+ T-cell activation level. We observed that the HCV clearance under pegIFNα/ribavirin therapy was associated with broader and higher cytokine responses in SVR than in NR.
Multianalyte assays make possible to analyze a wide an array of cytokines using a small sample volume rapidly. In the present study, the level of 25 cytokines and CD8+ T-cell activation were analyzed at baseline and week four after the initiation of pegIFNα/ribavirin therapy. Comparison between different multiplex bead assays has shown variable agreement among kits evidencing that absolute cytokine concentrations differ across commercial assays.27,28 As a consequence, multisite comparisons between cytokine concentrations obtained with different methods are difficult to interpret because of the lack of standardization. However, these methods are useful for longitudinal studies, and a similar rank order of cytokine concentrations between samples is preserved between the different commercial kits. Hence, we considered comparisons based on fold change under pegIFNα-ribavirin therapy as a valuable method for identifying a predictor of therapeutic success or failure. Even if interferon-free regimen progressively substitute pegIFNα-containing regimen for treatment of hepatitis C it remains of particular importance to describe immune response after initiation of pegIFNα. Indeed, IFNα remains the only drug approved for treatment of chronic hepatitis D and the only treatment used for a limited period for hepatitis B.
Baseline serum concentration of IP-10 and MCP-1 (monocyte chemotactic protein 1) were higher in NR than in SVR. A significant increase was observed after treatment initiation in 15 out of 25 soluble biomarkers tested. The strongest increased was observed for IFNα as an expectable consequence of the anti-HCV therapy, with more than 4-fold rise between week 0 and week 4. Increase in cytokine concentrations after pegIFNα-ribavirin administration was observed for pro-inflammatory- and Th1-related cytokines such as IL-12 or IP-10, but also for Th2-related cytokines such as IL-4 or IL-13. Activation of T cells accompanied this phenomenon as shown by the increased cell surface expression of CD38 on CD8+ T cells.
Based on therapeutic outcome distinct cytokine patterns were identified. Differences in cytokine concentrations were observed at baseline and following anti-HCV initiation. In agreement with previous studies in individuals coinfected we observed the pretreatment serum level of IP-10 was higher in the NR group than in the SVR group.19 Circulating IP-10 concentration is well correlated with intrahepatic IP-10 messenger RNA expression during chronic HCV infection.29 The high baseline hepatic IFN-stimulated genes expression is associated with a lower chance to be SVR to IFN-based therapy when HIV-uninfected.30–33 Interestingly, the pre-treatment MCP-1 concentration was also found at a higher concentration in the NR group. This pro-inflammatory chemokine is involved in the migration and infiltration of monocytes and CD4+ memory T lymphocytes. Intra-hepatic and serum MCP-1 levels are associated with liver inflammation and fibrosis during chronic HCV infection.34,35 MCP-1 plays a significant role in the recruitment of monocytes by interacting with their cell-surface adhesion-molecules that are over expressed during chronic HCV infection.36 Association between MCP-1 baseline level, and response to pegIFNα-ribavirin therapy has been recently reported in HIV-uninfected patients.37 Both chronic HCV and HIV infections induce expression and release of MCP-1 in humans.38,39 HIV proteins like gp12040 and transactivator protein Tat41,42 have shown strong immunomodulatory capacity through MCP-1 stimulation. MCP-1 has also been shown to promote Th2 responses by polarizing Th0 cells towards a Th2 phenotype43 and is thought to be one of the key factors involved in HIV pathogenesis.44 Hence, MCP-1 pretreatment level may be associated with the promotion of the Th1 dominant response and HCV clearance under pegIFNα-ribavirin therapy in HIV/HCV coinfected patients.
Cytokine response to pegIFNα/ribavirin therapy differed qualitatively between SVR and NR. The cytokine response appeared more restricted in NR than in SVR since only six versus 11 cytokines rise significantly following the commencement of anti-HCV therapy. As a possible consequence of high IP-10 and MCP-1 baseline level, the boost of circulating IP-10 and MCP-1 levels was not observed in the NR after four weeks of pegIFNα-ribavirin treatment. Circulating IP-10 is considered as a marker of the pre-therapeutic activation level of IFN-stimulated genes. The defect in IP-10 response observed four weeks after starting pegIFNα-ribavirin treatment can be viewed as a marker of the overexpression of IFN-stimulated genes leading to a poor capacity response to exogenous IFN injections.
Four weeks after initiation of pegIFNα-ribavirin, a higher MIP-1β (macrophage inflammatory protein-1β) and MCP-1 secretions were observed in SVR when compared to NR. Of interest, a trend for a paradoxical decrease of MIP-1β was also observed at the same time in the NR group. The concentration of MIP-1β was halved in the NR group at week four compared to baseline whereas MCP-1β level tends to increase in the SVR group. This observation is consistent with a previous study showing that HCV clearance is more frequent in patients experiencing strong MIP-1α and MIP-1β response to pegIFNα-based therapy.45,46 MIP-1β (also named CCL4) is a chemokine linked to the Th1 response. MIP-1β is involved in T cell recruitment mediated by CCR5 and CCR1 in the liver of HCV infected patients.38 The amplitude of MCP-1 variation in response to pegIFNα/RBV is probably dependent on baseline MCP-1 level as for IP-10. IL-8 dynamic response to pegIFNα-ribavirin therapy also appears different in the two groups. IL-8 serum levels tend to be elevated at baseline in NR and to diminish following pegIFNα-ribavirin initiation. High IL-8 mRNA is involved in liver inflammation47–49 and inhibition of the IFNα antiviral response in vitro.50 Pretreatment level of IL-8 has also been found associated with poor virological response to pegIFNα-ribavirin therapy.51 Evolution of IL-8 concentration may reflect the combined effect of pegIFN plus ribavirin since ribavirin induces its secretion through the activation of activator protein 1.14
In this study, relapse patients were not included in the group of patients that did not recover from HCV under pegIFNα-ribavirin. Subjects who experience relapse had patterns of virological response different from NR with a rapid decline in HCV RNA during the first week of treatment.52,53 Hence, relapse patients should be explored a part from NR and SNR groups.
Our study had limitations related to its retrospective nature and the small number of subjects included. HCV genotypes and fibrosis stage were not taken into account in the analysis and IL-28 polymorphism, which is also known to predict the hepatic responsiveness to IFN-based therapy, was not assessed. The study was not designed to establish clinical performances of immunological markers useful for HCV therapeutic prediction and/or monitoring but showed that different ongoing trends of cytokine response to pegIFNα/ribavirin can be observed in SVR and NR patients infected with HIV.
Conclusions
HIV/HCV coinfected patients, showing a high IP-10 and MCP-1 baseline level alongside with a reduced capacity to produce or to maintain broader secretion of cytokines, including MIP-1β, IL-4, IL-6 or IL-8, may have a poor response to regimens based on Peg-IFN and ribavirin. Anti-HCV treatment based on drugs having immunomodulating activities may benefits from immunomonitoring using multiplex techniques.
Acknowledgements
This work has been supported by grants from Agence Nationale de Recherche sur le SIDA et les hépatites virales (ANRS), UMR Inserm U1058, and the Ministère Français de la Coopération for Ph.D. grants of Tam Truong Nguyen.
References
- 1.Loko MA, Salmon D, Carrieri P, et al. The French national prospective cohort of patients co-infected with HIV and HCV (ANRS CO13 HEPAVIH): early findings, 2006–2010. BMC Infect Dis. 2010;10:303. doi: 10.1186/1471-2334-10-303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Larsen C, Chaix ML, Le Strat Y, et al. Gaining greater insight into HCV emergence in HIV-infected men who have sex with men: the HEPAIG Study. PLoS One. 2011;6:e29322. doi: 10.1371/journal.pone.0029322. [DOI] [PMC free article] [PubMed] [Google Scholar]; Davies A, Singh KP, Shubber Z, et al. Treatment outcomes of treatment-naïve Hepatitis C patients co-infected with HIV: a systematic review and meta-analysis of observational cohorts. PLoS One. 2013;8:e55373. doi: 10.1371/journal.pone.0055373. [DOI] [PMC free article] [PubMed] [Google Scholar]; Heim MH. Interferons and hepatitis C virus. Swiss Med Wkly. 2012;142:w13586. doi: 10.4414/smw.2012.13586. [DOI] [PubMed] [Google Scholar]
- 3.Luz PM, Bruyand M, Ribeiro S, et al. AIDS and non-AIDS severe morbidity associated with hospitalizations among HIV-infected patients in two regions with universal access to care and antiretroviral therapy, France and Brazil, 2000–2008: hospital-based cohort studies. BMC Infect Dis. 2014;21:278. doi: 10.1186/1471-2334-14-278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ingle SM, May MT, Gill MJ, et al. Antiretroviral Therapy Cohort Collaboration. Impact of risk factors for specific causes of death in the first and subsequent years of antiretroviral therapy among HIV-infected patients. Clin Infect Dis. 2014;59:287–297. doi: 10.1093/cid/ciu261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Thein HH1, Yi Q, Dore GJ, Krahn MD. Natural history of hepatitis C virus infection in HIV-infected individuals and the impact of HIV in the era of highly active antiretroviral therapy: a meta-analysis. AIDS. 2008;22:1979–1991. doi: 10.1097/QAD.0b013e32830e6d51. [DOI] [PubMed] [Google Scholar]
- 6.Danta M, Semmo N, Fabris P, et al. Impact of HIV on host-virus interactions during early hepatitis C virus infection. J Infect Dis. 2008;197:1558–1566. doi: 10.1086/587843. [DOI] [PubMed] [Google Scholar]
- 7.Loko MA, Bani-Sadr F, Winnock M, et al. Impact of HAART exposure and associated lipodystrophy on advanced liver fibrosis in HIV/HCV-coinfected patients. J Viral Hepat. 2011;18:307–314. doi: 10.1111/j.1365-2893.2010.01417.x. [DOI] [PubMed] [Google Scholar]
- 8.D’Arminio Monforte A, Cozzi-Lepri A, Castagna A, et al. Risk of developing specific AIDS-defining illnesses in patients coinfected with HIV and hepatitis C virus with or without liver cirrhosis. Clin Infect Dis. 2009;49:612–622. doi: 10.1086/603557. [DOI] [PubMed] [Google Scholar]
- 9.Ford N, Kirby C, Singh K, et al. Chronic hepatitis C treatment outcomes in low- and middle-income countries: a systematic review and meta-analysis. Bulletin of the World Health Organization. 2012;90:540–550. doi: 10.2471/BLT.11.097147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.European Association for Study of Liver. EASL Clinical Practice Guidelines: management of hepatitis C virus infection. J Hepatol. 2014;60:392–420. doi: 10.1016/j.jhep.2013.11.003. [DOI] [PubMed] [Google Scholar]
- 11.Cacciarelli TV, Martinez OM, Gish RG, et al. Immunoregulatory cytokines in chronic hepatitis C virus infection: pre- and posttreatment with interferon alfa. Hepatology. 1996;24:6–9. doi: 10.1002/hep.510240102. [DOI] [PubMed] [Google Scholar]
- 12.Tokumoto Y, Hiasa Y, Uesugi K, et al. Ribavirin regulates hepatitis C virus replication through enhancing interferon-stimulated genes and interleukin 8. J Infect Dis. 2012;205:1121–1130. doi: 10.1093/infdis/jis025. [DOI] [PubMed] [Google Scholar]
- 13.Rigopoulou EI, Abbott WG, Williams R, Naoumov NV. Direct evidence for immunomodulatory properties of ribavirin on T-cell reactivity to hepatitis C virus. Antiviral Res. 2007;75:36–42. doi: 10.1016/j.antiviral.2006.11.008. [DOI] [PubMed] [Google Scholar]
- 14.Brenndörfer ED, Brass A, Karthe J, et al. Cleavage of the T cell protein tyrosine phosphatase by the hepatitis C virus nonstructural 3/4A protease induces a Th1 to Th2 shift reversible by ribavirin therapy. J Immunol. 2014;192:1671–1680. doi: 10.4049/jimmunol.1301077. [DOI] [PubMed] [Google Scholar]
- 15.Falconer K, Askarieh G, Weis N, et al. IP-10 predicts the first phase decline of HCV RNA and overall viral response to therapy in patients co-infected with chronic hepatitis C virus infection and HIV. Scand J Infect Dis. 2010;42:896–901. doi: 10.3109/00365548.2010.498019. [DOI] [PubMed] [Google Scholar]
- 16.Zeremski M, Markatou M, Brown QB, et al. Interferon gamma-inducible protein 10: a predictive marker of successful treatment response in hepatitis C virus/HIV-coinfected patients. J Acquir Immune Defic Syndr. 2007;45:262–268. doi: 10.1097/QAI.0b013e3180559219. [DOI] [PubMed] [Google Scholar]
- 17.Thomson EC, Fleming VM, Main J, et al. Predicting spontaneous clearance of acute hepatitis C virus in a large cohort of HIV-1-infected men. Gut. 2011;60:837–845. doi: 10.1136/gut.2010.217166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Brockman MA, Kwon DS, Tighe DP, et al. IL-10 is up-regulated in multiple cell types during viremic HIV infection and reversibly inhibits virus-specific T cells. Blood. 2009;114:346–356. doi: 10.1182/blood-2008-12-191296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Porichis F, Hart MG, Zupkosky J, et al. Differential impact of PD-1 and/or interleukin-10 blockade on HIV-1-specific CD4 T cell and antigen-presenting cell functions. J Virol. 2014;88:2508–2518. doi: 10.1128/JVI.02034-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Grabmeier-Pfistershammer K, Steinberger P, Rieger A, et al. Identification of PD-1 as a unique marker for failing immune reconstitution in HIV-1-infected patients on treatment. J Acquir Immune Defic Syndr. 2011;56:118–124. doi: 10.1097/QAI.0b013e3181fbab9f. [DOI] [PubMed] [Google Scholar]
- 21.Spitsin S, Tustin NB, Riedel E, et al. Programmed death 1 receptor changes ex vivo in HIV-infected adults following initiation of highly active antiretroviral therapy. Clin Vaccine Immunol. 2012;19:752–756. doi: 10.1128/CVI.00093-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cockerham LR1, Jain V, Sinclair E, et al. Programmed death-1 expression on CD4? and CD8? T cells in treated and untreated HIV disease. AIDS. 2014;28:1749–1758. doi: 10.1097/QAD.0000000000000314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Rubbo PA, Van de Perre P, Tuaillon E. The long way toward understanding host and viral determinants of therapeutic success in HCV infection. Hepatol Int. 2012;6:436–440. doi: 10.1007/s12072-011-9339-y. [DOI] [PubMed] [Google Scholar]
- 24.Tuaillon E, Al Tabaa Y, Baillat V, et al. Close association of CD8+/CD38 bright with HIV-1 replication and complex relationship with CD4+ T-cell count. Cytometry B Clin Cytom. 2009;76:249–260. doi: 10.1002/cyto.b.20467. [DOI] [PubMed] [Google Scholar]
- 25.Djoba Siawaya JF, Roberts T, Babb C, et al. An evaluation of commercial fluorescent bead-based luminex cytokine assays. PloS ONE. 2008;3:e2535. doi: 10.1371/journal.pone.0002535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Dupuy AM, Kuster N, Lizard G, et al. Performance evaluation of human cytokines profiles obtained by various multiplexed-based technologies underlines a need for standardization. Clin Chem Lab Med. 2013;51:1385–1393. doi: 10.1515/cclm-2012-0648. [DOI] [PubMed] [Google Scholar]
- 27.Askarieh G, Alsio A, Pugnale P, et al. Systemic and intrahepatic interferon-gamma-inducible protein 10kDa predicts the first-phase decline in hepatitis C virus RNA and overall viral response to therapy in chronic hepatitis C. Hepathology. 2010;51:1523–1530. doi: 10.1002/hep.23509. [DOI] [PubMed] [Google Scholar]
- 28.Sarasin-Filipowicz M, Oakeley EJ, et al. Interferon signaling and treatment outcome in chronic hepatitis C. Proc Natl Acad Sci U S A. 2008;105:7034–7039. doi: 10.1073/pnas.0707882105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Feld JJ, Nanda S, Huang Y, et al. Hepatic gene expression during treatment with peginterferon and ribavirin: identifying molecular pathways for treatment response. Hepathology. 2007;46:1548–1563. doi: 10.1002/hep.21853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Dill MT, Duong FH, Vogt JE, et al. Interferon-induced gene expression is a stronger predictor of treatment response than IL28B genotype in patients with hepatitis C. Gastroenterology. 2011;140:1021–1031. doi: 10.1053/j.gastro.2010.11.039. [DOI] [PubMed] [Google Scholar]
- 31.Chen L, Borozan I, Feld J, et al. Hepatic gene expression discriminates responders and nonresponders in treatment of chronic hepatitis C viral infection. Gastroenterology. 2005;128:1437–1444. doi: 10.1053/j.gastro.2005.01.059. [DOI] [PubMed] [Google Scholar]
- 32.Micheloud D, Salcedo M, Banares R, et al. Serum levels of fibrosis biomarkers measured early after liver transplantation are associated with severe hepatitis C virus recurrence. Transpl Infect Dis. 2009;11:183–188. doi: 10.1111/j.1399-3062.2009.00370.x. [DOI] [PubMed] [Google Scholar]
- 33.Mühlbauer M, Bosserhoff AK, Hartmann A, et al. A novel MCP-1 gene polymorphism is associated with hepatic MCP-1 expression and severity of HCV-related liver disease. Gastroenterology. 2003;125:1085–1093. doi: 10.1016/S0016-5085(03)01213-7. [DOI] [PubMed] [Google Scholar]
- 34.El-Bassiouni NE, Mahmoud OM, El Ahwani EG, Ibrahim RA, El Bassiouny AE. Monocyte adhesion molecules expression in patients with chronic hepatitis C liver disease. Mediterr J Hematol Infect Dis. 2013;5(1):e2013054. doi: 10.4084/mjhid.2013.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Gu B, Ye B, Mao WL, Ye JL. Monocyte chemotactic protein-1 as possible prognostic markers of the efficacy of antiviral treatment in chronic hepatitis C. Hepatogastroenterology. 2014;61:55–58. [PubMed] [Google Scholar]
- 36.Heydtmann M, Adams DH. Chemokines in the immunopathogenesis of hepatitis C infection. Hepatology. 2009;49:676–688. doi: 10.1002/hep.22763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Tuyama AC, Hong F, Saiman Y, et al. Human immunodeficiency virus (HIV)-1 infects human hepatic stellate cells and promotes collagen I and monocyte chemoattractant protein-1 expression: implications for the pathogenesis of HIV/hepatitis C virus-induced liver fibrosis. Hepatology. 2010;52:612–22. doi: 10.1002/hep.23679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Hong F, Bansal MB. HIV gp120(X4) promotes hepatic stellate cell activation, fibrogenesis, and proliferation: a potential mechanism for rapid fibrosis progression in HIV/HCV coinfected patients. Hepathology. 2009;51:676–688. [Google Scholar]
- 39.Weiss JM, Nath A, Major EO, Berman JW. HIV-1 Tat induces monocyte chemoattractant protein-1-mediated monocyte transmigration across a model of the human blood-brain barrier and up-regulates CCR5 expression on human monocytes. J Immunol. 1999;163:2953–2959. [PubMed] [Google Scholar]
- 40.Kim BO, Liu Y, Zhou BY, He JJ. Induction of C chemokine XCL1 (lymphotactin/single C motif-1 alpha/activation-induced, T cell-derived and chemokine-related cytokine) expression by HIV-1 Tat protein. J Immunol. 2004;172:1888–1895. doi: 10.4049/jimmunol.172.3.1888. [DOI] [PubMed] [Google Scholar]
- 41.Gu L, Tseng S, Horner RM, et al. Control of TH2 polarization by the chemokine monocyte chemoattractant protein-1. Nature. 2000;404:407–411. doi: 10.1038/35006097. [DOI] [PubMed] [Google Scholar]
- 42.Ansari AW, Heiken H, Meyer-Olson D, Schmidt RE. CCL2: a potential prognostic marker and target of anti-inflammatory strategy in HIV/AIDS pathogenesis. Eur J Immunol. 2011;41:3412–3418. doi: 10.1002/eji.201141676. [DOI] [PubMed] [Google Scholar]
- 43.Zhang Y, Guo D, Zhao Y, et al. The effect of cytokine profiles on the viral response to re-treatment in antiviral-experienced patients with chronic hepatitis C virus infection. Antiviral Res. 2011;92:247–254. doi: 10.1016/j.antiviral.2011.08.009. [DOI] [PubMed] [Google Scholar]
- 44.Florholmen J, Kristiansen MG, Steigen SE, et al. A rapid chemokine response of macrophage inflammatory protein (MIP)-1a, MIP-1β and the regulated on activation, normal T expressed and secreted chemokine is associated with a sustained virological response in the treatment of chronic hepatitis C. Clin Microbiol Infect. 2011;17:204–209. doi: 10.1111/j.1469-0691.2010.03206.x. [DOI] [PubMed] [Google Scholar]
- 45.Shimoda K, Begum NA, Shibuta K, et al. Interleukin-8 and hIRH (SDF1-alpha/PBSF) mRNA expression and histological activity index in patients with chronic hepatitis C. Hepatology. 1998;28:108–115. doi: 10.1002/hep.510280116. [DOI] [PubMed] [Google Scholar]
- 46.Mahmood S, Sho M, Yasuhara Y, et al. Clinical significance of intrahepatic interleukin-8 in chronic hepatitis C patients. Hepatol Res. 2002;24:413–419. doi: 10.1016/S1386-6346(02)00136-5. [DOI] [PubMed] [Google Scholar]
- 47.Asselah T, Bièche I, Laurendeau I, et al. Liver gene expression signature of mild fibrosis in patients with chronic hepatitis C. Gastroenterology. 2005;129:2064–2075. doi: 10.1053/j.gastro.2005.09.010. [DOI] [PubMed] [Google Scholar]
- 48.Polyak SJ, Khabar KS, Paschal DM, et al. Hepatitis C virus nonstructural 5A protein induces interleukin-8, leading to partial inhibition of the interferon-induced antiviral response. J Virol. 2001;75:6095–6106. doi: 10.1128/JVI.75.13.6095-6106.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Akbar H, Idrees M, Butt S, et al. High baseline interleukine-8 level is an independent risk factor for the achievement of sustained virological response in chronic HCV patients. Infect Genet Evol. 2011;11:1301–1305. doi: 10.1016/j.meegid.2011.04.021. [DOI] [PubMed] [Google Scholar]
- 50.Feng B, Yang RF, Zhang HY, et al. Early predictive efficacy of core antigen on antiviral outcomes in genotype 1 hepatitis C virus infected patients. Braz J Infect Dis. 2015;19:390–398. doi: 10.1016/j.bjid.2015.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Dieterich DT1, Rizzetto M, Manns MP. Management of chronic hepatitis C patients who have relapsed or not responded to pegylated interferon alfa plus ribavirin. J Viral Hepat. 2009;16:833–843. doi: 10.1111/j.1365-2893.2009.01218.x. [DOI] [PubMed] [Google Scholar]