Acute lymphoblastic leukemia (ALL) is the most common hematologic malignancy in children1. Prolongation of therapy by incorporating a maintenance phase, containing 6-mercaptopurine (6-MP) as the backbone, has improved treatment outcomes in pediatric ALL2–4. However, 6-MP can cause critical side effects such as myelosuppression and abnormalities in liver function. This may lead to treatment interruptions, and subsequently contribute to an increased incidence of late relapse4,5. The pharmacogenetics of 6-MP has been studied extensively. Polymorphisms in the thiopurine methyltransferase (TPMT) gene have a well-established effect on myelosuppression from 6-MP. A clinical guideline for dose adjustment of 6-MP according to types of TPMT polymorphisms has been developed and is being used in most modern pediatric ALL protocols6. However, the frequencies of TPMT variants associated with thiopurine-induced myelosuppression in most Asian populations, including Thai, are low7–9. Determination of TPMT polymorphisms, therefore, has limited clinical benefits to children with ALL in most Asian countries. More recently, single nucleotide polymorphisms (SNPs) of inosine triphosphate pyrophosphohydrolase (ITPA) and nucleoside diphophate–linked moiety X-type motif 15 (NUDT15) have been shown to be associated with myelosuppression induced by 6-MP or other drugs in the thiopurine group, in both benign and malignant conditions10–14.
We present data on the association between the ITPA c.94C>A (rs1127354) and NUDT15 c.415C>T (rs116855232) and 6-MP induced myelosuppression in our cohort of pediatric ALL children. Patients aged between 1 and 15 years old, who received maintenance therapy and were followed-up at King Chulalongkorn Memorial Hospital between January 2014 and January 2015, were recruited. All patients were treated according to the Thai Pediatric Oncology Group (Thai-POG) protocol for childhood ALL15. The Thai-POG protocol is based on the modified Berlin-Frankfurt-Munster (BFM) protocol for childhood ALL. Patients were initially categorized into different risk groups: standard-risk, high-risk and very high-risk, using age at diagnosis and initial white blood cell count according to the National Cancer Institute (NCI)/Rome criteria for pediatric ALL risk classification, with the addition of cytogenetic information. In brief, the Thai-POG treatment regimen consists of five phases of therapy including remission induction, consolidation, interim maintenance, delayed intensification and maintenance phase. The total duration of therapy is two and a half years for girls and three and a half years for boys.
Essentially, medications given during the maintenance phase included monthly intravenous vincristine, a monthly pulse of prednisolone, weekly oral methotrexate, daily oral 6-MP and intrathecal methotrexate once every three months. The initial 6-MP dose was 50 mg/m2. A complete blood count (CBC) was performed at a 4-week interval. 6-MP was either increased by 25% of the previous dose or discontinued, to keep an absolute neutrophil count (ANC) of between 500–1,500 cells/μL.15 The ITPA c.94C>A and NUDT15 c.415C>T were evaluated by pyrosequencing using 5′-GACCAGCTTTTCTGGGGACTG-3′ and biotinylated 5′-GGCTGAAAGAGTGGGGGATAC-3′, and 5′-GACCAGCTTTTCTGGGGACTG-3′ and biotinylated 5′-GGCTGAAAGAGTGGGGGATAC-3′, respectively.
We retrospectively analyzed the myelosuppressive effect of 6-MP at months 2, 4 and 6 of the maintenance phase between the SNP groups in addition to comparing the median cumulative dose of 6-MP at each time point.
Out of 82 patients, the median age at diagnosis was 5.4 years old. The female to male ratio was 1.5 to 1. 50% of patients were standard-risk, whereas 30% and 20% of patients were high-risk and very high-risk, respectively. The median duration of follow-up in this study was 8 years (1 to 20 years) from diagnosis. Genotypic determination of the ITPA c.94C>A showed that 46 (56.1%), 33 (40.2%), and 3 (3.7%) patients were CC, CA, and AA, respectively. Of the NUDT15 c.415C>T, 70 (85.4%), 10 (12.2%), and 2 (2.4%) patients were CC, CT, and TT, respectively. These were in the Hardy-Weinberg equilibrium.
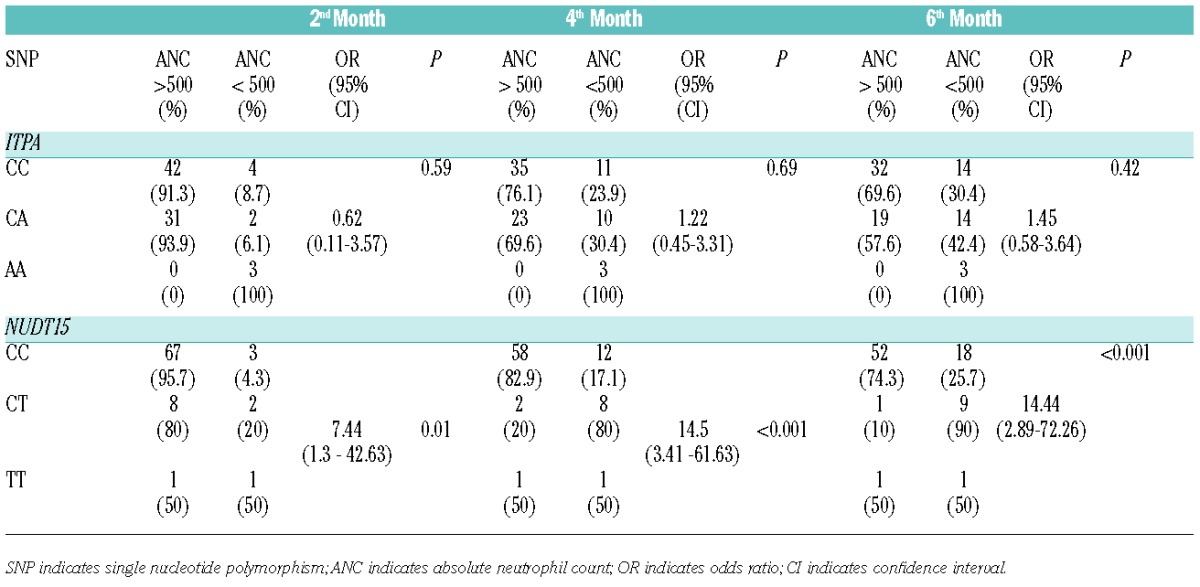
Genetic polymorphism of ITPA showed no significant difference between genotypes, and 6-MP induced myelosuppression at any time point during maintenance (Table 1). However, the NUDT15 c.415C>T was strongly associated with 6-MP induced early myelosuppression in the second, fourth, and sixth month. Patients with NUDT15 CT or TT had a significantly increased risk of neutropenia as early as two months after 6-MP administration with OR of 7.4 (95% CI 1.3–42.6). Longer follow-up duration at the fourth and sixth month showed even higher risks, with OR of 14.5 (95% CI 3.4–61.6) and 14.4 (95% CI 2.9–72.3), respectively (Table 1).
Table 1.
Associations between myelosuppression at the second, fourth and sixth month following the initiation of 6-MP during maintenance therapy in children with ALL and SNPs of the ITPA and NUDT15.
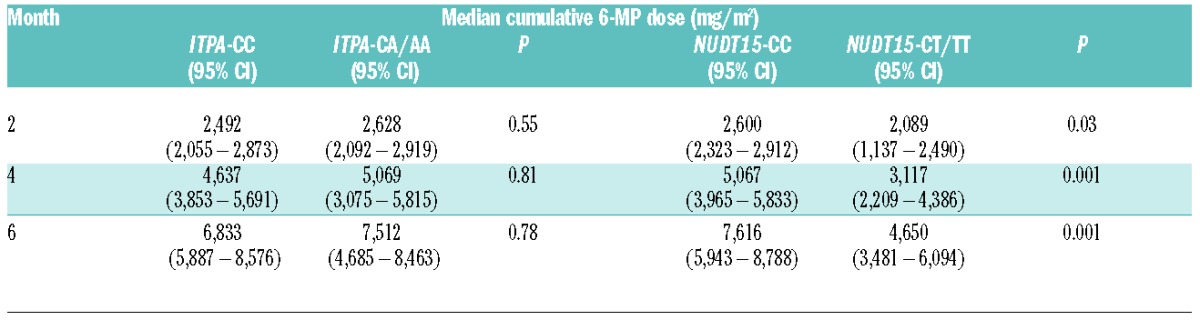
According to the Thai-POG ALL protocol, we adjusted 6-MP dosage according to ANC levels, which were determined every 4 weeks. Our patients with different ITPA polymorphisms had no significant difference in the median cumulative 6-MP dose at any time point. On the contrary, the median cumulative doses of 6-MP of 2, 4, and 6 months’ duration in the CT or TT groups of NUDT15 were 80.3%, 61.5%, and 61.1%, respectively, of those with the CC genotype (Table 2). For the 6-month duration of 6-MP administration, our patients with NUDT15 CT or TT received between 45.7% (3481/7616) and 80% (6094/7616) of the median cumulative 6-MP dose of those with the CC genotype.
Table 2.
Comparison of the median cumulative 6-MP doses (mg/m2) of 2, 4, and 6 months’ duration following 6-MP initiation among ALL patients with different genotypes of ITPA c.94C>A and NUDT15 c.415C>T.
Moreover, at the sixth month following 6-MP initiation, our patients with NUDT15 CT and TT were given a median 6-MP dose of 28 mg/m2/day (range: 6–52 mg/m2/day), which was only 56% of the standard 6-MP initial dose of 50 mg/m2/day in the Thai population. Notably, our two patients with the homozygous TT genotype received 6 and 36 mg/m2/day of 6-MP at the sixth month following 6-MP initiation, which were 12% and 72% of the standard 6-MP initial dose in the Thai ALL protocol. 6-MP was continued at 36 mg/m2/day in the latter patient throughout the treatment period without any additional episode of severe neutropenia. Interestingly, the homozygous TT patient who received 6 mg/m2/day of 6-MP was heterozygous for ITPA c.94C>A, while the other carried the wild-type CC genotype. The discrepancy of final 6-MP dosage between these two patients with homozygous TT genotype could be caused by multifactorial elements, including epistatic effects from other modifier genes such as ITPA. In addition, we found that a total of six patients had a history of relapse. Two out of these six patients demonstrated a heterozygous mutation of the NUDT15 c.415C>T. Due to the limited sample size, however, our study did not have enough resources to show a statistically significant correlation between NUDT15 polymorphism and the relapse rate.
Our study shows that NUDT15 c.415C>T has a strong effect on early myelosuppression from 6-MP in Asian children with ALL. The results were consistent with a recent study in Korean patients with inflammatory bowel disease, which showed that NUDT15 c.415C>T was associated with 5-azathioprine-induced leukopenia within 8 weeks of the initiation with an OR of 35.6.12 The 6-MP dose in the variant group of NUDT15 c.415C>T was significantly lower compared to that in the wild-type group, especially in the homozygous group. Recently, a genome-wide association study (GWAS) in children with ALL also showed that the NUDT15 c.415C>T was a determinant for 6-MP leukopenia. Their patients with the homozygous NUDT15 TT variant could tolerate only 8.3% of their standard 6-MP dose, which was 75 mg/m2/day.13 Moreover, a recent study in Japanese children with ALL showed significant 6-MP dose reductions of 42% and 82% in patients with the CT and TT genotypes, respectively.14
In conclusion, more studies on the role of NUDT15 c.415C>T in 6-MP metabolism, the proper dose adjustment in patients with heterozygous and homozygous variants, 6-MP pharmacogenetics in different ethnicities, as well as a systematic evaluation of the cost-effectiveness of NUDT15 screening are warranted.
Footnotes
Funding: this study was supported by the Ratchadapiseksomphot Endowment Fund of Chulalongkorn University (RES560530177-HR) and the Thailand Research Fund (RTA5680003).
Information on authorship, contributions, and financial & other disclosures was provided by the authors and is available with the online version of this article at www.haematologica.org.
References
- 1.Hunger SP, Loh ML, Whitlock JA, et al. Children’s Oncology Group’s 2013 blueprint for research: acute lymphoblastic leukemia. Pediatr Blood Cancer. 2013;60(6):957–963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Simone JV. The treatment of acute lymphoblastic leukaemia. Br J Haematol. 1980;45(1):1–4. [DOI] [PubMed] [Google Scholar]
- 3.Holland JF, Glidewell O. Chemotherapy of acute lymphocytic leukemia of childhood. Cancer. 1972;30(6):1480–1487. [DOI] [PubMed] [Google Scholar]
- 4.Schmiegelow K, Nielsen SN, Frandsen TL, Nersting J. Mercaptopurine/Methotrexate maintenance therapy of childhood acute lymphoblastic leukemia: clinical facts and fiction. J Pediatr Hematol Oncol. 2014;36(7):503–517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Relling MV, Hancock ML, Boyett JM, Pui CH, Evans WE. Prognostic importance of 6-mercaptopurine dose intensity in acute lymphoblastic leukemia. Blood. 1999;93(9):2817–2823. [PubMed] [Google Scholar]
- 6.Lennard L. Implementation of TPMT testing. Br J Clin Pharmacol. 2014;77(4):704–714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Chang JG, Lee LS, Chen CM, et al. Molecular analysis of thiopurine S-methyltransferase alleles in South-east Asian populations. Pharmacogenetics. 2002;12(3):191–195. [DOI] [PubMed] [Google Scholar]
- 8.Srimartpirom S, Tassaneeyakul W, Kukongviriyapan V. Thiopurine S-methyltransferase genetic polymorphism in the Thai population. Br J Clin Pharmacol. 2004;58(1):66–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Vannaprasaht S, Angsuthum S, Avihingsanon Y, et al. Impact of the heterozygous TPMT*1/*3C genotype on azathioprine-induced myelosuppression in kidney transplant recipients in Thailand. Clin Ther. 2009;31(7):1524–1533. [DOI] [PubMed] [Google Scholar]
- 10.Wan Rosalina WR, Teh LK, Mohamad N, et al. Polymorphism of ITPA 94C>A and risk of adverse effects among patients with acute lymphoblastic leukaemia treated with 6-mercaptopurine. J Clin Pharm Ther. 2012;37(2):237–241. [DOI] [PubMed] [Google Scholar]
- 11.Tanaka Y, Manabe A, Nakadate H, et al. The activity of the inosine triphosphate pyrophosphatase affects toxicity of 6-mercaptopurine during maintenance therapy for acute lymphoblastic leukemia in Japanese children. Leuk Res. 2012;36(5):560–564. [DOI] [PubMed] [Google Scholar]
- 12.Yang SK, Hong M, Baek J, et al. A common missense variant in NUDT15 confers susceptibility to thiopurine-induced leukopenia. Nat Genet. 2014;46(9):1017–1020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Yang JJ, Landier W, Yang W, et al. Inherited NUDT15 Variant Is a Genetic Determinant of Mercaptopurine Intolerance in Children With Acute Lymphoblastic Leukemia. J Clin Oncol. 2015; 33(11):1235–1242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tanaka Y, Kato M, Hasegawa D, et al. Susceptibility to 6-MP toxicity conferred by a NUDT15 variant in Japanese children with acute lymphoblastic leukemia. Br J Haematol. 2015;171(1):109–115. [DOI] [PubMed] [Google Scholar]
- 15.Kanjanapongkul S. Outcome of acute lymphoblastic leukemia treatment using national protocols at the Queen Sirikit National Institute of Child Health. J Med Assoc Thai. 2014;97(6):S1–5. [PubMed] [Google Scholar]


