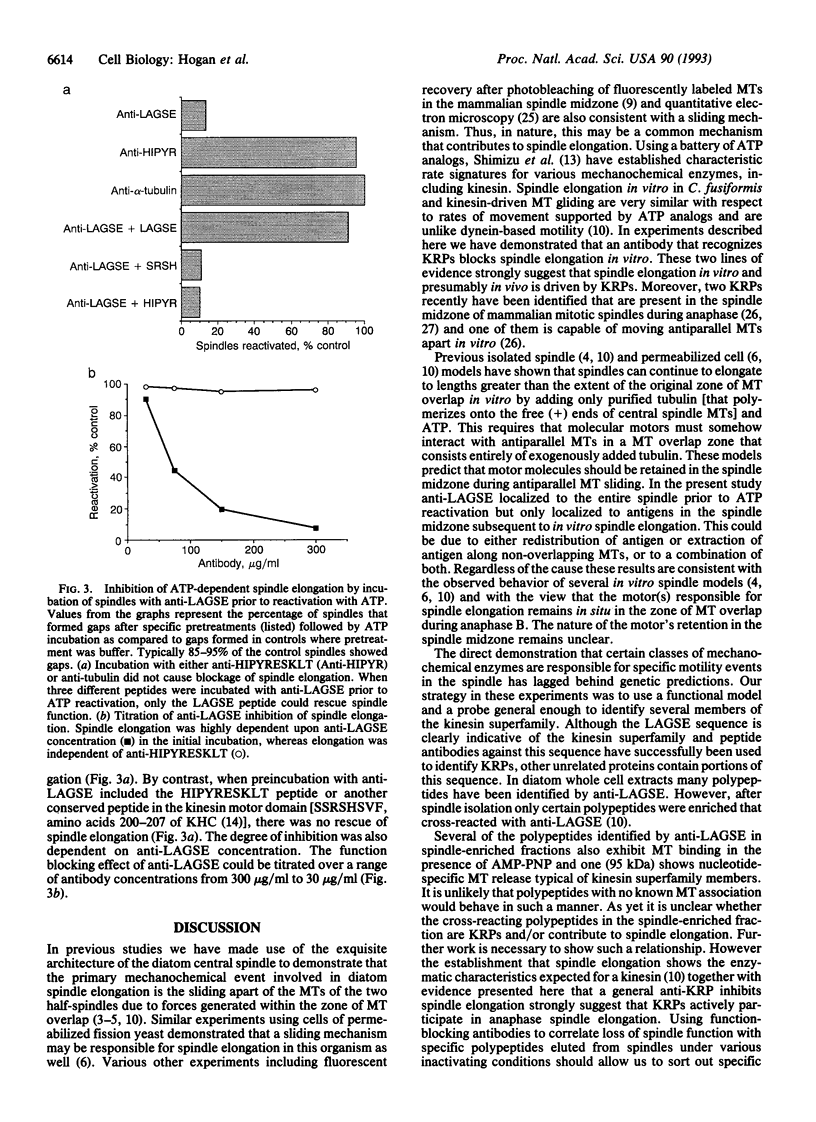
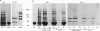Abstract
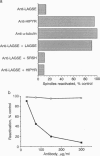
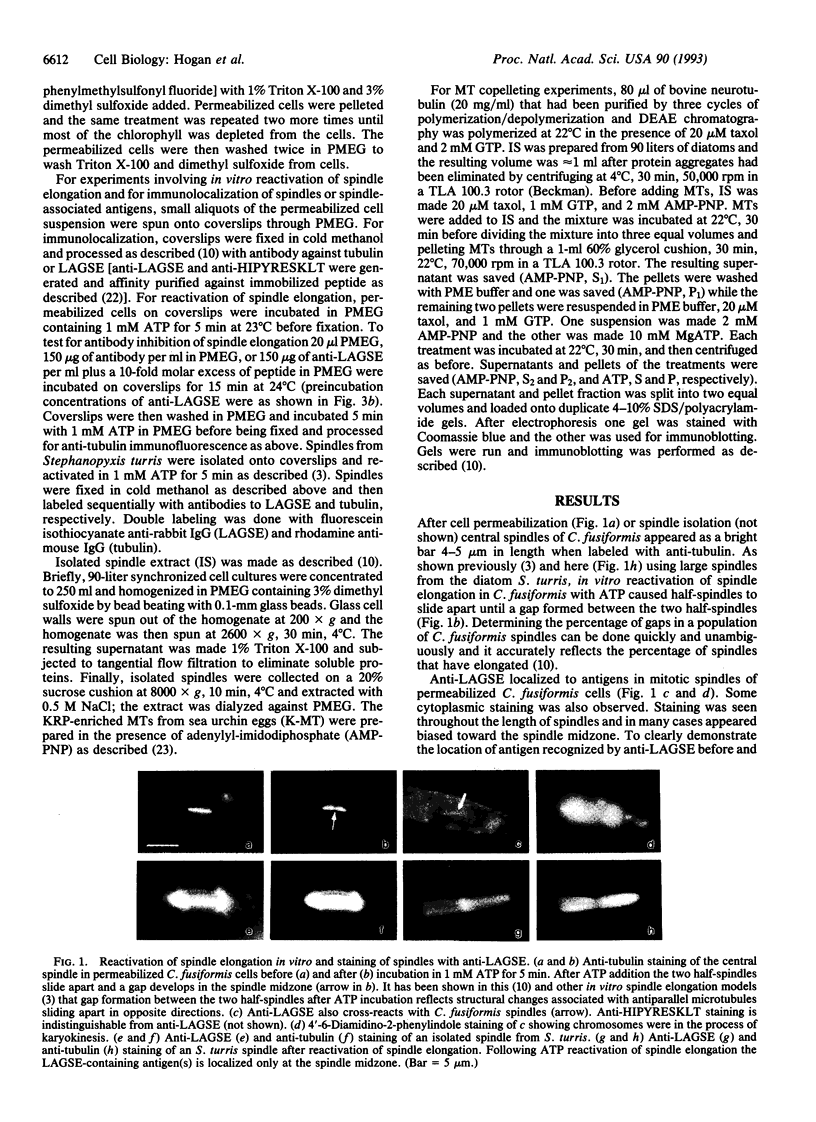
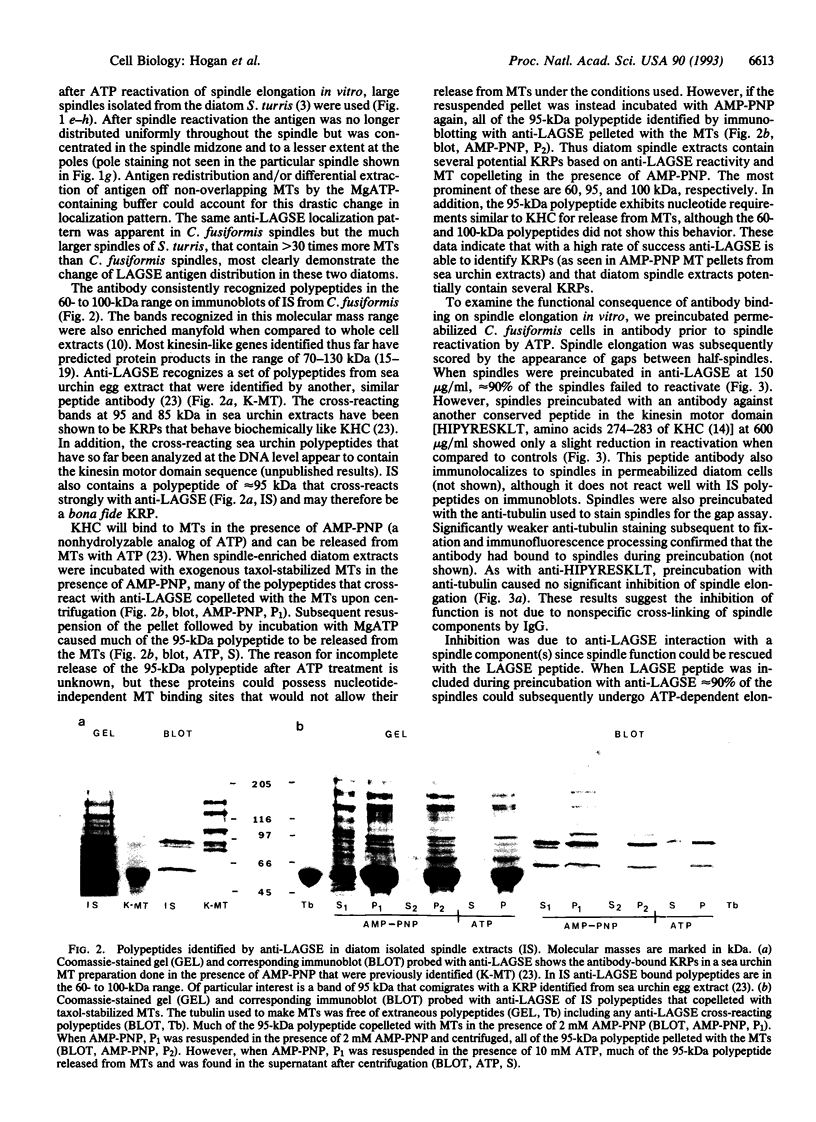
Isolated central spindles or spindles in detergent-permeabilized cells from the diatom Cylindrotheca fusiformis can undergo ATP-dependent reactivation of spindle elongation in vitro. We have used a peptide antibody raised against a 10-amino acid portion common to the kinesin superfamily motor domain to look for kinesin-like motor activity during anaphase B of mitosis. The peptide antibody localizes to central spindles. Upon ATP reactivation of spindle elongation, antigens recognized by the antibody are associated exclusively with the central spindle midzone where antiparallel microtubules of each half-spindle overlap. The antibody recognizes several polypeptides by immunoblot using isolated spindle extracts. One of these polypeptides behaves like kinesin with respect to nucleotide-specific binding to and release from taxol-stabilized microtubules. Preincubation of the spindle model with the peptide antibody inhibits subsequent ATP reactivation of spindle elongation. Coincubation of the peptide antibody with peptide antigen rescues spindle function. These results support a role for kinesin-related protein(s) in spindle elongation (anaphase B) of mitosis and suggest that one or several polypeptides that we have identified in spindle extracts may fulfill this function.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baskin T. I., Cande W. Z. Kinetic analysis of mitotic spindle elongation in vitro. J Cell Sci. 1990 Sep;97(Pt 1):79–89. doi: 10.1242/jcs.97.1.79. [DOI] [PubMed] [Google Scholar]
- Cande W. Z., McDonald K. L. In vitro reactivation of anaphase spindle elongation using isolated diatom spindles. Nature. 1985 Jul 11;316(6024):168–170. doi: 10.1038/316168a0. [DOI] [PubMed] [Google Scholar]
- Cande W. Z., McDonald K. Physiological and ultrastructural analysis of elongating mitotic spindles reactivated in vitro. J Cell Biol. 1986 Aug;103(2):593–604. doi: 10.1083/jcb.103.2.593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cande W. Z. Nucleotide requirements for anaphase chromosome movements in permeabilized mitotic cells: anaphase B but not anaphase A requires ATP. Cell. 1982 Jan;28(1):15–22. doi: 10.1016/0092-8674(82)90370-1. [DOI] [PubMed] [Google Scholar]
- Cole D. G., Cande W. Z., Baskin R. J., Skoufias D. A., Hogan C. J., Scholey J. M. Isolation of a sea urchin egg kinesin-related protein using peptide antibodies. J Cell Sci. 1992 Feb;101(Pt 2):291–301. doi: 10.1242/jcs.101.2.291. [DOI] [PubMed] [Google Scholar]
- Endow S. A., Hatsumi M. A multimember kinesin gene family in Drosophila. Proc Natl Acad Sci U S A. 1991 May 15;88(10):4424–4427. doi: 10.1073/pnas.88.10.4424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Endow S. A., Henikoff S., Soler-Niedziela L. Mediation of meiotic and early mitotic chromosome segregation in Drosophila by a protein related to kinesin. Nature. 1990 May 3;345(6270):81–83. doi: 10.1038/345081a0. [DOI] [PubMed] [Google Scholar]
- Enos A. P., Morris N. R. Mutation of a gene that encodes a kinesin-like protein blocks nuclear division in A. nidulans. Cell. 1990 Mar 23;60(6):1019–1027. doi: 10.1016/0092-8674(90)90350-n. [DOI] [PubMed] [Google Scholar]
- Hagan I., Yanagida M. Novel potential mitotic motor protein encoded by the fission yeast cut7+ gene. Nature. 1990 Oct 11;347(6293):563–566. doi: 10.1038/347563a0. [DOI] [PubMed] [Google Scholar]
- Hogan C. J., Cande W. Z. Antiparallel microtubule interactions: spindle formation and anaphase B. Cell Motil Cytoskeleton. 1990;16(2):99–103. doi: 10.1002/cm.970160203. [DOI] [PubMed] [Google Scholar]
- Hogan C. J., Stephens L., Shimizu T., Cande W. Z. Physiological evidence for involvement of a kinesin-related protein during anaphase spindle elongation in diatom central spindles. J Cell Biol. 1992 Dec;119(5):1277–1286. doi: 10.1083/jcb.119.5.1277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Masuda H., Cande W. Z. The role of tubulin polymerization during spindle elongation in vitro. Cell. 1987 Apr 24;49(2):193–202. doi: 10.1016/0092-8674(87)90560-5. [DOI] [PubMed] [Google Scholar]
- Masuda H., Hirano T., Yanagida M., Cande W. Z. In vitro reactivation of spindle elongation in fission yeast nuc2 mutant cells. J Cell Biol. 1990 Feb;110(2):417–425. doi: 10.1083/jcb.110.2.417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meluh P. B., Rose M. D. KAR3, a kinesin-related gene required for yeast nuclear fusion. Cell. 1990 Mar 23;60(6):1029–1041. doi: 10.1016/0092-8674(90)90351-e. [DOI] [PubMed] [Google Scholar]
- Nislow C., Lombillo V. A., Kuriyama R., McIntosh J. R. A plus-end-directed motor enzyme that moves antiparallel microtubules in vitro localizes to the interzone of mitotic spindles. Nature. 1992 Oct 8;359(6395):543–547. doi: 10.1038/359543a0. [DOI] [PubMed] [Google Scholar]
- Pickett-Heaps J. D., Tippit D. H. The diatom spindle in perspective. Cell. 1978 Jul;14(3):455–467. doi: 10.1016/0092-8674(78)90232-5. [DOI] [PubMed] [Google Scholar]
- Sawin K. E., Mitchison T. J., Wordeman L. G. Evidence for kinesin-related proteins in the mitotic apparatus using peptide antibodies. J Cell Sci. 1992 Feb;101(Pt 2):303–313. doi: 10.1242/jcs.101.2.303. [DOI] [PubMed] [Google Scholar]
- Saxton W. M., McIntosh J. R. Interzone microtubule behavior in late anaphase and telophase spindles. J Cell Biol. 1987 Aug;105(2):875–886. doi: 10.1083/jcb.105.2.875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimizu T., Furusawa K., Ohashi S., Toyoshima Y. Y., Okuno M., Malik F., Vale R. D. Nucleotide specificity of the enzymatic and motile activities of dynein, kinesin, and heavy meromyosin. J Cell Biol. 1991 Mar;112(6):1189–1197. doi: 10.1083/jcb.112.6.1189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stewart R. J., Pesavento P. A., Woerpel D. N., Goldstein L. S. Identification and partial characterization of six members of the kinesin superfamily in Drosophila. Proc Natl Acad Sci U S A. 1991 Oct 1;88(19):8470–8474. doi: 10.1073/pnas.88.19.8470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wright B. D., Henson J. H., Wedaman K. P., Willy P. J., Morand J. N., Scholey J. M. Subcellular localization and sequence of sea urchin kinesin heavy chain: evidence for its association with membranes in the mitotic apparatus and interphase cytoplasm. J Cell Biol. 1991 May;113(4):817–833. doi: 10.1083/jcb.113.4.817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yen T. J., Li G., Schaar B. T., Szilak I., Cleveland D. W. CENP-E is a putative kinetochore motor that accumulates just before mitosis. Nature. 1992 Oct 8;359(6395):536–539. doi: 10.1038/359536a0. [DOI] [PubMed] [Google Scholar]
- Zhang P., Knowles B. A., Goldstein L. S., Hawley R. S. A kinesin-like protein required for distributive chromosome segregation in Drosophila. Cell. 1990 Sep 21;62(6):1053–1062. doi: 10.1016/0092-8674(90)90383-p. [DOI] [PubMed] [Google Scholar]