Abstract
Cadmium (Cd), is one of the most hazardous metals found in the environment. Cd exposure through inhalation has been linked to various diseases in lungs. It was shown that Cd induces pro-inflammatory cytokines through oxidative stress mechanism. In this report, we studied the immuno-modulatory effect of a well known antioxidant, N-acetylcysteine (NAC) on cadmium chloride (CdCl2) treated human lung A549 cells through human cytokine array 6. The lung cells were treated with 0, or 75 μM CdCl2 alone, 2.5 mM NAC alone, or co-treated with 2.5 mM NAC and 75 μM CdCl2 for 24 h. The viability of cells was measured by crystal violet dye. The array results were validated by human IL-1alpha enzyme-linked immunosorbent assay (ELISA) kit. The viability of the 75 μM CdCl2 alone treated cells was decreased to 44.5%, while the viability of the co-treated cells with 2.5 mM NAC was increased to 84.1% in comparison to untreated cells. In the cell lysate of CdCl2 alone treated cells, 19 and 8 cytokines were up and down-regulated, while in the medium 15 and 3 cytokines were up and down-regulated in comparison to the untreated cells. In the co-treated cells, all these cytokines expression was modulated by the NAC treatment. The IL-1α ELISA result showed the same pattern of cytokine expression as the cytokine array. This study clearly showed the modulatory effect of NAC on cytokines and chemokines expression in CdCl2-treated cells and suggests the use of NAC as protective agent against cadmium toxicity.
Keywords: NAC, immune-modulation, cytokines, cadmium, human lung cells
Introduction
The tremendous global industrialization that led to the increased exploitation of Cadmium (Cd) containing compounds has resulted in the exposure of Cd not only to the industrial workers but also to the general population. Inhalation of Cd containing compounds in smoke from industrial waste is one of the major routes of Cd exposure in both humans and animals. Cigarette smoke also contains Cd and has been shown that humans that smoke one packet of cigarettes per day absorb approximately 0.5–3.0 μg Cd into the lungs (ATSDR, 2012). Cd causes pathologic changes to the airway system which is the cause of morbidity and mortality to the humans worldwide. Absorption of Cd into the lungs has been linked to major cause of chronic obstructive pulmonary diseases (COPD) (Cullinan, 2012; Oh et al., 2014), and even lung cancer in populations exposed occupationally or living in a Cd-polluted area (Nawrot et al., 2006, Park et al., 2012; Hartwig, 2013; Jarup et al., 1998).
Inflammation is a natural defense mechanism against the xenobiotics in mammalian systems. Cells produce small signaling molecules called cytokines that influx phagocytes or macrophages to the site of infection or damaged tissue. However, chronic inflammation induced by toxic chemicals may lead to permanent tissue damage through the continuous release of endogenous oxidants and toxic proteases by activated immune cells. Both animal and human epidemiologic data indicate that Cd exposure is associated with oxidative stress and chronic inflammation (Kirschvink et al., 2006; Kundu et al., 2011). Not only this, the over expression of cytokines as a result of chronic exposure of Cd in lungs, has been linked to various lung diseases in humans (Lag et al., 2010; Kundu et al., 2011; Rennolds et al., 2012).
NAC is a small molecule containing a thiol group (Ziment, 1988) and a precursor of reduced glutathione (GSH), a well-known anti-oxidant. It has been used as a mucolytic agent as well as an antioxidant in various clinical applications for more than 30 years (Cotgreave, 1997, Radomska-Lesniewska and Skopinski, 2012). The antioxidant role of NAC in reducing Cd toxicity was extensively studied in various cell lines and animal models (Odewumi et al., 2011; Luo et al., 2013; Wang et al., 2014). However, very few studies were reported on the immuno-modulatory effect of NAC in reducing Cd toxicity in liver cells (Dong et al., 1998; Souza et al., 2004) and so far, no study was reported in lung cells. In this study, we evaluated the modulatory effect of NAC on human A549 lung cells treated with CdCl2 through cytokines and chemokines (60) expression by human cytokine array analysis.
MATERIALS AND METHODS
Reagents
F12K medium (1x), penicillin-streptomycin anti-biotic solution (100x), fetal bovine serum (FBS), trypsin-EDTA solution (1x), phosphate buffered saline (PBS), cadmium chloride, 25% glutaraldehyde, and crystal violet were purchased from Sigma-Aldrich company (St. Louis, MO, USA). The Human cytokine array 6 and IL-1α ELISA kits were purchased from RayBiotech, Inc. (Norcross, GA, USA).
Maintenance of cell line
Human lung (A549, catalog number CCL-185) cell line was purchased from the American Type Culture Collection (ATCC, Manassas, VA, USA). The supplied frozen cells were cultured according to ATCC instructions. In brief, the cells were grown in 10 ml of F12K medium containing 100 units of penicillin per ml, 100 μg of streptomycin per ml, 0.025 μg of amphotericin B per ml, and 10% FBS in T-75 cm2 tissue culture flasks at 37 °C in a 5% CO2 incubator (Nuaire Inc, Plymouth, MN, USA).
Treatment of cells
To investigate the protective effect of NAC on CdCl2 treated lung cells, 1 × 105 cells/well were seeded into 24 well tissue culture plate in 900 μl of complete medium and incubated overnight in a 5% CO2 incubator at 37 °C for stabilization. Following the stabilization, the cells were treated with CdCl2 (0 or 75 μM) and with or without NAC (2.5 or 5 mM) in a final volume of 1 ml in triplicate wells and incubated for 24 h at 37 °C in a 5% CO2 incubator. The NAC stock (500 mM) was prepared in sterile water. Cells incubated with only culture medium without CdCl2 or NAC were served as the control cells.
Morphology
At the end of the treatment, morphology of untreated (control), CdCl2 alone treated, and co-treated lung cells with NAC were observed under a phase-contrast microscope and the images were taken with a Kodak digital camera under the Nikon Diaphot phase contrast microscope with 10x objective.
Cell Viability Test
The viability was evaluated by crystal violet dye uptake assay as reported earlier (Badisa et al., 2008). In brief, glutaraldehyde (400 μl of 0.25% to make 0.07% final concentration in the well) was added to each well and incubated for 30 min at room temperature to fix the viable cells. Following this, the plates were rinsed with water to wash off the dead cells and dried under airflow inside the laminar hood for 5–10 min. Crystal violet solution (400 μl of 0.1%) was added to each well and incubated for 15 min, followed by several washes and dried under airflow inside the laminar hood for 5–10 min. The dye was solubilized in each well with addition of 1 ml of 0.05 M sodium phosphate solution (monobasic) in 50% ethyl alcohol. The culture plates were read at 540 nm in a plate reader (Bio-Tek EL800 Plate Reader). The mean O.D. value of the control cells was taken as 100% and the other treated groups were calculated as a percent of the control.
Preparation of samples for cytokine array
To study the cytokines expression of the CdCl2 treated lung cells and the immuno-modulatory effect of NAC on these cells, approximately 3.9 × 106 cells were plated in T-75 cm2 flasks in complete F12K medium and allowed to stabilize overnight. The cells were then treated with CdCl2 alone (0, 75 μM) or co-treated with 2.5 mM NAC and 75 μM CdCl2 in triplicate T-75 cm2 flasks. The flasks were incubated for 24 h at 37 °C in a 5% CO2 incubator. After 24 h of incubation, the culture medium was transferred to 15 ml falcon tube and stored at −70 °C till it was used for cytokine array. The cells were trypsinized, pooled together and pelleted by centrifuging at 2,500 RPM for 5 min. The cell pellet was suspended in 1 ml of 1x cell lysis buffer (from the Cytokine Array Kit) and lysed by homogenization in a vial under ice for 15 s (3x) using a Polytron homogenizer. The homogenate was transferred to an eppendorff tube and centrifuged at 10,000 RPM for 10 min at 4 °C to remove the lysed cell membrane debris. The supernatant was transferred to fresh tube and cell lysate was stored at −80 °C for use in the cytokine array.
Protein estimation
The protein concentration was determined by the BCA method (Pierce company) using a BSA standard. Diluted albumin (BSA) standards and working reagent were prepared according to the kit instructions. Different concentrations of each standard and each unknown sample (25 μl) was pipetted in triplicate into appropriately labeled eppendorff tubes with 500 μl of working reagent and mixed well. The tubes were incubated at 37 °C for 30 min and then read at 562 nm in a Beckman spectrophotometer. From the standard curve, the protein concentrations of cell lysate were determined.
Cytokine Array Analysis
The cytokines expression was determined in cell lysate and cell culture medium using Ray Biotech’s Human Cytokine Antibody Array 6 kit, (catalog # AAH-CYT-6). The array study was carried out according to manual instructions with minor changes. The membranes were blocked for 30 min and then hybridized with 400 μg of cell lysate protein (total volume 1 ml) or 1 ml of culture medium for 2 h at room temperature. The membranes were washed with buffer I (3x) and buffer II (2x) for 5 min. Following this, the membranes were incubated with biotin-conjugated primary antibodies for 2 h at room temperature and then washed, followed by incubation with the HRP-conjugated streptavidin secondary antibodies at room temperature for 2 h. The arrays were washed again and developed by incubation with detection buffer for 5 min. The chemiluminiscence of the arrays was then detected using Alpha Innotech’s FluorChem FC2 machine and were analyzed by AlphaEaseFC software.
ELISA
Ray Bio IL-1α ELISA kit employs a specific cytokine IL-1α antibody coated on each well in a 96-well plate. Each well was incubated with 40 μg of cell lysate protein at 4 °C for overnight. The wells were washed with wash buffer (4x) and incubated with biotinylated antibody at room temperature for 2 h. Following this, the unbound biotinylated antibodies were washed away from the plate by washing with wash buffer and the wells were incubated with HRP-conjugated streptavidin. The wells were washed again and were developed with soluble HRP substrate 3, 3′, 5, 5′-tetramethylbenzidine (TMB) in buffered solution. The reaction was stopped with stop solution composed of 2 M sulfuric acid that changes the color from blue to yellow, and the intensity of the color in the 96 well plate wells was measured at 450 nm in a plate reader (Bio-Tek, Winooski, VT, USA). Cytokine level was expressed in treated groups in comparison to control cells cytokine expression (100%).
Statistical analysis
The results were presented as mean±standard error of the mean (SEM, n=3). The data were analyzed for significance by one-way analysis of variance (ANOVA) and then compared by Tukey’s multiple comparison tests using the GraphPad Prism software, version 3.00 (San Diego, CA, USA).
RESULTS
Protective effect of NAC on morphological changes in CdCl2 treated human lung A549 cells
The morphological changes of human lung A549 cells observed after 24 h treatment with CdCl2 alone (0, 75 μM), 2.5 mM NAC alone or co-treatment with 2.5 mM NAC and 75 μM CdCl2 were shown in Fig. 1. The control cells showed long spindle and narrow protrusions (Fig. 1A). The morphology of NAC exposed cells also resembled the untreated control cells (Fig. 1B). In the cells treated with 75 μM CdCl2 alone, many cells have retracted extensions and gradually became round (Fig. 1C). In the cells co-treated with NAC and 75 μM CdCl2 (Fig. 1D), the cell extensions that were altered by CdCl2 were restored and resemble the control cells (Fig. 1A). These results clearly showed the protective effect of NAC on the morphological changes in CdCl2 treated human lung A549 cells.
Fig. 1.
Protective effect of NAC on morphological changes in CdCl2 treated human lung A549 cells. The cells were treated with 0 CdCl2 (A), 2.5 mM NAC alone (B), 75 μM CdCl2 alone (C), or cotreated with 2.5 mM NAC and 75 μM CdCl2 (D) for 24 h.
Protective effect of NAC on the viability of CdCl2 treated human lung A549 cells
The protective effect of NAC against Cd toxicity was studied by measuring viability and the results were shown in Fig. 2. The viability of the cells treated with 75 μM CdCl2 alone was decreased significantly to 44.5 ± 2.7% in comparison to control cells (100%). However, the viability of the co-treated cells with CdCl2 and NAC (2.5 and 5 mM) was increased to 84.1± 2.7% and 60.7±2.1% respectively in comparison to the cells treated with 75 μM CdCl2 alone (44.5±2.7%). The 2.5 mM NAC didn’t show toxicity to human lung cells, but 5 mM NAC showed significant toxicity (77.3±3%) to human lung cells (Fig. 2). The viability data clearly showed the protective effect of 2.5 mM NAC against Cd toxicity in human lung A549 cells.
Fig. 2.
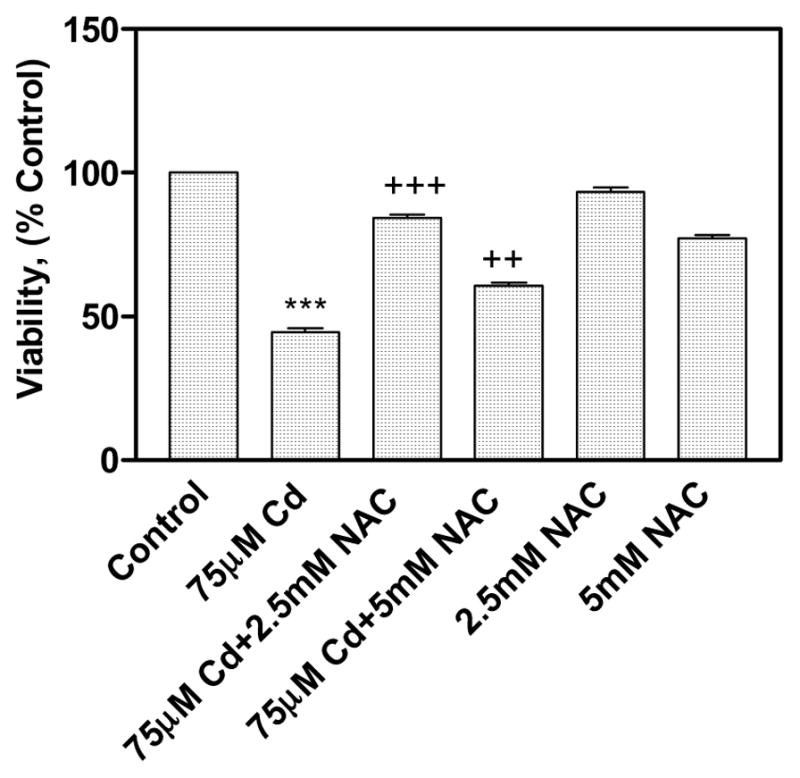
Protective effect of NAC on viability in CdCl2 treated human lung A549 cells. All values are mean ± standard deviation [S.D. (n = 6)]. Statistically (Tukey’s Multiple Comparison Test) different from the control (*** p < 0.001) and from the 75 μM CdCl2 alone (++ p < 0.01, +++ p < 0.001) were shown.
Immuno-modulatory effect of NAC on CdCl2 treated human lung A549 cells
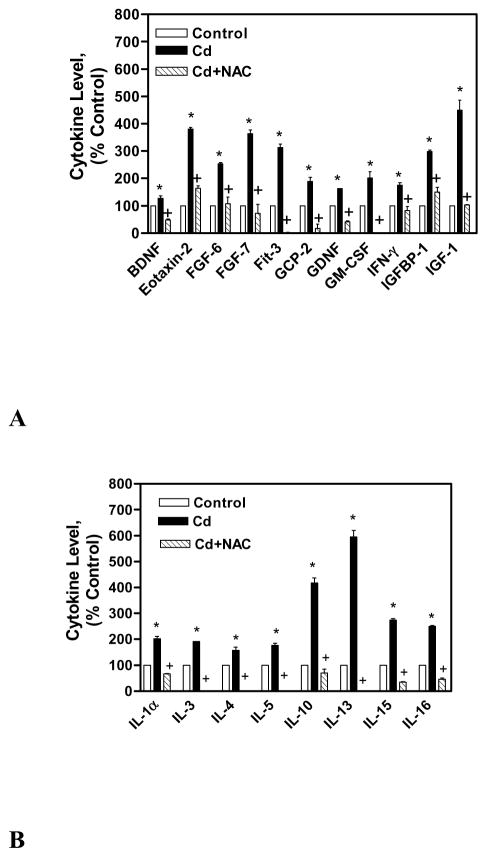
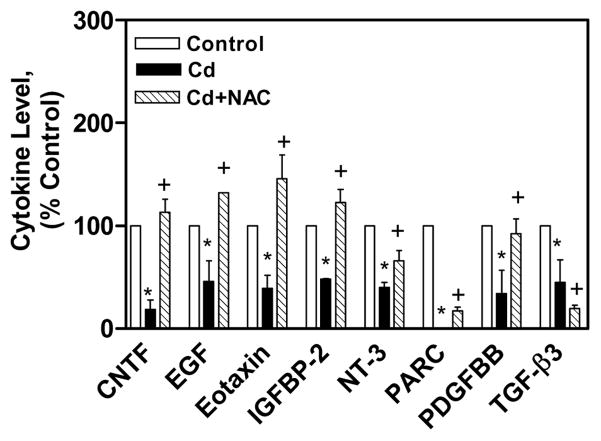
The immuno-modulatory effect of NAC on CdCl2 treated human lung A549 cells was studied by measuring various cytokines (60) expression through Human Ray Biotech cytokine array 6. The cytokine array analysis showed that 19 cytokines were up regulated (up regulation is considered as the expression level 30% above control cells expression) and 8 cytokines were down regulated (down regulation is considered as the expression level 30% below control cells expression) in the cell lysate of CdCl2 alone treated cells in comparison to untreated control cells’ lysate. The cytokines that were up regulated in the lysate of CdCl2 alone treated cells were BDNF, eotaxin-2, FGF-6, FGF-7, Fit-3 ligand, GCP-2, GDNF, GM-CSF, IFN-γ, IGFBP-1, IGF-1, IL-1α, IL-3, IL-4, IL-5, IL-10, IL-13, IL-15, and IL-16 (Figs. 3A, 3B). In the cell lysate of co-treated cells with NAC and CdCl2, all these cytokines expression was reduced in comparison to cell lysate of CdCl2 alone treated cells (Figs. 3A, 3B). The cytokines that were down regulated in the cell lysate of CdCl2 alone cells were CNTF, EGF, eotaxin, IGFBP-2, NT-3, PARC, PDGFBB, and TGF-β3 (Fig. 4). In the cell lysate of co-treated cells with NAC and CdCl2, all these cytokines expression was increased except TGF-β3 (Fig. 4).
Fig. 3.
Immuno-modulatory effect of NAC on up regulated cytokines (A, B) in CdCl2 treated human lung A549 cells’ lysate. All values are mean ± S.D. (n = 2). Statistically (Tukey’s Multiple Comparison Test) different from the control (* p < 0.05) and from the 75 μM CdCl2 alone (+ p < 0.05) were shown.
Fig. 4.
Immuno-modulatory effect of NAC on down regulated cytokines in CdCl2 treated human lung A549 cells’ lysate. All values are mean ± S.D. (n = 2). Statistically (Tukey’s Multiple Comparison Test) different from the control (* p < 0.05) and from the 75 μM CdCl2 alone (+ p < 0.05) were shown.
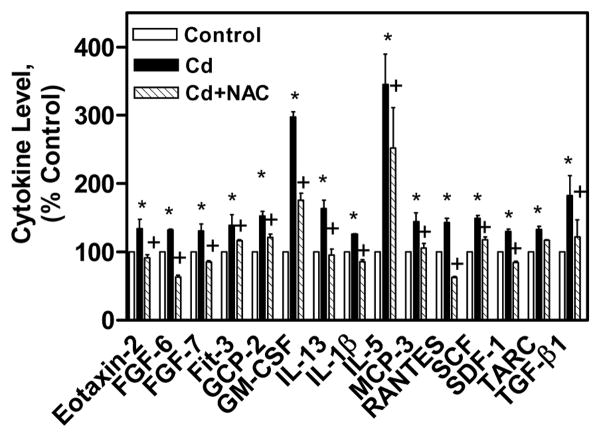
The cytokines expression was also measured in the cell culture medium of the above mentioned treatments. In cell culture medium of CdCl2 alone treated cells, 15 cytokines were up regulated and 3 cytokines were down regulated in comparison the control cells cell culture medium. The up regulated cytokines in the medium of CdCl2 alone treated cells were Eotaxin-2, FGF-6, FGF-7, Fit-3, GCP-2, GM-CSF, IL-13, IL-1β, IL-5, MCP-3, RANTES, SCF, SDF-1, TARC, and TGF-β1 (Fig 5). In the cell culture medium of co-treated cells with NAC and CdCl2, all these cytokines expression was reduced in comparison to the cytokines in the cell culture medium of CdCl2 alone treated cells (Fig 5). The down regulated cytokines in the cell culture medium of CdCl2 alone treated cells were IGFBP-4, and Angiogenin and NAP-2 (Fig 6). In the cell culture medium of co-treated cells, IGFBP-4, and Angiogenin cytokines expression were increased, while NAP-2 expression was decreased in comparison to cytokines in the cell culture medium of CdCl2 alone treated cells (Fig 6). These results clearly showed the immuno-modulatory effect of NAC on CdCl2 treated human lung A549 cells.
Fig. 5.
Immuno-modulatory effect of NAC on up regulated cytokines in CdCl2 treated human lung A549 cell culture medium. All values are mean ± S.D. (n = 2). Statistically (Tukey’s Multiple Comparison Test) different from the control (* p < 0.05) and from the 75 μM CdCl2 alone (+ p < 0.05) were shown.
Fig. 6.
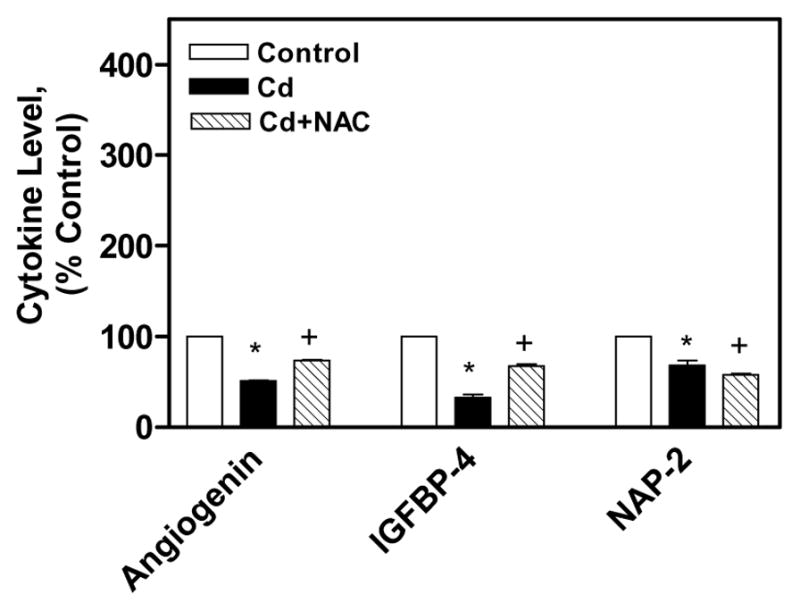
Immuno-modulatory effect of NAC on down regulated cytokines in CdCl2 treated human lung A549 cell culture medium. All values are mean ± S.D. (n = 2). Statistically (Tukey’s Multiple Comparison Test) different from the control (* p < 0.05) and from the 75 μM CdCl2 alone (+ p < 0.05) were shown.
Validation of array results by ELISA
The IL-1α ELISA was used to validate the array results. In the cell lysate of 75 μM CdCl2 alone treated cells, the cytokine IL-1α expression increased to 282.6 ± 3.1% in comparison to control cells (100%, Fig. 7). On the contrary, when the cells were co-treated with 2.5 mM NAC and 75 μM CdCl2, the expression of IL-1α was decreased to 126.1 ± 5.7% in comparison to the cell lysate of 75 μM CdCl2 alone treated cells (Fig. 7). This pattern of regulation of IL-1α expression in CdCl2 alone and co-treated cells was almost the same as the Ray Biotech array result pattern. Hence, the ELISA result clearly validated the cytokine array results.
Fig. 7.

Confirmation of array results by IL-1α ELISA. All values are mean ± S.D. (n = 3). Statistically (Tukey’s Multiple Comparison Test) different from the control (*** p < 0.001) and from the 75 μM CdCl2 alone (+++ p < 0.001) were marked.
DISCUSSION
Cd, a non-essential heavy metal that ranked seventh in the priority list of hazardous substances and environment toxicant causes severe health problems (Jarup and Åkesson, 2009; ATSDR, 2012). It is considered as multi-target toxicant, accumulating mostly in the lungs, liver and kidneys (Rikans and Yamano, 2000; Joseph, 2009; ATSDR, 2012). Even though humans are exposed to low doses of cadmium, it is accumulated in the tissues due to its extremely long half-life. Cd has a long biological half-life of 75–128 days in blood and 10–38 years in human tissues and hence accumulates in the body over period, particularly in the lungs, kidneys and liver (WHO, 2000). Cd exposure to humans occur occupationally by burning of fossil fuels as well as contaminated air from workplaces such as in battery manufacturing, metal soldering, or welding facilities (IARC, 1993; Klassen et al., 2009). Even though food (crabs, legumes, spinach etc) is the major source of Cd exposure for the general population, cigarette smoking significantly adds to the body burden of Cd in humans (Satarug and Moore, 2004; Klaassen et al., 1999). The Cd deposited in the lungs may persist for many years, as indicated in animal models (Rhoads and Sanders, 1985). Several mechanisms has been proposed for the toxic effect of cadmium in cells which include induction of free radicals, decrease in anti-oxidant potential, decrease in thiol status, activation of signaling pathways, inhibition of DNA repair and cell damage (Ikediobi et al., 2004; Badisa et al., 2008, Skrzycki et al., 2010). Previously in our laboratory, we studied the antioxidant effect of NAC on CdCl2 treated rat liver cells (Odewumi et al., 2011). In this report, we studied the protective effect of NAC on morphology, viability and immuno-modulatory effect of NAC through cytokines expression in CdCl2 treated human lung A549 cells.
It was observed that the cells treated with 75 μM CdCl2 lost their cell membrane integrity and became round (Fig. 1C). The loss in the cell membrane integrity in the Cd treated cells may be due to the lipid peroxidation induced by the Cd. This is supported by several previous reports showing that Cd causes lipid peroxidation (Ikediobi et al., 2004; Yadav and Khandelwal, 2006; Newairy et al., 2007). In the co-treated cells with NAC and 75 μM CdCl2 (Fig. 1F), the NAC restored the morphology of CdCl2 treated cells similar to the control cells (Fig. 1A). The suppression of lipid peroxidation by NAC may be responsible for the restoration of cell morphology in the co-treated lung cells with NAC and 75 μM CdCl2. It was shown earlier that NAC suppressed the lipid peroxidation in kidney cells (Wang et al., 2009).
The cytotoxicity was evaluated by simple, highly reproducible, and cost-effective crystal violet viability assay (Badisa et al., 2008). The 2.5 mM NAC showed better protection against Cd toxicity than 5 mM NAC in cell viability studies and therefore 2.5 mM NAC was used for cytokine array analysis studies (Fig. 2). The effect of NAC in preventing Cd-induced cell death may be achieved by its direct antioxidant effect or by increasing the antioxidant enzyme activities as well as its effect as a chelating agent. Various earlier studies showed that NAC treatment prevented cadmium or lead-induced cytotoxicity in various cell lines or animal models (Shaikh et al., 1999; Tandon et al., 2003; Odewumi et al., 2011). It is well known that NAC upon hydrolysis release cysteine which is a precursor for GSH synthesis. The GSH is a well known anti-oxidant which chelates the free radicals and thereby reduces the CdCl2 induced cytotoxicity.
The Ray Biotech cytokine array analysis is very valuable tool to study the expression of 60 cytokines simultaneously which saves tremendous amount of time and money. In this study, the cytokine levels were measured in the cell lysate and medium of the control, CdCl2 alone and co-treated cells with NAC and CdCl2 for 24 h. It was found that in the cell lysate and cultured medium of the cells treated with CdCl2 alone, eotaxin-2, FGF-6 & FGF-7, Fit-3 ligand, GCP-2, IL-5, IL-13 were highly up regulated (Figs. 3A, 3B, 5). Out of these cytokines, eotaxin-2, GCP-2 are chemokines, while FGF-6 & FGF-7, Fit-3 ligand, IL-5, IL-13 are involved in growth and proliferation of cells responsible for immunity. The BDNF, GDNF, GM-CSF, IFN-γ, IGFBP-1 IGF-1, IL-1α, IL-3, IL-4, IL-10, IL-15, IL-16 cytokines are highly up-regulated only in the cell lysate (not in cell cultured medium) of CdCl2 alone treated cells (Figs. 3A, 3B). The BDNF, GDNF, GM-CSF, IGFBP-1 IGF-1, IL-3, IL-4, and IL-15 are involved in growth of the cells. The IFN-γ, IL-1α-IL-10 show immunostimulatory and immunomodulatory effects, while IL-16 is a chemokine. The IL-1β, MCP-3, RANTES, SCF, SDF-1, TARC, and TGF-β1 cytokines are up-regulated only in the cultured medium of CdCl2 alone treated cells (Fig. 5). The IL-1β, SCF, TGF-β1 cytokines are involved in cell proliferation, differentiation, and apoptosis, while MCP-3, RANTES, SDF-1, TARC are chemokines. In the co-treated cells with NAC and CdCl2, all these cytokines levels are increased in comparison to CdCl2 alone treated cells which show the immuno-modulating effect of NAC on cytokines expression (Figs. 3A, 3B, 5). The immuno-modulating effect of NAC was supported by the earlier report where it was shown that NAC suppressed the stimulated chemokine production in dermal microvascular endothelial cells (Adhami et al., 2012).
It was found that CNTF, EGF, Eotaxin, IGFBP-2, NT-3, PARC, PDGFBB, TGF-β3 are down-regulated in the cell lysate of CdCl2 alone treated cells (Fig. 4). The CNTF, EGF, IGFBP-2, NT-3, PDGFBB, TGF-β3 cytokines are involved in cell growth, proliferation, and differentiation, while Eotaxin, PARC cytokines are chemokines. In the cell culture medium of CdCl2 alone treated cells, IGFBP-4, Angiogenin and NAP-2 were found down-regulated (Fig. 6). The IGFBP-4 is involved in growth of cells, while Angiogenin is a stress-activated ribonuclease and NAP-2 is a chemokine. In the cell lysate of co-treated cells with 2.5 mM NAC and CdCl2, all those down regulated cytokines in CdCl2 alone treated cells were increased except TGF-β3. In earlier reports, it was shown that NAC has been used to reduce inflammation in patients with chronic obstructive pulmonary disease and interstitial pulmonary fibrosis (Meyer et al., 1994; Day 2008). It is also effective in attenuating amiodarone-induced lung fibrosis (Leeder et al., 1994) and radiation-induced lung injury and fibrosis (Neal et al., 2003).
IL-1α is one of the highly modulated cytokines in CdCl2 alone treated cells and hence it was selected to validate the array results. It plays specific role in the onset of inflammatory process that regulates expression of other cytokines and chemokines in the cells against chemical insult (Barksby et al., 2007). The same pattern of IL-1α cytokine expression in the CdCl2 alone and co-treated cells in ELISA experiment as cytokine array validated the results obtained from Cytokine Antibody Array analysis (Fig. 7).
CONCLUSION
In conclusion, in this report, we showed that 2.5 mM NAC co-treatment restored the morphology and viability of CdCl2 treated human lung cells. Furthermore, we also showed that protection against CdCl2 toxicity was due to the immuno-modulatory effect of NAC on various cytokines expression in co-treated human lung cells with 2.5 mM NAC and 75 μM CdCl2. This study will suggest that NAC can be used to treat CdCl2 toxicity in humans after testing in the animal models and clinical trials.
Acknowledgments
We sincerely acknowledge the financial support of FAMU Title III, Dept. of Education (DOEHBGIPO31B40108-08), RCMI, National Institute of Health (G12RR03020, G12D007582) for this research work.
References
- Adhami K, Lee J, Levin L, Moquete R, Stohl LL, Ding W, Wong J, Schierl M, Zhou XK, Gordon JS, Perez E, Stock MB, Granstein RD. N-acetyl-S-farnesyl-l-cysteine suppresses chemokine production by human dermal microvascular endothelial cells. Exp Dermatol. 2012;21(9):700–705. doi: 10.1111/j.1600-0625.2012.01562.x. [DOI] [PubMed] [Google Scholar]
- Agency for Toxic Substances and Disease Registry (ATSDR) Toxicological Profile for Cadmium. Atlanta, GA: U.S. Department of Health and Human Services, Public Health Service; 2012. pp. 1–512. [Google Scholar]
- Badisa VL, Latinwo LM, Odewumi CO, Ikediobi CO, Badisa RB, Brooks-Walter A, Lambert AT, Nwoga J. Cytotoxicity and stress gene microarray analysis in cadmium-exposed CRL-1439 normal rat liver cells. Int J Mol Med. 2008;22:213–219. [PubMed] [Google Scholar]
- Barksby HE, Lea SR, Preshaw PM, Taylor JJ. The expanding family of interleukin-1 cytokines and their role in destructive inflammatory disorders. Clin Exp Immunol. 2007;149(2):217–225. doi: 10.1111/j.1365-2249.2007.03441.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cotgreave IA. N-acetylcysteine: pharmacological considerations and experimental and clinical applications. Adv Pharmacol. 1997;38:205–227. [PubMed] [Google Scholar]
- Cullinan P. Occupation and chronic obstructive pulmonary disease (COPD) Br Med Bull. 2012;104:143–161. doi: 10.1093/bmb/lds028. [DOI] [PubMed] [Google Scholar]
- Day BJ. Antioxidants as potential therapeutics for lung fibrosis. Antioxid Redox Signal. 2008;10:355–370. doi: 10.1089/ars.2007.1916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dong W, Simeonova PP, Gallucci R, Matheson J, Flood L, Wang S, Hubbs A, Luster MI. Toxic metals stimulate inflammatory cytokines in hepatocytes through oxidative stress mechanisms. Toxicol Appl Pharmacol. 1998;151(2):359–66. doi: 10.1006/taap.1998.8481. [DOI] [PubMed] [Google Scholar]
- Hartwig A. Cadmium and cancer. Met Ions Life Sci. 2013;11:491–507. doi: 10.1007/978-94-007-5179-8_15. [DOI] [PubMed] [Google Scholar]
- Ikediobi CO, Badisa VL, Ayuk-Takem LT, Latinwo LM, West J. Response of antioxidant enzymes and redox metabolites to cadmium-induced oxidative stress in CRL-1439 normal rat liver cells. Int J Mol Med. 2004;14:87–92. [PubMed] [Google Scholar]
- International Agency for Research on Cancer (IARC) Monographs on the evaluation of the carcinogenic risks to humans, Beryllium, Cadmium, Mercury and exposures in the glass manufacturing industry. IARC Scientific Publications; Lyon, France: 1993. pp. 119–238. [PMC free article] [PubMed] [Google Scholar]
- Järup L, Åkesson A. Current status of cadmium as an environmental health problem. Toxicol Appl Pharmacol. 2009;238:201–208. doi: 10.1016/j.taap.2009.04.020. [DOI] [PubMed] [Google Scholar]
- Järup L, Bellander T, Hogstedt C, Spång G. Mortality and cancer incidence in Swedish battery workers exposed to cadmium and nickel. Occup Environ Med. 1998;55(11):755–759. doi: 10.1136/oem.55.11.755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joseph P. Mechanisms of cadmium carcinogenesis. Toxicol Appl Pharmacol. 2009;238:272–279. doi: 10.1016/j.taap.2009.01.011. [DOI] [PubMed] [Google Scholar]
- Kirschvink N, Martin N, Fievez L, Smith N, Marlin D, Gustin P. Airway inflammation in cadmium-exposed rats is associated with pulmonary oxidative stress and emphysema. Free Radic Res. 2006;40:241–250. doi: 10.1080/10715760500494657. [DOI] [PubMed] [Google Scholar]
- Klaassen CD, Liu J, Choudhuri S. Metallothione an intracellular protein to protect against cadmium toxicity. Annu Pharmacol Rev Toxicol. 1999;39:267–294. doi: 10.1146/annurev.pharmtox.39.1.267. [DOI] [PubMed] [Google Scholar]
- Klaassen CD, Liu J, Diwan BA. Metallothionein protection of cadmium toxicity Toxicol. Appl Pharmacol. 2009;238:215–220. doi: 10.1016/j.taap.2009.03.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kundu S, Sengupta S, Bhattacharyya A. EGFR upregulates inflammatory and proliferative responses in human lung adenocarcinoma cell line (A549), induced by lower dose of cadmium chloride. Inhal Toxicol. 2011;23(6):339–348. doi: 10.3109/08958378.2011.572931. [DOI] [PubMed] [Google Scholar]
- Låg M, Rodionov D, Ovrevik J, Bakke O, Schwarze PE, Refsnes M. Cadmium-induced inflammatory responses in cells relevant for lung toxicity: Expression and release of cytokines in fibroblasts, epithelial cells and macrophages. Toxicol Lett. 2010;193(3):252–260. doi: 10.1016/j.toxlet.2010.01.015. [DOI] [PubMed] [Google Scholar]
- Leeder RG, Brien JF, Massey TE. Investigation of the role of oxidative stress in amiodarone-induced pulmonary toxicity in the hamster. Can J Physiol Pharmacol. 1994;72:613–621. doi: 10.1139/y94-087. [DOI] [PubMed] [Google Scholar]
- Luo YH, Wu SB, Wei YH, Chen YC, Tsai MH, Ho CC, Lin SY, Yang CS, Lin P. Cadmium-based quantum dot induced autophagy formation for cell survival via oxidative stress. Chem Res Toxicol. 2013;26(5):662–673. doi: 10.1021/tx300455k. [DOI] [PubMed] [Google Scholar]
- Meyer A, Buhl R, Magnussen H. The effect of oral N-acetylcysteine on lung glutathione levels in idiopathic pulmonary fibrosis. Eur Respir J. 1994;7:431–436. doi: 10.1183/09031936.94.07030431. [DOI] [PubMed] [Google Scholar]
- Nawrot T, Plusquin M, Hogervorst J, Roels HA, Celis H, Thijs L, Vangronsveld J, Van Hecke E, Staessen JA. Environmental exposure to cadmium and risk of cancer: a prospective population-based study. Lancet Oncol. 2006;7(2):119–126. doi: 10.1016/S1470-2045(06)70545-9. [DOI] [PubMed] [Google Scholar]
- Neal R, Matthews RH, Lutz P, Ercal N. Antioxidant role of N-acetyl cysteine isomers following high dose irradiation. Free Radic Biol Med. 2003;34:689–695. doi: 10.1016/s0891-5849(02)01372-2. [DOI] [PubMed] [Google Scholar]
- Newairy AA, El-Sharaky AS, Badreldeen MM, Eweda SM, Sheweita SA. The hepatoprotective effects of selenium against cadmium toxicity in rats. Toxicol. 2007;242(1–3):23–30. doi: 10.1016/j.tox.2007.09.001. [DOI] [PubMed] [Google Scholar]
- Odewumi CO, Badisa VL, Le UT, Latinwo LM, Ikediobi CO, Badisa RB, Darling-Reed SF. Protective effects of N-acetylcysteine against cadmium-induced damage in cultured rat normal liver cells. Int J Mol Med. 2011;27(2):243–248. doi: 10.3892/ijmm.2010.564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oh CM, Oh IH, Lee JK, Park YH, Choe BK, Yoon TY, Choi JM. Blood cadmium levels are associated with a decline in lung function in males. Environ Res. 2014;132:119–125. doi: 10.1016/j.envres.2014.04.008. [DOI] [PubMed] [Google Scholar]
- Park RM, Stayner LT, Petersen MR, Finley-Couch M, Hornung R, Rice C. Cadmium and lung cancer mortality accounting for simultaneous arsenic exposure. Occup Environ Med. 2012;69 (5):303–309. doi: 10.1136/oemed-2011-100149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Radomska-Lesniewska DM, Skopinski P. N-acetylcysteine as an antioxidant and anti-inflammatory drug and its some clinical applications. Cent Eur J Immun. 2012;37:57–66. [Google Scholar]
- Rennolds J, Malireddy S, Hassan F, Tridandapani S, Parinandi N, Boyaka PN, Cormet-Boyaka E. Curcumin regulates airway epithelial cell cytokine responses to the pollutant cadmium. Biochem Biophys Res Commun. 2012;417(1):256–261. doi: 10.1016/j.bbrc.2011.11.096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rhoads K, Sanders CL. Lung clearance, translocation, and acute toxicity of arsenic, beryllium, cadmium, cobalt, lead, selenium, vanadium, and ytterbium oxides following deposition in rat lung. Environ Res. 1985;36:359–378. doi: 10.1016/0013-9351(85)90031-3. [DOI] [PubMed] [Google Scholar]
- Rikans LE, Yamano T. Mechanisms of cadmium-mediated acute hepatotoxicity. J Biochem Mol Toxicol. 2000;14:110–117. doi: 10.1002/(sici)1099-0461(2000)14:2<110::aid-jbt7>3.0.co;2-j. [DOI] [PubMed] [Google Scholar]
- Satarug S, Moore MR. Adverse health effects of chronic exposure to low-level cadmium in foodstuffs and cigarette smoke. Environ Health Perspect. 2004;112:1099. doi: 10.1289/ehp.6751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shaikh ZA, Zaman K, Tang W, Vu T. Treatment of chronic cadmium nephrotoxicity by N-acetyl cysteine. Toxicol Lett. 1999;104(1–2):137–142. doi: 10.1016/s0378-4274(98)00358-0. [DOI] [PubMed] [Google Scholar]
- Skrzycki M, Czeczot H, Majewska M, Podsiad M, Karlik W, Grono D, Wiechetek M. Enzymatic antioxidant defense in isolated rat hepatocytes exposed to cadmium. Pol J Vet Sci. 2010;13(4):673–679. doi: 10.2478/v10181-010-0002-7. [DOI] [PubMed] [Google Scholar]
- Souza V, del Escobar Md MC, Gómez-Quiroz L, Bucio L, Hernández E, Cossio EC, Gutiérrez-Ruiz MC. Acute cadmium exposure enhances AP-1 DNA binding and induces cytokines expression and heat shock protein 70 in HepG2 cells. Toxicol. 2004;197(3):213–228. doi: 10.1016/j.tox.2004.01.006. [DOI] [PubMed] [Google Scholar]
- Tandon SK, Singh S, Prasad S, Khandekar K, Dwivedi VK, Chatterjee M, Mathur N. Reversal of cadmium induced oxidative stress by chelating agent, antioxidant or their combination in rat. Toxicol Lett. 2003;145(3):211–217. doi: 10.1016/s0378-4274(03)00265-0. [DOI] [PubMed] [Google Scholar]
- Wang J, Zhu H, Liu X, Liu Z. N-acetylcysteine protects against cadmium-induced oxidative stress in rat hepatocytes. J Vet Sci. 2014;15(4):485–493. doi: 10.4142/jvs.2014.15.4.485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang L, Chen D, Cao J, Liu Z. Protective effect of N-acetylcysteine on experimental chronic cadmium nephrotoxicity in immature female rats. Hum Exp Toxicol. 2009;28(4):221–229. doi: 10.1177/0960327109102365. [DOI] [PubMed] [Google Scholar]
- World Health Organization Cadmium (WHO) Air Quality Guidelines for Europe. 2. World Health Organization Regional Office for Europe; Copenhagen: 2000. http://www.euro.who.int/__data/assets/pdf_file/0005/74732/E71922.pdf. [Google Scholar]
- Yadav N, Khandelwal S. Effect of Picroliv on cadmium-induced hepatic and renal damage in the rat. Hum Exp Toxicol. 2006;25(10):581–591. doi: 10.1177/096032706072455. [DOI] [PubMed] [Google Scholar]
- Ziment I. Acetylcysteine: a drug with an interesting past and a fascinating future. Respiration. 1986;50(Suppl 1):26–30. doi: 10.1159/000195085. [DOI] [PubMed] [Google Scholar]