Abstract
Plant production and plant product quality strongly depend on the availability of mineral nutrients. Among them, sulfur (S) and iron (Fe) play a central role, as they are needed for many proteins of the respiratory chain. Plant mitochondria play essential bioenergetic and biosynthetic functions as well as they have an important role in signaling processes into the cell. Here, by comparing several transcriptomic data sets from plants impaired in their respiratory function with the genes regulated under Fe or S deficiencies obtained from other data sets, nutrient-responsive genes potentially regulated by hypothetical mitochondrial retrograde signaling pathway are evidenced. It leads us to hypothesize that plant mitochondria could be, therefore, required for regulating the expression of key genes involved both in Fe and S metabolisms.
Keywords: iron, mitochondria dysfunctions, nutrient-responsive genes, respiratory chain, sulfur
The interactions between Fe and S homeostasis in plants
Plant production and plant product quality strongly depend on the availability of mineral nutrients (Briat et al., 2015a,b). Among them, sulfur (S) and iron (Fe) play a central role, as they are needed for many proteins of essential metabolic processes. Indeed, Fe and S interact for the building of Fe–S clusters, which are essential prosthetic groups for photosynthesis, respiration, and many enzymatic reactions (Couturier et al., 2013). Fe and S interactions were documented both at physiological and molecular levels, although the mechanisms integrating the homeostasis of these two elements remain unknown. Leaf Fe concentration decreases in S-deficient tomato, which is consistent with the decrease activity of the root Fe uptake system in response to S starvation in tomato (Zuchi et al., 2009) and in Arabidopsis (Forieri et al., 2013). Reciprocally, Fe starvation modifies S homeostasis. In Fe-deficient Arabidopsis plants, S-metabolism-related genes (among which plasma membrane and tonoplast S transporters, and enzymes of the S assimilation pathway) are co-expressed with Fe-deficient genes (Schuler et al., 2011). For example, the SULTR1;1 S transporter gene is down-regulated in the absence of Fe (Forieri et al., 2013). It was not confirmed by Paolacci et al. (2014), showing that most of the group 2 and 4, and some of the group 1, tomato S transporter genes are up-regulated under Fe deficiency. In Graminaceous plants, S starvation decreases mugineic acid (Fe[III]-chelators) synthesis (Kobayashi and Nishizawa, 2012) and release (Kuwajima and Kawai, 1997; Astolfi et al., 2006). In contrast, YS1 (encoding a Fe[III]-mugineic acid transporter; Curie et al., 2009) gene expression increases in response to Fe deficiency (Astolfi et al., 2012). Fe deficiency-induced modifications of S metabolism were also investigated in durum wheat (Ciaffi et al., 2013). Genes encoding enzymes of the S assimilation pathway (APS reductase, ATP sulfurylase, sulfite reductase, and serine acetyltransferase), and activity of these enzymes, are up-regulated under Fe-deficient/S-sufficient conditions, as observed under S starvation conditions. However, not all the genes necessary for S assimilation are regulated in response to Fe scarcity. Some of them have their response to Fe or S deprivation uncoupled. For example, the SULTR1.1 transporter gene expression is strongly induced in response to S deficiency but unaffected by Fe starvation (Ciaffi et al., 2013). Recently, tomato plants grown under both S and Fe deficiencies were shown to display an even more increased expression of sulfate transporters in shoot and root than those of plant grown under a single nutrient deprivation (Zuchi et al., 2015). Such synergistic effect of S and Fe deficiency interaction was observed also at the metabolic level where the content of some metabolites (i.e., asparagine, fumaric acid, and malic acid) changed in the roots of plants grown under both S and Fe deficiencies comparatively to single nutrient deprivation (Zuchi et al., 2015). The interactions between Fe and S metabolisms have been started to be described at a molecular level but no information is yet available about the mechanisms regulating these cross-talks. Modulation of the Fe–S cluster biogenesis and their relative abundance in response to various nutritional stresses has, however, been suggested to potentially regulate Fe–S interactions (Couturier et al., 2013; Forieri et al., 2013).
Mitochondrion is one of the cellular compartments playing a central role in the Fe–S interaction, since it is a site where Fe–S cluster assembly takes place.
For detailed information about Fe–S clusters, the readers can refer to two recent reviews given by Couturier et al. (2013) and Balk and Schaedler (2014). Fe–S clusters are prosthetic groups composed of Fe atoms and acid-labile inorganic sulfide. Generally, Fe atom's coordination with the protein backbone occurs via thiol groups of cysteinyl residues. The most common clusters found in plant proteins are [Fe2S2] and [Fe4S4] clusters. Their cellular biogenesis is not spontaneous. In both eukaryotes and prokaryotes, Fe–S clusters are inserted co- or post-translationally into apo-proteins through specific assembly machineries. It allows a correct folding or stability of the protein. Schematically, the assembly process can be divided into two steps. First, Fe–S clusters are built on scaffold proteins interacting with iron- and sulfur-delivery proteins. Second, carrier proteins transfer the preformed Fe–S clusters to target apo-proteins. The nature of the Fe donor is still a matter of debate. Sulfur comes from cysteine through the activity of cysteine desulfurases that are associated with specific proteins to be fully active. A persulfide (S0) is produced onto an active site cysteine. Since sulfur is always present in the S2− oxidation state in Fe–S clusters, it explains why electrons are needed to reduce sulfane sulfur during the course of cluster assembly. A few additional proteins including ATP-hydrolyzing proteins or sulfur acceptors are also needed for this reaction. Fe–S cluster assembly machineries in plants are compartmented in three systems: the SUF (sulfur mobilization), ISC (iron–sulfur cluster), and CIA (cytosolic iron–sulfur cluster assembly) machineries for plastidial, mitochondrial, and cytosolic/nuclear Fe–S proteins, respectively. In addition, export machinery between mitochondria and the cytosol links the ISC and CIA machineries. The export and CIA machineries are specific to eukaryotes, whereas the SUF and/or ISC machineries are observed in most living organisms. Fe–S clusters perform a wide diversity of functions. It ranges from electron transfer to (de)hydration reactions, radical-generation, or disulfide cleavage. From biological point of view, the functionality of Fe–S proteins is required for sulfur and nitrogen assimilation, chlorophyll catabolism, DNA repair and replication, ribosome biogenesis, tRNA thio-modification, or co-enzyme (biotin, lipoic acid, and thiamine) synthesis.
The impact of Fe and S deficiencies on mitochondrial respiration
Mitochondria contain numerous Fe–S cluster-containing proteins participating in the respiratory chain. Indeed, one single respiratory chain unit requires at least 10 different Fe–S clusters corresponding to ~30 Fe atoms and 30 S atoms (Couturier et al., 2013; Balk and Schaedler, 2014).
Among the mitochondrial metabolic processes, respiration is of central importance. Indeed, the respiratory rate is a key determinant of the growth decrease under a range of abiotic stresses (Atkin et al., 2005; Atkin and Macherel, 2009; Van Aken et al., 2009; Jacoby et al., 2011). Therefore, understanding the control and regulation of the respiratory processes is vital to improve the rate of plant growth and biomass production (Jacoby et al., 2012).
Recently, Schwarzländer et al. (2012) demonstrated that the respiratory chain dysfunctions affect the expression of a great number of genes, suggesting the importance of mitochondria in the regulation of gene expression by retrograde signaling pathways. Although the nature of the retrograde signal(s) was not yet identified, the mitochondrial transport of electrons has been considered as a potential upstream stimulus for the regulation of nuclear gene expression (Schwarzländer et al., 2012).
Considering the essential role of Fe and S for mitochondrial respiration, a deficiency of these nutrients affects the function of mitochondria.
Iron-deficient cucumber plant showed decreased root Fe content by about 80% with respect to the control plant. In these plants, mitochondrial function was strongly affected and the specific activity of each respiratory chain complexes decreased as follows: Complex I – 95%, complex II – 77%, complex III – 56%, complex IV – 50%, and complex V – 52%. At the same time, the external type II NAD(P)H dehydrogenase (NDex) increased by about 100% in Fe-deficient plants (Vigani et al., 2009; Vigani and Zocchi, 2010). Similar changes were observed also in Hyoscyamus albus (Higa et al., 2010).
In S-deficient Arabidopsis plants, S content decreased by about 48% in leaf and 40% in root tissues. In these plants, the total mitochondrial respiration of complex I and complex IV capacities decreased, whereas the activity of NDx increased. These changes were accompanied by a lower ATP level and a more reduced state of leaf and root cells. Particularly, in S-deficient Arabidopsis plants, complex I capacity decreased by 40 and 25% in leaf and root tissues, respectively, whereas Complex IV capacity decreased by about 37 and 30% in leaf and root, respectively. At the same time, NDex increased by 45 and 20% in leaf and root, respectively (Ostaszewska et al., 2014).
Interestingly, such functional changes of the respiratory pathway observed under Fe or S deficiencies were associated to changes occurring at the ultrastructural level (Ostaszewska et al., 2014; Vigani et al., 2015a). Indeed, both nutrient deficiencies lead to a lower mitochondrial matrix density as well as to a lower cristae number compared to the control mitochondria. Particularly, such morphological changes were quantified in Fe-deficient plants: the number of cristae per mitochondria decreased by 56% and the relative intracristae space decreased by 46% in Fe-deficient mitochondria compared to the control ones. Such impaired ultrastructure might reflect the metabolic status of mitochondria with a decreased respiration rate (Vigani et al., 2015a).
An important question aroused when taking into consideration that (i) respiratory chain dysfunctions might be a source of retrograde signaling pathways and that (ii) Fe and S deficiencies affect respiratory chain: Are mitochondria involved in the regulation of Fe- and S-responsive genes?
In yeast, mitochondria play an important role in the regulation of Fe-responsive genes (Ueta et al., 2012). Indeed, Fe homeostasis in Saccharomyces cerevisae is controlled primarily by the transcriptional activator Aft1p (Rutherford and Bird, 2004). The activation/inactivation of Aft1p relies on its interaction with extra-mitochondrial monothiol glutaredoxins Grx3p and Grx4p. The Aft1p–Grxps complex senses the status of the mitochondrial Fe–S cluster biogenesis, and in turn regulates S. cerevisae genes required for Fe uptake and storage. Indeed, during Fe starvation, Fe–S cluster assembly in mitochondria as well as dimeric Grx3/4p with bound Fe–S clusters is limited, thereby decreasing the interaction of Grx3/4p with Aft1p. Therefore, Aft1p is able to bind its target promoters in order to increase the expression level of the iron regulon. In contrast, in the presence of Fe, Grx3/4p binds Fe–S clusters that require functional mitochondria in order to be correctly assembled. As a result, the Grx3/4p–Fe–S complex interacts with Aft1p, leading to its disassociation from its target promoters and to a down-regulation of the iron regulon (Ueta et al., 2012). These findings suggest that, at least in yeast, mitochondria play a central role in the regulation of cellular Fe homeostasis. However, these mechanisms do not seem to occur in plants (Bernard et al., 2009; Knuesting et al., 2015).
Although very few data are available concerning mitochondria retrograde signaling pathway in plants (Schwarzländer and Finkemeier, 2013), an involvement of mitochondria in such a pathway has been suggested to regulate Fe and S homeostasis (Wirtz et al., 2012; Vigani et al., 2013).
Indeed, under Fe-deficiency conditions, activities of Fe-containing enzymes are down-regulated, and the corresponding metabolite pools could be consequently modified. Therefore, organelle retrograde signals could be produced from post-transcriptional and/or post-translational-mediated metabolic changes, and transduced for subsequent regulation of nuclear genes important for Fe uptake and homeostasis (Vigani et al., 2013). Recently, it has been observed that knocking down Mitochondrial Iron Transporter (MIT) reprograms the transcriptome and the metabolome of rice plants, suggesting that a local induction of Fe deficiency in mitochondrial compartment affects the expression of several nuclear genes (Vigani et al., 2015b). Considering S, the crucial step of its assimilation in plant cells is the synthesis of cysteine. Such reaction occurs in cytosol, plastid, and mitochondria, since O-acetylserine-thiol lyase (OASTL) is localized in all these three compartments (Takahaschi et al., 2011). Interestingly, the mitochondrial isoform of OASTL is likely involved in the sensing of the cysteine status, and in turn in the sensing of S status (Wirtz et al., 2012). Therefore, a mitochondrial retrograde feedback signal(s) has also been proposed for the S assimilatory pathway (Forieri et al., 2013).
Genes differentially expressed in both Fe- and S-deficient plants and in mitochondrial-impaired plants
Considering the strong impact that Fe or S deficiencies have on mitochondrial functions, several biochemical processes related to this organelle might be a source to produce retrograde signals. In this context, the meta-analysis of transcriptomic data sets from Arabidopsis plants displaying mitochondrial dysfunction revealed interesting observations (Schwarzländer et al., 2012). This study considered 11 transcriptomic data sets from plants having either their mitochondrial functions genetically impaired or the mitochondrial respiratory chain inhibited with drugs at different points. The data sets from plants genetically impaired in their mitochondrial function corresponded to the following genetic backgrounds: aox1a (Giraud et al., 2008); ndufs4 and ndufa1 (Meyer et al., 2009); msh1xrecA (Shedge et al., 2010); AP3:u-ATP9 and AP9:u-ATP9 (Busi et al., 2011); msd1-RNAi and prxII F (Schwarzländer et al., 2012). On the other hand, some data sets were obtained from plants having their mitochondrial respiratory chain inhibited by the following chemical treatments: olygomicyn A and rotenone (Clifton et al., 2005), and Antimycin A (Schwarzländer et al., 2012).
Among the numerous genes affected in their expression in response to the various mitochondrial dysfunctions genetically or pharmacologically induced as mentioned above, many genes known to be regulated by Fe or S deficiencies can be observed.
Several genes exhibiting similar expression (up- or down-regulation) under Fe-deficiency (see list reported in Stein and Waters, 2012) conditions or under various mitochondrial dysfunction conditions were revealed. They are the transcription factor POPEYE (PYE, at3g47640); ZINC-INDUCED FACILITATOR (ZIF1, at5g13740); glutamate ammonia ligase (GLN1;4 at5g16570); a kelch repeat-containing protein (at3g07720); COPPER CHAPERONE (CCH, at3g56240); FERULIC ACID 5-HYDROXYLASE 1(FAH1, at4g36220); NICOTIANAMINE SYNTHASE 1 (NAS1, at5g04950); FERRITIN3 (FER3, at3g56090); BHLH039 (at3g56980); nodulin (at1g21140), and some genes encoding for unknown functions (at5g05250 and at3g56360) (Table 1). Furthermore, general mitochondrial dysfunctions affected other important Fe-responsive genes. They are BRUTUS (BTS at3g18290); FRO3 (at1g23020); OBP3-responsive gene (ORG1, at5g53450); OLIGOPEPTIDE TRANSPORTER (OPT3, at4g16370); S-ADENOSYLMETHIONINE SYNTHETASE (SAM1, at1g02500); METAL TOLERANCE PROTEIN A2 (MTPA2, at3g58810) (Table 1).
Table 1.
Genes differentially expressed under Fe deficiency (Stein and Waters, 2012) and under different mitochondrial impairments.
| Accession | Description | Mitochondrial dysfunctions | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| complex I | complex III | AOX | ATP synthase | MnSOD | MGR | |||||||
| −Fe | ROT | ndufs4 | ndufs1 | OLM | prxIIF + AA | aox1a | AP9:u-ATP9 | AP3:u-ATP9 | msd1-RNAi | msh1-recA | ||
| at3g18290 | EMB2454|EMB2454 protein binding/zinc ion binding BTS (BRUTUS) | |||||||||||
| at5g13740 | ZIF1|ZIF1 (ZINC INDUCED FACILITATOR 1) | |||||||||||
| at1g18910 | Protein binding/Zinc ion binding | |||||||||||
| at1g23020 | FRO3, ferric-chelate reductase | |||||||||||
| at5g53450 | ORG1 (OBP3-responsive gene 1); | |||||||||||
| at3g47640 | Basic helix-loop-helix (bHLH) family protein PYE (POPEYE) | |||||||||||
| at4g16370 | OPT3 (OLIGOPEPTIDE TRANSPORTER) | |||||||||||
| at5g16570 | GLN1;4; glutamate-ammonia ligase | |||||||||||
| at1g02500 | SAM1 (S-ADENOSYLMETHIONINE SYNTHETASE) | |||||||||||
| at5g49760 | Leucine-rich repeat family protein/protein kinase family protein | |||||||||||
| at3g07720 | Kelch repeat-containing protein | |||||||||||
| at5g01600 | FER1 ferric iron binding/iron ion binding | |||||||||||
| at3g46900 | COPT2; copper ion transmembrane transporter | |||||||||||
| at4g30490 | AFG1-like ATPase family protein | |||||||||||
| at5g59520 | ZIP2; transferase, transferring glycosyl groups/zinc ion transmembrane | |||||||||||
| at5g67370 | Unknown protein | |||||||||||
| at3g56240 | CCH (COPPER CHAPERONE); copper chaperone | |||||||||||
| at4g36220 | FAH1 (FERULIC ACID 5-HYDROXYLASE 1) | |||||||||||
| at4g38950 | Kinesin motor family protein | |||||||||||
| at5g26820 | ATIREG3 (IRON-REGULATED PROTEIN 3) | |||||||||||
| at3g56360 | Unknown protein | |||||||||||
| at1g80830 | NRAMP1 | |||||||||||
| at5g04950 | NAS1 (NICOTIANAMINE SYNTHASE 1) | |||||||||||
| at3g58810 | |MTPA2 (METAL TOLERANCE PROTEIN A2) | |||||||||||
| at3g09220 | LAC7 (laccase 7) | |||||||||||
| at2g29995 | Unknown protein | |||||||||||
| at1g01570 | Fringe-related protein | |||||||||||
| at5g36890 | BGLU42 (BETA GLUCOSIDASE 42) | |||||||||||
| at3g56090 | ATFER3 (ferritin 3) | |||||||||||
| at1g49000 | Unknown protein | |||||||||||
| at3g56980 | BHLH039, ORG3|BHLH039; DNA binding/transcription factor | |||||||||||
| at3g21240 | 4CL2 (4-COUMARATE:COA LIGASE 2) | |||||||||||
| at5g05250 | Unknown protein | |||||||||||
| at5g47910 | RBOHD (RESPIRATORY BURST OXIDASE HOMOLOG D) | |||||||||||
| at1g21140 | Nodulin, putative | |||||||||||
The data sets from plants genetically impaired in their mitochondrial function corresponded to the following genetic backgrounds: aox1a, (loss of alternative oxidase); ndufs4 and ndufa1 (loss of complex I); msh1xrecA, (mitochondrial genome rearrangement, MGR); AP3:u-ATP9 and AP9:u-ATP9 (loss of mitochondrial ATP synthase); msd1-RNAi (loss of mitochondrial manganese superoxide dismutase, MSD1, protein) and prxII F (inhibition of complex III) (Curley et al., 2009). The data sets from plants having their mitochondrial respiratory chain inhibited by the following chemical treatments: olygomicyn A (OLM) and rotenone (ROT) (Bales and Perkeybile, 2012), and Antimycin A (AA) (Curley et al., 2009). Up-regulated genes are in green and down-regulated genes are in red.
Among genes identified as similarly regulated under Fe deficiency and mitochondrial dysfunctions, PYE and BTS are of a particular interest. They characterize a transcriptional regulatory network to control Fe homeostasis in Arabidopsis (Long et al., 2010). PYE is a transcription factor necessary for the distribution of already imported Fe, whereas BTS, a functional RING E3 ubiquitin ligase, could be a post-translational regulator of the transcriptional regulatory network involved in the Fe-deficiency response (Selote et al., 2015). Among the potential targets of PYE identified in Long et al. (2010), 42 genes exhibited an expression level affected in the data sets from mitochondrial-impaired plants (Table 2). These observations underline the link between mitochondrial dysfunctions and the PYE/BTS regulatory system controlling Fe homeostasis. Interestingly, the expression of PYE and BTS is up-regulated when complex I is impaired. As Fe deficiency strongly affects complex I activity and protein synthesis (Vigani et al., 2009), mitochondria might be involved in the induction of such genes that are crucial for Fe homeostasis in plants.
Table 2.
PYE target genes (Long et al., 2010) affected by mitochondrial impairments (Schwarzländer et al., 2012).
| Accession | Description | Mitochondrial dysfunctions | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| complex I | complex III | AOX | ATP synthase | MnSOD | MGR | ||||||
| ROT | ndufs4 | ndufs1 | OLM | prxIIF + AA | aox1a | AP9:u-ATP9 | AP3:u-ATP9 | msd1-RNAi | msh1-recA | ||
| at5g13740 | ZIF1 (ZINC INDUCED FACILITATOR 1111); antiporter 668 FORWARD | ||||||||||
| at1g24400 | LHT2 (LYSINE HISTIDINE TRANSPORTER 2) | ||||||||||
| at1g23020 | FRO3; ferric-chelate reductase | ||||||||||
| at1g72440 | EDA25 (embryo sac development arrest 25) 7273596 REVERSE | ||||||||||
| at3g21640 | FKBP42, calmodulin binding | ||||||||||
| at5g14960 | DEL2 (DP-E2F-LIKE 2); DNA binding/transcription factor | ||||||||||
| at5g03210 | Unknown protein | ||||||||||
| at5g13730 | SIG4 (SIGMA FACTOR 4); DNA binding | ||||||||||
| at4g23010 | UTR2|UDP-galactose transporter-related REVERSE | ||||||||||
| at1g74790 | Catalytic | ||||||||||
| at3g13700 | RNA-binding protein, putative | ||||||||||
| at2g33710 | AP2 domain-containing transcription factor family | ||||||||||
| at5g24470 | PRR5|APRR5 transcription regulator/two-component response regulator | ||||||||||
| at3g02140 | TMAC2 (TWO OR MORE ABRESGENE 2) | ||||||||||
| at4g36920 | AP2 (APETALA 2); transcription factor | ||||||||||
| at5g04590 | SIR; sulfite reductase (ferredoxin)/sulfite reductase | ||||||||||
| at5g45410 | Unknown protein | ||||||||||
| at3g47650 | Bundle-sheath defective protein 2 family/bsd2 family | ||||||||||
| at5g14950 | GMII (GOLGI ALPHA-MANNOSIDASE II); | ||||||||||
| at1g72460 | Leucine-rich repeat transmembrane protein kinase | ||||||||||
| at4g00585 | Unknown protein | ||||||||||
| at1g56220 | Dormancy/Auxin associated family protein | ||||||||||
| at1g03090 | Symbols: MCCA|MCCA; methylcrotonoyl-CoA carboxylase | ||||||||||
| at2g46930 | Pectinacetylesterase, putative | ||||||||||
| at2g30090 | GCN5-related N-acetyltransferase (GNAT) family protein | ||||||||||
| at1g68580 | Agent domain-containing protein/Bromo-adjacent homology (BAH) | ||||||||||
| at1g23030 | Armadillo/Beta-catenin repeat family protein containing protein | ||||||||||
| at2g46920 | POL (poltergeist); protein serine/threonine phosphatase | ||||||||||
| at3g47420 | Glycerol-3-phosphate transporter, putative | ||||||||||
| at3g55430 | Glycosyl hydrolase family 17 protein | ||||||||||
| at3g15200 | Pentatricopeptide (PPR) repeat-containing protein | ||||||||||
| at5g03140 | Lectin protein kinase family protein | ||||||||||
| at3g47430 | PEX11B | ||||||||||
| at4g01730 | Zinc ion binding | ||||||||||
| at1g11840 | ATGLX1 (GLYOXALASE I HOMOLOG); | ||||||||||
| at1g56430 | NAS4 (NICOTIANAMINE SYNTHASE 4); | ||||||||||
| at3g57070 | Glutaredoxin family protein | ||||||||||
| at3g15210 | ERF4 | ||||||||||
| at5g45430 | Protein kinase, putative | ||||||||||
| at2g24550 | Unknown protein | ||||||||||
| at5g03230 | Unknown protein | ||||||||||
| at5g45310 | Unknown protein | ||||||||||
The data sets from plants genetically impaired in their mitochondrial function corresponded to the following genetic backgrounds: aox1a, (loss of alternative oxidase); ndufs4 and ndufa1 (loss of complex I); msh1xrecA, (mitochondrial genome rearrangement, MGR); AP3:u-ATP9 and AP9:u-ATP9 (loss of mitochondrial ATP synthase); msd1-RNAi (loss of mitochondrial manganese superoxide dismutase, MSD1, protein) and prxII F (inhibition of complex III) (Curley et al., 2009). The data sets from plants having their mitochondrial respiratory chain inhibited by the following chemical treatments: olygomicyn A (OLM) and rotenone (ROT) (Bales and Perkeybile, 2012), and Antimycin A (AA) (Curley et al., 2009). Up-regulated genes are in green and down-regulated genes are in red.
Similar to what was observed for Fe, several S-responsive genes (Maruyama-Nakashita et al., 2006) were also strongly up-regulated under mitochondrial dysfunctions. These genes are encoding for S transporters SULTR1;2 (at1g78000), SULTR3;4 (at3g15990), SULTR4;2 (at3g12520), and other crucial S-responsive genes such as LSU1 (at3g49580), BGLU28 (at2g44460), and SDI1 (at5g48850) (Table 3).
Table 3.
Genes differentially expressed under S deficiency (Maruyama-Nakashita et al., 2006) and under different mitochondrial impairments.
| Accession | Description | Mitochondrial dysfunctions | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| complex I | complex III | AOX | ATP synthase | MnSOD | MGR | |||||||
| −S | ROT | ndufs4 | ndufs1 | OLM | prxIIF + AA | aox1a | AP9:u-ATP9 | AP3:u-ATP9 | msd1-RNAi | msh1-recA | ||
| at5g23050 | AAE17 (ACYL-ACTIVATING ENZYME 17 | |||||||||||
| at1g12200 | Flavin-containing monooxygenase family protein| | |||||||||||
| at3g05400 | Sugar transporter, putative | |||||||||||
| at1g08920 | Sugar transporter, putative | |||||||||||
| at2g22330 | CYP79B3; electron carrier/heme binding/iron ion binding | |||||||||||
| at1g78000 | SULTR1;2 (SULFATE TRANSPORTER 1;2 | |||||||||||
| at3g15990 | SULTR3;4 (SULFATE TRANSPORTER 3;4) | |||||||||||
| at1g36370 | SHM7 (serine hydroxymethyltransferase 7) | |||||||||||
| at2g44460 | BGLU28 (BETA GLUCOSIDASE 28) | |||||||||||
| at5g40670 | PQ-loop repeat family protein/transmembrane family protein | |||||||||||
| at3g49580 | LSU1 (RESPONSE TO LOW SULFUR 1) | |||||||||||
| at5g26220 | ChaC-like family protein | |||||||||||
| at5g48180 | NSP5 (NITRILE SPECIFIER PROTEIN 5) | |||||||||||
| at3g47960 | Proton-dependent oligopeptide transport (POT) family protein | |||||||||||
| at1g64170 | ATCHX16 (CATION/H+ EXCHANGER 16 | |||||||||||
| at5g48850 | ATSDI1 (SULFUR DEFICIENCY-INDUCED 1) | |||||||||||
| at3g12520 | SULTR4;2; sulfate transmembrane transporter | |||||||||||
| at1g04770 | Male sterility MS5 family protein | |||||||||||
| at1g75280 | Isoflavone reductase, putative | |||||||||||
| at4g13430 | IIL1 (ISOPROPYL MALATE ISOMERASE LARGE SUBUNIT 1) | |||||||||||
| at3g56040 | UGP3 (UDP-GLUCOSE PYROPHOSPHORYLASE 3) | |||||||||||
| at5g37980 | NADP-dependent oxidoreductase, putative | |||||||||||
| at1g78370 | ATGSTU20 (GLUTATHIONE S-TRANSFERASE TAU 20); | |||||||||||
| at5g43780 | APS4; sulfate adenylyltransferase (ATP) | |||||||||||
| at4g25100 | FSD1 (Fe SUPEROXIDE DISMUTASE 1) | |||||||||||
| at4g39950 | CYP79B2; electron carrier/heme binding/iron ion binding monooxygenase/oxygen binding | |||||||||||
| at5g23020 | IMS2 (2-ISOPROPYLMALATE SYNTHASE 2) | |||||||||||
| at4g01430 | Nodulin MtN21 family protein | |||||||||||
| at4g31500 | CYP83B1 (CYTOCHROME P450 MONOOXYGENASE 83B1) | |||||||||||
| at5g23010 | MAM1 (METHYLTHIOALKYLMALATE SYNTHASE 1 | |||||||||||
The data sets from plants genetically impaired in their mitochondrial function corresponded to the following genetic backgrounds: aox1a, (loss of alternative oxidase); ndufs4 and ndufa1 (loss of complex I); msh1xrecA, (mitochondrial genome rearrangement, MGR); AP3:u-ATP9 and AP9:u-ATP9 (loss of mitochondrial ATP synthase); msd1-RNAi (loss of mitochondrial manganese superoxide dismutase, MSD1, protein) and prxII F (inhibition of complex III) (Curley et al., 2009). The data sets from plants having their mitochondrial respiratory chain inhibited by the following chemical treatments: olygomicyn A (OLM) and rotenone (ROT) (Bales and Perkeybile, 2012), and Antimycin A (AA) (Curley et al., 2009). Up-regulated genes are in green and down-regulated genes are in red.
SULTR1;2 is a high affinity transporter from Arabidopsis (group 1), which mediates sulfate uptake into roots (Gigolashvili and Kopriva, 2014). SULTR 3;4 is a member of group 3 of S-tranporters, which are likely to be involved in translocation from root to shoot, whereas SULTR4;2 (group 4) functions in vacuolar export of .
BGLU28 and SDI1 have received considerable attention. BGLU28 is the most strongly up-regulated gene characterized in several of the studies related to S deficiency. It encodes a protein hypothesized to act by releasing S from glucosinolates, a major potential S storage compound in the vacuole (Maruyama-Nakashita et al., 2003, 2006; Dan et al., 2007). SDI1 is annotated as a protein similar to male sterility family protein MS5, and recent evidences suggested that its expression level could act as a biosensor of S nutrient status (Howarth et al., 2009).
Interestingly, these S-responsive genes are strongly up-regulated under a specific mitochondrial impairment: rotenone treatment, which inhibits complex I activity (Schwarzländer et al., 2012). As recently reported, S deficiency affects complex I activity (Ostaszewska et al., 2014) likely because this complex has a high need of Fe–S cluster-containing proteins. Therefore, an inhibition of complex I might trigger the expression of S-responsive genes. Interestingly, such genes are crucial for S-sensing and signaling. Indeed SULTR1;2 has a major role in transport, and it has been recently suggested to act as a possible S-transceptor (Zheng et al., 2014). Furthermore, the transcriptional regulation of the SULTR1;2, BGLU28, and SDI1 genes is under the control of SLIM1, a central transcription factor regulating the S-response in plants. Therefore, under S deficiency, the affected complex I activity might participate in the regulation of such genes through an unknown retrograde signaling pathway.
Conclusion and perspective
It has been suggested that Fe and S deficiencies might trigger retrograde signaling pathways to regulate the expression of genes. However, no clear evidence demonstrating such hypothesis has been reported so far. Considering that both Fe and S deficiencies affect mitochondrial respiration, it can be hypothesized that the impaired respiratory chain might be at the origin of putative retrograde signals under such nutritional deficiencies. In agreement with such a hypothesis, we observed that the expression of several Fe- and S-responsive genes was affected in plants with an induced mitochondrial dysfunction, as reported in the study by Schwarzländer et al. (2012). Interestingly, these genes exhibited a similar regulation (up or down) under Fe or S deficiencies and respiratory chain impairments. These observations underline the possible role of the respiratory chain impairment under Fe and S deficiencies in the regulation of some Fe and S-responsive genes. Here, we hypothesize that Fe and S deficiencies, by triggering mitochondrial impairments, promote the generation of specific signal(s) targeted from this organelle to the nucleus, leading to the up-regulation of nuclear-encoded genes involved in the establishment of Fe and S homeostasis, respectively (Figure 1). However, mitochondria might be considered only as one of the players in the complex nutrient-sensing and signaling mechanisms, as other more direct regulations occur (Figure 1). Indeed, it has been proposed that direct binding of Fe to transcriptional regulators would be the primary Fe-sensing event in plant cells (Kobayashi and Nishizawa, 2014, 2015). Putative retrograde signaling pathways might, therefore, occur in a second time of nutrient sensing. Considering that mitochondrial impairments should occur on the long-term in response to Fe or S starvation, we suggest that mitochondria might come into the regulation of nutrient-responsive genes at a later stage. Plant responses to nutrient deficiencies, such as Fe or S, are more complex than the simple activation of nutrient uptake/translocation mechanisms. Indeed, a complex metabolic reprogramming occurs in the cell in order to adapt itself to the low nutrient availability, as in the case of Fe (Vigani, 2012).
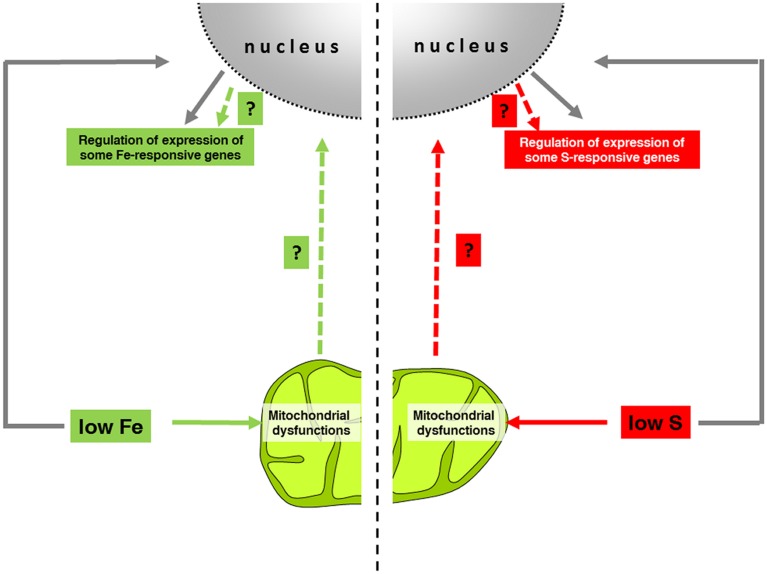
Figure 1.
Schematized model of the possible role of mitochondria in the regulation of nutrient-responsive genes. As Fe and S are essential elements for Fe–S cluster-containing proteins, deficiencies of such nutrients trigger mitochondrial dysfunctions mainly at the respiratory chain level. Through the comparison of different data sets (mitochondrial dysfunctions vs. Fe- and/or S-responsive genes), nutrient-responsive genes have been identified as potential candidates of hypothetical retrograde signaling pathways (PYE, BTS, FRO3; OPT3, SAM1 for Fe homeostasis and SULTR1;2 SULTR3;4, SULTR4;2, LSU1, BGLU28, SDI1 for S homeostasis). Mitochondrial impairments occur in long term of Fe and S starvations; therefore, such retrograde signaling would occur in a second phase of the regulation of nuclear gene expression, whereas the first phase (gray arrows) involved a more direct nutrient-sensing mechanisms.
Interestingly, among the Fe-responsive genes reported by Stein and Waters (2012), 45% of them were similarly regulated under Fe deficiency and mitochondrial dysfunctions. Furthermore, the expression of 45% of the PYE-regulated genes was affected by mitochondrial dysfunctions. Whereas among the S-responsive genes considered (Maruyama-Nakashita et al., 2006), 38% of them were similarly regulated under S deficiency and mitochondrial dysfunction. Complex I impairments seem to induce a differential expression of the majority of Fe- and S-responsive genes (Tables 1–3). It is well known that complex I deficiencies dramatically impact on cellular physiology, and numerous diseases have been linked with the impairment of complex I (Kühn et al., 2015). In humans, complex I is the main origin of diseases resulting from mitochondrial dysfunction (Nouws et al., 2012). However, in plants, the absence of complex I does not cause premature death as it occurs in humans (Kühn et al., 2015 and reference therein). Indeed, in plants, bypasses do exist for complex I (i.e., NDex), allowing even more metabolic flexibility than in human mitochondria. Recently, a central role of NDex in the regulation of cellular metabolism has been suggested. Suppression of the external mitochondrial NADPH dehydrogenase NDB1 affects global gene expression in Arabidopsis plants (Wallström et al., 2014), thereby suggesting that changes in nicotinamide redox level can selectively affect particular process of the cell. In fact, the existence of a NAD(P)H signaling process in plants has been suggested (Noctor, 2006). Under both Fe and S deficiencies, the increased activity of NDex along with the decreased activity of complex I might indicate that both nutrient deficiencies affect the nicotinamide redox level of the cell. Therefore, a possible NAD(P)H signaling pathway might be involved in the regulation of such nutrient-responsive genes. In this case, the metabolic status of mitochondria might produce non-specific signals. Therefore, the regulation of nutrient-responsive genes might result from integrated signaling pathways, involving specific and non-specific signals.
However, it cannot be ruled out that the above-reported overlap in the gene expression response to mitochondria dysfunctions or to Fe or S deficiencies could be simply be random and due to transcriptional noise, rather than indicative of a specific induction of nutrient starvation responses. Indeed, it is important to keep in mind that transcription is intrinsically stochastic and a small overlap may or may not be of physiological significance.
Furthermore, the transcriptomic data sets used here were obtained from different plant materials: cell culture, plant tissues, and at different plant developmental stages. It would be, therefore, important to investigate the possible role of mitochondria in the regulation of gene expression, specifically by differentiating cell culture from plant tissues and by differentiating root from shoot tissues of plant sampled at a given developmental stage. The cross-talk between mitochondria and plastids in roots should differ from that one between mitochondria and chloroplasts, and thereby the effect of respiratory chain impairment might be integrated in different signaling pathways in roots and leaves.
In order to clarify these points, it will be important to elucidate whether the impairment of the respiratory chain occurring under Fe and S deficiencies might affect the expression of some nutrient-regulated genes or whether it should be considered simply as a non-specific effect of the nutrient starvation. As the putative signals generated by impaired mitochondria might be numerous, the characterization of the retrograde pathway occurring under nutritional disorder could be complex. However, in order to elucidate the possible impact of the impaired respiratory chain on the expression of crucial Fe- and S-responsive genes, it would be helpful to evaluate the impact of respiratory chain inhibitors on the expression of Fe- and S-responsive genes in wild type and mutant plants that are defective in expression of important transcription factors (PYE/BTS, for Fe and SLIM, for S).
Author contributions
GV conceived the idea and wrote the draft of the manuscript. JFB critically reviewed for important intelectual content the manuscript.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
Research of GV was supported by Fondo per gli Investimenti della Ricerca di Base (FIRB) Futuro in Ricerca 2012 (project code RBFR127WJ9) founded by the Italian Ministry of Education (MIUR). Part of the work of JB related to this article was funded by the Agence Nationale de la Recherche (ANR “Fe–S traffic” n° ANR-13-BSV6-0002-02).
References
- Astolfi S., Cesco S., Zuchi S., Neumann G., Roemheld V. (2006). Sulfur starvation reduces phytosiderophores release by Fe-deficient barley plants. Soil Sci. Plant Nutr. 52, 80–85. 10.1111/j.1747-0765.2006.00010.x [DOI] [Google Scholar]
- Astolfi S., Zuchi S., Neumann G., Cesco S., Sanità di Toppi L., Pinton R. (2012). Response of barley plants to Fe defciency and Cd contamination as affected by S starvation. J. Exp. Bot. 63, 1241–1250. 10.1093/jxb/err344 [DOI] [PubMed] [Google Scholar]
- Atkin O. K., Bruhn D., Hurry V. M., Tjoelker M. G. (2005). The hot and the cold: unravelling the variable responses underpinning respiratory acclimation to the cold in Arabidopsis thaliana leaves. Funct. Plant Biol. 32, 87–105. 10.1071/FP03176 [DOI] [PubMed] [Google Scholar]
- Atkin O. K., Macherel D. (2009). The crucial role of plant mitochondria in orchestrating drought tolerance. Ann. Bot. 103, 581–597. 10.1093/aob/mcn094 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Balk J., Schaedler T. A. (2014). Iron cofactor assembly in plants. Annu. Rev. Plant Biol. 65, 125–153. 10.1146/annurev-arplant-050213-035759 [DOI] [PubMed] [Google Scholar]
- Bernard D. G., Cheng Y., Zhao Y., Balk J. (2009). An allelic mutant series of ATM3 reveals its key role in the biogenesis of cytosolic iron-sulfur proteins in Arabidospsis. Plant Physiol. 151, 590–602. 10.1104/pp.109.143651 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Briat J. F., Dubos C., Gaymard F. (2015a). Iron nutrition, biomass production, and plant product quality. Trends Plant Sci. 20, 33–40. 10.1016/j.tplants.2014.07.005 [DOI] [PubMed] [Google Scholar]
- Briat J. F., Rouached H., Tissot N., Gaymard F., Dubos C. (2015b). Integration of P, S, Fe, and Zn nutrition signals in Arabidospsis thaliana: potential involvement of PHOSPHATE STARVATION RESPONSE 1 (PHR1). Front. Plant Sci. 6:290. 10.3389/fpls.2015.00290 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Busi M. V., Gomez-Lobato M. E., Rius S. P., Turowski V. R., Casati P., Zabaleta E. J., et al. (2011). Effect of mitochondrial dysfunction on carbon metabolism and gene expression in flower tissues of Arabidopsis thaliana. Mol. Plant 4, 127–143. 10.1093/mp/ssq065 [DOI] [PubMed] [Google Scholar]
- Ciaffi M., Paolacci A. R., Celletti S., Catarcione G., Kopriva S., Astolfi S. (2013). Transcriptional and physiological changes in the S assimilation pathway due to single or combined S and Fe deprivation in durum wheat (Triticum durum L.) seedlings. J. Exp. Bot. 64, 1663–1675. 10.1093/jxb/ert027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clifton R., Liester R., Parker K. L., Sappl P. G., Elhafez D., Millar A. H., et al. (2005). Stress-induced co-expression of alternative respiratory chain components in Arabidopsis thaliana. Plant Mol. Biol. 58, 193–212. 10.1007/s11103-005-5514-7 [DOI] [PubMed] [Google Scholar]
- Couturier J., Touraine B., Briat J. F., Gaymard F., Rouhier N. (2013). The iron-sulfur cluster assembly machineries in plants: current knowledge and open questions. Front. Plant Sci. 4:259. 10.3389/fpls.2013.00259 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Curie C., Cassin G., Couch D., Divol F., Higuchi K., Le Jean M., et al. (2009). Metal movement within the plant: contribution of nicotianamine and yellow stripe 1-like transporters. Ann. Bot. 103, 1–11. 10.1093/aob/mcn207 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dan H., Yang G., Zheng Z. L. (2007). A negative regulatory role for auxin in sulphate deficiency response in Arabidopsis thaliana. Plant Mol. Biol. 63, 221–235. 10.1007/s11103-006-9084-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Forieri I., Wirtz M., Hell R. (2013). Towards new perspectives on the interaction of iron and sulfur metabolism in plants. Front. Plant Sci. 4:357. 10.3389/fpls.2013.00357 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gigolashvili T., Kopriva S. (2014). Transporters in plant sulfur metabolism. Front. Plant Sci. 5:442. 10.3389/fpls.2014.00442 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giraud E., Ho L. H., Clifton R., Carroll A., Estavillo G., Tan Y. F., et al. (2008). The absence of ALTERNATIVE OXIDASE1a in Arabidopsis results in acute sensitivity to combined light and drought stress. Plant Physiol. 147, 595–610. 10.1104/pp.107.115121 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Higa A., Mori Y., Kitamura Y. (2010). Iron deficiency induces changes in riboflavin secretion and the mitochondrial electron transport chain in hairy roots of Hyoscyamus albus. J. Plant Physiol. 167, 870–878. 10.1016/j.jplph.2010.01.011 [DOI] [PubMed] [Google Scholar]
- Howarth J. R., Pamar S., Barraclough P. B., Hawkesford M. J. (2009). A sulfur deficiency-induced gene, sdi1, involved in the utilization of stored sulphate pools under sulfur-limiting conditions has potential as a diagnostic indicator of sulfur nutritional status. Plant Biotechnol. J. 7, 200–209. 10.1111/j.1467-7652.2008.00391.x [DOI] [PubMed] [Google Scholar]
- Jacoby R. P., Li L., Huang S., Pong Lee C., Millar A. H., Taylor N. L. (2012). Mitochondrial composition, function and stress response in plants. J. Integr. Plant Biol. 54, 887–906. 10.1111/j.1744-7909.2012.01177.x [DOI] [PubMed] [Google Scholar]
- Jacoby R. P., Tajlor N. L., Millar A. H. (2011). The role of mitochondrial respiration in salinity tolerance. Trends Plant Sci. 16, 614–623. 10.1016/j.tplants.2011.08.002 [DOI] [PubMed] [Google Scholar]
- Knuesting J., Riondet C., Maria C., Kruse I., Becuwe N., Konig N., et al. (2015). Arabidopsis glutaredoxin S17 and its partner, the nuclear factor Y subunit C11/negative cofactor 2α, contribute to maintenance of the shoot apical meristem under long-day photoperiod. Plant Physiol. 167, 1643–1658. 10.1104/pp.15.00049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kobayashi T., Nishizawa N. K. (2012). Iron uptake, translocation, and regulation in higher plants. Annu. Rev. Plant Biol. 63, 131–152. 10.1146/annurev-arplant-042811-105522 [DOI] [PubMed] [Google Scholar]
- Kobayashi T., Nishizawa N. K. (2014). Iron sensors and signals in response to iron deficiency. Plant Sci. 224, 36–43. 10.1016/j.plantsci.2014.04.002 [DOI] [PubMed] [Google Scholar]
- Kobayashi T., Nishizawa N. K. (2015). Intracellular iron sensing by the direct binding of iron to regulators. Front. Plant Sci. 6:155. 10.3389/fpls.2015.00155 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kühn K., Obata T., Feher K., Bock R., Fernie A. R., Meyer E. H. (2015). Complete mitochondrial complex I deficiency induces an up-regulation of respiratory fluxes that is abolished by traces of functional complex I. Plant Physiol. 168, 1537–1549. 10.1104/pp.15.00589 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuwajima K., Kawai S. (1997). Relationship between sulfur metabolism and biosynthesis of phytosiderophores in barley roots, in Plant Nutrition for Sustainable Food Production and Environnment. Developments in Plant and Soil Sciences, Vol. 78, eds Ando T., Fujita K., Mae T., Matsumoto H., Mori S., Sekiya J. (Dordrech: Kluwer Academic Publishers; ), 285–286. [Google Scholar]
- Long T. A., Tsukagoshi H., Busch W., Lahner B., Salt D. E., Benfey P. N. (2010). The bHLH transcription factor POPEYE regulates response to iron deficiency in Arabidopsis roots. Plant Cell 22, 2219–2236. 10.1105/tpc.110.074096 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maruyama-Nakashita A., Inoue E., Watanabe-Takahashi A., Yamaya T., Takahaschi H. (2003). Transcriptome profiling of sulfur-responsive genes in Arabidopsis reveals global effects of sulfur nutrition on multiple metabolic pathways. Plant Physiol. 132, 597–605. 10.1104/pp.102.019802 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maruyama-Nakashita A., Nakamura Y., Tohge T., Saito K., Takahashi H. (2006). Arabidopsis SLIM1 is a central transcriptional regulator of plant sulfur response and metabolism. Plant Cell 18, 3235–3251. 10.1105/tpc.106.046458 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyer E. H., Tomaz T., Carroll A. J., Estavillo G., Delannoy E., Tanz S. K., et al. (2009). Remodelled respiration in ndufs4 with low phosphorylation efficiency suppresses Arabidopsis germination and growth and alters control of metabolism at night. Plant Physiol. 151, 603–619. 10.1104/pp.109.141770 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noctor G. (2006). Metabolic signalling in defence and stress: the central roles of soluble redox couples. Plant Cell Environ. 15, 473–497. 10.1111/j.1365-3040.2005.01476.x [DOI] [PubMed] [Google Scholar]
- Nouws J., Nijtmans L. G., Smeitink J. A., Vogel R. O. (2012). Assembly factors as a new class of desease genes for mitochondrial complex I deficiency: cause, pathology and treatment options. Brain 135, 12–22. 10.1093/brain/awr261 [DOI] [PubMed] [Google Scholar]
- Ostaszewska M., Juszczuk I. M., Kolodziejek I., Rychter A. M. (2014). Long-term sulfur starvation of Arabidopsis thaliana modifies mitochondrial ultrastructure and activity and changes tissues energy and redox status. J. Plant Physiol. 172, 549–558. 10.1016/j.jplph.2013.12.013 [DOI] [PubMed] [Google Scholar]
- Paolacci A. R., Celletti S., Catarcione G., Hawkesford M. J., Astolfi S., Ciaffi M. (2014). Iron deprivation results in a rapid but not sustained increase of the expression of genes involved in iron metabolism and sulfate uptake in tomato (Solanum lycopersicum L.) seedlings. J. Integr. Plant Biol. 56, 88–100. 10.1111/jipb.12110 [DOI] [PubMed] [Google Scholar]
- Rutherford J. C., Bird A. J. (2004). Metal-responsive transcription factors that regulate iron, zinc, and copper homeostasis in eukatyotic cells. Eukar. Cell 3, 1–13. 10.1128/EC.3.1.1-13.2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schuler M., Keller A., Backes C., Philippar K., Lenhof H. P., Bauer P. (2011). Transcriptome analysis by GeneTrail revealed regulation of functional categories in response to alterations of iron homeostasis in Arabidopsis thaliana. BMC Plant Biol. 11:87. 10.1186/1471-2229-11-87 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwarzländer M., Finkemeier I. (2013). Mitochondrial energy and redox signaling in plants. Antioxid. Redox Signal. 18, 2122–2143. 10.1089/ars.2012.5104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwarzländer M., Konig A. C., Sweetlove L. J., Finkemeier I. (2012). The impact of impaired mitochondrial function on retrograde signalling: a meta-analysis of transcriptomic responses. J. Exp. Bot. 63, 1735–1750. 10.1093/jxb/err374 [DOI] [PubMed] [Google Scholar]
- Selote D., Samire R., Matthiadis A., Gillikin J. W., Long T. A. (2015). Iron-binding E3 ligase mediates iron response in plants by targeting basic helix-loop-helix transcription factors. Plant Physiol. 167, 273–286. 10.1104/pp.114.250837 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shedge V., Davila J., Arrieta-Montiel M. P., Mohammed S., Mackenzie S. A. (2010). Extensive rearrangement of the Arabidopsis mitochondrial genome elicits cellular conditions for thermotolerance. Plant Physiol. 152, 1960–1970. 10.1104/pp.109.152827 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stein R. J., Waters B. M. (2012). Use of natural variation reveals core genes in the transcriptome of iron-deficient Arabidopsis thaliana roots. J. Exp. Bot. 63, 1039–1055. 10.1093/jxb/err343 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takahaschi H., Kopriva S., Giordano M., Saito K., Hell R. (2011). Sulfur Assimilation in Photosynthetic Organisms: molecular functions and regulations of transporters and assimilatory enzymes. Annu. Rev. Plant Biol. 62, 157–184. 10.1146/annurev-arplant-042110-103921 [DOI] [PubMed] [Google Scholar]
- Ueta R., Fujiwara N., Iwai K., Yamaguchi-Iwai Y. (2012). Iron-Induced dissociation of the Aft1p transcriptional regulator from target gene promoters is an initial event in iron-dependent gene suppression. Mol. Cell Biol. 32, 4998–5008. 10.1128/MCB.00726-12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Aken O., Zhang B., Carrie C., Uggalla V., Paynter E., Giraud E., et al. (2009). Defining the mitochondrial stress response in Arabidopsis thaliana. Mol. Plant 2, 1310–1324. 10.1093/mp/ssp053 [DOI] [PubMed] [Google Scholar]
- Vigani G. (2012). Discovering the role of mitochondria in the iron deficiency-induced metabolic responses of plants. J. Plant Physiol. 169, 1–11. 10.1016/j.jplph.2011.09.008 [DOI] [PubMed] [Google Scholar]
- Vigani G., Bashir K., Ishimaru Y., Lehmann M., Casiraghi F. M., Nakanishi H., et al. (2015b). Knocking down mitochondrial iron transporter (MIT) reprograms primary and secondary metabolism in rice plants. J. Exp. Bot. 10.1093/jxb/erv531. [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vigani G., Faoro F., Ferretti A. M., Cantele F., Maffi D., Marelli M., et al. (2015a). Three-dimensional reconstruction, by TEM tomography, of the ultrastructural modifications occurring in Cucumis sativus L. mitochondria under Fe deficiency. PLoS ONE 10:e0129141. 10.1371/journal.pone.0129141 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vigani G., Maffi D., Zocchi G. (2009). Iron availability affects the function of mitochondria in cucumber root. New Phytol. 182, 127–136. 10.1111/j.1469-8137.2008.02747.x [DOI] [PubMed] [Google Scholar]
- Vigani G., Zocchi G. (2010). Effect of Fe deficiency on mitochondrial alternative NAD(P)H dehydrogenases in cucumber roots. J. Plant Physiol. 167, 666–669. 10.1016/j.jplph.2009.12.006 [DOI] [PubMed] [Google Scholar]
- Vigani G., Zocchi G., Bashir K., Philippar K., Briat J. F. (2013). Signal from chloroplasts and mitochondria for iron homeostasis regulation. Trends Plant Sci. 18, 305–311. 10.1016/j.tplants.2013.01.006 [DOI] [PubMed] [Google Scholar]
- Wallström S. V., Florez-Sarasa I., Araujo W. L., Aidemark M., Fernandez-Fernandez M., Fernie A. R., et al. (2014). Suppression of the external mitochondrial NADPH dehydrogenase, NDB1, in Arabidopsis thaliana affects central metabolism and vegetative growth. Mol. Plant 2, 356–368. 10.1093/mp/sst115 [DOI] [PubMed] [Google Scholar]
- Wirtz M., Beard K. F., Lee C. P., Boltz A., Schwarzlander M., Fuchs C., et al. (2012). Mitochondrial cysteine synthase complex regulates O-acetylserine biosynthesis in plants. J. Biol. Chem. 287, 27941–27947. 10.1074/jbc.M112.372656 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng Z. L., Zhang B., Leustek T. (2014). Transceptors at the boundary of nutrient transporters and receptors: a new role for Arabidopsis SULTR1;2 in sulfur sensing. Front. Plant Sci. 5:710. 10.3389/fpls.2014.00710 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zuchi S., Cesco S., Varanini Z., Pinton R., Astolfi S. (2009). Sulfur deprivation limits Fe-deficiency responses in tomato plants. Planta 230, 85–94. 10.1007/s00425-009-0919-1 [DOI] [PubMed] [Google Scholar]
- Zuchi S., Watanabe M., Hubberten H. M., Bromke M., Osorio S., Fernie A. R., et al. (2015). The interplay between sulfur and iron nutrition in tomato. Plant Physiol. 169, 2624–2639. 10.1104/pp.15.00995 [DOI] [PMC free article] [PubMed] [Google Scholar]