Abstract
Purpose:
The goal of this study was to validate the ultrasonography (US) and cytopathological features that are used in the diagnosis of the follicular variant of papillary thyroid carcinoma (FVPTC) and to characterize the role of BRAFV600E mutation analysis in the diagnosis of FVPTC.
Methods:
From May 2012 to February 2014, 40 thyroid nodules from 40 patients (mean age, 56.2 years; range, 26 to 81 years) diagnosed with FVPTC were included in this study. The US features of the nodules were analyzed and the nodules were classified as probably benign or suspicious for malignancy. Twenty-three thyroid nodules (57.5%) underwent BRAFV600E mutation analysis. Clinical information and histopathologic results were obtained by reviewing the medical records of the patients.
Results:
Thirty nodules (75.0%) were classified as suspicious for malignancy, while 10 (25.0%) were classified as probably benign. Seven of the eight nodules (87.5%) with atypia of undetermined significance/follicular lesion of undetermined significance (AUS/FLUS) cytology showed suspicious US features, while one of the two nodules (50.0%) with follicular neoplasm cytology presented suspicious US features. Five of the 23 nodules (21.7%) that underwent BRAFV600E mutation analysis had positive results, all of which were diagnosed as suspicious for malignancy or malignant based on cytology. None of the nodules with benign, AUS/FLUS, or follicular neoplasm cytology were positive for the BRAFV600E mutation.
Conclusion:
US features allow nodules to be classified as suspicious for malignancy, and the presence of suspicious US features in nodules with ambiguous cytology may aid in the diagnosis of FVPTC. BRAFV600E mutation analysis is of limited value in the diagnosis of FVPTC.
Keywords: Thyroid gland; Ultrasonography; Carcinoma, papillary; Carcinoma, papillary, follicular; Biopsy, fine-needle
Introduction
Papillary thyroid carcinoma (PTC) is the most common malignancy of the thyroid gland [1,2], and numerous variants of PTC have been reported with distinct histopathological features [3]. Conventional or classical PTC is the most frequent type, while the follicular variant of papillary thyroid carcinoma (FVPTC) is the second most common variant; these variants comprise approximately 55%-66% and 11.8%-31.6% of well differentiated thyroid carcinomas, respectively [1-5]. Cytologically, FVPTC contains cells that show the nuclear features of PTC, such as ground glass nuclei, nuclear grooves, and overlapping nuclei with or without nuclear inclusions [2,6], with a predominantly follicular growth pattern [4,7]. Although FVPTC has been reported to have a lower rate of lymph node metastasis and extrathyroidal extension-than classical PTC [3,7,8], but a higher rate of lung and bone metastasis [2,9], it is generally accepted that the clinical behavior of FVPTC resembles that of PTC, with similar patient outcomes [3,4,8,10,11]. Since the prognosis and the long-term survival outcomes of patients with FVPTC are similar to those of patients with classical PTC, the same treatment strategies are currently applied to this variant form of thyroid carcinoma [3-5,11].
Many studies have evaluated the clinicopathological features and long-term outcomes of FVPTC alone or in comparison with classical PTC [3,4,6-8,10,11], whereas few studies have evaluated the ultrasonography (US) features of FVPTC [2,9]. Moreover, in light of the excellent sensitivity of BRAFV600E mutation analysis for detecting PTC, several recent studies have evaluated the role of BRAFV600E as a prognostic factor for FVPTC [1,12,13]. However, few studies have assessed correlations between US features and the cytological results of US-guided fine needle aspiration (US-FNA) in nodules diagnosed as FVPTC. In this study, we evaluated the preoperative US features, cytopathological characteristics, and BRAFV600E mutation analysis results in thyroid nodules diagnosed as FVPTC to determine whether any of those parameters are useful in predicting the diagnosis of FVPTC.
Materials and Methods
This retrospective study was approved by the Institutional Review Board of Severance Hospital, Seoul, Korea. Neither patient approval nor informed consent was required for the review of medical records or images. Written informed consent was obtained from all patients before US-FNA and before all surgical procedures.
Patients
From May 2012 to February 2014, 5,128 patients with 5,967 thyroid nodules underwent US-FNA at our institution. Among them, 40 thyroid nodules (0.7%) from 40 patients were histologically diagnosed as FVPTC after surgical excision. The mean age of the patients was 56.2 years (range, 26 to 81 years). Six (15.0%) were men and 34 (85.0%) were women. Of the 40 patients, 17 (42.5%) underwent lobectomy, while the remaining 23 (57.5%) underwent total thyroidectomy. Clinical information and histopathologic results were obtained by reviewing the medical records of the patients.
US, US-FNA, and BRAFV600E Mutation Analysis
US was performed using a 5-12-MHz linear array transducer (iU22; Philips Medical Systems, Bothell, WA, USA). Compound imaging was used to obtain all images. Real-time US and subsequent US-FNA were performed by one of seven board-certified radiologists (four faculty, three fellows) with 1-15 years of experience in thyroid imaging. US-FNA was performed on thyroid nodules showing suspicious US features or on the largest mass when none of the multiple thyroid nodules observed showed any suspicious US features.
The US features of each thyroid nodule were described according to the following categories: internal components, echogenicity, margin, calcifications, and shape [14]. Based on their internal components, nodules were classified as solid, predominantly solid (solid contents comprising more than 50% of the nodule), or predominantly cystic (solid contents comprising less than 50% of the nodule). Based on echogenicity, nodules were classified as hyperechoic or isoechoic (nodules showing hyperechogenicity or isoechogenicity when compared to the surrounding thyroid parenchyma), hypoechoic (nodules showing hypoechogenicity compared to the surrounding thyroid parenchyma), and markedly hypoechogenic (nodules showing hypoechogenicity compared to the adjacent strap muscle). The margins were classified as circumscribed or non-circumscribed (i.e., microlobulated or irregular margins). Based on the presence of calcifications, nodules were classified as having no calcifications, microcalcifications or mixed calcifications, or macrocalcifications including eggshell calcifications. The shape was classified as parallel or non-parallel (greater in the anteroposterior dimension than the transverse dimension, or ‘taller than wide’). Of these features, the US features considered indicative of malignancy were marked hypoechogenicity, non-circumscribed margins, microcalcifications or mixed calcifications, and a nonparallel shape, based on previously established criteria [14]. The final assessment of the thyroid nodules was ‘probably benign’ when none of the suspicious US features described above were present, and as ‘suspicious for malignancy’ when one or more of the suspicious US features were present.
US-FNA was performed at least twice from each thyroid nodule using a 23-gauge needle attached to a 2-mL disposable syringe without an aspirator. Local anesthesia was not routinely applied. The aspirated material was expelled onto glass slides and immediately placed in 95% ethanol for Papanicolaou staining. The remaining material in the syringe was rinsed in saline for cell block processing. The cytopathologists were not present during the procedures, and additional staining was performed on a case-by-case basis at the request of the cytopathologists. One of the seven cytopathologists specializing in thyroid pathology interpreted the cytology slides. During the period of the study, cytology reports involving US-FNA of the thyroid classified nodules according to the six categories in the Bethesda System for Reporting Cytopathology: non-diagnostic, benign, atypia of undetermined significance/follicular lesion of undetermined significance (AUS/FLUS), follicular neoplasm or suspicious for follicular neoplasm, suspicious for malignancy, and malignancy [15].
During the US-FNA procedures, aspiration was performed once more for BRAFV600E mutation analysis if requested by the referring physicians. Of the 40 thyroid nodules included in this study, 23 (57.5%) underwent BRAFV600E mutation analysis. The aspirated material was rinsed in 1 mL of normal saline, which was sent for BRAFV600E mutation analysis. Real-time polymerase chain reaction was used for detecting the BRAFV600E mutation during the study period at our institution [16].
Two of the 40 thyroid nodules underwent US-guided core needle biopsy due to repeated inconclusive cytology results. This procedure was performed using the freehand technique with a spring-activated 18-gauge Tru-Cut-type needle (Acecut, TSK Laboratory, Tochigi, Japan). At least three biopsy cores were obtained, and the specimen was fixed in formalin solution and sent to the pathology department.
Results
The size of the 40 thyroid nodules confirmed as FVPTC ranged from 4 to 53 mm (mean, 12.6±12.2 mm). Four nodules (10.0%) were palpable, while the remaining 36 (90.0%) were incidentally detected. Table 1 summarizes the clinical and pathological features of the 40 patients included in this study. Twenty-five nodules (62.5%) were encapsulated, while 15 (37.5%) were pathologically determined to be infiltrative. Six patients (15.0%) had multifocal lesions, and the remaining 34 (85.0%) had single lesions. Central or lateral cervical lymph node metastases were found in seven patients (17.5%) and two patients (5.0%), respectively.
Table 1.
Clinical and pathological features the follicular variant of papillary thyroid carcinoma, based on samples of 40 nodules from 40 patients
| Characteristic | No. (%) |
|---|---|
| Age (yr) | 56.2±13.3 |
| ≤45 | 10 (25.0) |
| >45 | 30 (75.0) |
| Sex | |
| Female | 34 (85.0) |
| Male | 6 (15.0) |
| Family history | |
| Absent | 35 (87.5) |
| Present | 5 (12.5) |
| Tumor size (cm) | 1.26±1.22 |
| <2 | 31 (77.5) |
| 2-4 | 6 (15.0) |
| >4 | 3 (7.5) |
| Histologic subtype | |
| Encapsulated | 25 (62.5) |
| Infiltrative | 15 (37.5) |
| Extrathyroid extension | |
| Absent | 34 (85.0) |
| Present | 6 (15.0) |
| Multicentricity | |
| Absent | 34 (85.0) |
| Present | 6 (15.0) |
| Central lymph node metastasis | |
| Absent | 33 (82.5) |
| Present | 7 (17.5) |
| Lateral lymph node metastasis | |
| Absent | 38 (95.0) |
| Present | 2 (5.0) |
Table 2 summarizes the US features of the 40 thyroid nodules diagnosed as FVPTC in this study. The common US features observed among the 40 thyroid nodules were as follows: solid composition (92.5%), hypoechogenicity (62.5%), microlobulated margins (55.0%), no calcifications (37.5%), and a parallel shape (67.5%). Thirty nodules (75.0%) were classified as suspicious for malignancy, while 10 (25.0%) were classified as probably benign.
Table 2.
Ultrasonography (US) features of the follicular variant of papillary thyroid carcinoma, based on 40 nodules from 40 patients
| US feature | No. (%) | |
|---|---|---|
| Composition | Solid | 37 (92.5) |
| Predominantly solid | 3 (7.5) | |
| Echogenicity | Hyperechoic to isoechoic | 9 (22.5) |
| Hypoechogenicity | 25 (62.5) | |
| Markedly hypoechogenicity | 6 (15.0) | |
| Margin | Well defined | 14 (35.0) |
| Microlobulated | 22 (55.0) | |
| Irregular | 4 (10.0) | |
| Calcifications | Microcalcifications and mixed calcifications | 13 (32.5) |
| Macrocalcifications and eggshell calcifications | 12 (30.0) | |
| Negative | 15 (37.5) | |
| Shape | Parallel | 27 (67.5) |
| Non-parallel | 13 (32.5) | |
| Parenchymal echogenicity | Homogeneous | 28 (70.0) |
| Heterogeneous | 12 (30.0) | |
| Final assessment | Probably benign | 10 (25.0) |
| Suspicious for malignancy | 30 (75.0) |
Table 3 shows the correlations of cytological characteristics with US features and the BRAFV600E mutation analysis results of the 40 FVPTC nodules. Of the 40 thyroid nodules, the preoperative cytology results were as follows: benign findings were observed in one nodule (2.5%) (Fig. 1), AUS/FLUS findings in eight nodules (20.0%) (Fig. 2), follicular neoplasm or suspicious for follicular neoplasm findings in two nodules (5.0%), a finding of suspicious for malignancy in 16 nodules (40.0%), and malignancy in 13 nodules (32.5%). Two nodules underwent US-guided core needle biopsy; both showed non-confirmative results, suggesting the possibility of follicular proliferative lesions. Ten nodules were assessed as probably benign, the cytology results were suspicious for malignancy or malignant in seven (70.0%), follicular neoplasm or suspicious for follicular neoplasm findings were observed in one nodule (10.0%), AUS/FLUS findings in one nodule (10.0%), and benign findings in one nodule (10.0%). Seven nodules (87.5%) with AUS/FLUS findings, one (50.0%) with a finding of follicular neoplasm or suspicious for follicular neoplasm, and 12 (75.0%) that were found to be suspicious for malignancy had suspicious US features. Five of the 23 nodules (21.7%) that underwent BRAFV600E mutation analysis had positive results, and all were diagnosed as suspicious for malignancy or malignant. None of the nodules with benign, AUS/FLUS, or follicular neoplasm cytology were positive for the BRAFV600E mutation.
Table 3.
Correlations of cytological characteristics with ultrasonography (US) features and the results of BRAFV600E mutation analysis in 40 nodules diagnosed as the follicular variant of papillary thyroid carcinoma, drawn from 40 patients
| Cytologya) | US feature |
BRAFb)
|
||||||
|---|---|---|---|---|---|---|---|---|
| Probably benign | Suspicious for malignancy | PPV (%) | Total | Negative | Positive | PPV (%) | Total | |
| Benign | 1 (10.0) | 0 | 0.0 | 1 (2.5) | 1 (5.6) | 0 | 0.0 | 1 (4.3) |
| AUS/FLUS | 1 (10.0) | 7 (23.3) | 87.5 | 8 (20.0) | 6 (33.3) | 0 | 0.0 | 6 (26.1) |
| FN | 1 (10.0) | 1 (3.3) | 50.0 | 2 (5.0) | 0 | 0 | 0.0 | 0 |
| SUSP | 4 (40.0) | 12 (40.0) | 75.0 | 16 (40.0) | 8 (44.4) | 2 (40.0) | 20.0 | 10 (43.5) |
| Malignancy | 3 (30.0) | 10 (33.3) | 76.9 | 13 (32.5) | 3 (16.7) | 3 (60.0) | 50.0 | 6 (26.1) |
Values are presented as number (%) unless otherwise indicated.
PPV, positive predictive value for US finding or BRAFV600E mutation result; AUS/FLUS, atypia of undetermined significance/follicular lesion of undetermined significance; FN, follicular neoplasm or suspicious for follicular neoplasm; SUSP, suspicious for malignancy.
According to the Bethesda System for Reporting Thyroid Cytopathology.
23 nodules that underwent BRAFV600E mutation analysis were included.
Fig. 1. An incidentally detected thyroid mass in a 32-year-old woman.
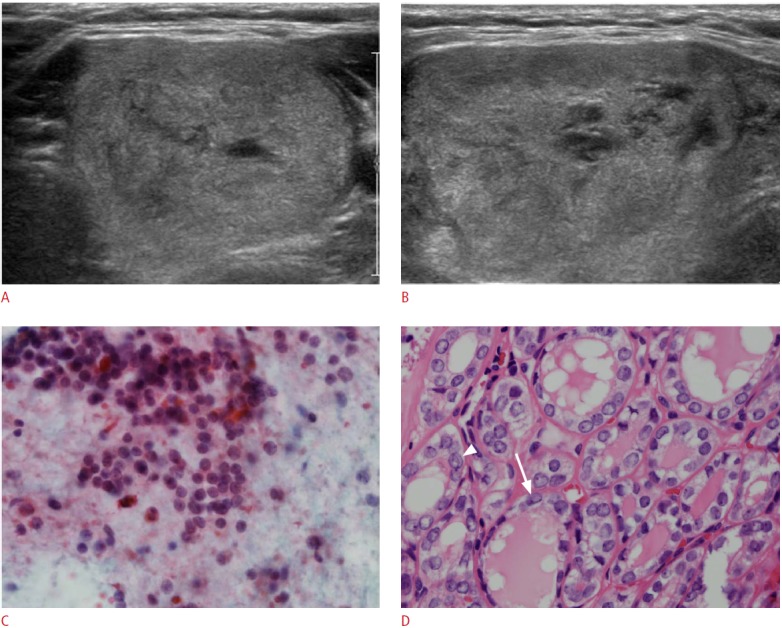
A, B. Ultrasonography (A, transverse; B, longitudinal) shows a 4.3-cm well-defined, predominantly solid mass with isoechoic features, a parallel shape, and no calcifications in the left thyroid. The final assessment of this thyroid mass was probably benign. C. Ultrasonographyguided fine needle aspiration cytology shows (H&E, ×400) sheets of follicular cells without significant atypia, and the results of BRAFV600E mutation analysis were negative. Diagnostic lobectomy was performed due to the large size of the mass, and follicular variant of papillary thyroid carcinoma was confirmed on pathology. D. Microscopic images of the tumor (H&E, ×400) shows neoplastic follicles containing eosinophilic colloid, lined by cells with irregular nuclei that show nuclear grooves (arrow) and suspicious pseudoinclusion (arrowhead).
Fig. 2. An incidentally detected thyroid mass in a 62-year-old woman.

A, B. Ultrasonography (A, transverse; B, longitudinal) shows a 2.3-cm hypoechoic solid mass with microlobulated margins, macrocalcifications, and a parallel shape in the left thyroid. The final assessment of this thyroid mass was suspicious for malignancy. C. Ultrasonography-guided fine needle aspiration (US-FNA) cytology (H&E, ×100) demonstrates a low cellular aspirate composed of microfollicles and scattered isolated cells, and the results BRAFV600E mutation analysis were negative. The patient underwent surgery due to the repeated presence of atypia of undetermined significance/follicular lesion of undetermined significance results on follow-up US-FNA, and this mass was confirmed as the follicular variant of papillary thyroid carcinoma on surgery. D. Microscopic images of the tumor (H&E, ×400) shows neoplastic follicles of variable size and shape, with lines near the cells containing eosinophilic colloid, lined by cells with irregular nuclei that show prominent nucleoli, nuclear grooves (arrows) and suspicious pseudoinclusion (arrowhead).
Discussion
FVPTC has been reported to have a relatively benign appearance compared to the typical US features seen in the classical variant of PTC [2,9]. The common US features of FVPTC observed in our study were a solid composition, hypoechogenicity, microlobulated margins, no calcifications, and a parallel shape, corresponding to the results of previous studies [2,9]. Each of these individual features is either a typically benign feature or is consistent with but not strongly suggestive of malignancy. Interestingly, although the most common US features of FVPTC in our study and in previous reports were typically benign, 75.0% (30 of 40 nodules) had more than one suspicious US feature. This observation indicates the importance of US features in predicting malignancy in thyroid nodules, which can be used as a guideline for patient management according to US-FNA cytology results.
US-FNA is considered the gold standard in the diagnosis of thyroid nodules, showing a high sensitivity of 83%-98% [8,12,14] and enabling benign nodules suitable for conservative follow-up to be differentiated from malignant nodules that require surgical intervention. However, US-FNA has limitations in the accurate diagnosis of FVPTC. The presence of a follicular arrangement of tumor cells is the cytological hallmark of FVPTC, and the presence of high cellularity, follicles and syncytial clusters, nuclear features of grooves, and enlarged nuclei with fine chromatin has been reported helpful in diagnosing FVPTC [3,17]. However, the characteristic cytological features described above are not always apparent, since these features may be focal or localized and not included during sampling procedures [3,16]. Moreover, it is difficult to distinguish between various follicular lesions of the thyroid, including benign adenomatous nodules, and follicular carcinomas based on cytological features alone [18]. The sensitivity of US-FNA cytology for the diagnosis of FVPTC has been reported to be lower than for the diagnosis of classical PTC [2,15]. Moreover, more suspicious cytological results are reported than malignant cytological results [2,3,19]. Our results follow this pattern; approximately 40.0% of the thyroid nodules had suspicious cytology results, compared to 32.5% with malignant cytology results. Seven of the eight nodules (87.5%) with AUS/FLUS results, one of the two nodules (50.0%) with a result of follicular neoplasm or suspicious for follicular neoplasm, and 12 of the 16 nodules (75.0%) found to be suspicious for malignancy showed suspicious US features. Based on these findings, we suggest that US features may help in the accurate diagnosis of FVPTC in thyroid nodules showing ambiguous cytology results by correctly guiding clinicians towards further diagnostic procedures or surgery.
Considering the difficulties involved in the definitive diagnosis of FVPTC based on cytological features alone, several reports have applied molecular studies including BRAFV600E mutation analysis to the diagnosis of FVPTC [1,12] in mutated lesions that already have suspicious or malignant cytology results [16]. Our results correspond to those of previous studies; five of the 23 nodules that underwent BRAFV600E mutation analysis were positive for the mutation, of which two had suspicious and three had malignant cytology results. None of the nodules with benign, AUS/FLUS, or follicular neoplasm cytology results showed positive results in BRAFV600E mutation analysis. Therefore, we suggest that BRAFV600E mutation analysis is of limited value for providing additional information in the differential diagnosis of FVPTC. Debate continues regarding the role of BRAFV600E mutations in the diagnosis of FVPTC, as FVPTC is known to be associated with a lower rate of the BRAFV600E mutation than classical PTC [20]. Moreover, several reports have shown a high frequency of RAS mutations in FVPTC [21], which may be useful in the preoperative diagnosis of FVPTC, although further research is required to reveal which molecular marker or markers are specific for FVPTC.
This study has some limitations. First, this was a retrospective study of lesions diagnosed as FVPTC after surgery, meaning that selection bias may have occurred. Larger prospective studies are anticipated in the future to validate our results. Second, seven cytopathologists were involved in the cytological slide review. Considerable interobserver variability exists in the diagnosis of FVPTC based on histology [12], but this issue was not considered in our study. Similarly, seven radiologists with different levels of experience in thyroid imaging reviewed the US images, meaning that interobserver variability may have affected those results. Fourth, only a limited number of nodules underwent BRAFV600E mutation analysis and core needle biopsy in this study. Further research involving a large number of cases will be required to conclusively establish the role of BRAFV600E mutation analysis and US-guided core needle biopsy in the diagnosis of FVPTC.
In conclusion, the US features of FVPTC have a relatively benign appearance, but most were ultimately classified as suspicious for malignancy. The presence of suspicious US features in nodules with ambiguous cytology may assist the accurate diagnosis of FVPTC. BRAFV600E mutation analysis is of limited value in the diagnosis of FVPTC, pending further validation studies.
Acknowledgments
This study was supported in part by the Research Fund of the Korean Society of Ultrasound in Medicine.
Footnotes
No potential conflict of interest relevant to this article was reported.
References
- 1.Chai YJ, Kim SJ, Kim SC, Koo DH, Min HS, Lee KE, et al. BRAF mutation in follicular variant of papillary thyroid carcinoma is associated with unfavourable clinicopathological characteristics and malignant features on ultrasonography. Clin Endocrinol (Oxf) 2014;81:432–439. doi: 10.1111/cen.12433. [DOI] [PubMed] [Google Scholar]
- 2.Yoon JH, Kim EK, Hong SW, Kwak JY, Kim MJ. Sonographic features of the follicular variant of papillary thyroid carcinoma. J Ultrasound Med. 2008;27:1431–1437. doi: 10.7863/jum.2008.27.10.1431. [DOI] [PubMed] [Google Scholar]
- 3.Ozdemir D, Ersoy R, Cuhaci N, Arpaci D, Ersoy EP, Korukluoglu B, et al. Classical and follicular variant papillary thyroid carcinoma: comparison of clinical, ultrasonographical, cytological, and histopathological features in 444 patients. Endocr Pathol. 2011;22:58–65. doi: 10.1007/s12022-011-9160-0. [DOI] [PubMed] [Google Scholar]
- 4.Burningham AR, Krishnan J, Davidson BJ, Ringel MD, Burman KD. Papillary and follicular variant of papillary carcinoma of the thyroid: initial presentation and response to therapy. Otolaryngol Head Neck Surg. 2005;132:840–844. doi: 10.1016/j.otohns.2005.02.002. [DOI] [PubMed] [Google Scholar]
- 5.Lang BH, Lo CY, Chan WF, Lam AK, Wan KY. Classical and follicular variant of papillary thyroid carcinoma: a comparative study on clinicopathologic features and long-term outcome. World J Surg. 2006;30:752–758. doi: 10.1007/s00268-005-0356-7. [DOI] [PubMed] [Google Scholar]
- 6.Hagag P, Hod N, Kummer E, Cohenpour M, Horne T, Weiss M. Follicular variant of papillary thyroid carcinoma: clinical-pathological characterization and long-term follow-up. Cancer J. 2006;12:275–282. doi: 10.1097/00130404-200607000-00005. [DOI] [PubMed] [Google Scholar]
- 7.Passler C, Prager G, Scheuba C, Niederle BE, Kaserer K, Zettinig G, et al. Follicular variant of papillary thyroid carcinoma: a long-term follow-up. Arch Surg. 2003;138:1362–1366. doi: 10.1001/archsurg.138.12.1362. [DOI] [PubMed] [Google Scholar]
- 8.Lin HW, Bhattacharyya N. Clinical behavior of follicular variant of papillary thyroid carcinoma: presentation and survival. Laryngoscope. 2010;120:712–716. doi: 10.1002/lary.20828. [DOI] [PubMed] [Google Scholar]
- 9.Kim DS, Kim JH, Na DG, Park SH, Kim E, Chang KH, et al. Sonographic features of follicular variant papillary thyroid carcinomas in comparison with conventional papillary thyroid carcinomas. J Ultrasound Med. 2009;28:1685–1692. doi: 10.7863/jum.2009.28.12.1685. [DOI] [PubMed] [Google Scholar]
- 10.Rhee SJ, Hahn SY, Ko ES, Ryu JW, Ko EY, Shin JH. Follicular variant of papillary thyroid carcinoma: distinct biologic behavior based on ultrasonographic features. Thyroid. 2014;24:683–688. doi: 10.1089/thy.2013.0351. [DOI] [PubMed] [Google Scholar]
- 11.Zidan J, Karen D, Stein M, Rosenblatt E, Basher W, Kuten A. Pure versus follicular variant of papillary thyroid carcinoma: clinical features, prognostic factors, treatment, and survival. Cancer. 2003;97:1181–1185. doi: 10.1002/cncr.11175. [DOI] [PubMed] [Google Scholar]
- 12.Rivera M, Ricarte-Filho J, Knauf J, Shaha A, Tuttle M, Fagin JA, et al. Molecular genotyping of papillary thyroid carcinoma follicular variant according to its histological subtypes (encapsulated vs infiltrative) reveals distinct BRAF and RAS mutation patterns. Mod Pathol. 2010;23:1191–1200. doi: 10.1038/modpathol.2010.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Proietti A, Giannini R, Ugolini C, Miccoli M, Fontanini G, Di Coscio G, et al. BRAF status of follicular variant of papillary thyroid carcinoma and its relationship to its clinical and cytological features. Thyroid. 2010;20:1263–1270. doi: 10.1089/thy.2009.0283. [DOI] [PubMed] [Google Scholar]
- 14.Kim EK, Park CS, Chung WY, Oh KK, Kim DI, Lee JT, et al. New sonographic criteria for recommending fine-needle aspiration biopsy of nonpalpable solid nodules of the thyroid. AJR Am J Roentgenol. 2002;178:687–691. doi: 10.2214/ajr.178.3.1780687. [DOI] [PubMed] [Google Scholar]
- 15.Cibas ES, Ali SZ. The Bethesda System For Reporting Thyroid Cytopathology. Am J Clin Pathol. 2009;132:658–665. doi: 10.1309/AJCPPHLWMI3JV4LA. [DOI] [PubMed] [Google Scholar]
- 16.Kwak JY, Han KH, Yoon JH, Kim EK, Moon HJ, Kim YL, et al. BRAFV600E mutation testing in fine needle aspirates of thyroid nodules: potential value of real-time PCR. Ann Clin Lab Sci. 2012;42:258–265. [PubMed] [Google Scholar]
- 17.Jogai S, Adesina AO, Temmim L, Al-Jassar A, Amir T, Amanguno HG. Follicular variant of papillary thyroid carcinoma: a cytological study. Cytopathology. 2004;15:212–216. doi: 10.1111/j.1365-2303.2004.00168.x. [DOI] [PubMed] [Google Scholar]
- 18.Manivannan P, Siddaraju N, Gopalakrishnan S. A systematic approach to assess the strengths and limitations of cytomorphology in the diagnosis of the follicular variant of papillary thyroid carcinoma. Cytopathology. 2014;25:190–198. doi: 10.1111/cyt.12124. [DOI] [PubMed] [Google Scholar]
- 19.Hahn SY, Shin JH, Han BK, Ko EY, Ko ES. Ultrasonography-guided core needle biopsy for the thyroid nodule: does the procedure hold any benefit for the diagnosis when fine-needle aspiration cytology analysis shows inconclusive results? Br J Radiol. 2013;86:20130007. doi: 10.1259/bjr.20130007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lee SR, Jung CK, Kim TE, Bae JS, Jung SL, Choi YJ, et al. Molecular genotyping of follicular variant of papillary thyroid carcinoma correlates with diagnostic category of fine-needle aspiration cytology: values of RAS mutation testing. Thyroid. 2013;23:1416–1422. doi: 10.1089/thy.2012.0640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Park JY, Kim WY, Hwang TS, Lee SS, Kim H, Han HS, et al. BRAF and RAS mutations in follicular variants of papillary thyroid carcinoma. Endocr Pathol. 2013;24:69–76. doi: 10.1007/s12022-013-9244-0. [DOI] [PubMed] [Google Scholar]


