Abstract
BACKGROUND:
Universal newborn hearing screening was implemented worldwide largely on modeled, not measured, long-term benefits. Comparative quantification of population benefits would justify its high cost.
METHODS:
Natural experiment comparing 3 population approaches to detecting bilateral congenital hearing loss (>25 dB, better ear) in Australian states with similar demographics and services: (1) universal newborn hearing screening, New South Wales 2003–2005, n = 69; (2) Risk factor screening (neonatal intensive care screening + universal risk factor referral), Victoria 2003–2005, n = 65; and (3) largely opportunistic detection, Victoria 1991–1993, n = 86. Children in (1) and (2) were followed at age 5 to 6 years and in (3) at 7 to 8 years. Outcomes were compared between states using adjusted linear regression.
RESULTS:
Children were diagnosed younger with universal than risk factor screening (adjusted mean difference –8.0 months, 95% confidence interval –12.3 to –3.7). For children without intellectual disability, moving from opportunistic to risk factor to universal screening incrementally improved age of diagnosis (22.5 vs 16.2 vs 8.1 months, P < .001), receptive (81.8 vs 83.0 vs 88.9, P = .05) and expressive (74.9 vs 80.7 vs 89.3, P < .001) language and receptive vocabulary (79.4 vs 83.8 vs 91.5, P < .001); these nonetheless remained well short of cognition (mean 103.4, SD 15.2). Behavior and health-related quality of life were unaffected.
CONCLUSIONS:
With new randomized trials unlikely, this may represent the most definitive population-based evidence supporting universal newborn hearing screening. Although outperforming risk factor screening, school entry language still lagged cognitive abilities by nearly a SD. Prompt intervention and efficacy research are needed for children to reach their potential.
What’s Known on This Subject:
Universal newborn hearing screening was implemented internationally to improve language and other outcomes of congenital hearing loss. However, our recent systematic review concluded that current evidence was insufficient to determine its long-term population effectiveness or value-for-money.
What This Study Adds:
Population language outcomes by the early school years benefited incrementally on moving from opportunistic detection to systematic risk factor screening to universal newborn hearing screening. However, with outcomes still well below population norms, much more remains to be done.
Few population screening programs are implemented or evaluated in light of adequate epidemiologic evidence. This would include known population costs versus benefits from exemplary randomized trials measuring long-term outcomes of screening, backed up by documented epidemiology of the condition’s natural history in whole-population cohorts assembled after, as well as before, the screening programs commenced.
This is as true for congenital hearing loss as for most programs. Hearing loss can have devastating effects on children’s lives1 and incur large societal costs.2 Therefore, despite methodologic flaws,1 studies in the 1990s linking earlier diagnosis and management to better preschool language3,4 drove large-scale implementation of new screening strategies (universal risk factor identification and universal newborn hearing screening [UNHS]) aiming to achieve much earlier detection and intervention than the existing opportunistic strategies, such as referral when a parent voiced concern about the child’s hearing or language.
Risk-factor screening involves systematically identifying and referring for audiologic testing all infants with risk factors for hearing loss,5 coupled with predischarge hearing screening of infants admitted to the NICU.5 Reports of the NICU-only6 and risk-factor-only7 components are available, but no published studies have reported outcomes of population-based risk-factor programs including both elements, presumably because they were never widely implemented.
In contrast, UNHS programs offer every newborn an objective hearing screen. This clearly leads to earlier amplification and intervention.8 Cost-effectiveness was supported by the only economic evaluation to consider long-term costs and outcomes of UNHS versus risk-factor screening but, in the absence of trials, this was based solely on projected improvements.2 Two quasi-randomized trials of UNHS have since been published. In the Wessex Study of 7- to 8-year-old English children offered UNHS in 1993–1996, neither expressive language nor speech clearly benefited; mean receptive language improved (effect size 0.56, 95% confidence interval [CI] 0.03–1.08) but remained nearly 2 SD below population norms.9 In the DECIBEL study, parents did not report better language outcomes in Dutch children born 2003 through 2005 in UNHS versus non-UNHS regions (at mean ages of 48 and 61 months, respectively).10 However, the Wessex study was not truly population based and was implemented before modernization of English audiologic services, whereas DECIBEL did not directly measure language outcomes.
Our recent systematic review of economic evaluations therefore questioned the evidence that UNHS represents a value-for-money proposition.11 However, gold standard randomized trials are now virtually impossible. We report an unusual whole-of-population natural experiment in which 1 Australian state offered a fully operational statewide UNHS program, while another offered the only full-fledged population risk factor program we know of.
We aimed to
compare population outcomes at age 5–6 years of UNHS versus risk factor screening,
explore program benefits by hearing loss severity, and
Compare UNHS and risk-factor screening outcomes with prospectively collected 7- to 8-year-old population outcomes in a comparable cohort exposed to opportunistic detection a decade earlier.
Methods
Study Design and Populations
The Statewide Comparison of Outcomes (SCOUT) study was a quasi-randomized trial targeting children born March 2003–February 2005 in 2 Australian states, New South Wales (offering UNHS) and Victoria (offering risk-factor screening). We also compared both groups with a 7- to 8-year-old population-based Victorian cohort, born January 1991 through July 1993 (when detection was largely opportunistic), from this research team’s earlier Children with Hearing Impairment in Victoria Outcome Study (CHIVOS).12,13
New South Wales and Victoria were otherwise similar in 2003–2005 on a range of potentially confounding factors, including socioeconomic advantage,14 ethnic composition,15 age of commencing school, and reading, writing, and numeracy performance across grades 3, 5, and 7.16 Both states offered government-funded diagnostic audiology. Access to specialist audiologic management was via Australian Hearing, a government-funded organization that fits and maintains hearing aids for all Australian children who need them at no cost to families. Its standardized pediatric protocols ensured national uniformity in assessment and amplification from the time of diagnosis, and its national database records every child who has a hearing aid fitted, as well as hearing acuity (via annual audiograms) and treatment during the preschool years. Both states offered the full range of early intervention services, cochlear implants at no cost to families, and health care via Australia’s universal Medicare system.
Children were eligible for the SCOUT study if they had bilateral hearing loss believed to be congenital of >25 dB pure tone average (decibels hearing level [dB HL]) in the better ear and were fitted with hearing aids and/or cochlear implants by 4 years. We excluded children whose (1) parents had insufficient English to participate, as judged by the treating Australian Hearing audiologist or by the researcher at the initial contact call; (2) hearing loss was unilateral, acquired (as judged by Australian Hearing records) and/or conductive, or currently in the normal range; and/or (3) recruitment was considered inappropriate (eg, living overseas, severe known social/disability barriers). The earlier CHIVOS cohort additionally excluded children with known intellectual disability.
Detection Programs
Universal Newborn Hearing Screening, 2003–2005
Since December 2002, New South Wales has offered automated auditory brainstem response screening to its ∼84 500 newborns annually; infants failing 2 successive screens are referred for diagnostic audiology, and infants with hearing risk factors are referred for diagnostic audiology at age 8 to 12 months. Since May 2003, it has consistently achieved >95% population coverage and >95% follow-up for referred infants.17
Risk-Factor Screening, 2003–2005
From February 2003 to February 2005, all newborns admitted to Victoria’s 4 NICUs and associated special care nurseries were systematically offered predischarge automated auditory brainstem response hearing screening (71.1% capture attained over this period18) followed by diagnostic audiology referral. All other newborns were offered a risk factor ascertainment and behavioral hearing screening program (see next paragraph).
Opportunistic Detection, 1991–1993
Detection in Victoria was wholly opportunistic until December 1992. Thereafter, a 2-stage program was implemented whereby maternal and child health nurses administered (1) a hearing risk factor questionnaire at 2 weeks and 8 months (attended by >97% and 83% of Victorian infants respectively), and (2) a standardized, modified Ewing Distraction hearing screen19 at 8 to 10 months. Infants with risk factors and/or who failed 2 successive distraction tests were referred to diagnostic audiology.20 We included as “opportunistic” children born spanning (ie, both before and after) this program’s implementation because previous evaluation showed little impact on detection.7
Sampling
The Australian Hearing database provided a uniform sampling frame for all 3 groups. Between July 2008 and November 2009, researchers at the National Acoustic Laboratories (Australian Hearing’s research division) enumerated the UNHS and risk factor birth cohorts. A 2-stage opt in process was then required. First, the local Australian Hearing audiologist caring for a potentially eligible child had to agree to an approach being made on their behalf to the family. Then the National Acoustic Laboratories sent a brief approach letter on Australian Hearing letterhead on the audiologist’s behalf, inviting parents to contact the researchers directly to express interest. CHIVOS followed similar procedures a decade earlier, via the Victorian Infant Hearing Screening Program and Australian Hearing.
Procedures
When a family expressed interest, the project officer phoned and then mailed an information statement, consent form, and questionnaire, and a speech pathology or psychology researcher conducted a 2-hour home visit. Children used their usual amplification; all children using amplification are offered an annual appointment with Australian Hearing at which hearing aid gain is verified and adjustments made as clinically indicated. As far as possible, researchers were blind to hearing loss severity and whether screening had actually occurred, but the state-based design precluded blinding to program.
Of the SCOUT children, 37% (n = 50) were already participating in the Longitudinal Outcomes of Children with Hearing Impairment (LOCHI) study. We prospectively harmonized the studies and preferentially accessed any LOCHI results to avoid duplication. Parents also consented to our accessing any direct assessment measures administered elsewhere in the preceding 6 months (eg by speech pathologists).
The studies were approved by the Ethics Committees of Melbourne’s Royal Children’s Hospital (SCOUT, CHIVOS) and National Acoustic Laboratories (SCOUT, LOCHI). Parents provided written informed consent.
Measures
Main Outcomes
See Table 1 for details. At age 5 to 6 years, SCOUT children were directly assessed on standardized measures administered in English of language, receptive vocabulary, letter knowledge, and nonverbal IQ. The parent reported their child’s behavior and their own and their child’s health-related quality of life. CHIVOS similarly assessed language, receptive vocabulary, and non-verbal IQ at age 7 to 8 years. Children unable to be directly assessed because of documented, parent-reported, or apparent intellectual disability were assigned the basal (or lowest possible) outcome scores as follows: receptive/expressive language (basal score = 50; n = 12, 9.0%), receptive vocabulary (basal score = 20; n = 21, 15.7%), letter knowledge (basal score = 0; n = 17, 12.7%), and nonverbal IQ (basal score = 30; n = 19, 14.2%). Using dates of birth, we calculated ages at diagnosis and hearing aid fitting from the dates of first Australian Hearing and first fitting appointments respectively.
TABLE 1.
Key measures for the SCOUT cohorts 2003-2005
| Construct | Source and Measure | Additional Information |
|---|---|---|
| Age of diagnosis | Australian Hearing Database | Age at first Australian Hearing appointment. |
| Receptive and expressive language | Direct assessment: Preschool Language Scale—4, Australian Language Adaptation21 | Auditory Comprehension (receptive language) and Expressive Communication (expressive language) scales. Standardized: mean 100, SD 15, range 50–140.a |
| Receptive vocabulary | Direct assessment: The Peabody Picture Vocabulary Tes—422 | 228 items. Standardized: mean 100, SD 15, range 20–160.b |
| Letter knowledge | Direct assessment: Phonological Abilities Test23 | Letter Knowledge subtest measures letter name/sound recall. Raw score range 0–26. |
| Health-related quality of life | Parent report: Health Utility Index 3 parent self-report (HUI23S4En.15Q); parent-proxy child report (HUI23P4En.15Q)24 | 15 item multiattribute health status and utility system including hearing and speech dimensions. Two overall health-related quality of life scores. Range: 0.00 = conventional dead to 1.00 = perfect health. |
| Parent report: Pediatric Quality of Life Inventory 4.025 | 23 items. The total score is the sum of physical, social, emotional, and school-functioning scores. Range: 0 = worst health to 100 = best possible health. | |
| Behavior and emotion | Parent report: Strengths and Difficulties Questionnaire: Australian version for 4–10 y olds26 | 25 items. Conduct problems, emotional symptoms, hyperactivity, peer relationships, prosocial behavior scales. Total Difficulties score is the sum of all difficulties scales but not the prosocial scale; range: 0–40. |
| Nonverbal cognition (IQ) | Direct assessment: Wechsler Nonverbal Scale of Ability27 | Two-subtest version (Matrices and Recognition). Children taking part in LOCHI study who had been administered the 4-subtest version had their results rescored. Standardized: mean 100, SD 15, range 30–170. |
Because the 1991–1993 opportunistic cohort were slightly older, their language was measured using the Clinical Evaluation of Language Fundamentals. Like the Preschool Language Scale—4, this yields normative means of 100 [SD 15] for receptive and expressive language.
The 1991-3 opportunistic cohort were assessed by using the Peabody Picture Vocabulary Test—3.
Potential a priori confounders were nonverbal IQ and hearing loss severity, defined as the most recent 3-frequency pure-tone average dB HL (for most children within the year preceding the assessment) across 0.5, 1, and 2 kHz in the better ear. Demographic measures included child gender, parent tertiary level education, household income category, English as the child’s second language, and the census-based Disadvantage Index for home postcode (a continuous measure; higher scores reflect greater advantage).28
Statistical Analysis
Outcomes of UNHS Versus Risk-Factor Programs (Aim 1)
Mean differences were estimated using linear regression. Potential confounders were included in a stepwise approach: first sociodemographic factors, then nonverbal IQ, and finally current hearing acuity; age was also included for letter knowledge. Analyses were conducted including all children, then for children without intellectual disability, defined as a nonverbal IQ that was either <70 or could not be assessed for the reasons noted earlier. This restricted the sample by 29 (21.6%): 20 (29.0%) in the UNHS group and 9 (16.1%) in the risk factor group.
Outcomes by hearing loss severity (Aim 2):
Using fractional polynomials, we examined graphically whether benefits of UNHS versus risk factor screening may differ by hearing loss severity among children without intellectual disability.
Comparison Among UNHS, Risk Factor, and Opportunistic Detection (Aim 3)
Mean between-program differences were estimated using linear regression, adjusting for confounders as in aim 1 and excluding children with intellectual disability to enable comparability with the opportunistic cohort. Tests for trends in outcomes, moving from opportunistic to risk factor to UNHS, were conducted with these 3 categories as a linear predictor term in the regression models.
Other Considerations
Stata version 12 was used throughout. To account for missing potential confounders, analyses were conducted with and without multiple imputation using the multivariate normal regression model and assuming data were missing at random. Results were similar, so the latter are reported.
Results
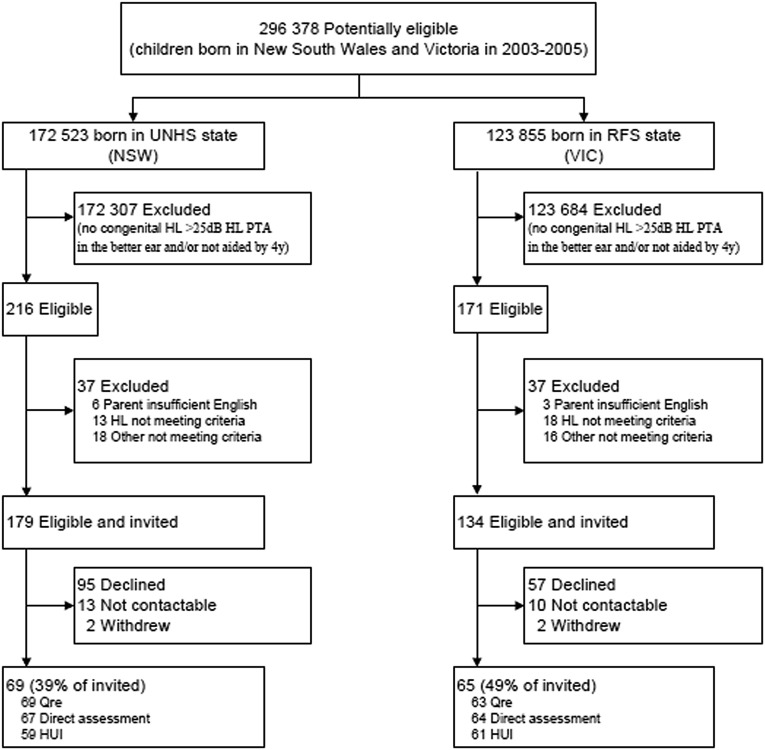
Figure 1 summarizes participant recruitment. Between March 2003 and February 2005, 172 523 babies were born in New South Wales and 123 855 in Victoria. The National Acoustic Laboratories considered 313 to be eligible, comprising 179 in New South Wales (0.10% of the total births) and 134 in Victoria (0.11%). The 179 nonparticipants had similar initial (59 vs 63 dB HL, P = .30) and current (60 vs 65 dB HL, P = .24) hearing as the 134 participants (69 New South Wales, 65 Victoria; 43% response).
FIGURE 1.
Study recruitment. HL, hearing loss; HUI, Health Utilities Index; NSW, New South Wales; PTA, pure tone average; Qre, questionnaire; RFS, risk-factor screening; VIC, Victoria. [medium]
Children in the 2 states (Table 2) had, on average, similar initial and current hearing loss and nonverbal IQ. Since New South Wales parents reported slightly lower educational levels and less advantaged neighborhoods, these factors became potential confounders. Table 2 also summarizes characteristics of the opportunistic sample, which have been reported in detail elsewhere.12,13
TABLE 2.
Sample Characteristics
| UNHS (NSW 2003–2005) (n = 69a) | Risk Factor (VIC 2003–2005) (n = 65a) | Opportunistic (VIC 1991–1993) (n = 86b) | |||
|---|---|---|---|---|---|
| Child | |||||
| Age at assessment, mo, mean (SD) | 65.3 (6.6) | 62.8 (4.9) | 95.3 (5.3) | ||
| Male gender, % | 57 | 55 | 62 | ||
| Initial severity of hearing impairment, mean (SD) | 61.0 (27.1) | 64.6 (31.6) | 64.5 (24.8) | ||
| Current hearing impairment severity, mean (SD) | 65.2 (30.2) | 63.8 (32.4) | 57.3 (21.8) | ||
| Nonverbal IQ,c mean (SD) | |||||
| Whole sample | 89.7 (31.9) | 93.0 (28.0) | 104.6 (16.7) | ||
| Children without intellectual disability | 103.4 (15.2) | 102.0 (15.5) | 104.6 (16.7) | ||
| Family | |||||
| Language other than English household, % | 8 | 10 | 11 | ||
| Disadvantage Index, mean (SD) | 1004.4 (71.4) | 1019.5 (60.7) | 1019.4 (68.1) | ||
| Income category, % | |||||
| <$33 800 per year | 20 | 16 | — | ||
| $33 800–$51 999 per year | 25 | 27 | — | ||
| $52 000–$103 999 per year | 29 | 29 | — | ||
| ≥$104 000 per year | 26 | 28 | — | ||
| Parent with tertiary-level education, % | 31 | 44 | 28 | ||
NSW, New South Wales; VIC, Victoria; --, data not available.
Percent missing data (UNHS and risk factor combined sample): initial severity 3.0%, current severity 3.0%, nonverbal IQ 6.0%, languages other than English (LOTE) 3.7%, income 9.7%, educational level 6.0%, all other measures complete.
Percent missing data (opportunistic): age at assessment 1.2%, initial severity 4.7%, current severity 15.1%, nonverbal IQ 1.2%, disadvantage index 1.2%, all other measures complete.
Nonverbal IQ including unassessable children for whom the basal standard score (30) was imputed.
Outcomes of UNHS Versus Risk-Factor Programs (Aim 1)
Having taken account of sociodemographic differences, hearing loss was estimated to be diagnosed on average 8.0 months earlier (95% CI –12.3 to –3.7; Model 1) in the UNHS state (Table 3). There was some evidence of these children having better expressive language, receptive vocabulary, and letter knowledge. Although these effects strengthened with adjustment for nonverbal IQ and 3-frequency average hearing loss, statistically significant differences did not emerge. This appeared to reflect the greater number of low-functioning children in the UNHS state. Their strong clustering of scores around the lowest possible “basal” values lowered their mean scores and prevented discernment of any possible UNHS benefit.
TABLE 3.
Outcomes of UNHS Versus Risk-Factor Screening
| Outcome | Normative | Demographically Adjusted Meana | Fully Adjusted Meanb | ||||
|---|---|---|---|---|---|---|---|
| Mean (SD) | UNHS | Risk Factor | Mean Diff (95% CI) | P | Mean Diff (95% CI) | P | |
| All childrenc | |||||||
| Age diagnosed, mo | — | 7.9 | 15.9 | −8.0 (–12.3 to –3.7) | <.001 | −6.8 (–11.1 to –2.6) | .002 |
| Language | |||||||
| Receptive language | 100 (15)21 | 80.9 | 78.0 | 2.9 (–5.2 to 11.0) | .48 | 3.4 (–2.5, 9.3) | .26 |
| Expressive language | 100 (15)21 | 81.1 | 76.0 | 5.1 (–3.3 to 13.5) | .23 | 5.3 (–1.1 to 11.7) | .11 |
| Receptive vocabulary | 100 (15)22 | 79.7 | 73.9 | 5.8 (–4.9 to 16.6) | .29 | 6.2 (–0.3 to 12.7) | .06 |
| Letter knowledge | — | 12.2 | 9.0 | 3.3 (–0.3 to 6.9) | .08 | 2.7 (–0.4 to 5.9) | .09 |
| Behavior problems | 6.9 (5.1)29 | 9.6 | 9.6 | 0.0 (–2.3 to 2.4) | .97 | −0.1 (–2.6 to 2.5) | .94 |
| Health-related quality of life | |||||||
| PedsQL | 81.9 (12.6)25 | 76.7 | 75.5 | 1.2 (–5.1 to 7.6) | .70 | 2.3 (–4.3 to 9.0) | .49 |
| HUI (child) | 0.90 (0.13)30 | 0.72 | 0.66 | 0.06 (–0.04 to 0.16) | .20 | 0.04 (–0.04 to 0.13) | .33 |
| HUI (parent) | 0.86 (0.31)31 | 0.87 | 0.87 | 0.00 (–0.06 to 0.07) | .96 | 0.01 (–0.06 to 0.07) | .88 |
| Without intellectual disabilityc | |||||||
| Age diagnosed, months | — | 7.0 | 16.3 | −9.3 (–14.3 to –4.3) | <.001 | −8.4 (–13.1 to –3.7) | .001 |
| Language | |||||||
| Receptive language | 100 (15)21 | 87.5 | 82.7 | 4.8 (–3.2 to 12.9) | .24 | 5.2 (–1.9 to 12.3) | .15 |
| Expressive language | 100 (15)21 | 87.8 | 80.0 | 7.7 (–0.9 to 16.4) | .08 | 8.2 (0.5 to 15.9) | .04 |
| Receptive vocabulary | 100 (15)22 | 90.8 | 82.9 | 7.9 (0.0 to 15.7) | .05 | 8.1 (0.8 to 15.4) | .03 |
| Letter knowledge | — | 13.2 | 10.9 | 2.3 (–1.6 to 6.2) | .25 | 1.8 (–1.9 to 5.5) | .34 |
| Behavior problems | 6.9 (5.1)29 | 9.9 | 9.2 | 0.7 (–1.9 to 3.3) | .61 | 0.8 (–1.9 to 3.5) | .56 |
| Health-related quality of life | |||||||
| PedsQL | 81.9 (12.6)25 | 79.0 | 76.2 | 2.9 (–3.9 to 9.6) | .40 | 2.7 (–4.3 to 9.7) | .45 |
| HUI (child) | 0.90 (0.13)30 | 0.76 | 0.72 | 0.04 (–0.04 to 0.12) | .33 | 0.04 (–0.04 to 0.12) | .29 |
| HUI (parent) | 0.86 (0.31)31 | 0.87 | 0.87 | 0.00 (–0.07 to 0.07) | 1.00 | 0.00 (–0.08 to 0.07) | .91 |
HUI, Health Utilities Index; PedsQL, Pediatric Quality of Life Inventory; --, (Letter Knowledge): no standardized normative mean; --, (age of diagnosis): N/A.
Adjusted for parent education, English as a second language, disadvantage index, gender (and age, for letter knowledge).
With additional adjustment for nonverbal IQ and current hearing loss.
Sample n varied between 101 and 120 for demographically adjusted model, and 93 to 111 for fully adjusted model. When children with intellectual disability were excluded, the sample n varied from 87 to 96 for the demographically adjusted model and 85 to 93 for the fully adjusted model.
In children without intellectual disability, clear benefits were associated with UNHS for expressive language (fully adjusted mean difference 8.2 points, 95% CI 0.5 to 15.9) and receptive vocabulary (8.1, 95% CI 0.8 to 15.4), with some evidence of better receptive language (5.2, 95% CI –1.9 to 12.3). Letter knowledge, behavior, and parent and child health-related quality of life were similar between states.
Outcomes by Hearing Loss Severity (Aim 2)
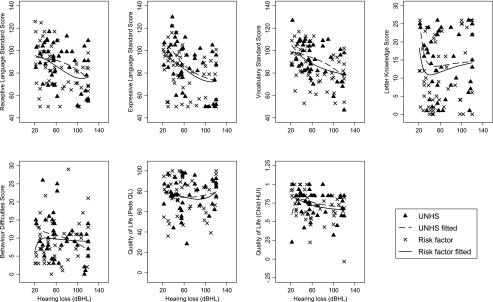
Among children without intellectual disability, Fig 2 illustrates that language and vocabulary scores fell with increasing severity of hearing loss, and letter knowledge scores were better in those with mild than more severe losses (Fig 2). Benefits of UNHS appeared maximal in the mild-moderate range for letter knowledge, severe range for receptive vocabulary and profound range for receptive language; benefits to expressive language appeared unrelated to severity. Child behavior difficulties and health-related quality of life were largely independent of both severity and screening program.
FIGURE 2.
Population outcomes by hearing acuity in children without intellectual disability for UNHS and risk-factor screening. [large]
Comparison Among UNHS, Risk Factor, and Opportunistic Detection (Aim 3)
Outcomes on moving from opportunistic, to risk factor, to UNHS detection showed significant stepwise gains in children without intellectual disability (Table 4). Having adjusted for sociodemographic factors, nonverbal IQ and level of hearing loss, the mean age of diagnosis was estimated as 14.4 months earlier in the UNHS than the opportunistic cohort (95% CI –19.3 to –9.6) and, despite delays, fitting still occurred 10.5 months earlier (both P for trend <.001). Adjusted mean receptive language, expressive language, and receptive vocabulary scores were 7.0 points (95% CI 0.2 to 13.8), 14.4 points (95% CI 7.3 to 21.5), and 12.1 points (95% CI 5.9 to 18.4) higher in the UNHS than the opportunistic cohort, respectively.
TABLE 4.
Population Outcomes of UNHS Versus Risk Factor Versus Opportunistic Screening in Children Without Intellectual Disability
| Outcome | Fully Adjusteda Mean for Each Program | Fully Adjusted Mean Difference (95% CI)a | P (Trend) | ||||
|---|---|---|---|---|---|---|---|
| Opportunisticb | Risk Factorb | UNHSb | Opportunisticb | Risk Factorb | UNHSb | ||
| Milestones to early intervention (mo) | |||||||
| Age diagnosed | 22.5 | 16.2 | 8.1 | Ref | −6.4 (–11.0 to –1.8) | −14.4 (–19.3 to –9.6) | <.001 |
| Age hearing aid fitted | 24.0 | 17.9 | 13.5 | Ref | −6.1 (–11.0 to –1.1) | −10.5 (–15.7 to –5.3) | <.001 |
| Time between diagnosis and fitting | 1.4 | 1.7 | 5.4 | Ref | 0.3 (–1.7 to 2.3) | 4.0 (1.8–6.1) | .001 |
| Language | |||||||
| Receptive language | 81.8 | 83.0 | 88.9 | Ref | 1.1 (–5.4 to 7.7) | 7.0 (0.2–13.8) | .05 |
| Expressive language | 74.9 | 80.7 | 89.3 | Ref | 5.8 (–1.0 to 12.6) | 14.4 (7.3–21.5) | <.001 |
| Receptive vocabulary | 79.4 | 83.8 | 91.5 | Ref | 4.5 (–1.4 to 10.4) | 12.1 (5.9–18.4 | <.001 |
Adjusted for parent education, English as a second language, disadvantage index, gender, nonverbal IQ, current hearing loss (and age, for letter knowledge).
Sample n varied between 69 and 71 for opportunistic, 51 and 52 for risk factor, and 41 and 42 for UNHS.
Nonetheless, language, receptive vocabulary, behavior, and child health-related quality of life scores remained well below population normative mean values after introduction of UNHS (Table 3, all Ps ≤ .001). Parent health-related quality of life was similar to normative mean values regardless of screening program.
Discussion
Principal Findings
This is the first population study to contrast directly assessed, comparable, long-term outcomes of 3 approaches to detecting bilateral congenital hearing loss. There was strong evidence of incremental benefits to age of diagnosis, receptive and expressive language, and receptive vocabulary on moving from opportunistic to risk factor to universal hearing screening in children without intellectual disability (the large majority). Improvements occurred across the severity spectrum, implicating earlier access to useful hearing via both hearing aids and cochlear implantation.
Nonetheless, language and vocabulary remained well below population means and the children’s cognitive potential, and there was little benefit to behavior or health-related quality of life. Reflecting the relative newness of the UNHS program, ages of diagnosis and hearing aid fitting still fell well short of the goal of “appropriate intervention at no later than 6 months of age” even in the UNHS state.32
Strengths of the Study
Of relevance to public health,33 we demonstrated mean population improvements regardless of actual receipt of screen and saw no evidence of harm to parent quality of life or child psychosocial well-being. Because the 2003–2005 cohorts shared postdiagnostic protocols, amplification, and early intervention opportunities, we are confident in attributing these benefits to the UNHS program itself. We measured outcomes and potential confounders using directly assessed, reliable measures with normative standard scores for outcomes and potential confounders.
Limitations
Although clinically important in size, the study was underpowered to confirm the gains observed with universal versus risk-factor screening, reflecting the 43% uptake from the unavoidable 2-stage opt-in process. Our measures were insufficiently fine-grained for low-functioning children. Imputing basal scores for children with intellectual disabilities allowed us to retain this important group but precluded detecting improvements they may have experienced and skewed whole-cohort comparisons toward null values.
Because characteristics were similar in participants and nonparticipants in both states, differential selection or response bias seem unlikely. However, our results may not generalize to families experiencing stressors or poor English skills that precluded recruitment. Nor was the study powered for subgroup analysis (eg, cochlear implantation). Lack of blinding to program should have been partially offset by the standardized assessments.
Finally, the 1991–1993 cohort excluded children with intellectual disability and, because they were slightly older, their language was measured using the Clinical Evaluation of Language Fundamentals 3. Like the Preschool Language Scale 4, this yields normative means of 100 (SD 15), and our long-term multiwave follow-up to 17 to 19 years indicates high stability of language scores (unpublished data), indicating it should be a good proxy for the 5- to 6-year-old measures.
Interpretation in Light of Other Studies
These findings broadly agree with, but go beyond, other recent reports.9,10 The steady improvement in language-related outcomes with each screening advance makes sense, unlike the DECIBEL study in which benefits to gross motor and social, but not language, skills were difficult to explain.10 Our results also preserve both the expected gradients in language-related outcomes by hearing loss severity and congruence across receptive language, expressive language, and receptive vocabulary. These results are dissimilar to the Wessex study, which reported a major and surprising discordance in children exposed to UNHS of nearly 1.2 SD between their surprisingly low receptive language (–1.89 SD below population norms in the UNHS group) and much better expressive language scores (–0.74 SD below population norms).9
Meaning of the Study
With new randomized trials now unlikely, this study may represent the most definitive population-based evidence that UNHS leads to meaningful improvements in language and related outcomes. However, it is just 1 important incremental step, buying, on average, a third to a half of an SD in language scores by school entry over comprehensive risk factor screening. We draw parallels with other public health issues for which the spectacular gains of the past half century, such as smoking, road deaths, childhood cancer, cystic fibrosis, have occurred in modest, steady steps, never resting on a single innovation.
Conclusions
This study has important forward implications. First, intervention should follow detection much faster than was possible in New South Wales in 2003–2005. In Australia, children are now routinely fitted with hearing aids in the first 6 months of life,34 but many jurisdictions lag behind. Second, research should focus on the science of intervention, amplification, and hearing restoration. Population-based randomized trials, which are sorely lacking in this field, could optimize postdiagnostic interventions for children with hearing impairment, with directly assessed language and other measures as outcomes. Third, long-term follow-up of the existing cohorts could confirm the societal benefits of UNHS, especially if combined into an adequately powered prospective meta-analysis. Finally, new population-based birth cohorts must be recruited to document secular improvements and guard against complacency; this study’s 3 cohorts could provide a benchmark against which to measure these future gains.
Acknowledgments
We thank all children, parents, and researchers involved in the SCOUT study. We also acknowledge the assistance of the LOCHI study and the early intervention agencies and cochlear implant clinics in the collection of outcomes data.
Glossary
- CHIVOS
Children with Hearing Impairment in Victoria Outcome Study
- CI
confidence interval
- dB HL
decibels hearing level
- LOCHI
Longitudinal Outcomes of Children with Hearing Impairment
- SCOUT
Statewide Comparison of Outcomes
- UNHS
Universal Newborn Hearing Screening
Footnotes
Dr Wake conceptualized and designed the study; she takes overall responsibility for all aspects of the study and contributed to and critically reviewed the final manuscript; Dr Ching assisted with the design the study, provided guidance regarding the data collection instruments, facilitated the recruitment of participants, and reviewed and revised the manuscript; Dr Wirth was responsible for acquisition of the data, provided guidance regarding the data collection instruments, carried out the data analysis, and drafted and revised the manuscript; Drs Poulakis and Rickards conceptualized and designed the study, provided guidance regarding the data collection instruments, and reviewed and revised the manuscript; Dr Mensah was responsible for the analysis and interpretation of the data and reviewed and revised the manuscript; Dr Gold conceptualized and designed the study, provided guidance regarding economic evaluation, and reviewed and revised the manuscript; Ms King assisted with the design the study, facilitated the recruitment of participants, and reviewed and revised the manuscript; Ms Bryson contributed to the data analysis and drafted and revised the manuscript; Dr Reilly conceptualized and designed the study, provided guidance regarding the data collection instruments, and reviewed and revised the manuscript; and all authors approved the final manuscript as submitted.
FINANCIAL DISCLOSURE: The authors have indicated they have no financial relationships relevant to this article to disclose.
FUNDING: This study was funded by Australian National Health and Medical Research Council (NHMRC) project grant 491228. Professor Wake was supported by NHMRC Population Health Career Development grant 546405 and Senior Research Fellowship 1046518; Dr Mensah by NHMRC Population Health Capacity Building grant 436914 and Early Career Fellowship 1037449; Dr Lisa Gold by NHMRC Population Health Capacity Building grant 425855 and NHMRC Early Career Fellowship 1035100; and Professor Reilly by NHMRC Practitioner Fellowship 491210. Research at the National Acoustic Laboratories is supported by the Australian Government through the Office of Hearing Services and the HEARing Co-operative Research Centre. Part of the work included in this study was supported by National Institutes of Health funding (R01DC008080) awarded to Dr Ching. Research at the Murdoch Childrens Research Institute is supported by the Victorian Government’s Operational Infrastructure Support Program. The funding organizations are independent of all researchers and had no role in the design and conduct of the study; in the collection, analysis, and interpretation of the data; or in the decision to submit the article for publication or in the preparation, review, or approval of the manuscript.
POTENTIAL CONFLICT OF INTEREST: The authors have indicated they have no potential conflicts of interest to disclose.
References
- 1.Helfand M, Thompson DC, Davis R. Newborn Hearing Screening (Systematic Evidence Review Number 5; Contract 290-97-0018 to the Oregon Health & Science University Evidence-based Practice Center, Portland, Oregon). AHRQ Publication No. 02-S001. Rockville, MD: Agency for Healthcare Research and Quality; 2001 [PubMed] [Google Scholar]
- 2.Keren R, Helfand M, Homer C, McPhillips H, Lieu TA. Projected cost-effectiveness of statewide universal newborn hearing screening. Pediatrics. 2002;110(5):855–864 [DOI] [PubMed] [Google Scholar]
- 3.Yoshinaga-Itano C, Sedey AL, Coulter DK, Mehl AL. Language of early- and later-identified children with hearing loss. Pediatrics. 1998;102(5):1161–1171 [DOI] [PubMed] [Google Scholar]
- 4.Moeller MP. Early intervention and language development in children who are deaf and hard of hearing. Pediatrics. 2000;106(3):E43 [DOI] [PubMed] [Google Scholar]
- 5.Joint Committee on Infant Hearing . Year 2000 position statement: principles and guidelines for early hearing detection and intervention programs. Am J Audiol. 2000;9(1):9–29 [PubMed] [Google Scholar]
- 6.van Straaten HLM, Hille ETM, Kok JH, Verkerk PH; Dutch NICU Neonatal Hearing Screening Working Group . Implementation of a nation-wide automated auditory brainstem response hearing screening programme in neonatal intensive care units. Acta Paediatr. 2003;92(3):332–338 [PubMed] [Google Scholar]
- 7.Russ SA, Rickards F, Poulakis Z, Barker M, Saunders K, Wake M. Six year effectiveness of a population based two tier infant hearing screening programme. Arch Dis Child. 2002;86(4):245–250 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Nelson HD, Bougatsos C, Nygren P; 2001 US Preventive Services Task Force . Universal newborn hearing screening: systematic review to update the 2001 US Preventive Services Task Force Recommendation. Pediatrics. 2008;122(1):e266–e276 [DOI] [PubMed] [Google Scholar]
- 9.Kennedy CR, McCann DC, Campbell MJ, et al. Language ability after early detection of permanent childhood hearing impairment. N Engl J Med. 2006;354(20):2131–2141 [DOI] [PubMed] [Google Scholar]
- 10.Korver AM, Konings S, Dekker FW, et al. ; DECIBEL Collaborative Study Group . Newborn hearing screening vs later hearing screening and developmental outcomes in children with permanent childhood hearing impairment. JAMA. 2010;304(15):1701–1708 [DOI] [PubMed] [Google Scholar]
- 11.Colgan S, Gold L, Wirth K, et al. The cost-effectiveness of universal newborn screening for bilateral permanent congenital hearing impairment: systematic review. Acad Pediatr. 2012;12(3):171–180 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wake M, Poulakis Z, Hughes EK, Carey-Sargeant C, Rickards FW. Hearing impairment: a population study of age at diagnosis, severity, and language outcomes at 7–8 years. Arch Dis Child. 2005;90(3):238–244 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wake M, Hughes EK, Poulakis Z, Collins C, Rickards FW. Outcomes of children with mild-profound congenital hearing loss at 7 to 8 years: a population study. Ear Hear. 2004;25(1):1–8 [DOI] [PubMed] [Google Scholar]
- 14.Australian Bureau of Statistics Socio-Economic Indexes for Areas (SEIFA). Volume 1.1. Canberra, Australia: Australian Bureau of Statistics; 2001 [Google Scholar]
- 15.Australian Bureau of Statistics Migration, Australia (Catalogue no. 3412.0). Canberra, Australia: Australian Bureau of Statistics; 2006 [Google Scholar]
- 16.Productivity Commission Report on Government Services 2007. Melbourne, Australia: Productivity Commission; 2007 [Google Scholar]
- 17.Murphy E, Leigh-Erin R. Bridging the Gap Between Universal Newborn Hearing and Screening (UNHS) and Audiological Assessment: Achieving Above 95% Follow-up to Audiological Assessment—The NSW Health SWISH Program. Presented at the 2006 International Conference on Newborn Hearing Screening: Beyond New Born Hearing Screening: Infant and Childhood Hearing in Science and Clinical Practice; May 31–June 3, 2016; Cernobbio, Italy; 2006 [Google Scholar]
- 18.Barker MJ, Hughes EK, Wake M. NICU-only versus universal screening for newborn hearing loss: Population audit. J Paediatr Child Health. 2013;49(1):E74–E79 [DOI] [PubMed] [Google Scholar]
- 19.Ewing IR, Ewing AWG. The ascertainment of deafness in infancy and early childhood. J Laryngol Otol. 1944;59:309–333 [Google Scholar]
- 20.Russ SA, Poulakis Z, Wake M, et al. The distraction test: the last word? J Paediatr Child Health. 2005;41(4):197–200 [DOI] [PubMed] [Google Scholar]
- 21.Zimmerman IL, Steiner VG, Pond RE. Preschool Language Scale. 4th ed. London, England: Harcourt Assessment; 2002 [Google Scholar]
- 22.Dunn LM, Dunn DM. The Peabody Picture Vocabulary Test. 4th ed. Minneapolis, MN: Pearson; 2007 [Google Scholar]
- 23.Muter V, Hulme C, Snowling M. Phonological Abilities Test. London, UK: Harcourt Assessment; 1997 [Google Scholar]
- 24.Feeny D. The Health Utilities Index: A Tool for Assessing Health Benefits. PRO Newsletter. 2005;34:2–6 [Google Scholar]
- 25.Varni JW, Burwinkle TM, Seid M, Skarr D. The PedsQL 4.0 as a pediatric population health measure: feasibility, reliability, and validity. Ambul Pediatr. 2003;3(6):329–341 [DOI] [PubMed] [Google Scholar]
- 26.Goodman R. Psychometric properties of the strengths and difficulties questionnaire. J Am Acad Child Adolesc Psychiatry. 2001;40(11):1337–1345 [DOI] [PubMed] [Google Scholar]
- 27.Wechsler D, Naglieri JA. Wechsler Nonverbal Scale of Ability. San Antonio, TX: Harcourt Assessment; 2006 [Google Scholar]
- 28.Australian Bureau of Statistics Socio-Economic Indexes for Areas (SEIFA) (Technical Paper 2006). Canberra, Australia: Australian Bureau of Statistics; 2008.
- 29.Hawes DJ, Dadds MR. Australian data and psychometric properties of the Strengths and Difficulties Questionnaire. Aust N Z J Psychiatry. 2004;38(8):644–651 [DOI] [PubMed] [Google Scholar]
- 30.Wake M, Levickis P, Tobin S, et al. Improving outcomes of preschool language delay in the community: protocol for the Language for Learning randomised controlled trial. BMC Pediatr. 2012;12(96):96 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.HUI Mark2 and Mark3 Reference Population Data (“HUI2 and HUI3 Population Norms”). Available at: http://www.healthutilities.com. Accessed November 30, 2012
- 32.American Academy of Pediatrics, Joint Committee on Infant Hearing . Year 2007 position statement: principles and guidelines for early hearing detection and intervention programs. Pediatrics. 2007;120(4):898–921 [DOI] [PubMed] [Google Scholar]
- 33.Raffle AE, Muir Gray JA. Screening: evidence and practice. New York, NY: Oxford University Press; 2007 [Google Scholar]
- 34.Australian Hearing Demographic Details of Persons Under the Age of 21 years With a Hearing Impairment Who Are Fitted With a Hearing Aid or Cochlear Implant—2011. Canberra, Australia: Australian Hearing; 2012 [Google Scholar]