Abstract
Background and objectives
Hemodialysis (HD) per se entails vascular dysfunction in patients with ESRD. Endothelial dysfunction is a key step in atherosclerosis and is characterized by impaired flow–mediated dilation (FMD). Interventional studies have shown that cocoa flavanol (CF)–rich supplements improve vascular function. Aim of this study was to investigate the effect of flavanol–rich bioactive food ingredients on acute and chronic HD–induced vascular dysfunction in ESRD.
Design, setting, participants, & measurements
We conducted a randomized, double–blind, placebo–controlled trial from 2012 to 2013. Fifty-seven participants were enrolled, ingested CF-rich beverages (900 mg CF per study day), and were compared with those ingesting CF-free placebo. This included (1) a baseline cross–over acute study to determine safety and efficacy of CF and (2) a subsequent chronic parallel group study with a 30-day follow-up period to study effects of CF on HD–mediated vascular dysfunction entailing (3) an acute substudy during HD in flavanol-naive patients and (4) an acute on chronic study during HD. Primary and secondary outcome measures included changes in FMD and hemodynamics.
Results
CF ingestion was well tolerated. Acute ingestion improved FMD by 53% (3.2±0.6% to 4.8±0.9% versus placebo, 3.2±0.7% to 3.3±0.8%; P<0.001), with no effects on BP or heart rate. A 30-day ingestion of CF led to an increase in baseline FMD by 18% (3.4±0.9% to 3.9±0.8% versus placebo, 3.5±0.7% to 3.5±0.7%; P<0.001), with reduced diastolic BP (73±12 to 69±11 mmHg versus placebo, 70±11 to 73±13 mmHg; P=0.03) and increased heart rate (70±12 to 74±13 bpm versus placebo, 75±15 to 74±13 bpm; P=0.01). No effects were observed for placebo. Acute ingestion of CF during HD alleviated HD–induced vascular dysfunction (3.4±0.9% to 2.7±0.6% versus placebo, 3.5±0.7% to 2.0±0.6%; P<0.001). This effect was sustained throughout the study (acute on chronic, 3.9±0.9% to 3.0±0.7% versus placebo, 3.5±0.7% to 2.2±0.6; P=0.01).
Conclusions
Dietary CF ingestion mitigates acute HD–induced and chronic endothelial dysfunction in patients with ESRD and thus, improves vascular function in this high-risk population. Larger clinical trials are warranted to test whether this translates into an improved cardiovascular prognosis in patients with ESRD.
Keywords: cocoa-flavanols; endothelial function; hemodialysis; vascular functions; nutrition; blood pressure; double-blind method; heart rate; hemodynamics; humans; kidney failure, chronic
Introduction
Patients with renal insufficiency form one of the fastest growing populations with a high prevalence for cardiovascular diseases (CVD) (1). ESRD together with diabetes, smoking, arterial hypertension, and dyslipidemia constitute major cardiovascular risk factors (2). This leads to a dramatic increase in morbidity and mortality in this population, which is eight times greater than in the general population (3).
Atherosclerosis is characterized by atheromatous plaques with blood flow restrictions and tissue ischemia and leads to altered vascular compliance because of distorted conduit function and increased arterial stiffness (4,5). Macrovascular prognostic implications include endothelial dysfunction, which is an important and early step in the development of atherosclerosis (6). Flow-mediated vasodilation (FMD) of the brachial artery was identified as a prognostic relevant surrogate marker in patients with cardiovascular risk factors, established CVD, and ESRD (7–9). Microvascular changes in ESRD entail small arterial structural and functional alterations, characterizing the peripheral resistance vasculature, which directly contributes to the development of hypertension common in ESRD (10).
Vascular dysfunctions are associated with heart failure and sudden cardiac death. Elevated cardiac troponin levels reflect this and are an important predictor of all-cause mortality in ESRD (11). Nontraditional and interrelated risk factors that promote atherosclerosis include chronic inflammation and oxidative stress (12,13). Vascular dysfunctions ascribed to patients with ESRD are further perpetuated by impaired nitric oxide (NO) bioavailability. Importantly, hemodialysis (HD) itself acutely impairs endothelial function through reduction of NO bioactivity (14).
Therapeutic approaches counteracting atherosclerosis and CVD in this collective seem insufficient. Therefore, interventions to target key steps in atherogenesis are of great importance. An increasing body of evidence supports the concept that a healthy diet rich in fruits and vegetables is associated with a lower incidence of morbidity and mortality from CVD in nonrenal patients (15). Cocoa flavanols (CFs), like epicatechin and catechin as well as the procyanides, represent a subgroup of plant-derived polyphenols; are, in particular, present in tea, wine, cocoa, and berries; and have been substantiated as biologic active food constituents by the European Food Safety Authority (16). Interventional studies have shown that CFs exert beneficial effects regarding the regulation of BP and recovery of endothelial function (17,18). The mechanisms by which CFs mediate their effects are not fully understood. An enhancement in NO bioactivity through increases in NO synthase activity has been shown along with enhanced endothelial homeostatic vascular repair capabilities and activation of signaling pathways (19,20).
Although interventional trials have been conducted in healthy volunteers and patients with hypertension and diabetes, highlighting potential health benefits through flavanol ingestion regimens, studies investigating the effect on vascular dysfunction in patients with ESRD are sparse (21–23). We hypothesized that CF ingestion improves chronic and HD–induced vascular dysfunction in patients with ESRD. To test this hypothesis, we conducted this randomized, double–blind, and placebo–controlled trial, including the assessment of acute and longer-term effects.
Materials and Methods
We conducted this randomized, double–blind, placebo–controlled trial in accordance with Good Clinical Practice to investigate the effect of a flavanol–rich dietary supplement in patients with ESRD (Figure 1) (Clinicaltrials.gov no. NCT01412320). This was an independent investigator–initiated trial without any commercial interest. The Institutional Review Board and Ethics Committee approved the protocol, and the study was performed in accordance with the Declaration of Helsinki and the European Union Clinical Trial Directive (2001/20/EC). Patients were enrolled at the DaVita Renal Center, Dusseldorf from 2012 to 2013. Participants meeting inclusion criteria of ESRD, age >18 years old, chronic HD, and a nonfistula brachial artery were eligible for the trial. Exclusion criteria were acute renal failure, acute infection, heart failure, pregnancy, or anuria (Supplemental Table 1). An external independent data monitoring committee monitored the study. Participants provided written informed consent, were blinded, and were randomly assigned to the treatment group with a 1:1 allocation. Randomization sequences were computer generated centrally at the coordinating center using permuted blocks of one to four participants. Baseline assessments of trial participants included medical history, laboratory analyses, and current medications.
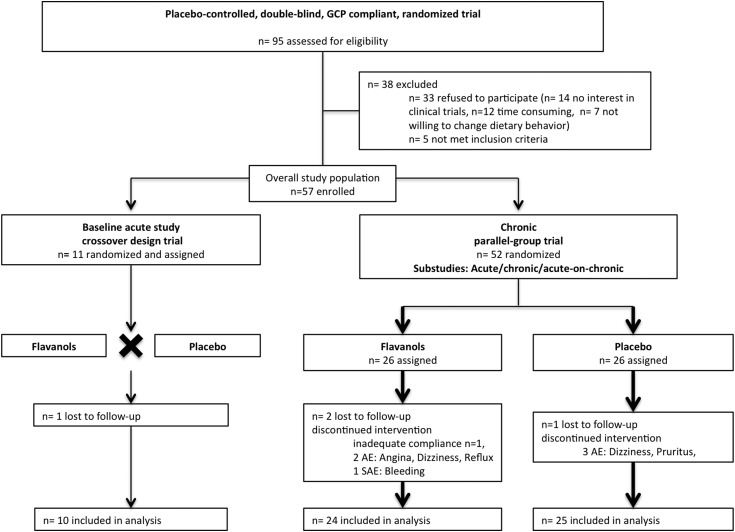
Figure 1.
Consolidated Standards of Reporting Trials (CONSORT) diagram. CONSORT diagram depicting flow of study participants. AE, adverse event; GCP, good clinical practice; SAE, serious adverse event.
The CF–containing test materials were low–calorie fruit–flavored beverage mixes supplied as agglomerated powders by Mars Symbioscience (Germantown, MD) and standardized for their CF content. CF and placebo drinks were closely matched for equal content of macronutrients, micronutrients, caloric load, theobromine, and caffeine (Supplemental Table 2). All drinks were similar in taste and supplied in opaque sachets labeled with an anonymized alphanumeric identifier code. One CF sachet contained 450 mg total CF, and patients were advised to dissolve both sachets in a minimum of 150 ml water and consume two sachets in the acute study and two sachets per each study day in the chronic study. Background diet was assessed by dietary interviews at baseline, during the study, and at the follow-up visit.
FMD of the brachial artery was measured by a noninvasive technique to assess endothelial function as previously described (24,25). Briefly, with the use of a 12-MHz linear–array transducer and the Vivid I System (GE Healthcare), the brachial artery diameter was acquired proximal of the antecubital fossa before and immediately at 20, 40, 60, and 80 seconds after cuff deflation of 5-minute forearm arterial occlusion at 250 mmHg pressure with a 12.5-cm-wide cuff. End diastolic frames were obtained and analyzed with an automated analysis system (Brachial Analyzer; Medical Imaging Applications, Iowa City, IA). Nitroglycerin-mediated vasodilation (i.e., endothelium-independent vasodilation) was measured at 4 minutes after 400 μg sublingual nitroglycerin in patients with adequate BP (26). FMD was determined as the maximal percentage diameter change of the arterial diameter measurement relative to the baseline measurement.
Detailed methods relative to determination of pulse wave velocity and BP, analysis of epicatechin and its related metabolites, nitrite and nitrate, advanced glycation end product carboxymethyl lysine, and markers of inflammation high-sensitivity IL-6 and oxidative stress (oxidized LDL) are provided in Supplemental Material.
Results are expressed as means±SDs. Previous studies yielded an effect size of 0.8 for the primary outcome measure FMD in our laboratory (22,23). Sample size calculations on the basis of a 5% two-sided significance, 80% power, and 5% dropout indicated that 26 patients in each group needed to be enrolled. Paired t test was used for statistical analyses within the groups; unpaired t test was used for comparing the groups on change from baseline. Comparisons using area under the curve calculations are described in Supplemental Material. All statistical tests were conducted using R and Prism 5.0 (GraphPad). P values <0.05 were regarded as statistically significant.
Results
The flow of study participants in this randomized, double–blind, and placebo–controlled trial is depicted in Figure 1. Of 95 patients initially screened, 57 were included in the study. Initially, in 2012, 11 patients were included in the acute study. Of those, five patients were unavailable for the chronic study because of hospitalization and change of the dialysis center. Subsequently, in 2013, 52 participants were enrolled in the chronic study. All patients were on chronic HD with a median dialysis vintage of 43 (interquartile range, 20–59) months and three times per week dialysis treatments. The mean age was 65±13 years old (74% men), with a body mass index of 29±5 kg/m2. Underlying renal diseases included hypertensive (32%) and diabetic (23%) nephropathy along with GN (19%) and polycystic kidney disease (18%). Additional baseline demographic, clinical, and biochemical characteristics are shown in Table 1.
Table 1.
Baseline patient characteristics
| Characteristics | Overall study group (n=57) | Chronic Study (n=52) | |
|---|---|---|---|
| Cocoa Flavanol (n=26) | Placebo (n=26) | ||
| Men, n | 42 | 18 | 20 |
| Age (yr), mean±SD | 65±13 | 65±14 | 66±14 |
| Height (cm), mean±SD | 174±10 | 173±9 | 174±11 |
| Weight (kg), mean±SD | 88±17 | 85±21 | 90±16 |
| Body mass index (kg/m2), mean±SD | 29±5 | 28±6 | 29±6 |
| Renal diagnosis, n | |||
| Hypertensive/large vessel | 18 | 7 | 10 |
| Diabetic nephropathy | 13 | 4 | 7 |
| Polycystic kidney disease | 10 | 4 | 5 |
| GN | 11 | 8 | 3 |
| Other/miscellaneous | 5 | 3 | 1 |
| Dialysis vintage (mo), median (IQR) | 43 (20–59) | 48 (23–63) | 34 (20–58) |
| Hypertension, n | 52 | 23 | 25 |
| Diabetes, n | 21 | 6 | 13 |
| Current smoker, n | 13 | 8 | 4 |
| Hypercholesterinemia, n | 24 | 10 | 14 |
| CVD, n | 20 | 7 | 11 |
| Medication, n | |||
| Acetylsalicylic acid | 28 | 11 | 15 |
| Statin | 26 | 12 | 12 |
| AT blocker | 12 | 6 | 10 |
| ACE-I | 11 | 6 | 3 |
| β-Blocker | 30 | 13 | 16 |
| Ca channel blocker | 22 | 11 | 10 |
| Diuretics | 39 | 16 | 19 |
Baseline characteristics for the overall study group (n=57) and the chronic study (cocoa flavanol and placebo; n=26 each). Acute study characteristics are given in Supplemental Table 3. Other/miscellaneous refers to amyloidosis, renal carcinoma, hepatorenal syndrome, and unknown origin. IQR, interquartile range; CVD, cardiovascular disease; AT, angiotensin; ACE-I, angiotensin–converting enzyme inhibitor.
Acute CF Effects on Conduit Artery Function
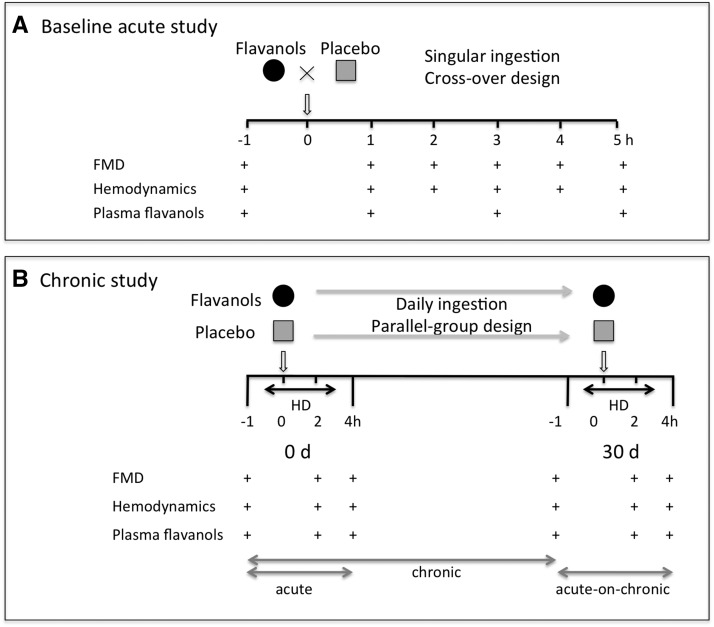
In a baseline acute study, we determined the safety and efficacy of a singular CF ingestion at the end of the weekend interval in 10 patients with ESRD compared with a nutrient-matched placebo in a crossover design from October to November of 2012 (baseline characteristics are given in Supplemental Table 3). Before intake of the CF-containing drinks, baseline FMD measurements, plasma flavanol metabolites, and hemodynamics were obtained. These examinations were repeated hourly (Figure 2A). Study days were separated by 1 week to avoid potential carryover effects.
Figure 2.
Study design. (A) Baseline acute study: placebo–controlled, double–blind, randomized study design to test acute effects of a single–occasion oral intake of cocoa flavanols (CFs) in patients with ESRD investigated at the end of the weekend interval. Flow-mediated dilation (FMD), hemodynamics, and CF metabolites were measured before and up to 4 hours after consumption of a single flavanol drink. Ingestion was separated by at least 1 week to avoid potential carryover effects. (B) Chronic study: placebo–controlled, double–blind, randomized, parallel group study design to investigate the sustained effects of daily CF intake on endothelial function in patients with ESRD undergoing hemodialysis (HD). FMD, hemodynamic parameters, and plasma CF metabolites were measured before, during, and after HD, with ingestion at the beginning of dialysis.
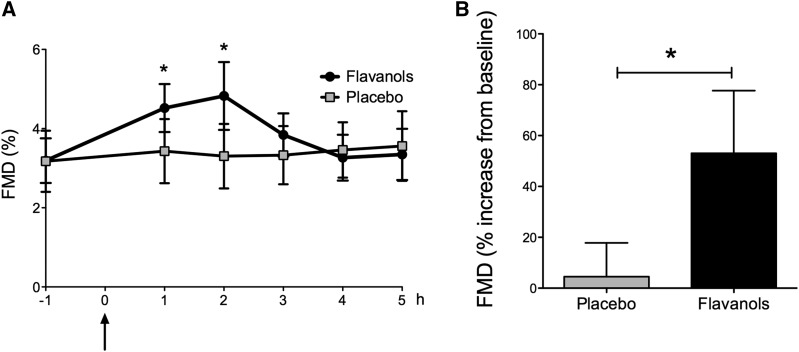
Acute CF ingestion in patients with ESRD led to an improved conduit artery function with enhanced FMD and a maximum response at 2 hours after CF intake compared with placebo (CF from 3.2±0.6% to 4.8±0.9%; P<0.001; placebo from 3.2±0.7% to 3.3±0.8%; P=0.72; unpaired P<0.001) (Figure 3A), corresponding to a flavanol–induced FMD increase of 53% (Figure 3B) with no change in nitroglycerin-mediated vasodilation (Supplemental Figure 1). Importantly, CF ingestion was well tolerated. We found no differences in plasma potassium or pH levels and no effect on BP or heart rate (HR) (Supplemental Figure 2).
Figure 3.
Baseline acute study. Acute effect of dietary flavanols on endothelial function in patients with ESRD. (A) Time course in flow-mediated dilation (FMD) in patients receiving either the flavanol-containing drink or placebo. Arrow denotes ingestion of cocoa flavanols or placebo at 0 hour. (B) Maximum increase in FMD from baseline in patients with ESRD receiving cocoa flavanols versus placebo. Data are given as means±SDs. *P<0.05 (n=10).
The acute improvement in vascular function after ingestion of the CF-containing beverage was associated with an increase in plasma flavanol metabolites (Supplemental Figure 3).
Endothelial Functions after Chronic CF Intake
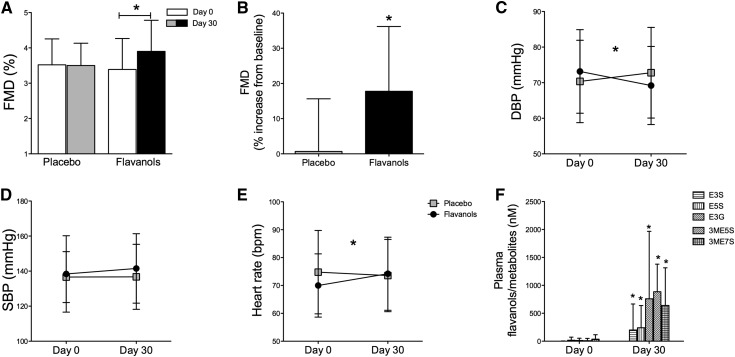
To determine the longer-term effects of CF on vascular functions in patients with ESRD, we conducted a subsequent chronic study on the basis of a parallel group design (Figure 2B) from January to November of 2013; 3 of 52 patients enrolled were lost to follow-up and included only in the acute substudy. Baseline patient characteristics (Table 1) and dialysis intervals as well as FMD were balanced between the study groups. After a 30-day ingestion of CF, we observed improved macrovascular functions. Baseline FMD increased compared with placebo (CF from 3.4±0.9% to 3.9±0.8%; P<0.001; placebo from 3.5±0.7% to 3.5±0.7%; P=0.72; unpaired P<0.001) (Figure 4A, Supplemental Figure 4), corresponding to an FMD increase by 18% (P<0.001) (Figure 4B). No difference was observed for endothelial independent function (Supplemental Figure 5). Improved endothelial function through CF ingestion was paralleled by improved microvascular functions with a decrease in diastolic BP (DBP) and an associated increase in HR (DBP: CF, 73±12 to 69±11 mmHg; P=0.004; placebo, 70±11 to 73±13 mmHg; P=0.39; unpaired P=0.03 [Figure 4C]; HR: CF, 70±12 to 74±13 bpm; P<0.01; placebo, 75±15 to 74±13 bpm; P=0.16; unpaired P=0.01 [Figure 4E]). Chronic ingestion of CF increased structurally related epicatechin metabolites (Figure 4F). No effect was observed on aortic stiffness (pulse wave velocity), intima-media thickness, plasma nitrate and nitrite, serum markers of inflammation (C-reactive protein and IL-6), oxidized LDL, myocardial ischemia (high-sensitivity troponin), or other clinical routine parameters between groups after the 30-day ingestion period (Table 2).
Figure 4.
Chronic study. Chronic effects of dietary flavanols in patients with ESRD. (A and B) Chronic effect on endothelial function in flavanol-naïve patients after a 30-day ingestion period of cocoa flavanols or placebo (flow-mediated dilation [FMD]). (C–E) Effects on hemodynamics. An increase in heart rate and a reduction in diastolic BP (DBP) are observed after a 30-day ingestion period, with no effect on systolic BP (SBP). (F) Plasma concentration of structurally related epicatechin metabolites (SREMs) at baseline and follow-up. Metabolites quantified correspond to epicatechin-3′-β-d-glucuronide (E3G), (−)-epicatechin-3′-sulfate (E3S), (−)-epicatechin-5-sulfate (E5S), 3′-O-methyl-(−)-epicatechin-7-sulfate (3ME7S), and 3′-O-methyl-(−)-epicatechin-5-sulfate (3ME5S). Data are expressed as nanomolar (nanomoles of SREM per liter of plasma). Data are given as means±SDs. *P<0.05 (n=24–25).
Table 2.
Aortic stiffness, nitric oxide species, markers of oxidative stress and inflammation, and chemistry panel
| Parameters | Cocoa Flavanol | Placebo | P Value | ||
|---|---|---|---|---|---|
| Day 0 | Day 30 | Day 0 | Day 30 | ||
| Aortic stiffness | |||||
| PWV (m/s) | 11.9±1.1 | 11.0±0.9 | 12.4±0.9 | 12.0±1.1 | 0.32 |
| Carotid IMT (mm) | 0.81±0.02 | 0.79±0.02 | 0.81±0.02 | 0.81±0.02 | 0.28 |
| Nitric oxide interleukin | |||||
| Nitrite (µM) | 0.31±0.05 | 0.33±0.05 | 0.34±0.05 | 0.35±0.09 | 0.49 |
| Nitrate (µM) | 57.5±4.9 | 71.7±11.8 | 84.5±11.8 | 76.8±8.0 | 0.12 |
| Oxidative stress | |||||
| OxLDL (units per liter) | 50.9±3.6 | 51.2±3.6 | 51.1±2.9 | 50.1±1.8 | 0.93 |
| Inflammation | |||||
| AGE CML (ng/ml) | 3240±561 | 3438±310 | 2503±325 | 3189±324 | 0.35 |
| CRP (mg/dl) | 0.6±0.1 | 0.7±0.2 | 0.9±0.4 | 0.8±0.5 | 0.77 |
| IL-6 (pg/ml) | 2.6±0.4 | 3.3±0.4 | 3.6±0.4 | 3.3±0.6 | 0.25 |
| Myocardial ischemia | |||||
| Hs-troponin (ng/ml) | 62.6±9.4 | 66.6±10.3 | 61.5±8.0 | 62.9±8.1 | 0.93 |
| Chemistry panel | |||||
| Creatinine (mg/dl) | 8.3±0.5 | 8.4±0.6 | 7.5±0.5 | 7.8±0.5 | 0.42 |
| Cystatin C (mg/dl) | 5.2±0.3 | 5.3±0.2 | 5.6±0.2 | 5.3±0.2 | 0.86 |
| Potassium (mEq/L) | 5.1±0.2 | 5.1±0.2 | 4.9±0.2 | 5.0±0.1 | 0.06 |
| Homocysteine (mg/dl) | 44.6±4.7 | 48.0±4.4 | 36.2±1.7 | 39.6±2.1 | 0.53 |
| Total protein (mg/dl) | 7.0±0.1 | 6.9±0.1 | 6.9±0.2 | 6.9±0.1 | 0.65 |
| Phosphate (mg/dl) | 5.3±0.3 | 5.3±0.3 | 5.0±0.3 | 5.0±0.3 | 0.34 |
Unpaired t test was used for comparing the groups on change from baseline. PWV, pulse wave velocity; IMT, intima-media thickness; OxLDL, oxidized LDL; AGE CML, carboxymethyl lysine advanced glycation end products; CRP, C-reactive protein; Hs-troponin, high-sensitivity troponin.
Safety of chronic CF ingestion was assessed by adverse events (AEs) monitoring. In the CF group, one serious AE (bleeding) and two AEs (one patient with angina and one with dizziness and reflux) occurred, whereas three AEs were noted in the placebo group (pruritus and dizziness two times).
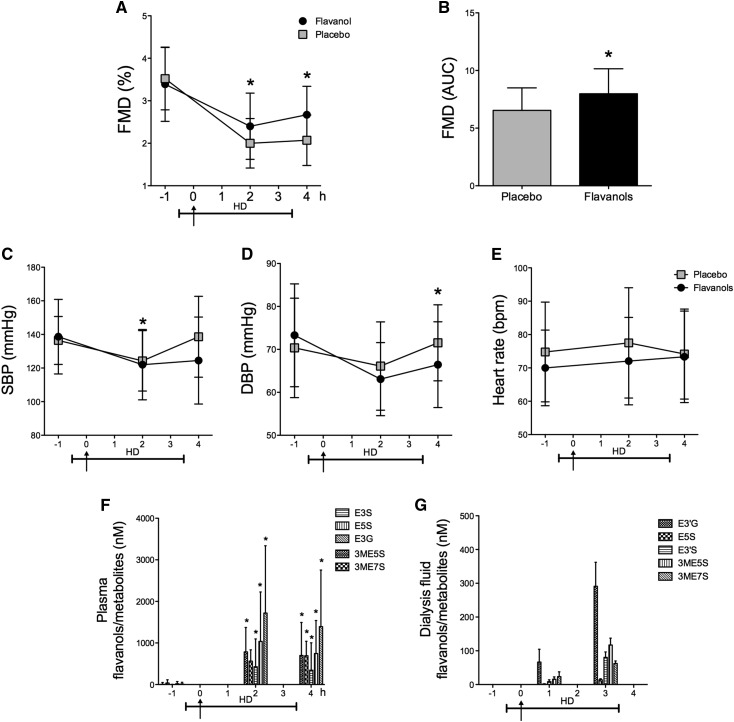
Effects on HD–Induced Vascular Dysfunction
To investigate the potential interaction and effect of CF on HD–induced acute vascular dysfunction, we performed measurements before, during, and after a single HD session (Figure 2B). HD was associated with an acute impairment in endothelial functions during and after HD (Figure 5, A and B). Notably, a single–occasion CF ingestion alleviated HD–induced vascular dysfunction in patients with ESRD during and after HD (during HD decrease of FMD: CF, 3.4±0.9% to 2.4±0.8%; P<0.001; placebo, 3.5±0.7% to 2.0±0.5%; P<0.001; unpaired P<0.01; during HD ΔFMD: CF, −1.0±0.6% versus placebo, −1.5±0.6%; P<0.001; after HD decrease of FMD: CF, 3.4±0.9% to 2.7±0.6%; P<0.001; placebo, 3.5±0.7% to 2.0±0.6%; P<0.001; unpaired P<0.001) (Figure 5, A and B, respectively). CF intake was associated with reductions in systolic BP (SBP) during HD and DBP after HD (SBP: flavanols, 139±22 to 122±21 mmHg; P<0.001; placebo, 136±14 to 124±18 mmHg; P=0.002; unpaired P=0.03; DBP: CF, 73±12 to 66±10 mmHg; P=0.01; placebo, 70±12 to 72±9 mmHg; P=0.64; unpaired P=0.04) (Figure 5, C and D, respectively). No effect was noted for HR between groups (Figure 5E). No difference in intradialytic hypotensive episodes was noted between groups. Epicatechin metabolites were increased after ingestion and persisted throughout the dialysis session (Figure 5F). Importantly, CF metabolites were found in the dialysis fluid after ingestion (Figure 5G).
Figure 5.
Chronic study. Acute cocoa flavanols effects on hemodialysis (HD) in flavanol-naive patients with ESRD on day 0 of the chronic study phase. (A and B) Improved endothelial function in ESRD under HD and after HD. AUC, area under the curve; FMD, flow-mediated dilation. (C–E) A reduction in systolic BP (SBP) during HD and a reduction in diastolic BP (DBP) after HD are observed with no effect on heart rate. (F and G) Plasma and dialysis fluid concentration of structurally related epicatechin metabolites. Metabolites quantified correspond to epicatechin-3′-β-d-glucuronide (E3G), (−)-epicatechin-3′-sulfate (E3S), (−)-epicatechin-5-sulfate (E5S), 3′-O-methyl-(−)-epicatechin-7-sulfate (3ME7S), and 3′-O-methyl-(−)-epicatechin-5-sulfate (3ME5S). Arrows denote ingestion of cocoa flavanols or placebo. Data are given as means±SDs. *P<0.05 (n=25–26).
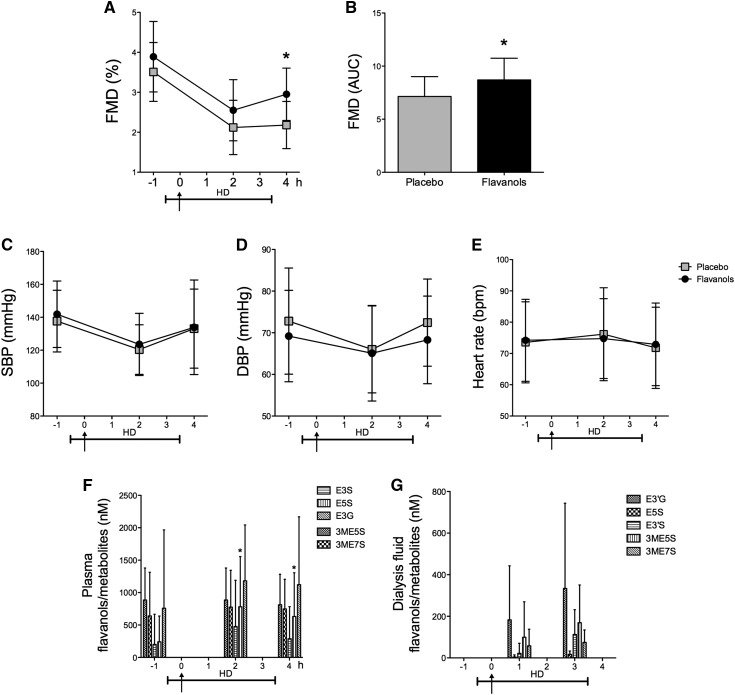
After the 30-day ingestion period, we also determined the potential acute on chronic effect of CF during HD (Figure 2B). Ingestion of CF showed a sustained effect on endothelial functions, even after HD (during HD decrease of FMD: CF, 3.9±0.9% to 2.5±0.8%; P<0.001; placebo, 3.5±0.7% to 2.1±0.7%; P<0.001; unpaired P=0.60; after HD decrease of FMD: CF, 3.9±0.9% to 3.0±0.7%; P<0.001; placebo, 3.5±0.7% to 2.2±0.6; P<0.001; unpaired P=0.01) (Figure 6, A and B). No effect was observed regarding SBP and DBP or HR in this acute on chronic setting (SBP: CF, 141±20 to 134±29 mmHg; P=0.05; placebo, 138±19 to 133±24 mmHg; P=0.37; unpaired P=0.60; DBP: CF, 69±11 to 68±11; P=0.69; placebo, 73±13 to 72±10 mmHg; P=0.86; unpaired P=0.86; HR: CF, 74±13 to 73±13 bpm; P=0.58; placebo, 74±13 to 72±13 bpm; P=0.38; unpaired P=0.85) (Figure 6, C–E). Epicatechin metabolites were increased after the chronic ingestion regimen and showed sustained increase after a single CF intake only in terms of (−)-epicatechin-5-sulfate (Figure 6F). Epicatechin metabolites were detected in the dialysis fluid before and after ingestion of the flavanol-containing beverage (Figure 6G).
Figure 6.
Chronic study. Acute on chronic effects of dietary flavanols in flavanol-pretreated patients with ESRD on day 30 of the chronic study phase. (A and B) Sustained improvement on endothelial function after hemodialysis (HD). AUC, area under the curve; FMD, flow-mediated dilation. (C–E) No effect on systolic BP (SBP), diastolic BP (DBP) or heart rate. (F and G) Plasma and dialysis fluid concentrations of structurally related epicatechin metabolites. Metabolites quantified correspond to epicatechin-3′-β-d-glucuronide (E3G), (−)-epicatechin-3′-sulfate (E3S), (−)-epicatechin-5-sulfate (E5S), 3′-O-methyl-(−)-epicatechin-7-sulfate (3ME7S), and 3′-O-methyl-(−)-epicatechin-5-sulfate (3ME5S). Arrows denote ingestion of cocoa flavanols or placebo. Data are given as means±SDs. *P<0.05 (n=24–25).
Discussion
The main findings of this study are: (1) CF ingestion at 900 mg/d is well tolerated in patients with ESRD; (2) Acute and chronic CF ingestion reverses, in part, impairment of endothelial function; (3) A chronic dietary CF intake reduces DBP in ESRD without affecting markers of aortic stiffness; and (4) CF intake mitigates HD–induced vascular dysfunction acutely in flavanol-naive patients and exerts sustained effects after the chronic ingestion period.
ESRD and Atherosclerosis
Patients with ESRD suffer from increased morbidity and mortality because of higher incidences of cardiovascular events (1,27). This increased cardiovascular mortality is mirrored by elevated cardiac high–sensitivity troponins in our study. Classic therapeutic approaches seem insufficient, and therefore, timely fashioned interventions against the atherosclerotic alterations at the level of conduit vessels are of increasing importance (28). The accelerated atherosclerotic process of ESRD involves several interrelated processes, which include nondialysis- as well as dialysis-related factors. Intimal-medial thickening, increased aortic stiffness and endothelial dysfunction, oxidative stress, and the milieu of constant low–grade inflammation are mainly held responsible (29,30). Endothelial dysfunction is a key feature in the development of atherosclerosis and has been associated with higher incidence of cardiac events (7,12,31). The mechanism of endothelial dysfunction induced by HD is multifactorial. Decreased NO formation by accumulated endogenous NO synthase inhibitors or increased inactivation and scavenging of NO is considered responsible (14,32–36). Corroborating previous findings, we observed an acute decrease in endothelium-dependent vasodilation after a single HD session, and we showed additional impairment, even during HD (14,37).
Flavanol Intake in Patients with ESRD
Dietary intervention studies have shown that foods rich in flavanols are associated with a reduced cardiovascular risk (38). We have shown that CF can acutely alleviate endothelial dysfunction in individuals with established cardiovascular risk factors (39). Moreover, flavanol–rich bioactive food ingredients improve vascular dysfunction in patients suffering from diabetes, with an inverse relationship between the intake of CF and BP, insulin resistance, and glucose tolerance (22). To limit effects of other cocoa constituents, we now use a test drink closely matched for ingredients, like caffeine and theobromine. We here show an acute attenuation of endothelial dysfunctions in patients with ESRD after a singular ingestion of CF. The improved endothelial function persisted during the chronic 30-day ingestion period, which is in line with a previous study in patients with ESRD after green tea consumption (21). Notably, the improvement in endothelial function was observed even during an HD session, and additionally, a sustained effect was evident after the chronic ingestion period. This suggests an acute beneficial effect on HD–induced endothelial cell injury through CF ingestion. Impressively, the degree of reversion of endothelial dysfunction is comparable with the effects observed in ESRD through administration of statins, use of angiotensin–converting enzyme inhibitors in patients with cardiovascular risk, or use of other dietary intervention and lifestyle modification studies (40–43). The effects on the conduit arteries with improved endothelial functions did not translate into reduced arterial stiffness indices after this 30-day ingestion period.
We further observed an effect on the vasculature with a decrease in DBP. In CKD, an optimal DBP of ≥70 mmHg is associated with reduced mortality (44). Our observations are in line with published data showing improvements in microvascular functions after flavanol ingestion (45). This could benefit patients with hypertension, which is common in ESRD (18). Importantly, no difference was observed for intra- and postdialytic hypotensive episodes in our study. Although autonomic dysfunction and lower HR variability are known for patients with ESRD, the increased HR observed in our study might develop as a compensational mechanism (46).
Until now, vascular effects of CF in healthy individuals were considered to be related to the kinetics of the appearance in plasma of epicatechin metabolites (47). After a single CF ingestion, we observed an increase in circulating epicatechin metabolites and no clear decrease in the observation period. Our findings in this study population seem to contradict the pharmacokinetics compared with healthy individuals (47). This is in line with the notion of altered metabolism, accumulation, and excretion of certain metabolites and uremic solutes in ESRD (48). Recent data suggest that the elimination of CF is dependent on the urinary excretion of their corresponding metabolites (49). We now show that metabolites are dialyzable, which opens new insights into pharmacokinetics. The exact mechanisms through which CF exerts putative cardioprotective effects still remain incompletely elucidated. Effects on NO homeostasis with increased nitrosothiol plasma levels, cellular signal cascades, altered gene expression, and enzyme activity are considered potential pathways (19,47). In this study, we did not observe differences in plasma nitrite or nitrate levels, possibly because of small sample size or activation of inducible NO synthase as suggested for patients with ESRD (50). Experimental evidence has shown that CF exerts anti-inflammatory effects, affecting cellular TNF-α levels (51). The plasma markers of oxidative stress or inflammation were not different after the 30-day interventional period. Determination of cellular TNF-α or IL-2 levels in this population or longer–term dietary interventions might yield different results.
Limitations
We designed this study with surrogate outcomes instead of secondary prevention–specific hard end points, like cardiac and cerebrovascular events. We included patients on maintenance HD and did not preselect ESRD caused by hypertensive or large-vessel disease, in which CF might exert more profound effects. However, our intention was not to study a highly preselected cohort but investigate individuals with characteristics representative for ESRD in general, emphasizing the relevance of our findings for patients at high risk for CVD.
Despite the limitations reported above, our results show that CF ingestion is well tolerated and improves endothelial functions in patients with ESRD. CFs mitigate chronic and HD–induced vascular dysfunction in ESRD. CFs may, thus, have the potential to ameliorate vascular dysfunction in this high-risk population. Clinical end points–based clinical trials have to be conducted to investigate effects in ESRD.
Disclosures
None.
Supplementary Material
Acknowledgments
We thank Dr. Hagen Schroeter (Mars Symbioscience) and Dr. Javier Ottaviani (University of California, Davis) for critical discussions and fruitful comments. We also thank Dr. Ottaviani for analytical support of epicatechin metabolites.
T.R. is a Heisenberg Professor funded by German Research Foundation Grant Ra969/7-2. This work was, in part, funded by European Commission Flaviola Grant FP7-KBBE 226588 (to T.R., C.H., and M.K.). Mars Symbioscience provided the cocoa flavanol test products and analytical standards.
Footnotes
Present address: Prof. Tienush Rassaf, Dr. Christos Rammos, and Dr. Ulrike B. Hendgen-Cotta, Department of Cardiology, West German Heart and Vascular Center Essen, University Hospital Essen, Essen, Germany.
Published online ahead of print. Publication date available at www.cjasn.org.
See related editorial, “Cocoa Flavanols: A Magic Potion for Protecting the Endothelium in Kidney Failure?,” on pages 9–11.
This article contains supplemental material online at http://cjasn.asnjournals.org/lookup/suppl/doi:10.2215/CJN.05560515/-/DCSupplemental.
References
- 1.Go AS, Chertow GM, Fan D, McCulloch CE, Hsu CY: Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N Engl J Med 351: 1296–1305, 2004 [DOI] [PubMed] [Google Scholar]
- 2.Goodman WG, Goldin J, Kuizon BD, Yoon C, Gales B, Sider D, Wang Y, Chung J, Emerick A, Greaser L, Elashoff RM, Salusky IB: Coronary-artery calcification in young adults with end-stage renal disease who are undergoing dialysis. N Engl J Med 342: 1478–1483, 2000 [DOI] [PubMed] [Google Scholar]
- 3.Parfrey PS: Cardiac disease in dialysis patients: Diagnosis, burden of disease, prognosis, risk factors and management. Nephrol Dial Transplant 15[Suppl 5]: 58–68, 2000 [DOI] [PubMed] [Google Scholar]
- 4.London GM, Marchais SJ, Guerin AP, Metivier F, Adda H: Arterial structure and function in end-stage renal disease. Nephrol Dial Transplant 17: 1713–1724, 2002 [DOI] [PubMed] [Google Scholar]
- 5.Verbeke F, Van Biesen W, Honkanen E, Wikström B, Jensen PB, Krzesinski JM, Rasmussen M, Vanholder R, Rensma PL, CORD Study Investigators : Prognostic value of aortic stiffness and calcification for cardiovascular events and mortality in dialysis patients: Outcome of the calcification outcome in renal disease (CORD) study. Clin J Am Soc Nephrol 6: 153–159, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ross R: Atherosclerosis—An inflammatory disease. N Engl J Med 340: 115–126, 1999 [DOI] [PubMed] [Google Scholar]
- 7.Perticone F, Ceravolo R, Pujia A, Ventura G, Iacopino S, Scozzafava A, Ferraro A, Chello M, Mastroroberto P, Verdecchia P, Schillaci G: Prognostic significance of endothelial dysfunction in hypertensive patients. Circulation 104: 191–196, 2001 [DOI] [PubMed] [Google Scholar]
- 8.Yeboah J, Crouse JR, Hsu FC, Burke GL, Herrington DM: Brachial flow-mediated dilation predicts incident cardiovascular events in older adults: The Cardiovascular Health Study. Circulation 115: 2390–2397, 2007 [DOI] [PubMed] [Google Scholar]
- 9.Verbeke FH, Pannier B, Guérin AP, Boutouyrie P, Laurent S, London GM: Flow-mediated vasodilation in end-stage renal disease. Clin J Am Soc Nephrol 6: 2009–2015, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Luksha N, Luksha L, Carrero JJ, Hammarqvist F, Stenvinkel P, Kublickiene K: Impaired resistance artery function in patients with end-stage renal disease. Clin Sci (Lond) 120: 525–536, 2011 [DOI] [PubMed] [Google Scholar]
- 11.Apple FS, Murakami MM, Pearce LA, Herzog CA: Predictive value of cardiac troponin I and T for subsequent death in end-stage renal disease. Circulation 106: 2941–2945, 2002 [DOI] [PubMed] [Google Scholar]
- 12.Heitzer T, Schlinzig T, Krohn K, Meinertz T, Münzel T: Endothelial dysfunction, oxidative stress, and risk of cardiovascular events in patients with coronary artery disease. Circulation 104: 2673–2678, 2001 [DOI] [PubMed] [Google Scholar]
- 13.Stenvinkel P: Inflammation in end-stage renal disease: The hidden enemy. Nephrology (Carlton) 11: 36–41, 2006 [DOI] [PubMed] [Google Scholar]
- 14.Meyer C, Heiss C, Drexhage C, Kehmeier ES, Balzer J, Mühlfeld A, Merx MW, Lauer T, Kühl H, Floege J, Kelm M, Rassaf T: Hemodialysis-induced release of hemoglobin limits nitric oxide bioavailability and impairs vascular function. J Am Coll Cardiol 55: 454–459, 2010 [DOI] [PubMed] [Google Scholar]
- 15.Sofi F, Cesari F, Abbate R, Gensini GF, Casini A: Adherence to Mediterranean diet and health status: Meta-analysis. BMJ 337: a1344, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Agostoni C, Bresson J-L, Fairweather-Tait S, Flynn A, Golly I, Korhonen H, Lagiou P, Løvik M, Marchelli R, Martin A, Moseley B, Neuhäuser-Berthold M, Przyrembe H, Salminen S, Yolanda Sanz SJJS, Strobel S, Inge Tetens DT, Loveren HV, Verhagen H: Scientific opinion on the substantiation of a health claim related to cocoa flavanols and maintenance of normal endothelium-dependent vasodilation pursuant to Article 13(5) of Regulation (EC) No 1924/2006. EFSA J 10: 2809, 2012 [Google Scholar]
- 17.Heiss C, Jahn S, Taylor M, Real WM, Angeli FS, Wong ML, Amabile N, Prasad M, Rassaf T, Ottaviani JI, Mihardja S, Keen CL, Springer ML, Boyle A, Grossman W, Glantz SA, Schroeter H, Yeghiazarians Y: Improvement of endothelial function with dietary flavanols is associated with mobilization of circulating angiogenic cells in patients with coronary artery disease. J Am Coll Cardiol 56: 218–224, 2010 [DOI] [PubMed] [Google Scholar]
- 18.Taubert D, Roesen R, Lehmann C, Jung N, Schömig E: Effects of low habitual cocoa intake on blood pressure and bioactive nitric oxide: A randomized controlled trial. JAMA 298: 49–60, 2007 [DOI] [PubMed] [Google Scholar]
- 19.Ramirez-Sanchez I, Maya L, Ceballos G, Villarreal F: (-)-Epicatechin activation of endothelial cell endothelial nitric oxide synthase, nitric oxide, and related signaling pathways. Hypertension 55: 1398–1405, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Benito S, Lopez D, Sáiz MP, Buxaderas S, Sánchez J, Puig-Parellada P, Mitjavila MT: A flavonoid-rich diet increases nitric oxide production in rat aorta. Br J Pharmacol 135: 910–916, 2002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Park CS, Kim W, Woo JS, Ha SJ, Kang WY, Hwang SH, Park YW, Kim YS, Ahn YK, Jeong MH, Kim W: Green tea consumption improves endothelial function but not circulating endothelial progenitor cells in patients with chronic renal failure. Int J Cardiol 145: 261–262, 2010 [DOI] [PubMed] [Google Scholar]
- 22.Balzer J, Rassaf T, Heiss C, Kleinbongard P, Lauer T, Merx M, Heussen N, Gross HB, Keen CL, Schroeter H, Kelm M: Sustained benefits in vascular function through flavanol-containing cocoa in medicated diabetic patients a double-masked, randomized, controlled trial. J Am Coll Cardiol 51: 2141–2149, 2008 [DOI] [PubMed] [Google Scholar]
- 23.Heiss C, Kleinbongard P, Dejam A, Perré S, Schroeter H, Sies H, Kelm M: Acute consumption of flavanol-rich cocoa and the reversal of endothelial dysfunction in smokers. J Am Coll Cardiol 46: 1276–1283, 2005 [DOI] [PubMed] [Google Scholar]
- 24.Rammos C, Hendgen-Cotta UB, Sobierajski J, Adamczyk S, Hetzel GR, Kleophas W, Dellanna F, Kelm M, Rassaf T: Macrophage migration inhibitory factor is associated with vascular dysfunction in patients with end-stage renal disease. Int J Cardiol 168: 5249–5256, 2013 [DOI] [PubMed] [Google Scholar]
- 25.Rassaf T, Heiss C, Mangold S, Leyendecker T, Kehmeier ES, Kelm M, Lauer T: Vascular formation of nitrite after exercise is abolished in patients with cardiovascular risk factors and coronary artery disease. J Am Coll Cardiol 55: 1502–1503, 2010 [DOI] [PubMed] [Google Scholar]
- 26.Rassaf T, Heiss C, Hendgen-Cotta U, Balzer J, Matern S, Kleinbongard P, Lee A, Lauer T, Kelm M: Plasma nitrite reserve and endothelial function in the human forearm circulation. Free Radic Biol Med 41: 295–301, 2006 [DOI] [PubMed] [Google Scholar]
- 27.Mann JF, Gerstein HC, Pogue J, Bosch J, Yusuf S: Renal insufficiency as a predictor of cardiovascular outcomes and the impact of ramipril: The HOPE randomized trial. Ann Intern Med 134: 629–636, 2001 [DOI] [PubMed] [Google Scholar]
- 28.Nolan CR: Strategies for improving long-term survival in patients with ESRD. J Am Soc Nephrol 16[Suppl 2]: S120–S127, 2005 [DOI] [PubMed] [Google Scholar]
- 29.Locatelli F, Canaud B, Eckardt KU, Stenvinkel P, Wanner C, Zoccali C: Oxidative stress in end-stage renal disease: An emerging threat to patient outcome. Nephrol Dial Transplant 18: 1272–1280, 2003 [DOI] [PubMed] [Google Scholar]
- 30.Giachelli CM: Vascular calcification mechanisms. J Am Soc Nephrol 15: 2959–2964, 2004 [DOI] [PubMed] [Google Scholar]
- 31.Suwaidi JA, Hamasaki S, Higano ST, Nishimura RA, Holmes DR, Jr., Lerman A: Long-term follow-up of patients with mild coronary artery disease and endothelial dysfunction. Circulation 101: 948–954, 2000 [DOI] [PubMed] [Google Scholar]
- 32.Vallance P, Leone A, Calver A, Collier J, Moncada S: Accumulation of an endogenous inhibitor of nitric oxide synthesis in chronic renal failure. Lancet 339: 572–575, 1992 [DOI] [PubMed] [Google Scholar]
- 33.Kielstein JT, Böger RH, Bode-Böger SM, Schäffer J, Barbey M, Koch KM, Frölich JC: Asymmetric dimethylarginine plasma concentrations differ in patients with end-stage renal disease: Relationship to treatment method and atherosclerotic disease. J Am Soc Nephrol 10: 594–600, 1999 [DOI] [PubMed] [Google Scholar]
- 34.Ferraro B, Galli F, Frei B, Kingdon E, Canestrari F, Rice-Evans C, Buoncristiani U, Davenport A, Moore KP: Peroxynitrite-induced oxidation of plasma lipids is enhanced in stable hemodialysis patients. Kidney Int 63: 2207–2213, 2003 [DOI] [PubMed] [Google Scholar]
- 35.Hasdan G, Benchetrit S, Rashid G, Green J, Bernheim J, Rathaus M: Endothelial dysfunction and hypertension in 5/6 nephrectomized rats are mediated by vascular superoxide. Kidney Int 61: 586–590, 2002 [DOI] [PubMed] [Google Scholar]
- 36.Annuk M, Zilmer M, Lind L, Linde T, Fellström B: Oxidative stress and endothelial function in chronic renal failure. J Am Soc Nephrol 12: 2747–2752, 2001 [DOI] [PubMed] [Google Scholar]
- 37.Miyazaki H, Matsuoka H, Itabe H, Usui M, Ueda S, Okuda S, Imaizumi T: Hemodialysis impairs endothelial function via oxidative stress: Effects of vitamin E-coated dialyzer. Circulation 101: 1002–1006, 2000 [DOI] [PubMed] [Google Scholar]
- 38.Fisher ND, Hollenberg NK: Aging and vascular responses to flavanol-rich cocoa. J Hypertens 24: 1575–1580, 2006 [DOI] [PubMed] [Google Scholar]
- 39.Heiss C, Dejam A, Kleinbongard P, Schewe T, Sies H, Kelm M: Vascular effects of cocoa rich in flavan-3-ols. JAMA 290: 1030–1031, 2003 [DOI] [PubMed] [Google Scholar]
- 40.Rammos C, Hendgen-Cotta UB, Sobierajski J, Bernard A, Kelm M, Rassaf T: Dietary nitrate reverses vascular dysfunction in older adults with moderately increased cardiovascular risk. J Am Coll Cardiol 63: 1584–1585, 2014 [DOI] [PubMed] [Google Scholar]
- 41.Dod HS, Bhardwaj R, Sajja V, Weidner G, Hobbs GR, Konat GW, Manivannan S, Gharib W, Warden BE, Nanda NC, Beto RJ, Ornish D, Jain AC: Effect of intensive lifestyle changes on endothelial function and on inflammatory markers of atherosclerosis. Am J Cardiol 105: 362–367, 2010 [DOI] [PubMed] [Google Scholar]
- 42.Han SH, Kang EW, Yoon SJ, Yoon HS, Lee HC, Yoo TH, Choi KH, Han DS, Kang SW: Combined vascular effects of HMG-CoA reductase inhibitor and angiotensin receptor blocker in non-diabetic patients undergoing peritoneal dialysis. Nephrol Dial Transplant 26: 3722–3728, 2011 [DOI] [PubMed] [Google Scholar]
- 43.Flammer AJ, Sudano I, Hermann F, Gay S, Forster A, Neidhart M, Künzler P, Enseleit F, Périat D, Hermann M, Nussberger J, Luscher TF, Corti R, Noll G, Ruschitzka F: Angiotensin-converting enzyme inhibition improves vascular function in rheumatoid arthritis. Circulation 117: 2262–2269, 2008 [DOI] [PubMed] [Google Scholar]
- 44.Kovesdy CP, Bleyer AJ, Molnar MZ, Ma JZ, Sim JJ, Cushman WC, Quarles LD, Kalantar-Zadeh K: Blood pressure and mortality in U.S. veterans with chronic kidney disease: A cohort study. Ann Intern Med 159: 233–242, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Weseler AR, Ruijters EJ, Drittij-Reijnders MJ, Reesink KD, Haenen GR, Bast A: Pleiotropic benefit of monomeric and oligomeric flavanols on vascular health--a randomized controlled clinical pilot study. PLoS One 6: e28460, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Brotman DJ, Bash LD, Qayyum R, Crews D, Whitsel EA, Astor BC, Coresh J: Heart rate variability predicts ESRD and CKD-related hospitalization. J Am Soc Nephrol 21: 1560–1570, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Schroeter H, Heiss C, Balzer J, Kleinbongard P, Keen CL, Hollenberg NK, Sies H, Kwik-Uribe C, Schmitz HH, Kelm M: (-)-Epicatechin mediates beneficial effects of flavanol-rich cocoa on vascular function in humans. Proc Natl Acad Sci U S A 103: 1024–1029, 2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Rhee EP, Souza A, Farrell L, Pollak MR, Lewis GD, Steele DJ, Thadhani R, Clish CB, Greka A, Gerszten RE: Metabolite profiling identifies markers of uremia. J Am Soc Nephrol 21: 1041–1051, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Ottaviani JI, Momma TY, Kuhnle GK, Keen CL, Schroeter H: Structurally related (-)-epicatechin metabolites in humans: Assessment using de novo chemically synthesized authentic standards. Free Radic Biol Med 52: 1403–1412, 2012 [DOI] [PubMed] [Google Scholar]
- 50.Schmidt S, Westhoff TH, Krauser P, Zidek W, van der Giet M: The uraemic toxin phenylacetic acid increases the formation of reactive oxygen species in vascular smooth muscle cells. Nephrol Dial Transplant 23: 65–71, 2008 [DOI] [PubMed] [Google Scholar]
- 51.Mao TK, van de Water J, Keen CL, Schmitz HH, Gershwin ME: Modulation of TNF-alpha secretion in peripheral blood mononuclear cells by cocoa flavanols and procyanidins. Dev Immunol 9: 135–141, 2002 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.