Abstract
Purpose
Low socioeconomic status (SES) is associated with adverse outcomes among unrelated donor hematopoietic stem cell transplant (HCT) recipients, but the biological mechanisms contributing to this health disparity are poorly understood. Therefore, we examined whether social environment affects expression of a stress-related gene expression profile known as the conserved transcriptional response to adversity (CTRA), which involves up-regulation of pro-inflammatory genes and down-regulation of genes involved in type I IFN response and antibody synthesis.
Experimental Design
We compared pre-transplant leukocyte CTRA gene expression between a group of 78 high vs. low SES recipients of unrelated donor HCT for acute myelogenous leukemia in first remission. Post hoc exploratory analyses also evaluated whether CTRA gene expression was associated with poor clinical outcomes.
Results
Peripheral blood mononuclear cells collected pre-HCT from low SES individuals demonstrated significant CTRA up-regulation compared to matched HCT recipients of high SES. Promoter-based bioinformatics implicated distinct patterns of transcription factor activity including increased CREB signaling and decreased IRF and GR signaling. High expression of the CTRA gene profile was also associated with increased relapse risk and decreased leukemia-free survival.
Conclusions
Low SES is associated with increased expression of the CTRA gene profile, and CTRA gene expression is associated with adverse HCT clinical outcomes. These findings provide a biologic framework within which to understand how social environmental conditions may influence immune function and clinical outcomes in allogeneic HCT.
Keywords: socioeconomic status, hematopoietic stem cell transplantation, gene expression, clinical outcomes
Introduction
Low socioeconomic status (SES), independent of race, has a negative impact on unrelated (1) and related (2) donor hematopoietic stem cell transplant (HCT) outcomes, including worse overall survival (OS) and higher transplant-related mortality (TRM). This effect has been demonstrated in multiple other solid tumor populations as well (3, 4). The biological, psychosocial, and environmental mechanisms accounting for the relationship between SES and outcomes in cancer are poorly understood. While health disparities between patients with varying SES levels are in part attributable to differences in health behaviors, biological processes resulting in altered central nervous system and immune activation also contribute (5). There is an increasing literature linking biobehavioral factors more prevalent in those of lower SES, such as chronic stressors, depressed mood, and lower levels of social integration and support (6), to higher levels of inflammatory burden (7), potentially through physiologic stress mechanisms (8). SES is inversely related to morbidity and mortality in a variety of diseases and populations, with inflammation implicated as a key biological pathway mediating this association (7, 9).
Adverse social conditions are associated with worse outcomes and cancer progression in several populations (10, 11), including HCT (12). However, while interactions between behavioral, neural, and immune function continue to be identified in other cancer populations (13), these pathways are just beginning to be explored among HCT recipients (14, 15). Social environmental influences on human health are well established, with previous studies indicating that circulating immune cells demonstrate a systematic shift in basal gene expression profiles during extended periods of stress, threat, or uncertainty (16–19). This shift, termed the “conserved transcriptional response to adversity” (CTRA), is characterized by increased expression of genes involved in inflammation (e.g., pro-inflammatory cytokines such as IL1B, IL6, IL8, and TNF) and decreased expression of genes involved in type I IFN antiviral responses (e.g., IFI-, OAS-, and MX- family genes) and IgG antibody synthesis (e.g., IGJ) (17, 20, 21).
There are several biological pathways through which CTRA gene expression dynamics might influence HCT-related biology and clinical outcomes. Inflammation plays a role in acute and chronic graft-versus-host-disease (a/cGVHD) (22), whose incidence has been described as increased among HCT recipients with low SES (2). Inflammation is also associated with other adverse health outcomes post-transplant including graft rejection and failure (23), severe infection (24), and increased symptom burden (25). Type I IFN and IgG1 antibody responses are also implicated in post-transplant complications (26, 27).
Previous research demonstrates increased CTRA gene expression profiles in circulating leukocytes from low SES individuals in healthy populations (7, 16); however, it is not known how such results might apply in the complex biologic milieu of HCT. Given previous work demonstrating the impact of SES on HCT outcomes, the present study sought to determine whether biologic factors, such as CTRA expression profiles, may partially explain the relationship between SES and outcomes. Therefore, we hypothesized that CTRA gene expression profiles from pre-transplant blood samples would be increased among HCT recipients of low SES. Post-hoc exploratory analyses evaluated whether SES and CTRA profiles were associated with adverse clinical outcomes. Given the extreme immunologic perturbations HCT recipients endure, it is important to understand how pre-existing social environmental factors such as SES may impact clinical outcomes.
Methods
Data Sources
The CIBMTR is a research affiliation of the International Bone Marrow Transplant Registry (IBMTR), Autologous Blood and Marrow Transplant Registry (ABMTR), and the National Marrow Donor Program Office of Research (NMDP) established in 2004 that comprises a voluntary working group of more than 450 transplantation centers worldwide that contribute detailed data on consecutive allogeneic and autologous hematopoietic cell transplantation to the Coordinating Center at the Medical College of Wisconsin in Milwaukee and the NMDP Coordinating Center in Minneapolis. Participating centers are required to report all transplants consecutively; compliance is monitored by on-site audits. Patients are followed longitudinally. Computerized checks for discrepancies, physicians’ review of submitted data, and on-site audits of participating centers ensure data quality. Observational studies conducted by the CIBMTR are performed in compliance with all applicable federal regulations pertaining to the protection of human research participants. Protected Health Information used in the performance of such research is collected and maintained in CIBMTR’s capacity as a Public Health Authority under the HIPAA Privacy Rule.
The CIBMTR collects data at two levels: Transplant Essential Data (TED) and Comprehensive Report Form (CRF) data. TED data include disease type, age, sex, pre-transplant disease stage and chemotherapy-responsiveness, date of diagnosis, graft type (bone marrow- and/or blood-derived stem cells), high-dose conditioning regimen, post-transplant disease progression and survival, development of a new malignancy, and cause of death. All CIBMTR teams contribute TED data. More detailed disease, and pre- and post-transplant clinical information are collected on a subset of registered patients selected for CRF data by a weighted randomization scheme. TED and CRF level data are collected pre-transplant, 100 days and six months post transplant, annually until year 6 post-transplant, and biannually thereafter until death.
Participants
The present sample of 78 participants was selected from the population evaluated in the study by Baker et al. that identified SES as an independent predictor of OS and TRM among unrelated donor allogeneic transplant recipients (1). The Baker et al. population included 6207 patients with acute or chronic leukemia or myelodysplastic syndrome who underwent an unrelated donor transplant in the United States using either a bone marrow (BM) or peripheral blood stem cell (PBSC) source with a myeloablative preparative regimen between 1995 and 2004 and who also had an available residential postal zip code. Patient income was estimated by the mean household income of their residential zip code from the 2004 U.S. Census. The package “Zip Code Deluxe” (28) was used to obtain income and location data by zip code. To achieve a homogeneous population for comparison purposes in the present study as well as to control for factors that may affect inflammation, our further inclusion criteria limited eligible subjects to adults ages 20–59 with a diagnosis of acute myelogenous leukemia (AML) in their first complete remission (CR1) who had an available pre-transplant whole blood sample and complete data for body mass index (BMI; height and weight), comorbid conditions, sex, age, race, Karnofsky performance status (KPS), and residential distance from transplant center. Pre-transplant/baseline blood samples are routinely collected and banked at the NMDP for all US allogeneic transplant recipients. The relevant samples for the current study were withdrawn and immediately sent to UCLA. Subjects with pre-existing comorbid autoimmune disorders were excluded to remove the potential for extreme influence on inflammation. Finally, we restricted the cohort to those individuals from the lowest SES quartile (<$34,700) and a comparison group of individuals from the highest SES quartile (>$56,300) (quartiles as per Baker et al., 2009). Patient race was reported by transplant centers and was categorized according to the U.S. Office of Management and Budget classification as White, African American, Hispanic, or Asian/Pacific-Islander. The initial plan was to analyze racial groups separately, however there were insufficient samples meeting the above criteria to warrant separate analysis, so the analysis is limited to whites only. This selection resulted in 100 individuals.
We then performed a matching procedure using the MatchIt R package (29) among these 100 eligible participants to ensure similar groups with respect to variables that could affect inflammation. We matched individuals from the lowest SES quartile with a comparison group of individuals from the highest SES quartile based on BMI (BMI<30 vs. BMI≥30) and age (closest within 10 years). Additional matching on other variables such as comorbid conditions, KPS, or gender was not feasible because of limited numbers. The final population available to be analyzed for gene transcription meeting the above criteria included 39 low SES and 39 high SES recipients matched for BMI group and age within 10 years. For the current sample size, the detectable fold changes in gene expression levels between patients with low- and high-SES were calculated based on the method described by Jung (30). We assume testing of 20,000 genes, of which approximately 200 are differentially expressed. We target 90% average sensitivity, or the expected proportion of differentially expressed genes which are identified as significant. The univariate significance level corresponding to this setting and a 5% false discovery rate is alpha=0.009. The current sample size on average will detect 90% of differentially expressed genes with fold changes of approximately 1.5 to 2 depending on the coefficients of variation. Further, the current sample of 39 in each group is larger than sample sizes that have evaluated gene expression as a function of psychosocial factors and yielded hundreds of differentially expressed genes that generate statistically significant results in higher-order bioinformatics (18, 31). All study procedures were approved by the institutional review board of the Medical College of Wisconsin.
Transcriptome Analysis
Genome-wide transcriptional profiling was performed on isolated peripheral blood mononuclear cells (PBMCs) from all 78 participants in one batch. Assays were conducted as previously described (32), with PBMCs isolated by density gradient centrifugation and total RNA extracted (RNeasy; Qiagen), tested for suitable mass (Nanodrop ND1000) and integrity (Bioanalyzer; Agilent), and converted to fluorescent cRNA for hybridization to Illumina Human HT-12 v4 BeadArrays following the manufacturer’s standard protocol in the University of California, Los Angeles, Neuroscience Genomics Core Laboratory. Gene expression values were quantile-normalized, log2-transformed, and subject to general linear model analyses relating the expression of each assayed gene to SES (0 = high SES, 1 = low SES; results are interpretable in terms of the effects of low SES) while also controlling for age, sex, BMI, and presence of pre-defined co-existing medical conditions at the time of preparative regimen. Primary analyses tested an a priori-defined contrast score representing the 53-gene CTRA profile of up-regulated expression of 19 pro-inflammatory genes (IL1A, IL1B, IL6, IL8, TNF, PTGS1, PTGS2, FOS, FOSB, FOSL1, FOSL2, JUN, JUNB, JUND, NFKB1, NFKB2, REL, RELA, and RELB) and down-regulated expression of 31 genes involved in type I interferon (IFN) responses (GBP1, IFI16, IFI27, IFI27L1-2, IFI30, IFI35, IFI44, IFI44L, IFI6, IFIH1, IFIT1-3, IFIT5, IFIT1L, IFITM1-3, IFITM4P, IFITM5, IFNB1, IRF2, IRF7-8, MX1-2, OAS1-3, and OASL) and three genes involved in antibody synthesis (IGJ, IGLL1, and IGLL3) (17, 20, 21, 33) to evaluate whether low SES might be associated with increased expression of this CTRA profile. Contrast coefficient-weighted association statistics were averaged to summarize the magnitude of association over the entire CTRA gene set, and standard errors were derived from 200 cycles of bootstrap resampled residual vectors (to account for potential correlation among residuals across genes) (34).
To identify transcription control pathways that may mediate observed transcriptional differences, initial “low-level” genome-wide analyses identified all transcripts showing a model-adjusted point estimate of ≥20% difference in expression between low- vs. high-SES HCT recipients. Those putatively associated genes were subject to Transcription Element Listening System (TELiS) promoter-based bioinformatic analysis (35) to assess activity of NF-κB, AP-1, IRF, and CREB and GR family transcription factors previously linked to CTRA transcriptional dynamics (TRANSFAC V$CREL_01, V$AP1_Q4, V$ISRE_01, V$CREB_Q4, V$GR_Q6) (35), with results averaged over nine parametric variations of MatInspector scan stringency and promoter length (35). To ensure that results were not confounded by individual differences in the prevalence of specific leukocyte subtypes within the PBMC pool (36), analyses also controlled for the prevalence of transcripts marking T lymphocyte subsets (CD3D, CD3E, CD4, CD8A), B lymphocytes (CD19), natural killer cells (CD16/FCGR3A, CD56/NCAM1), and monocytes (CD14) (18). Transcript origin analysis (TOA) was applied to the low-level association data to identify the specific PBMC subtypes mediating the observed differences in gene expression, as previously described (32). Low-level transcript-phenotype associations were estimated solely as inputs into high-level TELiS and TOA gene set expression analyses and are not tested for statistical reliability at the level of individual genes.
Clinical Outcome Study Definitions
While the aim of the primary analysis was to understand the relationship between SES and CTRA gene expression, exploratory secondary analyses assessed the impact of SES and CTRA expression on clinical outcomes including neutrophil engraftment at Day +28, acute and chronic GVHD, TRM, relapse, leukemia-free survival (LFS), and OS. Neutrophil engraftment at Day +28 was defined as the presence or absence of absolute neutrophil count (ANC) > 0.5 × 109/L sustained for three consecutive days. Patients were assessed for aGVHD and cGVHD by standard criteria (37, 38). TRM was defined as death in complete remission. Relapse was defined as disease recurrence at any site. LFS was defined as survival in complete remission after HCT. For OS, death from any cause was considered an event.
HLA matching status was categorized as well-matched, partially matched, or mismatched based on the NMDP classification of HLA matching status that allows adequate adjustment for donor-recipient HLA compatibility while accounting for best available resolution of typing. Well-matched patients had no identified mismatches at HLA-A, -B, -C, and –DRB1 with low-intermediate- or high-resolution data available at HLA-A, -B and high-resolution –DRB1. Partially matched patients had a single locus mismatch at any of the 4 loci and/or missing HLA-C data. Mismatched patients had 2 or more allele or antigen mismatches.
Statistical Analysis of Patient Characteristics and Outcomes
The chi-square statistic was used to compare categorical patient, disease, and HCT-related variables and the Kruskal-Wallis test was used to compare continuous variables. Due to the dramatically different variances among gene expression values (often >10-fold range), we stabilized their variance by converting each gene expression value into a single z-score. Z-scores for each individual were then averaged over the CTRA genes to compute a single gene expression composite score for each individual. These values were then split at the median and dichotomized as low vs. high CTRA expression, as no biologically-based cutpoints have yet been defined. CTRA profiles were unable to be compared to historic non-diseased populations as the normalization process performed within a given set of assays removes differences between sets of assayed samples. Clinical outcome variables were compared between the low vs. high SES groups as well as the low vs. high CTRA expression groups. Probabilities of neutrophil recovery, acute and chronic GVHD, TRM, and relapse were calculated by the cumulative-incidence function method and compared between groups using Gray’s test. Probabilities of LFS and OS were calculated using the Kaplan-Meier method and compared between groups using the log-rank test. Multivariate analyses were not performed due to sample size limitations. We also conducted stratified log-rank tests to account for the potential impact of matching, but the results were similar to the unstratified tests so they are not reported.
Results
Patient Characteristics
Seventy-eight HCT recipients from the population sampled met study inclusion and matching criteria. Table 1 describes patient, disease, and treatment characteristics for low vs. high SES. All patients had AML in first CR. Only distance to transplant center was significantly different between the two groups, with individuals of lower SES significantly more likely to live further away from the transplant center (p<0.001). Table 2 describes the cohort’s characteristics by low vs. high CTRA expression level. Individuals with lower levels of CTRA gene expression were more likely to be older (p=0.04) and have received a higher infused cell dose if receiving a peripheral blood HCT (p=0.02).
Table 1.
Patient, disease, and treatment characteristics by low vs. high-socioeconomic status
| A | B | C | D |
|---|---|---|---|
| High SES (%) | Low SES (%) | p-value | |
| Age, median (range), years | 44 (21–59) | 40 (21–59) | 0.26 |
| Age | 0.71 | ||
| 20–29 | 6 (15) | 10 (26) | |
| 30–39 | 10 (26) | 10 (26) | |
| 40–49 | 16 (41) | 13 (33) | |
| 50–59 | 7 (18) | 6 (15) | |
| Sex | 0.36 | ||
| Male | 21 (54) | 17 (44) | |
| Female | 18 (46) | 22 (56) | |
| Median income, 2000* | 61814 | 30259 | <.001 |
| Distance to transplant center | <.001 | ||
| <17 miles | 13 (33) | 8 (21) | |
| 17–55 miles | 15 (38) | 4 (10) | |
| 55–150 miles | 3 (8) | 17 (44) | |
| >150 miles | 8 (21) | 10 (26) | |
| KPS | 0.08 | ||
| >=90 | 32 (82) | 29 (74) | |
| <90 | 2 (6) | 8 (20) | |
| Missing | 5 (12) | 2 (6) | |
| Comorbid conditions | 1 | ||
| No | 21 (54) | 21 (54) | |
| Yes | 18 (46) | 18 (46) | |
| BMI, median (range), kg/m^2 | 26 (20–41) | 26 (18–51) | 0.9 |
| BMI, kg/m^2 | 0.47 | ||
| 0 < 18.5 | 0 (0) | 2 (5) | |
| 18.5 – 24.9 | 11 (28) | 14 (36) | |
| 25 – 29.9 | 19 (48) | 14 (36) | |
| >=30 | 9 (24) | 9 (23) | |
| CTRA profile expression | |||
| High | 14 (36) | 25 (64) | 0.02 |
| Low | 25 (64) | 14 (36) | |
| Cytogenetics | 0.8 | ||
| Good risk | 1 (2) | 0 (0) | |
| Intermediate risk | 19 (48) | 18 (46) | |
| Poor/adverse risk | 11 (28) | 10 (26) | |
| Unknown | 8 (20) | 11 (28) | |
| HLA match status | 0.59 | ||
| Well matched | 23 (48) | 24 (62) | |
| Partially matched | 9 (24) | 11 (28) | |
| Mismatched | 7 (18) | 4 (10) | |
| Race match (donor/recipient) | 0.83 | ||
| Match | 31 (80) | 33 (84) | |
| Mismatch | 4 (10) | 3 (8) | |
| Unknown | 4 (10) | 3 (8) | |
| Donor age, median (range), years | 33 (21–56) | 37 (20–56) | 0.5 |
| Donor age at transplant, years | 0.45 | ||
| 18–29 | 14 (36) | 11 (28) | |
| 30–39 | 13 (33) | 15 (38) | |
| 40–49 | 6 (15) | 10 (26) | |
| >=50 | 6 (15) | 3 (8) | |
| Sex match (donor/recipient) | 0.16 | ||
| Male/male | 12 (30) | 15 (38) | |
| Male/female | 10 (26) | 12 (30) | |
| Female/male | 9 (24) | 2 (6) | |
| Female/female | 8 (20) | 10 (26) | |
| CMV match (donor/recipient) | 0.82 | ||
| Negative/negative | 12 (31) | 14 (36) | |
| Negative/positive | 14 (36) | 11 (28) | |
| Positive/negative | 5 (13) | 5 (13) | |
| Positive/positive | 8 (20) | 8 (20) | |
| Unknown | 0 (0) | 1 (3) | |
| Year of transplant | 0.64 | ||
| 1995–1999 | 13 (34) | 15 (38) | |
| 2000–2004 | 26 (66) | 24 (62) | |
| Conditioning regimen | 0.25 | ||
| Bu + Cy +/− other | 7 (18) | 5 (13) | |
| Cy + TBI +/− other | 28 (72) | 26 (67) | |
| TBI +/− other | 0 (0) | 4 (10) | |
| Flu +/− other | 3 (8) | 4 (10) | |
| Other | 1 (2) | 0 (0) | |
| GVHD prophylaxis | 0.46 | ||
| T-cell depletion | 7 (18) | 6 (15) | |
| CsA + MTX +/− others | 13 (33) | 18 (46) | |
| CsA +/− others | 0 (0) | 2 (6) | |
| Tacrolimus + MTX | 15 (38) | 10 (26) | |
| Tacrolimus | 4 (10) | 3 (8) | |
| Graft type | 1 | ||
| Bone marrow | 28 (72) | 28 (72) | |
| Peripheral blood | 11 (28) | 11 (28) | |
| Infused BM cell dose | 0.18 | ||
| > 2 × 10^8 | 17 (39) | 12 (43) | |
| <= 2 × 10^8 | 11 (61) | 16 (57) | |
| Missing | 0 (0) | 0 (0) | |
| Infused PB cell dose | 0.45 | ||
| > 5 × 10^8 | 6 (27) | 9 (1) | |
| <= 5 × 10^8 | 3 (55) | 1 (9) | |
| Missing | 2 (18) | 9(1) | |
| Time from diagnosis to transplant, median (range), months | 5 (3–17) | 6 (3–12) | 0.82 |
| Donor search time, median (range), months | |||
| Diagnosis to preliminary search | 2 (<1–10) | 2 (<1–10) | 0.62 |
| Preliminary search to formal search | <1 (<1–12) | <1 (0–3) | 0.29 |
| Formal search to transplant | 3 (2–11) | 3 (2–6) | 0.25 |
| Follow-up of survivors, median (range), months | 28 (12–86) | 36 (12–132) | 0.53 |
| Cause of Death | 0.43 | ||
| Infection | 4 (10) | 6 (15) | |
| Interstitial pneumonia | 4 (10) | 2 (6) | |
| aGVHD | 3 (8) | 4 (10) | |
| Recurrence or persistence of primary disease | 3 (8) | 6 (16) | |
| Organ failure | 5 (12) | 5 (12) | |
| Hemorrhage | 4 (10) | 0 (0) | |
| Other | 16 (42) | 16 (41) |
BM, bone marrow; Bu, busulfan; CMV, cytomegalovirus; CSA, cyclosporine; CTRA, conserved transcriptional response to adversity; Cy, cyclophosphamide; Flu, fludarabine; GVHD, graft-versus-host disease; HLA, human leukocyte antigen; KPS, Karnofsky Performance Status; MTX, methotrexate; PB, peripheral blood; TBI, total body irradiation
Based on 2004 Census tract data linking income to residential ZIP code.
Table 2.
Patient, disease, and treatment characteristics by low vs. high-CTRA gene expression profile
| A | B | C | D |
|---|---|---|---|
| High CTRA (%) | Low CTRA (%) | p-value | |
| Age, median (range), years | 37 (21–59) | 44 (21–59) | 0.04 |
| Age | 0.16 | ||
| 20–29 | 10 (26) | 6 (15) | |
| 30–39 | 13 (33) | 7 (18) | |
| 40–49 | 11 (28) | 18 (46) | |
| 50–59 | 5 (13) | 8 (21) | |
| Sex | 0.65 | ||
| Male | 18 (46) | 20 (52) | |
| Female | 21 (54) | 19 (48) | |
| Median income, 2000* | 32300 | 58257 | 0.02 |
| Distance to transplant center | 0.58 | ||
| <17 miles | 12 (33) | 9 (23) | |
| 17–55 miles | 7 (18) | 12 (33) | |
| 55–150 miles | 11 (28) | 9 (23) | |
| >150 miles | 9 (23) | 9 (23) | |
| KPS | 0.38 | ||
| >=90 | 33 (84) | 28 (72) | |
| <90 | 4 (10) | 6 (16) | |
| Missing | 2 (6) | 5 (12) | |
| Comorbid conditions | 0.17 | ||
| No | 24 (62) | 18 (46) | |
| Yes | 15 (38) | 21 (54) | |
| BMI, median (range), kg/m^2 | 27 (18–51) | 26 (20–44) | 0.9 |
| BMI, kg/m^2 | 0.68 | ||
| 0 < 18.5 | 2 (5) | 0 (0) | |
| 18.5 – 24.9 | 13 (33) | 12 (30) | |
| 25 – 29.9 | 16 (41) | 17 (44) | |
| >=30 | 8 (21) | 10 (26) | |
| SES | |||
| High | 14 (36) | 25 (64) | 0.02 |
| Low | 25 (64) | 14 (36) | |
| Cytogenetics | 0.64 | ||
| Good risk | 0 (0) | 1 (2) | |
| Intermediate risk | 19 (48) | 18 (46) | |
| Poor/adverse risk | 9 (24) | 12 (30) | |
| Unknown | 11 (28) | 8 (20) | |
| HLA match status | 0.95 | ||
| Well matched | 23 (59) | 24 (62) | |
| Partially matched | 10 (26) | 10 (26) | |
| Mismatched | 6 (15) | 5 (13) | |
| Race match (donor/recipient) | 0.87 | ||
| Match | 32 (82) | 32 (82) | |
| Mismatch | 3 (8) | 4 (10) | |
| Unknown | 4 (10) | 3 (8) | |
| Donor age, median (range), years | 37 (20–56) | 35 (21–56) | 0.97 |
| Donor age at transplant, years | 0.48 | ||
| 18–29 | 14 (36) | 11 (28) | |
| 30–39 | 11 (28) | 17 (44) | |
| 40–49 | 10 (26) | 6 (15) | |
| >=50 | 4 (10) | 5 (13) | |
| Sex match (donor/recipient) | 0.78 | ||
| Male/male | 13 (33) | 14 (36) | |
| Male/female | 13 (33) | 9 (23) | |
| Female/male | 5 (13) | 6 (15) | |
| Female/female | 8 (21) | 10 (26) | |
| CMV match (donor/recipient) | 0.71 | ||
| Negative/negative | 14 (36) | 12 (33) | |
| Negative/positive | 11 (28) | 14 (36) | |
| Positive/negative | 6 (15) | 4 (10) | |
| Positive/positive | 7 (18) | 9 (23) | |
| Unknown | 1 (3) | 0 (0) | |
| Year of transplant | 0.64 | ||
| 1995–1999 | 15 (38) | 13 (33) | |
| 2000–2004 | 24 (62) | 26 (67) | |
| Conditioning regimen | 0.74 | ||
| Bu + Cy +/− Other | 5 (12) | 7 (18) | |
| Cy + TBI +/− other | 28 (72) | 26 (67) | |
| TBI +/− other | 3 (8) | 1 (3) | |
| Flu +/− other | 3 (8) | 4 (10) | |
| Other | 0 (0) | 1 (3) | |
| GVHD prophylaxis | 0.84 | ||
| T-cell depletion | 7 (18) | 6 (15) | |
| CSA + MTX +/− others | 17 (44) | 14 (36) | |
| CSA +/− others | 1 (3) | 1 (3) | |
| Tacrolimus + MTX | 12 (31) | 13 (33) | |
| Tacrolimus | 2 (5) | 5 (12) | |
| Graft type | 0.31 | ||
| Bone marrow | 30 (77) | 26 (67) | |
| Peripheral blood | 9 (31) | 13 (33) | |
| Infused BM cell dose | 0.41 | ||
| > 2 × 10^8 | 14 (47) | 15 (58) | |
| <= 2 × 10^8 | 16 (53) | 11 (42) | |
| Missing | 0 (0) | 0 (0) | |
| Infused PB cell dose | 0.02 | ||
| > 5 × 10^8 | 4 (44) | 11 (85) | |
| <= 5 × 10^8 | 4 (44) | 0 (0) | |
| Missing | 1 (11) | 2 (15) | |
| Time from diagnosis to transplant, median (range), months | 6 (3–17) | 5 (3–15) | 0.28 |
| Donor search time, median (range), months | |||
| Diagnosis to preliminary search | 2 (<1–10) | 2 (<1–10) | 0.45 |
| Preliminary search to formal search | <1 (0–12) | <1 (0–3) | 0.44 |
| Formal search to transplant | 3 (2–8) | 3 (2–11) | 0.45 |
| Follow-up of survivors, median (range), months | 28 (12–81) | 36 (23–132) | 0.52 |
| Cause of Death | 0.18 | ||
| Infection | 5 (13) | 5 (13) | |
| Interstitial pneumonia | 3 (8) | 3 (8) | |
| aGVHD | 3 (8) | 4 (10) | |
| Recurrence or persistence of primary disease | 8 (21) | 1 (3) | |
| Organ failure | 5 (13) | 5 (13) | |
| Hemorrhage | 3 (8) | 1 (3) | |
| Other | 12 (31) | 20 (51) |
BM, bone marrow; Bu, busulfan; CMV, cytomegalovirus; CSA, cyclosporine; CTRA, conserved transcriptional response to adversity; Cy, cyclophosphamide; Flu, fludarabine; GVHD, graft-versus-host disease; HLA, human leukocyte antigen; KPS, Karnofsky Performance Status; MTX, methotrexate; PB, peripheral blood; TBI, total body irradiation
Based on 2004 Census tract data linking income to residential ZIP code.
CTRA Transcriptome Profile
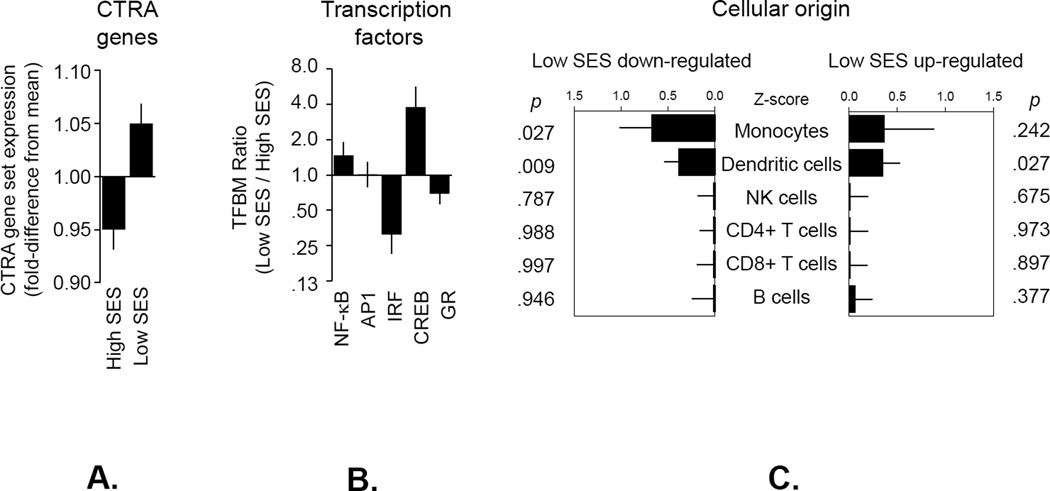
CTRA gene expression varied significantly as a function of SES, with HCT recipients of lower SES expressing the CTRA gene profile significantly more than those of high SES (Figure 1A) (p=0.009). Follow-up analysis of specific gene subsets identified down-regulated expression of the type I IFN response genes as the primary driver of the overall reduction in CTRA gene expression in low SES (p=0.012); pro-inflammatory and antibody genes were not significantly associated with low SES (p=0.418 and 0.481, respectively). The magnitude of the within-sample differences (e.g., by SES) are similar to those observed in other studies (7, 16), though direct comparison is not valid as previously described.
Figure 1.
Expression of the CTRA gene set, transcription control pathways and cellular origin. (A) Linear model-based estimates of fold-difference from the mean in expression in a 53-gene CTRA contrast score in PBMCs from individuals with low vs. high SES (adjusting for age, gender, race, BMI, number of comorbid conditions at time of transplant, and mRNA markers of major leukocyte subsets). Genes showing ≥ 20% difference in expression between HCT recipients of low vs. high SES were tested for (B) differential activity of specific transcription factors as indicated by TELiS analysis of transcription factor-binding motifs (TFBM) in proximal promoter sequences of up- vs. down-regulated genes (35) and (C) PBMC cell type of origin as indicated by TOA cell-type diagnosticity z-scores (32). Error bars indicate standard error.
Transcription Control Pathways
To assess the role of immunoregulatory transcription factors previously implicated in CTRA-related gene expression (pro-inflammatory NF-κB, activator protein 1 (AP-1), and cAMP response element-binding protein (CREB) factors as well as interferon response factor (IRF) and glucocorticoid receptor (GR) activity) (17, 20, 21), we applied TELiS promoter-based bioinformatics analyses (35) to all genes showing ≥20% difference in average expression between the low vs. high SES groups (Figure 1B). Consistent with findings from the CTRA analyses, results showed significantly decreased activity of IRF transcription factors (the primary mediators of Type I interferon response; p=0.003) but no significant alterations in NF-κB or AP-1 activity in the low SES group. With respect to neural/endocrine processes that might regulate these immunobiologic pathways, we also saw indications of increased CREB activity (which mediates β-adrenergic signaling from the sympathetic nervous system (17); p<0.001) and decreased activity of the GR (decreased activity often occurs secondary to stress-induced desensitization of the GR via β-adrenergic signaling (17); p=0.068) in this group that was not statistically significant.
Cellular Origins
To determine whether the transcriptional correlates of SES might occur within the same leukocyte subpopulations (i.e., monocytes, dendritic cells, and B lymphocytes) previously shown to mediate the CTRA transcriptional effects of other adverse life circumstances (20, 21, 31, 32), we conducted transcript origin analysis (TOA) (32) on differentially expressed genes (Figure 1C). These results indicate that differentially expressed genes derived predominately from dendritic cells (both up-regulated and down-regulated genes) and from monocytes (down-regulated genes). To ensure that differential prevalence of these subsets did not confound analyses of transcription factor activity, all CTRA gene set analyses and transcription factor bioinformatics results reported above were adjusted for leukocyte subset marker transcripts (CD3D/E, CD4, CD8A, CD19, CD16/FCGR3A, CD56/NCAM1, and CD14).
Clinical Outcomes
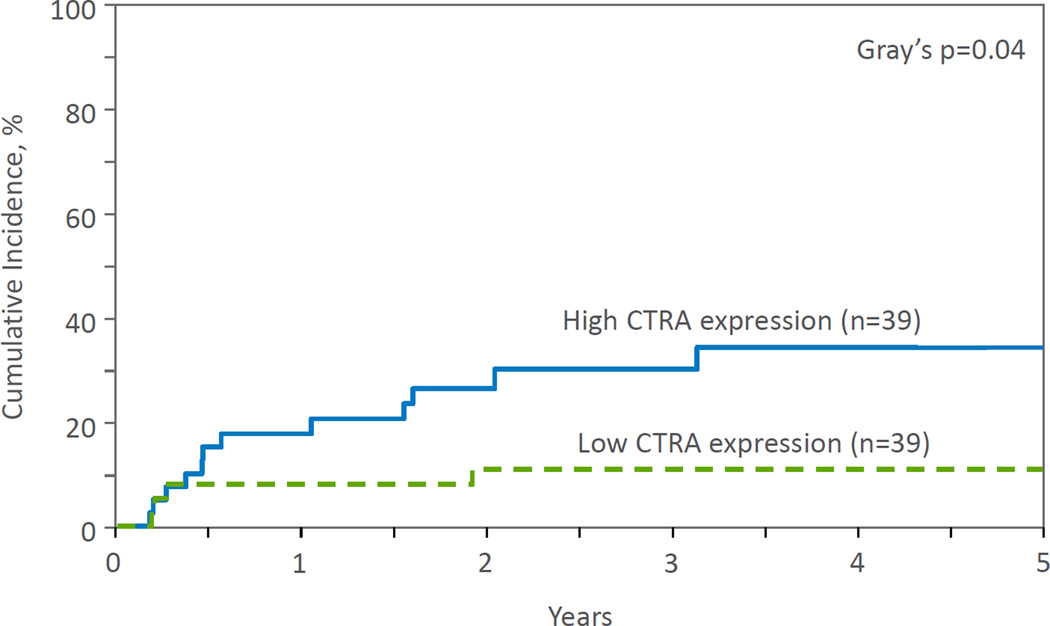
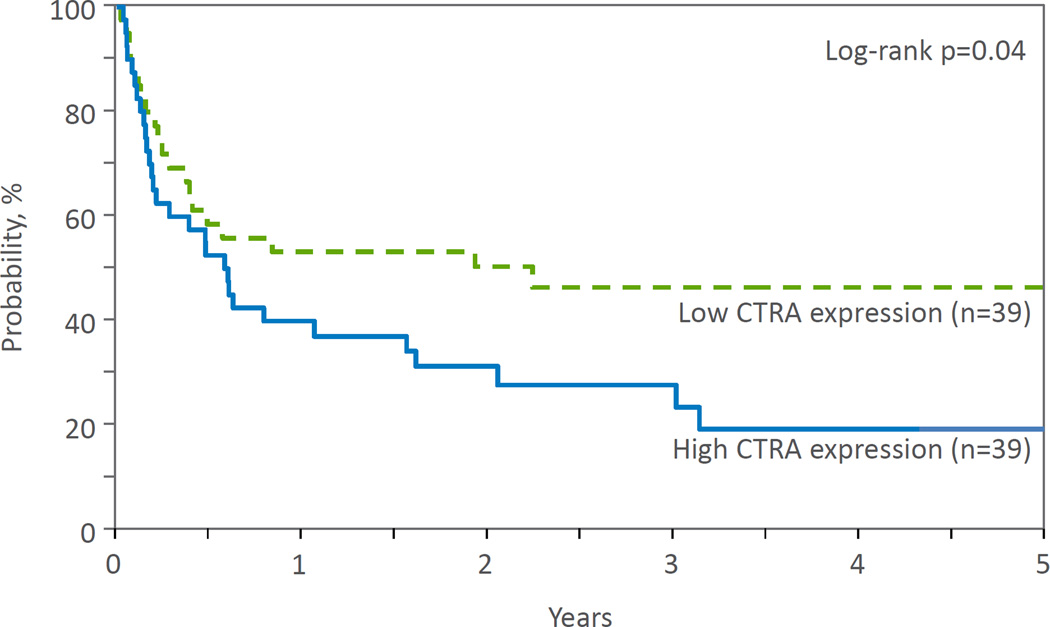
Increased expression of the CTRA gene profile was significantly associated with increased overall relapse (p=.04; Figure 2) and decreased overall LFS (p=.04; Figure 3) as compared to the low CTRA group in unadjusted analyses (Table 3). Relapse at 3 years was 31% and 11% in the high and low CTRA groups, respectively, while LFS at 3 years was 26% and 45%. Exploratory analyses demonstrated no significant differences between low vs. high SES groups on any of the clinical outcome variables.
Figure 2.
Cumulative incidence of relapse by CTRA expression.
Figure 3.
Probability of LFS by CTRA expression.
Table 3.
Clinical outcomes as a function of high- vs. low-CTRA gene expression
| A | B | C | D |
|---|---|---|---|
| Outcome | High CTRA expression |
Low CTRA expression |
p-value |
| [% of cohort, 95% CI] |
[% of cohort, 95% CI] |
||
| N=39 | N=39 | ||
| Neutrophil recovery @ 100 days | 92 (83–98) | 85 (72–94) | 0.27 |
| aGVHD @ 100 days | 49 (33–64) | 62 (46–76) | 0.25 |
| cGVHD | 0.08 | ||
| @ 1 year | 30 (16–45) | 35 (21–51) | 0.61 |
| @ 3 years | 30 (16–45) | 47 (31–63) | 0.13 |
| TRM | 0.61 | ||
| @ 100 days | 33 (20–49) | 23 (12–38) | 0.33 |
| @ 1 year | 44 (29–59) | 40 (25–56) | 0.74 |
| @ 3 years | 44 (29–59) | 44 (28–60) | 0.98 |
| Relapse | 0.04 | ||
| @ 1 year | 18 (8–31) | 8 (2–19) | 0.2 |
| @ 3 years | 31 (17–46) | 11 (3–23) | 0.04 |
| LFS | 0.04 | ||
| @ 1 year | 38 (24–54) | 52 (36–68) | 0.23 |
| @ 3 years | 26 (13–41) | 45 (29–62) | 0.09 |
| OS | 0.09 | ||
| @ 1 year | 44 (29–59) | 54 (38–69) | 0.36 |
| @ 3 years | 25 (12–42) | 47 (31–63) | 0.14 |
CTRA, conserved transcriptional response to adversity; GVHD, graft-versus-host disease (a=acute, c=chronic); LFS, leukemia-free survival; OS, overall survival; TRM, transplant-related mortality
Discussion
The results of this study show that low SES is associated with a marked activation of the CTRA gene expression profile in leukocytes from hematologic cancer patients receiving HCTs from unrelated donors. These results are consistent with studies of other stressed populations suggesting that extended exposure to life adversity is associated with up-regulated expression of the CTRA gene profile. While increased CTRA gene expression has been observed among healthy individuals of low SES (7, 16), these are the first data to demonstrate the robustness of this relationship in an immunologically compromised and diseased population. These data are the first to suggest a potential neuroimmune mechanism for the previously reported adverse clinical outcomes among HCT recipients of lower SES (1). Descriptive analyses suggest that greater CTRA gene expression is associated with increased relapse and decreased LFS, which is the first indication in the literature to directly link the CTRA profile with clinical outcomes.
The provocative differences between CTRA among those with low vs. high SES may explain differences in outcomes, and could lead to mechanistic targets for future interventions. β-Adrenergic signaling is known to affect the transcription pathways differentially expressed between low vs. high SES groups in this study, including up-regulation of CREB activity, inhibition of IRF signaling, and desensitization of the GR (16). Therefore, this profile of results is consistent with the possibility that increased sympathetic nervous system activity associated with exposure to chronic stress may contribute to the differential gene expression profiles observed. β-Adrenergic signaling may regulate multiple cellular processes contributing to cancer progression (39). Retrospective epidemiologic studies have linked the use of β-adrenergic antagonists (β-blockers) to reduced rates of progression for several solid tumors (40–42). Preclinical pharmacologic and biomarker studies in animals support translation of β-blockade as a novel adjuvant to existing therapeutic strategies in clinical oncology, linking the use of β-blockers with reduced disease progression (16, 40–46). This effect has been demonstrated in malignant hematologic cells as well (16, 45). The identification of altered gene transcription patterns among HCT recipients exposed to varying levels of chronic stress may provide a biological target for the novel use of β-blockers in the HCT setting.
In addition to β-adrenergic signaling, prostaglandin synthesis may be another mechanistic drug target for stress-associated cancer control. Animal models demonstrate that COX-2 inhibitors attenuate tumor-promoting effects of surgery (43, 47), though gene expression studies have not been done in this setting. Finally, behavioral interventions targeted at stress management can modulate leukocyte transcriptional activity among cancer patients (20). The CTRA gene profile may represent a useful molecular biomarker for assessing pharmacologic or behavioral intervention treatment response.
Though this study was not powered to examine the relationship between CTRA expression and clinical outcomes in multivariate analysis, the data are provocative in identifying a molecular signature that could serve as a cumulative stress biomarker. Such a biomarker may be useful in stratifying disease risk. Social and physical stressors other than SES may affect overall gene transcription as well as specific expression of the CTRA gene profile; this, in addition to low power, may explain why CTRA expression, but not SES, was associated with clinical outcomes in our exploratory analyses. While SES was significantly associated with OS and TRM in the Baker et al. study (1), this finding was not replicated in the present study either due to lack of statistical power or to the fact that, by design, the present sample was purposefully selected as a homogeneous subset and therefore not representative of the Baker cohort. The significant age difference between the high vs. low CTRA expression groups is contrary to what might be expected given the known positive association between aging and inflammation. If this confounded the present findings it should have been in the direction of the null hypothesis. Further studies assessing clinical outcomes while controlling for age as well as other patient-, disease-, and treatment-related variables that impact HCT outcomes (including cytogenetics; HLA match; donor age; donor/recipient match on race, gender, and CMV status; year of transplant; conditioning regimen; GVHD prophylaxis; graft type; cell dose; and time from diagnosis to transplant) are needed.
This study found differences in relapse and LFS, whereas TRM and OS were different between SES groups in the Baker study (1). This difference may be explained by the subset selection or the different independent variables used for analysis (CTRA rather than SES). SES is only one etiology of chronic stress, and the biological sequelae may be similar but not identical to that predicted by an overall stress biomarker. The significant clinical outcomes in the present analysis are, however, consistent with the pathophysiology of β-adrenergic mediated tumor progression as previously discussed. Finally, the sample size in this subgroup was not powered to evaluate clinical outcomes, therefore these interesting findings require validation in adequately powered multivariate analyses.
This study’s findings are limited in several respects. These results are from a cross-sectional analysis, and it is possible the observed associations are attributable to factors other than SES. This study focuses on a one-time assessment of leukocyte gene expression in a Caucasian-only sample; future studies are needed to determine whether the same gene expression effects exist in other races, vary between racial groups, change throughout the transplant process, or are affected by social environmental factors experienced by the donor. The present findings are from extreme SES quartiles and may overestimate the magnitude of SES-related differences within the population as a whole; robustness of this relationship should be confirmed in future studies assessing SES as a continuous variable. We were unable to control for smoking status, though previous research has failed to demonstrate an effect of smoking status on outcomes among unrelated donor recipients for chronic myelogenous leukemia (48). Since inflammation is a large component of the CTRA profile, and low SES is associated with higher levels of smoking (49), it is feasible this could have artificially enhanced the relationship between SES and CTRA expression. However, IFN and antibody expression are also significant contributors to the overall CTRA profile and there is substantially less research available to suggest a significant relationship between IFN/antibody responses and SES or smoking. Further, our results are consistent with those from other studies of social adversity that were able to control for smoking (16–19). Finally, it will also be important to validate the present bioinformatic indications of monocyte and dendritic cell involvement in future studies using physical isolation of cell subsets.
This study demonstrates significant SES-related differences in immune system gene expression among unrelated donor HCT recipients consistent with that of other populations confronting significant life adversity (46). Given prior evidence for worse overall survival and transplant-related mortality among low SES transplant recipients (1) and possible association between CTRA gene expression and relapse and leukemia-free survival, these findings provide a molecular framework within which to understand potential social environmental influences on immune function and clinical outcomes in the setting of cancer and HCT.
Statement of Translational Relevance.
Low socioeconomic status (SES) accounts for a 15–21% difference in overall survival and transplant-related mortality among HCT recipients as compared to recipients of high SES. SES-related health disparities persist after controlling for differences in access to care and health behaviors. Lifestyle and stress associated with low SES can activate psychobiological processes leading to altered neural, endocrine and immune activation. Here, we identify that pre-transplant gene expression profiles of allogeneic HCT recipients from lower SES backgrounds display significantly increased expression of the conserved transcriptional response to adversity (CTRA) gene expression profile. Further, our post-hoc analyses indicate that this increased CTRA expression is associated with a greater likelihood of relapse and decreased leukemia-free survival. These transcriptomic differences provide useful therapeutic biological targets to improve outcomes in socially at-risk populations through targeted pharmacologic intervention.
Acknowledgements
The CIBMTR is supported by Public Health Service Grant/Cooperative Agreement 5U24-CA076518 from the National Cancer Institute (NCI), the National Heart, Lung and Blood Institute (NHLBI) and the National Institute of Allergy and Infectious Diseases (NIAID); a Grant/Cooperative Agreement 5U10HL069294 from NHLBI and NCI; a contract HHSH250201200016C with Health Resources and Services Administration (HRSA/DHHS); two Grants N00014-13-1-0039 and N00014-14-1-0028 from the Office of Naval Research; and grants from *Actinium Pharmaceuticals; Allos Therapeutics, Inc.; *Amgen, Inc.; Anonymous donation to the Medical College of Wisconsin; Ariad; Be the Match Foundation; *Blue Cross and Blue Shield Association; *Celgene Corporation; *Chimerix, Inc.; Fred Hutchinson Cancer Research Center; Fresenius-Biotech North America, Inc.; *Gamida Cell Teva Joint Venture Ltd.; Genentech, Inc.; *Gentium SpA; Genzyme Corporation; *Gilead Sciences, Inc.; GlaxoSmithKline; Health Research, Inc. Roswell Park Cancer Institute; HistoGenetics, Inc.; Incyte Corporation; Jeff Gordon Children’s Foundation; Kiadis Pharma; The Leukemia & Lymphoma Society; Medac GmbH; The Medical College of Wisconsin; Merck & Co, Inc.; *Mesoblast; *Millennium: The Takeda Oncology Co.; *Milliman USA, Inc.; *Miltenyi Biotec, Inc.; National Marrow Donor Program; Neovii Biotech NA, Inc.; Novartis Pharmaceuticals Corporation; Onyx Pharmaceuticals; Optum Healthcare Solutions, Inc.; Osiris Therapeutics, Inc.; Otsuka America Pharmaceutical, Inc.; Perkin Elmer, Inc.; *Remedy Informatics; *Sanofi US; Seattle Genetics; Sigma-Tau Pharmaceuticals; Soligenix, Inc.; Spectrum Pharmaceuticals, Inc.; St. Baldrick’s Foundation; StemCyte, A Global Cord Blood Therapeutics Co.; Stemsoft Software, Inc.; *Strakan, Inc.; *Sunesis Pharmaceuticals, Inc.; Swedish Orphan Biovitrum; *Tarix Pharmaceuticals; *TerumoBCT; *Teva Neuroscience, Inc.; *THERAKOS, Inc.; University of Minnesota; and *Wellpoint, Inc. The views expressed in this article do not reflect the official policy or position of the National Institute of Health, the Department of the Navy, the Department of Defense, Health Resources and Services Administration (HRSA) or any other agency of the U.S. Government. This work was also supported in part by a Medical College of Wisconsin Institutional Research Grant #86-004-26 from the American Cancer Society as well as NIA P30 AG017265, USC/UCLA Center on Biodemography and Population Health.
The views expressed in this article do not reflect the official policy or position of the National Institutes of Health, the Department of the Navy, the Department of Defense, or any other agency of the U.S. Government.
*Corporate Members
Footnotes
J.M.K designed and performed research, analyzed data, and wrote the paper. J.D.R. designed research and assisted with writing the paper. B.R.L. designed research, contributed vital analytical tools, analyzed data, and assisted with writing the paper. T.W. designed research and analyzed data. J.M.G.A. and J.M. performed research. S.W.C. designed and performed research, contributed vital analytical tools, analyzed data, and assisted with writing the paper.
Financial disclosure: This work was presented in part at the Annual Meetings of the American Psychosomatic Society, the Society for Biological Psychiatry, and the Academy of Psychosomatic Medicine, 2014.
References
- 1.Baker KS, Davies SM, Majhail NS, Hassebroek A, Klein JP, Ballen KK, et al. Race and socioeconomic status influence outcomes of unrelated donor hematopoietic cell transplantation. Biol Blood Marrow Transplant. 2009;15(12):1543–1554. doi: 10.1016/j.bbmt.2009.07.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Silla L, Fischer GB, Paz A, Daudt LE, Mitto I, Katz B, et al. Patient socioeconomic status as a prognostic factor for allo-SCT. Bone Marrow Transplant. 2009;43(7):571–577. doi: 10.1038/bmt.2008.358. [DOI] [PubMed] [Google Scholar]
- 3.Aziz Z, Iqbal J, Akram M, Anderson BO. Worsened oncologic outcomes for women of lower socio-economic status (SES) treated for locally advanced breast cancer (LABC) in pakistan. Breast. 2010;19(1):38–43. doi: 10.1016/j.breast.2009.10.005. [DOI] [PubMed] [Google Scholar]
- 4.Clegg LX, Reichman ME, Miller BA, Hankey BF, Singh GK, Lin YD, et al. Impact of socioeconomic status on cancer incidence and stage at diagnosis: Selected findings from the surveillance, epidemiology, and end results: National longitudinal mortality study. Cancer Causes Control. 2009;20(4):417–435. doi: 10.1007/s10552-008-9256-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Owen N, Poulton T, Hay FC, Mohamed-Ali V, Steptoe A. Socioeconomic status, C-reactive protein, immune factors, and responses to acute mental stress. Brain Behav Immun. 2003;17(4):286–295. doi: 10.1016/s0889-1591(03)00058-8. [DOI] [PubMed] [Google Scholar]
- 6.Williams DR. Socioeconomic differentials in health: A review and redirection. Soc Psychol Q. 1990:81–99. [Google Scholar]
- 7.Miller GE, Chen E, Fok AK, Walker H, Lim A, Nicholls EF, et al. Low early-life social class leaves a biological residue manifested by decreased glucocorticoid and increased proinflammatory signaling. Proceedings of the National Academy of Sciences. 2009;106(34):14716–14721. doi: 10.1073/pnas.0902971106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Black PH. The inflammatory consequences of psychologic stress: Relationship to insulin resistance, obesity, atherosclerosis and diabetes mellitus, type II. Med Hypotheses. 2006;67(4):879–891. doi: 10.1016/j.mehy.2006.04.008. [DOI] [PubMed] [Google Scholar]
- 9.Gruenewald TL, Cohen S, Matthews KA, Tracy R, Seeman TE. Association of socioeconomic status with inflammation markers in black and white men and women in the coronary artery risk development in young adults (CARDIA) study. Soc Sci Med. 2009;69(3):451–459. doi: 10.1016/j.socscimed.2009.05.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Reiche EM, Nunes SO, Morimoto HK. Stress, depression, the immune system, and cancer. Lancet Oncol. 2004;5(10):617–625. doi: 10.1016/S1470-2045(04)01597-9. [DOI] [PubMed] [Google Scholar]
- 11.Antoni MH, Lutgendorf SK, Cole SW, Dhabhar FS, Sephton SE, McDonald PG, et al. The influence of bio-behavioural factors on tumour biology: Pathways and mechanisms. Nat Rev Cancer. 2006;6(3):240–248. doi: 10.1038/nrc1820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Knight JM, Moynihan JA, Lyness JM, Xia Y, Tu X, Messing S, et al. Peri-transplant psychosocial factors and neutrophil recovery following hematopoietic stem cell transplantation. PloS one. 2014;9(6):e99778. doi: 10.1371/journal.pone.0099778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cole SW. Nervous system regulation of the cancer genome. Brain Behav Immun. 2013;30:S10–S18. doi: 10.1016/j.bbi.2012.11.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Knight JM, Lyness JM, Sahler OJZ, Liesveld JL, Moynihan JA. Psychosocial factors and hematopoietic stem cell transplantation: Potential biobehavioral pathways. Psychoneuroendocrinology. 2013;38(11):2383–2393. doi: 10.1016/j.psyneuen.2013.06.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Costanzo ES, Juckett MB, Coe CL. Biobehavioral influences on recovery following hematopoietic stem cell transplantation. Brain Behav Immun. 2012 doi: 10.1016/j.bbi.2012.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Powell ND, Sloan EK, Bailey MT, Arevalo JM, Miller GE, Chen E, et al. Social stress up-regulates inflammatory gene expression in the leukocyte transcriptome via beta-adrenergic induction of myelopoiesis. Proc Natl Acad Sci U S A. 2013;110(41):16574–16579. doi: 10.1073/pnas.1310655110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Irwin MR, Cole SW. Reciprocal regulation of the neural and innate immune systems. Nature Reviews Immunology. 2011;11(9):625–632. doi: 10.1038/nri3042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Cole SW, Hawkley LC, Arevalo JM, Sung CY, Rose RM, Cacioppo JT. Social regulation of gene expression in human leukocytes. Genome Biol. 2007;8(9):R189. doi: 10.1186/gb-2007-8-9-r189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Cacioppo JT, Hawkley LC. Social isolation and health, with an emphasis on underlying mechanisms. Perspect Biol Med. 2003;46(3 Suppl):S39–S52. [PubMed] [Google Scholar]
- 20.Antoni M, Lutgendorf S, Blomberg B, Stagl J, Carver C, Lechner S, et al. Transcriptional modulation of human leukocytes by cognitive-behavioral stress management in women undergoing treatment for breast cancer. Biol Psychiatry. 2012;71(4):366–372. doi: 10.1016/j.biopsych.2011.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Cole SW, Conti G, Arevalo JM, Ruggiero AM, Heckman JJ, Suomi SJ. Transcriptional modulation of the developing immune system by early life social adversity. Proceedings of the National Academy of Sciences. 2012;109(50):20578–20583. doi: 10.1073/pnas.1218253109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Tanaka J, Imamura M, Kasai M, Hashino S, Kobayashi S, Noto S, et al. The important balance between cytokines derived from type 1 and type 2 helper T cells in the control of graft-versus-host disease. Bone Marrow Transplant. 1997;19(6):571–576. doi: 10.1038/sj.bmt.1700708. [DOI] [PubMed] [Google Scholar]
- 23.Nagler A, Or R, Nisman B, Kalickman I, Slavin S, Barak V. Elevated inflammatory cytokine levels in bone marrow graft rejection. Transplantation. 1995;60(9):943–948. [PubMed] [Google Scholar]
- 24.Fietze E, Prösch S, Reinke P, Stein J, Döcke WD, Staffa G, et al. Cytomegalovirus infection in transplant recipients. the role of tumor necrosis factor. Transplantation. 1994;58(6):675. [PubMed] [Google Scholar]
- 25.Wang XS, Shi Q, Williams LA, Cleeland CS, Mobley GM, Reuben JM, et al. Serum interleukin-6 predicts the development of multiple symptoms at nadir of allogeneic hematopoietic stem cell transplantation. Cancer. 2008;113(8):2102–2109. doi: 10.1002/cncr.23820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Karimi MH, Daneshmandi S, Pourfathollah AA, Geramizadeh B, Ramzi M, Yaghobi R, et al. The IFN-gamma allele is correlated to moderate-to-severe acute graft-versus-host disease after allogeneic stem cell transplant. Exp Clin Transplant. 2010;8(2):125–129. [PubMed] [Google Scholar]
- 27.Lim SH, Coleman S, Bull A, O’Callaghan U, Evely R, Booth M. Cyclosporin A/alpha interferon-induced autologous graft-versus-host disease following peripheral blood stem cell transplant for chronic myeloid leukaemia: A clinico-pathological study. Bone Marrow Transplant. 1997;20(1):83–85. doi: 10.1038/sj.bmt.1700845. [DOI] [PubMed] [Google Scholar]
- 28.Zip code database-deluxe database specifications.[homepage on the Internet] [Google Scholar]
- 29.Ho D, Imai K, King G, Stuart E. Matchit: Nonparametric preprocessing for parametric causal inference. Journal of Statistical Software. 2007 [Google Scholar]
- 30.Jung SH. Sample size for FDR-control in microarray data analysis. Bioinformatics. 2005;21(14):3097–3104. doi: 10.1093/bioinformatics/bti456. [DOI] [PubMed] [Google Scholar]
- 31.Miller GE, Chen E, Sze J, Marin T, Arevalo JMG, Doll R, et al. A functional genomic fingerprint of chronic stress in humans: Blunted glucocorticoid and increased NF-[kappa] B signaling. Biol Psychiatry. 2008;64(4):266–272. doi: 10.1016/j.biopsych.2008.03.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Cole SW, Hawkley LC, Arevalo JMG, Cacioppo JT. Transcript origin analysis identifies antigen-presenting cells as primary targets of socially regulated gene expression in leukocytes. Proceedings of the National Academy of Sciences. 2011;108(7):3080. doi: 10.1073/pnas.1014218108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Fredrickson BL, Grewen KM, Coffey KA, Algoe SB, Firestine AM, Arevalo JM, et al. A functional genomic perspective on human well-being. Proc Natl Acad Sci U S A. 2013;110(33):13684–13689. doi: 10.1073/pnas.1305419110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Efron B, Tibshirani RJ. An introduction to the bootstrap. CRC press. 1994 [Google Scholar]
- 35.Cole SW, Yan W, Galic Z, Arevalo J, Zack JA. Expression-based monitoring of transcription factor activity: The TELiS database. Bioinformatics. 2005;21(6):803–810. doi: 10.1093/bioinformatics/bti038. [DOI] [PubMed] [Google Scholar]
- 36.Cole SW. Elevating the perspective on human stress genomics. Psychoneuroendocrinology. 2010;35(7):955–962. doi: 10.1016/j.psyneuen.2010.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sullivan KM, Agura E, Anasetti C, Appelbaum F, Badger C, Bearman S, et al. Chronic graft-versus-host disease and other late complications of bone marrow transplantation. Semin Hematol. 1991;28(3):250–259. [PubMed] [Google Scholar]
- 38.Przepiorka D, Weisdorf D, Martin P, Klingemann HG, Beatty P, Hows J, et al. 1994 consensus conference on acute GVHD grading. Bone Marrow Transplant. 1995;15(6):825–828. [PubMed] [Google Scholar]
- 39.Cole SW, Sood AK. Molecular pathways: Beta-adrenergic signaling in cancer. Clinical Cancer Research. 2012;18(5):1201–1206. doi: 10.1158/1078-0432.CCR-11-0641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Aydiner A, Ciftci R, Karabulut S, Kilic L. Does beta-blocker therapy improve the survival of patients with metastatic non-small cell lung cancer? Asian Pac J Cancer Prev. 2013;14(10):6109–6114. doi: 10.7314/apjcp.2013.14.10.6109. [DOI] [PubMed] [Google Scholar]
- 41.Barron TI, Connolly RM, Sharp L, Bennett K, Visvanathan K. Beta blockers and breast cancer mortality: A population- based study. J Clin Oncol. 2011;29(19):2635–2644. doi: 10.1200/JCO.2010.33.5422. [DOI] [PubMed] [Google Scholar]
- 42.De Giorgi V, Grazzini M, Gandini S, Benemei S, Lotti T, Marchionni N, et al. Treatment with beta-blockers and reduced disease progression in patients with thick melanoma. Arch Intern Med. 2011;171(8):779–781. doi: 10.1001/archinternmed.2011.131. [DOI] [PubMed] [Google Scholar]
- 43.Benish M, Bartal I, Goldfarb Y, Levi B, Avraham R, Raz A, et al. Perioperative use of beta-blockers and COX-2 inhibitors may improve immune competence and reduce the risk of tumor metastasis. Ann Surg Oncol. 2008;15(7):2042–2052. doi: 10.1245/s10434-008-9890-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Lin X, Luo K, Lv Z, Huang J. Beta-adrenoceptor action on pancreatic cancer cell proliferation and tumor growth in mice. Hepatogastroenterology. 2012;59(114):584–588. doi: 10.5754/hge11271. [DOI] [PubMed] [Google Scholar]
- 45.Lamkin DM, Sloan EK, Patel AJ, Chiang BS, Pimentel MA, Ma JCY, et al. Chronic stress enhances progression of acute lymphoblastic leukemia via β-adrenergic signaling. Brain Behav Immun. 2012 doi: 10.1016/j.bbi.2012.01.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Cole SW. Social regulation of human gene expression: Mechanisms and implications for public health. Am J Public Health. 2013;103(S1):S84–S92. doi: 10.2105/AJPH.2012.301183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Neeman E, Zmora O, Ben-Eliyahu S. A new approach to reducing postsurgical cancer recurrence: Perioperative targeting of catecholamines and prostaglandins. Clin Cancer Res. 2012;18(18):4895–4902. doi: 10.1158/1078-0432.CCR-12-1087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Marks DI, Ballen K, Logan BR, Wang Z, Sobocinski KA, Bacigalupo A, et al. The effect of smoking on allogeneic transplant outcomes. Biology of Blood and Marrow Transplantation. 2009;15(10):1277–1287. doi: 10.1016/j.bbmt.2009.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Chuang YC, Cubbin C, Ahn D, Winkleby MA. Effects of neighbourhood socioeconomic status and convenience store concentration on individual level smoking. J Epidemiol Community Health. 2005;59(7):568–573. doi: 10.1136/jech.2004.029041. [DOI] [PMC free article] [PubMed] [Google Scholar]