Abstract
Background
Monocentric cohorts suggested that radiation-induced CD8 T-lymphocyte apoptosis (RILA) can predict late toxicity after curative intent radiotherapy (RT). We assessed the role of RILA as a predictor of breast fibrosis (bf +) after adjuvant breast RT in a prospective multicenter trial.
Methods
A total of 502 breast-cancer patients (pts) treated by conservative surgery and adjuvant RT were recruited at ten centers. RILA was assessed before RT by flow cytometry. Impact of RILA on bf + (primary endpoint) or relapse was assessed using a competing risk method. Receiver–operator characteristic (ROC) curve analyses were also performed in intention to treat. This study is registered with ClinicalTrials.gov, number NCT00893035 and final analyses are presented here.
Findings
Four hundred and fifty-six pts (90.8%) were included in the final analysis. One hundred and eight pts (23.7%) received whole breast and node irradiation. A boost dose of 10–16 Gy was delivered in 449 pts (98.5%). Adjuvant hormonotherapy was administered to 349 pts (76.5%). With a median follow-up of 38.6 months, grade ≥ 2 bf + was observed in 64 pts (14%). A decreased incidence of grade ≥ 2 bf + was observed for increasing values of RILA (p = 0.012). No grade 3 bf + was observed for patients with RILA ≥ 12%. The area under the ROC curve was 0.62. For cut-off values of RILA ≥ 20% and < 12%, sensitivity and specificity were 80% and 34%, 56% and 67%, respectively. Negative predictive value for grade ≥ 2 bf + was equal to 91% for RILA ≥ 20% and positive predictive value was equal to 22% for RILA < 12% where the overall prevalence of grade ≥ 2 bf + was estimated at 14%. A significant decrease in the risk of grade ≥ 2 bf + was found if patients had no adjuvant hormonotherapy (sHR = 0.31, p = 0.007) and presented a RILA ≥ 12% (sHR = 0.45, p = 0.002).
Interpretation
RILA significantly predicts the risk of breast fibrosis. This study validates the use of RILA as a rapid screening test before RT delivery and will change definitely our daily clinical practice in radiation oncology.
Funding
The French National Cancer Institute (INCa) through the “Program Hospitalier de Recherche Clinique (PHRC)”.
Keywords: Apoptosis, Lymphocyte, Radiotherapy, Breast fibrosis, Prediction
Highlights
-
•
High value of radiation-induced lymphocyte apoptosis (RILA) was clearly associated with no risk of severe toxicities.
-
•
RILA is done rapidly (72 h) and from a single blood sample without any tissue biopsy.
-
•
RILA will change the practice and has to be integrated in multivariate models to optimize the evaluation of the risk.
Of original research, some reports from our group focused on the development of the radiation-induced lymphocyte apoptosis assay. High values of this test were shown to be correlated with the absence of late effect after curative intent radiotherapy. For breast cancer, surgery and immediate reconstruction could be offered instead of conserving-surgery and radiotherapy. Reduced volume as in partial breast radiotherapy could also be proposed in the case of low-risk tumors to protect healthy tissues without carcinologic risk. In addition, low values of this test will be arguments against breast reconstruction. If no alternatives are available new radiotherapy techniques have to be considered.
1. Introduction
Severe but also moderate toxicities after curative-intent radiotherapy (RT), such as a poor cosmetic outcome following breast cancer can have a negative impact on quality of life and a marked effect on subsequent psychological outcome (Al-Ghazal et al., 1999). A number of factors are known to increase the risk of radiation toxicity including intrinsic radiosensitivity (Azria et al., 2012). While toxicity risks for populations of patients are known, the determination of an individual's normal tissue radiosensitivity is seldom possible before treatment. Therefore, current practice standards commonly prescribe radiation dose according to clinical scenarios, without regard to the genotype or phenotype of the individual being irradiated.
In that context, we (Ozsahin et al., 2005) and others (Bordon et al., 2010, Foro et al., 2014, Schnarr et al., 2009) have developed retrospectively or prospectively in small cohorts a rapid (72 h) radiosensitivity assay based on flow cytometric assessment of radiation-induced CD8 T-lymphocyte apoptosis (RILA). An excellent negative predictive value was found in the case of high RILA value and less grade ≥ 2 late toxicity (Ozsahin et al., 2005). In addition, all severe side-effects (grade ≥ 2) were observed in patients with low values of RILA. We assumed that the assay had clear potential as a useful biomarker for selecting individuals likely to display an increased probability of toxicity to RT.
In 2006, we obtained a PHRC (Programme Hospitalier de Recherche Clinique) grant from the French National Cancer Institute to improve the level of evidence of the RILA assay. We then started two prospective multicenter trials (NCT00893035) to evaluate RILA as a predictor of late effects after adjuvant RT of breast cancer (trial 1, n = 502) or after curative intent RT in prostate cancer (trial 2, n = 372). We report here the final results of trial 1 with a median follow-up of 38.6 months.
2. Methods
2.1. Patients
Ten French centers included 502 patients in this prospective multicenter study evaluating the predictive value of RILA on the development of breast fibrosis (bf +). The National Ethical Committee approved this protocol and the study was registered in the NCI PDQ database (NCT00893035). The eligibility criteria included adult patients (age ≥ 18 years) with invasive breast cancer confirmed pathologically; breast-conserving surgery; negative surgical margins; T1/T2 tumors; negative sentinel lymph node or negative/positive axillary node resection (pN −/pN +); negative/positive hormonal receptor tumor expression; post-surgery symptoms < grade 2 (CTCAE v3.0); patient affiliated to the French social security; dated and signed written informed consent. Exclusion criteria are presented in the protocol document.
2.2. Inclusion and Masking
After obtaining informed consent, investigators provided by fax the necessary information to the data center in Montpellier. After an eligibility check, patients were included by the Biometrics Unit of Montpellier. The data center registered enrolment, assigned a unique sequential numbering for each patient, and answered by fax to the respective investigators. RILA was performed in the Montpellier radiobiology laboratory (Institut de Recherche en Cancérologie de Montpellier, IRCM, INSERM U1194, Montpellier, France) and results were concealed from the clinicians before final statistical analyses.
2.3. Radiation-induced CD8 T-lymphocyte Apoptosis (RILA) Procedure
The protocol was adapted from our previous studies (Ozsahin et al., 2005). Briefly, before RT one blood sample was collected from each patient in a 5-ml heparinized tube. 200 μL of blood was aliquoted into a 6-well plate. All tests were carried out in triplicate for both 0 and 8 Gy. Irradiations (single dose of 8 Gy in a 25 cm × 25 cm field size at a dose rate of 1 Gy/min) were delivered after 24 h (H24) using a linear accelerator (2100 EX, 200 UM/min, Varian, US) in the Radiation Department. Control cells were removed from the incubator and placed for the same period of time under the Linac but without radiation treatment. After irradiation, the flasks were immediately incubated at 37 °C (5% CO2). After a further forty-eight hours (H72), it was labeled with anti-human CD8-FITC antibody (10 μL/tests, Becton Dickinson, USA). After addition of lysis buffer (Becton Dickinson, USA), propidium iodide (Sigma, France) and RNAse (Qiagen, France) was added to each tube and prepared for flow cytometry (FACS).
2.4. Preparation and Delivery of Radiotherapy
RT was delivered in the supine position to ensure reproducibility during simulation and treatment. The planning target volume included the whole breast (WB) and the regional lymph nodes (RLN) if necessary. Only photons were allowed for WB irradiation thus allowing standardisation of treatment across centers.
A median dose of 50 Gy to the target volume was recommended. The field arrangement involved the use of an anterior photon field in the supraclavicular region and a combination of anterior electrons/photons to the internal mammary nodes at 44–50 Gy. A daily dose of 50 Gy to the WB was delivered by two opposed tangential fields; a boost in the surgical bed up to 10–16 Gy was given when necessary. Fractionation was 2 Gy per fraction, 5 days a week. Calculation used 3-D dosimetry. The ICRU report 62 prescription points were used for prescribing dose. As a minimum, on-line portal imaging was obtained each day for the first three days and once a week during the rest of the course of treatment.
2.5. Adjuvant Systemic Therapies
Chemotherapy (CT) regimen when indicated consisted either of 6 cycles of FEC 100 [5 FU (500 mg/m2), epirubicin (100 mg/m2), cyclophosphamide (500 mg/m2)] on day 1 and repeated every 21 days or 3 cycles of FEC 100 followed by 3 cycles of docetaxel (100 mg/m2) every three weeks. In case of HER2 overexpression or gene amplification, trastuzumab (beginning with a loading dose of 8 mg/kg) was added to the protocol (6 mg/kg every 3 weeks for 1 year). Hormonotherapy (HT: tamoxifen or aromatase inhibitor) was started after surgery or after the end of RT and given daily for five years.
2.6. End-point Assessments
The primary objective was the predictive role of RILA in radiation-induced grade ≥ 2 bf +. Secondary objectives were the incidence of acute side effects, local recurrence, relapse-free survival (RFS), breast fibrosis-free survival (BF-FS), breast fibrosis-relapse-free survival (BF-RFS) and overall survival (OS). Acute and late side effects were assessed and graded according to the CTC v3.0 scale (Trotti et al., 2003).
Toxicity evaluations were performed at baseline, every week during RT, one, three and six months after the last RT fraction, every 6 months up to month 36. Each evaluation was assessed by the physicians blinded for RILA. The most severe late bf + observed from 12 weeks to 3 years post RT was considered as the primary endpoint. The most severe late effects (skin, lung, cardiac) observed from 12 weeks to 3 years post RT and the most severe acute side effects (skin and lung mainly) observed from the start of RT to 12 weeks post RT were considered as the secondary endpoints. Toxicities were evaluated using all the possible definitions described in the scale “Dermatology/skin area”, “pulmonary/upper respiratory” and “cardiac general” (Trotti et al., 2003).
All endpoints were defined as the interval between the start of RT and following the first events: death for OS, local or contralateral or distant recurrence or death for RFS, grade ≥ 2 bf + for BF-FS, and first event of RFS and BF-FS for BF-RFS (Peto et al., 1977). Censoring patients were patients alive at the last follow-up visit for OS, patients alive and without relapse for RFS, patients alive who never experienced a grade ≥ 2 bf + for BF-FS and patients alive who never experienced grade ≥ 2 bf + or relapse for BF-RFS.
2.7. Sample Size Calculation and Statistical Analysis
To test the prognostic value of RILA rate on the occurrence of breast fibrosis in breast cancer, we started from the results of our preliminary study (Azria et al., 2004). Details are presented in the protocol document. Briefly, based on σ2 = 0.54, an estimated complication rate of ψ = 15% with a two-tailed α error of 0.05 and a β error of 0.05 (power = 0.95), 430 patients had to be included. The number of patients was increased by at least 15% (n = 494) to take into account loss to follow-up and the impact of the boost on bf +.
The cumulative incidences of complications as a function of the prognostic variables were calculated using a non-parametric model (Pepe and Mori, 1993). The main statistical procedure included a multivariate analysis using the Fine et al. model of competing risks (Fine, 2001) for the assessment of the impact of RILA rate on the occurrence of bf + in the presence of other events (such as relapse or death) that are considered as competing risk events in this pathology. For multivariate analysis, selected factors were the baseline parameters with a p value less than 0.20 in univariate analysis. Final model was defined using backward stepwise selection (p < 0.15) and a step by step method was used to include only the significant parameters (p < 0.05).
Data were summarized by frequency and percentage for categorical variables and by median and range for continuous variables. Absolute changes in RILA counts before and after irradiation were evaluated as continuous and categorical variables. Three categories were constructed around the 33% quantiles (< 12, 12–20, and ≥ 20) and then merged in two categories (< 12 and ≥ 12).
OS, RFS, BF-FS and BF-RFS rates were estimated by the Kaplan-Meier method. Ninety-five percent confidence intervals (95%CI) were also determined.
Univariate analysis and multivariate analysis were performed using the Cox proportional hazard's regression model to estimate the hazard ratio including baseline characteristics and treatment parameters. Comparisons were performed using the log-rank test for univariate analysis. Independent effects were evaluated from the likelihood ratio statistics.
Impact of RILA on breast fibrosis-relapse-free survival (BF-RFS) was assessed. The cumulative incidence of breast fibrosis and relapse or death were estimated from a competing risk model using estimates obtained from the cause-specific hazard functions and the composite RFS and BF-FS distribution (Arriagada et al., 1992) and compared using Gray's test.
Median follow-up was estimated with the inverse Kaplan-Meier method. A p value less than 0.05 was regarded as significant. All statistical tests were two sided. Stata was used for all statistical analyses (version 13.0) and the SAS macro %cif was used for Gray's test.
To complement analysis, receiver–operator characteristic (ROC) curve analyses for RILA were performed to identify patients who experienced at least a grade 2 bf + within three years (Kramar et al., 2001). The empirical areas under the ROC curves (AUC) and the respective 95%CI were used for RILA to determine the sensitivity, specificity, positive (PPV), and negative predictive value (NPV).
2.8. Role of the Funding Source
The funder of the study had no role in the study design, data collection, data analysis, data interpretation, or writing of the report. DA, FC and SG had access to the raw data. The report was written by the authors, who had unrestricted access to the study data and are responsible for the accuracy and completeness of the reported analyses. The corresponding author had full access to all of the data and the final responsibility to submit for publication.
3. Results
3.1. Trial Profile
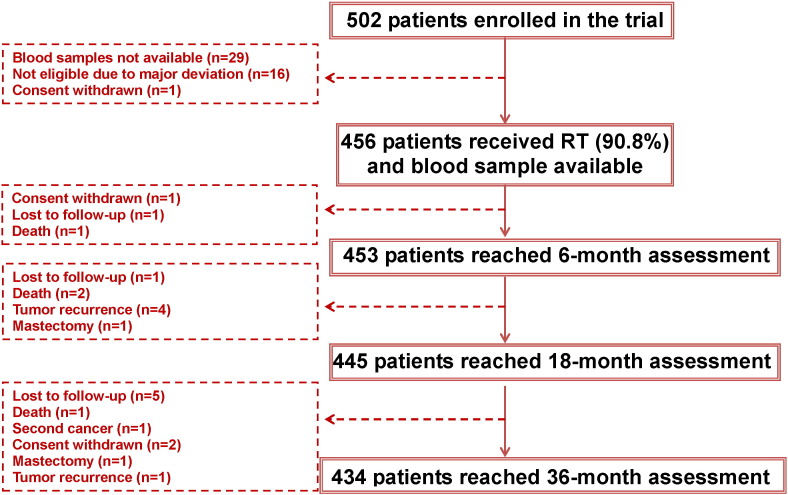
Fig. 1 shows the trial profile. From Jan 15 2007, to Jul 11 2011, 502 patients were included in this trial by 10 centers in France. At baseline, 16 patients were excluded for major deviation, one patient withdrew consent before any treatment and blood samples were not technically available for 29 participants. All these patients (n = 46) were treated according to current guidelines and were not included for analysis since no data were collected. Thus, RILA and complete RT were performed for 456 patients (90.8%) before entering follow-up. 434 (86.5%) patients were followed for at least 36 months according to the protocol. The 22 other patients interrupted the planned follow-up before 36 months (Fig. 1).
Fig. 1.
Trial profile.
3.2. Acute and Late Toxicities
A total of 455 patients experienced an acute side effect (Table 1). Dermatitis was the most common skin toxicity graded 1, 2, or 3 in 217 (47.6%), 210 (46.1%), and 25 (5.5%) patients, respectively. Only one grade 2 pneumonitis was observed. Symptoms rapidly settled in most patients and in univariate analysis a significant association was found between early side effects and RILA (p = 0.003, Table 1). In addition, a positive correlation was found between grade ≥ 2 acute and late effects (p = 0.03).
Table 1.
Early and late side effects.
| Grade 0 | Grade 1 | Grade 2 | Grade 3 | |
|---|---|---|---|---|
| Early side effects | ||||
| Number of patients | 1 (0.2%) | 197 (43.2%) | 230 (50.4%) | 28 (6.1%) |
| RILA | ||||
| < 12% | 1 (100%) | 62 (31.5%) | 97 (42.2%) | 9 (32.1%) |
| 12–20% | 0 | 63 (32.0%) | 61 (26.5%) | 16 (57.1%) |
| ≥ 20% | 0 | 72 (36.6%) | 72 (31.3%) | 3 (10.7%) |
| Late side effects | ||||
| Number of patients | 62 (13.6%) | 253 (55.5%) | 129 (28.3%) | 12 (2.6%) |
| RILA | ||||
| < 12% | 22 (35.5%) | 89 (35.2%) | 54 (41.9%) | 4 (33.3%) |
| 12–20% | 19 (30.6%) | 76 (30.0%) | 40 (31.0%) | 5 (41.7%) |
| ≥ 20% | 21 (33.9%) | 88 (34.8%) | 35 (27.1%) | 3 (25.0%) |
RILA = radiation-induced CD8 T-lymphocyte apoptosis.
All types of late side effects were evaluated for 434 patients (95.2%) at 3 years as mentioned in Fig. 1. A total of 394 (86.4%) patients experienced a late side effect (Table 1). A total of 218 (47.8%), 61 (13.4%), and 3 (0.6%) experienced grade 1, 2, and 3 bf +, respectively, including 3 patients with grade 2 at baseline but not included in the primary endpoint. Grade ≥ 2 bf + occurred 6 months after the end of RT in 38 patients (62.3%). No late lung fibrosis or cardiac toxicity was observed.
3.3. Patients, Disease and Treatment Characteristics According to Breast Fibrosis
Patient and disease characteristics of the 456 patients according to grade ≥ 2 bf + and RILA are presented in Table 2, Table 3, respectively. Overall, median age was 56 years (range 29–88), 69 (15.8%) patients were active smokers and 312 (69.2%) patients were postmenopausal. Baseline characteristics were well balanced for age, menopausal status, breast volume, node status, and hormone receptor expression between bf + and bf − groups. More women smoked in the bf + group than in the bf − group. In addition, more T2 stage and grade 1 tumors were shown in the bf + group (Table 2).
Table 2.
Baseline patient and disease characteristics according to breast fibrosis.
| Grade ≥ 2 fibrosis |
Grade ≤ 1 fibrosis |
||
|---|---|---|---|
| N = 61 (%) | N = 395 (%) | p | |
| Median age (years, range) | 55 (38–86) | 56 (29–88) | 0.91 |
| Tobacco smoking | |||
| Non-smokers | 32 (52.5) | 257 (65.1) | 0.04 |
| Active/former smokers | 28 (45.9) | 119 (30.1) | |
| NA | 1 (1.6) | 19 (4.8) | |
| ECOG | |||
| 0 | 58 (95.1) | 365 (92.4) | 0.41 |
| 1 | 3 (4.9) | 14 (3.5) | |
| 2 | 0 | 1 (0.3) | |
| NA | 0 | 15 (3.8) | |
| Menopausal status | |||
| Premenopausal | 20 (32.8) | 119 (30.1) | 0.56 |
| Postmenopausal | 40 (65.6) | 272 (68.9) | |
| NA | 1 (1.6) | 4 (1.0) | |
| Breast volume | |||
| Small | 14 (23.0) | 121 (30.6) | 0.47 |
| Large | 36 (59.0) | 205 (51.9) | |
| NA | 11 (18.0) | 69 (17.5) | |
| T stage | |||
| 0 | 2 (3.3) | 3 (0.8) | 0.05 |
| 1 | 46 (75.4) | 344 (87.1) | |
| 2 | 13 (21.3) | 46 (11.6) | |
| NA | 0 | 2 (0.5) | |
| N stage | |||
| 0 | 52 (85.2) | 337 (85.3) | 0.81 |
| 1 | 9 (14.8) | 50 (12.7) | |
| 2 | 0 | 6 (1.5) | |
| 3 | 0 | 1 (0.2) | |
| NA | 0 | 1 (0.2) | |
| Hormonal receptors | |||
| ER −/PR − | 2 (3.3) | 39 (9.9) | 0.05 |
| ER +/PR − | 12 (19.7) | 58 (14.7) | |
| ER −/PR − | 2 (3.3) | 2 (0.5) | |
| ER +/PR + | 44 (72.1) | 273 (69.1) | |
| NA | 1 (1.6) | 23 (5.8) | |
| Histological grade (SBR) | |||
| I | 26 (42.6) | 109 (27.6) | 0.01 |
| II | 22 (36.1) | 209 (52.9) | |
| III | 13 (21.3) | 61 (15.4) | |
| NA | 0 | 16 (4.1) |
Data are n or median (range). NA = not available. SBR = Scarff–Bloom–Richardson. ER and PR = estrogen and progesterone receptors. ECOG = Eastern Cooperative Oncology Group.
Table 3.
Baseline patient and disease characteristics according to RILA.
| RILA < 12% |
RILA ≥ 12% |
||
|---|---|---|---|
| N = 169 (%) | N = 287 (%) | p | |
| Median age (years, range) | 56 (37–79) | 57 (29–88) | 0.32 |
| Tobacco smoking | |||
| Non-smokers | 102 (60.4) | 187 (65.1) | 0.37 |
| Active/former smokers | 61 (36.1) | 86 (30.0) | |
| NA | 6 (3.6) | 12 (4.9) | |
| ECOG | |||
| 0 | 156 (92.3) | 267 (93.0) | 0.70 |
| 1 | 6 (3.6) | 11 (3.8) | |
| 2 | 1 (0.6) | 0 | |
| NA | 6 (3.5) | 9 (3.1) | |
| Menopausal status | |||
| Premenopausal | 53 (31.4) | 86 (30.0) | 0.28 |
| Postmenopausal | 116 (68.6) | 196 (68.3) | |
| NA | 0 | 5 (1.7) | |
| Breast volume | |||
| Small | 44 (26.0) | 91 (31.7) | 0.42 |
| Large | 95 (56.2) | 146 (50.9) | |
| NA | 30 (17.8) | 50 (17.4) | |
| T stage | |||
| 0 | 2 (1.2) | 3 (1.0) | 0.41 |
| 1 | 140 (82.8) | 250 (87.1) | |
| 2 | 27 (16.0) | 32 (11.2) | |
| NA | 0 | 2 (0.7) | |
| N stage | |||
| 0 | 145 (85.8) | 244 (85.0) | 1.00 |
| 1 | 22 (13.0) | 37 (12.9) | |
| 2 | 2 (1.2) | 4 (1.4) | |
| 3 | 0 | 1 (0.3) | |
| NA | 0 | 1 (0.3) | |
| Hormonal receptors | |||
| ER −/PR − | 17 (10.1) | 24 (8.4) | 0.29 |
| ER +/PR − | 32 (18.9) | 38 (13.2) | |
| ER −/PR + | 2 (1.2) | 2 (0.7) | |
| ER +/PR + | 112 (66.3) | 205 (71.4) | |
| NA | 6 (3.6) | 18 (6.3) | |
| Histological grade (SBR) | |||
| I | 44 (26.0) | 91 (31.7) | 0.001 |
| II | 76 (45.0) | 155 (54.0) | |
| III | 43 (25.4) | 31 (10.8) | |
| NA | 6 (3.6) | 10 (3.5) |
NA = not available. SBR = Scarff–Bloom–Richardson. ER and PR = estrogen and progesterone receptors. ECOG = Eastern Cooperative Oncology Group.
Table 4 shows details of all treatments. Quadrantectomy and adjuvant hormonotherapy was performed more often in the bf + group than the bf − group. Overall, hormonotherapy was given to 349 (76.5%) patients. Chemotherapy (n = 143, 31.4%) and trastuzumab (n = 17, 3.7%) were similar between groups. In all patients (n = 456), 50 Gy were delivered to the WB. A localized 16-Gy boost (range 6–24.8) was delivered to 449 (98.5%) patients to a median total dose of 66 Gy (range 48–74.8). Median treatment duration was 49 days (range 35–65). Node irradiation (supraclavicular (SC) only and both SC and mammary chain) was delivered to 108 (23.7%) patients and was more frequently used in the bf + group than the bf − group.
Table 4.
Treatments and RILA according to breast fibrosis.
| Grade ≥ 2 fibrosis (n = 61) |
Grade ≤ 1 fibrosis (n = 395) |
|||
|---|---|---|---|---|
| N | % | N | % | |
| Type of initial surgery | ||||
| Tumorectomy | 44 | 72.1 | 337 | 85.3 |
| Quadrantectomy | 17 | 27.9 | 58 | 14.7 |
| Histology | ||||
| LIC | 3 | 4.9 | 53 | 13.4 |
| DIC | 55 | 90.2 | 325 | 82.3 |
| LIC + DIC | 1 | 1.6 | 4 | 1.0 |
| Other | 2 | 3.3 | 13 | 3.3 |
| Margin | ||||
| Negative | 59 | 96.7 | 382 | 96.7 |
| Positive | 2 | 3.3 | 11 | 2.8 |
| NA | 0 | 2 | 0.5 | |
| Surgical area | ||||
| < 50 cm3 | 26 | 42.6 | 207 | 52.4 |
| ≥ 50 cm3 | 34 | 55.8 | 183 | 46.3 |
| NA | 1 | 1.6 | 5 | 1.3 |
| Adjuvant chemotherapy | ||||
| No | 36 | 59.0 | 277 | 70.1 |
| Yes | 25 | 41.0 | 118 | 29.9 |
| Adjuvant trastuzumab | ||||
| No | 58 | 95.1 | 381 | 96.5 |
| Yes | 3 | 4.9 | 14 | 3.5 |
| Adjuvant hormonotherapy | ||||
| No | 6 | 9.8 | 101 | 25.6 |
| Yes | 55 | 90.2 | 294 | 74.4 |
| Node irradiation | ||||
| Mammary gland only | 39 | 63.9 | 309 | 78.2 |
| Supraclavicular +/− IMC | 22 | 36.1 | 86 | 21.8 |
| Boost | ||||
| No | 0 | 7 | 1.8 | |
| Yes | 61 | 100.0 | 388 | 98.2 |
| Boost technique | ||||
| Photon | 48 | 78.7 | 292 | 73.9 |
| Electron | 9 | 14.8 | 46 | 11.6 |
| Brachytherapy | 0 | 1 | 0.3 | |
| Photon + electron | 3 | 4.9 | 47 | 11.9 |
| NA | 1 | 1.6 | 9 | 2.3 |
| RILA (percent, range) | 10.9 | (1.3–35.9) | 15.6 | (0.7–52.8) |
Data are n or median (range). DIC = ductal invasive carcinoma. LIC = lobular invasive carcinoma. NA = not available. IMC = internal mammary chain. RILA = radiation-induced CD8 T-lymphocyte apoptosis.
A total of 293 (64.8%) patients interrupted their RT with a median interruption of 3 days (range 1–12) mainly due to programmed maintenance or short flaws in the machines. More interruptions were observed in bf + group (n = 46, 76.7%) compared to bf − group (n = 247, 63.0%, p = 0.04) but no specific reason was statistically found.
3.4. Radiation-induced CD8 T-lymphocyte Apoptosis (RILA) and Breast Fibrosis
Overall, median RILA was 15.2 (range 0.7–52.8) and was significantly lower (p = 0.004) in the bf + group (10.9, range 1.3–35.9) than in the bf − group (15.6, range 0.7–52.8). There was an inverse relationship between the incidence of bf + and RILA. A decreased percentage of grade ≥ 2 bf + was observed for increasing values of RILA (p = 0.001). No grade 3 bf + were observed for patients with RILA > 12%.
A ROC analysis was performed among the 435 patients (95.4%) who were followed up to 3 years (n = 434) or who presented a grade ≥ 2 bf + but were lost to follow-up before 3 years (n = 1). The area under the ROC curve was 0.62 (95%CI 0.54–0.70). For RILA ≥ 20% and < 12% sensitivity and specificity were 80% and 34%, 56% and 67%, respectively. The PPV was equal to 22% for RILA < 12% and the NPV was equal to 91% for RILA ≥ 20%, where the overall prevalence of grade ≥ 2 bf + was estimated at 14%.
3.5. Competing Risk Model
There were 12 relapses (2.6%) and 5 patients died (1.1%). As a first event, there were 61 late skin side effects (13.4%), 9 relapses (2%) and 4 deaths (0.9%). At 3 years, 366 patients were event-free. Three-year OS and RFS were 99.1% (95%CI 97.6–99.7) and 97.1% (95%CI 95.1–98.3), respectively. The 3-year BF-FS rate was 87.8% (95%CI 84.4–90.5). Prognostic factors for BF-FS are described in Table 5. RILA, tobacco smoking, T stage, tumor location in the breast, hormonal status, tumor grade, type of surgery, surgical breast volume, adjuvant chemotherapy and hormonotherapy, node irradiation, and boost were included in the multivariate analysis. In the final model, adjuvant hormonotherapy and RILA remained the two independent factors for BF-RFS, adjusted for tobacco smoking (Table 6). A significant decrease in the risk of grade ≥ 2 bf + was found if patients did not receive adjuvant hormonotherapy (subdistribution HR, sHR = 0.31, 95%CI 0.13–0.73, p = 0.007) and presented a RILA ≥ 12 (sHR = 0.45, 95%CI 0.27–0.75, p = 0.002). A high trend was observed if they were non-smokers (sHR = 0.62, 95%CI 0.37–1.05, p = 0.074).
Table 5.
Prognostic factors for complication-free survival: univariate analysis.
| N event/N total | 3-year CFS (%) | HR | 95%CI | p value | ||
|---|---|---|---|---|---|---|
| RILA (%) | Continuous | 0.93–0.99 | 0.005 | |||
| < 12 | 34/169 | 80.7 | 1 | 0.001 | ||
| ≥ 12 | 27/287 | 91.9 | 0.44 | 0.26–0.73 | ||
| < 12 | 34/169 | 80.7 | 1 | 0.005 | ||
| 12–20 | 15/140 | 91.3 | 0.49 | 0.27–0.91 | ||
| ≥ 20 | 12/147 | 92.5 | 0.38 | 0.20–0.74 | ||
| Age (years) | ≤ 55 | 31/216 | 88.3 | 1 | 0.620 | |
| > 55 | 30/240 | 87.3 | 0.88 | 0.53–1.46 | ||
| Tobacco smoking | Non-smokers | 32/289 | 89.8 | 1 | 0.020 | |
| Active or former smokers | 28/147 | 82.8 | 1.83 | 1.10–3.04 | ||
| NA | 20 | |||||
| Menopausal status | Premenopausal | 20/139 | 89.8 | 1 | 0.782 | |
| Postmenopausal | 40/312 | 87.0 | 0.93 | 0.54–1.59 | ||
| NA | 5 | |||||
| Breast volume | Small | 14/135 | 91.7 | 1 | 0.216 | |
| Large | 36/241 | 85.8 | 1.46 | 0.79–2.71 | ||
| NA | 80 | |||||
| T stage | 0 | 2/5 | 80.0 | 1 | 0.037 | |
| 1 | 46/390 | 89.4 | 0.28 | 0.07–1.17 | ||
| 2 | 13/59 | 77.0 | 0.60 | 0.13–2.64 | ||
| NA | 2 | |||||
| N stage | 0 | 52/389 | 88.0 | 1 | 0.931 | |
| 1–2–3 | 9/66 | 86.3 | 1.03 | 0.51–2.09 | ||
| NA | 1 | |||||
| Hormonal status | ER − PR − | 2/41 | 97.6 | 1 | 0.080 | |
| ER + PR − | 12/70 | 83.8 | 3.61 | 0.81–16.13 | ||
| ER − PR + | 2/4 | 50.0 | 12.94 | 1.82–92.01 | ||
| ER + PR + | 44/317 | 87.3 | 2.86 | 0.69–11.79 | ||
| NA | 24 | |||||
| Histological grade (SBR) | I | 26/135 | 82.0 | 1 | 0.018 | |
| II | 22/231 | 91.8 | 0.47 | 0.27–0.83 | ||
| III | 13/74 | 83.4 | 0.95 | 0.49–1.85 | ||
| NA | 16 | |||||
| Type of surgery | Tumorectomy | 44/381 | 89.9 | 1 | 0.012 | |
| Quadrantectomy | 17/75 | 77.0 | 2.16 | 1.23–3.79 | ||
| Histology | LIC | 3/56 | 96.4 | 1 | 0.216 | |
| DIC | 55/380 | 86.7 | 2.85 | 0.89–9.11 | ||
| LIC + DIC | 1/5 | 80.0 | 3.94 | 0.41–38.04 | ||
| Other | 2/15 | 87.0 | 2.61 | 0.44–15.59 | ||
| Margin | Negative | 59/441 | 87.8 | 1 | 0.791 | |
| Positive | 2/13 | 85.0 | 1.22 | 0.30–4.98 | ||
| NA | 2 | |||||
| Surgical area | < 50 cm3 | 26/233 | 90.0 | 1 | 0.151 | |
| ≥ 50 cm3 | 34/217 | 85.5 | 1.45 | 0.87–2.42 | ||
| NA | 6 | |||||
| Adjuvant CT | No | 36/313 | 89.7 | 1 | 0.060 | |
| Yes | 25/143 | 83.5 | 1.64 | 0.98–2.73 | ||
| Adjuvant trastuzumab | No | 58/439 | 87.8 | 1 | 0.580 | |
| Yes | 3/17 | 88.2 | 1.41 | 0.44–4.50 | ||
| Adjuvant HT | No | 6/107 | 87.8 | 1 | 0.580 | |
| Yes | 55/349 | 88.2 | 1.41 | 0.44–4.50 | ||
| Node RT | No | 39/348 | 90.1 | 1 | 0.019 | |
| Yes | 22/108 | 80.3 | 1.91 | 1.14–3.23 | ||
| Boost | No | 0/7 | − | 1 | 0.158 | |
| Yes | 61/449 | 87.6 | − | − | ||
| Boost technique | Photon | 48/340 | 87.2 | 1 | 0.281 | |
| Electron | 9/55 | 85.0 | 1.21 | 0.59–2.47 | ||
| Brachytherapy | 0 | – | – | – | ||
| Photons + electrons | 3/50 | 93.9 | 0.41 | 0.13–1.31 | ||
| NA | 11 | |||||
| Acute side-effects | Grade 0–1 | 17/198 | 91.7 | 1 | 0.030 | |
| Grade 2 | 39/230 | 85.1 | 2.06 | 1.16–3.64 | ||
| Grade 3 | 5/28 | 82.0 | 2.09 | 0.77–5.68 | ||
CFS = complication-free survival. NA = not available. HR = hazard ratio. N = number. DIC = ductal invasive carcinoma. LIC = lobular invasive carcinoma. SBR = Scarff–Bloom–Richardson. ER = estrogen receptor. PR = progesterone receptor. + = positive. − = negative. RT = radiotherapy. HT = hormonotherapy. CT = chemotherapy. RILA = radiation-induced CD8 T-lymphocyte apoptosis. The logrank test was used for all p-values.
Table 6.
Multivariate regression analysis for CRFS and competing risk analysis.
| Fibrosis and relapse (Cox regression) |
Fibrosis and relapse (competing risks) |
|||||
|---|---|---|---|---|---|---|
| HR | 95%CI | p value | sHR | 95%CI | p value | |
| RILA in subclasses | ||||||
| RILA | ||||||
| < 12 | 1 | 1 | ||||
| ≥ 12 | 0.45 | 0.27–0.74 | 0.002 | 0.45 | 0.27–0.75 | 0.002 |
| Tobacco Smoking | ||||||
| No | 1 | 1 | ||||
| Active/former | 1.61 | 0.97–2.68 | 0.068 | 1.60 | 0.96–2.69 | 0.074 |
| Hormonotherapy | ||||||
| No | 1 | 1 | ||||
| Yes | 3.17 | 1.36–7.40 | 0.008 | 3.21 | 1.37–7.53 | 0.007 |
| RILA as continuous variable | ||||||
| RILA | 0.96 | 0.93–0.99 | 0.012 | 0.96 | 0.92–0.99 | 0.025 |
| Tobacco Smoking | ||||||
| No | 1 | 1 | ||||
| Active/former | 1.56 | 0.93–2.06 | 0.091 | 1.56 | 0.93–2.61 | 0.093 |
| Hormonotherapy | ||||||
| No | 1 | 1 | ||||
| Yes | 3.17 | 1.36–7.39 | 0.008 | 3.18 | 1.36–7.44 | 0.008 |
HR = hazard ratio estimated by Cox regression. sHR = subdistribution hazard ratio estimated by competing risk method. RILA = radiation-induced CD8 T-lymphocyte apoptosis. CRFS = complication-relapse-free survival.
Interactions between significant parameters were tested and remained not significant. A similar model was obtained using RILA as a continuous parameter.
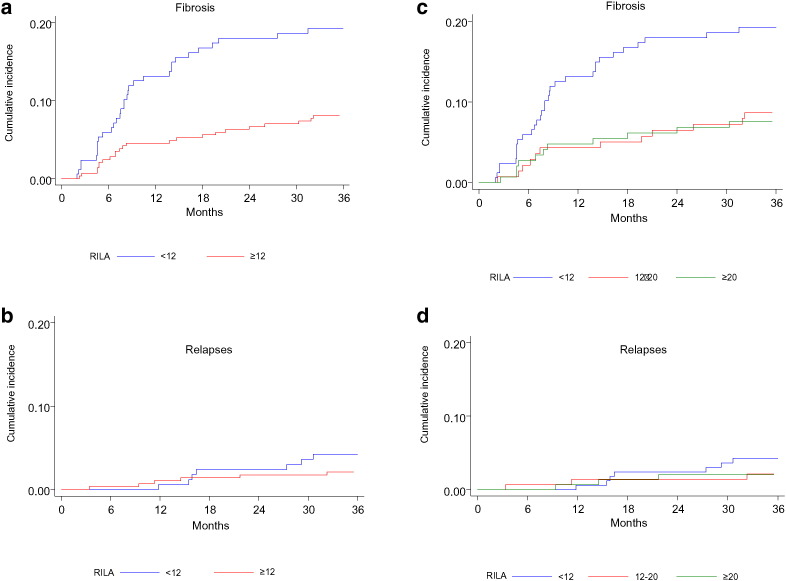
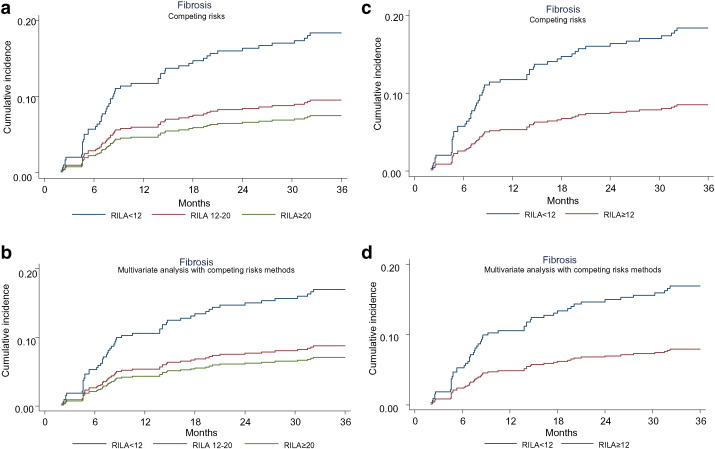
The 3-year cumulative incidence rates for grade ≥ 2 bf + were 19.3%, 8.7%, and 7.5% in RILA < 12, 12–20, and ≥ 20, respectively (Table 7, Fig. 2) (p = 0.005). The relapse component of the composite RFS and BF-FS rates was similarly distributed between the three categories of RILA with estimated cumulative relapse incidence of 4%, 2%, and 2%, respectively (Table 7, Fig. 3).
Table 7.
Number and cumulative incidence of first event according to RILA.
| Total number of events | 3-year cumulative incidence of grade ≥ 2 breast fibrosis | p value | 3-year cumulative incidence of relapse | p value | |
|---|---|---|---|---|---|
| RILA | |||||
| < 12 | 41 | 19.3 | 4.2 | ||
| 12–20 | 17 | 8.7 | 2.2 | ||
| ≥ 20 | 16 | 7.5 | 0.005 | 2.0 | 0.764 |
Event = first event of complication (grade ≥ 2 breast fibrosis) or relapse. RILA = radiation-induced CD8 T-lymphocyte apoptosis.
Fig. 2.
Cumulative incidence of grade ≥ 2 late side effects (BF-FS, breast fibrosis-free survival) and relapses (RFS, relapse-free survival). Breast fibrosis component (a) and relapse component (b) for two categories (< 12 and ≥ 12) of radiation-induced lymphocyte apoptosis (RILA) are presented. Breast fibrosis component (c) and relapse component (d) for three categories (< 12, 12–20, and ≥ 20) of RILA are presented.
Fig. 3.
Cumulative incidence of first events according to radiation-induced lymphocyte apoptosis (RILA). Univariate (a) and multivariate (b) analyses for three categories (< 12, 12–20, and ≥ 20) of RILA are presented. Univariate (c) and multivariate (d) analyses for two categories (< 12 and ≥ 12) of RILA are presented.
In multivariate analysis, no correlation was found between acute effects and RILA or other clinically relevant parameters.
4. Discussion
In the present multicenter study, we confirmed prospectively that a decreased percentage of grade ≥ 2 bf + was observed for increasing values of RILA with an excellent NPV of 91%. RILA identified independently and with more than 90% accuracy patients who will not develop severe late fibrosis among other factors like hormonotherapy and in a lesser extent tobacco smoking. In addition, we observed no grade 3 bf + in patients with RILA ≥ 12%. In multivariate analysis, no correlation between RILA and acute events was found. Finally, the recurrence rates were higher in the RILA < 12% group but without statistical significance.
This trial evaluated the predictive impact of an individual radiosensitivity biomarker in accordance with the methodology published by the REMARK international committee (Altman et al., 2012). Before the current trial, we and others presented promising results of correlation between RILA and late side-effects but only in retrospective manner or in a prospective single center study (Azria et al., 2010, Foro et al., 2014, Ozsahin et al., 2005, Schnarr et al., 2009). Here, the level of evidence of RILA was highly improved and allowed a rapid implementation of the RILA assay in daily practice.
The main findings of this trial confirmed that a biological assay by itself will not be sufficient to predict the risk of late effects. To predict if a patient is at risk or not of developing severe effects after curative intent RT, many factors have to be added to the biological determinant, namely systemic treatments and tobacco smoking. In addition, RILA taken as a continuous variable is strongly predictive of late fibrosis and reinforces the usefulness of this test in multifactorial support systems integrating clinical, physical and biological factors to decide the optimal treatment strategy (Lambin et al., 2013).
In the current study, adjuvant hormonotherapy but not chemotherapy was found to be an independent factor of late fibrosis. We and others identified that risk in previous cohorts but mainly with tamoxifen (Azria et al., 2010, Azria et al., 2004, Bentzen et al., 1996, Johansen et al., 2007) and probably through synergistic secretion of profibrotic cytokines such as transforming growth factor β (Canney and Dean, 1990). New chemotherapy regimens including taxanes seem to reduce the risk compared to the CMF protocol (cyclophosphamide, methotrexate, and 5-FU) (Johansen et al., 2007).
Tobacco smoking was also a potential factor that may increase the risk of late fibrosis by 60% but with only a statistical trend. Nevertheless, we are still convinced that this factor has to be considered in a multifactorial model as it alters recovery after RT and/or surgery and impairs cosmetic outcome in case of breast reconstruction (Kern et al., 2015).
The absence of correlation between RILA and acute toxicities in the multifactorial analysis is in concordance with our preliminary trial (Ozsahin et al., 2005). More than 60% of late fibrosis appeared 6 months after the end of RT, suggesting that mechanisms other than DNA repair are represented by the RILA assay. Correlation between cellular radiosensitivity and early skin reactions is highly controversial (Begg et al., 1993, Johansen et al., 1996) and could not explain by itself the late reactions observed clinically in tissues. In lymphocytes from 26 patients with locally advanced breast carcinoma, an inverse correlation was found between initial damage to DNA and RILA (Pinar et al., 2010). It is still unknown why the lymphocyte and particularly the CD8 subtype present this property to be the mirror of healthy tissues. Ongoing works within a European consortium (West et al., 2014) are trying to identify if some genetic polymorphisms (Azria et al., 2008, Seibold et al., 2015) may explain the incorrect healing of the irradiated tissue. Recently, we started the BIORISE project to evaluate the underlying mechanisms using a proteomics approach (Lacombe et al., 2013). Interesting preliminary results show protein overexpression and significant ROS production in lymphocytes of patients with radiation-induced severe late effects and low RILA (manuscript in preparation). Research to understand how lymphocytes can predict late RT-toxicity has now been launched in Montpellier.
Breast is of course not the only site for which RILA is useful. In our first clinical trial (Ozsahin et al., 2005), many tumor sites were included and RILA predicted late rectal, bladder, or cervical toxicities after RT. It was then confirmed by others in prostate (Foro et al., 2014), head and neck (Bordon et al., 2010), and cervix (Bordon et al., 2011). PHRC trial 2 (NCT00893035) dedicated to prostate is now closed to recruitment and will be presented with longer follow-up. The mechanism underlying the predictive property of RILA seems to be ubiquitous whatever the irradiated site.
The clinical utility of RILA is high as mentioned in the STROGAR guidelines (Kerns et al., 2014). For example, patients identified as low risk of late effects might be offered a higher dose with modern techniques or radiotherapy in combination with systemic therapies with the aim of improving local control. In some cases radiotherapy may be avoided completely for individuals with a high risk of toxicity, provided that an effective alternative exists. For example, for breast cancer, surgery and immediate reconstruction could be offered instead of conserving-surgery and radiotherapy. Reduced volume as in partial breast RT could also be proposed in case of low-risk tumors to protect healthy tissues without compromising carcinologic outcomes. In addition, adjuvant hormonotherapy, low level of RILA, and smoking habits will be arguments against breast reconstruction. In cases where therapeutic alternatives to radiotherapy are not available, a high risk of toxicity could lead to the patient being considered for new radiotherapy techniques.
We did not observe statistical correlation between RILA and tumor recurrences but the number of events is too small to draw any conclusions. In cancers with higher recurrences like locally advanced prostate (Foro et al., 2014) or cervix (Ordonez et al., 2014), such correlations have recently been suggested but this needs to be confirmed in larger prospective cohorts.
In conclusion, this trial is strong enough to support the use of RILA as a predictor of the risk of late effects after RT. This biological assay has to be integrated in a multivariate model to optimise the global evaluation of the risk. We are now entering a new era of personalized treatment and radiation oncology is surely a good example with the development of a biomarker like RILA. Other disciplines like radiology, nuclear medicine, surgery, interventional cardiology or rheumatology that use ionising radiation in their practice have to be involved in further development of this test to better protect patients and professionals.
Author contributions
D.A., A.K., C.H., A.P., S.G., and E.M.O. were involved in the conception and design of the study. D.A., O.R., T.D.N., K.P., C.L., J-L.L., Y.K, E.L., Y.B., C.B., S.R., G.N., S.C., and F.M. were involved in the provision of patients, data acquisition and helped to draft the report.
D.A. and S.G. supervised the study. F.C. and S.G. were in charge of the statistical analysis of the study. D.A., F.C., and S.G. had access to the raw data and analyzed and interpreted the data. P.F. was the referent physicist and coordinated the technical radiotherapy part of the study. D.A., F.C., A.P., S.G., and E.M.O. were involved in writing the report, which was corrected and approved by all authors.
Declaration
ICM Val d'Aurelle (Montpellier) received an unrestricted educational grant (PHRC 2005) to cover the expenses of the investigators for undertaking this trial. David Azria, Mahmut Ozsahin and Sophie Gourgou have a patent protection document submitted pending. The other authors have no conflict of interests related to this article.
Acknowledgments
The authors thank Ms. Carole Jouet for data monitoring and Mr. Michael Coelho for his excellent technical assistance (lymphocyte assay).
The French National Cancer Institute (INCa) through the “Programme Hospitalier de Recherche Clinique (PHRC 2005)” funded this trial. It was also approved and labeled by the French Society of Radiation Oncology (SFRO). The funding agency has no role in the actual experimental design, analysis, or writing of this manuscript.
Footnotes
Presented in oral communication at the 57th Annual Meeting of the American Society for Radiation Oncology (ASTRO), San Antonio, TX, October 18–21, 2015. This work received the Basic/Translational Science Award by the Scientific Committee of ASTRO 2015 and has been selected as one of the few abstracts to be discussed at the 2015 Best of ASTRO meeting, November 13–14, 2015, San Diego.
References
- Al-Ghazal S.K., Fallowfield L., Blamey R.W. Does cosmetic outcome from treatment of primary breast cancer influence psychosocial morbidity? Eur. J. Surg. Oncol. 1999;25(6):571–573. doi: 10.1053/ejso.1999.0708. (Epub 1999/11/11) [DOI] [PubMed] [Google Scholar]
- Altman D.G., McShane L.M., Sauerbrei W., Taube S.E. Reporting recommendations for tumor marker prognostic studies (REMARK): explanation and elaboration. PLoS Med. 2012;9(5) doi: 10.1371/journal.pmed.1001216. (Epub 2012/06/08) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arriagada R., Rutqvist L.E., Kramar A., Johansson H. Competing risks determining event-free survival in early breast cancer. Br. J. Cancer. 1992;66(5):951–957. doi: 10.1038/bjc.1992.391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Azria D., Gourgou S., Sozzi W.J. Concomitant use of tamoxifen with radiotherapy enhances subcutaneous breast fibrosis in hypersensitive patients. Br. J. Cancer. 2004;91(7):1251–1260. doi: 10.1038/sj.bjc.6602146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Azria D., Ozsahin M., Kramar A. Single nucleotide polymorphisms, apoptosis, and the development of severe late adverse effects after radiotherapy. Clin. Cancer Res. 2008;14(19):6284–6288. doi: 10.1158/1078-0432.CCR-08-0700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Azria D., Belkacemi Y., Romieu G. Concurrent or sequential adjuvant letrozole and radiotherapy after conservative surgery for early-stage breast cancer (CO-HO-RT): a phase 2 randomised trial. Lancet Oncol. 2010;11(3):258–265. doi: 10.1016/S1470-2045(10)70013-9. (Epub 2010/02/09) [DOI] [PubMed] [Google Scholar]
- Azria D., Betz M., Bourgier C., Jeanneret Sozzi W., Ozsahin M. Identifying patients at risk for late radiation-induced toxicity. Crit. Rev. Oncol. Hematol. 2012;84(Suppl. 1):e35–e41. doi: 10.1016/j.critrevonc.2010.08.003. (Epub 2010/09/28) [DOI] [PubMed] [Google Scholar]
- Begg A.C., Russell N.S., Knaken H., Lebesque J.V. Lack of correlation of human fibroblast radiosensitivity in vitro with early skin reactions in patients undergoing radiotherapy. Int. J. Radiat. Biol. 1993;64(4):393–405. doi: 10.1080/09553009314551581. [DOI] [PubMed] [Google Scholar]
- Bentzen S.M., Skoczylas J.Z., Overgaard M., Overgaard J. Radiotherapy-related lung fibrosis enhanced by tamoxifen. J. Natl. Cancer Inst. 1996;88(13):918–922. doi: 10.1093/jnci/88.13.918. [DOI] [PubMed] [Google Scholar]
- Bordon E., Henriquez-Hernandez L.A., Lara P.C. Prediction of clinical toxicity in locally advanced head and neck cancer patients by radio-induced apoptosis in peripheral blood lymphocytes (PBLs) Radiat. Oncol. 2010;5:4. doi: 10.1186/1748-717X-5-4. (Epub 2010/01/30) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bordon E., Henriquez-Hernandez L.A., Lara P.C., Pinar B., Rodriguez-Gallego C., Lloret M. Role of CD4 and CD8 T-lymphocytes, B-lymphocytes and Natural Killer cells in the prediction of radiation-induced late toxicity in cervical cancer patients. Int. J. Radiat. Biol. 2011;87(4):424–431. doi: 10.3109/09553002.2010.537433. (Epub 2010/12/15) [DOI] [PubMed] [Google Scholar]
- Canney P.A., Dean S. Transforming growth factor beta: a promotor of late connective tissue injury following radiotherapy? Br. J. Radiol. 1990;63(752):620–623. doi: 10.1259/0007-1285-63-752-620. [DOI] [PubMed] [Google Scholar]
- Fine J.P. Regression modeling of competing crude failure probabilities. Biostatistics. 2001;2(1):85–97. doi: 10.1093/biostatistics/2.1.85. [DOI] [PubMed] [Google Scholar]
- Foro P., Algara M., Lozano J. Relationship between radiation-induced apoptosis of T lymphocytes and chronic toxicity in patients with prostate cancer treated by radiation therapy: a prospective study. Int. J. Radiat. Oncol. Biol. Phys. 2014;88(5):1057–1063. doi: 10.1016/j.ijrobp.2014.01.002. (Epub 2014/03/26) [DOI] [PubMed] [Google Scholar]
- Johansen J., Bentzen S.M., Overgaard J., Overgaard M. Relationship between the in vitro radiosensitivity of skin fibroblasts and the expression of subcutaneous fibrosis, telangiectasia, and skin erythema after radiotherapy. Radiother. Oncol. 1996;40(2):101–109. doi: 10.1016/0167-8140(96)01777-x. [DOI] [PubMed] [Google Scholar]
- Johansen J., Overgaard J., Overgaard M. Effect of adjuvant systemic treatment on cosmetic outcome and late normal-tissue reactions after breast conservation. Acta Oncol. 2007;46(4):525–533. doi: 10.1080/02841860701291698. (Epub 2007/05/15) [DOI] [PubMed] [Google Scholar]
- Kern P., Zarth F., Kimmig R., Rezai M. Impact of age, obesity and smoking on patient satisfaction with breast implant surgery — a unicentric analysis of 318 implant reconstructions after mastectomy. Geburtshilfe Frauenheilkd. 2015;75(6):597–604. doi: 10.1055/s-0035-1546171. (Epub 2015/07/15) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kerns S.L., de Ruysscher D., Andreassen C.N. STROGAR — STrengthening the Reporting Of Genetic Association studies in Radiogenomics. Radiother. Oncol. 2014;110(1):182–188. doi: 10.1016/j.radonc.2013.07.011. (Epub 2013/09/03) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kramar A., Faraggi D., Fortune A., Reiser B. mROC: a computer program for combining tumour markers in predicting disease states. Comput. Methods Prog. Biomed. 2001;66(2-3):199–207. doi: 10.1016/s0169-2607(00)00129-2. [DOI] [PubMed] [Google Scholar]
- Lacombe J., Azria D., Mange A., Solassol J. Proteomic approaches to identify biomarkers predictive of radiotherapy outcomes. Expert Rev. Proteomics. 2013;10(1):33–42. doi: 10.1586/epr.12.68. (Epub 2013/02/19) [DOI] [PubMed] [Google Scholar]
- Lambin P., van Stiphout R.G., Starmans M.H. Predicting outcomes in radiation oncology—multifactorial decision support systems. Nat. Rev. Clin. Oncol. 2013;10(1):27–40. doi: 10.1038/nrclinonc.2012.196. (Epub 2012/11/21) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ordonez R., Henriquez-Hernandez L.A., Federico M. Radio-induced apoptosis of peripheral blood CD8 T lymphocytes is a novel prognostic factor for survival in cervical carcinoma patients. Strahlenther. Onkol. 2014;190(2):210–216. doi: 10.1007/s00066-013-0488-x. (Epub 2013/12/24) [DOI] [PubMed] [Google Scholar]
- Ozsahin M., Crompton N.E., Gourgou S. CD4 and CD8 T-lymphocyte apoptosis can predict radiation-induced late toxicity: a prospective study in 399 patients. Clin. Cancer Res. 2005;11(20):7426–7433. doi: 10.1158/1078-0432.CCR-04-2634. [DOI] [PubMed] [Google Scholar]
- Pepe M.S., Mori M. Kaplan–Meier, marginal or conditional probability curves in summarizing competing risks failure time data? Stat. Med. 1993;12(8):737–751. doi: 10.1002/sim.4780120803. [DOI] [PubMed] [Google Scholar]
- Peto R., Pike M.C., Armitage P. Design and analysis of randomized clinical trials requiring prolonged observation of each patient. II. Analysis and examples. Br. J. Cancer. 1977;35(1):1–39. doi: 10.1038/bjc.1977.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pinar B., Henriquez-Hernandez L.A., Lara P.C. Radiation induced apoptosis and initial DNA damage are inversely related in locally advanced breast cancer patients. Radiat. Oncol. 2010;5:85. doi: 10.1186/1748-717X-5-85. (Epub 2010/09/28) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schnarr K., Boreham D., Sathya J., Julian J., Dayes I.S. Radiation-induced lymphocyte apoptosis to predict radiation therapy late toxicity in prostate cancer patients. Int. J. Radiat. Oncol. Biol. Phys. 2009;74(5):1424–1430. doi: 10.1016/j.ijrobp.2008.10.039. (Epub 2009/01/27) [DOI] [PubMed] [Google Scholar]
- Seibold P., Behrens S., Schmezer P. XRCC1 polymorphism associated with late toxicity after radiation therapy in breast cancer patients. Int. J. Radiat. Oncol. Biol. Phys. 2015;92(5):1084–1092. doi: 10.1016/j.ijrobp.2015.04.011. (Epub 2015/06/15) [DOI] [PubMed] [Google Scholar]
- Trotti A., Colevas A.D., Setser A. CTCAE v3.0: development of a comprehensive grading system for the adverse effects of cancer treatment. Semin. Radiat. Oncologia. 2003;13(3):176–181. doi: 10.1016/S1053-4296(03)00031-6. [DOI] [PubMed] [Google Scholar]
- West C., Azria D., Chang-Claude J. The REQUITE project: validating predictive models and biomarkers of radiotherapy toxicity to reduce side-effects and improve quality of life in cancer survivors. Clin. Oncol. (R. Coll. Radiol.) 2014;26(12):739–742. doi: 10.1016/j.clon.2014.09.008. (Epub 2014/10/01) [DOI] [PubMed] [Google Scholar]