Abstract
Rocaglates are natural products that inhibit protein synthesis in eukaryotes and exhibit anti-neoplastic activity. In vitro biochemical assays, affinity chromatography experiments coupled with mass spectrometry analysis, and in vivo genetic screens have identified eukaryotic initiation factor (eIF) 4A as a direct molecular target of rocaglates. eIF4A is the RNA helicase subunit of eIF4F, a complex which mediates cap-dependent ribosome recruitment to mRNA templates. The eIF4F complex has been implicated in tumor initiation and maintenance through elevated levels or increased phosphorylation status of its cap-binding subunit, eIF4E, thus furthering the interest towards developing rocaglates as anti-neoplastic agents. Recent experiments have indicated that rocaglates also interact with prohibitins (PHB) 1 and 2; proteins implicated in c-Raf-MEK-ERK signaling. Since increased ERK signaling stimulates eIF4E phosphorylation status, rocaglates are also expected to inhibit eIF4E phosphorylation status, a point that has not been thoroughly investigated. It is currently unknown whether the effects on translation observed with rocaglates are solely through eIF4A inhibition or also a feature of blocking eIF4E phosphorylation. Here, we show that rocaglates inhibit translation through an eIF4E-phophorylation independent mechanism.
Keywords: Rocaglates, eIF4A, eIF4E, Mnk, translational control, chemical biology
INTRODUCTION
Rocaglates are a family of natural products characterized by a common cyclopenta[b]furan skeleton exclusively found in the Aglaia genus of the Meliaceae family of angiosperms (1). Many members of this family are potent inhibitors of translation initiation and exhibit single agent anti-neoplastic activity in pre-clinical cell and mouse models (2–4) as well as the ability to modulate chemo-responsiveness (5–9). A significant body of evidence indicates that these compounds inhibit translation initiation by disabling eIF4F and interfering with ribosome recruitment to mRNA templates (2, 5, 9).
eIF4F is a hetero-trimeric complex consisting of eIF4E, a cap-binding protein; eIF4A, the RNA helicase target of rocaglates; and eIF4G, a large scaffolding protein. eIF4F is required to unwind cap-proximal secondary structure within the mRNA 5' untranslated region (UTR) as a prelude to 43S pre-initiation complex recruitment. Of all the initiation factors, eIF4E is the least abundant (10, 11) and mRNAs must compete for the limiting amounts of eIF4F during the initiation process. One determinant of competitive efficiency is the presence of structural barriers (e.g. stem-loop structures, protein-RNA interactions) within the mRNA 5' UTR, with elevated levels associated with poorly initiating mRNAs. Consequently, translation of these weaker mRNAs is most affected upon eIF4F inhibition, and hence by rocaglates (2, 12–14).
eIF4A is an abundant factor that exists as a free form (eIF4Af) or as part of the eIF4F complex (eIF4Ac). Biochemical assays using recombinant eIF4A (2, 5, 9), affinity chromatography experiments using immobilized epi-silvestrol (15), and chemogenomic profiling in yeast (16) have identified eIF4A as a predominant target of rocaglates. Rocaglates are thought to restrict efficient recycling of eIF4A through the eIF4F complex by increasing non-specific RNA binding of eIF4A (Fig. 1A) (8).
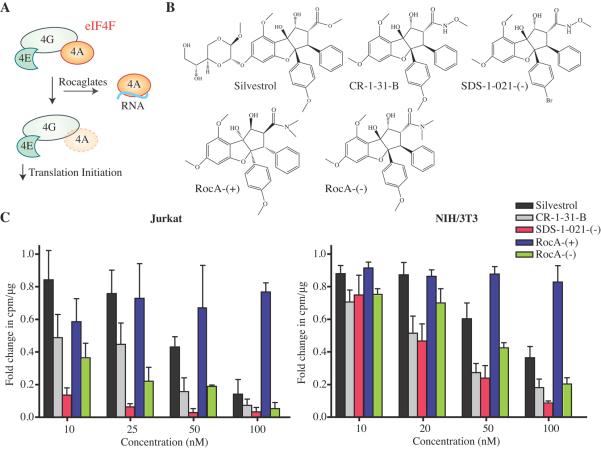
Figure 1.
A. Proposed mechanism of action of rocaglates on eIF4A recycling through the eIF4F complex. In this model, rocaglates stimulate eIF4A RNA binding, rendering it unavailable to enter into the eIF4F complex. B. Structures of rocaglates used in this study. C. Dose-dependent inhibition of translation by rocaglates in Jurkat and NIH/3T3 cells. Cells were incubated in the presence of compound for a total of 2 hours and protein synthesis rates were determined as described in the Materials and Methods. The relative rates are of translation are calculated by normalizing to DMSO. n = 4; error bars represent the error of the mean.
An alternative mechanism of rocaglate activity on translation initiation has been proposed based on their interactions with prohibitins (PHB) 1 and 2 (17). PHB1/2 are involved in a wide variety of cellular processes, including activation of the MAPK signaling cascade through direct interaction with c-RAF (18). In the presence of rocaglates, the PHB1/2:c-RAF interaction is inhibited, leading to dampened signaling through MEK and ERK 1/2 (17). Since MNK are activated by ERK signaling and phosphorylate eIF4E on S209, rocaglates are expected to inhibit eIF4E S209 phosphorylation. However, the effect of rocaglates on eIF4E phosphorylation status has yet to be assessed. If correct, this mechanism of action would have profound consequences on our understanding of the anti-neoplastic effects of these compounds since eIF4E phosphorylation is essential to its oncogenic activity (19, 20). As well, transcriptome-wide studies attributing alterations in mRNA translational efficiency to eIF4A inhibition by rocaglates would have to be re-interpreted if inhibition of eIF4E phosphorylation was a significant biological property of rocaglates (12, 13).
However, there are several lines of evidence inconsistent with inhibition of eIF4E phosphorylation contributing to the biological activity of rocaglates. Firstly, rocaglates are potent inhibitors of eIF4A-dependent translation in vitro where RAS/MEK/ERK signaling is not maintained (5). Secondly, rocaglates have been shown to inhibit encephalomyocarditis (EMC) IRES-driven translation (5), an event that is eIF4A, but not eIF4E, dependent (21). Thirdly, rocaglates are potent inhibitors of global cap-dependent translation (2, 5), whereas loss of eIF4E S209 phosphorylation leads to more selective inhibition of mRNA translation (20, 22–24). It therefore remains an open question as to whether the reported suppression of RAS/MEK/ERK signaling by rocaglates represents an essential feature of these compounds. Herein, we report that the biological activity of rocaglates cannot be explained by modulation of eIF4E phosphorylation.
MATERIALS AND METHODS
General Methods and Reagents
Jurkat (in 2002) and NIH/3T3 (in 2005) cells were obtained from the American Type Culture Collection (ATCC). Mnk1+/+Mnk2+/+ and Mnk1−/−Mnk2−/− MEFs were obtained from Dr. Nahum Sonenberg in 2010 (McGill University, Montreal, Quebec, Canada). RAS-transformed NIH/3T3 cells were generated by infection with pBABE-Puro.H-Ras(V12) in 2015. Cells lines were not further authenticated. Jurkat, NIH/3T3, RAS-transformed NIH/3T3, and Mnk1+/+Mnk2+/+ and Mnk1−/−Mnk2−/− MEFs (Mouse Embryonic Fibroblasts) were grown in RPMI 1640 and DMEM supplemented with 10% FBS and 100 U/ml penicillin/streptomycin at 37°C and 5% CO2. Cell extracts were prepared in lysis buffer (20 mM HEPES [pH 7.5], 150 mM NaCl, 1% Triton-X100, 10% glycerol, 1 mM EDTA, 10 mM tetrasodium pyrophosphate, 100 mM NaF, 17.5 mM β-glycerophosphate, 1mM PMSF, 4 μg/ml aprotinin, 2 μg/ml leupeptin, 2 μg/ml pepstatin). Extracts from Jurkat cells were prepared by lysing cells in 1X NuPAGE LDS sample buffer (26.5 mM Tris HCl [pH 8.5], 35.25 mM Tris Base, 0.5% LDS, 2.5 % glycerol, 0.1275 mM EDTA). Protein samples were fractionated on 10% polyacrylamide gels, and transferred to PVDF membranes (Bio-Rad). Antibodies used in this study were directed against: p-eIF4E (Ser209) (#9741, Cell Signaling), eIF4E (#9742, Cell Signaling), p-ERK 1/2 (#9106, Cell Signaling), ERK 1/2 (#9102, Cell Signaling), eEF2 (#2332, Cell Signaling), and GAPDH (ab8245, Abcam). SDS-1-021-(−), Roc A-(−), and Roc A-(+) were synthesized using biomimeic kinetic resolution of chiral, racemic aglain ketone precursors as recently published (25) followed by amide formation (26). Silvestrol and CR-1-31-B were synthesized as previously reported (9, 27). The concentrations of compound used were based on our experience with this class of small molecules, as well as previously published studies (2, 5, 6, 9, 12).
Cell Labeling and TCA Precipitations
35S-methionine/cysteine protein labeling was performed as previously described (11). Essentially, the day prior to metabolic labeling, Jurkat cells were seeded at 500,000 cells/ml and NIH 3T3, Mnk1+/+Mnk2+/+, and Mnk1−/−Mnk2−/− MEFs were seeded at 250 cells/mm2. On the day of labeling, cells were exposed to the indicated concentrations of rocaglates for 2 hours. During the last 30 minutes of incubation, [35S]-methionine/cysteine was added (150–200 μCi/ml; 1175 Ci/mmole) (Perkin Elmer, Waltham, MA) and the labeling reactions were terminated with the addition of RIPA lysis buffer (20 mM Tris [pH 7.6], 100 mM NaCl, 1 mM EDTA, 1 mM EGTA, 1% NP40, 0.5% Sodium Deoxycholate, 0.1% SDS, 1 mM PMSF, 4 μg/ml aprotinin, 2 μg/ml leupeptin, 2 μg/ml pepstatin). Lysates were then spotted onto 3 MM Whatman paper (1.5 cm2 squares) that had been preblocked with MEM Non-Essential Amino Acid mixture (Gibco). After drying, filters were submerged in ice-cold 10% trichloroacetic acid (TCA)/ 0.1% L-methionine for 20 minutes, followed by boiling in 5% TCA for 15 minutes. The Whatman squares were washed twice with cold 5% TCA and then twice more with 95% ethanol, with each wash lasting one minute. Filter squares were dried and the amount of radiolabelled precipitated protein was quantitated by scintillation counting (Beckman Coulter). Counts were standardized to total protein content that had been previously determined using the DC protein assay (Bio-Rad).
RNA Binding assay
Body-labeled [32P]-labeled RNA was produced by in vitro transcription of pSP/CAT (linearized with PvuII) using SP6 RNA polymerase. Recombinant eIF4AI was purified as previously reported (28). Binding assays were performed by incubating [32P]-labeled RNA (35000 cpm) with recombinant eIF4AI in binding buffer (25 mM Tris [pH 7.5], 1 mM DTT, 100 mM KCl, 1 mM ATP, 5 mM MgCl2) in the presence of 5 μM of the indicated compound or 0.1% DMSO for 10 minutes at 37 °C, as previously noted (9). No pre-incubations were performed and when reactions were assembled rocaglates were added pentultimately before the addition of [32P]-labeled RNA. Reactions were terminated by the addition of 1 mL stop buffer (25 mM Tris [pH 7.5], 100 mM KCl, 3 mM MgCl2) and then filtered through nitrocellulose (45 μM HA Millipore) (preblocked with 0.1% sodium pyrophosphate). Filters were washed 3 times with 1 mL stop buffer, dried, and the amount of retained [32P]-labeled RNA quantitated by liquid scintillation counting.
RESULTS
Our previous work investigating the consequences of silvestrol (2, 5), CR-1-31-B [a.k.a. hydroxamate (−)-9] (9) and SDS-1-021-(−) (RC., unpublished observations) (Fig. 1B) on translation indicate that these compounds target eIF4A and prevent its entry/recycling into the eIF4F complex (Fig. 1A). Li-Weber and colleagues (17) have reported that rocaglates, such as RocA (Fig. 1B) can also inhibit PHB1/2 to downregulate ERK activation, although the downstream effects on eIF4E S209 phosphorylation were never reported. We therefore decided to investigate this potential relationship and also took the opportunity to synthesize enantio-enriched preparations of RocA to tease out possible biological differences between the stereoisomers (Fig. 1B).
To assess the relative potencies of the rocaglates on translation, we performed a series of titrations in Jurkat and NIH/3T3 cells and measured relative levels of protein synthesis using [35S]-methionine/cysteine metabolic labeling. To ensure that the measured values reflected the immediate effects of the compounds on translation, cells were exposed to the indicated rocaglate for only 2 hours, which was also the shortest incubation period used by Li-Weber and colleagues to assess translation inhibition (17). Concentrations tested ranged from 10 nM to 100 nM, as rocaglates have been previously reported to be effective inhibitors of translation at nanomolar concentrations (2). Based on the relative IC50s, as defined by the concentration of compound required to inhibit translation by 50%, SDS-1-021-(−) was found to be the most potent rocaglate (IC50 < 10 nM) in Jurkat cells, followed by RocA-(−) and CR-1-31-B (Fig. 1C). In NIH/3T3 cells, SDS-1-021-(−) and CR-1-31-B showed similar potencies, with IC50s ~20 nM for translation inhibition under our test conditions (Fig. 1C). Silvestrol and RocA-(−) were slightly less potent with IC50s of ~50 nM. RocA-(+) did not inhibit translation in either cell line highlighting the importance of compound stereochemistry for biological activity, as previously noted (9). In sum, these results indicate that individual rocaglates exert differences in their ability to inhibit translation across cell lines, as previously reported (5, 9), and that within this small series, SDS-1-021-(−) is the most potent compound tested (Fig. 1C).
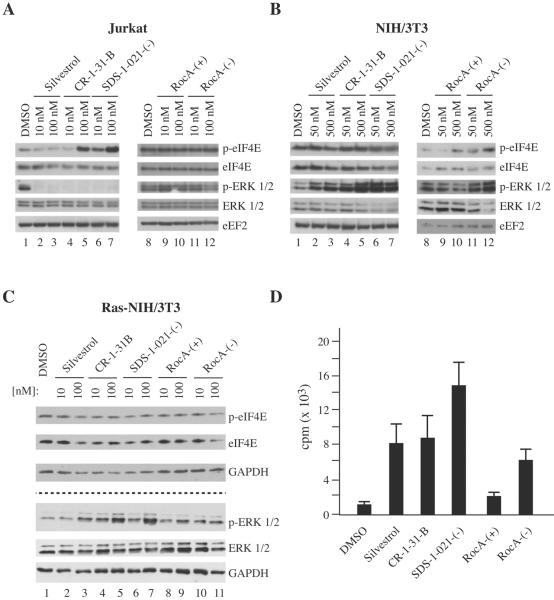
RocA has been shown to inhibit ERK 1/2 phosphorylation in Jurkat cells (17), although its effects on eIF4E phosphorylation have never been reported. We found that, with the exception of RocA-(+), all other rocaglates suppressed ERK 1/2 phosphorylation in Jurkat cells (Fig. 2A). Silvestrol, CR-1-31-B, and SDS-1-021-(−) completely blocked phosphorylation and modest inhibition was observed at 100 nM RocA-(−) (Fig. 2A). Surprisingly, eIF4E phosphorylation was not inhibited and, contrary to expectations, was stimulated by CR-1-31-B and SDS-1-021-(−). In contrast, in NIH/3T3 cells we observed stimulation, not inhibition, of ERK 1/2 phosphorylation by all rocaglates tested, with the exception of the inactive RocA-(+) enantiomer, compared to vehicle-treated cells (Fig. 2B). None of the tested rocaglates affected phospho-eIF4E status in NIH/3T3 cells under the tested conditions. To exclude the possibility that the absence of diminished eIF4E phosphorylation is a consequence of low levels of MEK/ERK activity in NIH/3T3 cells, we transformed the cells with RAS in order to elevate ERK signaling and treated with the rocaglate series (Fig. 2C). All rocaglates tested stimulated p-ERK levels in RAS-transformed NIH/3T3 cells with little effect on p-eIF4E status(Fig. 2C). This is consistent with what was noted in non-transformed NIH/3T3 cells and we have not further investigated the underlying molecular mechanism. Nonetheless, there is little correlation between ERK and eIF4E phosphorylation status with the inhibition of translation documented above (Fig. 1C). One well-characterized activity of rocaglates is stimulation of the RNA-binding activity of eIF4A (2, 5, 9). To assess whether the rocaglate series under evaluation retained this activity, we performed RNA filter binding assays in vitro using 32P-labelled RNA in the presence of 5 μM of each compound, which is within the concentration range of silvestrol previously shown to stimulate RNA binding of eIF4A (2) (Fig. 2D). With the exception of the inactive RocA-(+) enantiomer, all rocaglates stimulated binding of eIF4A to RNA, with SDS-1-021-(−) being the most potent compound (Fig. 2D).
Figure 2.
A. Effects of rocaglates on eIF4E phosphorylation in Jurkat cells. Cells were incubated in the presence of the indicated compounds for 2 hours, lysed, fractionated on a 10% NuPAGE Bis-Tris gel, and transferred to PVDF membranes for western blotting. Blots were probed using antibodies directed to the proteins indicated to the right of the panel. B. Effects of rocaglates on eIF4E phosphorylation status in NIH/3T3 cells. Cells were treated, proteins fractionated, transferred to PVDF membranes, and western blots analyzed as described for Panel A. C. Effects of rocaglates on eIF4E phosphorylation status in RAS-transformed NIH/3T3 cells. Cells were treated, proteins fractionated, transferred to PVDF membranes, and western blots analyzed as described for Panel A. The dashed line indicates that probing for eIF4E and ERK were performed on different membranes. GAPDH levels were used as an internal standard to account for variations in extract levels between lanes. D. Stimulation of eIF4A:RNA binding by rocaglates. Recombinant eIF4A (1.3 μM) was incubated with 35 000 cpm of 32P-labelled RNA in the presence of 5 μM rocaglate and processed as described in the Materials and Methods. eIF4A:RNA complexes retained on nitrocellulose filters were quantitated by scintillation counting. N = 3; error bars represent error of the mean.
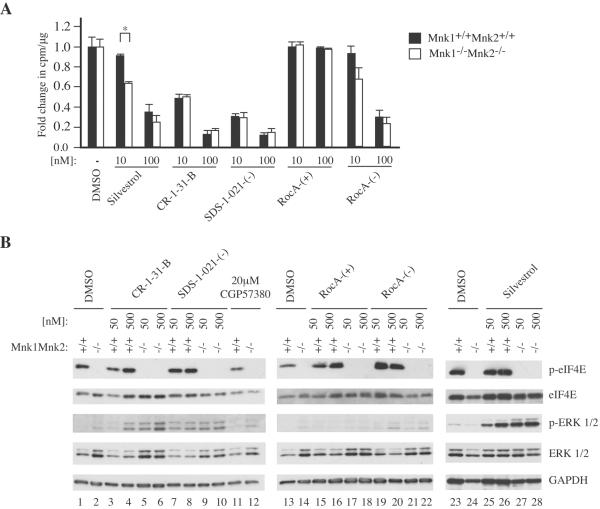
To further support the notion that eIF4E phosphorylation status is inconsequential to the inhibition of translation observed with rocaglates, we quantitated the effects of rocaglates on protein synthesis in Mnk1−/−Mnk2−/− MEFs, where eIF4E cannot be phosphorylated (Fig. 3A) (29). Whereas we observed a slight difference in the sensitivities between Mnk1−/−Mnk2−/− and Mnk1+/+Mnk2+/+ MEFs (~40%) towards silvestrol, there was no significant difference on translation exerted by the other tested rocaglates in these two cell types (Fig. 3A). Consistent with our results in Jurkat and NIH/3T3 cells, SDS-1-021-(−) was the most potent inhibitor among the series tested (Fig. 3A). Western blots of extracts confirmed the complete absence of eIF4E phosphorylation in the Mnk1−/−Mnk2−/− MEFs as well as the absence of diminished eIF4E phosphorylation in Mnk1+/+Mnk2+/+ MEFs (Fig. 3B). As a positive control, we included the Mnk1 inhibitor, CGP57380, and observed reduced phospho-eIF4E levels in Mnk1+/+Mnk2+/+ MEFs (Fig. 3B) (30).
Figure 3.
A. Rocaglates inhibit translation in Mnk1+/+Mnk2+/+ and Mnk1−/−Mnk2−/− MEFs. Protein synthesis rates were determined as described in the Materials and Methods. The relative rates of 35S-Met incorporation are normalized to DMSO. N = 4; error bars represent error of the mean. *, p < 0.001 (Student's t-test). B. Effects of rocaglates on eIF4E phosphorylation in Mnk1+/+Mnk2+/+ and Mnk1−/−Mnk2−/− cells. Cells were incubated in the presence of the indicated compounds for two hours, lysed, fractionated on a 10% polyacrylamide gel, and transferred to PVDF membranes for western blotting. Blots were probed with antibodies directed to the proteins indicated to the right of the panel. GAPDH levels were used as an internal standard to account for variations in extract levels between lanes. Note that lane 24 is slightly underloaded based on the GAPDH internal standard.
DISCUSSION
In this study, we report that translation inhibition by rocaglates is independent of eIF4E phosphorylation status. Although we have not directly tested the ability of our rocaglate series to inhibit PHB1/2:c-RAF association, RocA has been previously shown to block this interaction (17). We find that the effects of rocaglates on p-ERK 1/2 and p-eIF4E status appear cell-type dependent and overall do not correlate with rocaglate-induced translation inhibition (Fig. 2). It is clear that in the complete absence of eIF4E phosphorylation, the ability of CR-1-31-B, SDS-1-021-(−), and RocA-(−) to inhibit protein synthesis is unperturbed (Fig. 3). Silvestrol, but not CR-1-31-B, is a known Pgp-1 multi-drug transporter substrate (31) (R.C. and J.P., unpublished observations). Whether the increased sensitivity of Mnk1−/−Mnk2−/− cells to silvestrol is due to reduced expression of Pgp-1 and/or other drug response modifiers remains to be evaluated (Fig. 3A).
As reported by Li-Weber and colleagues (17), we also find that rocaglates inhibit ERK 1/2 phosphorylation in Jurkat cells (Fig. 2A). However, in NIH/3T3 cells we observed stimulation, not inhibition, of this post-translational modification (Fig. 2B, C). The increase in p-eIF4E that we observed with the more potent SDS-1-021-(−) and CR-1-31-B compounds in Jurkat cells is unlinked to p-ERK 1/2 status and may reflect activation of a stress kinase response—an effect that has been previously documented with other translation inhibitors including anisomycin, onnamide A, and theopederin B (32, 33). This increase in p-eIF4E levels is contrary to what would be expected upon ERK 1/2 inhibition, which one would expect to stimulate, not inhibit, selective mRNA translation (20, 22–24).
Our results do not rule out the possibility that some rocaglate family members not tested here can block eIF4E phosphorylation given the appropriate context. Indeed, the compound RocAR has been reported to exhibit this activity in HTLV-infected ATL (adult T-cell leukemia/lymphoma) cells (34). However, our data indicate that this is not a general feature of this class of compounds, and that rocaglate-induced translation inhibition is independent of eIF4E phosphorylation status. Taken together with previous data indicating that these compounds also do not inhibit translation by increasing eIF2α phosphorylation (5), the consequences of rocaglates on translation appear best explained by their effects on eIF4A activity.
Acknowledgments
This work was supported by grants from the Canadian Institutes of Health Research to J. Pelletier (MOP-106530) and the National Institutes of Health (R01 GM073855 to J.A.Porco, Jr.). Research at the Center for Chemical Methodology and Library Development at Boston University (CMLD-BU) was supported by NIH grant GM-067041.
Footnotes
None of the authors have any potential financial conflict of interest related to this manuscript
REFERENCES
- 1.Ebada SS, Lajkiewicz N, Porco JA, Jr, Li-Weber M, Proksch P. Chemistry and biology of rocaglamides (= flavaglines) and related derivatives from aglaia species (meliaceae) Prog Chem Org Nat Prod. 2011;94:1–58. doi: 10.1007/978-3-7091-0748-5_1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Cencic R, Carrier M, Galicia-Vazquez G, Bordeleau ME, Sukarieh R, Bourdeau A, et al. Antitumor activity and mechanism of action of the cyclopenta[b]benzofuran, silvestrol. PLoS One. 2009;4:e5223. doi: 10.1371/journal.pone.0005223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lucas DM, Edwards RB, Lozanski G, West DA, Shin JD, Vargo MA, et al. The novel plant-derived agent silvestrol has B-cell selective activity in chronic lymphocytic leukemia and acute lymphoblastic leukemia in vitro and in vivo. Blood. 2009;113:4656–66. doi: 10.1182/blood-2008-09-175430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Alachkar H, Santhanam R, Harb JG, Lucas DM, Oaks JJ, Hickey CJ, et al. Silvestrol exhibits significant in vivo and in vitro antileukemic activities and inhibits FLT3 and miR-155 expressions in acute myeloid leukemia. Journal of hematology & oncology. 2013;6:21. doi: 10.1186/1756-8722-6-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bordeleau ME, Robert F, Gerard B, Lindqvist L, Chen SM, Wendel HG, et al. Therapeutic suppression of translation initiation modulates chemosensitivity in a mouse lymphoma model. J Clin Invest. 2008;118:2651–60. doi: 10.1172/JCI34753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Robert F, Roman W, Bramoulle A, Fellmann C, Roulston A, Shustik C, et al. Translation initiation factor eIF4F modifies the dexamethasone response in multiple myeloma. Proc Natl Acad Sci U S A. 2014;111:13421–6. doi: 10.1073/pnas.1402650111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Giaisi M, Kohler R, Fulda S, Krammer PH, Li-Weber M. Rocaglamide and a XIAP inhibitor cooperatively sensitize TRAIL-mediated apoptosis in Hodgkin's lymphomas. Int J Cancer. 2012;131:1003–8. doi: 10.1002/ijc.26458. [DOI] [PubMed] [Google Scholar]
- 8.Chu J, Pelletier J. Targeting the eIF4A RNA helicase as an anti-neoplastic approach. Biochim Biophys Acta. 2015;1849:781–791. doi: 10.1016/j.bbagrm.2014.09.006. [DOI] [PubMed] [Google Scholar]
- 9.Rodrigo CM, Cencic R, Roche SP, Pelletier J, Porco JA. Synthesis of rocaglamide hydroxamates and related compounds as eukaryotic translation inhibitors: synthetic and biological studies. J Med Chem. 2012;55:558–62. doi: 10.1021/jm201263k. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Duncan R, Hershey JW. Identification and quantitation of levels of protein synthesis initiation factors in crude HeLa cell lysates by two-dimensional polyacrylamide gel electrophoresis. J Biol Chem. 1983;258:7228–35. [PubMed] [Google Scholar]
- 11.Galicia-Vazquez G, Cencic R, Robert F, Agenor AQ, Pelletier J. A cellular response linking eIF4AI activity to eIF4AII transcription. RNA. 2012;18:1373–84. doi: 10.1261/rna.033209.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wolfe AL, Singh K, Zhong Y, Drewe P, Rajasekhar VK, Sanghvi VR, et al. RNA G-quadruplexes cause eIF4A-dependent oncogene translation in cancer. Nature. 2014;513:65–70. doi: 10.1038/nature13485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Rubio CA, Weisburd B, Holderfield M, Arias C, Fang E, DeRisi JL, et al. Transcriptome-wide characterization of the eIF4A signature highlights plasticity in translation regulation. Genome Biol. 2014;15:476. doi: 10.1186/s13059-014-0476-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Liu T, Nair SJ, Lescarbeau A, Belani J, Peluso S, Conley J, et al. Synthetic silvestrol analogues as potent and selective protein synthesis inhibitors. J Med Chem. 2012;55:8859–78. doi: 10.1021/jm3011542. [DOI] [PubMed] [Google Scholar]
- 15.Chambers JM, Lindqvist LM, Webb A, Huang DC, Savage GP, Rizzacasa MA. Synthesis of biotinylated episilvestrol: highly selective targeting of the translation factors eIF4AI/II. Org Lett. 2013;15:1406–9. doi: 10.1021/ol400401d. [DOI] [PubMed] [Google Scholar]
- 16.Sadlish H, Galicia-Vazquez G, Paris CG, Aust T, Bhullar B, Chang L, et al. Evidence for a functionally relevant rocaglamide binding site on the eIF4A-RNA complex. ACS Chem Biol. 2013;8:1519–27. doi: 10.1021/cb400158t. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Polier G, Neumann J, Thuaud F, Ribeiro N, Gelhaus C, Schmidt H, et al. The natural anticancer compounds rocaglamides inhibit the Raf-MEK-ERK pathway by targeting prohibitin 1 and 2. Chemistry & biology. 2012;19:1093–104. doi: 10.1016/j.chembiol.2012.07.012. [DOI] [PubMed] [Google Scholar]
- 18.Rajalingam K, Wunder C, Brinkmann V, Churin Y, Hekman M, Sievers C, et al. Prohibitin is required for Ras-induced Raf-MEK-ERK activation and epithelial cell migration. Nat Cell Biol. 2005;7:837–43. doi: 10.1038/ncb1283. [DOI] [PubMed] [Google Scholar]
- 19.Topisirovic I, Ruiz-Gutierrez M, Borden KL. Phosphorylation of the eukaryotic translation initiation factor eIF4E contributes to its transformation and mRNA transport activities. Cancer Res. 2004;64:8639–42. doi: 10.1158/0008-5472.CAN-04-2677. [DOI] [PubMed] [Google Scholar]
- 20.Wendel HG, Silva RL, Malina A, Mills JR, Zhu H, Ueda T, et al. Dissecting eIF4E action in tumorigenesis. Genes Dev. 2007;21:3232–7. doi: 10.1101/gad.1604407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Pestova TV, Shatsky IN, Hellen CU. Functional dissection of eukaryotic initiation factor 4F: the 4A subunit and the central domain of the 4G subunit are sufficient to mediate internal entry of 43S preinitiation complexes. Mol Cell Biol. 1996;16:6870–8. doi: 10.1128/mcb.16.12.6870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Furic L, Rong L, Larsson O, Koumakpayi IH, Yoshida K, Brueschke A, et al. eIF4E phosphorylation promotes tumorigenesis and is associated with prostate cancer progression. Proc Natl Acad Sci U S A. 2010;107:14134–9. doi: 10.1073/pnas.1005320107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Robichaud N, Del Rincon SV, Huor B, Alain T, Petruccelli LA, Hearnden J, et al. Phosphorylation of eIF4E promotes EMT and metastasis via translational control of SNAIL and MMP-3. Oncogene. 2014 doi: 10.1038/onc.2014.146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ueda T, Sasaki M, Elia AJ, Chio, Hamada K, Fukunaga R, et al. Combined deficiency for MAP kinase-interacting kinase 1 and 2 (Mnk1 and Mnk2) delays tumor development. Proc Natl Acad Sci U S A. 2010;107:13984–90. doi: 10.1073/pnas.1008136107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Stone SD, Lajkiewicz NJ, Whitesell L, Hilmy A, Porco JA., Jr Biomimetic kinetic resolution: highly enantio- and diastereoselective transfer hydrogenation of aglain ketones to access flavagline natural products. J Am Chem Soc. 2015;137:525–30. doi: 10.1021/ja511728b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Gerard B, Sangji S, O'Leary DJ, Porco JA., Jr Enantioselective photocycloaddition mediated by chiral Bronsted acids: asymmetric synthesis of the rocaglamides. J Am Chem Soc. 2006;128:7754–5. doi: 10.1021/ja062621j. [DOI] [PubMed] [Google Scholar]
- 27.Gerard B, Cencic R, Pelletier J, Porco JA., Jr Enantioselective synthesis of the complex rocaglate (−)-silvestrol. Angew Chem Int Ed Engl. 2007;46:7831–4. doi: 10.1002/anie.200702707. [DOI] [PubMed] [Google Scholar]
- 28.Cencic R, Galicia-Vazquez G, Pelletier J. Inhibitors of translation targeting eukaryotic translation initiation factor 4A. Methods Enzymol. 2012;511:437–61. doi: 10.1016/B978-0-12-396546-2.00020-6. [DOI] [PubMed] [Google Scholar]
- 29.Ueda T, Watanabe-Fukunaga R, Fukuyama H, Nagata S, Fukunaga R. Mnk2 and Mnk1 Are Essential for Constitutive and Inducible Phosphorylation of Eukaryotic Initiation Factor 4E but Not for Cell Growth or Development. Mol Cell Biol. 2004;24:6539–49. doi: 10.1128/MCB.24.15.6539-6549.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Tschopp C, Knauf U, Brauchle M, Zurini M, Ramage P, Glueck D, et al. Phosphorylation of eIF-4E on Ser 209 in response to mitogenic and inflammatory stimuli is faithfully detected by specific antibodies. Mol Cell Biol Res Commun. 2000;3:205–11. doi: 10.1006/mcbr.2000.0217. [DOI] [PubMed] [Google Scholar]
- 31.Cencic R, Robert F, Galicia-Vazquez G, Malina A, Ravindar K, Somaiah R, et al. Modifying chemotherapy response by targeted inhibition of eukaryotic initiation factor 4A. Blood cancer journal. 2013;3:e128. doi: 10.1038/bcj.2013.25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wang X, Flynn A, Waskiewicz AJ, Webb BL, Vries RG, Baines IA, et al. The phosphorylation of eukaryotic initiation factor eIF4E in response to phorbol esters, cell stresses, and cytokines is mediated by distinct MAP kinase pathways. J Biol Chem. 1998;273:9373–7. doi: 10.1074/jbc.273.16.9373. [DOI] [PubMed] [Google Scholar]
- 33.Lee KH, Nishimura S, Matsunaga S, Fusetani N, Horinouchi S, Yoshida M. Inhibition of protein synthesis and activation of stress-activated protein kinases by onnamide A and theopederin B, antitumor marine natural products. Cancer Sci. 2005;96:357–64. doi: 10.1111/j.1349-7006.2005.00055.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bleumink M, Kohler R, Giaisi M, Proksch P, Krammer PH, Li-Weber M. Rocaglamide breaks TRAIL resistance in HTLV-1-associated adult T-cell leukemia/lymphoma by translational suppression of c-FLIP expression. Cell Death Differ. 2011;18:362–70. doi: 10.1038/cdd.2010.99. [DOI] [PMC free article] [PubMed] [Google Scholar]