Abstract
Background
Cadaveric studies have examined disc degeneration at the L4-L5 and L5-S1 motion segments; however, we are not aware of another study that has examined the relationship between bilateral spondylolysis and its effect on degenerative disc disease at those levels. This may have been overlooked by researchers owing to the majority of spondylolysis occurring at the L5 vertebra.
Questions/purposes
Using osteologic specimens from a collection that included individuals who died in one city in the USA between 1893 and 1938, we asked: (1) do specimens with bilateral spondylolysis (bilateral pars defects) have increased levels of disc degeneration, at their respective motion segments, when compared with matched controls without spondylolysis, and (2) is the finding of a bilateral pars defect associated with more severe arthritis at L4-L5 than at L5-S1?
Methods
An observational study was performed on 665 skeletal lumbar spines from the Hamann-Todd Osteologic Collection at the Cleveland Museum of Natural History (Cleveland, OH, USA). The specimens included 534 males and 131 females ranging from 17 to 87 years old, with a nearly bell-shaped distribution of ages for males and a larger proportion of younger ages in the female specimens. Of those with spondylolysis, 81 had a defect at L5 and 14 had a defect at L4. The gross specimens were examined subjectively for evidence of arthrosis. At the time of examination, specific attention was not paid to the coexisting presence or absence of spondylolysis nor was the examiner blinded to the age of the specimens. Disc degeneration was measured by the classification of Eubanks et al., a modified version of the Kettler and Wilke classification. Linear regression was performed to derive a formula that would predict the amount of disc degeneration at L4-L5 and L5-S1 for the normal control population given a specimen’s age, sex, and race. We then used this formula to evaluate the difference in disc degeneration at the corresponding level of the pars defect that is greater than the predicted amount for a control without spondylolysis. This allowed us to conclude that any significant differences found between the L4-L5 and L5-S1 cohorts were attributable to factors not simply inherent to their functional position in the spine of an individual without a bilateral pars defect.
Results
L4 spondylolysis and L5 spondylolysis showed greater amounts of degeneration compared with that of matched controls (L4 controls: mean = 1.52, SD = 0.74; L4 spondylolysis: mean = 3.21, SD = 0.87; p < 0.001; L5 controls: mean = 0.97, SD = 0.48; L5 spondylolysis: mean = 2.06, SD = 0.98; p < 0.001). When we controlled for the expected amount of degenerative disc disease at each level in controls, the observed degeneration was more severe at L4-L5 than at L5-S1 (p = 0.008, R-squared = 18.6).
Conclusions
L4-L5 and L5-S1 bilateral spondylolysis groups had increased presence of degenerative disc disease compared with those without bilateral spondylolysis. For the same degree of spondylolysis, the observed amount of disc degeneration was greater at the L4-5 motion segment compared with L5-S1.
Clinical Relevance
Although not as common as the spondylolysis at L5-S1, we believe that our findings support that patients with L4-L5 spondylolysis can expect a greater degree of degenerative disc disease and increasing clinical symptoms. Multiple factors in the sacropelvic geometry of an individual, facet morphologic features at L4-L5, and the absence of the iliolumbar ligament at this level are possible contributing factors to the findings of this study.
Introduction
Spondylolysis is defined as a defect in the pars interarticularis of the vertebra [5]. While the incidence of a pars interarticularis defect of the lumbar vertebrae is effectively zero at birth [18, 44, 51, 52], prospective, long-term followup population studies found the incidence in adolescents to be 4%, with this number increasing to 6% in adulthood. Greater than 70% of the spondylolytic lesions were at the L5 vertebra, whereas approximately 15% were located at L4 [18]. Additional studies have verified these incidences in adults [25, 38]. Forty-five-year followup determined that the spondylolysis was most common bilaterally and was associated with instability at the corresponding level [6]. Patients with spondylolysis are more likely to have degenerative disc disease develop below the level of the associated pars defect [12]. The process of disc degeneration has been defined as an aberrant, cell-mediated response to progressive structural failure, for which there are many underlying factors at play [1]. However, we are not aware of any study that has examined the relationship between spondylolysis and disc degeneration at these lumbar levels. Owing to the majority of bilateral pars defects occurring at L5, it appears that the L4 involvement has been overlooked by previous researchers.
The defect in the pars at L5 can lead to an isthmic type spondylolisthesis at the L5-S1 level, which in turn, may contribute to disc degeneration (Fig. 1). Even so, patients with bilateral pars defects typically do not have instability develop at this level for many years. This may be the result of the added stability of the iliolumbar ligament at L5. The iliolumbar ligament connects the transverse process of the fifth lumbar vertebra to the ilium, stabilizing onto the pelvis [32]. Aihara et al. [3] showed that the integrity of the iliolumbar ligament may determine the amount of stability at the lumbosacral junction and the amount of forward slipping of the L5 vertebra. To further assess the amount of degenerative disc disease present in patients with bilateral pars defects at either L4 or L5, we made the assumption that changes in the skeletal anatomy of the adjacent vertebrae represent later stages of underlying soft tissue derangement, and therefore would give us an accurate estimation of the amount of degenerative disc disease in these respective motion segments for patients today with similar lesions.
Fig. 1.
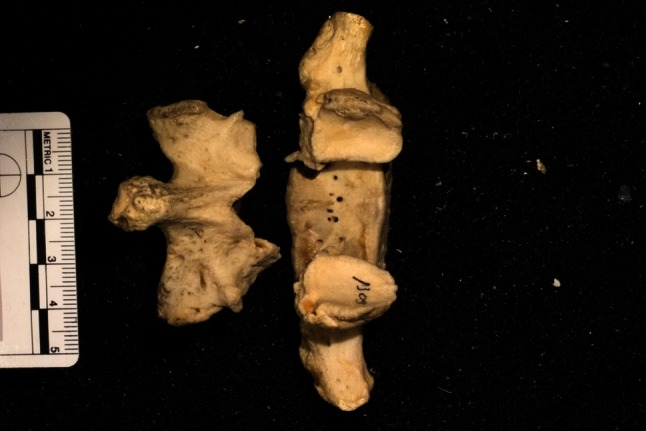
These specimens show bilateral spondylolysis at the L5 vertebra.
Using osteologic specimens from a collection that included individuals who died in one city in the USA between 1893 and 1938, we asked: (1) do specimens with bilateral spondylolysis (bilateral pars defects) have increased levels of disc degeneration, at their respective motion segments, compared with matched controls without spondylolysis, and (2) is the finding of a bilateral pars defect associated with more severe arthritis at L4-L5 than at L5-S1?
Methods
An observational study was performed on 665 cadaveric specimens from the Hamann-Todd Osteologic Collection at the Cleveland Museum of Natural History (Cleveland, OH, USA). The collection includes only individuals who died in Cleveland between 1893 and 1938.
Of 3100 cadaveric specimens from the Hamann-Todd Collection, we chose 665 skeletal lumbar spines for examination. These specimens were chosen from the entire collection depending on their collective proximity in two rows of the collection and their ease of access (no ladder necessary to take them off of the shelves). The selection was random insofar as the specimens in the collection are arranged only in the order in which they were added to the collection. At the time of examination, specific attention was not paid to the coexisting presence or absence of spondylolysis nor was the examiner blinded to the age of the specimens. We included 534 males and 131 females ranging from 17 to 87 years old, with a nearly bell-shaped distribution of ages for males and a larger proportion of younger ages in the female specimens. Linear regression was performed to derive a formula that would predict the amount of disc degeneration at L4-L5 and L5-S1 for the normal control population taking into account a specimen’s age, sex, and race. We then compared the degree of disc degeneration in the populations with L4 and L5 spondylolysis with the predicted level for a normal control (without bilateral spondylolysis), correcting for age, race, and sex.
Of these cadaveric specimens, we identified 120 (of 665, 18%) with spondylolysis. Four were excluded owing to incomplete or degraded skeletal elements, 18 were excluded for having unilateral defects only, and three were excluded because they had spondylolysis at a different lumbar level. Of the remaining specimens, 81 (of 95, 85%) had a bilateral defect at L5 and 14 (of 95, 15%) had a defect at L4. The gross specimens were examined subjectively by one examiner (JE) for evidence of arthrosis. At the time of examination, specific attention was not paid to the coexisting presence or absence of spondylolysis, nor was the examiner blinded to the age of the specimens (Table 1).
Table 1.
Specimen demographics
| Demographic | Years/number |
|---|---|
| Age | |
| Mean | 50 |
| SD | 16 |
| Sex | |
| Male | 534 |
| Female | 131 |
| Race | |
| White | 408 |
| Black | 257 |
Statistical Analysis
Disc degeneration was measured by the classification of Eubanks et al. [17], a modified version of the Kettler and Wilke [29] classification (Table 2). In this classification, degenerative disc disease was graded from no arthrosis to complete ankyloses, from Grade 0 to Grade IV (Fig. 2). Using linear regression analysis to correct for age, race, and sex, we compared disc degeneration at L4-L5 in L4 pars defects with the degree of degeneration seen at L5-S1 from a L5 pars defect (Fig. 3). Multiple regression analysis of the difference between the amount of degenerative disc disease seen in spondylolysis at each level, compared with the expected amount of disc degeneration in the specimens with no spondylolysis, also was performed. To determine that the increase in disc degeneration seen in specimens with L4 spondylolysis was not simply a representation of the overall increase in disc degeneration at L4-L5 in the control population, regression analysis was performed again with the control specimens (without spondylolysis) to create a formula to determine the expected amount of disc degeneration at L4-L5 and L5-S1, given the age, sex, and race of an individual specimen. We then compared the degree of disc degeneration in the populations with L4 and L5 spondylolysis with the predicted level for a normal control (without bilateral spondylolysis), correcting for age, race, and sex. The corresponding motion segment in question for specimens with bilateral pars defect at L4 is L4-L5 and L5-S1 motion segment for a L5 pars defect.
Table 2.
Classification of disc degeneration
| Grade | Classification | Description |
|---|---|---|
| 0 | No degeneration | Normal endplate |
| 1 | Mild degeneration | Osteophyte reaction involving < 50% of the endplate |
| 2 | Moderate degeneration | Osteophyte reaction involving between 50% and 100% of the endplate |
| 3 | Severe degeneration | Osteophyte reaction involving 100% of the endplate |
| 4 | Complete ankylosis | Fusion |
Fig. 2.

The anterior and posterior views of the specimens show bilateral spondylolysis at L5 with Grade 2 degeneration at the L5-S1 motion segment.
Fig. 3.

These specimens show the L4 vertebra with bilateral spondylolysis on the left and an intact L5 vertebra on the right. The joint surface of L4 and L5 are pointed at the reader, showing a Grade 3 L4-L5 motion segment.
Statgraphics® XVI software (Statpoint Technologies Inc, Warrenton, VA, USA) was used for statistical analysis.
Results
L4 spondylolysis and L5 spondylolysis showed greater amounts of degeneration compared with matched controls (L4 controls: mean = 1.52, SD = 0.74; L4 spondylolysis: mean = 3.21, SD = 0.87, p < 0.001; L5 controls: mean = 0.97, SD = 0.48. L5 spondylolysis: mean = 2.06, SD = 0.98, p < 0.001) (Table 3).
Table 3.
Degenerative disc disease at L4-L5 and L5-S1
| Group | Mean | 95% CI |
|---|---|---|
| L4 controls | 1.52 | 1.09–1.95 |
| L4 spondylolysis | 3.21 | 2.71–3.72 |
| L5 controls | 0.97 | 0.87–1.08 |
| L5 spondylolysis | 2.06 | 1.85–2.28 |
After controlling for degree of spondylolysis, age, race, and sex, we found that the L4 specimens had a larger than anticipated increase in degeneration compared with the L5 group (R-squared = 19.7; p < 0.001). To be sure that the increased amount of degenerative disc disease we found at L4-L5 compared with L5-S1 was associated with the bilateral spondylolysis and not attributable to the inherent properties of the motion segment at baseline, using our formula derived from the nonspondylolysis control group, we corrected for this and still found an increased amount of degenerative disc disease at L4-L5, greater than what could be predicted and accounted for in the normal control population (R-squared = 18.6; p = 0.008).
Discussion
Lumbar spondylosis or degenerative spine disease is the most common etiology of low-back pain and can have a substantial effect on an individual’s quality of life [40]. The medical costs associated with back pain in the US have doubled to more than $100 billion dollars during the past 7 years, with a 65% increase in national expenditure on back-related issues [33]. Owing to this increasingly common presentation, it would behoove clinicians to have a greater understanding about which characteristics, such as a bilateral pars defect in the lumbar spine, need to be more closely monitored for progression of lumbar degenerative disc disease. We are not aware of another study that has examined the relationship between bilateral spondylolysis and disc degeneration for the L4 and L5 vertebrae and their respective motion segments. Pars defects are most commonly found at L5 [4, 6, 7, 18, 20, 22, 25, 53], and because of this majority, most research has focused on the L5-S1 motion segment, while a bilateral pars defect at L4 and its effect on L4-L5 has been overlooked. Our findings show that bilateral pars defects of L4 and L5 cause increased amounts of degenerative disc disease at their respective motion segments. In addition, even after correcting for the predicted amount of degenerative disc disease and spondylolysis expected at each level based on a control cohort, we found an increase in the amount of degeneration at L4-L5 compared with L5-S1 owing to their respective bilateral pars defects. This supports the notion that patients with the less common L4 spondylolysis are at risk for more rapid progression of their disease along with increasing clinical symptoms attributable to the greater degree of degenerative disc disease.
This study should be interpreted in light of its limitations. First, skeletal specimens lack the soft tissue that would allow us to make inferences regarding what effects certain surrounding tissues such as the iliolumbar ligament may have had on the stability of the L4-L5 and L5-S1 motion segments. We also acknowledge that one of the current standards for assessing the degree of degenerative disc disease, MRI, could not be used in our cadaveric study. During the past 30 years, MRI has become the most accurate test for observing abnormal degenerative morphologic features of the spine [15, 19, 27, 36, 37]. Assuming the changes in the skeletal anatomy of adjacent vertebrae represent the later stages of underlying soft tissue derangement, our conclusions would seem to accurately reflect alterations in the soft tissues that make up the intervertebral spaces. We also could not evaluate sacropelvic parameters for each specimen, such as sacral inclination, pelvic lordosis, and pelvic incidence, because not all specimens had complete skeletal elements available. Toy et al. [49] found sacral inclination and degenerative disc disease, and pelvic incidence and degenerative disc disease, to be associated at the L5-S1 level. Pelvic incidence has been shown to correlate with spondyloarthrosis [14], and spondylolysis and spondylolisthesis [23, 30, 34, 42, 43]. However, none of these previous articles mentioned the effect that pelvic incidence had on the L4-L5 motion segment. Second, we had only one examiner (JE) grading the specimens and the examiner was not blinded to the age of the specimens, which could lead to bias as far as tending to associate a higher grade of degeneration to older patients. However, looking carefully at our data, many of the younger patients with bilateral spondylolysis were given higher grades and the opposite also was true for older control specimens in which little degeneration seen.
We also are not able to discuss the clinical picture and symptoms that these people may have presented with to a modern-day spine clinic owing to the nature of a cadaveric study. Studies on causality between degenerative disc disease and low back pain have been mixed. van Tulder et al. [50] performed a systematic review and reported that lumbar disc degeneration is a risk factor for low back pain in adults. Several studies have shown an association between degenerative disc disease and low back pain [16, 39, 48]. Some researchers have suggested a causal relationship between early-onset degenerative disc disease and recurrent low back pain throughout a patient’s lifetime [45, 46]. However, substantial research supports the fact that degenerative changes may be seen in asymptomatic patients [8, 28, 41], and these findings may not be predictive of development nor duration of low-back pain in long-term followup [9].
Another interesting question is how our specimens compare with that of the modern 21st century patient we see today. We would expect the BMI of a sample population from today to be approximately 25% greater than that of our specimens [24]. Previous research indicates that increasing BMI is not only a risk factor for degenerative disc disease and low back pain [13], but also spondylolysis [47]. Although we cannot make clinical correlations based on our cadaveric specimens, based on previous data we might reasonably infer that a sample population from today would have, if anything, increased prevalence of degenerative disc disease, spondylolysis, and low back pain based on the changes in body habitus that have developed during the last century or more.
Our data showed specimens with bilateral pars defects had increased amounts of disc degeneration compared with their matched controls without spondylolysis. We believe this is an important finding as most studies until now have focused on the L5-S1 motion segment and its degenerative changes [2, 3, 10, 11]. Researchers who have examined the L4-L5 motion segment have not done so in patients or specimens with bilateral pars defects, and therefore these effects have not been well studied [35].
Previous research has shown that bilateral spondylolytic defects in patients are much more likely to progress to spondylolisthesis and subsequent disc degeneration [11, 26, 49]. The general conclusions provided from such studies, along with the results we obtained, support the notion that the expected amount of degenerative disc disease in a patient with bilateral pars defects at a given level, should exceed that of someone without those lesions. This may be important to clinicians who want to look for these characteristics when deciding which patients with low-back pain need to be followed more closely for progression of disease. Even after correcting for confounding factors, we found that the L4-L5 motion segment showed increasing amounts of degenerative disc disease, greater than what was predicted, compared with the resultant amount of degenerative disc disease at L5-S1 from L5 lesions. The L4 spondylolysis predisposes individuals to a larger degree of disc degeneration at an early age compared with the matched controls (Fig. 4A). As age increases, the difference between the two groups stays rather constant. However, for the L5 group, we see an early increased amount of disc degeneration compared with the normal controls. As age increases, the disparity between the two closes (Fig. 4B).
Fig. 4A–B.
Disc degeneration at the (A) L4-L5 and (B) L5-SI motion segments is shown. The solid line represents the trend line for degree of degeneration and the dashed line represents the trend line.
One hypothesis for why we have come to these findings is the effect that the iliolumbar ligament has on the stability of the L5-S1 motion segment. Grobler et al. [21] performed biomechanical experiments on six specimens to examine why isthmic spondylolisthesis progressed more quickly at L4-L5 than it did at L5-S1. Their concluded that the L4-L5 spondylolytic lesion was more unstable, even when compared with L5 lesions with the iliolumbar ligament transected, possibly owing to the sagittal development of the L4-L5 facets. This was contrary to previous studies with similar design [10, 31, 54]. Aihara et al. [2] showed that a strong iliolumbar ligament in a patient with bilateral pars defects would stabilize the lumbosacral junction against spondylolisthesis and degeneration of the disc. Grobler et al. [21] hypothesized that other mechanical and anatomic factors such as musculature, ligaments, and the discs also must contribute, along with the iliolumbar ligament, to the stability of L5-S1 in the presence of bilateral spondylolysis. Another factor is the role an individual’s sacropelvic geometry may play in these outcomes. Pelvic incidence has been shown to be associated with spondylolysis, progression of spondylolisthesis, and degree of degenerative disc disease at L5-S1 [14, 23, 30, 34, 42, 43]. It could be possible that pelvic incidence or sacral inclination may have the same effect on spondylolysis at L4 and may account for some of the greater than predicted amount of degenerative disc disease we found in specimens with L4 lesions. Unfortunately, we did not have access to these sacropelvic measurements for all of our specimens, but this is an interesting question for a followup study.
Our study supports the idea that individuals with bilateral pars defects at L4 or L5 are likely to have increasing amounts of degenerative changes at their respective motion segments, compared with individuals who have intact pars. We also showed that after correcting for the spondylolysis, there still is a larger degree of degenerative disc disease in patients with bilateral L4 pars defects compared with those with lesions at L5, and this is greater than what is accounted for based on the normal population. Degenerative spine disease is the most common etiology of low-back pain [40], and as this becomes an increasing source of disability and national spending [33], clinicians will need to be able to triage patients appropriately based on what can be expected for their disease progression. We found that patients with bilateral spondylolysis may be at an increased risk to have degenerative disc disease develop and special attention must be paid to patients with L4 spondylolysis, as they may have a more rapidly progressive and severe disease course.
Acknowledgments
We thank Jason Eubanks MD, Lyman Jellema MA, and the staff at the Cleveland Museum of Natural History for assistance with using the Hamann-Todd Collection.
Footnotes
Each author certifies that he or she, or a member of his or her immediate family, has no funding or commercial associations (eg, consultancies, stock ownership, equity interest, patent/licensing arrangements, etc) that might pose a conflict of interest in connection with the submitted article.
All ICMJE Conflict of Interest Forms for authors and Clinical Orthopaedics and Related Research ® editors and board members are on file with the publication and can be viewed on request.
Each author certifies that his or her institution approved the human protocol for this investigation, that all investigations were conducted in conformity with ethical principles of research, and that informed consent for participation in the study was obtained.
This work was performed at the University Hospitals Case Medical Center and the Cleveland Museum of Natural History, Cleveland, OH, USA.
References
- 1.Adams MA, Roughley PJ. What is intervertebral disc degeneration, and what causes it? Spine (Phila Pa 1976). 2006;31:2151–2161. [DOI] [PubMed]
- 2.Aihara T, Takahashi K, Yamagata M, Moriya H, Shimada Y. Does the iliolumbar ligament prevent anterior displacement of the fifth lumbar vertebra with defects of the pars? J Bone Joint Surg Br. 2000;82:846–850. doi: 10.1302/0301-620X.82B6.10302. [DOI] [PubMed] [Google Scholar]
- 3.Aihara T, Takahashi K, Yamagata M, Moriya H, Tamaki T. Biomechanical functions of the iliolumbar ligament in L5 spondylolysis. J Orthop Sci. 2000;5:238–242. doi: 10.1007/s007760050158. [DOI] [PubMed] [Google Scholar]
- 4.Al-Khawashki H, Wasef Al-Sebai M. Combined dysplastic and isthmic spondylolisthesis: possible etiology. Spine (Phila Pa 1976). 2001;26:E542–546. [DOI] [PubMed]
- 5.American Academy of Orthopaedic Surgery. OrthoInfo: Glossary. Available at: http://orthoinfo.aaos.org/glossary.cfm#S. Accessed January 25, 2015.
- 6.Beutler WJ, Fredrickson BE, Murtland A, Sweeney CA, Grant WD, Baker D. The natural history of spondylolysis and spondylolisthesis: 45-year follow-up evaluation. Spine (Phila Pa 1976). 2003;28:1027–1035; discussion 1035. [DOI] [PubMed]
- 7.Blackburne JS, Velikas EP. Spondylolisthesis in children and adolescents. J Bone Joint Surg Br. 1977;59:490–494. doi: 10.1302/0301-620X.59B4.925059. [DOI] [PubMed] [Google Scholar]
- 8.Boden SD, Davis DO, Dina TS, Patronas NJ, Wiesel SW. Abnormal magnetic-resonance scans of the lumbar spine in asymptomatic subjects: a prospective investigation. J Bone Joint Surg Am. 1990;72:403–408. [PubMed] [Google Scholar]
- 9.Borenstein DG, O’Mara JW, Jr, Boden SD, Lauerman WC, Jacobson A, Platenberg C, Schellinger D, Wiesel SW. The value of magnetic resonance imaging of the lumbar spine to predict low-back pain in asymptomatic subjects: a seven-year follow-up study. J Bone Joint Surg Am. 2001;83:1306–1311. doi: 10.2106/00004623-200109000-00002. [DOI] [PubMed] [Google Scholar]
- 10.Chow DH, Luk KD, Leong JC, Woo CW. Torsional stability of the lumbosacral junction: significance of the iliolumbar ligament. Spine (Phila Pa 1976). 1989;14:611–615. [DOI] [PubMed]
- 11.Cyron BM, Hutton WC, Troup JD. Spondylolytic fractures. J Bone Joint Surg Br. 1976;58-B:462–466. [DOI] [PubMed]
- 12.Dai LY. Disc degeneration in patients with lumbar spondylolysis. J Spinal Disord. 2000;13:478–486. doi: 10.1097/00002517-200012000-00003. [DOI] [PubMed] [Google Scholar]
- 13.Dario AB, Ferreira ML, Refshauge KM, Lima TS, Ordoñana JR, Ferreira PH. The relationship between obesity, low back pain, and lumbar disc degeneration when genetics and the environment are considered: a systematic review of twin studies. Spine J. 2015;15:1106–1117. doi: 10.1016/j.spinee.2015.02.001. [DOI] [PubMed] [Google Scholar]
- 14.Duval-Beaupère G, Schmidt C, Cosson P. A barycentremetric study of the sagittal shape of spine and pelvis: the conditions required for an economic standing position. Ann Biomed Eng. 1992;20:451–462. doi: 10.1007/BF02368136. [DOI] [PubMed] [Google Scholar]
- 15.Edelman RR, Shoukimas GM, Stark DD, Davis KR, New PF, Saini S, Rosenthal DI, Wismer GL, Brady TJ. High-resolution surface-coil imaging of lumbar disk disease. AJR Am J Roentgenol. 1985;144:1123–1129. doi: 10.2214/ajr.144.6.1123. [DOI] [PubMed] [Google Scholar]
- 16.Erkintalo MO, Salminen JJ, Alanen AM, Paajanen HE, Kormano MJ. Development of degenerative changes in the lumbar intervertebral disk: results of a prospective MR imaging study in adolescents with and without low-back pain. Radiology. 1995;196:529–533. doi: 10.1148/radiology.196.2.7617872. [DOI] [PubMed] [Google Scholar]
- 17.Eubanks JD, Lee MJ, Cassinelli E, Ahn NU. Does lumbar facet arthrosis precede disc degeneration? A postmortem study. Clin Orthop Relat Res. 2007;464:184–189. doi: 10.1097/BLO.0b013e3181583d4e. [DOI] [PubMed] [Google Scholar]
- 18.Fredrickson BE, Baker D, McHolick WJ, Yuan HA, Lubicky JP. The natural history of spondylolysis and spondylolisthesis. J Bone Joint Surg Am. 1984;66:699–707. [PubMed] [Google Scholar]
- 19.Gibson MJ, Buckley J, Mawhinney R, Mulholland RC, Worthington BS. Magnetic resonance imaging and discography in the diagnosis of disc degeneration: a comparative study of 50 discs. J Bone Joint Surg Br. 1986;68:369–373. doi: 10.1302/0301-620X.68B3.3733797. [DOI] [PubMed] [Google Scholar]
- 20.Green TP, Allvey JC, Adams MA. Spondylolysis: bending of the inferior articular processes of lumbar vertebrae during simulated spinal movements. Spine (Phila Pa 1976). 1994;19:2683–2691. [PubMed]
- 21.Grobler LJ, Novotny JE, Wilder DG, Frymoyer JW, Pope MH. L4-5 isthmic spondylolisthesis: a biomechanical analysis comparing stability in L4-5 and L5-S1 isthmic spondylolisthesis. Spine (Phila Pa 1976). 1994;19:222–227. [PubMed]
- 22.Grobler LJ, Robertson PA, Novotny JE, Pope MH. Etiology of spondylolisthesis: assessment of the role played by lumbar facet joint morphology. Spine (Phila Pa 1976). 1993;18:80–91. [PubMed]
- 23.Hanson DS, Bridwell KH, Rhee JM, Lenke LG. Correlation of pelvic incidence with low- and high-grade isthmic spondylolisthesis. Spine (Phila Pa 1976). 2002;27:2026–2029. [DOI] [PubMed]
- 24.Helmchen LA, Henderson RM. Changes in the distribution of body mass index of white US men, 1890–2000. Ann Hum Biol. 2004;31:174–181. doi: 10.1080/03014460410001663434. [DOI] [PubMed] [Google Scholar]
- 25.Hensinger RN. Spondylolysis and spondylolisthesis in children and adolescents. J Bone Joint Surg Am. 1989;71:1098–1107. [PubMed] [Google Scholar]
- 26.Hutton WC, Stott JR, Cyron BM. Is spondylolysis a fatigue fracture? Spine (Phila Pa 1976). 1977;2:202–209.
- 27.Jarvik JG, Maravilla KR, Haynor DR, Levitz M, Deyo RA. Rapid MR imaging versus plain radiography in patients with low back pain: initial results of a randomized study. Radiology. 1997;204:447–454. doi: 10.1148/radiology.204.2.9240534. [DOI] [PubMed] [Google Scholar]
- 28.Jensen MC, Brant-Zawadzki MN, Obuchowski N, Modic MT, Malkasian D, Ross JS. Magnetic resonance imaging of the lumbar spine in people without back pain. N Engl J Med. 1994;331:69–73. doi: 10.1056/NEJM199407143310201. [DOI] [PubMed] [Google Scholar]
- 29.Kettler A, Wilke HJ. Review of existing grading systems for cervical or lumbar disc and facet joint degeneration. Eur Spine J. 2006;15:705–718. doi: 10.1007/s00586-005-0954-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Labelle H, Roussouly P, Berthonnaud E, Transfeldt E, O’Brien M, Chopin D, Hresko T, Dimnet J. Spondylolisthesis, pelvic incidence, and spinopelvic balance: a correlation study. Spine (Phila Pa 1976). 2004;29:2049–2054. [DOI] [PubMed]
- 31.Leong JC, Luk KD, Chow DH, Woo CW. The biomechanical functions of the iliolumbar ligament in maintaining stability of the lumbosacral junction. Spine (Phila Pa 1976). 1987;12:669–674. [DOI] [PubMed]
- 32.Luk KD, Ho HC, Leong JC. The iliolumbar ligament: a study of its anatomy, development and clinical significance. J Bone Joint Surg Br. 1986;68:197–200. doi: 10.1302/0301-620X.68B2.3958002. [DOI] [PubMed] [Google Scholar]
- 33.Martin BI, Deyo RA, Mirza SK, Turner JA, Comstock BA, Hollingworth W, Sullivan SD. Expenditures and health status among adults with back and neck problems. JAMA. 2008;299:656–664. doi: 10.1001/jama.299.6.656. [DOI] [PubMed] [Google Scholar]
- 34.Marty C, Boisaubert B, Descamps H, Montigny JP, Hecquet J, Legaye J, Duval-Beaupère G. The sagittal anatomy of the sacrum among young adults, infants, and spondylolisthesis patients. Eur Spine J. 2002;11:119–125. doi: 10.1007/s00586-001-0349-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Miller JA, Schmatz C, Schultz AB. Lumbar disc degeneration: correlation with age, sex, and spine level in 600 autopsy specimens. Spine (Phila Pa 1976). 1988;13:173–178. [PubMed]
- 36.Modic MT. Degenerative disc disease and back pain. Magn Reson Imaging Clin N Am. 1999;7:481–491, viii. [PubMed]
- 37.Modic MT, Steinberg PM, Ross JS, Masaryk TJ, Carter JR. Degenerative disk disease: assessment of changes in vertebral body marrow with MR imaging. Radiology. 1988;166:193–199. doi: 10.1148/radiology.166.1.3336678. [DOI] [PubMed] [Google Scholar]
- 38.Muschik M, Hähnel H, Robinson PN, Perka C, Muschik C. Competitive sports and the progression of spondylolisthesis. J Pediatr Orthop. 1996;16:364–369. doi: 10.1097/01241398-199605000-00014. [DOI] [PubMed] [Google Scholar]
- 39.Paajanen H, Erkintalo M, Kuusela T, Dahlstrom S, Kormano M. Magnetic resonance study of disc degeneration in young low-back pain patients. Spine (Phila Pa 1976). 1989;14:982–985. [DOI] [PubMed]
- 40.Parker SL, Godil SS, Mendenhall SK, Zuckerman SL, Shau DN, McGirt MJ. Two-year comprehensive medical management of degenerative lumbar spine disease (lumbar spondylolisthesis, stenosis, or disc herniation): a value analysis of cost, pain, disability, and quality of life: clinical article. J Neurosurg Spine. 2014;21:143–149. doi: 10.3171/2014.3.SPINE1320. [DOI] [PubMed] [Google Scholar]
- 41.Powell MC, Wilson M, Szypryt P, Symonds EM, Worthington BS. Prevalence of lumbar disc degeneration observed by magnetic resonance in symptomless women. Lancet. 1986;2:1366–1367. doi: 10.1016/S0140-6736(86)92008-8. [DOI] [PubMed] [Google Scholar]
- 42.Rajnics P, Templier A, Skalli W, Lavaste F, Illés T. The association of sagittal spinal and pelvic parameters in asymptomatic persons and patients with isthmic spondylolisthesis. J Spinal Disord Tech. 2002;15:24–30. doi: 10.1097/00024720-200202000-00004. [DOI] [PubMed] [Google Scholar]
- 43.Roussouly P, Gollogly S, Berthonnaud E, Labelle H, Weidenbaum M. Sagittal alignment of the spine and pelvis in the presence of L5-s1 isthmic lysis and low-grade spondylolisthesis. Spine (Phila Pa 1976). 2006;31:2484–2490. [DOI] [PubMed]
- 44.Rowe GG, Roche MB. The etiology of separate neural arch. J Bone Joint Surg Am. 1953;35:102–110. [PubMed] [Google Scholar]
- 45.Salminen JJ, Erkintalo M, Laine M, Pentti J. Low back pain in the young: a prospective three-year follow-up study of subjects with and without low back pain. Spine (Phila Pa 1976). 1995;20:2101–2107; discussion 2108. [DOI] [PubMed]
- 46.Salminen JJ, Erkintalo MO, Pentti J, Oksanen A, Kormano MJ. Recurrent low back pain and early disc degeneration in the young. Spine (Phila Pa 1976). 1999;24:1316–1321. [DOI] [PubMed]
- 47.Sonne-Holm S, Jacobsen S, Rovsing HC, Monrad H, Gebuhr P. Lumbar spondylolysis: a life long dynamic condition? A cross sectional survey of 4.151 adults. Eur Spine J. 2007;16:821–828. doi: 10.1007/s00586-006-0250-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Tertti MO, Salminen JJ, Paajanen HE, Terho PH, Kormano MJ. Low-back pain and disk degeneration in children: a case-control MR imaging study. Radiology. 1991;180:503–507. doi: 10.1148/radiology.180.2.1829844. [DOI] [PubMed] [Google Scholar]
- 49.Toy JO, Tinley JC, Eubanks JD, Qureshi SA, Ahn NU. Correlation of sacropelvic geometry with disc degeneration in spondylolytic cadaver specimens. Spine (Phila Pa 1976). 2012;37:E10–15. [DOI] [PubMed]
- 50.van Tulder MW, Assendelft WJ, Koes BW, Bouter LM. Spinal radiographic findings and nonspecific low back pain: a systematic review of observational studies. Spine (Phila Pa 1976). 1997;22:427–434. [DOI] [PubMed]
- 51.Wiltse LL. Spondylolisthesis in children. Clin Orthop Relat Res. 1961;21:156–163. [PubMed] [Google Scholar]
- 52.Wiltse LL. The etiology of spondylolisthesis. J Bone Joint Surg Am. 1962;44:539–560. [PubMed] [Google Scholar]
- 53.Wiltse LL, Winter RB. Terminology and measurement of spondylolisthesis. J Bone Joint Surg Am. 1983;65:768–772. [PubMed] [Google Scholar]
- 54.Yamamoto I, Panjabi MM, Oxland TR, Crisco JJ. The role of the iliolumbar ligament in the lumbosacral junction. Spine (Phila Pa 1976). 1990;15:1138–1141. [DOI] [PubMed]



