Abstract
Rationale
Whilst the FDA-approved anorectic, lorcaserin and various 5-hydroxytryptamine (5-HT)6 receptor antagonists reduce feeding, a direct assessment of their impact upon feeding behaviour is less clear. We therefore examined the action of lorcaserin and the clinical-stage developmental candidate 5-HT6 receptor antagonist, SB-742457, upon microstructural analysis of licking behaviour. Such analysis provides a rich source of information about the mechanisms controlling food intake.
Objectives
The objective of the present study was to gain insight into the influence upon feeding behaviour of the 5-HT2C receptor agonist, lorcaserin and the developmental 5-HT6 receptor antagonist, SB-742457.
Methods
The impact of lorcaserin and SB-742457 upon licking behaviour of non-deprived rats for a glucose solution was assessed using microstructural analysis.
Results
Lorcaserin (0.1–3.0 mg/kg) displayed a dose-dependent ability to reduce glucose consumption via reduction in the number of bouts of licking. A similar action was evident with SB-742457, but only at the lowest dose tested (3.0 mg/kg).
Conclusions
The behavioural actions of both lorcaserin and SB-742457 demonstrate they directly promote satiety.
Keywords: Lorcaserin, 5-HT2C receptor, 5-HT6 receptor, Obesity, Feeding behaviour, Microstructural analysis
Introduction
Obesity is a major public health problem in many countries and gives rise to various co-morbidities including type 2 diabetes, hypertension and dyslipidemia. These sequelae create a considerable burden to the healthcare system (e.g. Ogden et al. 2014) and at least in the USA are second only to tobacco use as a cause of death in under 70 year olds (Danaei et al. 2009). Against this background, the requirement for safe and efficacious anti-obesity drugs is clear.
There is a considerable history of drugs targeting the 5-hydroxytryptamine (5-HT) serotonin system influencing appetite and feeding in pre-clinical models and patients (e.g. Blundell 1977; Vickers and Dourish 2004). For instance, drugs that increase 5-HT neurotransmission such as fenfluramine (and its d-enantiomer, dexfenfluramine) alone or in combination with phentermine (Fen-Phen; e.g. Weintraub et al. 1984), and subsequently, sibutramine (which also increases synaptic levels of noradrenaline and dopamine; McNeely and Goa 1998; Balcioglu and Wurtman 2000) possess clear therapeutic anorectic efficacy in patients (for review, see Halford et al. 2011); however, their adverse effects on the cardiovascular system (e.g. valvulopathy, pulmonary hypertension; Fitzgerald et al. 2000; Rothman et al. 2000; Launay et al. 2002) eventually led to their removal from the market. Investigation concerning which 5-HT receptors mediate the therapeutic actions of drugs like fenfluramine and sibutramine suggested the 5-HT2C receptor as having at least a major role (e.g. Jackson et al. 1997; Vickers et al. 1999; Dutton and Barnes 2006; Higgs et al. 2011; Berglund et al. 2013). Fortunately, the activation of the 5-HT2C receptor does not appear to be associated with the adverse cardiovascular effects allowing direct 5-HT2C receptor agonists to be evaluated for the anorectic efficacy. However, there is a need to avoid concomitant 5-HT2A or 5-HT2B receptor activation as the former results in neuropsychiatric events (e.g. hallucinations; Nichols 2004) and the latter receptor may be responsible for some of the cardiovascular complications arising from drugs like fenfluramine (Fitzgerald et al. 2000; Rothman et al. 2000; Launay et al. 2002). The pharmacophores of the 5-HT2A, 5-HT2B and 5-HT2C receptors are structurally similar (for reviews, see Meltzer and Roth 2013; Michino et al. 2015) and hence it has proved challenging to develop 5-HT2C receptor agonists with presumed sufficient selectivity. One such proposed agent, however, is lorcaserin, which displays around 100- and 19-fold selectivity as an agonist for the human 5-HT2C receptor compared to the human 5-HT2B receptor and human 5-HT2A receptor, respectively (functional selectivity assessed by inositol phosphate accumulation via the appropriate receptor; Smith et al. 2008; Thomsen et al. 2008), although a lower selectivity is apparent (12- and 7-fold, respectively) when the relative affinities are compared (Thomsen et al. 2008; it should be noted that in these studies, lorcaserin was a partial agonist at the human recombinant 5-HT2A receptor with an intrinsic activity of 0.75). This discrepancy between the selectivity for the three 5-HT2 receptors when comparing agonist potency or receptor affinity may indicate lorcaserin displays biased agonism and/or there were differential levels of receptor reserve apparent when using recombinant expression of the different 5-HT2 receptors. Nevertheless, lorcaserin is now approved by the FDA as a prescription medication to assist weight loss regimens (trade name, Belviq®; http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm309993.htm), although a caveat to the approval is the need to perform post-marketing studies to, for example, better examine the risk of major adverse cardiac events. However, despite this recent approval, there are a relatively small number of papers in the literature reporting the actions of lorcaserin in pre-clinical models of obesity and feeding (Smith et al. 2008; Thomsen et al. 2008; Fletcher et al. 2009; Higgins et al. 2014; Burke et al. 2014) and these have only reported lorcaserin’s effect on the quantity of food intake (acute lorcaserin administration) or on body weight (chronic lorcaserin administration). Given the complexity of 5-HT’s action upon feeding, in an attempt to gain further insight into the action of lorcaserin upon feeding behaviour, in the present study, we report the microstructural analysis of lorcaserin’s action upon licking behaviour of rats to ingest a liquid diet. Analysis of licking responses provides a rich source of information concerning the principal factors (including satiety), which determine food intake (e.g. Davis and Smith 1992; Davis et al. 2001). To establish the behavioural mechanisms that underlie drug-induced changes in food intake, the effects of pharmacological manipulations can be compared with available reference standards that display distinctive effects on licking patterns associated with satiety or orosensory variables (e.g. Davis and Smith 1992; Davis et al. 2001). We have previously used this approach successfully to reveal the effects of diverse pharmacological agents on eating with high resolution. For example, we have found that whilst both the cannabinoid CB1 receptor antagonist, rimonabant and the 5-HT mimetic, sibutramine, both decreased total intake of glucose, this was achieved by different mechanisms; rimonabant primarily decreased licking bout duration (Higgs et al. 2003) whereas sibutramine decreased bout number (Higgs et al. 2011). Such data indicates that cannabinoid neurotransmission is important for food palatability whereas 5-HT neurotransmission influences satiety. Identification of such mechanistic differentiation highlights the usefulness of microstructural analysis of feeding behaviour to better understand the mechanisms of drug-induced changes in food intake.
This present report is extended further to also assess in the behavioural action of a selective 5-HT6 receptor antagonist, SB-742457, using the microstructural analysis of licking behaviour. The 5-HT6 receptor has also been proposed as a suitable therapeutic target to reduce obesity although the peer-reviewed reports simply assess the ability of 5-HT6 receptor ligands to modify the quantity of food intake or body weight (Woolley et al. 2001; Fisas et al. 2006; Garfield et al. 2014).
Materials and methods
Animals
For each compound tested, 12 drug naïve adult male hooded Lister rats (Charles River, UK), weighing approximately 200 g at the beginning of training, were housed in groups of four. Rats were maintained on a 12:12 h light/dark cycle (lights on at 0700 hours), at a constant temperature of 21 ± 1 °C (humidity 50 %). Food pellets (rat and mouse expanded ground maintenance diet; Special Diet services, Cambridge, UK) and water were available ad libitum, except when the animals were in the test chambers. All testing was conducted in the light phase between 0900 and 1700 hours. The experiments were carried out in accordance with the terms of the UK Animals (Experimental Procedures) Act, 1986.
Drugs
The selective 5-HT2C receptor agonist, lorcaserin (custom synthesis performed by Axon Medchem) was prepared for injection by dissolving in 0.9 % saline and was administered via the i.p. route. The selective 5-HT6 receptor antagonist, SB-742457 (Axon Medchem), was homogenised in 1 % methylcellulose and administered orally. Both drugs were administered in a volume of 1.0 ml/kg.
Apparatus
Testing was carried out using an MS80 multistation lick analysis system (Dilog Instruments, Tallahassee, FL, USA), which has been described in detail previously (Higgs and Cooper 1997). Briefly, rats were placed in a Perspex chamber that had an opening in the centre of the front wall allowing access to a drinking spout that was connected to a plastic 50-ml measuring tube containing the glucose solution (200 mM glucose dissolved in distilled water 200 mM), which is palatable to rats. The lickometer was connected to an amplifier that passed less than 60 nA through the rat every time tongue contact was made with the spout. The current was fed to a standard personal computer (Opus Technology, Surrey, UK), which stored the time of each tongue contact to the nearest millisecond.
Training
A non-deprivation, free-feeding paradigm was used to avoid the possibility that deprivation might modify licking responses that would complicate identification of independent drug effects. During the training phase, each rat was placed in the test chamber daily with access to the drinking spout to obtain the glucose solution. The rats were left in the test apparatus for 20 min from the time of the first lick at the drinking spout for the glucose solution. This training was repeated daily until each rat achieved a consistent number of total licks (typically achieved by day 5). Although 12 rats in each study (lorcaserin or SB-742457) began training, 1 and 2 rats failed to achieve baseline levels of licking in the lorcaserin and SB-742457 studies, respectively; these rats were not studied further.
To familiarise the rats with the drug administration procedure, on the 2 days before drug testing began, each rat received saline administration via the appropriate route of administration.
Testing
Administration of vehicle or drug was made using a repeated-measures design, with the order of treatment randomised. After injection, each rat was returned to the home cage before being placed individually in a test chamber, where it had access to the glucose solution for 20 mins. The vehicle/drug administration test interval for the lorcaserin study was 30 and 60 min for the SB-742457 study. ‘Wash-out’ between successive administration of drug/vehicle was at least 48 h.
Analysis
The accumulated lick data were grouped into bouts defined as the licking occurring with an upper inter-lick interval of less than 400 ms, which corresponds to an interval that is just above twice the average inter-lick interval and is consistent with our previous studies (Higgs and Cooper 1996, 1997, 1998, 2000). Drug impact was examined on four microstructural variables: the number of bouts, the duration of a bout, the intra-bout lick rate (defined as the average number of licks per second within a bout) and the latency to begin licking (timed from the shutter opening allowing access to the drinking spout to the first lick). Analysis of data was by one-way analysis of variance (ANOVA) for repeated measures. The effects of drug relative to vehicle were assessed using a post hoc t test corrected for multiple comparisons. For the number of bouts of licking, values were obtained for each rat, and then mean values and standard error of the mean (SEM) were calculated for each treatment. For bout duration and intra-bout lick rate, values across the test period for each rat were averaged, and the mean and SEM were subsequently calculated for treatment groups. Three of the 11 rats in the lorcaserin study failed to register a lick at the highest dose of lorcaserin tested (3.0 mg/kg) and so could not contribute to the microstructural analysis. The data were analysed both without the data from these rats and using multiple imputation to substitute for the missing values. The pattern of results was similar in both cases. For brevity, we just report the results with the affected data points excluded. The latency data were not normally distributed and so the data were square root transformed before analysis. Statistical tests were performed using Sigma Stat (Jandel Corporation, San Rafael, CA, USA, 1992–1994). A result was considered statistically significant if p < 0.05.
Results
Dose-dependent effect of lorcaserin on licking for glucose solution
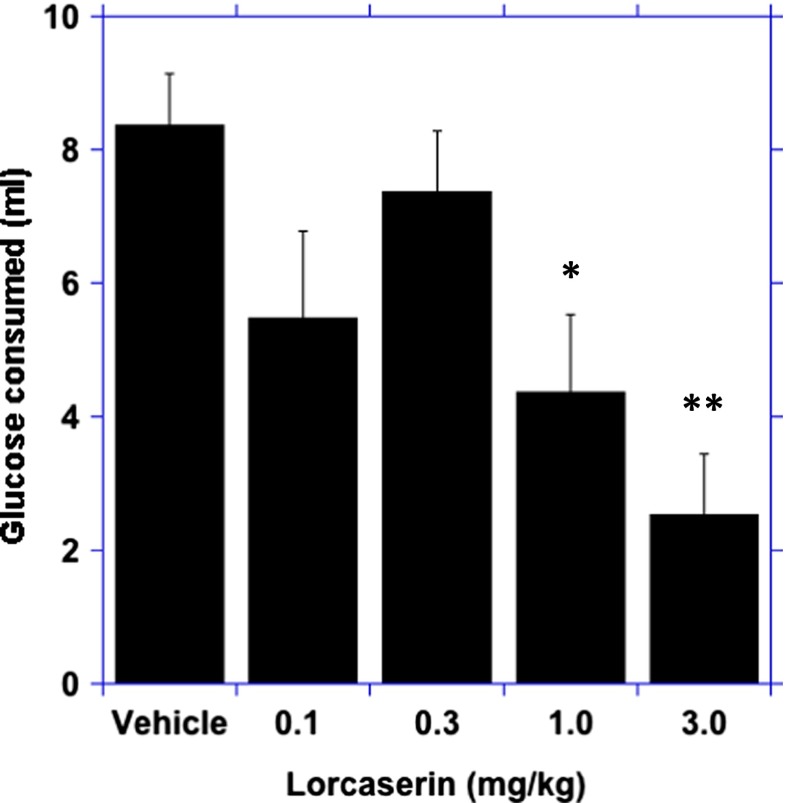
There was a main effect of lorcaserin on the amount of glucose solution consumed ((4 ,28) = 6, p < 0.001). In comparison to the vehicle response, there was a significant reduction in glucose solution consumed after the 1.0 mg/kg (p < 0.05) and 3.0 mg/kg doses of lorcaserin (p < 0.01; Fig. 1).
Fig. 1.
Dose-dependent ability of lorcaserin to reduce the consumption of glucose. Data represent the mean ± SEM, n = 12, *p < 0.05, **p < 0.01
Microstructural analysis
There was a main effect of lorcaserin on total licks for the glucose solution (F (4, 28) = 6; p < 0.01; Table 1). In comparison to the vehicle response, there was a significant reduction in the total licks for the glucose solution after the 1.0 and 3.0 mg/kg doses of lorcaserin (p < 0.05; Table 1); the lorcaserin-induced reduction in total licks was due to a reduction in the number of lick bouts (F(4, 28) = 5.7; p < 0.001; Table 1) rather than an effect on lick bout duration (F(4, 28) = 1.9; p = 0.1; Table 1). There was a main effect of lorcaserin on intra-bout lick rate (F(4, 28) = 6; p < 0.01) with a significant reduction in intra-bout lick rate but only at the highest dose tested of lorcaserin (3.0 mg/kg; p < 0.01; Table 1). There was also a significant effect of lorcaserin on the latency to lick (F(4,28) = 4.6, p < 0.01); post hoc analysis indicated that latency was significantly increased at the highest does tested (3.0 mg/kg; Table 1).
Table 1.
Total number of licks and microstructural data after lorcaserin. The microstructural data are shown as means. Standard error difference between means is shown in brackets
| Microstructure analysis | Vehicle | Lorcaserin | |||
|---|---|---|---|---|---|
| 0.1 mg/kg | 0.3 mg/kg | 1.0 mg/kg | 3.0 mg/kg | ||
| Number of licks | 2091.4 (192.4) | 1366.3 (325.7) | 1837.3 (231.0) | 1087.4* (292.7) | 635.3**(224.5) |
| Bout duration (s) | 3.4 (0.4) | 3.5 (0.5) | 3.3 (0.4) | 2.4 (0.4) | 2.4 (0.4) |
| Bout number | 98.0 (10.1) | 60.5* (13.8) | 89.5 (12.1) | 59.5 (15.5)* | 36.0**(11.4) |
| Intra-bout lick rate (licks/s) | 6.3 (0.2) | 6.2 (0. 2) | 6.3 (0. 2) | 6.6 (0.2) | 5.8* (0.2) |
| Latency (s) | 31.5 (17.9) | 26.8 (11.2) | 25.0 (8.6) | 39.7 (18.2) | 262.5** (130.1) |
Asterisks indicate significantly different from vehicle control condition (*p < 0.05; **p < 0.01)
Effect of SB-742457 on licking for glucose solution
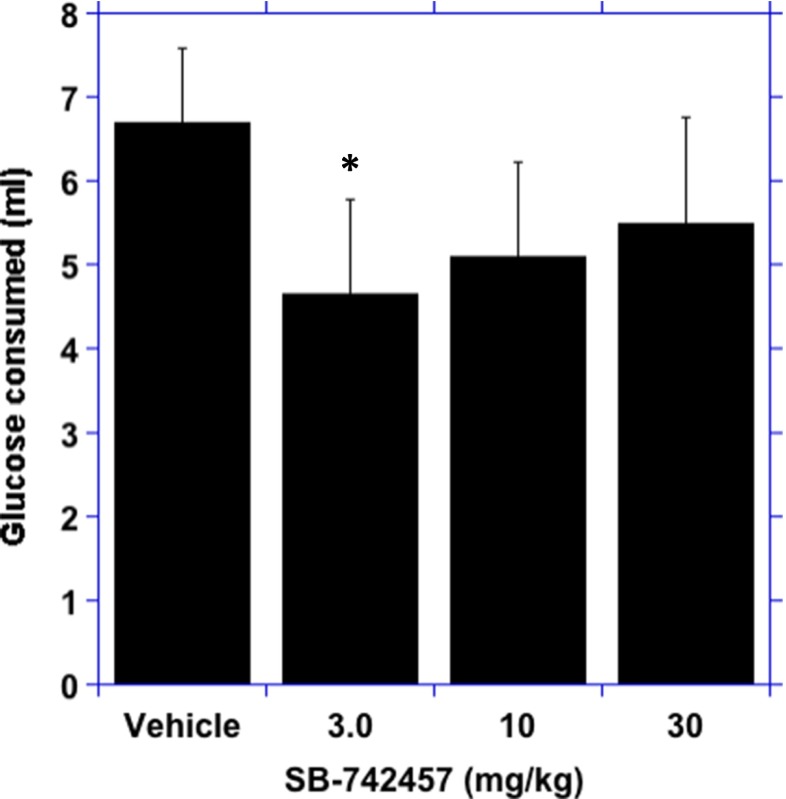
There was a main effect of SB-742457 on the amount of glucose solution consumed (F(3, 27) = 3.4; p < 0.05). In comparison to the vehicle response, there was a significant reduction in glucose solution consumed after the 3.0 mg/kg dose of SB-742457 (p < 0.05; Fig. 2).
Fig. 2.
Ability of SB-742457 to reduce consumption of glucose. Data represent the mean ± SEM, n = 12, *p < 0.05
Microstructural analysis
There was a significant reduction in total licks for glucose solution after SB-742457 (F(3, 27) = 3.3; p < 0.05) but this was only significant at the lowest dose tested (3.0 mg/kg; p < 0.01; Table 2). The reduction in total licks was due to a decrease in the number of lick bouts (F(3, 27) = 2.8; p < 0.05; Table 2) rather than bout duration (F(3, 27) = 1.8; p = 0.2; Table 2). There was a significant main effect of SB-742457 on intra-bout lick rate (F(3, 27) = 4.8; p < 0.01) but the only significant reduction was at the highest dose tested (30 mg/kg; p < 0.01; Table 2). There was no significant effect of SB-742457 on latency to start licking (F(3, 27) = 0.14, p = 0.9; Table 2).
Table 2.
Total number of licks and microstructural data after SB-742457. The microstructural data are shown as means. Standard error difference between means is shown in brackets
| Microstructure analysis | Vehicle | SB-742457 | ||
|---|---|---|---|---|
| 3.0 mg/kg | 10 mg/kg | 30 mg/kg | ||
| Number of licks | 1456.7 (199.9) | 992.8* (255.2) | 1130.7 (249.9) | 1238.9 (278.4) |
| Bout duration (s) | 2.7 (0.5) | 1.9 (0.1) | 2.1 (0.3) | 2.0 (0.3) |
| Bout number | 97.2 (12.8) | 73.5* (17.2) | 80.1 (13.9) | 114.5 (27.3) |
| Intra-bout lick rate (licks/s) |
6.4 (0.1) | 6.4 (0.2) | 6.3 (0.2) | 5.9* (0.1) |
| Latency (s) | 84.7 (46.9) | 66.1 (28.0) | 76.1 (20.7) | 62.7 (23.9) |
Asterisks indicate significantly different from vehicle control condition (*p < 0.05)
Discussion
Previous studies exploring the anti-obesity actions of the 5-HT2C receptor agonist, lorcaserin (Smith et al. 2008; Thomsen et al. 2008; Higgins et al. 2014; Burke et al. 2014) or 5-HT6 receptor antagonists (Woolley et al. 2001; Garfield et al. 2014) in animal models have utilised food intake or body weight to measure their impact. The present study utilised microstructural analysis of licking behaviour in rats for a liquid diet to offer direct insight into the behavioural mechanisms influenced by lorcaserin and the selective 5-HT6 receptor antagonist, SB-742457. By examining the pattern of licking behaviour predictions can be made concerning the influence of drug upon satiety and the palatability of food (Davis and Smith 1992; Breslin et al. 1996; Higgs and Cooper 1996, 1997, 1998; Davis et al. 2001; Cooper and Higgs 2005; Higgs et al. 2011).
In the present study, lorcaserin treatment resulted in a selective reduction in the number of bouts of licking behaviour that was responsible for the reduced food intake. Importantly, the effect on bout number was also observed at doses that had no non-specific effects on licking latency or lick rate. Rats lick at a constant rate of about 6–7 licks/s (Stellar and Hill 1952) and drug-induced disruption of this rate would suggest that a pharmacological treatment might be affecting sensorimotor coordination, as distinct from a specific effect on the controls of ingestion. For example, Davis and Smith (1992) showed that intra-bout lick rate is affected by moving the drinking spout progressively further away from an animal, but not by altering the solution concentration. Hence, the current pattern of results suggests that since lorcaserin was able to decrease bout number in the absence of a reduced intra-lick rate, a modulation of motoric activity did not complicate interpretation. However, it should be noted that the effect was not linear in that the 0.3 mg/kg dose has less of an effect on bout number than the 0.1 mg/kg dose. Similar effects on licking microstructure are observed when delaying negative feedback post-ingestive signals, by adding the non-digestible sugar mannitol to glucose solutions (Davis and Levine 1977). Conversely, the addition of the non-nutritive sweetener saccharin to glucose, which selectively increases palatability, increases bout duration whilst having minimal effects on bout number (Breslin et al. 1996). Hence, selective action of lorcaserin on bout number supports promotion of satiety by the drug rather than an effect on taste palatability, which is consistent with the mechanism of action of some less-selective 5-HT2C receptor agonists in rodents (e.g. Ro 60–0175; Clifton et al. 2000) and humans (Thomas et al. 2014) as well as the 5-HT releasing agents, fenfluramine (d-fenfluramine) and sibutramine (Tallett et al. 2009; Higgs et al. 2011; Burke et al. 2014).
One study that has examined the behavioural effects of lorcaserin assessed its effect on the behavioural satiety sequence (Higgins et al. 2014). It was reported that lorcaserin (0.3–1.0 mg/kg) advanced the behavioural satiety sequence in non-deprived rats consuming a sweetened mash diet over 1 h, consistent with the effect on satiety reported here. Lorcaserin was also reported to reduce deprivation-induced food intake, operant responding for food- and nicotine-motivated behaviours (Higgins et al. 2012). These data suggest that lorcaserin has effects to reduce responding for both food and drug rewards. One explanation for the overall pattern of behavioural responding after lorcaserin is that the drug has a dual mechanism of action to modulate neuronal systems involved in both (1) the processing of metabolic signals related to nutritional state and (2) reward-related processing (Asin et al. 1992; Higgins et al. 2012). Indeed, it may be that both these mechanisms are critical for the observed effects of lorcaserin on food intake. The pleasantness of food is known to decline as food is eaten, a phenomenon known as alliesthesia (Cabanac 1971). It is possible that lorcaserin enhances alliesthesia, such that food becomes less rewarding at a faster rate during a meal, thereby enhancing satiety. This suggestion requires further testing, but such a unifying explanation of the anorectic effects of lorcaserin is consistent with other evidence that points towards important interactions between homeostatic and reward processes in appetite control (Berthoud 2011).
In terms of the underlying neuronal mechanisms for lorcaserin and SB-742457 to promote satiety, elegant neurobiological studies have demonstrated a common population of pro-opiomelanocortin (POMC) neurones within the arcuate nucleus of the hypothalamus that are activated by 5-HT2C receptor agonists (Berglund et al. 2013; Burke et al. 2014) and also d-fenfluramine and sibutramine to signal their anorectic actions (Heisler et al. 2002, 2006; Lam et al. 2008; Xu et al. 2008, 2010; Berglund et al. 2013; Burke et al. 2014); and is consistent with their effects upon satiety (Hillebrand et al. 2002). Results from the Heisler laboratory have also implicated the paraventricular nucleus (PVN) within the hypothalamus as a target for a hypophagic dose of the selective 5-HT6 receptor antagonist, SB-399855 (Garfield et al. 2014). Hence, the arcuate-PVN circuit would appear to be important to mediate the hypophagic actions of both 5-HT2C receptor agonists and 5-HT6 receptor antagonists; the latter potentially increasing neuronal activity via disinhibition (Garfield et al. 2014, 2015). 5-HT2C receptors are also located on GABA neurons in the VTA and activation of these receptors has been reported to decrease DA release in the nucleus accumbens and frontal cortex (Di Matteo et al. 2002). This latter mechanism may underlie the effects of lorcaserin on reward-related responding.
Lorcaserin’s positive results from three phase III trials (Smith et al. 2010; Fidler et al. 2011; O’Neil et al. 2012; for meta-analysis of the data, see Chan et al. 2013) in the relative absence of adverse side effects, importantly including only a very small additional risk of cardiovascular events (Weissman et al. 2013), led to the FDA approving lorcaserin when combined with a reduced calorie diet and exercise regimen for obese (BMI ≥30) or overweight adults (BMI ≥27) who also have at least one co-morbid condition (e.g. hypertension, type-2 diabetes, dyslipidemia). Despite approval as a medication, there is still much to understand about lorcaserin’s effects on eating behaviour processes; in particular, its effects on reward processes and the relationship of such actions upon satiety. Knowledge of the specific behavioural mechanisms will be helpful in guiding advice and support for patients prescribed the drug, for example in educating them about what to expect from treatment.
Lorcaserin’s impact upon feeding behaviour of rats identified in the present study is consistent with the self-reporting of decreased appetite and hunger of patients receiving the drug (Martin et al. 2011), further supporting the use of microstructural analysis of licking behaviour in pre-clinical models to predict clinical outcome.
Whilst an early report indicated that activation of the 5-HT6 receptor may offer therapeutic potential to promote weight loss (Fisas et al. 2006), evidence from the use of various 5-HT6 receptor antagonists (Woolley et al. 2001; Garfield et al. 2014; for review, see Heal et al. 2008 that also includes a review of data presented at conferences) and knock-down of 5-HT6 receptor gene expression (Woolley et al. 2001) indicates inhibition of 5-HT6 receptor signalling possesses the clearest anorectic potential from targeting this receptor. This effect arising from a blockade of 5-HT’s actions contrasts the dogma of 5-HT’s influence upon feeding. Similar to the situation with lorcaserin, data arising from the use of 5-HT6 receptor antagonists in feeding studies use quantity of food intake (acute drug administration studies) or body weight (chronic drug administration studies) as the response readouts (Woolley et al. 2001; Garfield et al. 2014; for review, see Heal et al. 2008), although data presented at a conference indicates that the 5-HT6 receptor antagonist, PRX-07034 promotes satiety (Gannon et al. 2006; reviewed in Heal et al. 2008). In the present study, we assessed the action of the selective 5-HT6 receptor antagonist, SB-742457, using the microstructural analysis of licking behaviour paradigm. SB-742457 was selected for study as it is already a clinical candidate (for Alzheimer’s disease; e.g. Maher-Edwards et al. 2011) and hence would allow relatively rapid translation of the present pre-clinical study into clinical trial. SB-742457 at a dose likely to antagonise central 5-HT6 receptors (Idris et al. 2010; Witten et al. 2012) decreased the number of bouts of licking behaviour that was responsible for the reduction in food intake, demonstrating a drug-induced increase in satiety. This effect, however, was only evident at the lowest dose tested, indicating the presence of an inverted bell-shaped response curve for this drug, which has been reported when investigating the action of this drug on other types of behaviour (de Bruin et al. 2013). Similar to the data obtained with lorcaserin, an effect of SB-742457 upon the number of bouts of licking was apparent without an effect upon licking latency or lick rate, excluding motoric effects from influencing the response to drug.
In summary, in the present study the microstructural analysis of ingestive behaviour identified that both the relatively selective 5-HT2C receptor agonist, lorcaserin and the 5-HT6 receptor antagonist, SB-742457, displayed an anorectic action by primarily reducing the number of bouts of licking behaviour, which indicates a direct promotion of satiety by both drugs. A better understanding of the behavioural mechanism of action of anti-obesity drugs allows informed decisions to select drug combinations in the search for more efficacious anti-obesity therapies. In addition, for the lorcaserin study, the reverse translation of supporting data from the clinic further validates the predictive value of this behavioural model.
Acknowledgments
The authors are grateful to Dave Barber for assistance with data collection. This work was funded by the University of Birmingham.
Compliance with ethical standards
Conflict of interest
The authors declare that they have no competing interests.
References
- Asin KE, Davis JD, Bednarz L. Differential effects of serotonergic and cathecholaminergic drugs on ingestive behaviour. Psychopharmacology. 1992;109:415–421. doi: 10.1007/BF02247717. [DOI] [PubMed] [Google Scholar]
- Balcioglu A, Wurtman RJ. Sibutramine, a serotonin uptake inhibitor, increases dopamine concentrations in rat striatal and hypothalamic extracellular fluid. Neuropharmacology. 2000;39:2352–2359. doi: 10.1016/S0028-3908(00)00083-6. [DOI] [PubMed] [Google Scholar]
- Berglund ED, Liu C, Sohn JW, Kim MH, Lee CE, Vianna CR, Williams KW, Xu Y, Elmquist JK. Serotonin2C receptors in pro-opiomelanocortin neurons regulate energy and glucose homeostasis. J Clin Invest. 2013;123:5061–5070. doi: 10.1172/JCI70338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berthoud HR. Metabolic and hedonic drives in the neural control of appetite: who is the boss? Curr Opin Neurobiol. 2011;21:888–896. doi: 10.1016/j.conb.2011.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blundell JE. Is there a role for serotonin (5-hydroxytryptamine) in feeding? Int J Obes. 1977;1:15–42. [PubMed] [Google Scholar]
- Breslin PAS, Davis JD, Rosenak R. Saccharin increases the effectiveness of glucose in stimulating ingestion in rats but has little effect on negative feedback. Physiol Behav. 1996;60:411–416. doi: 10.1016/S0031-9384(96)80012-6. [DOI] [PubMed] [Google Scholar]
- Burke LK, Doslikova B, D’Agostino G, Garfield AS, Farooq G, Burdakov D, Low MJ, Rubinstein M, Evans ML, Bilups B, Heisler LK. 5-HT obesity medication via POMC-activation in maintained during ageing. Endocrinology. 2014;155:3732–3738. doi: 10.1210/en.2014-1223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cabanac M. Physiological role of pleasure. Science. 1971;173:1103–1107. doi: 10.1126/science.173.4002.1103. [DOI] [PubMed] [Google Scholar]
- Chan EW, He Y, Chui CS, Wong AY, Lau WC, Wong IC. Efficacy and safety of lorcaserin in obese adults: a meta-analysis of 1-year randomised controlled trials (RCTs) and narrative review on short-term RCTs. Obes Rev. 2013;14:383–392. doi: 10.1111/obr.12015. [DOI] [PubMed] [Google Scholar]
- Clifton PG, Lee MD, Dourish CT. Similarities in the action of Ro 60–0175, a 5-HT2C receptor agonist, and d-fenfluramine on feeding patterns in the rat. Psychopharmacology. 2000;152:256–267. doi: 10.1007/s002130000504. [DOI] [PubMed] [Google Scholar]
- Cooper SJ, Higgs S. Benzodiazepine effects on licking responses for sodium chloride solutions in water-deprived male rats. Physiol Behav. 2005;85:252–258. doi: 10.1016/j.physbeh.2005.03.027. [DOI] [PubMed] [Google Scholar]
- Danaei G, Ding EL, Mozaffarian D, Taylor B, Rehm J, Murray CJL, Ezzati M. The preventable causes of death in the United States: comparative risk assessment for dietary, lifestyle, and metabolic risk factors. Plos Med. 2009;6 doi: 10.1371/journal.pmed.1000058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis JD, Levine MW. A model for the control of ingestion. Psychol Rev. 1977;84:379–412. doi: 10.1037/0033-295X.84.4.379. [DOI] [PubMed] [Google Scholar]
- Davis JD, Smith GP. Analysis of the microstructure of the rhythmic tongue movements of rats ingesting maltose and sucrose solutions. Behav Neurosci. 1992;106:217–228. doi: 10.1037/0735-7044.106.1.217. [DOI] [PubMed] [Google Scholar]
- Davis JD, Smith GP, Singh B, McCann DL. The impact of sucrose-derived unconditioned and conditioned negative feedback on the microstructure of ingestive behaviour. Physiol Behav. 2001;72:393–402. doi: 10.1016/S0031-9384(00)00442-X. [DOI] [PubMed] [Google Scholar]
- de Bruin NM, van Drimmelen M, Kops M, van Elk J, Wetering MM, Schwienbacher I. Effects of resperidone, clozapine and the 5-HT6 antagonist GSK-742457 on PCP-induced deficits in reversal learning in the two-lever operant task in male Sprague Dawley rats. Behav Brain Res. 2013;244:15–28. doi: 10.1016/j.bbr.2013.01.035. [DOI] [PubMed] [Google Scholar]
- Di Matteo V, Cacchio M, Di Giulio C, Esposito E. Role of serotonin 2C receptors in the control of brain dopaminergic function. Pharmacol Biochem Behav. 2002;71:727–734. doi: 10.1016/S0091-3057(01)00705-5. [DOI] [PubMed] [Google Scholar]
- Dutton AC, Barnes NM. Anti-obesity pharmacotherapy: future perspectives utilising 5-HT2C receptor agonists. Drug Discov Today: Ther Strategies. 2006;3:577–583. [Google Scholar]
- Fidler MC, Sanchez M, Raether B, Weissman NJ, Smith SR, Shanahan WR, Anderson CM. A one-year randomised trial of lorcaserin for weight loss in obese and overweight adults: the BLOSSOM trial. J Clin Endocrinol Metab. 2011;96:3067–3077. doi: 10.1210/jc.2011-1256. [DOI] [PubMed] [Google Scholar]
- Fisas A, Codony X, Romero G, Dordal A, Giraldo J, Merce R, Holenz J, Heal D, Buschmann H, Pauwels PJ. Chronic 5-HT6 receptor modulation by E-6837 induces hypophagia and sustained weight loss in diet-induced obese rats. Br J Pharmacol. 2006;148:973–983. doi: 10.1038/sj.bjp.0706807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fitzgerald LW, Burn TC, Brown BS, Patterson JP, Corjay MH, Valentine PA, Sun JH, Link JR, Abbaszade I, Hollis JM, Largent BL, Hartig PR, Hollis GF, Meunier PC, Robichaud AJ, Robertson DW. Possible role of valvular serotonin 5-HT2B receptors in the cardiopathy associated with fenfluramine. Mol Pharmacol. 2000;57:75–81. [PubMed] [Google Scholar]
- Fletcher PJ, Tampakeras M, Sinyard J, Slassi A, Isaac M, Higgins GA. Characterizing the effects of 5-HT2C receptor ligands on motor activity and feeding behaviour in 5-HT2C receptor knockout mice. Neuropharmacology. 2009;57:259–267. doi: 10.1016/j.neuropharm.2009.05.011. [DOI] [PubMed] [Google Scholar]
- Gannon KS, Heal DJ, Cheetham SC, Jackson HC, Seeley RJ, Melendez R et al. PRX-07034, a potent and selective 5-HT6 receptor antagonist, reduces food intake and body weight. Proceedings of the Serotonin Club Sixth IUPHAR Satellite Meeting on Serotonin, Hokkaido, Japan. 2006
- Garfield AS, Burke LK, Shaw J, Evans ML, Heisler LK. Distribution of cells responsive to 5-HT6 receptor antagonist-induced hypophagia. Behav Bran Res. 2014;266:201–206. doi: 10.1016/j.bbr.2014.02.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garfield AS, Li C, Madara JC, Shah BP, Webber E, Steger JS, Campbell JN, Gavrilova O, Lee CE, Olson DP, Elmquist JK, Tannous BA, Krashes MJ, Lowell BB. A neural basis for melanocortin-4 receptor-regulated appetite. Nat Neurosci. 2015;18:863–871. doi: 10.1038/nn.4011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halford JC, Boyland EJ, Lawton CL, Blundell JE, Harrold JA. Serotonergic anti-obesity agents: past experience and future prospects. Drugs. 2011;71:2247–2255. doi: 10.2165/11596680-000000000-00000. [DOI] [PubMed] [Google Scholar]
- Heal DJ, Smith SL, Fisas A, Codony X, Buschmann H. Selective 5-HT6 receptor ligands: progress in the development of a novel pharmacological approach to the treatment of obesity and related metabolic disorders. Pharmacol Ther. 2008;117:207–231. doi: 10.1016/j.pharmthera.2007.08.006. [DOI] [PubMed] [Google Scholar]
- Heisler LK, Cowley MA, Tecott LH, Fan W, Low MJ, Smart JL, Rubinstein M, Tatro JB, Marcus JN, Holstege H, Lee CE, Cone RD, Elmquist JK. Activation of central melanocortin pathways by fenfluramine. Science. 2002;297:609–611. doi: 10.1126/science.1072327. [DOI] [PubMed] [Google Scholar]
- Heisler LK, Jobst EE, Sutton GM, Zhou L, Borok E, Thornton-Jones Z, Liu HY, Zigman JM, Balhasar N, Kishi T, Lee CE, Aschkenasi CJ, Zhang CY, Yu J, Boss O, Mountjoy KG, Clifton PG, Lowell BB, Friedman JM, Horvath T, Butler AA, Elmquist JK, Cowley MA. Serotonin reciprocally regulates melanocortin neurons to modulate food intake. Neuron. 2006;51:239–249. doi: 10.1016/j.neuron.2006.06.004. [DOI] [PubMed] [Google Scholar]
- Higgins GA, Silenieks LB, Roßmann A, Rizos Z, Noble K, Soko AD, Fletcher PJ (2012) The 5-HT2C receptor agonist lorcaserin reduces nicotine self-administration, discrimination, and reinstatement: relationship to feeding behavior and impulse control. Neuropsychopharmacology 37(5):1177–1191 [DOI] [PMC free article] [PubMed]
- Higgins GA, Desnoyer J, van Niekerk A, Silenieks LB, Lau W, Thevarkunnel S, Izhakova J, DeLannoy IAM, Fletcher PJ, DeLay J, Dobson H. Characterization of the 5-HT2C receptor agonist lorcaserin on efficacy and safety measures in a rat model of diet-induced obesity. Pharmacol Res Perspect. 2014;3 doi: 10.1002/prp2.84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Higgs S, Cooper SJ. Effects of the benzodiazepine receptor inverse agonist. Ro 15–4513 on the ingestion of sucrose and sodium saccharin solutions: a microstructural analysis of licking behavior. Behav Neurosci. 1996;110:559–566. doi: 10.1037/0735-7044.110.3.559. [DOI] [PubMed] [Google Scholar]
- Higgs S, Cooper SJ. Midazolam induced rapid changes in licking behaviour: evidence for involvement of endogenous opioid peptides. Psychopharmacology. 1997;131:278–286. doi: 10.1007/s002130050294. [DOI] [PubMed] [Google Scholar]
- Higgs S, Cooper SJ. Effects of benzodiazepine receptor ligands on the ingestion of sucrose, intralipid, and maltodextrin: an investigation using a microstructural analysis of licking behavior in a brief contact test. Behav Neurosci. 1998;112:447–457. doi: 10.1037/0735-7044.112.2.447. [DOI] [PubMed] [Google Scholar]
- Higgs S, Cooper SJ (2000) The effect of the dopamine D2 receptor antagonist raclopride on the pattern of licking microstructure induced by midazolam in the rat. Eur J Pharmacol 409(1):73–80 [DOI] [PubMed]
- Higgs S, Williams CM, Kirkham TC. Cannabinoid influences on palatability: microstructural analysis of sucrose drinking after Δ9-THC, anandamide, 2-AG and SR141716. Psychopharmacology. 2003;165:370–377. doi: 10.1007/s00213-002-1263-3. [DOI] [PubMed] [Google Scholar]
- Higgs S, Cooper AJ, Barnes NM. Reversal of sibutramine-induced anorexia with a selective 5-HT2C receptor antagonist. Psychopharmacology. 2011;214:941–947. doi: 10.1007/s00213-010-2106-2. [DOI] [PubMed] [Google Scholar]
- Hillebrand JJG, de Wied D, Adan RAH. Neuropeptides, food intake and body weight regulation: a hypothalamic focus. Peptides. 2002;23:2283–2306. doi: 10.1016/S0196-9781(02)00269-3. [DOI] [PubMed] [Google Scholar]
- Idris N, Neill J, Grayson B, Bang-Andersen B, Witten LM, Tottrup L, Arnt J. Sertindole improves sub-chronic PCP-induced reversal learning and episodic memory deficits in rodents: involvement of 5-HT6 and 5-HT2A receptor mechanisms. Psychopharmacology. 2010;208:23–36. doi: 10.1007/s00213-009-1702-5. [DOI] [PubMed] [Google Scholar]
- Jackson HC, Bearham MC, Hutchins LJ, Mazurkiewicz SE, Needham AM, Heal DJ. Investigation of the mechanisms underlying the hypophagic effects of the 5-HT and noradrenaline reuptake inhibitor, sibutramine, in the rat. Br J Pharmacol. 1997;121:1613–1618. doi: 10.1038/sj.bjp.0701311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lam DD, Przydzial MJ, Ridley SH, Yeo GS, Rochford JJ, O’Rahilly S, Heisler LK. Serotonin 5-HT2C receptor agonist promotes hypophagia via downstream activation of melanocortin 4 receptors. Endocrinology. 2008;149:1323–1328. doi: 10.1210/en.2007-1321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Launay JM, Herve P, Peoc’h K, Tournois C, Callebert J, Nebigil CG, Etienne N, Drouet L, Humbert M, Simonneau G, Maroteaux L. Function of the serotonin 5-hydroxytryptmine2B receptor in pulmonary hypertension. Nat Med. 2002;8:1129–1135. doi: 10.1038/nm764. [DOI] [PubMed] [Google Scholar]
- Maher-Edwards G, Dixon R, Hunter J, Gold M, Hopton G, Jacobs G, Hunter J, Williams P. SB-742457 and donepezil in Alzheimer disease: a randomised, placebo-controlled study. Int J Geriatr Psychiatry. 2011;26:536–544. doi: 10.1002/gps.2562. [DOI] [PubMed] [Google Scholar]
- Martin CK, Redman LM, Zhang J, Sanchez M, Anderson CM, Smith SR, Ravussin E. Lorcaserin, a 5-HT2C receptor agonist, reduces body weight by decreasing energy intake without influencing energy expenditure. J Clin Endocrinol Metab. 2011;96:837–845. doi: 10.1210/jc.2010-1848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McNeely W, Goa KL. Sibutramine: a review of its contribution to the management of obesity. Drugs. 1998;56:1093–1124. doi: 10.2165/00003495-199856060-00019. [DOI] [PubMed] [Google Scholar]
- Meltzer HY, Roth BL. Lorcaserin and pimavanserin: emerging selectivity of serotonin receptor subtype-targeted drugs. J Clin Invest. 2013;123:4986–4991. doi: 10.1172/JCI70678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Michino M, Beuming T, Donthamsetti P, Newman AH, Javitch JA, Shi L. What can crystal structures of aminergic receptors tell us about designing subtype-selective ligands? Pharmacol Rev. 2015;67:198–213. doi: 10.1124/pr.114.009944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nichols DE. Hallucingens. Pharmacol Ther. 2004;101:131–181. doi: 10.1016/j.pharmthera.2003.11.002. [DOI] [PubMed] [Google Scholar]
- O’Neil PM, Smith SR, Weissman NJ, Fidler MC, Sanchez M, Zhang J, Raether B, Anderson CM, Shanahan WR. Randomized placebo-controlled clinical trial of lorcaserin for weight loss in type 2 diabetes mellitus: the BLOOM-DM study. Obesity. 2012;20:1426–1436. doi: 10.1038/oby.2012.66. [DOI] [PubMed] [Google Scholar]
- Ogden CL, Carroll MD, Kit BK, Flegal KM. Prevalence of childhood and adult obesity in the United States, 2011–2012. JAMA. 2014;311:806–814. doi: 10.1001/jama.2014.732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rothman RB, Baumann MH, Savage JE, Rauser L, McBride A, Hufeisen SJ, Roth BL. Evidence for possible involvement of 5-HT2B receptors in the cardiac valvulopathy associated with fenfluramine and other serotonergic medications. Circulation. 2000;102:2836–2841. doi: 10.1161/01.CIR.102.23.2836. [DOI] [PubMed] [Google Scholar]
- Smith BM, Smith JM, Tsai JH, Schultz JA, Gilson CA, Estrada SA, Chen RR, Park DM, Prieto EB, Gallardo CS, Sengupta D, Dosa PI, Covel JA, Ren A, Webb RR, Beeley NRA, Martin M, Morgan M, Espitia S, Saldana HR, Bjenning C, Whelan KT, Grottick AJ, Menzaghi F, Thomsen WJ. Discovery and structure-activity relationship of (1R)-8-chloro-2,3,4,5-tetrahydro-1-methyl-1H-3-benzazepine (lorcaserin), a selective serotonin 5-HT2C receptor agonist for the treatment of obesity. J Med Chem. 2008;51:305–313. doi: 10.1021/jm0709034. [DOI] [PubMed] [Google Scholar]
- Smith SR, Weissman NJ, Anderson CM, Sanchez M, Chuang E, Stubbe S, Bays H, Shanahan WR. Multicenter. Placebo-controlled trial of lorcaserin for weight management. N Eng J Med. 2010;363:245–256. doi: 10.1056/NEJMoa0909809. [DOI] [PubMed] [Google Scholar]
- Stellar E, Hill JH. The rat’s rate of drinking as a function of water deprivation. J Comp Physiol Psychol. 1952;5:96–102. doi: 10.1037/h0062150. [DOI] [PubMed] [Google Scholar]
- Tallett AJ, Blundell JE, Rodgers RJ. Sibutramine-induced anorexia: potent, dose-dependent and behaiourally-selective profile in male rats. Behav Brain Res. 2009;198:359–365. doi: 10.1016/j.bbr.2008.11.011. [DOI] [PubMed] [Google Scholar]
- Thomas JM, Dourish CT, Tomlinson JW, Hassan-Smith Z, Higgs S. Effects of the 5-HT2C receptor agonist meta-chlorophenylpiperazine on appetite, food intake and emotional processing in healthy volunteers. Psychopharmacology. 2014;231(12):2449–2459. doi: 10.1007/s00213-013-3409-x. [DOI] [PubMed] [Google Scholar]
- Thomsen WJ, Grottick AJ, Menzaghi F, Reyes-Saldana H, Espitia S, Yuskin D, Whelan K, Martin M, Morgan M, Chen W, Al-Shamma H, Smith B, Chalmers D, Behan D. Lorcaserin, a novel selective human 5-hydroxytryptamine2C agonist: in vitro and in vivo pharmacological characterization. J Pharmacol Exp Ther. 2008;325:577–587. doi: 10.1124/jpet.107.133348. [DOI] [PubMed] [Google Scholar]
- Vickers SP, Dourish CT. Serotonin receptor ligands and the treatment of obesity. Curr Opin Investig Drugs. 2004;5:377–388. [PubMed] [Google Scholar]
- Vickers SP, Clifton PG, Dourish CT, Tecott LH. Reduced satiating effect of d-fenfluramine in serotonin 5-HT2C receptor mutant mice. Psychopharmacology. 1999;143:309–314. doi: 10.1007/s002130050952. [DOI] [PubMed] [Google Scholar]
- Weintraub M, Hasday JD, Mushlin AI, Lockwood DH. A double-blind clinical trial in weight control: use of fenfluramine and phentermine alone and in combination. Arch Intern Med. 1984;144:1143–1148. doi: 10.1001/archinte.1984.00350180055008. [DOI] [PubMed] [Google Scholar]
- Weissman NJ, Sanchez M, Koch GG, Smith SR, Shanahan WR, Anderson CM. Echocardiographic assessment of cardiac valular regurgitation with lorcaserin from analysis of 3 phase 3 clinical trials. Circ Cardiovasc Imaging. 2013;6:560–567. doi: 10.1161/CIRCIMAGING.112.000128. [DOI] [PubMed] [Google Scholar]
- Witten L, Bang-Andersen B, Nielsen SM, Miller S, Christoffersen CT, Stensbol TB, Brennum LT, Arnt J. Characterization of [3H]Lu AE60157 ([3H]8-(4-methylpiperazin-1-yl)-3-phenylsulfonylquinoline) binding to 5-hydroxytryptamine6 (5-HT6) receptors in vivo. Eur J Pharmacol. 2012;676:6–11. doi: 10.1016/j.ejphar.2011.11.029. [DOI] [PubMed] [Google Scholar]
- Woolley ML, Bentley JC, Sleight AJ, Marsden CA, Fone KC. A role for 5-ht6 receptors in retention of spatial learning in the Morris water maze. Neuropharmacology. 2001;41:210–219. doi: 10.1016/S0028-3908(01)00056-9. [DOI] [PubMed] [Google Scholar]
- Xu Y, Jones JE, Kohno D, Williams KW, Lee CE, Choi MJ, Anderson JG, Heisler LK, Zigman JM, Lowell BB, Elmquist JK. 5-HT2CRs expressed by pro-opiomelanocortin neurons regulate energy homeostasis. Neuron. 2008;60:582–589. doi: 10.1016/j.neuron.2008.09.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu Y, Jones JE, Lauzon DA, Anderson JG, Balthasar N, Heisler LK, Zinn AR, Lowell BB, Elmquist JK. A serotonin and melanocortin circuit mediates D-defenfluramine anorexia. J Neurosci. 2010;30:14630–14634. doi: 10.1523/JNEUROSCI.5412-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]