Abstract
The surge of luteinizing hormone triggers the genomic reprogramming, cell differentiation, and tissue remodeling of the ovulated follicle, leading to the formation of the corpus luteum. During this process, called luteinization, follicular granulosa cells begin expressing a new set of genes that allow the resulting luteal cells to survive in a vastly different hormonal environment and to produce the extremely high amounts of progesterone (P4) needed to sustain pregnancy. To better understand the molecular mechanisms involved in the regulation of luteal P4 production in vivo, the transcription factors GATA4 and GATA6 were knocked down in the corpus luteum by crossing mice carrying Gata4 and Gata6 floxed genes with mice carrying Cre recombinase fused to the progesterone receptor. This receptor is expressed exclusively in granulosa cells after the luteinizing hormone surge, leading to recombination of floxed genes during follicle luteinization. The findings demonstrated that GATA4 and GATA6 are essential for female fertility, whereas targeting either factor alone causes subfertility. When compared to control mice, serum P4 levels and luteal expression of key steroidogenic genes were significantly lower in conditional knockdown mice. The results also showed that GATA4 and GATA6 are required for the expression of the receptors for prolactin and luteinizing hormone, the main luteotropic hormones in mice. The findings demonstrate that GATA4 and GATA6 are crucial regulators of luteal steroidogenesis and are required for the normal response of luteal cells to luteotropins.
Keywords: corpus luteum, female infertility, GATA, luteinizing hormone (LH/LH receptor), progesterone/progesterone receptor
INTRODUCTION
The corpus luteum is one of the most remarkable endocrine glands in the human body and, in proportion to its mass, is the most active steroid-secreting tissue. The formation of the corpus luteum is triggered by the midcycle surge of luteinizing hormone (LH), which stimulates the final maturation of the oocyte and its release at ovulation and converts the ovulated follicle into a new endocrine gland that produces almost exclusively progesterone (P4), a steroid hormone. Luteal P4 promotes uterine receptivity for blastocyst implantation. Despite the vital role of the corpus luteum in female fertility, the molecular mechanisms involved in the maintenance of luteal function and steroidogenesis are not fully understood.
Luteal cells from rodents, farm animals, and humans express the transcription factors GATA4 and GATA6 [1–5]. Insights into the role of GATA4 in luteal steroidogenesis are exclusively provided by in vitro transactivation experiments showing that this factor can target the promoter of steroidogenic genes, including steroidogenic acute regulatory protein (STAR) [2, 6–9], cholesterol side-chain cleavage (CYP11A1) [10], 3β-hydroxysteroid dehydrogenase (HSD3B1) [11], aromatase (CYP19A1) [9, 12–14], inhibin alpha (INHA) [2, 9, 15, 16], anti-Müllerian hormone [9], and others (for a review, see [17]). However, transactivation experiments do not necessarily recapitulate the in vivo regulation of luteal function. The only in vivo evidence suggesting the involvement of GATA on the regulation of luteal steroidogenesis is provided by the colocalization of GATA4/6 and CYP11A1 to the corpus luteum of rats [1]. Finally, in vitro experiments using small interference RNA (siRNA) in porcine luteal cells showed that GATA6 knockdown reduces Star mRNA and P4 levels by 60%, whereas knockdown of GATA4 has no effect on basal and cAMP-induced P4 production or Star expression [18]. These findings suggest that GATA6 plays a more dominant role on the regulation of luteal P4 production; nevertheless, functional in vivo studies are needed to support this conclusion and to determine the role of GATA4 and GATA6 on the regulation of luteal function.
Previously, we demonstrated that the conditional knockdown of GATA4 and GATA6 in granulosa cells disrupts follicle development and ovulation [19, 20]. This block in follicular growth prevents the formation of corpora lutea; therefore, studies to examine the roles of GATA4 and GATA6 in luteal function were not possible. Here, GATA4 and GATA6 functions in luteal cells and the molecular mechanisms involved in the regulation of luteal P4 synthesis in vivo were examined by targeting the Gata4 and Gata6 genes during luteinization using the Cre/lox system. This approach demonstrated that GATA4 and GATA6 are essential for luteal P4 synthesis and consequently for the maintenance of pregnancy in mice.
MATERIALS AND METHODS
Reagents
DMEM/F12 medium, equine chorionic gonadotropin (eCG), human chorionic gonadotropin (hCG), P4, and all other media components were purchased from Sigma unless otherwise specified.
Animals
Animals expressing Cre recombinase fused to the progesterone receptor (PRcre) were previously described [21]. Gata4f/f mice were kindly provided by Dr. William Pu (Department of Cardiology, Children's Hospital Boston). Gata6f/f mice were obtained from the Jackson Laboratory. The mice carrying floxed alleles of the Gata4 and/or the Gata6 genes were previously described [20]. PRcre and Gata4f/f, Gata6f/f, or Gata4/6f/f mice were crossed to generate PRcre:Gata4f/f, PRcre:Gata6f/f, and PRcre:Gata4/6f/f mice. Only PR+/cre animals were used because PRcre/cre animals are infertile [21]. The animals were treated in accordance with the National Institutes of Health Guide for Care and Use of Laboratory Animals, and all the protocols were approved by the University of Illinois at Chicago Animal Care Committee.
Estrous Cycling
Vaginal smears were collected daily from adult females, that is, ∼Day 55 (∼d55), over the course of 14 days. The vaginal smears were evaluated microscopically to determine the stage of the cycle.
Ovulation Assay
Adult females (d90–d100), were injected subcutaneously with 7.5 international units (IU) of eCG for 48 h followed by treatment with 7.5 IU of hCG to induce ovulation. Next, 17 h after the hCG injection, the animals were euthanized and oocytes from each oviduct were collected and counted.
Immunohistochemistry
Immunohistochemistry was performed as previously described [20]. Slides were developed using a VECTASTAIN Elite ABC kit and 3,3′-diamidinobenzidine (Vector Laboratories) following the manufacturer's recommendations and then counterstained with Gill hematoxylin.
Western Blot Analysis
The control and PRcre:Gata4/6f/f mice were treated with 7.5 IU of eCG followed by 7.5 IU of hCG for 96 h to induce ovulation and luteinization. Corpora lutea were isolated and then homogenized in ice-cold RIPA lysis buffer using a Dounce tissue grinder, followed by sonification. Samples were centrifuged to eliminate cellular debris, and the protein lysate was collected. Protein concentration was determined using the Pierce BCA Protein Kit Assay (Life Technologies) with bovine serum albumin as the standard. The samples were denatured using sample buffer followed by boiling at 90°C for 10 min. Twelve micrograms of protein were separated on 12% Bis-Tris-PAGE gels. Samples were transferred to nitrocellulose membranes followed by incubation in 5% nonfat dry milk for 2 h at room temperature to block nonspecific binding. Membranes were washed with 2 mM Tris, 15 mM NaCl, and 0.1% Tween 20, and incubated overnight at 4°C with anti-GATA4 (sc9053, 1:500 dilution; Santa Cruz), anti-GATA-6 (sc9055, 1:100 dilution; Santa Cruz), STAR (1:1000 dilution, kindly donated by Dr. D.B. Hales), and anti-β-actin (ACTB) (ab8227, 1:500 dilution; Abcam). Membranes were washed and incubated with an anti-rabbit secondary antibody conjugated to horseradish peroxidase (1:10 000 dilution; Jackson ImmunoResearch) for 2 h at room temperature. Protein-antibody complexes were visualized using SuperSignal West Pico Chemiluminescent Substrate (Thermo Scientific) or SuperSignal West Femto Maximum Sensitivity Substrate (Thermo Scientific) depending on the abundance of the protein and the sensitivity of the antibody.
RNA Isolation and Quantitative Real-Time PCR Analysis
The control, PRcre:Gata4f/f, PRcre:Gata6f/f, and PRcre:Gata4/6f/f mice were treated with 7.5 IU of eCG followed by 7.5 IU of hCG for 36, 48, 72, or 96 h to induce ovulation and luteinization. Corpora lutea were isolated and then homogenized in 1 ml of TRIzol reagent (Invitrogen) using a Dounce tissue grinder. Total RNA was isolated using TRIzol reagent (Invitrogen) following the manufacturer's instructions. One microgram of total RNA was reverse transcribed at 42°C for 1 h using Moloney murine leukemia virus reverse transcriptase (Invitrogen). The cDNA was diluted to 100 μl in water, and 5 μl of this dilution was used for each reaction. The identity and size of all the PCR products were confirmed by sequencing and gel analysis. The results are expressed as the ratio between the copies per nanogram of total RNA of the gene of interest and ribosomal protein L19 (Rpl19). Primer sequences are provided in Table 1.
TABLE 1.
Primers used for quantitative real-time PCR.
Progesterone Assessment
Truncal blood was collected from the control, PRcre:Gata4f/f, PRcre:Gata6f/f, or PRcre:Gata4/6f/f mice under two conditions: 1) control, PRcre:Gata4f/f, PRcre:Gata6f/f, or PRcre:Gata4/6f/f mice were injected with 7.5 IU of eCG for 48 h followed by treatment with 7.5 IU of hCG for 17, 24, 36, 48, 72 or 96 h to induce ovulation and luteinization or 2) control or PRcre:Gata4/6f/f female mice were mated with males of proven fertility until confirmation of copulation by the presence of a vaginal plug. Truncal blood was collected from the mice at the time of harvest. Plasma was isolated by centrifugation of blood at 3000 × g for 5 min. Progesterone plasma levels were determined using a DRG Progesterone ELISA kit. Progesterone levels were determined in the control plasma samples at a 1:20 dilution and in the PRcre:Gata4f/f, PRcre:Gata6f/f, and PRcre:Gata4/6f/f plasma samples at a 1:5 dilution. The sensitivity of the P4 assays used was 0.045 ng/ml.
Luteal Cell Culture
Luteinizing granulosa cells were isolated from preovulatory follicles from immature 23-day-old Gata4/6f/f mice treated with eCG for 48 h followed by hCG treatment for 7 h. Cells were infected with adenoviral Cre recombinase (AdCRE) (University of Iowa Gene Vector Transfer Core) or virus-expressing green fluorescent protein (GFP) at a multiplicity of infection of 10. For RNA expression, cells were treated with AdCRE or AdGFP for 48 h prior to treatments with 1 mM N6,2′-O-dibutyryladenosine 3′,5′-cyclic monophosphate (dbcAMP) or 50 ng/ml hCG; dbcAMP is a cell-permeable cAMP analog that activates cAMP-dependent protein kinase (PKA). Cells were cultured for 48 h after treatments and then harvested for RNA isolation.
Statistics
Data are expressed as the mean ± SEM. Multiple group statistical analyses were performed by one-way ANOVA followed by the Tukey test for multiple comparisons. Two-group comparisons were performed using a t-test for independent samples. Differences between groups were considered to be statistically significant at P < 0.05.
RESULTS
Deletion of GATA4 and GATA6 During Luteinization Leads to Female Infertility
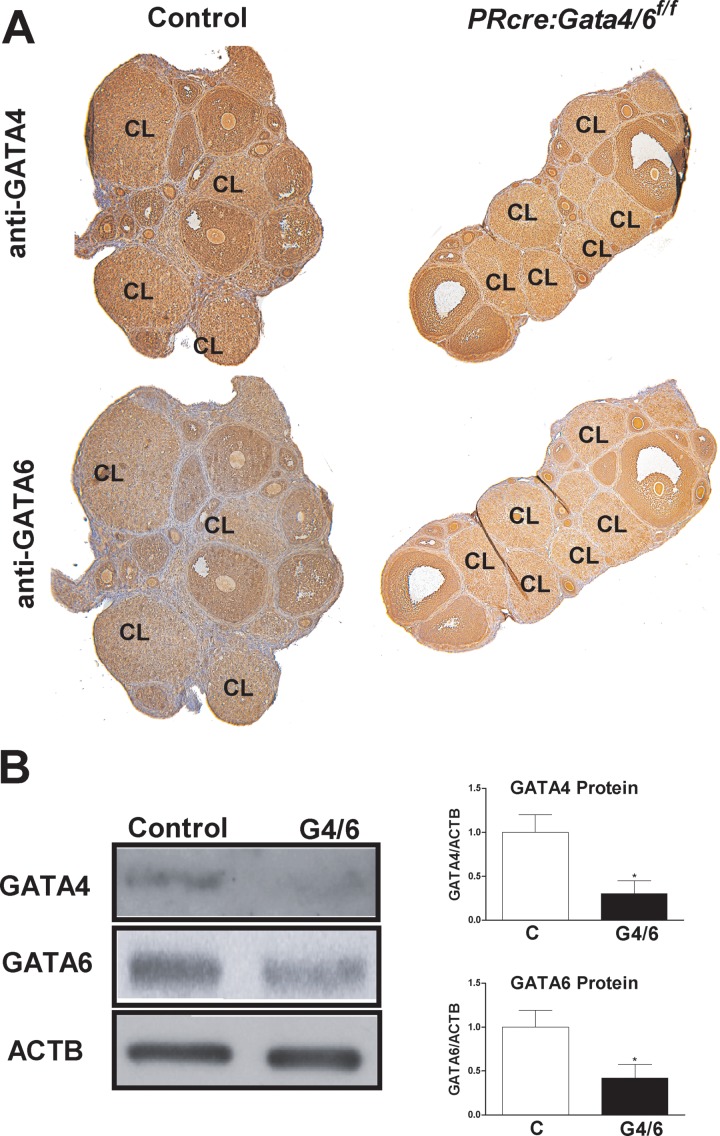
To examine the roles of GATA4 and GATA6 in corpus luteum function, mice bearing floxed Gata4 and Gata6 genes were crossed with PRcre mice. In PRcre mice, Cre recombinase is expressed only in granulosa cells during the luteinization process and leads to the targeted deletion of floxed genes in the corpus luteum [21]. Using this approach, Gata4 and Gata6 were targeted alone or together via the generation of PRcre:Gata4f/f, PRcre:Gata6 f/f, and PRcre:Gata4/6f/f mice, respectively. As controls, PRcre:Gata4/6 f/+ mice were used. The control and PRcre:Gata4/6f/f mice were treated sequentially with eCG and hCG as described in the Materials and Methods section. Immunohistochemical analysis of the ovaries of these mice revealed that GATA4 and GATA6 proteins were decreased in the corpus luteum 96 h after hCG treatment (Fig. 1A). Western blot analysis showed significantly diminished expression of GATA4 and GATA6 proteins in the corpus luteum of PRcre:Gata4/6f/f mice when compared with the controls (Fig. 1B).
FIG. 1.
Expression of GATA4 and GATA6 in PRcre:Gata4/6f/f mice. Control (C) or PRcre:Gata4/6f/f mice were treated with 7.5 IU of eCG for 48 h, followed by treatment with 7.5 IU of hCG for 96 h. A) Immunohistochemical analysis of GATA4 and GATA6 protein expression in whole ovary sections. CL: corpus luteum. B) Corpora lutea isolated from the control and PRcre:Gata4/6f/f (G4/6) mice were used for the isolation of total protein for Western blot analysis to detect GATA4, GATA6, and β-actin (ACTB) (*P < 0.05 vs. C, t-test, n = 5).
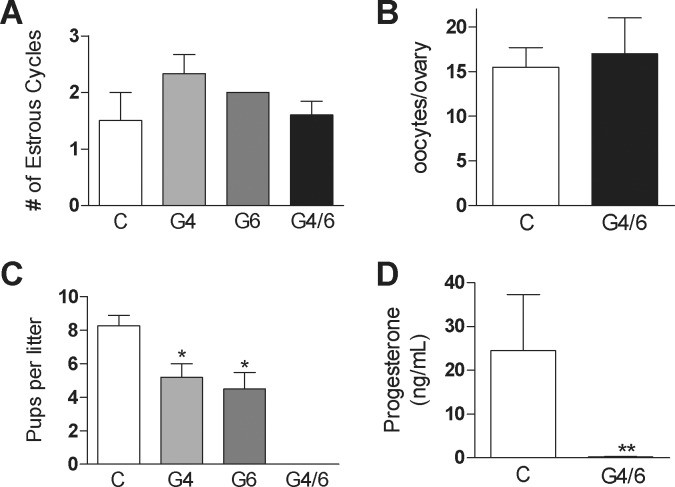
Next, we evaluated the main reproductive functions of the control and GATA conditional knockdown mice. No differences in the estrous cycle length or ovulation rate were detected between the control and PRcre:Gata4f/f, PRcre:Gata6f/f, or PRcre:Gata4/6f/f female mice (Fig. 2, A and B, and data not shown). In addition, vaginal plugs were found in the control and experimental females after mating with wild-type males (data not shown).
FIG. 2.
Fertility, cyclicity, and ovulation of GATA-deficient mice. A) Number of estrous cycles over a 14-day period in PRcre (C), PRcre:Gata4f/f (G4), PRcre:Gata6f/f (G6), and PRcre:Gata4/6f/f (G4/6) mice. B) Number of ovulated oocytes in control and G4/6 females 17 h following hCG treatment. C) Average number of pups per litter in C, G4, G6, and G4/6 female mice after continuous mating over a 6-mo period with males of proven fertility (*P < 0.05 vs. C, one-way ANOVA, n = 3). D) Serum P4 levels in female mice 9 days postcoitum (**P < 0.01 vs. control, t-test, n = 5).
The mating of the control and experimental animals with males of proven fertility for 6 mo demonstrated that PRcre:Gata4f/f and Prcre:Gata6f/f females are subfertile, characterized by a significant decrease in the number of pups per litter when compared to the control mice (Fig. 2C). In marked contrast to the single knockouts, PRcre:Gata4/6f/f females were infertile (Fig. 2C). This result agrees with serum P4 levels determined 9 days after copulation, which showed that PRcre:Gata4/6f/f females had significantly lower levels of P4 in circulation when compared with the control mice (Fig. 2D). These results indicate that the infertility of PRcre:Gata4/6f/f females is not due to ovarian acyclicity, anovulation, or abnormal sexual behavior, but is most probably due to luteal dysfunction.
GATA Factors Are Required for Luteal P4 Production
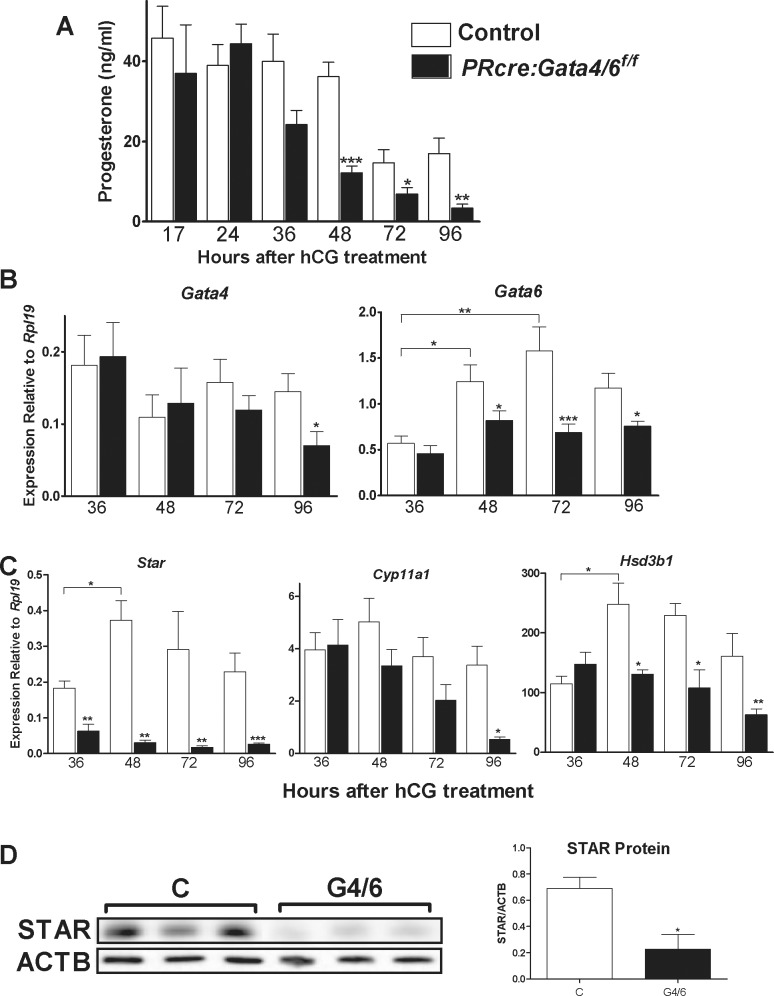
Because only PRcre:Gata4/6f/f mice are infertile and produce extremely low levels of P4, we next examined luteal function in the PRcre:Gata4/6f/f mice, and P4 levels were measured in the serum of animals treated sequentially with eCG and hCG. As shown in Figure 3A, a time-course experiment revealed that P4 levels in the PRcre:Gata4/6f/f mice were comparable to those found in the control animals at 17 and 24 h after hCG treatments. At 36 h, the P4 levels in the PRcre:Gata4/6f/f mice tended to be lower than in the controls, although the difference was not statistically significant (P = 0.08). However, P4 levels were significantly lower in the PRcre:Gata4/6f/f mice when compared with the controls at 48, 72, and 96 h after hCG treatment.
FIG. 3.
Serum P4 and luteal steroidogenic gene expression in PRcre:Gata4/6f/f mice. A) Serum P4 levels were measured in control (C) or PRcre:Gata4/6f/f (G4/6) female mice treated with eCG followed by treatment with hCG for 17, 24, 36, 48, 72, or 96 h (*P < 0.05, **P < 0.01, ***P < 0.001 vs. C, one-way ANOVA, n = 5). B and C) Gata4, Gata6, Star, Cyp11a1, and Hsd3b1 mRNA levels in C and G4/6 female mice treated with eCG and hCG for 36, 48, 72, or 96 h. Quantification of mRNA expression was performed using quantitative PCR (*P < 0.05, **P < 0.01, ***P < 0.001 vs. C or vs. 36 h as indicated by t-test, n = 8). D) Total protein isolated from the corpus luteum of C or G4/6 female mice treated with hCG for 96 h. Western blot analysis was used to detect STAR total protein levels. Quantification was performed as a ratio of STAR to ACTB as a loading control (*P < 0.05 vs. C, t-test, n = 5).
In the control mice, Gata4 mRNA expression tended to decrease over time after hCG treatment (Fig. 3B, left). However, Gata4 mRNA levels were not significantly different between the control and PRcre:Gata4/6f/f mice 36 and 48 h after treatment with hCG. Gata4 mRNA levels tended to be lower in the PRcre:Gata4/6f/f mice than in the controls 72 h after hCG. A significant decrease in Gata4 mRNA levels was observed in the PRcre:Gata4/6f/f mice 96 h after hCG treatment when compared with the control animals.
In contrast to Gata4, the Gata6 mRNA levels in the control mice increased significantly 48 and 72 h after hCG when compared to 36 h for the control group (Fig. 3B, right). These increases in luteal Gata6 expression did not occur in the PRcre:Gata4/6f/f mice. Thus, Gata6 mRNA levels tended to be lower at 36 h and were significantly lower in the PRcre:Gata4/6f/f mice after 48, 72, and 96 h when compared to the controls.
To determine the chronological development of P4 synthesis inhibition, the luteal mRNA content of Star, Cyp11a1, and Hsd3b1, three genes essential for P4 synthesis, was quantified 36, 48, 72, and 96 h following hCG treatment. In the control mice, Star mRNA levels increased from 36 to 48 h after hCG (Fig. 3C). However, the steady-state levels of Star mRNA were significantly decreased in the PRcre:Gata4/6f/f mice at all times after hCG (Fig. 3C). The percentage of Star decrease in the PRcre:Gata4/6f/f mice was only 50% at 36 h, whereas the inhibitions at 48, 72, and 96 h were 92%, 94%, and 96%, respectively. In contrast, no significant differences in Cyp11a1 mRNA levels were observed between the control and PRcre:Gata4/6f/f mice 36, 48, and 72 h after hCG. However, a significant decrease in Cyp11a1 mRNA levels was observed 96 h after hCG treatment in the PRcre:Gata4/6f/f mice when compared with the controls.
As observed for Gata6 and Star mRNA levels, Hsd3b1 mRNA levels significantly increased from 36 to 48 h after hCG in the control animals. This increase did not take place in the PRcre:Gata4/6f/f mice. Consequently, although the mRNA levels of Hsd3b1 were not different at 36 h between the control and PRcre:Gata4/6f/f mice, they were significantly lower in the PRcre:Gata4/6f/f mice when compared with the controls at all later times (Fig. 3B).
The early inhibition of Star suggests that it may be a direct target of GATA factors in vivo; therefore, we performed Western blot analyses to examine whether the STAR protein is also affected. In the PRcre:Gata4/6f/f mice, a 70% reduction in luteal STAR protein content was observed when compared to the controls (Fig. 3D).
Effect of P4 Replacement on Implantation in PRcre:Gata4/6f/f Mice
Thus far, our results indicate that the recombination of Gata4 and Gata6 floxed genes during luteinization results in the inhibition of luteal P4 synthesis. To determine whether the lack of luteal P4 production is the primary cause of infertility, PRcre:Gata4/6f/f female mice at d90–d100 were bred with males of proven fertility until copulation was confirmed by the presence of vaginal plugs. Starting the day after vaginal plugs were found, the females were injected daily with 3 mg of P4 or sesame oil for 13 days. On Day 14, implantation was assessed. The PRcre:Gata4/6f/f mice injected with sesame oil alone showed no indication of implantation (n = 3). In contrast, in the PRcre:Gata4/6f/f mice injected with P4, implantation sites were observed in three out of five mice (data not shown).
Role of GATA4 and GATA6 in the Expression of Luteotropic Hormone Receptors
It is well known that hCG strongly stimulates luteal P4 synthesis [22]. However, in the PRcre:Gata4/6f/f mice, hCG was unable to stimulate serum P4 levels or the expression of Star and Hsd3b1. These results suggest that diminished expression of GATA4 and GATA6 may affect the response of luteal cells to LH. To test this possibility, the expression of the receptor for LH (LHR) at different times after the administration of hCG was examined. As shown in Figure 4, a significant decrease in Lhr mRNA levels was observed in the PRcre:Gata4/6f/f mice when compared to the control animals at 48, 72, and 96 h after hCG. Moreover, in the control mice, Lhr mRNA levels increased progressively from 36 to 48 h after hCG treatment and remained high until 96 h after treatment. In contrast, in the PRcre:Gata4/6f/f mice, a progressive decrease in Lhr expression from 36 to 96 h was observed (Fig. 4).
FIG. 4.
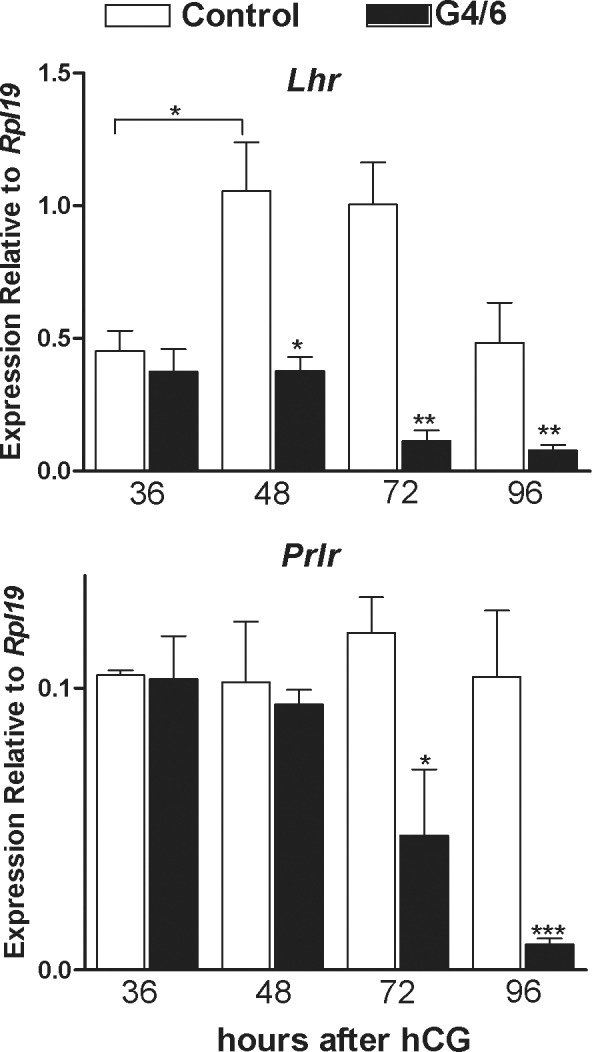
Role of GATA4/6 knockdown on luteotropic receptor expression. Control (C) or PRcre:Gata4/6f/f (G4/6) mice were treated with 7.5 IU of eCG for 48 h followed by treatment with 7.5 IU of hCG for 36, 48, 72, or 96 h. Total RNA was isolated from the corpus luteum of the C or G4/6 female mice treated with hCG for 96 h. Quantification of gene expression of Lhr and Prlr was performed using quantitative PCR (*P < 0.05, **P < 0.01, ***P < 0.001 vs. C, t-test, n = 8).
Because prolactin is essential for normal luteal function in rodents [22], prolactin receptor (Prlr) mRNA levels in the control and PRcre:Gata4/6f/f mice were examined. In contrast to Lhr, the mRNA levels of Prlr were not affected 36 and 48 h after hCG. However, a significant decrease in the steady-state levels of Prlr mRNA was observed 72 and 96 h after hCG (Fig. 4).
GATA4 and GATA6 Contribution to Luteal Steroidogenesis and Lhr Expression
Next, we determined the individual contribution of GATA4 and GATA6 to the regulation of Star, Cyp11a1, Hsd3b1, and Lhr mRNA levels. For this purpose, the control, PRcre:Gata4f/f, and PRcre:Gata6f/f females were treated with eCG for 48 h, followed by treatment with hCG. Animals were killed 96 h after hCG administration. As shown in Figure 5, Lhr, Star, and Hsd3b1 mRNA levels were significantly decreased in all the experimental animals when compared to the controls. In contrast, the mRNA of Cyp11a1 was significantly decreased only in the PRcre:Gata4f/f mice. Cyp11a1 mRNA levels tended to be lower in the PRcre:Gata6f/f mice; however, this decrease did not reach statistical significance. Additionally, serum P4 levels were significantly decreased in the PRcre:Gata4f/f and PRcre:Gata6 f/f mice when compared to the control mice.
FIG. 5.
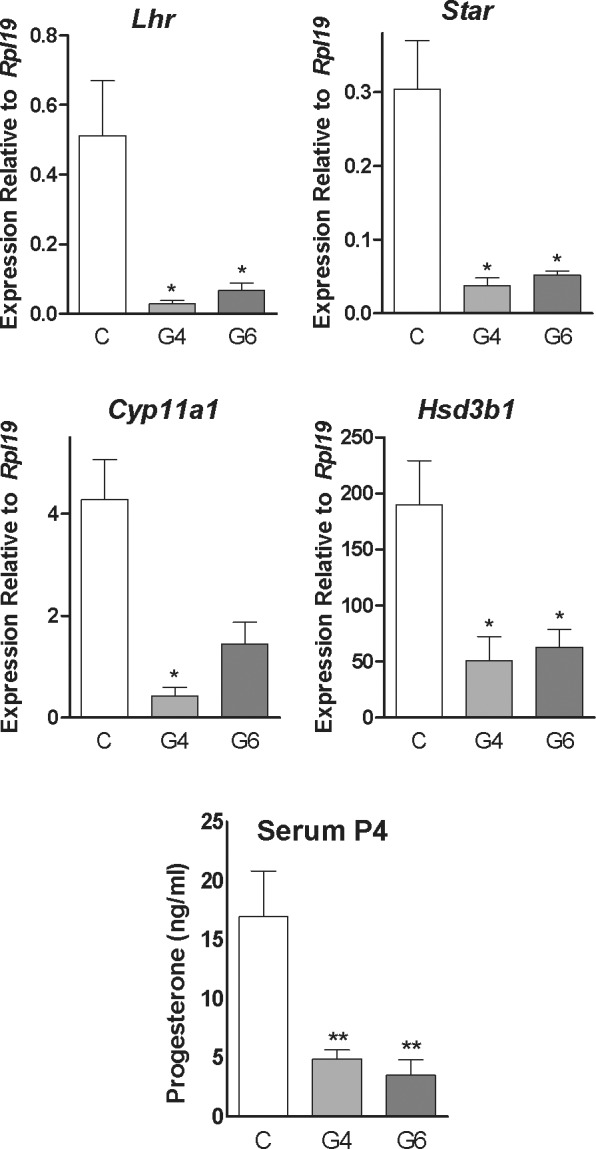
Contribution of GATA4 and GATA6 to luteal steroidogenesis and Lhr expression. Control (C), PRcre:Gata4f/f (G4), or PRcre:Gata6f/f (G6) mice were treated with 7.5 IU of eCG for 48 h followed by treatment with 7.5 IU of hCG. A) Total RNA was isolated from the corpus luteum of the C, G4, or G6 female mice treated with hCG for 96 h. Quantification of gene expression of Lhr, Star, Cyp11a1, and Hsd3b1 was performed using quantitative PCR (*P < 0.05 vs. C, n = 8, t-test). B) Serum P4 levels were measured in the C, G4, and G6 female mice treated with hCG for 96 h (**P < 0.01 vs. C, t-test, n = 4).
Direct and Indirect Effects of GATA on Luteal Steroidogenesis
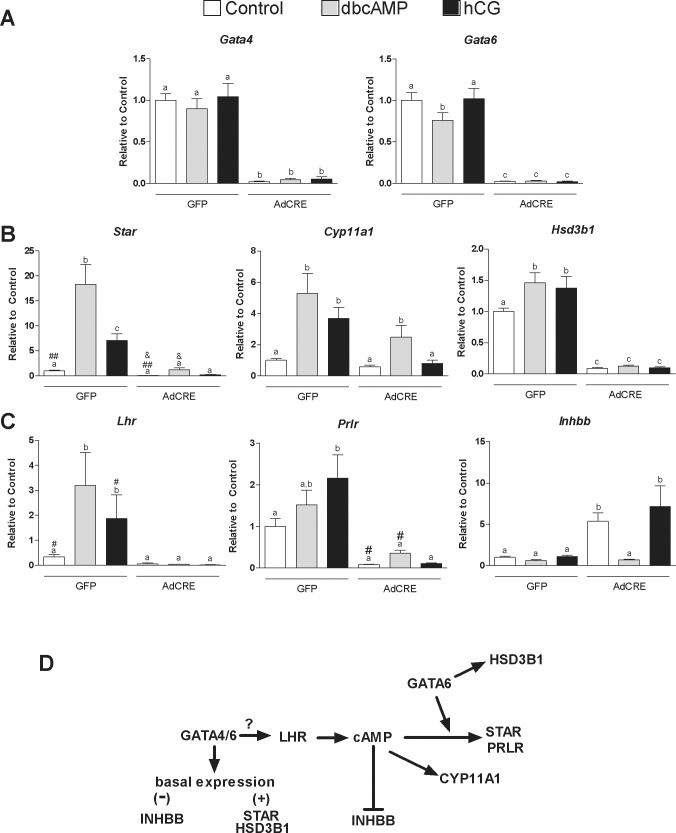
Our findings reveal a decrease in the steady-state levels of the mRNAs for Star, Cyp11a1, Hsd3b1, Prlr, and Lhr in mice with reduced expression of GATA4 and GATA6 in the corpus luteum. However, because LH stimulates Star, Cyp11a1, and Hsd3b1 expression [23], the downregulation of these genes could be due to a reduction in Lhr expression. To explore this possibility, we examined whether treatment with an analog of cAMP, the main second messenger of the LHR, restores Star, Cyp11a1, and Hsd3b1 mRNA levels in GATA4/6-deficient cells. For this purpose, the Gata4/6f/f immature female mice were treated with eCG for 48 h followed by hCG. Luteal cells were isolated 7 h after hCG administration at a time when luteinization had been initiated [24]. To ensure full recombination of the floxed genes in vitro, the cells were infected with AdCRE. As controls, cells were infected with a virus-expressing GFP. The overexpression of Cre reduced Gata4 and Gata6 mRNA to almost undetectable levels, whereas in cells infected with the control virus, Gata4 and Gata6 expression was not affected (Fig. 6A).
FIG. 6.
Role of LHR on the regulation of the steady-state levels of mRNA by GATA factors. A, B, and C) Control or Gata4/6f/f mice were treated with 7.5 IU of eCG for 48 h followed by treatment with 7.5 IU of hCG for 7 h to induce luteinization. Luteal cells were cultured for 48 h with a Cre recombinase expression adenovirus (AdCRE) to induce recombination or a GFP expression vector as a control at a multiplicity of infection of 10. Cells were then treated for 48 h with vehicle, dibutyryl cAMP (dbcAMP) (1 mM), or hCG (50 ng/ml). Quantification of the expression of Gata4, Gata6, Star, Cyp11a1, Hsd3b1, Lhr, Prlr, and Inhbb was performed using quantitative PCR. Columns represent the mean ± SEM of four different samples. Columns with different letters differ significantly (P < 0.05; one-way ANOVA, Tukey test, n = 3). Paired columns labeled with #, ##, or & differ significantly from each other by t-test (P < 0.05, n = 3). D) General scheme indicating genes regulated directly by GATA factors or indirectly via the inhibition of LHR expression. The question mark (?) denotes possible direct or indirect effects of GATA4/6 on LHR expression.
Next, we assessed the response of control and GATA4/6-deficient luteal cells to hCG or dbcAMP, an analog of cAMP. In the control cells, dbcAMP and hCG had no effect on Gata4 or Gata6 mRNA levels. However, as expected, hCG and dbcAMP treatment increased the steady-state levels of Star, Cyp11a1, and Hsd3b1 mRNA levels (Fig. 6B).
In untreated GATA4 and GATA6-deficient cells, the mRNA levels of Star and Hsd3b1 were significantly reduced when compared to the control cells. Moreover, Star, Cyp11a1, and Hsd3b1 mRNA levels were not stimulated by hCG in the GATA4/6-deficient cells. In marked contrast, in cells lacking GATA factors, treatment with dbcAMP caused a 16-fold increase in Star mRNA levels, which is similar in magnitude to the 18-fold increase induced by dbcAMP in control cells. However, dbcAMP-stimulated Star mRNA levels in the control cells were 18 times higher than in GATA4/6-deficient cells. Similarly, dbcAMP stimulated Cyp11a1 to nearly the same extent in the control cells (5.2-fold) and the GATA4/6-deficient cells (4.25-fold), although in the absence of GATA4 and GATA6, Cyp11a1 mRNA levels were approximately 50% lower when compared to the control cells. Treatment with dbcAMP had no effect on Hsd3b1 mRNA expression in the GATA4/6-deficient cells.
In the control cells, dbcAMP and hCG significantly increased Lhr mRNA levels, whereas Prlr mRNA levels showed only a tendency to increase in dbcAMP-treated cells, although Prlr was significantly stimulated by hCG (Fig. 6C). In GATA4/6-deficient cells, hCG was unable to increase Lhr or Prlr mRNA expression, whereas dbcAMP increased Prlr mRNA but had no effect on Lhr mRNA levels. The increase in Prlr mRNA levels by dbcAMP was significant by t-test when compared to the controls.
Finally, we examined the mRNA levels of inhibin βA (Inhba) and inhibin βB (Inhbb), the expression of which decreases during corpus luteum formation [25, 26]. No changes in the steady-state mRNA levels of Inhba were observed regardless of cell genotype or treatment (data not shown). On the other hand, in untreated GATA4/6-deficient cells, the mRNA levels of Inhbb were significantly higher when compared to the control cells (Fig. 6C). Interestingly, Inhbb expression decreased in GATA4/6-deficient cells treated with dbcAMP but not in cells treated with hCG, suggesting that GATA4 and GATA6 are not required for the downregulation of Inhbb induced by cAMP.
DISCUSSION
This report demonstrates that GATA4 and GATA6 are crucial for normal luteal function and consequently for female fertility. Previously, we reported that GATA4 and GATA6 are required for follicle growth beyond the early antral stage [19, 20]. Here, the Gata4 and Gata6 genes were targeted during the luteinization process after follicle maturation. In the PRcre:Gata4/6f/f mice, ovulation was not impaired, indicating normal follicular function; however, these mice were infertile. Our results show that in experimental animals, luteal steroidogenesis is significantly reduced within 48 h of the onset of luteinization and implantation could be rescued by treatment with P4. These findings suggest that diminished luteal P4 synthesis in PRcre:Gata4/6 f/f mice is, at least in part, responsible for the infertility observed in these animals.
The transient expression of the progesterone receptor is tightly coupled with the preovulatory LH surge and localizes exclusively to the granulosa cells, but it is absent from the theca cells of ovulating follicles [27]. It is therefore expected that in our experimental approach, the knockdown of GATA4 and GATA6 takes place only in granulosa cells. As theca cells form part of the corpus luteum in mice, the decrease in serum P4 and the infertility of the PRcre:Gata4/6f/f mice suggest that the luteal cells derived from the granulosa cell layer are the main contributors to circulating P4. However, because luteinizing granulosa cells may produce factors necessary for the differentiation of theca cells, we cannot rule out defects in theca cell luteinization in PRcre:Gata4/6f/f mice.
We demonstrated that PRcre:Gata4/6f/f mice are infertile; however, in the PRcre:Gata4f/f and PRcre:Gata6f/f animals, only a significant decrease in the number of pups per litter was observed. This finding suggests that GATA4 and GATA6 partially compensate for each other. In follicles, the lack of GATA6 can be fully compensated for by GATA4 [19, 20]. In sharp contrast, no full compensation takes place in the corpus luteum, as indicated by the subfertility of the PRcre:Gata6f/f mice. A previous in vitro study suggested a more prominent role of GATA6 on luteal cell function in pigs [18]. This notion is further supported by the increase in the Gata6 mRNA levels that we observed in the control mice after hCG treatment, which was accompanied by a simultaneous decrease in GATA4. Moreover, previous reports demonstrated that during the postovulatory luteinization period, GATA4 levels decline and remain low in the corpus luteum [1–3, 6, 28], whereas GATA6 expression is fairly strong during the postovulatory period [2, 3, 28]. In good agreement with this expression pattern, our in vivo time-course experiment indicates that the decrease in Star, Hsd3b1, and Lhr expression and in serum P4 levels correlates more closely with the reduction of Gata6 expression than with Gata4 mRNA levels. However, findings from the PRcre:Gata4f/f and PRcre:Gata6f/f mice indicate that both factors are equally required for the luteal expression of Star, Hsd3b1, Cyp11a1, and Lhr and for maintaining serum P4 levels. Therefore, although GATA6 seems to be more important, the GATA6-targeted genes required to maintain luteal cell function remain to be revealed.
Recent reports have demonstrated an essential role of GATA4 in the regulation of steroidogenesis in testicular cells. For instance, Schrade et al. [29] demonstrated that siRNA-mediated silencing of GATA4 in a murine Leydig tumor cell line downregulates the genes needed for androgen biosynthesis, including Star, Cyp11a1, Hsd3b1, and Cyp17a1. Similar findings were found with GATA4 knockdown in primary cultures of Leydig cells using the Cre/lox system. In good agreement with our findings, that report also showed a decrease in the expression of LHR in the presence of siRNA against GATA4. Interestingly, GATA4 silencing also impairs glycolysis in Leydig cells [29], and it remains possible that this pathway is targeted in luteal cells. In addition, Bergeron et al. [30] recently reported that GATA4 knockdown in mouse MA-10 Leydig tumor cells was associated with reduced expression of Star, Cyp11a1, and Hsd3b1. These previous in vitro findings and our current in vivo and in vitro results support the notion that GATA4 is a key transcriptional regulator of steroidogenesis in testicular and ovarian cells.
Our in vivo and in vitro findings, however, revealed that Star and Hsd3b1 are highly sensitive to GATA6 depletion. For instance, in vivo time-course experiments demonstrated that Star and Hsd3b1 mRNAs were significantly decreased as early as 48 h after hCG treatment, at the time when a significant decrease in Gata6 expression was observed. Strikingly, we observed a 50% reduction in Star mRNA levels 36 h after hCG, at the time when Gata6 was only slightly lower than in the controls. Moreover, in vitro, the basal expression of Star and Hsd3b1 was reduced to almost undetectable levels in cells that expressed minimal levels of GATA. In contrast, the mRNA levels of Cyp11a1 were significantly lower compared to the controls only 96 h after hCG treatment, at the time when the expression of GATA4 and GATA6 becomes significantly lower in conditional knockout animals. Taken together, these findings suggest that GATA6 is essential for the normal expression of Star and Hsd3b1 in the corpus luteum.
Our findings also suggest that the mechanisms involved in the regulation of luteal Star, Cyp11a1, and Hsd3b1 expression may be different for each gene. For instance, the Star and Hsd3b1 genes are directly regulated by GATA6, whereas both GATA4 and GATA6 seem to indirectly regulate Cyp11a1. In fact, a novel finding of this report is an early block on Lhr expression in luteinizing granulosa cells of PRcre:Gata4/6f/f mice. Therefore, we evaluated to what extent diminished LHR signaling contributes to the block in steroidogenesis. The findings suggested the presence of three groups of genes based on whether or not their stimulation by cAMP requires GATA4/6 or whether GATA factors only enhance the effects of LH on gene expression. Thus, cAMP stimulation of Hsd3b1 and Lhr requires GATA. On the other hand, cAMP is able to stimulate Star and Prlr in the absence of GATA4 and GATA6, but both GATA factors are necessary for the full stimulation of these genes. In contrast, the stimulation of Cyp11a1 by cAMP does not require GATA (Fig. 6D). The findings suggest that in mice the downregulation of Lhr leads to a decrease in Cyp11a1 expression and to a diminished induction of Star and Hsd3b1 by hCG.
LHR is crucial for luteal formation and function [31, 32]. It is well known that after the LH surge, Lhr mRNA and protein levels drop due to a decrease in Lhr mRNA half-life [33–37]. However, this drop lasts for approximately 36 h, and the receptor becomes highly expressed in luteal cells [22, 34, 35, 38]. Our findings demonstrate that in luteal cells expressing low levels of GATA4 and GATA6, Lhr mRNA drops to almost undetectable levels, suggesting that GATA factors play a key role in the upregulation of LHR in the corpus luteum. It remains to be determined whether GATA inhibits the mechanisms involved in Lhr mRNA degradation or directly stimulates the activity of the Lhr promoter in luteal cells (question mark in Fig. 6D).
Our findings also indicate that silencing of the Inhbb gene in luteal cells does not require GATA factors. Inhbb is highly expressed in preovulatory granulosa cells and is downregulated during luteinization by the action of LH [25, 26]. Interestingly, we observed that knockdown of GATA4 and GATA6 increases the basal expression of Inhbb, whereas treatment with cAMP, but not hCG, decreases the elevated levels of Inhbb mRNA found in luteal cells lacking GATA4 and GATA6. These results suggest that GATA stimulation of LHR is required for the downregulation of Inhbb that takes place during luteinization. Of great interest, Bergeron et al. [30] recently demonstrated that Inha and Inhba are decreased by the knockdown of GATA4 in mouse MA-10 Leydig tumor cells, whereas we found no effect of GATA4/6 knockdown on Inhba. These findings suggest that the effect of GATA on the inhibin system are cell specific.
The main function of the corpus luteum is to synthesize high levels of P4, which is critical for the maintenance of pregnancy. To evaluate whether the loss in P4 synthesis is the primary cause of infertility in PRcre:Gata4/6f/f mice, we attempted to rescue pregnancy in these animals by administrating P4. Progesterone replacement rescued implantation in PRcre:Gata4/6 f/f females, although it was not effective in every animal. However, none of the PRcre:Gata4/6f/f animals that received sesame oil showed signs of implantation. These findings suggest that luteal dysfunction is one of the main causes of infertility in PRcre:Gata4/6f/f mice. Nevertheless, we cannot discard defects in the oviducts and/or uterus. In fact, despite the fact that the knockdown of either GATA4 or GATA6 equally affects steroidogenesis, only PRcre:Gata4/6f/f mice are infertile. Therefore, the ultimate cause of infertility remains elusive, and further experiments are needed to determine whether GATA4/6 regulate oviductal and uterine function.
In summary, our findings demonstrate that GATA4 and GATA6 are essential for luteal P4 synthesis in mice. The knockdown of GATA4 and GATA6 severely reduces the response of luteal cells to gonadotropins. Although the mechanisms by which GATA4 and GATA6 control Lhr and Prlr expression remain to be elucidated, our findings provide novel in vivo evidence indicating that GATA factors are crucial regulators of luteal steroidogenesis and gonadotropin receptor expression.
ACKNOWLEDGMENT
We thank Dr. William Pu (Department of Cardiology, Children's Hospital Boston, Boston, MA) for providing the Gata4 floxed mice.
Footnotes
Current Address: Division of Reproductive & Developmental Sciences, Oregon National Primate Research Center, Oregon Health & Science University, Beaverton, OR 97006.
This work was supported by NIH grant numbers R01HD057110 and R21HD066233 (C.S.); S.M.C. and S.C.B. were also supported by NIH training grant number T32HL07692.
These authors contributed equally to this study.
REFERENCES
- Lavoie HA, McCoy GL, Blake CA. Expression of the GATA-4 and GATA-6 transcription factors in the fetal rat gonad and in the ovary during postnatal development and pregnancy Mol Cell Endocrinol 2004. 227 31– 40 [DOI] [PubMed] [Google Scholar]
- Gillio-Meina C, Hui YY, LaVoie HA. GATA-4 and GATA-6 transcription factors: expression, immunohistochemical localization, and possible function in the porcine ovary Biol Reprod 2003. 68 412– 422 [DOI] [PubMed] [Google Scholar]
- Heikinheimo M, Ermolaeva M, Bielinska M, Rahman NA, Narita N, Huhtaniemi IT, Tapanainen JS, Wilson DB. Expression and hormonal regulation of transcription factors GATA-4 and GATA-6 in the mouse ovary Endocrinology 1997. 138 3505– 3514 [DOI] [PubMed] [Google Scholar]
- Taniguchi H, Komiyama J, Viger RS, Okuda K. The expression of the nuclear receptors NR5A1 and NR5A2 and transcription factor GATA6 correlates with steroidogenic gene expression in the bovine corpus luteum Mol Reprod Dev 2009. 76 873– 880 [DOI] [PubMed] [Google Scholar]
- Laitinen MP, Anttonen M, Ketola I, Wilson DB, Ritvos O, Butzow R, Heikinheimo M. Transcription factors GATA-4 and GATA-6 and a GATA family cofactor, FOG-2, are expressed in human ovary and sex cord-derived ovarian tumors J Clin Endocrinol Metab 2000. 85 3476– 3483 [DOI] [PubMed] [Google Scholar]
- Silverman E, Eimerl S, Orly J. CCAAT enhancer-binding protein beta and GATA-4 binding regions within the promoter of the steroidogenic acute regulatory protein (StAR) gene are required for transcription in rat ovarian cells J Biol Chem 1999. 274 17987– 17996 [DOI] [PubMed] [Google Scholar]
- Tremblay JJ, Viger RS. Novel roles for GATA transcription factors in the regulation of steroidogenesis J Steroid Biochem Mol Biol 2003. 85 291– 298 [DOI] [PubMed] [Google Scholar]
- Tremblay JJ, Viger RS. Transcription factor GATA-4 is activated by phosphorylation of serine 261 via the cAMP/protein kinase a signaling pathway in gonadal cells J Biol Chem 2003. 278 22128– 22135 [DOI] [PubMed] [Google Scholar]
- Tremblay JJ, Viger RS. GATA factors differentially activate multiple gonadal promoters through conserved GATA regulatory elements Endocrinology 2001. 142 977– 986 [DOI] [PubMed] [Google Scholar]
- Sher N, Yivgi-Ohana N, Orly J. Transcriptional regulation of the cholesterol side chain cleavage cytochrome P450 gene (CYP11A1) revisited: binding of GATA, cyclic adenosine 3′,5′-monophosphate response element-binding protein and activating protein (AP)-1 proteins to a distal novel cluster of cis-regulatory elements potentiates AP-2 and steroidogenic factor-1-dependent gene expression in the rodent placenta and ovary Mol Endocrinol 2007. 21 948– 962 [DOI] [PubMed] [Google Scholar]
- Peng L, Huang Y, Jin F, Jiang SW, Payne AH. Transcription enhancer factor-5 and a GATA-like protein determine placental-specific expression of the type I human 3beta-hydroxysteroid dehydrogenase gene, HSD3B1 Mol Endocrinol 2004. 18 2049– 2060 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stocco C, Kwintkiewicz J, Cai Z. Identification of regulatory elements in the Cyp19 proximal promoter in rat luteal cells J Mol Endocrinol 2007. 39 211– 221 [DOI] [PubMed] [Google Scholar]
- Kwintkiewicz J, Cai Z, Stocco C. Follicle-stimulating hormone-induced activation of Gata4 contributes in the up-regulation of Cyp19 expression in rat granulosa cells Mol Endocrinol 2007. 21 933– 947 [DOI] [PubMed] [Google Scholar]
- Cai Z, Kwintkiewicz J, Young ME, Stocco C. Prostaglandin E2 increases cyp19 expression in rat granulosa cells: implication of GATA-4 Mol Cell Endocrinol 2007. 263 181– 189 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anttonen M, Parviainen H, Kyronlahti A, Bielinska M, Wilson DB, Ritvos O, Heikinheimo M. GATA-4 is a granulosa cell factor employed in inhibin-{alpha} activation by the TGF-{beta} pathway J Mol Endocrinol 2006. 36 557– 568 [DOI] [PubMed] [Google Scholar]
- Robert NM, Miyamoto Y, Taniguchi H, Viger RS. LRH-1/NR5A2 cooperates with GATA factors to regulate inhibin alpha-subunit promoter activity Mol Cell Endocrinol 2006. 257-258 65– 74 [DOI] [PubMed] [Google Scholar]
- Viger RS, Guittot SM, Anttonen M, Wilson DB, Heikinheimo M. Role of the GATA family of transcription factors in endocrine development, function, and disease Mol Endocrinol 2008. 22 781– 798 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hui YY, Lavoie HA. GATA4 reduction enhances 3′,5′-cyclic adenosine 5′-monophosphate-stimulated steroidogenic acute regulatory protein messenger ribonucleic acid and progesterone production in luteinized porcine granulosa cells Endocrinology 2008. 149 5557– 5567 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bennett J, Baumgarten SC, Stocco C. GATA4 and GATA6 silencing in ovarian granulosa cells affects levels of mRNAs involved in steroidogenesis, extracellular structure organization, IGF-I activity, and apoptosis Endocrinology 2013. 154 4845– 4858 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bennett J, Wu YG, Gossen J, Zhou P, Stocco C. Loss of GATA-6 and GATA-4 in granulosa cells blocks folliculogenesis, ovulation, and follicle stimulating hormone receptor expression leading to female infertility Endocrinology 2012. 153 2474– 2485 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soyal SM, Mukherjee A, Lee KY, Li J, Li H, DeMayo FJ, Lydon JP. Cre-mediated recombination in cell lineages that express the progesterone receptor Genesis 2005. 41 58– 66 [DOI] [PubMed] [Google Scholar]
- Stocco C, Telleria C, Gibori G. The molecular control of corpus luteum formation, function, and regression Endocr Rev 2007. 28 117– 149 [DOI] [PubMed] [Google Scholar]
- Sandhoff TW, Hales DB, Hales KH, McLean MP. Transcriptional regulation of the rat steroidogenic acute regulatory protein gene by steroidogenic factor 1 Endocrinology 1998. 139 4820– 4831 [DOI] [PubMed] [Google Scholar]
- Richards JS. Hormonal control of gene expression in the ovary Endocr Rev 1994. 15 725– 751 [DOI] [PubMed] [Google Scholar]
- Makanji Y, Zhu J, Mishra R, Holmquist C, Wong WP, Schwartz NB, Mayo KE, Woodruff TK. Inhibin at 90: from discovery to clinical application, a historical review Endocr Rev 2014. 35 747– 794 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knight PG, Satchell L, Glister C. Intra-ovarian roles of activins and inhibins Mol Cell Endocrinol 2012. 359 53– 65 [DOI] [PubMed] [Google Scholar]
- Park OK, Mayo KE. Transient expression of progesterone receptor messenger RNA in ovarian granulosa cells after the preovulatory luteinizing hormone surge Mol Endocrinol 1991. 5 967– 978 [DOI] [PubMed] [Google Scholar]
- Anttonen M, Ketola I, Parviainen H, Pusa AK, Heikinheimo M. FOG-2 and GATA-4 are coexpressed in the mouse ovary and can modulate Müllerian-inhibiting substance expression Biol Reprod 2003. 68 1333– 1340 [DOI] [PubMed] [Google Scholar]
- Schrade A, Kyronlahti A, Akinrinade O, Pihlajoki M, Hakkinen M, Fischer S, Alastalo TP, Velagapudi V, Toppari J, Wilson DB, Heikinheimo M. GATA4 is a key regulator of steroidogenesis and glycolysis in mouse Leydig cells Endocrinology 2015. 156 1860– 1872 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bergeron F, Nadeau G, Viger RS. GATA4 knockdown in MA-10 Leydig cells identifies multiple target genes in the steroidogenic pathway Reproduction 2015. 149 245– 257 [DOI] [PubMed] [Google Scholar]
- Stouffer RL. Progesterone as a mediator of gonadotrophin action in the corpus luteum: beyond steroidogenesis Hum Reprod Update 2003. 9 99– 117 [DOI] [PubMed] [Google Scholar]
- Duncan WC, McNeilly AS, Fraser HM, Illingworth PJ. Luteinizing hormone receptor in the human corpus luteum: lack of down-regulation during maternal recognition of pregnancy Hum Reprod 1996. 11 2291– 2297 [DOI] [PubMed] [Google Scholar]
- LaPolt PS, Oikawa M, Jia XC, Dargan C, Hsueh AJ. Gonadotropin-induced up- and down-regulation of rat ovarian LH receptor message levels during follicular growth, ovulation and luteinization Endocrinology 1990. 126 3277– 3279 [DOI] [PubMed] [Google Scholar]
- Segaloff DL, Wang HY, Richards JS. Hormonal regulation of luteinizing hormone/chorionic gonadotropin receptor mRNA in rat ovarian cells during follicular development and luteinization Mol Endocrinol 1990. 4 1856– 1865 [DOI] [PubMed] [Google Scholar]
- Peegel H, Randolph J, Jr, Midgley AR, Menon KM. In situ hybridization of luteinizing hormone/human chorionic gonadotropin receptor messenger ribonucleic acid during hormone-induced down-regulation and the subsequent recovery in rat corpus luteum Endocrinology 1994. 135 1044– 1051 [DOI] [PubMed] [Google Scholar]
- Lu DL, Peegel H, Mosier SM, Menon KM. Loss of lutropin/human choriogonadotropin receptor messenger ribonucleic acid during ligand-induced down-regulation occurs post transcriptionally Endocrinology 1993. 132 235– 240 [DOI] [PubMed] [Google Scholar]
- Menon B, Sinden J, Franzo-Romain M, Botta RB, Menon KM. Regulation of LH receptor mRNA binding protein by miR-122 in rat ovaries Endocrinology 2013. 154 4826– 4834 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu F, Stouffer RL, Muller J, Hennebold JD, Wright JW, Bahar A, Leder G, Peters M, Thorne M, Sims M, Wintermantel T, Lindenthal B. Dynamics of the transcriptome in the primate ovulatory follicle Mol Hum Reprod 2011. 17 152– 165 [DOI] [PMC free article] [PubMed] [Google Scholar]