Abstract
The primary cilium of renal epithelia acts as a transducer of extracellular stimuli. Polycystin (PC)1 is the protein encoded by the PKD1 gene that is responsible for the most common and severe form of autosomal dominant polycystic kidney disease (ADPKD). PC1 forms a complex with PC2 via their respective carboxy-terminal tails. Both proteins are expressed in the primary cilia. Mutations in either gene affect the normal architecture of renal tubules, giving rise to ADPKD. PC1 has been proposed as a receptor that modulates calcium signals via the PC2 channel protein. The effect of PC1 dosage has been described as the rate-limiting modulator of cystic disease. Reduced levels of PC1 or disruption of the balance in PC1/PC2 level can lead to the clinical features of ADPKD, without complete inactivation. Recent data show that ADPKD resulting from inactivation of polycystins can be markedly slowed if structurally intact cilia are also disrupted at the same time. Despite the fact that no single model or mechanism from these has been able to describe exclusively the pathogenesis of cystic kidney disease, these findings suggest the existence of a novel cilia-dependent, cyst-promoting pathway that is normally repressed by polycystin function. The results enable us to rethink our current understanding of genetics and cilia signaling pathways of ADPKD.
Keywords: Autosomal dominant polycystic kidney disease, Calcium, Cilia, Polycystin-1, Polycystin-2
Introduction
It has been reported that abnormalities in the structure or function of primary cilia result in kidney cyst growth in animal models and human genetic diseases collectively known as ciliopathies [1], [2], [3]. Polycystin (PC)1 has been hypothesized to form a mechanosensitive cation channel complex with PC2 in the primary cilia [4], [5], [6]. Functional defects in this complex caused by mutation of PKD1 or PKD2 result in autosomal dominant polycystic kidney disease (ADPKD) [7], [8]. Primary cilia are membrane-enclosed hair-like projections from the apical surface of renal epithelial cells, facing into the tubule lumen (Fig. 1). Primary cilia are microtubule-based organelles that are ideally positioned to detect extracellular stimuli and to transduce these signals into the cell to elicit physiological responses [6], [9]. Experimental evidence that flow-mediated deflection of the cilia induces an increase in intracellular calcium has fostered the hypothesis that cilia may sense flow in the kidney tubule lumens [10], [11], [12]. Intraflagellar transport (IFT) is a general ciliary component transport mechanism required for assembly and steady-state maintenance of cilia [13], [14]. IFT plays an additional role in regulating cell-cycle progression independent of its function in cilia formation [1]. Despite these data, there remains some lack of clarity on the relationship between PCs and cilia function. Recent studies have generated novel information regarding the genetic and molecular implications of ADPKD, its pathogenesis, and new potential strategies for targeted treatment. In this commentary, new signaling activity of interconnectedness between primary cilia and PCs, and dosage effects of PC are highlighted.
Figure 1.
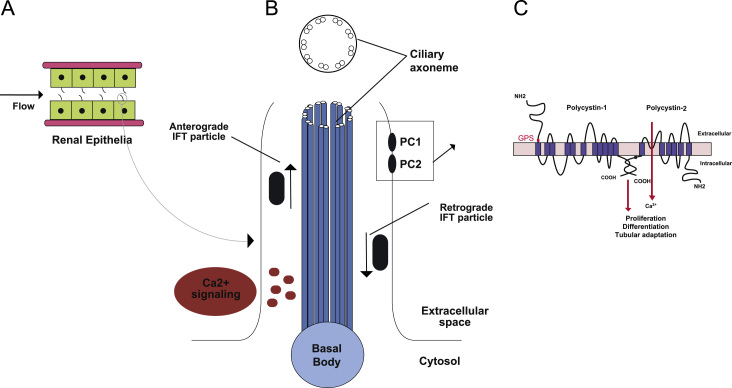
PCs and cilia. (A) Renal tubular epithelium depicted as cuboidal cells with apical primary cilia projections subjected to luminal flow. (B) Schematic of 9 + 0 primary cilia. As a microtubule-based structure that imports proteins via anterograde IFT and returns proteins to the cell body via retrograde IFT transport. PC1 and PC2 reside in the cilium membrane and are hypothesized to subsume a local calcium signaling process that may be modulated by mechanical or ligand stimuli. (C) Schematic representations of PC1 and PC2 showing their respective topologies and interaction via coiled coil domains in their carboxy termini. PC1 has the properties of a receptor and undergoes cleavage at the indicated (red letters) GPS sites.
GPS site, whereas PC2 is a calcium channel of the TRP family. GPS, G protein coupled receptor proteolytic site; IFT, intraflagellar transport; PC, polycystin; TRP, transient receptor potential. (For interpretation of the references to color in this figure legend, the reader is referred to the web version of this article.)
ADPKD
ADPKD is a common, single-gene multi-system disorder. Its prevalence at birth is between 1:400 and 1:1,000 and it affects >12 million people worldwide, without regard to sex or ethnicity [5], [7], [8], [15]. The disease is characterized by the development of renal cysts and various extrarenal manifestations. ADPKD results from excessive proliferation of renal tubular epithelial cells and remodeling of surrounding structures, giving rise to growth of epithelial-lined cysts accompanied by fibrosis and accumulation of extracellular matrix. As the disease progresses, this leads to destruction of the normal renal parenchyma, massive renal enlargement, deterioration of renal function, and eventually renal failure in >50% of affected individuals by late adulthood [5], [15]. A two-hit model has been proposed to explain the focal nature of renal cysts and the variability in cyst size in both orthologous mouse models and in humans [16], [17], [18]. In this model, a germline and a somatic mutation inactivate the PKD alleles separately. The first hit, a germline mutation inherited from the affected parent, is predisposing but not sufficient for cyst formation. The first hit exists in all cells in the body. The second hit, a somatic mutation in an individual cell, inactivates the normal PKD1 or PKD2 allele, and when it occurs in kidney tubule cells, is thought to cause abnormal focal proliferation and tissue remodeling, giving rise to cyst formation [19], [20]. Conditional knockout of Pkd1 in mice indicates that the timing of the somatic second hit mutation affects the severity of cyst progression. Inactivation of Pkd1 in mice prior to a developmental switch occurring prior to postnatal Day 13 results in severely cystic kidneys within 3 weeks, whereas inactivation at Day 14 and later results in cysts only after 5 months [21]. Multiple genetic mechanisms that result in an imbalance in the expression of either PC1 or PC2 below a critical threshold without complete loss can also cause cyst formation [22], [23]. The presence of somatic PKD2 mutations detected in cystic patients with PKD1 germline mutations and increased disease severity in patients with heterozygous germline mutation in both PKD1 and PKD2 offer further support for the two-hit model and the dosage effect hypothesis for cyst formation [24], [25].
ADPKD genes and proteins – PKD1, PKD2, and PC1 and PC2
PKD1 mutations account for ~85% of the clinically ascertained disease burden. The PKD1 gene product, PC1, is a large protein consisting of 4,302 amino acids with a large extracellular amino terminus, 11 transmembrane domains, and a short cytosolic carboxy terminus (Fig. 1C) [5], [15]. PC1 is possibly expressed in most nephron segments, despite expression at the tissue level being difficult to determine due to its low protein levels. At the subcellular level, it has been reported that PCs are expressed in several subcellular compartments of renal epithelial cells, remarkably primary cilia, lateral cell to cell junctions, and the endoplasmic reticulum. Investigations in other experiments have demonstrated that the localization of PC fractions to primary cilia is thought to be required for the function of cilia as mechanosensors [2], [3], [6], [20], [26].
PKD2, the gene mutated in 15% of polycystic kidney disease cases, encodes PC2 [5], [27]. PC2 has been described as a nonselective, calcium-permeable cation channel belonging to the transient receptor potential (TRP) polycystic subfamily of TRP channels [28], [29]. PC2 is an integral membrane protein with six transmembrane segments, and both the amino and carboxy termini face the cytoplasmic compartment [30], [31]. PC2 contains several trafficking motifs: the RVxP motif at the amino terminus of PC2 is necessary and sufficient for the ciliary location of the protein [30], whereas a region at the carboxy terminus may restrict the membrane location of PC2 to the endoplasmic reticulum and cilia. In the kidney, PC2 is found in all nephron segments with the exception of the glomeruli. PC1 is thought to function as a mechanosensor or chemosensor at the primary cilia that coordinates the activity of the PC2 calcium channel [15], [32], [33].
Disruption of primary cilia or mutations in cilia-associated proteins leads to renal epithelial proliferation and cyst growth in animal models and many human genetic diseases that are classified as ciliopathies [2], [3], [34], [35]. Primary cilia have an axonemal structure consisting of a circular array of nine pairs of microtubules (9+0 arrangement; Fig. 1B) [3]. Primary cilia are single nonmotile projections on the apical surface of renal epithelial cells toward the lumen [9]. They have a plasma membrane but do not have subcellular organelles and protein-synthetic capacity. Primary cilia, which are usually nonmotile, are thought to serve either a chemosensory or mechanosensory function [4], [6]. The phenotypes of ciliopathies in mammals include cystic kidneys, abnormal formation of the left–right body axis, abnormalities in neural tube closure and patterning, skeletal defects such as polydactyly, liver, and pancreatic cysts and fibrosis, and obesity [34], [35]. In addition to their potential role as mechanosensors, primary cilia have important functions in sensing the external environment and are essential to processes as diverse as photoreception, olfaction, and developmental signaling [35].
Many cellular pathways have been proposed as effector pathways in ciliary and cystic diseases. They include planar cell polarity, Wnt, mammalian target of rapamycin, cyclic adenosine monophosphate (cAMP), G-protein-coupled receptor, cystic fibrosis transmembrane conductance regulator, epidermal growth factor receptor, mitogen-activated protein kinase (MAPK), cellular calcium, and the cell cycle [5]. Experimental evidence supports that cAMP stimulates MAPK/extracellular signal-regulated kinase (ERK) signaling [36], [37], and promotes cyst growth. Functional loss of PC1 at the cilium results in reduced cellular calcium signaling [6]. This in turn results in increased adenylate cyclase and decreased phosphodiesterase activity, leading to increased levels of intracellular cAMP. Increased cAMP in cystic epithelium increases the activity of protein kinase A, which can result in increased cell proliferation and chloride-driven fluid secretion; features associated with cyst growth [5], [36]. Lacking through all of these proposed functional pathway hypotheses is an understanding of the relationship of PCs with other ciliary component proteins and a clear definition of the direct role of PC signaling within cilia.
PC1 dosage in ADPKD
Studies on a possible gene-dosage effect for PCs have been described in trans heterozygous states in mice [16]. Mutations in Pkd1 resulting in reduction of functional PC1 have suggested that reduced activity is sufficient to cause cyst formation in some situations [38], [39], [40]. Hence, PC1 dosage has been described as a modulator of cystic disease. These authors generated an intronic insertion of a neomycin cassette into Pkd1, which had a profound effect on splicing efficiency. Although the hypomorphic Pkd1nl/nl mice survive, they develop severe cystic kidneys from the collecting duct, thick ascending limb, and distal tubules. Another study [41] further supports that diminished expression of native PC1 is sufficient to induce renal cystic lesions of ADPKD. Studies have demonstrated that 50% reduction in PC1 levels, as occurs in true heterozygote ADPKD patients, does not lead to overt cyst formation, but 80% reduction may be sufficient to result in a polycystic phenotype [42]. A recent study demonstrating that the decrease in the steady-state levels of PC1 that can be partially rescued in a temperature-sensitive manner also points to the importance of PC1 dosage in the modulation of cyst burden [43]. Separately, high copy number Pkd1 transgenic mouse lines have reported the presence of kidney cysts [44], suggesting that massively increased dosage of PC1 also leads to cyst formation. Although the mechanism for the latter is unknown, PC1 overexpression may generate cysts by disrupting the steady-state balance of the PC1/PC2 complex, thereby altering its signaling [44].
A comprehensive study of the mechanisms underlying isolated ADPKD has clearly demonstrated the central role of PC dosage in kidney cyst formation [23], [45]. The two genes responsible for isolated polycystic liver disease, PRKCSH and SEC63, are both involved in modulating protein biogenesis and quality control for integral membrane and secreted proteins in the endoplasmic reticulum. Inactivation of either of these genes results in inefficient maturation of the PCs and cyst formation in both the liver and kidney in mouse models. This effect can be exacerbated by reduced gene dosage for PKD1 or PKD2 [23]. Overexpression of Pkd1, but not Pkd2, can overcome the tendency toward cyst formation in Prkcsh and Sec63 mutants, thereby establishing PC1 as the rate-limiting component whose dosage determines both the rate and extent of cyst formation in ADPKD [23], [45]. In a proof of principle experiment, inhibition of proteasomal degradation using the proteasome inhibitor carfilzomib was able to ameliorate polycystic disease progression in a manner analogous to increased PC1 expression [23], [45]. Based on these data, PC1 dosage is one of the effector mechanisms of cyst formation in both mouse and human ADPKD.
Novel inter-relationship between primary cilia and PCs
Defects in primary cilia are associated with increased proliferation of cells characteristic of polycystic kidney disease [34], [46]. IFT machinery is required to traffic component proteins into and out of cilia (Fig. 1B) [3], [13], [47]. Among the many proteins delivered to cilia by IFT are PC1 and PC2, loss of either of which results in ADPKD [4], [48], [49]. Kidney cysts also arise in mice following the disruption of cilia by targeted inactivation of genes encoding IFT components, such as the heterotrimeric kinesin component Kif3a [50] and IFT proteins, Ift20 [47] and Ift88 [11], [51], [52]. It is generally hypothesized that the primary cilium of kidney epithelial cells acts as a sensory organelle and that PC1 and PC2 form a receptor–channel sensory complex in the cilium.
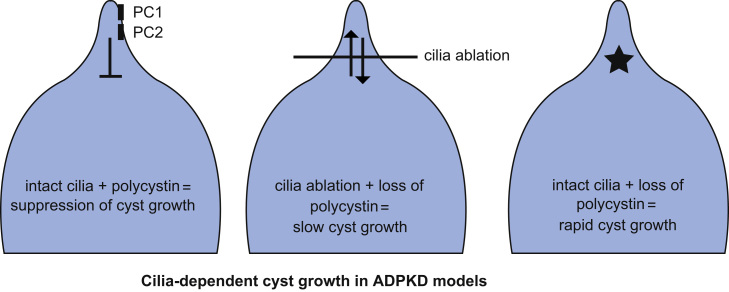
A recent study from our laboratory based on genetic manipulation of polycystins and cilia in mutant mice has the potential to recalibrate significantly the concepts about the inter-relationship between PC and cilia function [53]. Inactivation of PCs or cilia alone can lead to the formation of kidney cysts. However, it has not been clear whether it is the loss of PCs or persistence of intact cilia under conditions of loss of PCs that is responsible for initiation of the signaling involved in cyst growth. We evaluated these possible mechanisms by using tissue-specific and inducible knockout of Pkd1 and Pkd2 alone or in combination with knockout of Kif3a and Ift20 in mouse models. We found that disruption of cilia markedly delayed cyst growth caused by the loss of PCs. Concomitant loss of Pkd1 or Pkd2 along with either Kif3a or Ift20 resulted in much milder progression of polycystic disease than was seen with inactivation of either PC alone (Fig. 2).
Figure 2.
Schematic representation of the cilia dependent cyst promoting pathways in ADPKD. When PCs are normally expressed in cilia, renal tubular architecture is preserved (left panel). When PCs are lost, intact cilia are responsible for signal (depicted by the star) that promotes rapid cyst growth (right panel). When both PCs and cilia on non-functioning (center panel), indolent cyst growth results.
ADPKD, autosomal dominant polycystic kidney disease; PC, polycystin
We further tested whether disease severity was affected by a time-dependent persistence of cilia following PC loss. We found that the shorter the time interval until cilia disappearance following initial loss of PCs, the greater the protection conferred against cyst progression.
Therefore, the severity of ADPKD is directly affected by the duration of the persistence of cilia in the absence of PCs. These findings suggest that PCs provide baseline tonic inhibition of a cilium-dependent signal that promotes cyst growth when unchecked following inactivation of PCs. As part of normal physiology, this inhibitory function of PCs is likely altered based on mechanical flow or ligand-mediated regulation of PC1 that is currently not understood. Neither reduced Pkd1 nor increased Pkd1 gene dosage affects cyst formation following cilia ablation, suggesting albeit not proving, that polycystic disease following cilia inactivation occurs by PC-independent mechanisms [53]. Taken together, the data show that loss of PCs causes kidney cyst formation in a cilium-dependent manner, but loss of cilia causes cyst formation independent of PC function (Fig. 2).
The data suggest that the molecular mechanisms of the cilium-dependent cyst promoting activity following loss of polycystins are likely to be novel. We examined the activities of the MAPK/ERK, mammalian target of rapamycin, and cAMP pathways, all of which have been implicated in cystic mouse models and therapeutic targets in human clinical trials, as candidate effectors for this novel cilium-dependent process. Although there was increased activity of both the MAPK/ERK and cAMP pathways in cystic mice, both activities were primarily confined to distal nephron. These pathways were therefore unable to account for the ubiquitous inhibition of cyst growth observed following cilia ablation in all nephron segments, including proximal tubule. Our data strongly suggest the presence of a new, cilium-dependent cyst-promoting pathway, which is suppressed by functional PCs. Our in vivo studies prove the principle that targeting of the cilium-dependent, PC-inhibited pathway will be effective in slowing the progression of ADPKD. This novel cilium-dependent pathway is now proposed as the central homeostatic mechanism of polycystins and cilium signaling in ADPKD. Defining components of this pathway will lead to improved targets for potential therapies in ADPKD.
Summary
The polycystin proteins are thought to be part of the signaling system of the cilium required to maintain normal renal structural and functional homeostasis. Both of the functioning polycystins and intact cilia seem to be required for tubular adaptation and maintaining the architecture of renal epithelial tubules. The steady state of the physiological roles of PC1 and PC2 regulates this cilium-dependent pathway and elicits responses of tubule adaptation to either chemical or mechanical signals. The cyst-suppressive role of polycystins requires either mechanical or ligand-mediated activation to affect calcium-dependent inhibition of cyst growth [46], [54], [55], but the precise nature of these signals remains unknown. Our findings strongly suggest that the components of cilium-dependent cyst growth signals reside in cilia and require intact cilia for activity. PC1 dosage can regulate cyst progression in ADPKD by having inadequate levels to suppress fully the cilium-dependent cyst-promoting pathway. This would explain the hypothesis that cyst growth lies along a continuum from physiological changes in tubule luminal diameter in response to discrete stimuli, to slow cyst grow in the setting of reduced PC1 function, and to more rapid cyst growth in the setting of completely absent PC1 function. Altering PC1 levels by chemical chaperone therapy may slow down cyst progression in a subset of patients in whom reduced, rather than absent function is the cause of ADPKD. Our understanding of ADPKD has evolved to include reduced PC dosage or PC loss as a cause of ADPKD and have provided genetic evidence of the existence of yet to be discovered novel targets for ADPKD therapy that hold promise for the future wellbeing of these patients with this complex disease.
Conflicts of interest
None.
References
- 1.Zhou J. Polycystins and primary cilia: primers for cell cycle progression. Annu Rev Physiol. 2009;71:83–113. doi: 10.1146/annurev.physiol.70.113006.100621. [DOI] [PubMed] [Google Scholar]
- 2.Hildebrandt F, Benzing T, Katsanis N. Ciliopathies. N Engl J Med. 2011;364:1533–1543. doi: 10.1056/NEJMra1010172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Berbari NF, O׳Connor AK, Haycraft CJ, Yoder BK. The primary cilium as a complex signaling center. Curr Biol. 2009;19:R526–R535. doi: 10.1016/j.cub.2009.05.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Yoder BK, Hou X, Guay-Woodford LM. The polycystic kidney disease proteins, polycystin-1, polycystin-2, polaris, and cystin, are co-localized in renal cilia. J Am Soc Nephrol. 2002;13:2508–2516. doi: 10.1097/01.asn.0000029587.47950.25. [DOI] [PubMed] [Google Scholar]
- 5.Harris PC, Torres VE. Polycystic kidney disease. Annu Rev Med. 2009;60:321–337. doi: 10.1146/annurev.med.60.101707.125712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Nauli SM, Alenghat FJ, Luo Y, Williams E, Vassilev P, Li X, Elia AE, Lu W, Brown EM, Quinn SJ, Ingber DE, Zhou J. Polycystins 1 and 2 mediate mechanosensation in the primary cilium of kidney cells. Nat Genet. 2003;33:129–137. doi: 10.1038/ng1076. [DOI] [PubMed] [Google Scholar]
- 7.Igarashi P, Somlo S. Genetics and pathogenesis of polycystic kidney disease. J Am Soc Nephrol. 2002;13:2384–2398. doi: 10.1097/01.asn.0000028643.17901.42. [DOI] [PubMed] [Google Scholar]
- 8.Igarashi P, Somlo S. Polycystic kidney disease. J Am Soc Nephrol. 2007;18:1371–1373. doi: 10.1681/ASN.2007030299. [DOI] [PubMed] [Google Scholar]
- 9.Satir P, Christensen ST. Structure and function of mammalian cilia. Histochem Cell Biol. 2008;129:687–693. doi: 10.1007/s00418-008-0416-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Low SH, Vasanth S, Larson CH, Mukherjee S, Sharma N, Kinter MT, Kane ME, Obara T, Weimbs T. Polycystin-1, STAT6, and P100 function in a pathway that transduces ciliary mechanosensation and is activated in polycystic kidney disease. Dev Cell. 2006;10:57–69. doi: 10.1016/j.devcel.2005.12.005. [DOI] [PubMed] [Google Scholar]
- 11.Grimm DH, Cai Y, Chauvet V, Rajendran V, Zeltner R, Geng L, Avner ED, Sweeney W, Somlo S, Caplan MJ. Polycystin-1 distribution is modulated by polycystin-2 expression in mammalian cells. J Biol Chem. 2003;278:36786–36793. doi: 10.1074/jbc.M306536200. [DOI] [PubMed] [Google Scholar]
- 12.Patel A: The primary cilium calcium channels and their role in flow sensing. Pflugers Arch - Eur J Physiol.doi:10.1007/s00424-014-1516-0. Epub 2014 Apr 26 [DOI] [PubMed]
- 13.Silverman MA, Leroux MR. Intraflagellar transport and the generation of dynamic, structurally and functionally diverse cilia. Trends Cell Biol. 2009;19:306–316. doi: 10.1016/j.tcb.2009.04.002. [DOI] [PubMed] [Google Scholar]
- 14.Kim S, Dynlacht BD. Assembling a primary cilium. Curr Opin Cell Biol. 2013;25:506–511. doi: 10.1016/j.ceb.2013.04.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gallagher AR, Germino GG, Somlo S. Molecular advances in autosomal dominant polycystic kidney disease. Adv Chronic Kidney Dis. 2010;17:118–130. doi: 10.1053/j.ackd.2010.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wu G, Tian X, Nishimura S, Markowitz GS, D׳Agati V, Park JH, Yao L, Li L, Geng L, Zhao H, Edelmann W, Somlo S. Trans-heterozygous Pkd1 and Pkd2 mutations modify expression of polycystic kidney disease. Hum Mol Genet. 2002;11:1845–1854. doi: 10.1093/hmg/11.16.1845. [DOI] [PubMed] [Google Scholar]
- 17.Brasier JL, Henske EP. Loss of the polycystic kidney disease (PKD1) region of chromosome 16p13 in renal cyst cells supports a loss-of-function model for cyst pathogenesis. J Clin Invest. 1997;99:194–199. doi: 10.1172/JCI119147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Qian F, Watnick TJ, Onuchic LF, Germino GG. The molecular basis of focal cyst formation in human autosomal dominant polycystic kidney disease type I. Cell. 1996;87:979–987. doi: 10.1016/s0092-8674(00)81793-6. [DOI] [PubMed] [Google Scholar]
- 19.Arnaout MA. Molecular genetics and pathogenesis of autosomal dominant polycystic kidney disease. Annu Rev Med. 2001;52:93–123. doi: 10.1146/annurev.med.52.1.93. [DOI] [PubMed] [Google Scholar]
- 20.Pei Y. A “two-hit” model of cystogenesis in autosomal dominant polycystic kidney disease? Trends Mol Med. 2001;7:151–156. doi: 10.1016/s1471-4914(01)01953-0. [DOI] [PubMed] [Google Scholar]
- 21.Piontek K, Menezes LF, Garcia-Gonzalez MA, Huso DL, Germino GG. A critical developmental switch defines the kinetics of kidney cyst formation after loss of Pkd1. Nat Med. 2007;13:1490–1495. doi: 10.1038/nm1675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Koptides M, Mean R, Demetriou K, Pierides A, Deltas CC. Genetic evidence for a trans-heterozygous model for cystogenesis in autosomal dominant polycystic kidney disease. Hum Mol Genet. 2000;9:447–452. doi: 10.1093/hmg/9.3.447. [DOI] [PubMed] [Google Scholar]
- 23.Fedeles SV, Tian X, Gallagher AR, Mitobe M, Nishio S, Lee SH, Cai Y, Geng L, Crews CM, Somlo S. A genetic interaction network of five genes for human polycystic kidney and liver diseases defines polycystin-1 as the central determinant of cyst formation. Nat Genet. 2011;43:639–647. doi: 10.1038/ng.860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hughes J, Ward CJ, Peral B, Aspinwall R, Clark K, San Millan JL, Gamble V, Harris PC. The polycystic kidney disease 1 (PKD1) gene encodes a novel protein with multiple cell recognition domains. Nat Genet. 1995;10:151–160. doi: 10.1038/ng0695-151. [DOI] [PubMed] [Google Scholar]
- 25.Harris PC. What is the role of somatic mutation in autosomal dominant polycystic kidney disease? J Am Soc Nephrol. 2010;21:1073–1076. doi: 10.1681/ASN.2010030328. [DOI] [PubMed] [Google Scholar]
- 26.Scheffers MS, van der Bent P, Prins F, Spruit L, Breuning MH, Litvinov SV, de Heer E, Peters DJ. Polycystin-1, the product of the polycystic kidney disease 1 gene, co-localizes with desmosomes in MDCK cells. Hum Mol Genet. 2000;9:2743–2750. doi: 10.1093/hmg/9.18.2743. [DOI] [PubMed] [Google Scholar]
- 27.Foggensteiner L, Bevan AP, Thomas R, Coleman N, Boulter C, Bradley J, Ibraghimov-Beskrovnaya O, Klinger K, Sandford R. Cellular and subcellular distribution of polycystin-2, the protein product of the PKD2 gene. J Am Soc Nephrol. 2000;11:814–827. doi: 10.1681/ASN.V115814. [DOI] [PubMed] [Google Scholar]
- 28.Petri ET, Celic A, Kennedy SD, Ehrlich BE, Boggon TJ, Hodsdon ME. Structure of the EF-hand domain of polycystin-2 suggests a mechanism for Ca2+-dependent regulation of polycystin-2 channel activity. Proc Natl Acad Sci U S A. 2010;107:9176–9181. doi: 10.1073/pnas.0912295107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Koulen P, Cai Y, Geng L, Maeda Y, Nishimura S, Witzgall R, Ehrlich BE, Somlo S. Polycystin-2 is an intracellular calcium release channel. Nat Cell Biol. 2002;4:191–197. doi: 10.1038/ncb754. [DOI] [PubMed] [Google Scholar]
- 30.Geng L, Okuhara D, Yu Z, Tian X, Cai Y, Shibazaki S, Somlo S. Polycystin-2 traffics to cilia independently of polycystin-1 by using an N-terminal RVxP motif. J Cell Sci. 2006;119:1383–1395. doi: 10.1242/jcs.02818. [DOI] [PubMed] [Google Scholar]
- 31.Celic A, Petri ET, Demeler B, Ehrlich BE, Boggon TJ. Domain mapping of the polycystin-2 C-terminal tail using de novo molecular modeling and biophysical analysis. J Biol Chem. 2008;283:28305–28312. doi: 10.1074/jbc.M802743200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Celic AS, Petri ET, Benbow J, Hodsdon ME, Ehrlich BE, Boggon TJ. Calcium-induced conformational changes in C-terminal tail of polycystin-2 are necessary for channel gating. J Biol Chem. 2012;287:17232–17240. doi: 10.1074/jbc.M112.354613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ferreira FM, Oliveira LC, Germino GG, Onuchic JN, Onuchic LF. Macromolecular assembly of polycystin-2 intracytosolic C-terminal domain. Proc Natl Acad Sci U S A. 2011;108:9833–9838. doi: 10.1073/pnas.1106766108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Waters AM, Beales PL. Ciliopathies: an expanding disease spectrum. Pediatr Nephrol. 2011;26:1039–1056. doi: 10.1007/s00467-010-1731-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ware SM, Aygun MG, Hildebrandt F. Spectrum of clinical diseases caused by disorders of primary cilia. Proc Am Thorac Soc. 2011;8:444–450. doi: 10.1513/pats.201103-025SD. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Yamaguchi T, Nagao S, Wallace DP, Belibi FA, Cowley BD, Pelling JC, Grantham JJ. Cyclic AMP activates B-Raf and ERK in cyst epithelial cells from autosomal-dominant polycystic kidneys. Kidney Int. 2003;63:1983–1994. doi: 10.1046/j.1523-1755.2003.00023.x. [DOI] [PubMed] [Google Scholar]
- 37.Yamaguchi T, Pelling JC, Ramaswamy NT, Eppler JW, Wallace DP, Nagao S, Rome LA, Sullivan LP, Grantham JJ. cAMP stimulates the in vitro proliferation of renal cyst epithelial cells by activating the extracellular signal-regulated kinase pathway. Kidney Int. 2000;57:1460–1471. doi: 10.1046/j.1523-1755.2000.00991.x. [DOI] [PubMed] [Google Scholar]
- 38.Pei Y, Paterson AD, Wang KR, He N, Hefferton D, Watnick T, Germino GG, Parfrey P, Somlo S, St, George-Hyslop P. Bilineal disease and trans-heterozygotes in autosomal dominant polycystic kidney disease. Am J Hum Genet. 2001;68:355–363. doi: 10.1086/318188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Jiang ST, Chiou YY, Wang E, Lin HK, Lin YT, Chi YC, Wang CK, Tang MJ, Li H. Defining a link with autosomal-dominant polycystic kidney disease in mice with congenitally low expression of Pkd1. Am J Pathol. 2006;168:205–220. doi: 10.2353/ajpath.2006.050342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Lantinga-van Leeuwen IS, Dauwerse JG, Baelde HJ, Leonhard WN, van de Wal A, Ward CJ, Verbeek S, Deruiter MC, Breuning MH, de Heer E, Peters DJ. Lowering of Pkd1 expression is sufficient to cause polycystic kidney disease. Hum Mol Genet. 2004;13:3069–3077. doi: 10.1093/hmg/ddh336. [DOI] [PubMed] [Google Scholar]
- 41.Rossetti S, Kubly VJ, Consugar MB, Hopp K, Roy S, Horsley SW, Chauveau D, Rees L, Barratt TM, van׳t Hoff WG, Niaudet P, Torres VE, Harris PC. Incompletely penetrant PKD1 alleles suggest a role for gene dosage in cyst initiation in polycystic kidney disease. Kidney Int. 2009;75:848–855. doi: 10.1038/ki.2008.686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Wang E, Hsieh-Li HM, Chiou YY, Chien YL, Ho HH, Chin HJ, Wang CK, Liang SC, Jiang ST. Progressive renal distortion by multiple cysts in transgenic mice expressing artificial microRNAs against Pkd1. J Pathol. 2010;222:238–248. doi: 10.1002/path.2765. [DOI] [PubMed] [Google Scholar]
- 43.Hopp K, Ward CJ, Hommerding CJ, Nasr SH, Tuan HF, Gainullin VG, Rossetti S, Torres VE, Harris PC. Functional polycystin-1 dosage governs autosomal dominant polycystic kidney disease severity. J Clin Invest. 2012;122:4257–4273. doi: 10.1172/JCI64313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Pritchard L, Sloane-Stanley JA, Sharpe JA, Aspinwall R, Lu W, Buckle V, Strmecki L, Walker D, Ward CJ, Alpers CE, Zhou J, Wood WG, Harris PC. A human PKD1 transgene generates functional polycystin-1 in mice and is associated with a cystic phenotype. Hum Mol Genet. 2000;9:2617–2627. doi: 10.1093/hmg/9.18.2617. [DOI] [PubMed] [Google Scholar]
- 45.Fedeles SV, Gallagher AR, Somlo S. Polycystin-1: a master regulator of intersecting cystic pathways. Trends Mol Med. 2014;20:251–260. doi: 10.1016/j.molmed.2014.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Gerdes JM, Davis EE, Katsanis N. The vertebrate primary cilium in development, homeostasis, and disease. Cell. 2009;137:32–45. doi: 10.1016/j.cell.2009.03.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Jonassen JA, San Agustin J, Follit JA, Pazour GJ. Deletion of IFT20 in the mouse kidney causes misorientation of the mitotic spindle and cystic kidney disease. J Cell Biol. 2008;183:377–384. doi: 10.1083/jcb.200808137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Pazour GJ, San Agustin JT, Follit JA, Rosenbaum JL, Witman GB. Polycystin-2 localizes to kidney cilia and the ciliary level is elevated in orpk mice with polycystic kidney disease. Curr Biol. 2002;12:R378–R380. doi: 10.1016/s0960-9822(02)00877-1. [DOI] [PubMed] [Google Scholar]
- 49.Huang K, Diener DR, Mitchell A, Pazour GJ, Witman GB, Rosenbaum JL. Function and dynamics of PKD2 in Chlamydomonas reinhardtii flagella. J Cell Biol. 2007;179:501–514. doi: 10.1083/jcb.200704069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Lin F, Hiesberger T, Cordes K, Sinclair AM, Goldstein LS, Somlo S, Igarashi P. Kidney-specific inactivation of the KIF3A subunit of kinesin-II inhibits renal ciliogenesis and produces polycystic kidney disease. Proc Natl Acad Sci U S A. 2003;100:5286–5291. doi: 10.1073/pnas.0836980100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Haycraft CJ, Swoboda P, Taulman PD, Thomas JH, Yoder BK. The C. elegans homolog of the murine cystic kidney disease gene Tg737 functions in a ciliogenic pathway and is disrupted in osm-5 mutant worms. Development. 2001;128:1493–1505. doi: 10.1242/dev.128.9.1493. [DOI] [PubMed] [Google Scholar]
- 52.Davenport JR, Watts AJ, Roper VC, Croyle MJ, van Groen T, Wyss JM, Nagy TR, Kesterson RA, Yoder BK. Disruption of intraflagellar transport in adult mice leads to obesity and slow-onset cystic kidney disease. Curr Biol. 2007;17:1586–1594. doi: 10.1016/j.cub.2007.08.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Ma M, Tian X, Igarashi P, Pazour GJ, Somlo S. Loss of cilia suppresses cyst growth in genetic models of autosomal dominant polycystic kidney disease. Nat Genet. 2013;45:1004–1012. doi: 10.1038/ng.2715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Yoshiba S, Shiratori H, Kuo IY, Kawasumi A, Shinohara K, Nonaka S, Asai Y, Sasaki G, Belo JA, Sasaki H, Nakai J, Dworniczak B, Ehrlich BE, Pennekamp P, Hamada H. Cilia at the node of mouse embryos sense fluid flow for left-right determination via Pkd2. Science. 2012;338:226–231. doi: 10.1126/science.1222538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Tanaka Y, Okada Y, Hirokawa N. FGF-induced vesicular release of Sonic hedgehog and retinoic acid in leftward nodal flow is critical for left-right determination. Nature. 2005;435:172–177. doi: 10.1038/nature03494. [DOI] [PubMed] [Google Scholar]