Abstract
One important aspect of mesenchymal stromal cells (MSCs)-mediated immunomodulation is the recruitment and induction of regulatory T (Treg) cells. However, we do not yet know whether MSCs have similar effects on the other subsets of Treg cells. Herein, we studied the effects of MSCs on CD8+CD28− Treg cells and found that the MSCs could not only increase the proportion of CD8+CD28− T cells, but also enhance CD8+CD28−T cells' ability of hampering naive CD4+ T-cell proliferation and activation, decreasing the production of IFN-γ by activated CD4+ T cells and inducing the apoptosis of activated CD4+ T cells. Mechanistically, the MSCs affected the functions of the CD8+CD28− T cells partially through moderate upregulating the expression of IL-10 and FasL. The MSCs had no distinct effect on the shift from CD8+CD28+ T cells to CD8+CD28− T cells, but did increase the proportion of CD8+CD28− T cells by reducing their rate of apoptosis. In summary, this study shows that MSCs can enhance the regulatory function of CD8+CD28− Treg cells, shedding new light on MSCs-mediated immune regulation.
Keywords: antigens, CD28, CD8-positive T cells, mesenchymal stromal cells, regulatory T cells
Introduction
Mesenchymal stromal cells (MSCs) can be isolated from various tissues, such as bone marrow, adipose tissue, dental pulp, pancreas and spleen.1 MSCs show enormous expansion potential when cultured in vitro and can differentiate into osteoblasts, adipocytes and chondrocytes, which play important roles in tissue regeneration and damage repair.2 Due to the recent exciting discoveries that MSCs have immunomodulatory properties, these cells have attracted significant attention for their potential to treat immune related diseases, such as graft-versus-host disease (GVHD), liver diseases, autoimmune diseases and tissue inflammation.3,4,5 MSCs have been reported to suppress T cell responses, including activation, proliferation and inflammatory cytokine production.6 Some studies have suggested that MSCs also inhibit the cytotoxic activity of natural killer cells, regulate the maturation and function of B cells and help determine the polarization of macrophages.7 Moreover, MSCs possess the anti-inflammatory and immunomodulatory effects and have been used to treat autoimmune diseases in clinical trials.8
Regulatory T (Treg) cells play important roles in controlling immunologic tolerance and immune system homeostasis.9 Aggarwal and Pittenger7 reported that CD4+CD25+ Treg cells were increased when peripheral blood mononuclear cells (PBMCs) were cocultured with MSCs. Subsequently, Di Ianni et al.10 extensively cocultured human MSCs (hMSCs) with different T-cell subsets, including CD4+CD25+, CD4+CD25+CD45RA+ and CD4+CD25+CD45RO+ T cells, and found that hMSCs could recruit, modulate and maintain the function and phenotype of Treg cells over time. Mechanistically, MSCs-derived TGF-β and PGE2 are key regulators involved in Treg cell induction.11,12 Recently, Mougiakakos et al.13 found that hMSCs could induce non-FOXP3-expressing CD4+ Treg cells, IL-10+ type 1 Treg cells and TGF-β+ Treg cell subsets in an HO-1-dependent fashion.
Although the best characterized Treg cells have the CD4+CD25+ phenotype, CD8+ T-cell populations have also been shown to possess immunosuppressive function, including CD8+CD28−, CD8+CD25+ and CD8+IL-10+ T cells, which can inhibit the activation and proliferation of lymphocytes.12,14,15 CD8+CD28− T cells have been reported to be antigen-specific, limited in their proliferative capacity and susceptible to programmed cell death.16,17 These cells accumulate with aging, probably because of the continuous exposure to foreign antigens.18,19,20 CD8+CD28− T cells have recently attracted interest for their critical roles in various chronic immune disorders, such as Graves's disease, rheumatoid arthritis and type 1 diabetes mellitus.21,22,23 Growing evidence have also suggested that CD8+CD28− T cells can induce tolerance in transplantation, displaying immunosuppressive functions in renal or liver transplantations, as well as other organ transplantations.24,25
The induction of Treg cells in CD4+T cell subsets by MSCs has been studied extensively; however, limited information exists concerning the effects of MSCs on CD8+CD28− Treg cells.12,26 Furthermore, although our previous clinical study demonstrated that the MSC treatment-induced clinical improvement of chronic GVHD (cGVHD) was accompanied by upregulation of CD8+CD28− T cells,27 the mechanisms underlying MSCs affecting the frequency and function of CD8+CD28− Treg cells are still poorly understood.
Hence, we explored the effects and potential mechanisms of MSCs on the proportion and immunomodulatory functions of CD8+CD28− Treg cells.
Materials and methods
MSCs isolation, expansion and characterization
Ethical approval for this study was obtained from the ethics committee of Zhongshan Medical School (Sun Yat-sen University). Heparin-treated bone marrow was obtained by an iliac crest aspiration from healthy donors with informed consent. The MSCs were cultured and characterized as described previously.28 The MSCs differentiated into osteoblasts and adipocytes under special culture conditions (Supplementary Figure 1a–c) and expressed CD29, CD44, CD73, CD90, CD105, but not CD34 or CD45 (all relevant monoclonal antibodies were purchased from BD Biosciences Pharmingen, Franklin Lakes, NJ, USA; Supplementary Figure 1d). All the MSCs used in this study were at passage 5.
Antibodies and flow cytometry
The cells were stained with fluorochrome-coupled monoclonal antibodies (mAbs) according to the manufacturers' recommendations. The following mAbs were obtained from BD Bioscience (San Jose, CA, USA): FITC-conjugated anti-CD3, PE-Cy7-conjugated anti-CD28, PE-conjugated anti-CD4, APC-Cy7-conjugated anti-CD25, APC-conjugated anti-CD8, V500-conjugated anti-CD8, PE-conjugated anti-tumor necrosis factor-related apoptosis inducing ligand (TRAIL), APC-conjugated anti-cytotoxic T-lymphocyte antigen 4 (CTLA-4) and APC-conjugated anti-IL-10. FITC Annexin V Apoptosis Detection Kit was purchased from BD Pharmingen (San Jose, CA, USA). The Alexa Fluor 488-conjugated anti-TGF-β was purchased from R&D Systems (Minneapolis, MN, USA). The PE-conjugated anti-FasL was purchased from eBioscience (San Diego, CA, USA). The flow cytometry analyses were performed using a Gallios flow cytometer (Beckman Coulter, Fullerton, CA, USA) and a BD Bioscience Influx cell sorter, and the data were analyzed with the Kaluza software (Beckman Coulter) and FlowJo7.5 (Treestar, Ashland, OR, USA).
PBMCs isolation and cell sorting
Heparinized peripheral blood samples were acquired from healthy donors and cGVHD patients with written informed consent. The PBMCs were collected by Ficoll-Hypaque density gradient centrifugation and resuspended in tissue culture medium (RPMI-1640 medium (Hyclone, Logan, UT, USA) supplemented with 10% (v/v) fetal bovine serum, 50 U/ml penicillin, and 50 U/ml streptomycin). For the cell sorting, the PBMCs were stained with FITC-conjugated anti-CD3, PE-Cy7-conjugated anti-CD28, PE-conjugated anti-CD4, APC-Cy7-conjugated anti-CD25 or APC-conjugated anti-CD8 antibody. The CD3+ T cells, CD3+CD4+CD25− T cells and CD3+CD8+CD28− T cells were sorted by FACS (BD Bioscience Influx, Franklin Lakes, NJ, USA). The purities of the sorted cells were >95%.
Coculture experiments
The MSCs (1×105 cells/well) were seeded in 24-well flat-bottom plates and incubated for 24 h. T cells were added at different ratios and the increased effect was found optimal for the MSCs/T ratio of 1∶5. The CD3+ T cells (5×105 cells/well) were added on the following day and cells were cocultured for 3 and 6 days. In order to exclude the effects of CD4+ T help cells,29 we cocultured the CD3+CD8+ T cells (5×105 cells/well) with MSCs (1×105 cells/well) for 3 and 6 days. The cells were harvested and stained with APC-conjugated anti-CD8 and PE-Cy7-conjugated anti-CD28 antibodies, and the CD8+CD28− T-cell subpopulation was detected by flow cytometry. For the investigation of CD8+CD28− T cells' function, CD8+CD28− T cells were purified using FACS (BD Bioscience Influx) from CD3+CD8+ T cells cultured with or without MSCs. All the lymphocytes were cultured in RPMI-1640 containing 10% (v/v) fetal bovine serum, 50 U/ml penicillin and 50 U/ml streptomycin.
Proliferation assays
Purified CD3+CD4+CD25− T cells (naive T cells) were stained with 5 µM 5-(and-6)-carboxyfluorescein diacetate succinimidyl ester (CFSE, CellTrace; Invitrogen, Carlsbad, CA, USA). The CFSE-labeled naive T cells (2×105 cells/well) and CD3+CD8+CD28− T cells from the conditional cultures incubated with or without MSCs were seeded into 96-well microplates, treated with 200 ng/ml anti-CD3 mAb at 4 °C overnight, at ratios of 1∶0, 1∶1, 2∶1, 5∶1 and 10∶1 in a total volume of 200 µl of complete medium in the presence of 1 µg/ml CD28 mAb. After 4 days, the percentage of proliferating CD4+T cells was detected by CFSE dilution, as described above. In each experiment, the naive CD4+ T cells and CD8+CD28− T cells were obtained from the same donor. To investigate the activation of the naive T cells, we evaluated two early activation markers of CD4+ T cells, CD69 and CD25, in the similar coculture system of naive CD4+ T cells and CD8+CD28− T cells at different ratios with anti-CD3 mAb 200 ng/ml and IL2 100 U/ml for 24 h.
Cytokine assays
For the analysis of intracellular cytokine production, the CD4+CD25− T cells were cocultured with CD8+CD28− T cells that had been exposed to the conditional cultures in the presence or absence of MSCs. The cells were seeded into 96-well microplates, treated with 200 ng/ml anti-CD3 mAb at 4 °C overnight, at ratios of 1∶0, 1∶1, 2∶1, 5∶1 and 10∶1. The cells were then restimulated with the leukocyte activation cocktail (GolgiPlug; BD Pharmingen) for 6 h and IFN-γ production by CD4+ T cells was detected by flow cytometry according to gating on CD4+ T cells.
Apoptosis assays
Purified CD3+CD4+CD25− T cells (naive CD4+ T cells) were seeded in pre-treated plates (as described for the proliferation assays), stimulated with 100 U/ml IL-2 for 2 days and then harvested. The CD25+ T cells (activated CD4+ T cells) and CD25− T cells (non-activated CD4+ T cells) were isolated by FACS (BD Bioscience Influx). The activated CD4+ T cells (1×105 cells/well) were incubated with sorted CD8+CD28− T cells or MSCs-pre-treated CD8+CD28− T cells in 96-well V-bottom plates at ratios of 1∶0, 1∶1, 2∶1, 5∶1 and 10∶1. After 6 h, the cells were harvested and labeled with APC-conjugated anti-CD8, FITC-conjugated anti-Annexin V antibody and propidium iodide (BD Biosciences). The cytotoxicity of the CD8+CD28−T cells was evaluated as the percentage of Annexin V-positive CD4+ T cells.
The apoptosis ratio of CD8+CD28− T cells was also evaluated by hypodiploid DNA content analysis.30 Purified CD8+CD28− T cells cultured in the presence or absence of MSCs were harvested, fixed with cold 75% ethanol overnight and resuspended in PBS contained 0.2% saponin, 50 μg/ml propidium iodide and 10 μg/ml RNaseA, then analyzed by flow cytometry after incubation for 30 min in the dark.
Blocking studies
The neutralizing antibodies, anti-IL-10 (10 µg/ml) and anti-TGF-β (1 µg/ml), were added at the start of the culture periods for the proliferation, activation and cytokine assays. For the apoptosis assays, the anti-FasL (10 µg/ml) or anti-PD-L1 (10 µg/ml) neutralizing antibodies were added. Additionally, 1 mM 1-MT (Sigma, St Louis, MO, USA), 10 µg/ml anti-IL-6 mAb (R&D Systems), 1 µM Indomethacin (Sigma), 1 µM NS398 (Sigma) or anti-TGF-β (1 µg/ml) was separately added to the coculture system of the CD3+CD8+ T cells and MSCs.
Chronic GVHD patients' characteristic and MSCs infusion
Patients with chronic GVHD met the National Institutes of Health consensus criteria for diagnosis and classification of chronic GVHD patients for clinical trials that detail described in our previous study.28 Clinical data of all patients are summarized in Supplementary Table 1. In addition to the original immunosuppressive therapy, the recruited patients received three infusions (at 4-week intervals) of MSCs derived from third-party healthy donors. The dose of MSCs was 1×106 cells/kg/infusion. Heparinized peripheral blood was obtained from these patients before MSCs treatment and 3 months after the first MSCs infusion. All patients were provided written informed consent.
Statistical analysis
The results were given as the means±s.d. of the independent experiments. The statistical analyses were performed using SPSS v. 17.0 (SPSS Inc. Chicago, IL, USA). The data obtained on the effects of the MSCs on CD8+CD28− T cells under different culture conditions were compared using paired t-tests.
Results
MSCs increase the frequency of CD8+CD28− T cells
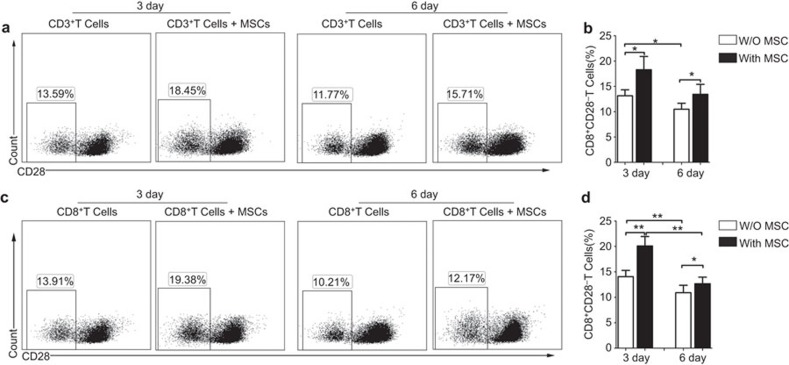
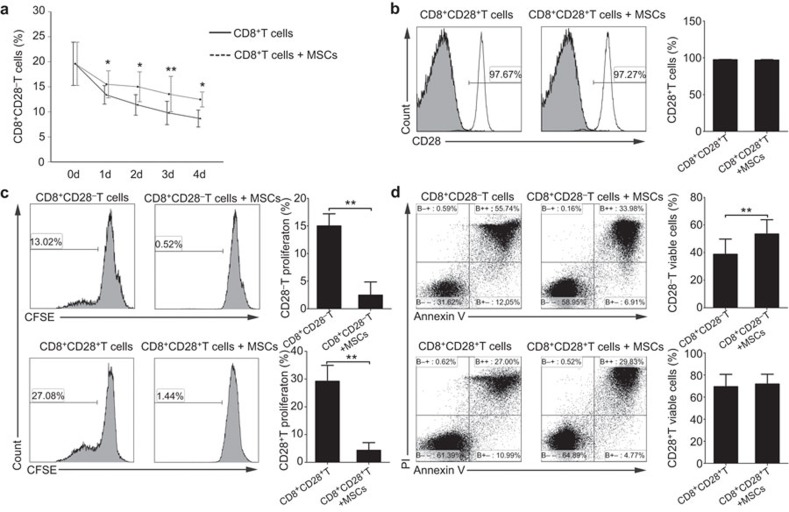
To assess the effects of the MSCs on the proportion of CD8+CD28− T cells in culture, purified CD3+ T cells were cultured with or without MSCs at a ratio of 5∶1. We found that MSCs significantly increased the frequency of CD8+CD28− T cells in the CD3+ T-cell population after 3 days of coculture (18.28%±2.64% versus 13.14%±1.18% of CD3+ T cells alone, P=0.029). The frequency of CD8+CD28− T cells after 6 days culture was still higher when the cells were cultured in the presence of MSCs (13.44%±1.97%% versus 10.49%±1.17% of CD3+ T cells alone, P=0.036) (Figure 1a and b). To avoid the possible interference of CD4+ T cells in the coculture system,29 Purified CD3+CD8+ T cells were cocultured with or without MSCs. Consistently, the much higher frequency of the CD8+CD28− T cells was exhibited in the presence of MSCs (20.08%±1.86%) after 3 days than that in absence of MSCs (14.04%±1.23% P=0.001). In addition, the frequency of the CD8+CD28− T cells presented the same tendency after 6-days culture (12.68%±1.27% with MSCs versus 10.9%±1.46% without MSCs, P=0.038) (Figure 1c and d).
Figure 1.
MSCs increase the percentage of CD8+CD28− T cells. Representative flow cytometry dot plots of the percentage of CD28− T cells by gating on CD8+ T cells among the purified CD3+ T cells (a) or among the purified CD3+CD8+ T cells (c) in the absence or presence of MSCs 3 or 6 days at the ratio of 5∶1, and higher percentages of CD28− T cells were observed when cocultured with MSCs (b, d). The results are representative of four independent experiments. The bar graphs indicate the means±s.d., statistically significant differences are indicated as follows: *P<0.05 and **P<0.01. MSC, mesenchymal stem cell.
MSCs enhance the immunosuppressive functions of CD8+CD28− T cells on inhibiting the proliferation and activation of CD4+CD25− T cells
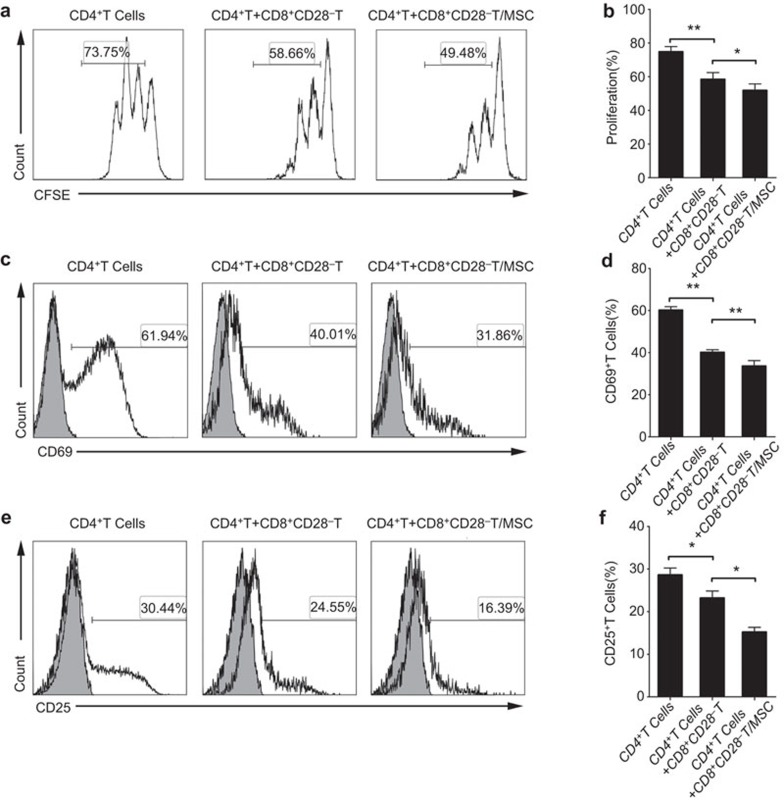
Previous studies have demonstrated that CD8+CD28− Treg cells possessed immune regulatory effects on T-cell responses, and displayed immunosuppressive roles in autoimmune diseases.17,21,31 To investigate whether the MSCs could affect the immunosuppressive function of the CD8+CD28− T cells, we set up a coculture system for evaluating regulatory effect on T-cell proliferation, using CD4+CD25− T cells as responder cells. As shown in Supplementary Figure 2a, CD4+CD25− T cells were well proliferated at on the day 4 in the presence of anti-CD3 mAb and anti-CD28 mAb. And the proliferation was inhibited when the CD8+CD28− T cells were added (Supplementary Figure 2b). The CD8+CD28− T cells strongly suppressed the CD4+ T cell proliferation at a ratio of 1∶1 (CD4+CD25− T responder cells, 72.13%±1.97% versus 56.76%±2.03% P=0.003), but showed a weaker (and non-significant) effect at a ratio of 1∶10 (CD4+CD25− T responder cells, 72.13%±1.97% versus 70.73%±1.86% P=0.567) (Supplementary Figure 2b). Next, we pre-treated the CD8+ T cells with MSCs for 3 days at a ratio of 5∶1, and then sorted the CD8+CD28− T cells, which was followed by coculturing with the CD4+CD25− T responder cells at a ratio of 1∶1. The MSCs-pre-treated CD8+CD28− T cells yielded a significantly lower percentage of proliferating CD4+ T cells (51.84%±3.86%) compared with the untreated CD8+CD28− T cells (58.48%±3.95%, P=0.038) (Figure 2a and b).
Figure 2.
The proliferation and activation of naive CD4+ T cells is markedly inhibited by the CD8+CD28− T cells educated by MSCs. The T-cell proliferative response was evaluated by the CFSE dilution of the CFSE-labeled CD4+ T cells in the presence of anti-CD3 (200 ng/ml) and anti-CD28 (1 μg/ml). The CD8+CD28− T cells and MSCs-pre-treated CD8+CD28− T cells were added respectively at the ratio of 1∶1, and their inhibitory effects on T-cell proliferation were analyzed (a, b). The CD8+CD28− T cells and MSCs-pre-treated CD8+CD28− T cells were cocultured with naive CD4+ T cells at a ratio of 1∶1 in the presence of a stimulus for 24 h, and their effects on T-cell activation were determined by the expression of CD69 (c, d) and CD25 (e, f) on CD4+ T cells. The gray histograms correspond to the isotype controls. The bar graphs indicate the means±s.d. Statistically significant differences are indicated as follows: *P<0.05 and **P<0.01, n=3. CFSE, 5-(and-6)-carboxyfluorescein diacetate succinimidyl ester; MSC, mesenchymal stem cell.
Treg cells show the ability of inhibiting the activation of T cells when they response to foreign or self-antigens.32 Therefore, we next used CD69 and CD25, which were generally regarded as surface-activation markers of T cells,33 to examine the effects of the CD8+CD28− T cells on the activation of naive CD4+ T cells. As shown in Supplementary Figure 2c and d, the expression of CD69 and CD25 on CD4+ T cells reached a high level 24 h after stimulation, and they were significantly and dose-dependently reduced through cocultured with CD8+CD28− T cells. Furthermore, MSCs-pre-treated CD8+CD28− T cells significantly inhibited the expression of CD69 (CD8+CD28− T cells after MSCs pre-treatment, 32.95%±1.42% versus 40.12%±1.21% P=0.005) and CD25 (CD8+CD28− T cells after MSCs pre-treatment, 15.54%±1.14% versus 23.19%±1.67% P=0.024) on CD4+T cells compared with CD8+CD28− T cells that had not been exposed to the MSCs (Figure 2c–f).
Taken together, these findings suggest that the MSCs may enhance the ability of CD8+CD28− T cells to inhibit the proliferation and activation of the CD4+CD25− T cells.
MSCs enhance the ability of CD8+CD28− T cells to inhibit IFN-γ production by responder CD4+ T cells
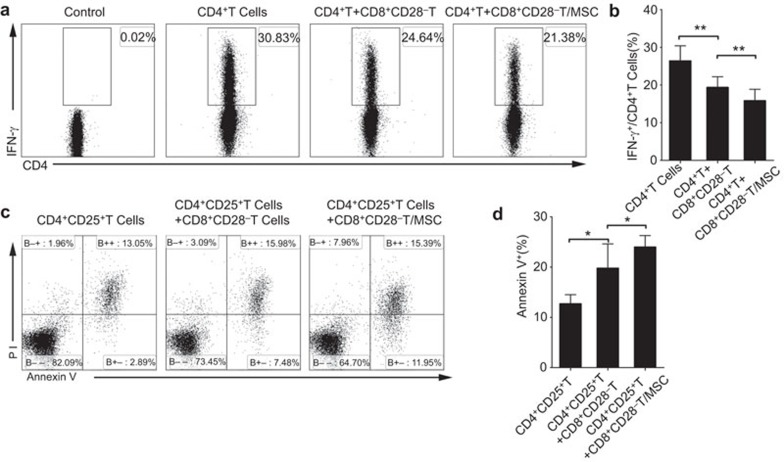
Inhibiting inflammatory cytokine production by CD4+ T cells is one of the immunomodulatory functions of CD8+CD28− T cells.31 To investigate whether MSCs can enhance the immunosuppressive ability of CD8+CD28− T cells, we assessed the ability of MSCs-pre-treated and monocultured CD8+CD28− T cells to modulate the production of IFN-γ by CD4+ T cells. As shown in Supplementary Figure 3a, CD8+CD28− T cells dose-dependently inhibited the IFN-γ production by activated CD4+ T cells, with the maximum inhibition observed at a ratio of 1∶1 (activated CD4+ T cells, 26.37%±4.03% versus 19.34%±4.96% P=0.007). Furthermore, CD4+ T cells produced even less IFN-γ when cocultured with MSCs-pre-treated CD8+CD28− T cells, compared with CD8+CD28− T cells that had not been exposed to MSCs (CD8+CD28− T cells after MSCs pre-treatment, 15.8%±5.27% versus 19.34%±4.96% P=0.003) (Figure 3a and b).
Figure 3.
MSCs enhance the ability of CD8+CD28− T cells to inhibit IFN-γ production by responder CD4+ T cells and induce the apoptosis of activated CD4+ T cells. Flow cytometry plots represent the levels of IFN-γ secreted by the CD4+ T cells. The CD8+CD28− T cells and MSCs-pre-treated CD8+CD28− T cells were added respectively at a ratio of 1∶1, and IFN-γ production was significantly downregulated by MSCs-pre-treated CD8+CD28− T cells (a, b). The purified activated T cells (CD4+CD25+ T cells) were cocultured with CD8+CD28− T cells and MSCs-pre-treated CD8+CD28− T cells at a ratio of 1∶1, and the cytotoxity of CD8+CD28− T cells was evaluated by Annexin V-positive cells within the total CD4+ T-cell population (c, d) The bar graphs indicate the means±s.d.; statistically significant differences are indicated as follows: *P<0.05 and **P<0.01, n=3. MSC, mesenchymal stem cell.
MSCs enhance the capacity of CD8+CD28− T cells to induce apoptosis in activated CD4+ T cells
Because CD4+CD25+Foxp3+ Treg cells could induce apoptosis of responder T cells,34 we then studied the ability of CD8+CD28− T cells on inducing apoptosis of activated CD4+ T cells, and further tested whether MSCs could enhance this function. As shown in Supplementary Figure 3b, the CD8+CD28− T cells dose-dependently induced apoptosis in the activated CD4+CD25+vT cells (as assessed by Annexin V positivity). In contrast, the CD4+CD25− non-activated T cells were resistant to the CD8+CD28− T cell-mediated cytotoxicity (Supplementary Figure 3c). Compared with the CD8+CD28− T cells, the MSCs-pre-treated CD8+CD28− T cells induced apoptosis in a larger proportion of the activated CD4+ T cells (19.77%±4.83% versus 23.98%±3.97% of CD8+CD28− T cells after MSCs pre-treatment; P=0.017) (Figure 3c and d).
MSCs enhance the immune regulatory functions of CD8+CD28− T cells by increasing the expression levels of IL-10 and FasL
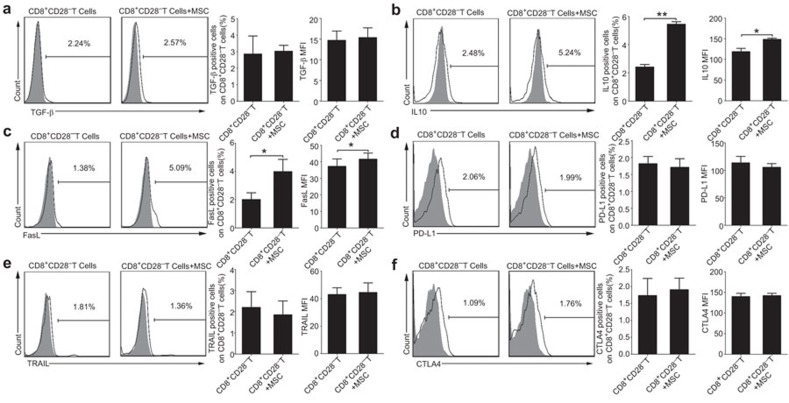
The above findings showed that MSCs efficiently enhanced the immunosuppressive activity of CD8+CD28− T cells. As previous studies have shown that TGF-β or IL-10 involved in the functions of Treg cells,31 we used blocking antibodies against TGF-β (1 µg/ml) or IL-10 (10 µg/ml) to detect the CD8+CD28− Treg cells activity on the proliferation, activation, and IFN-γ production of responder CD4+ T cells. Our results showed that CD8+CD28− T cells partially lost their immunosuppressive ability when anti-TGF-β or anti-IL-10 neutralizing antibody was added to the coculture system respectively (Supplementary Figure 4a–h). Based on these findings, we further evaluated the expression levels of TGF-β and IL-10 in the CD8+CD28− T cells obtained from the CD8+ T cells cocultured with MSCs. We found that there was no difference in the levels of TGF-β expression with or without MSCs coculture (3.02%±0.36% versus 2.85%±1.09% of CD8+ T cells alone; P=0.77) (Figure 4a). However, IL-10 production was higher in the CD8+CD28− T cells obtained from the MSCs-pre-treated CD8+ T cells compared with the monocultured CD8+ T cells (percentage: 5.44%±0.18% versus 2.41%±0.18% of CD8+ T cells alone; P=0.003; MFI: 148.43±2.93 versus 118.62±8.01 of CD8+ T cells alone; P=0.011) (Figure 4b).
Figure 4.
MSCs affect the functions of the CD8+CD28− T cells partially by upregulating the expression of IL-10 and FasL. An analysis of the effector molecules that expressed by the CD8+CD28− T cells in the presence or absence of MSCs, including TGF-β (a), IL-10 (b), FasL (c), PD-L1 (d), TRAIL (e) and CTL-A4 (f). The frequency and mean fluorescence intensity (MFI) of IL-10 and FasL were moderately increased by MSCs (b, c). The results are representative of three independent experiments. The bar graphs indicate the means±s.d. Statistically significant differences are indicated as follows: *P<0.05 and **P<0.01. CTLA-4, cytotoxic T-lymphocyte antigen 4; MSC, mesenchymal stem cell; TRAIL, tumor necrosis factor-related apoptosis inducing ligand.
Because the FasL/Fas and PD-L1/PD-1 pathways represent common apoptotic signaling pathways in many cell types,35,36 we hypothesized that these pathways might be involved in the CD8+CD28− T cell-mediated the apoptosis of CD4+T cells. We thus added blocking antibodies against FasL and PD-L1 to coculture system and performed cytotoxicity experiments. The apoptosis of CD4+ T cells was reduced by the treatment of CD8+CD28−T cells with anti-FasL neutralizing antibody, rather than anti-PD-L1 (Supplementary Figure 4i and j). We also evaluated the expression of FasL and PD-L1 on the CD8+CD28− T cells cultured with or without MSCs, and found that FasL expression was higher in the presence of the MSCs (percentage: 3.98%±0.87% versus 2.0%±0.48% of CD8+ T cells alone; P=0.045; MFI: 41.59±3.7 versus 37.22±4.53 of CD8+ T cells alone; P=0.02) (Figure 4c). In contrast, the PD-L1 expression did not differ significantly in the presence or absence of the MSCs (1.81%±0.0.21% versus 1.71%±0.25% of CD8+ T cells alone; P=0.10) (Figure 4d). We also detected the other factors potentially involved in the killing activity of Treg cells, such as the CTLA-4 and the TRAIL, and found no obvious change in the presence or absence of the MSCs (Figure 4e and f).
To gain insight into the mechanism of how MSCs-mediated the expressions of IL-10 and FasL on CD8+CD28− T cells, we tested several potential mediators using monoclonal neutralizing antibodies specific for IL-6 and TGF-β, specific inhibitors for IDO (1-MT) or cyclooxygenase (NS398 for COX2 or Indomethacin for COX1 and COX2). Unfortunately, as shown in Supplementary Figures 5 and 6, neither inhibitors nor neutralizing antibodies tested showed obvious effects on the expressions of IL-10 or FasL of CD8+CD28− T cells after cocultured with MSCs.
MSCs increase the percentage of CD8+CD28− T cells by decreasing their rate of apoptosis partially through IL-6
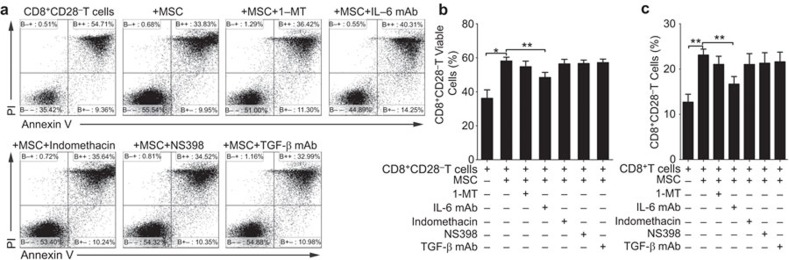
To further understand how MSCs increased the proportion of CD8+CD28− T cells, we performed a kinetic experiment to monitor the changes in the percentage of CD8+CD28− T cells among the total CD8+ T-cell population as culturing CD3+CD8+ T cells with or without MSCs for 4 days. Interestingly, although the frequency of CD8+CD28− T cells decreased gradually over time both in the presence and absence of MSCs, the proportion of CD8+CD28− T cells was higher in the coculture system with the MSCs at the corresponding time point, and showed a significant difference on the third day (Figure 5a). Consequently, we explored the possible reasons for the MSCs affecting increase of proportion CD8+CD28− T cells. Firstly, we investigated whether the MSCs could shift the CD8+CD28+ T cells toward CD8+CD28− T cells, and we cocultured purified CD3+CD8+CD28+ T cells with or without MSCs (ratio of 5∶1) for 3 days. The percentage of CD28+T cells did not significantly differ between these cultures, suggesting that the MSCs may not directly shift the CD8+CD28+ T cells to the regulatory phenotype (Figure 5b). Secondly, we addressed whether MSCs could promote the proliferation of the CD8+CD28− T cells. CFSE-labeled CD8+CD28− T cells and CD8+CD28+ T cells were cultured with or without MSCs (5∶1) in the presence of PHA (5 µg/ml) for 4 days. However, MSCs inhibited the proliferation of the CD8+CD28− T and CD8+CD28+ T cells to similar degrees (Figure 5c). Thus, we then examined the influence of the MSCs on the apoptosis of the CD8+CD28− T and CD8+CD28+ T cells. The percentage of viable cells (those negative for Annexin V and propidium iodide staining) was significantly higher in the CD8+CD28− T cells cocultured with MSCs compared with the monocultures, whereas the viability of the CD8+CD28+ T cells was similar in the cultures with and without the MSCs (Figure 5d). To further confirm this result, we detected the apoptosis of CD8+CD28− T cells by hypodiploid DNA content analysis30 and observed similar result (Supplementary Figure 7). Taken together, MSCs significantly inhibit CD8+CD28− T cells apoptosis. To explore the factors involved in the MSCs-enhanced survival of the CD8+CD28− T cells, we cocultured CD8+T cells and MSCs in the presence of neutralizing antibodies against IL-6 or TGF-β or specific inhibitors of cyclooxygenase (NS398 and Indomethacin) or IDO (1-MT). We found that both the MSCs-mediated decrease of CD8+CD28− T cells apoptosis (Figure 6a and b) and the increase in the frequency of CD8+CD28− T cells (Figure 6c) could be partially blocked by the neutralizing antibody against IL-6, rather than the other tested agents. Collectively, our data strongly suggest that MSCs can increase the frequency of CD8+CD28− T cells by decreasing their apoptosis partially through the IL-6 pathway.
Figure 5.
MSCs increase the percentage of CD8+CD28− T cells by decreasing the apoptosis. The kinetics of the relative percentages of CD8+CD28− T cells were detected in purified CD8+ T cells cocultured with or without MSCs (a). MSCs increased the percentages of CD8+CD28− T cells neither shift CD8+CD28+ T cells to CD8+CD28− T cells (b), nor prior to promote the proliferation of CD8+CD28− T cells (c), but decreased the apoptosis of CD8+CD28− T cells (d). The bar graphs indicate the means±s.d.; statistically significant differences are indicated as follows: *P<0.05 and **P<0.01, n=3.
Figure 6.
MSCs-mediated increase in the frequency of CD8+CD28− T cells and the decrease of CD8+CD28− T cells apoptosis were partially via IL-6. The frequency of viable CD8+CD28− T cells was increased after cocultured with MSCs, and anti-IL-6 mAb partially reversed the MSCs-mediated effects on CD8+CD28− T cell survival (a, b). Similarly, the frequency of CD8+CD28− T cells among purified CD8+ T cells were increased in the presence of MSCs, and that increase was partially blocked by anti-IL-6 mAb (c). The bar graphs indicate the means±s.d.; statistically significant differences are indicated as follows: *P<0.05 and **P<0.01, n=3. mAb, monoclonal antibody; MSC, mesenchymal stem cell.
Clinical efficacy of hMSCs correlates with the frequency of CD8+CD28− T cells in cGVHD patients
To investigate the effects of MSCs on CD8+CD28− T cells in vivo, samples of 13 cGVHD patients who received three infusions of MSCs were analyzed. In the calculation of the maximum response for the 6-month follow-up period, three patients had a complete response (CR), seven patients had a partial response (PR) and three patients had a non-response (NR) (Supplementary Table 1). Because the best therapeutic effects were observed 3 months after the first MSCs infusion, we analyzed the changes of CD8+CD28− T cells at this time point. Compared with the pre-treatment, the frequency of CD8+CD28− T cells within the CD8+ T-cell population was markedly increased (from 31.84%±12.48% to 42.59%±15.71%, P=0.005) (Supplementary Figure 8a and b), whereas no significant change was observed in the NR cGVHD patients (Supplementary Figure 9). A detailed analysis of the expression of IL-10 and FasL on CD8+CD28− T cells was conducted in CR/PR patients. As shown in Supplementary Figure 8c–f, after MSCs treatment, the frequency of CD8+CD28− IL-10+ T cells was increased (from 1.7%±0.62% to 6.21%±1.04%, P=0.02), and the frequency of CD8+CD28−FasL+ T cells was increased from 1.15%±0.39% to 4.43%±0.96% (P=0.012). Taken together, these results suggest that the clinical improvement is accompanied by an increase of CD8+CD28− T cells in the CR/PR cGVHD patients.
Discussion
The main findings of this study are that MSCs can effectively enhance the immunomodulatory function of CD8+CD28− T cells by inhibiting naive CD4+ T-cell proliferation and activation, decreasing the production of IFN-γ by activated CD4+ T cells and inducing apoptosis in activated CD4+T cells. Moreover, our results indicate that the MSCs increase the proportion of the CD8+CD28− T cells by reducing their rate of apoptosis.
Accumulating evidence have demonstrated that the CD8+CD28− T-cell subpopulation displayed a regulatory role in diverse diseases or conditions, such as cancer,37 transplant rejection,38 autoimmune diseases and experimentally induced inflammatory bowel disease.21 Functional studies have reported that CD8+CD28− T cells can dose-dependently inhibit the proliferation and IFN-γ production of responder CD4+ T cells.31,39 Our observation also demonstrated that the CD8+CD28− T cells showed an immunosuppressive function. Moreover, this study demonstrated that CD8+CD28− T cells could induce apoptosis in activated CD4+ T cells and decrease the expression levels of the early activated antigens, CD25 and CD69, on CD4+ T cells, which suggested that the CD8+CD28− T cells possessed pleiotropic immunosuppressive effects similar to those of traditional Treg cells.34
Most of the in vitro and in vivo studies have suggested that MSCs performed the immunomodulatory function by the induction and expansion of Treg cells,40 but few studies have addressed whether MSCs could affect the immunomodulatory functions of Treg cells. Although Prevosto et al.12 reported that the MSCs-mediated immunomodulation was not associated with enhanced Treg cell activity, our study has demonstrated for the first time that MSCs could augment the CD8+CD28− Treg cell activity by inhibiting naive CD4+ T-cell proliferation and activation, decreasing IFN-γ production by the activated CD4+ T cells and inducing apoptosis in the activated CD4+ T cells. These results were similar to the findings that injury-induced increase in CD4+CD25+ Treg cell activity by suppressing T-cell proliferation and Th1-type cytokine production.41 Additionally, Choileain41 et al. found that the injury-induced upregulation in Treg cell activity was cell-contact dependent and mediated partially by increased TGF-β1, rather than IL-10, expression.41 Menager-Marcq et al.31 found that the soluble immunosuppressive factors, TGF-β and IL-10, played crucial roles in the ability of CD8+CD28− Treg lymphocytes to prevent murine colitis. Additionally, CD8+CD28− Treg cells incapable of producing IL-10 lost the functional ability to prevent colitis. Consistent with these findings, we also observed that the CD8+CD28− T-cell inhibition of the CD4+ T-cell proliferation was mediated by both TGF-β and IL-10. In contrast, IL-10 alone rather than TGF-β participated in the inhibition of naïve CD4+ T-cell activation and inflammatory cytokine secretion, which was supported by the observation that only anti-IL-10, and not anti-TGF-β antibodies, blocked the related ability of the CD8+CD28− Treg cells. Furthermore, when CD8+CD28− Treg cells were cultured with MSCs, the expression of IL-10, rather than TGF-β was increased moderately, which implied that the MSCs might enhance the immunosuppressive activity of the CD8+CD28− Treg cells through increased IL-10 production.
The decrease of cytotoxic cells is thought to be another mechanism of Treg cells suppression by the induction of apoptosis in effector cells. FasL, a type-II transmembrane protein which belongs to the tumor necrosis factor family, is mostly expressed on activated T cells and induces apoptosis upon binding with its receptor.34 Meanwhile, the PD-1/PD-L1 pathway performs a crucial role in inhibiting T-cell responses, enhancing T-cell tolerance and preventing immune disorders.42 Other factors potentially involved in the killing activity of Treg cells include the CTLA-4 and the TRAIL.43,44 The question is which pathways are related with the CD8+CD28− T cell-mediated CD4+ T-cell apoptosis? In the present work, blocking the Fas/FasL interaction decreased the apoptosis of activated CD4+ T cells when cultured with CD8+CD28− T cells in vitro, whereas an abrogation of the PD-1/PD-L1 interaction did not show any obvious suppression of the apoptosis of the responder T cells. Furthermore, the MSCs enhanced the killing activity of the CD8+CD28− Treg cells through an upregulation of FasL, rather than PD-L1 expression. Collectively, this evidence supported the hypothesis that MSCs might enhance the regulatory function of the CD8+CD28− T cells by increasing the expression of IL-10 and FasL, and future studies should address the potential mechanisms of CD8+CD28− Treg cells function after the treatment with MSCs.
Another question is how the MSCs increase the proportion of CD8+CD28− T cells in the coculture system. Although, several groups have been reported that MSCs affect the percentage of CD8+CD28− T cells after cocultured with CD3+CD8+ T cells, but the mechanism still need to be elucidated.29,45 CD8+CD28− T cells are considered to be antigen-specific, terminally differentiated, senescent and with shortened telomeres. The present concept that CD8+CD28− T cells lack the ability of proliferation after antigen stimulation has been challenged by several groups who have demonstrated that these T cells also possess the ability of proliferation under certain stimulation conditions. We verified for the first time that the coculture with MSCs did not promote the proliferation of CD8+CD28− T cells or the shift from CD8+CD28+ T cells to a CD8+CD28− T cells phenotype, whereas the MSCs increased the percentage of CD8+CD28− T cells by reducing their apoptosis. This observation were similar with previous study that CD8+CD28− T cells were prone to apoptosis46 because their telomerase activity is decreased by repeated antigenic stimulation.47 The kinetic analysis of the proportion of CD8+CD28− T cells in the CD8+ T cell population showed that the frequency of CD8+CD28− T cells decreased over time, possibly due to their shortened telomeres. Moreover, the protective action of MSCs on CD8+CD28− T cells occurred partially through the action of IL-6, which has been demonstrated to play a crucial role in the suppression of lymphocyte apoptosis by MSCs.48 Additionally, IL-6 has pleiotropic function on preventing the death of resting T cells and activated T cells under certain stimulus.49,50 However, blocking IL-6 did not fully abrogate the anti-apoptotic function of the MSCs on the CD8+CD28− T cells, indicating that some other mediators may not have been identified yet.
Increasing studies showed that the CD8+CD28− T cells played an important role in various immune disorders such as cancer, chronic infections, autoimmune diseases and allogeneic transplantation. Our previous clinical study showed that the MSC treatment-induced clinical improvement of cGVHD was accompanied by the increasing ratio of CD8+CD28−/CD8+CD28+ T cells.19 In this follow-up prospective observation of the MSC treatment of cGVHD patients, we also found an increase in CD8+CD28− T cells in the CR/PR groups, but not in the NR group. Interestingly, clinical improvement was also accompanied by an increase of IL-10 and FasL expression on CD8+CD28− T cells. However, a more complete understanding of the MSCs-induced enhancement of CD8+CD28− Treg cell activity may be of major significance for elucidating the therapeutic mechanisms of MSCs for the treatment of cGVHD.
In conclusion, we herein demonstrate a new immunomodulatory activity of hMSCs: the modulation of CD8+CD28− Treg cell function. MSCs can upregulate the percentage of CD8+CD28− T cells, apparently by protecting them from apoptosis. Thus, our novel findings may facilitate the design of clinical studies and help establish novel monitoring procedures to reproducibly assess the efficacy of MSCs-based treatments in the future.
Author contributions
Qiuli Liu, Haiqing Zheng, Qiquan Sun and Andy Peng Xiang designed the study, analyzed and interpreted the data and wrote the paper, Xiaoyong Chen, Yanwen Peng and Weijun Huang performed cell isolations. Xiaobo Li, Gang Li and Wenjie Xia performed Flow Cytometery analysis of lymphocytes.
Acknowledgments
This study was supported by the National Basic Research Program of China (2012CBA01302, 2010CB945400), the National Natural Science Foundation of China (31171398, 81271265, 81425016), the Key Scientific and Technological Projects of Guangdong Province (2007A032100003), the Natural Science Foundation of Guangdong Province (S2013030013305), the Key Scientific and Technological Program of Guangzhou City (201400000003-3, 201300000089, 2010U1-E00551) and Guangdong Department of Science & Technology Translational Medicine Center grant (2011A080300002).
The authors declare that no conflict of interest exists.
Footnotes
Supplementary Information accompanies the paper on Cellular & Molecular Immunology's website. (http://www.nature.com/cmi).
Supplementary Information
References
- 1da Silva Meirelles L, Chagastelles PC, Nardi NB. Mesenchymal stem cells reside in virtually all post-natal organs and tissues. J Cell Sci 2006; 119: 2204–2213. [DOI] [PubMed] [Google Scholar]
- 2Shi Y, Hu G, Su J, Li W, Chen Q, Shou P, et al. Mesenchymal stem cells: a new strategy for immunosuppression and tissue repair. Cell Res 2010; 20: 510–518. [DOI] [PubMed] [Google Scholar]
- 3Shi M, Liu ZW, Wang FS. Immunomodulatory properties and therapeutic application of mesenchymal stem cells. Clin Exp Immunol 2011; 164: 1–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4Sensebe L, Krampera M, Schrezenmeier H, Bourin P, Giordano R. Mesenchymal stem cells for clinical application. Vox Sang 2010; 98: 93–107. [DOI] [PubMed] [Google Scholar]
- 5Lin H, Xu R, Zhang Z, Chen L, Shi M, Wang FS. Implications of the immunoregulatory functions of mesenchymal stem cells in the treatment of human liver diseases. Cell Mol Immunol 2011; 8: 19–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6Tse WT, Pendleton JD, Beyer WM, Egalka MC, Guinan EC. Suppression of allogeneic T-cell proliferation by human marrow stromal cells: implications in transplantation. Transplantation 2003; 75: 389–397. [DOI] [PubMed] [Google Scholar]
- 7Aggarwal S, Pittenger MF. Human mesenchymal stem cells modulate allogeneic immune cell responses. Blood 2005; 105: 1815–1822. [DOI] [PubMed] [Google Scholar]
- 8Bouffi C, Bony C, Courties G, Jorgensen C, Noel D. IL-6-dependent PGE2 secretion by mesenchymal stem cells inhibits local inflammation in experimental arthritis. PLoS One 2010; 5: e14247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9Sakaguchi S. Naturally arising CD4+ regulatory t cells for immunologic self-tolerance and negative control of immune responses. Annu Rev Immunol 2004; 22: 531–562. [DOI] [PubMed] [Google Scholar]
- 10Di Ianni M, del Papa B, de Ioanni M, Moretti L, Bonifacio E, Cecchini D et al. Mesenchymal cells recruit and regulate T regulatory cells. Exp Hematol 2008; 36: 309–318. [DOI] [PubMed] [Google Scholar]
- 11English K, Ryan JM, Tobin L, Murphy MJ, Barry FP, Mahon BP. Cell contact, prostaglandin E2 and transforming growth factor beta 1 play non-redundant roles in human mesenchymal stem cell induction of CD4+CD25High forkhead box P3+ regulatory T cells. Clin Exp Immunol 2009; 156: 149–160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12Prevosto C, Zancolli M, Canevali P, Zocchi MR, Poggi A. Generation of CD4+ or CD8+ regulatory T cells upon mesenchymal stem cell-lymphocyte interaction. Haematologica 2007; 92: 881–888. [DOI] [PubMed] [Google Scholar]
- 13Mougiakakos D, Jitschin R, Johansson CC, Okita R, Kiessling R, Le Blanc K. The impact of inflammatory licensing on heme oxygenase-1-mediated induction of regulatory T cells by human mesenchymal stem cells. Blood 2011; 117: 4826–4835. [DOI] [PubMed] [Google Scholar]
- 14Roncarolo MG, Gregori S, Battaglia M, Bacchetta R, Fleischhauer K, Levings MK. Interleukin-10-secreting type 1 regulatory T cells in rodents and humans. Immunol Rev 2006; 212: 28–50. [DOI] [PubMed] [Google Scholar]
- 15Fontenot JD, Rasmussen JP, Gavin MA, Rudensky AY. A function for interleukin 2 in Foxp3-expressing regulatory T cells. Nat Immunol 2005; 6: 1142–1151. [DOI] [PubMed] [Google Scholar]
- 16Brenchley JM, Karandikar NJ, Betts MR, Ambrozak DR, Hill BJ, Crotty LE et al. Expression of CD57 defines replicative senescence and antigen-induced apoptotic death of CD8+ T cells. Blood 2003; 101: 2711–2720. [DOI] [PubMed] [Google Scholar]
- 17Arosa FA. CD8+CD28− T cells: certainties and uncertainties of a prevalent human T-cell subset. Immunol Cell Biol 2002; 80: 1–13. [DOI] [PubMed] [Google Scholar]
- 18Merino J, Martinez-Gonzalez MA, Rubio M, Inoges S, Sanchez-Ibarrola A, Subira ML. Progressive decrease of CD8high+ CD28+ CD57− cells with ageing. Clin Exp Immunol 1998; 112: 48–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19Weng NP, Akbar AN, Goronzy J. CD28− T cells: their role in the age-associated decline of immune function. Trends Immunol 2009; 30: 306–312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20Mikulkova Z, Praksova P, Stourac P, Bednarik J, Strajtova L, Pacasova R et al. Numerical defects in CD8+CD28− T-suppressor lymphocyte population in patients with type 1 diabetes mellitus and multiple sclerosis. Cell Immunol 2010; 262: 75–79. [DOI] [PubMed] [Google Scholar]
- 21Tulunay A, Yavuz S, Direskeneli H, Eksioglu-Demiralp E. CD8+CD28−, suppressive T cells in systemic lupus erythematosus. Lupus 2008; 17: 630–637. [DOI] [PubMed] [Google Scholar]
- 22Sun Z, Zhong W, Lu X, Shi B, Zhu Y, Chen L et al. Association of Graves' disease and prevalence of circulating IFN-gamma-producing CD28− T cells. J Clin Immunol 2008; 28: 464–472. [DOI] [PubMed] [Google Scholar]
- 23Scarsi M, Ziglioli T, Airo P. Decreased circulating CD28-negative T cells in patients with rheumatoid arthritis treated with abatacept are correlated with clinical response. J Rheumatol 2010; 37: 911–916. [DOI] [PubMed] [Google Scholar]
- 24Lin YX, Wang LL, Yan LN, Cai P, Li B, Wen TF et al. Analysis of CD8+CD28− T-suppressor cells in living donor liver transplant recipients. Hepatobiliary Pancreat Dis Int 2009; 8: 241–246. [PubMed] [Google Scholar]
- 25Klaus G, Mostert K, Reckzeh B, Mueller TF. Phenotypic changes in lymphocyte subpopulations in pediatric renal-transplant patients after T-cell depletion. Transplantation 2003; 76: 1719–1724. [DOI] [PubMed] [Google Scholar]
- 26Hof-Nahor I, Leshansky L, Shivtiel S, Eldor L, Aberdam D, Itskovitz-Eldor J et al. Human mesenchymal stem cells shift CD8+ T cells towards a suppressive phenotype by inducing tolerogenic monocytes. J Cell Sci 2012; 125: 4640–4650. [DOI] [PubMed] [Google Scholar]
- 27Weng JY, Du X, Geng SX, Peng YW, Wang Z, Lu ZS et al. Mesenchymal stem cell as salvage treatment for refractory chronic GVHD. Bone Marrow Transplant 2010; 45: 1732–1740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28Peng Y, Chen X, Liu Q, Zhang X, Huang K, Liu L et al. Mesenchymal stromal cells infusions improve refractory chronic graft versus host disease through an increase of CD5+ regulatory B cells producing interleukin 10. Leukemia 2014; in press. [DOI] [PubMed]
- 29Engela AU, Baan CC, Litjens NH, Franquesa M, Betjes MG, Weimar W et al. Mesenchymal stem cells control alloreactive CD8+ CD28− T cells. Clin Exp Immunol 2013; 174: 449–458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30Wang R, Shi YF. A simplified protocol for apoptosis assay by DNA content analysis. Biotechniques 2002; Suppl: 88–91. [PubMed] [Google Scholar]
- 31Menager-Marcq I, Pomie C, Romagnoli P, van Meerwijk JP. CD8+CD28− regulatory T lymphocytes prevent experimental inflammatory bowel disease in mice. Gastroenterology 2006; 131: 1775–1785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32Levings MK, Sangregorio R, Roncarolo MG. Human CD25+CD4+ T regulatory cells suppress naive and memory T cell proliferation and can be expanded in vitro without loss of function. J Exp Med 2001; 193: 1295–1302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33Johannisson A, Thuvander A, Gadhasson IL. Activation markers and cell proliferation as indicators of toxicity: a flow cytometric approach. Cell Biol Toxicol 1995; 11: 355–366. [DOI] [PubMed] [Google Scholar]
- 34Pandiyan P, Zheng L, Ishihara S, Reed J, Lenardo MJ. CD4+CD25+Foxp3+ regulatory T cells induce cytokine deprivation-mediated apoptosis of effector CD4+ T cells. Nat Immunol 2007; 8: 1353–1362. [DOI] [PubMed] [Google Scholar]
- 35Gu YZ, Xue Q, Chen YJ, Yu GH, Qing MD, Shen Y et al. Different roles of PD-L1 and FasL in immunomodulation mediated by human placenta-derived mesenchymal stem cells. Hum Immunol 2013; 74: 267–276. [DOI] [PubMed] [Google Scholar]
- 36Nagata S. Fas-mediated apoptosis. Adv Exp Med Biol 1996; 406: 119–124. [DOI] [PubMed] [Google Scholar]
- 37Filaci G, Fenoglio D, Fravega M, Ansaldo G, Borgonovo G, Traverso P et al. CD8+ CD28− T regulatory lymphocytes inhibiting T cell proliferative and cytotoxic functions infiltrate human cancers. J Immunol 2007; 179: 4323–4334. [DOI] [PubMed] [Google Scholar]
- 38Colovai AI, Mirza M, Vlad G, Wang S, Ho E, Cortesini R et al. Regulatory CD8+CD28− T cells in heart transplant recipients. Hum Immunol 2003; 64: 31–37. [DOI] [PubMed] [Google Scholar]
- 39Ceeraz S, Hall C, Choy EH, Spencer J, Corrigall VM. Defective CD8+ CD28− regulatory T cell suppressor function in rheumatoid arthritis is restored by tumour necrosis factor inhibitor therapy. Clin Exp Immunol 2013; 174: 18–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40Burr SP, Dazzi F, Garden OA. Mesenchymal stromal cells and regulatory T cells: the Yin and Yang of peripheral tolerance? Immunol Cell Biol 2013; 91: 12–18. [DOI] [PubMed] [Google Scholar]
- 41Ni Choileain N, MacConmara M, Zang Y, Murphy TJ, Mannick JA, Lederer JA. Enhanced regulatory T cell activity is an element of the host response to injury. J Immunol 2006; 176: 225–236. [DOI] [PubMed] [Google Scholar]
- 42Keir ME, Butte MJ, Freeman GJ, Sharpe AH. PD-1 and its ligands in tolerance and immunity. Annu Rev Immunol 2008; 26: 677–704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43Ren X, Ye F, Jiang Z, Chu Y, Xiong S, Wang Y. Involvement of cellular death in TRAIL/DR5-dependent suppression induced by CD4+CD25+ regulatory T cells. Cell Death Differ 2007; 14: 2076–2084. [DOI] [PubMed] [Google Scholar]
- 44Wing K, Onishi Y, Prieto-Martin P, Yamaguchi T, Miyara M, Fehervari Z et al. CTLA-4 control over Foxp3+ regulatory T cell function. Science 2008; 322: 271–275. [DOI] [PubMed] [Google Scholar]
- 45Zhang W, Ge W, Li CH, You SG, Han Q, Deng WM et al. [Inhibition effect of bone marrow mesenchymal stem cells on T-lymphocyte proliferation through up-regulation of CD8+CD28− T cells]. Zhongguo Shi Yan Xue Ye Xue Za Zhi 2004; 12: 666–669. Chinese. [PubMed] [Google Scholar]
- 46Vallejo AN. CD28 extinction in human T cells: altered functions and the program of T-cell senescence. Immunol Rev 2005; 205: 158–169. [DOI] [PubMed] [Google Scholar]
- 47Strioga M, Pasukoniene V, Characiejus D. CD8+ CD28− and CD8+ CD57+ T cells and their role in health and disease. Immunology 2011; 134: 17–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48Xu G, Zhang Y, Zhang L, Ren G, Shi Y. The role of IL-6 in inhibition of lymphocyte apoptosis by mesenchymal stem cells. Biochem Biophys Res Commun 2007; 361: 745–750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49Teague TK, Marrack P, Kappler JW, Vella AT. IL-6 rescues resting mouse T cells from apoptosis. J Immunol 1997; 158: 5791–5796. [PubMed] [Google Scholar]
- 50Ayroldi E, Zollo O, Cannarile L, D'Adamio F, Grohmann U, Delfino DV, et al. Interleukin-6 (IL-6) prevents activation-induced cell death: IL-2-independent inhibition of Fas/fasL expression and cell death. Blood 1998; 92: 4212–4219. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.