Abstract
The vacuolar-type H+ adenosine triphosphatase (V-ATPase) plays an important role in cellular acidification and bone resorption by osteoclasts. However, the direct effect of bovine parathyroid hormone (bPTH) on V-ATPase has not yet been elucidated. The aim of the present study was to assess the effects of bPTH on V-ATPase and osteoclasts. Osteoclasts from bone marrow (BM)-derived monocytes of C57BL/6 mice were cultured with or without bPTH. The mRNA and protein expression levels of the V-ATPase a3-subunit and d2-subunit (by RT-qPCR and western blot analysis), V-ATPase activity (using the V type ATPase Activity Assay kit) and the bone resorption function of osteoclasts (by bone resorption assay) were examined following treatment with various concentrations of bPTH (0.1, 1.0, 10 and 100 ng/ml) alone or with bPTH and its inhibitor, bafilomycin A1. Furthermore, the expression of parathyroid hormone (PTH) receptors in osteoclasts was also detected. The results revealed that the mRNA and protein expression levels of V-ATPase a3-subunit and d2-subunit increased in a dose-dependent manner, paralleling the level of bPTH present. In addition, an increase in the concentration of bPTH was accompanied by the increased resorption capability of osteoclasts, whereas bone resorption was inhibited in the presence of bafilomycin A1. In addition, we confirmed the existence of parathyroid hormone 1 receptor (PTH1R) in osteoclasts using three different methods (RT-qPCR, western blot analysis and immunofluorescence staining). We found that bPTH enhanced the bone resorption capability of osteoclasts by modulating the expression of V-ATPase subunits, intracellular acidification and V-ATPase activity. Thus, we propose that PTH has a direct effect on osteoblasts and osteoclasts, and that this effect is mediated through PTH1R, thus contributing to bone remodeling.
Keywords: parathyroid hormone, vacuolar-type H+ adenosine triphosphatase, osteoclast, bone resorption, proton pump
Introduction
The parathyroid hormone (PTH) family, including PTH-related peptide (PTHrP), regulates calcium and bone homeostasis and a multitude of developmental processes through its receptors (the PTHRs). Two PTHRs have been identified as PTH family ligands in mammals, designated as parathyroid hormone 1 receptor (PTH1R) and parathyroid hormone 2 receptor (PTH2R), and three in teleostei fish or other non-mammalian vertebrates, including zebrafish, seabream and chicken (1,2). PTH1R mediates the actions of PTH and PTHrP in mammals. Human PTH2R, which is found in the brain, pancreas, heart and kidneys, is activated by PTH and tuberoinfundibular peptide of 39 residues (1,3,4). PTH1R is a G protein-coupled member of the secretin receptor family that interacts with the NH2-terminal 34 amino acids of the ligand (5). PTH1R signaling, which is mediated through the α-subunit of the stimulatory G-protein promotes cyclic adenosine monophosphate production and subsequently activates protein kinase A or protein kinase C (6,7).
Thus far, the opinion of the majority of researchers is that osteoclasts do not express PTHR; it is widely accepted that PTH has an indirect effect on osteoclastogenesis by activating PTH1R in osteoblasts or osteocytes (8–10). Only a few researchers have considered the possibility that PTH1R has a direct effect on osteoclasts (11,12). PTH increases osteoclast formation and bone resorption through the regulation of receptor activator of nuclear factor-kappa B ligand (RANKL)/osteoprotegerin expressed by osteoblasts (13).
Vacuolar-type H+ adenosine triphosphatase (V-ATPase), a type of proton pump, is widely present in eukaryotic cells, and participates in various physiological processes, particularly in the control of intracellular pH (14). V-ATPase is highly expressed in osteoclasts and plays an important role in bone resorption (15). The core structure of V-ATPase has been defined: it consists of V1 and V0 domains and an auxiliary subunit AC45, M8-9 (16). The V1 domain is an approximately 640-kDa peripheral complex on the cytoplasmic side of the membrane (17). This domain is organized into several subunits (A, B1, B2, C1, C2, D, E1, E2, F, G1, G2, G3 and H) and is responsible for ATP hydrolysis (16). The V0 domain is approximately 260 kDa and is membrane-embedded. V0 consists of 10 subunits (a1, a2, a3, a4, c, c′, c″, d1, d2 and e) and mediates proton transport across the membrane. In mammals, the V0 domain contains one of four isoforms (a1, a2, a3 and a4) of the a-subunit, which is a large integral protein and contributes to the proton pore (18). The a1 isoform is most highly expressed in brain and myocardial cells; a2 is most highly expressed in the acrosomal membrane in sperm; the a3 isoform is highly expressed by osteoclasts, microglia and pancreatic β cells, whereas the a4 isoform is highly expressed by renal intercalated cells (19). Disruption of the mouse a3 encoding gene has been noted to result in severe osteopetrosis and mutations of the a3 isoform in humans and also leads to a condition known as auto-somal recessive osteopetrosis including infantile malignant osteopetrosis. This compelling genetic evidence suggests that the a3 isoform is essential for, and specific to, osteoclastic bone resorption (15,17,20). In addition, the d1 isoform is ubiquitously expressed, whereas the d2 isoform is expressed predominantly in the kidneys and osteoclasts. The d2 isoform is an essential component of the osteoclast-specific proton pump that mediates the extracellular acidification in bone resorption (21). In d2 gene knockout mice, failure of pre-osteoclast fusion into mature multinucleated cells was noted, as was the osteopetrosis phenotype (22). Mutations in the d2-subunit of V-ATPase result in reduced tartrate-resistant acid phosphatase (TRAP) expression and decreased fusion of osteoclast precursors (23). These data suggest that the d2-subunit has dual functions in relation to the regulation of osteoclast maturation and osteoclast extracellular acidification. Thus, our study focuses on the expression of the a3-subunit and d2-subunit, where they serve as a component of V-ATPase in mature osteoclasts. Furthermore, we wished to determine whether PTH has a direct effect on osteoclasts and whether this effect is linked to V-ATPase.
Materials and methods
Reagents
C57BL/6 mice were provided by the Laboratory Animal Center of Sun Yat-Sen University (Guangzhou, China). Six 6–10-week-old and three 7-day-old female mice were used (type C57BL/6), typically weighting 25 g and 6.0 g, respectively. All animal experiments were approved by the Institutional Animal Care and Use Committee at the School of Life Sciences at Sun Yat-Sen University. All experimental procedures were performed in accordance with the Guide for the Care and Use of Laboratory Animals and the Institutional Ethical Guidelines for Animal Experiments. C57BL/6 mice were employed for cell cultures. Calvarial osteoblasts from postnatal and bone marrow (BM) cells from adult C57BL/6 mice were collected.
All primers were designed and synthesized by Invitrogen Life Technologies (Carlsbad, CA, USA). The reverse transcription reagent kit and the real-time polymerase chain reaction (PCR) reagents were purchased from Takara Bio (Dalian, China). A BCA Protein Assay kit and BCECF-AM solution were acquired from Beyotime Institute of Biotechnology (Nanjing, China). The TRAP staining kit was purchased from Sigma-Aldrich (St. Louis, MO, USA). The AKP staining kit was purchased from Jiancheng Bioengineering Institute (Jiancheng, Nanjing, China). Bovine PTH [bPTH-(1–34)] was purchased from Bachem Bioscience Inc. (King of Prussia, PA, USA). Soluble murine RANKL and macrophage colony-stimulating factor (M-CSF) were purchased from R&D Systems (Minneapolis, MN, USA). Antibodies to the V-ATPase a3-subunit (Cat. no. H00010312-K) and d2-subunit (Cat. no. H00245972-M01) were purchased from Abnova, Inc. (Taipei, Taiwan). Anti-rabbit IgG-FITC antibody (Cat. no. sc-2012), and PTH1R (Cat. no. sc-20749) and PTH2R (Cat. no. sc-30005) antibodies were purchased from Santa Cruz Biotechnology, Inc. (Santa Cruz, CA, USA).
Administration of bPTH
The amino acid sequences of murine and bovine PTH are highly conserved, as has been previously observed (24). bPTH is frequently used in research to intervene in cell multiplication and differentiation processes (25–27). In the present study, bPTH-(1–34) powder was dissolved in 0.1% bovine serum albumin (BSA) and stored at −20°C. The control cultures were treated with the same concentration of BSA. In our experiments, the treated groups were incubated with bPTH from day 5 to 6. The final concentration of bPTH in the culture medium was 0.1, 1.0, 10 and 100 ng/ml.
Culture of primary mouse monocytes and differentiation of osteoclasts
BM cells were isolated from the femurs of 6–10-week-old C57BL/6 mice by flushing the shaft with phosphate-buffered saline (PBS) using needles, and the cells were further dispersed several times by gentle, repeated pipetting with a sterile pipette. The isolated cells were cultured for 24 h in α-MEM containing 100 U/ml penicillin, 100 µg/ml streptomycin and 10% FCS. Non-adherent BM-derived monocytes were collected and cultured for 48 h in α-MEM (Invitrogen, Shanghai, China) containing 100 U/ml penicillin (Gibco, Shanghai, China), 100 µg/m streptomycin (Gibco), 10% FCS (Biological Industries, Kibbutz Beit-Haemek, Israel) and 25 ng/ml recombinant M-CSF as previously described (28). After the BM-derived monocytes were induced to differentiate into mononuclear macrophages, the stromal cells and lymphocytes which cannot adhere to the suspension culture dish were removed and the adherent cells were collected, pelleted and counted before being seeded at 104 cells/cm2 in the presence of recombinant RANKL. Fresh medium containing 35 ng/ml RANKL was added every 2 days until multinucleated osteoclasts formed (approximately 5 days). After the mononuclear macrophages were cultured for 5 days, the cells were fixed and stained using the TRAP kit based on the manufacturer's instructions (Sigma-Aldrich). TRAP-positive multinucleated cells containing 3 or more nuclei were identified as osteoclasts.
RNA isolation and RT-qPCR
The osteoclasts were induced in 6-well plates as mentioned above and cultured with bPTH from day 5 to 6. Fresh medium containing a different concentration of bPTH was replaced every 2 days. Following culture for 6 days, total RNA was isolated from the osteoclasts using TRIzol reagent. For RT-qPCR, cDNA was synthesized from 1 µg total RNA using reverse transcriptase and oligo(dT) primers in a volume of 10 µl, and the reaction mixture was finally adjusted to 50 µl with TE buffer for PCR. The cDNA amplification reaction mixture was initially incubated at 95°C for 30 sec to denature DNA. Amplification was performed for 40 cycles of 95°C for 5 sec and 60°C for 34 sec, respectively. qPCR was performed under the following conditions: 95°C for 2 min, then 40 cycles of 95°C, 30 sec; 60°C, 1 min; 72°C, 2 min. The specificity of the PCR products was verified by melting curve analysis. Data were normalized using the GAPDH housekeeping gene as an endogenous control. All primers were derived against murine sequences. The following primer sets were used: GAPDH forward, CCATGTTTGTGATGGGTGTGAACC and reverse, TGTGAGGGAGATGCTCAGTGTTGG; V-ATPase a3-subunit (Atp6v0a3) forward, GAGA CCTCAACGAATCCGTGA and reverse, CGATCCGTTTCCTCCTGGA; V-ATPase d2-subunit (Atp6v0d2) forward, CTGGTTCGAGGATGCAAAGC and reverse, GTTGCCATAGTCCGTGGTCTG; PTH1R forward, GCACACAGCAGCCAACATAA and reverse, CGCAGCATAAACGACAGGAA; and PTH2R forward, GGCTGATTCTCAGTAGCTGTCT and reverse, GGGCCAACAAATGATCCCATC.
Western blot analysis
The osteoclasts were induced in 12-cm petri dishes. Following 48 h of co-culture with bPTH (0.1, 1.0, 10 and 100 ng/ml), protein lysates of osteoclasts were prepared in RIPA buffer (Beyotime Institute of Biotechnology). Cell lysates (30 µg) were electrophoresed on 12% polyacrylamide-SDS gels. Proteins were then transferred onto a nitrocellulose membrane and incubated with the following rabbit anti-mouse antibodies (0.2 µg/ml): anti-V-ATPase a3-subunit, anti-V-ATPase d2-subunit, anti-PTH1R, anti-PTH2R and anti-GAPDH as an internal control, followed by 1 µg/ml anti-rabbit immunoglobulin G-horseradish peroxide conjugate. The bands were scanned, and the intensity was measured using a digital gel electrophoresis image processing and analysis system (Tanon, Shanghai, China).
Determination of intracellular pH
The osteoclasts were induced in 24-well plates and treated with 0.1, 1, 10 and 100 ng/ml bPTH from day 5 to 6. Following culture for 6 days, the cells were harvested and treated with 20 mM ammonium chloride for 15 min and washed twice with α-MEM free of phenol red and then placed into a 24-well plate with cover slips. The cells were then stained with the dyeing agent, BCECF-AM, a fluorescent probe that binds specifically to hydrogen ions, and incubated at 37°C for 30 min. A total of 10 osteoclasts from each well were selected randomly to determine the fluorescence intensity (excitation at 488 nm, emission detected at 535 nm) under a laser scanning confocal microscope (Leica SP5-FCS; Leica Microsystems GmbH, Wetzlar, Germany), with the help of the image analyzing software Image-Pro Plus 6.0 (Media Cybernetics, Inc., Houston, TX, USA).
V-ATPase activity assay
To investigate V-ATPase activity, protein samples were extracted from 5 groups with the method described in the study by Koizumi et al (29). The protein concentration was determined using the BCA Protein Assay kit. V-ATPase activity assay was performed using the c (Genmed Scientifics, Inc., Arlington, MA, USA). In the presence or absence of bafilomycin A1 (APExBIO Technology, Houston, TX, USA), which is a sensitive inhibitor of V-ATPase, V-ATPase hydrolyzed the substrate ATP in a pyruvate kinase and lactate dehydrogenase (LDH) system. In this reaction, NADH was converted to NAD, and the activity of V-ATPase was calculated according to the change of absorbance at 340 nm. The unit of measurement is µmol ATP/min/mg. These assay protocols were executed according to the manufacturer's instructions.
Bone resorption assay
The osteoclasts were induced using the foregoing method and grown in each well of Osteo Assay Surface multiple-well plates (Corning, NY, USA) for 6 days. Various concentrations of bPTH were added to the culture from day 5 to 6; 10 nM bafilomycin A1 was used to block V-ATPase. Following culture for 6 days, the plates were washed with saline and a solution of 5% sodium hypochlorite was added for 5 min in order to detach the cells. The cells were then stained with 1% toluidine blue. The resorption area was examined under a microscope and each group was compared using ImageJ software (National Institutes of Health, Bethesda, MD, USA).
Detection of PTHRs in osteoclasts by immunofluorescence staining
The primary antibodies used were polyclonal rabbit anti-mouse PTH1R (Cat. no. sc-20749) and PTH2R (200 µg/ml; Cat. no. sc-30005) (both from Santa Cruz Biotechnology, Inc.) diluted at 1/50. The secondary antibodies were FITC-conjugated goat anti-rabbit (Cat. no. sc-2012; Santa Cruz Biotechnology, Inc.). DAPI (1 mg/ml) was used to identify the nuclei. The cells were fixed in 4% PFA prior to antibody staining. After being washed with PBS (0.01 M, pH 7.4) solution 3 times, glass cover slips were saturated with washing solution (5% BSA) for 30 min. The cells were incubated with the primary antibody diluted in washing solution for 30 min and then rinsed in the same solution for 15 min prior to incubation with the secondary conjugated antibodies (1:100 dilution) for 30 min at room temperature. The cells were then washed for 15 min and incubated with DAPI (1:1,000 dilution) for 5 min. Finally, the cells were washed for 15 min. Primary mouse calvarial osteoblasts were isolated from 7-day-old C57BL/6 mice using the method described by Wang et al (30). In brief, osteoblasts were isolated from the calvaria of 7-day-old C57BL/6 mice by 4 sequential 15-min enzyme digestions at 37°C in solution containing 0.05% trypsin-EDTA and 0.1% collagenase P (Gibco). The cells released from the second to fourth digestions were pooled, centrifuged, resuspended and plated at 1.5×104/cm2 in 25cm2 culture plates in DMEM (Invitrogen, Shanghai, China) containing 10% (v/v) FCS (Biological Industries), 100 U/m penicillin (Gibco), 100 µg/mL streptomycin (Gibco) and non-essential amino acids (100 µM). The plated cells became confluent around days 5–7. Then the culture medium was changed to differentiation medium. The osteoblasts were then cultured in plastic dishes or plates containing α-MEM supplemented with 10% (v/v) FCS, 100 U/ml penicillin (Gibco), 100 µg/ml streptomycin (Gibco) and 2 mM glutamine (Amresco, Solon, OH, USA). Following 48 h of culture, the cells were stained as positive controls. Immunofluorescence and confocal microscopy were performed immediately. No specific staining was observed if the primary antibodies were omitted.
Statistical analysis
The results are represented as the means ± the standard deviation (SD). Data were statistically analyzed by one-way analysis of variance (ANOVA) using SPSS 13.0 software (SPSS Inc., Chicago, IL, USA). All statistical tests were two-sided; a P-value <0.05 was considered to indicate a statistically significant difference.
Results
Impact of bPTH on osteoclast differentiation
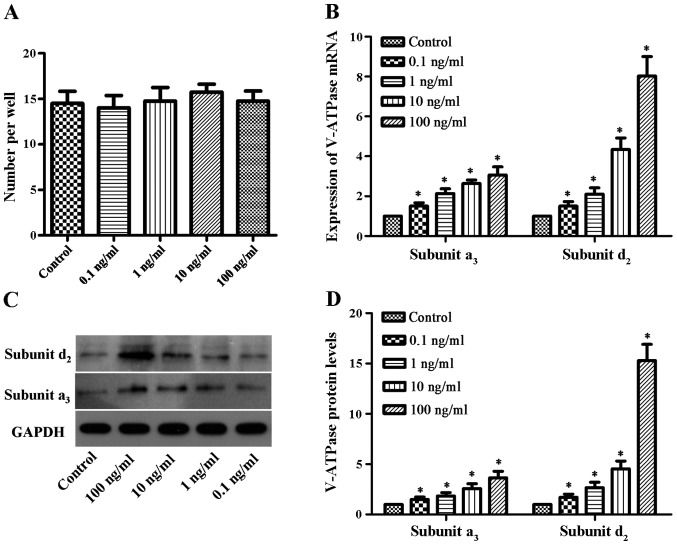
After the osteoclasts were induced in 96-well plates and cultured with various concentrations of bPTH for 5 days, the number of mature osteoclasts was determined (Fig. 1A). The number of mature osteoclasts in the control group and bPTH-treated groups (0.1, 1, 10 and 100 ng/ml) was 14.5±2.6, 14.0±2.7, 14.8±3.0, 15.8±1.7 and 14.7±2.2, respectively. There were no significant differences observed between the different groups. These results suggest that bPTH does not have a great impact on osteoclast differentiation.
Figure 1.
Impact of bovine parathyroid hormone (bPTH) on osteoclast differentiation. (Α) The number of mature osteoclasts was calculated in each group. Results were from 3 individual experiments performed in triplicate. There was no significant difference between these groups. A P-value <0.05 denoted a statistically significant difference. (B) Effect of bPTH on vacuolar-type H+ adenosine triphosphatase (V-ATPase). Osteoclasts were treated with 0.1, 1, 10 and 100 ng/ml bPTH in medium for 48 h. RT-qPCR was performed, and the expression of V-ATPase a3-subunit and d2-subunit was normalized to GAPDH. Results are from 3 independent experiments performed in triplicate, and are displayed as relative expression calibrated to the control samples. Data are represented as the means ± SEM, as shown by the vertical bars. *P<0.05 vs. control. (C) Western blot analysis was used to detect the protein expression of V-ATPase a3-subunit and d2-subunit. (D) Densitometric scan analysis of western blot analysis. Data are expressed as the means from triplicate determinations (means ± SEM) shown by the vertical bars (*P<0.05).
Effect of bPTH on V-ATPase expression
RT-qPCR was used to measure the mRNA expression levels of the V-ATPase a3-subunit and d2-subunit. Compared to the control group, the mRNA expression of V-ATPase was found to be 1.5-3-fold higher for the a3-subunit and 1.5-8-fold higher for the d2-subunit (Fig. 1B). The protein expression levels increased by 1.4-3.6-fold and 1.7-15-fold for the V-ATPase a3-subunit and d2-subunit, respectively, in the bPTH-treated groups compared to the control (Fig. 1C and D). The mRNA and protein expression levels of V-ATPase differed significantly between the bPTH-treated groups and the control (P<0.05). These results indicated that the mRNA and protein expression of the V-ATPase a3-subunit and the d2-subunit were elevated in a dose-dependent manner (with increasing concentrations of bPTH).
Effect of bPTH on intracellular pH in osteoclasts
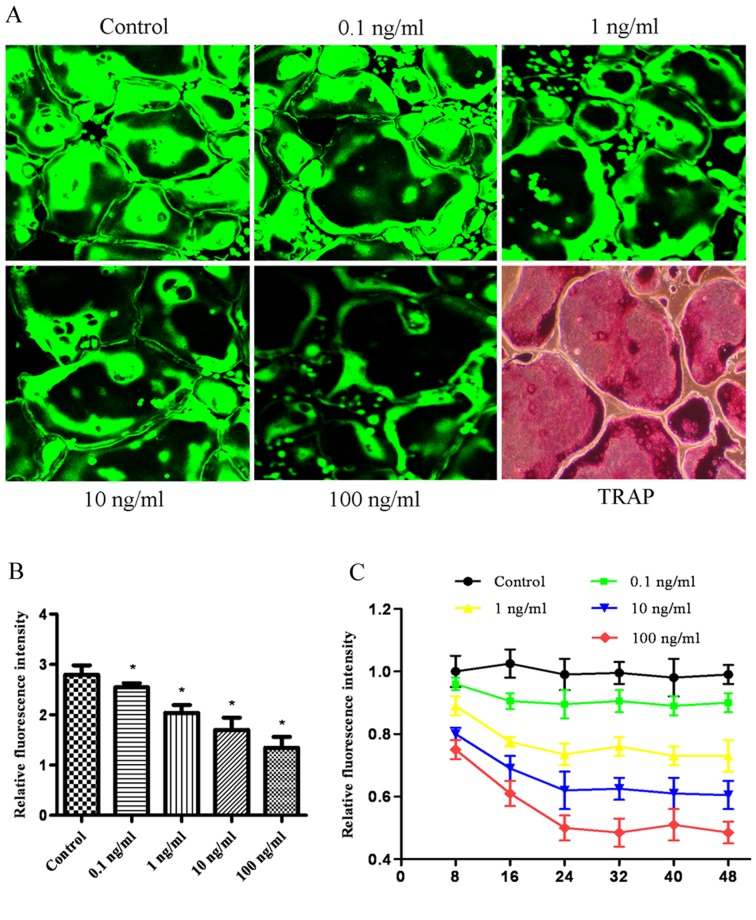
BCECF-AM is capable of sensing intracellular pH in living cells, with the fluorescence intensity decreasing as the pH value decreases from 9.5 to 6.2. The effect of bPTH on the intracellular pH value in osteoclasts was assessed by laser scanning confocal microscopy. Treatment with increasing concentrations of bPTH, from 0.1 to 100 ng/ml, decreased the fluorescence intensity (Fig. 2). The fluorescence intensity decreased with time in the first 24 h and reached a platform thereafter.
Figure 2.
Effect of bPTH on the intracellular pH of osteoclasts. (A) Osteoclasts were stained with BCECF-AM, and the fluorescence intensity was examined under a laser scanning confocal microscope. The green fluorescence intensity declined, with a decrease of intracellular pH value. The bottom right panel depicts tartrate-resistant acid phosphatase (TRAP) staining of osteoclasts. Mature osteoclasts were identifiable as large, multinucleated giant cells that express TRAP. (B) Relative fluorescence intensity per cell. Data are expressed as the means from triplicate determinations (means ± SEM), represented by vertical bars (*P<0.05). (C) The time course for the effect of bPTH on intracellular pH value. Fluorescence intensity was examined as mentioned above.
Effect of bPTH on V-ATPase activity
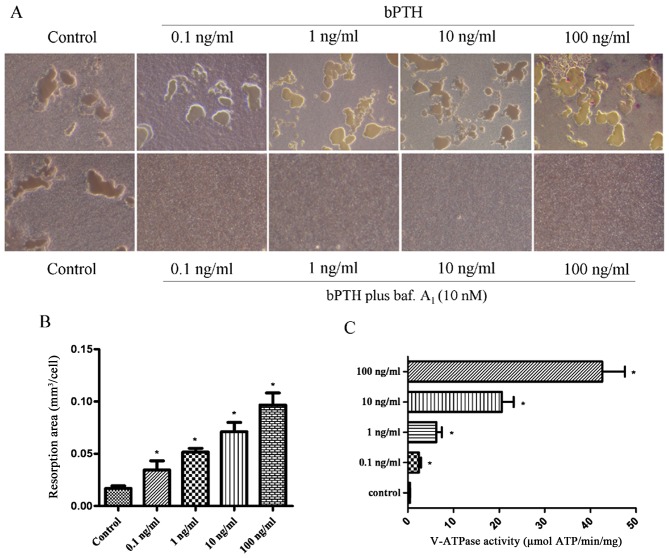
In this study, a significant increase in V-ATPase activity was observed in the bPTH-treated groups compared with the control group. The increase in V-ATPase activity indicated a positive association with the concentrations of bPTH (Fig. 3C). V-ATPase activity was 42.59±9.97, 20.58±5.17, 6.20±2.46, 2.41±0.91 and 0.42±0.12 (µmol ATP/min/mg) in the treated groups (100, 10, 1.0 and 0.1 ng/ml) and the control, respectively.
Figure 3.
Effect of bovine parathyroid hormone (bPTH) on bone resorption. Osteoclasts were cultured in Osteo Assay Surface multiple-well plates and treated with various concentrations of bPTH for 2 days. (A) The absorbed areas on the plates were captured at 100× magnification with a digital camera attached to the microscope. Resorption planes from 10 random fields for each well were analyzed using ImageJ software. Upper panels, bPTH group; lower panels, bPTH plus bafilomycin A1 (baf. A1; 10 nM) group. (B) The concentrations of bPTH were 0.1, 1.0, 10 and 100 ng/ml. The bone resorption area of osteoclasts. Each value of bone resorption indicates the means ± SEM. *P<0.05 vs. value for the controls. (C) Vacuolar-type H+ adenosine triphosphatase (V-ATPase) activity in response to bPTH. Osteoclasts were bPTH-treated or vehicle-treated. Data are expressed as the means from triplicate determinations (means ± SEM) shown by the horizontal bars (*P<0.05).
Effect of bPTH on bone resorption
As a culture dish with its bottom coated with calcium-phosphate thin film that would become a substrate for osteoclasts, the Osteo Assay Surface multiple-well plate (Corning, NY, USA) is designed to measure the bone resorption activity of osteoclasts. The bone resorption activity of the osteoclasts can be measured by direct observation under a phase contrast microscope without using a scanning electron microscope (SEM). The osteoclasts were induced and cultured on plates covered with hydroxyapatite crystals. Following culture for 6 days, the bone resorption area of osteoclasts in the bPTH-treated groups increased by 2–6-fold, in parallel with the increasing concentration of bPTH in the culture medium (Fig. 3). The bone resorption area of the control group and bPTH-treated groups was 0.050±0.007, 0.103±0.026, 0.155±0.011, 0.213±0.027 and 0.290±0.035 mm2, respectively. In the osteoclast groups treated with bPTH plus bafilomycin A1, bone resorption was completely inhibited (Fig. 3A). This implies that exposure to bPTH enhances the bone resorption capability of osteoclasts.
Expression of PTHRs in osteoclasts
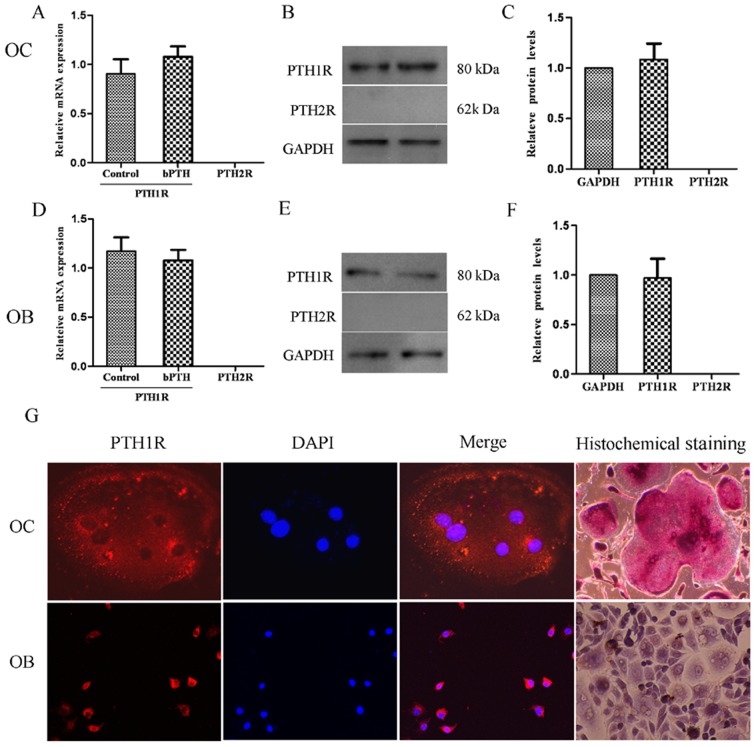
The expression of PTH1R and PTH2R was analyzed using RT-qPCR, western blot analysis and immunofluorescence staining (Fig. 4). The results revealed that the osteoclasts expressed PTH1R mRNA both in the control group and the bPTH-treated group (1 ng/ml), but PTH2R mRNA was not detectable. It seemed that the mRNA expression of PTH1R was not markedly affected by the low dose of bPTH, and neither was PTH1R protein expression, as was also detected by western blot analysis. Moreover, positive staining for PTH1R protein was observed in both osteoblasts and osteoclasts by laser scanning confocal microscopy. These results suggest that PTH exerts its effects on both osteoblasts and osteoclasts by binding to PTH1R and that downstream signaling pathways are initiated to maintain the balance of osteogenesis and osteolysis. However, this balance would be broken by a high serum level of PTH, which exists in pathalogical states, such as hyperparathyroidism.
Figure 4.
Detection of PTHRs. (A) Relative mRNA expression of parathyroid hormone 1 receptor (PTH1R) and parathyroid hormone 2 receptor (PTH2R) in osteoclasts. RT-qPCR was used to analyze the mRNA expression of PTH1R and PTH2R in osteoclasts (normalized to GAPDH). Data are expressed as the means from triplicate determinations (means ± SEM) shown by the vertical bars (*P<0.05). (B) Western blot analysis of PTH1R and PTH2R protein expression in osteoclasts normalized to GAPDH. Representative blots from three independent experiments are shown. (C) Densitometric scan analysis for western blot analysis. Data are expressed as the means from triplicate determinations (means ± SEM) shown by the vertical bars (*P<0.05). (D) Relative mRNA expression of PTH1R and PTH2R in osteoblasts. (E) Western blot analysis of PTH1R and PTH2R protein expression in osteoblasts normalized to GAPDH. (F) Densitometric scan analysis for western blot analysis. (G) Immunofluorescence detection of PTH1R. Cells were cultured in plastic plates with a cover slip placed inside wells before seeding. Nuclei were stained in blue and PTH1R was stained in red. Positive staining for PTH1R in osteoclasts (OC) was observed with a laser scanning confocal microscope at 40× magnification. Osteoblasts (OB) that express PTH1R were stained as positive controls. As shown in the upper-right corner of the image, mature osteoclasts were identifiable as large multinucleated giant cells that express tartrate-resistant acid phosphatase (TRAP). After 10 days of culture, osteoblasts were also dyed using AKP histochemical staining to confirm the origin of the cells. Red-brown granules appeared in mature osteoblast cytoplasm and nuclei were stained in purple.
Discussion
Osteoclasts are multinucleated cells fused from BM-derived monocytes-macrophages, which are specialized for and involved in bone resorption (31). In patients with end-stage renal disease, the excessive secretion of PTH has been shown to increase osteoclast activity, stimulate bone resorption, mobilize bone calcium and disrupt the balance between osteoclasts and osteoblasts (32). Generalized bone loss from increased osteoclast activity also contributes to renal osteodystrophy and a significant reduction in the quality of life (33). Generally, the unique ability of osteoclasts to degrade skeletal tissue depends on the formation of a resorptive microenvironment between osteoclasts and the bone surface (34). V-ATPase plays a crucial role in this skeleton remodeling process; nevertheless, the underlying mechanisms remain poorly understood. The findings of the present study revealed that the exposure of osteoclasts to bPTH induced the high expression of the V-ATPase a3-subunit and d2-subunit, promoted V-ATPase activity, accelerated intracellular acidification and increased the bone-resorption capability of osteoclasts. We also found that PTH1R exists in BM-derived osteoclasts. Based on these results, we hypothesized that bPTH binds to PTH1R and increases the bone resorption capability of osteoclasts by increasing the expression of the V-ATPase a3-subunit and d2-subunit. PTH not only promotes the formation of osteoblasts, but also accelerates bone resorption by osteoclasts by combining with PTH1R (Fig. 5). Therefore, V-ATPase and PTH1R may be novel treatment targets in bone disease (35).
Figure 5.
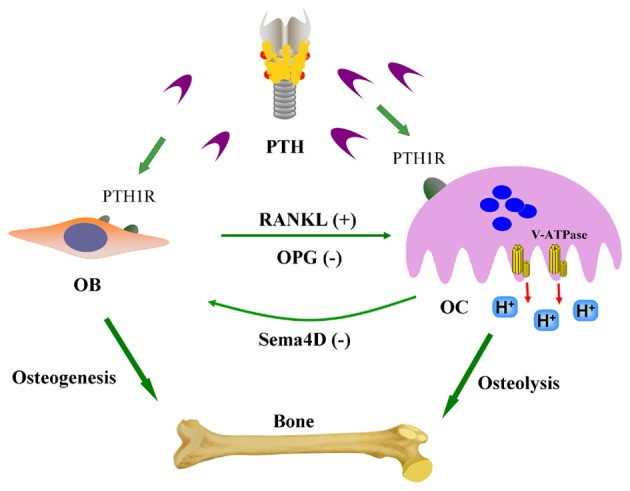
A model of parathyroid hormone (PTH)-mediated cellular interactions in bone remodeling. PTH stimulates osteoblasts (OB) to secrete receptor activator of nuclear factor-kappa B ligand (RANKL) and enhance the activity of osteoclasts (OC). RANKL interacts with its receptor (RANK), which is expressed on the surface of osteoclast precursors, to induce terminal differentiation of these cells to mature bony resorbing osteoclasts. Osteoprotegerin (OPG), a decoy receptor, inhibits the RANKL-RANK interaction by binding RANKL. In the presence of RANKL, osteoclasts also release semaphorin 4D (Sema4D) to inhibit osteogenesis. PTH may simultaneously have a direct effect on osteoblasts and osteoclasts through its receptor (PTH1R). The interactions between the cellular cytokines contribute to maintain the balance between bone formation and resorption.
The formation of hydrogen ions and bicarbonate by osteoclasts is catalyzed by carbonic anhydrase II from carbon dioxide (36). The chloride-bicarbonate (Cl−/HCO3−) exchange also plays a role in the intracellular acidification of osteoclasts (37). Previous studies have confirmed that PTH directly stimulates the acidification of osteoclasts (38,39). It has also been verified that PTH promotes the expression of carbonic anhydrase II in murine marrow cells (40). In this study, bPTH induced a reduction in osteoclast intracellular pH, which correlated with a decrease in fluorescence intensity. We hypothesized that the decrease in intracellular pH in osteoclasts was related to the carbonic anhydrase II or Cl−/HCO3− exchange, but not V-ATPase. Intracellular acidification may provide adequate hydrogen ions for V-ATPase to transfer to the extracellular microenvironment. We suggest that this change contributes to the bone resorption capability of osteoclasts once osteoclasts are activated by attaching to the bone surface.
The effect of PTH on osteoclasts has seldom been studied before, as the opinion that osteoclasts do not express PTHRs is widely accepted. In fact, PTHR expression in osteoclasts has yet to be confirmed (41–43). Langub et al (42) detected PTH1R mRNA by in situ hybridization in osteoclasts in sections of iliac crest biopsies. mRNA levels of PTH1R in patients with secondary hyperparathyroidism were higher than those in normal individuals (42). Immunostaining also revealed PTH1R protein in osteoclasts from diseased tissue (42). This suggests that PTH1R expression may be weak in normal situations and increased in pathological states. Conversely, due to its short half-life of approximately 4 min, PTH can be rapidly cleared in the blood (44). Indeed, as also previously demonstrated, cell-surface bound PTH was rapidly cleared in approximately 20 min when fluorescent-tagged PTH was introduced to cultures of osteoclasts (45). Rapid clearance contributes to the difficulty of localizing the receptor using conventional detection methods. In a previous study, multinucleated osteoclasts were obtained from longitudinally split animal long bones. However, despite using milder procedures, the isolated osteoclasts were damaged, as evidenced by their failure to exude trypan blue (46). With fragile cells, significant cellular disintegration prevents us from detecting membrane receptors. However, osteoclasts induced from BM-derived monocytes are generated from a different source and cultured under relatively stable conditions and can be stained in a very short time. This may improve the positive rate of the detection of PTHRs.
It should be noted that the results of this study were based on BM-derived osteoclasts, not primary osteoclasts derived from long bones of mice. Notwithstanding this limitation, this study suggests a dual regulatory mechanism, whereby PTH acts both directly on osteoblasts and also via osteoclasts. The identification of a new mode of cell-cell communication mediated by PTH and PTH1R has added another level of information to our knowledge of the regulatory balance between bone formation and resorption.
Acknowledgments
We would like to thank the physicians in the Department of Nephrology for their helpful comments.
References
- 1.Guerreiro PM, Renfro JL, Power DM, Canario AV. The parathyroid hormone family of peptides: structure, tissue distribution, regulation, and potential functional roles in calcium and phosphate balance in fish. Am J Physiol Regul Integr Comp Physiol. 2007;292:R679–R696. doi: 10.1152/ajpregu.00480.2006. [DOI] [PubMed] [Google Scholar]
- 2.On JS, Chow BK, Lee LT. Evolution of parathyroid hormone receptor family and their ligands in vertebrate. Front Endocrinol (Lausanne) 2015;6:28. doi: 10.3389/fendo.2015.00028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Dobolyi A, Palkovits M, Usdin TB. The TIP39-PTH2 receptor system: unique peptidergic cell groups in the brainstem and their interactions with central regulatory mechanisms. Prog Neurobiol. 2010;90:29–59. doi: 10.1016/j.pneurobio.2009.10.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Potthoff SA, Janus A, Hoch H, Frahnert M, Tossios P, Reber D, Giessing M, Klein HM, Schwertfeger E, Quack I, et al. PTH-receptors regulate norepinephrine release in human heart and kidney. Regul Pept. 2011;171:35–42. doi: 10.1016/j.regpep.2011.06.002. [DOI] [PubMed] [Google Scholar]
- 5.Pioszak AA, Xu HE. Molecular recognition of parathyroid hormone by its G protein-coupled receptor. Proc Natl Acad Sci USA. 2008;105:5034–5039. doi: 10.1073/pnas.0801027105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Abou-Samra AB, Jüppner H, Force T, Freeman MW, Kong XF, Schipani E, Urena P, Richards J, Bonventre JV, Potts JT., Jr Expression cloning of a common receptor for parathyroid hormone and parathyroid hormone-related peptide from rat osteoblast-like cells: a single receptor stimulates intracellular accumulation of both cAMP and inositol trisphosphates and increases intracellular free calcium. Proc Natl Acad Sci USA. 1992;89:2732–2736. doi: 10.1073/pnas.89.7.2732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Silva BC, Bilezikian JP. Parathyroid hormone: anabolic and catabolic actions on the skeleton. Curr Opin Pharmacol. 2015;22:41–50. doi: 10.1016/j.coph.2015.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lupp A, Klenk C, Röcken C, Evert M, Mawrin C, Schulz S. Immunohistochemical identification of the PTHR1 parathyroid hormone receptor in normal and neoplastic human tissues. Eur J Endocrinol. 2010;162:979–986. doi: 10.1530/EJE-09-0821. [DOI] [PubMed] [Google Scholar]
- 9.Romero G, Sneddon WB, Yang Y, Wheeler D, Blair HC, Friedman PA. Parathyroid hormone receptor directly interacts with dishevelled to regulate beta-Catenin signaling and osteoclastogenesis. J Biol Chem. 2010;285:14756–14763. doi: 10.1074/jbc.M110.102970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tohmonda T, Yoda M, Mizuochi H, Morioka H, Matsumoto M, Urano F, Toyama Y, Horiuchi K. The IRE1α-XBP1 pathway positively regulates parathyroid hormone (PTH)/PTH-related peptide receptor expression and is involved in pth-induced osteoclastogenesis. J Biol Chem. 2013;288:1691–1695. doi: 10.1074/jbc.C112.424606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Dempster DW, Hughes-Begos CE, Plavetic-Chee K, Brandao-Burch A, Cosman F, Nieves J, Neubort S, Lu SS, Iida-Klein A, Arnett T, Lindsay R. Normal human osteoclasts formed from peripheral blood monocytes express PTH type 1 receptors and are stimulated by PTH in the absence of osteoblasts. J Cell Biochem. 2005;95:139–148. doi: 10.1002/jcb.20388. [DOI] [PubMed] [Google Scholar]
- 12.Jilka RL. Are osteoblastic cells required for the control of osteoclast activity by parathyroid hormone? Bone Miner. 1986;1:261–266. [PubMed] [Google Scholar]
- 13.Del Fattore A, Teti A, Rucci N. Osteoclast receptors and signaling. Arch Biochem Biophys. 2008;473:147–160. doi: 10.1016/j.abb.2008.01.011. [DOI] [PubMed] [Google Scholar]
- 14.Ludwig J, Kerscher S, Brandt U, Pfeiffer K, Getlawi F, Apps DK, Schägger H. Identification and characterization of a novel 9.2-kDa membrane sector-associated protein of vacuolar proton-ATPase from chromaffin granules. J Biol Chem. 1998;273:10939–10947. doi: 10.1074/jbc.273.18.10939. [DOI] [PubMed] [Google Scholar]
- 15.Su Y, Zhou A, Al-Lamki RS, Karet FE. The a-subunit of the V-type H+-ATPase interacts with phosphofructokinase-1 in humans. J Biol Chem. 2003;278:20013–20018. doi: 10.1074/jbc.M210077200. [DOI] [PubMed] [Google Scholar]
- 16.Qin A, Cheng TS, Pavlos NJ, Lin Z, Dai KR, Zheng MH. V-ATPases in osteoclasts: structure, function and potential inhibitors of bone resorption. Int J Biochem Cell Biol. 2012;44:1422–1435. doi: 10.1016/j.biocel.2012.05.014. [DOI] [PubMed] [Google Scholar]
- 17.Toro EJ, Ostrov DA, Wronski TJ, Holliday LS. Rational identification of enoxacin as a novel V-ATPase-directed osteoclast inhibitor. Curr Protein Pept Sci. 2012;13:180–191. doi: 10.2174/138920312800493151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Drory O, Nelson N. The emerging structure of vacuolar ATPases. Physiology (Bethesda) 2006;21:317–325. doi: 10.1152/physiol.00017.2006. [DOI] [PubMed] [Google Scholar]
- 19.Manolson MF, Yu H, Chen W, Yao Y, Li K, Lees RL, Heersche JN. The a3 isoform of the 100-kDa V-ATPase subunit is highly but differentially expressed in large (≥10 nuclei) and small (≤nuclei) osteoclasts. J Biol Chem. 2003;278:49271–49278. doi: 10.1074/jbc.M309914200. [DOI] [PubMed] [Google Scholar]
- 20.Niikura K. Vacuolar ATPase as a drug discovery target. Drug News Perspect. 2006;19:139–144. doi: 10.1358/dnp.2006.19.3.977442. [DOI] [PubMed] [Google Scholar]
- 21.Nishi T, Kawasaki-Nishi S, Forgac M. Expression and function of the mouse V-ATPase d subunit isoforms. J Biol Chem. 2003;278:46396–46402. doi: 10.1074/jbc.M303924200. [DOI] [PubMed] [Google Scholar]
- 22.Wu H, Xu G, Li YP. Atp6v0d2 is an essential component of the osteoclast-specific proton pump that mediates extracellular acidification in bone resorption. J Bone Miner Res. 2009;24:871–885. doi: 10.1359/jbmr.081239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lee SH, Rho J, Jeong D, Sul JY, Kim T, Kim N, Kang JS, Miyamoto T, Suda T, Lee SK, et al. v-ATPase V0 subunit d2-deficient mice exhibit impaired osteoclast fusion and increased bone formation. Nat Med. 2006;12:1403–1409. doi: 10.1038/nm1514. [DOI] [PubMed] [Google Scholar]
- 24.Bell O, Silver J, Naveh-Many T. Parathyroid hormone, from gene to protein. In: Naveh-Many T, editor. Molecular Biology of the Parathyroid. Landes Bioscience and Kluwer Academic; New York: 2005. pp. 8–28. [DOI] [Google Scholar]
- 25.Huang JC, Sakata T, Pfleger LL, Bencsik M, Halloran BP, Bikle DD, Nissenson RA. PTH differentially regulates expression of RANKL and OPG. J Bone Miner Res. 2004;19:235–244. doi: 10.1359/JBMR.0301226. [DOI] [PubMed] [Google Scholar]
- 26.Lee SK, Lorenzo JA. Parathyroid hormone stimulates TRANCE and inhibits osteoprotegerin messenger ribonucleic acid expression in murine bone marrow cultures: correlation with osteoclast-like cell formation. Endocrinology. 1999;140:3552–3561. doi: 10.1210/endo.140.8.6887. [DOI] [PubMed] [Google Scholar]
- 27.Rhee Y, Bivi N, Farrow E, Lezcano V, Plotkin LI, White KE, Bellido T. Parathyroid hormone receptor signaling in osteocytes increases the expression of fibroblast growth factor-23 in vitro and in vivo. Bone. 2011;49:636–643. doi: 10.1016/j.bone.2011.06.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Lamothe B, Lai Y, Xie M, Schneider MD, Darnay BG. TAK1 is essential for osteoclast differentiation and is an important modulator of cell death by apoptosis and necroptosis. Mol Cell Biol. 2013;33:582–595. doi: 10.1128/MCB.01225-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Koizumi K, Ito Y, Kojima K, Fujii T. Isolation and characterization of the plasma membranes from rat ascites hepatomas and from normal rat livers, including newborn, regenerating, and adult livers. J Biochem. 1976;79:739–748. doi: 10.1093/oxfordjournals.jbchem.a131126. [DOI] [PubMed] [Google Scholar]
- 30.Wang YH, Liu Y, Buhl K, Rowe DW. Comparison of the action of transient and continuous PTH on primary osteoblast cultures expressing differentiation stage-specific GFP. J Bone Miner Res. 2005;20:5–14. doi: 10.1359/JBMR.041016. [DOI] [PubMed] [Google Scholar]
- 31.Udagawa N, Takahashi N, Akatsu T, Tanaka H, Sasaki T, Nishihara T, Koga T, Martin TJ, Suda T. Origin of osteoclasts: mature monocytes and macrophages are capable of differentiating into osteoclasts under a suitable microenvironment prepared by bone marrow-derived stromal cells. Proc Natl Acad Sci USA. 1990;87:7260–7264. doi: 10.1073/pnas.87.18.7260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Blair HC, Athanasou NA. Recent advances in osteoclast biology and pathological bone resorption. Histol Histopathol. 2004;19:189–199. doi: 10.14670/HH-19.189. [DOI] [PubMed] [Google Scholar]
- 33.Cannata-Andía JB, Rodriguez García M, Gómez Alonso C. Osteoporosis and adynamic bone in chronic kidney disease. J Nephrol. 2013;26:73–80. doi: 10.5301/jn.5000212. [DOI] [PubMed] [Google Scholar]
- 34.Zou W, Teitelbaum SL. Integrins, growth factors, and the osteoclast cytoskeleton. Ann N Y Acad Sci. 2010;1192:27–31. doi: 10.1111/j.1749-6632.2009.05245.x. [DOI] [PubMed] [Google Scholar]
- 35.Yuan FL, Li X, Lu WG, Li CW, Li JP, Wang Y. The vacuolar ATPase in bone cells: a potential therapeutic target in osteoporosis. Mol Biol Rep. 2010;37:3561–3566. doi: 10.1007/s11033-010-0004-7. [DOI] [PubMed] [Google Scholar]
- 36.Oksala N, Levula M, Pelto-Huikko M, Kytömäki L, Soini JT, Salenius J, Kähönen M, Karhunen PJ, Laaksonen R, Parkkila S, Lehtimäki T. Carbonic anhydrases II and XII are up-regulated in osteoclast-like cells in advanced human atherosclerotic plaques-Tampere Vascular Study. Ann Med. 2010;42:360–370. doi: 10.3109/07853890.2010.486408. [DOI] [PubMed] [Google Scholar]
- 37.Josephsen K, Praetorius J, Frische S, Gawenis LR, Kwon TH, Agre P, Nielsen S, Fejerskov O. Targeted disruption of the Cl−/HCO3− exchanger Ae2 results in osteopetrosis in mice. Proc Natl Acad Sci USA. 2009;106:1638–1641. doi: 10.1073/pnas.0811682106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Hunter SJ, Schraer H, Gay CV. Characterization of isolated and cultured chick osteoclasts: the effects of acetazolamide, calcitonin, and parathyroid hormone on acid production. J Bone Miner Res. 1988;3:297–303. doi: 10.1002/jbmr.5650030308. [DOI] [PubMed] [Google Scholar]
- 39.Gay CV, Kief NL, Bekker PJ. Effect of estrogen on acidification in osteoclasts. Biochem Biophys Res Commun. 1993;192:1251–1259. doi: 10.1006/bbrc.1993.1551. [DOI] [PubMed] [Google Scholar]
- 40.Wang BL, Liang H, Zheng F, Li XX, Liu YB, Dai CL. Recombinant soluble receptor activator of nuclear factor-kappaB inhibits parathyroid hormone-induced osteoclastogenesis in vitro. Sheng Li Xue Bao. 2007;59:169–174. [PubMed] [Google Scholar]
- 41.Gay CV, Zheng B, Gilman VR. Co-detection of PTH/PTHrP receptor and tartrate resistant acid phosphatase in osteoclasts. J Cell Biochem. 2003;89:902–908. doi: 10.1002/jcb.10579. [DOI] [PubMed] [Google Scholar]
- 42.Langub MC, Monier-Faugere MC, Qi Q, Geng Z, Koszewski NJ, Malluche HH. Parathyroid hormone/parathyroid hormone-related peptide type 1 receptor in human bone. J Bone Miner Res. 2001;16:448–456. doi: 10.1359/jbmr.2001.16.3.448. [DOI] [PubMed] [Google Scholar]
- 43.Faucheux C, Horton MA, Price JS. Nuclear localization of type I parathyroid hormone/parathyroid hormone-related protein receptors in deer antler osteoclasts: evidence for parathyroid hormone-related protein and receptor activator of NF-kappaB-dependent effects on osteoclast formation in regenerating mammalian bone. J Bone Miner Res. 2002;17:455–464. doi: 10.1359/jbmr.2002.17.3.455. [DOI] [PubMed] [Google Scholar]
- 44.Libutti SK, Alexander HR, Bartlett DL, Sampson ML, Ruddel ME, Skarulis M, Marx SJ, Spiegel AM, Simmonds W, Remaley AT. Kinetic analysis of the rapid intraoperative parathyroid hormone assay in patients during operation for hyperparathyroidism. Surgery. 1999;126:1145–1150. doi: 10.1067/msy.2099.101835. discussion 1150–1151. [DOI] [PubMed] [Google Scholar]
- 45.Niendorf A, Dietel M, Arps H, Childs GV. A novel method to demonstrate parathyroid hormone binding on unfixed living target cells in culture. J Histochem Cytochem. 1988;36:307–309. doi: 10.1177/36.3.2830332. [DOI] [PubMed] [Google Scholar]
- 46.Hefley TJ, Stern PH. Isolation of osteoclasts from fetal rat long bones. Calcif Tissue Int. 1982;34:480–487. doi: 10.1007/BF02411289. [DOI] [PubMed] [Google Scholar]