Abstract
AIM: To investigate the expression of NF-κBp65 protein and human telomerase reverse transcriptase (hTERT) and their correlation in gastric cancer and precancerous lesions.
METHODS: Forty-one patients with primary gastric cancer, 15 with dysplasia, 23 intestinal metaplasia and 10 with normal gastric mucosa were included in this study. Expression of NF-κBp65 protein, hTERT mRNA and protein were determined by immunohistochemistry and in situ hybridization.
RESULTS: The rate of p65 expression in normal gastric mucosa, intestinal metaplasia, dysplasia and carcinoma was 0%, 34.78%, 53.33% and 60.98%, respectively, while the rate of hTERT mRNA expression was 10.00%, 39.13%, 66.67% and 85.37% and the rate of hTERT protein expression was 0%, 30.43%, 60.00% and 78.05%, respectively. All the three parameters were significantly increased in dysplasia and carcinoma compared to normal mucosa, while the expression levels were also significantly higher in carcinoma than in intestinal metaplasia (P < 0.05). In gastric cancer tissues, nuclear staining rates of p65 and hTERT protein were both significantly associated with the degree of differentiation, lymph node metastasis, clinical stage and invasion depth (P < 0.05). However, hTERT mRNA expression was only significantly associated with clinical stage. There was a positive correlation between p65 and hTERT mRNA (rs = 0.661 - 0.752, P < 0.01), and between hTERT protein and hTERT mRNA (rs = 0.609-0.750, P < 0.01).
CONCLUSION: NF-κBp65 and hTERT expressions are upregulated at the early stage of gastric carcinogenesis. NF-κB activation may contribute to hTERT expression and thereby enhance telomerase activity, which represents an important step in carcinogenesis progress.
Wang W, Luo HS, Yu BP. Expression of NF-κB and human telomerase reverse transcriptase in gastric cancer and precancerous lesions.
INTRODUCTION
NF-κB is a family of dimeric transcription factors that play a critical role in host defense by regulating the expression of immune and inflammatory genes[1-3]. The most common dimer is RelA (p65)/NF-κB1 (p50) heterodimer. In resting cells, NF-κB is localized in the cytoplasm, which is noncovalently associated with the cytoplasmic inhibitory protein IκB. Upon stimulation with a variety of pathogenic inducers such as viruses, mitogens, bacteria, agents providing oxygen radicals, and inflammatory cytokines, IκB is phosphorylated, ubiquitinated, and degraded in the cytoplasm, and the NF-κB complex migrates into the nucleus and binds to DNA recognition sites in the regulatory regions of target genes[4]. Recently, there were several reports on the role of NF-κB gene products in cell proliferation, transformation, and tumor development[5-9]. Recent studies have also indicated that NF-κB is constitutively activated in several tumors[10-13]. However, the biological significance of NF-κB activation remains unclear in gastric carcinogenesis although gastric cancer is one of the most aggressive forms of cancer.
Telomerase is a key enzyme that catalyzes the synthesis of telomere DNA participating in cell immortalization through stabilization of chromosomal structure[14]. Telomerase is expressed in germ tissues as well as in majority of human tumors, including gastrointestinal carcinomas, but is low and difficult to detect in somatic cells generally[15-19]. So it may be a useful molecular marker for cancer diagnosis and therapeutic strategies. Human telomerase reverse transcriptase (hTERT) has been identified as a putative catalytic subunit of human telomerase[20]. Recent reconstitution experiments, both in vitro and in vivo, also strongly suggest that hTERT is the major determinant of human telomerase activity[21,22]. Overexpression of hTERT in cancer cells is thought to contribute to tumor development and angiogenesis[23-25]. However, the mechanism by which hTERT is overexpressed in cancer cells remains unclear. There are evidences that hTERT expression may be regulated by the highly inducible NF-κB transcription factor[26]. So it is interesting to investigate the relation between NF-κB and hTERT at cellular level.
We undertook the present study to determine whether NF-κB was constitutively activated in precancerous and cancerous tissues of the stomach, to examine whether expression of hTERT gene correlated with NF-κB activation, and to evaluate the relationship between clinicopathological features and NF-κB activation as well as hTERT gene expression.
MATERIALS AND METHODS
Tissue samples
Gastric tissues were obtained by surgical resection from 89 patients: 10 with normal gastric mucosa, 23 with intestinal metaplasia, 15 with dysplasia and 41 with gastric cancer. Patients with gastric cancer were admitted to Renmin Hospital of Wuhan University from February to December in 2000, and had not been treated with chemotherapy or radiation therapy before operation. Histological examinations were performed, according to the criteria of the Japanese Gastric Cancer Association[27]. All specimens were fixed in 10 % buffered neutral formalin and embedded in paraffin. Serial tissue sections (4.5 μm thick) were placed on glass slides coated with 3-aminopropyltriethoxysilane (Sigma, USA), and then used in the following experiments.
Immunohistochemistry for NF-κBp65 and hTERT proteins
Immunostaining was performed as described previously with slight modification[28]. Briefly, slides were treated overnight at 4 °C with either anti-p65 mAb (4 μg/ml) or anti-hTERT polyclonal antibody (3 μg/ml). Both antibodies were from Santa Cruz Biotechnology (USA). Then, slides were incubated with secondary antibody by using a streptavidin-peroxidase kit (Maixin-Bio, Fujian). Finally, the antigen sites were visualized by incubating with diaminobendizine (DAB) solution (Zhongshan Biotechnical Co, Beijing), and the nuclei were weakly counterstained with Mayer’s hematoxylin (Maixin-Bio, Fujian). The specificity of immunostaining was determined by replacement of the primary antibody with PBS. Positive reaction was detected as nuclear stain presenting in brown-yellow color. Slides positively stained were further stratified from 1+ to 3+ based on the overall intensity and percentage of the stained tumor cells, with an estimated scale of < 25% cell positive = 1+, 25% to 50% = 2+, and > 50% = 3+. Staining was defined as negative if positive cells were < 5%.
In situ hybridization for hTERT mRNA
hTERT mRNA ISH detection kit and antisense polyoligonucleotide probe (digoxin-labeled) were purchased from Boster Biological Technology Ltd. (Wuhan). In brief, deparaffinized sections were incubated with 3% hydrogen peroxide for 30 min and then with 1 μg/ml pepsin for 15 min. The prehybridization was performed at 37 °C for 2 h, and the hybridization was conducted in a 42 °C water bath for 18 h with each section covered with a soil coverslip. After thorough washing, tissue sections were preblocked for 20 min with blocking solution. Then, rabbit anti-digoxin antibody was added for 60 min at 37 °C. After washed in PBS, the sections were visualized according to the manufacturer’s instructions. A negative control was prepared by using a hybridization solution without the probe. A positive reaction was detected as plasmatic stain presenting in brown-yellow color. Slides positively stained were further stratified from 1+ to 3+ as described above.
Statistics
Statistical analysis was performed using chi-squared test, Fisher’s exact test, and spearman rank test. A P value < 0.05 (one-sided) was accepted as statistically significant.
RESULTS
Staining of NF-κBp65 protein, hTERT mRNA and protein
P65 immunostaining was significantly enhanced both in cytoplasm and in nuclei of the tumor cells in comparison to that in normal epithelial cells (Figure 1). Since nuclear staining, which indicated nuclear transportation of p65, was considered as a marker of NF-κB activation, we only counted the number of nuclear stained cells and took them into calculation of the percentage of positively stained cells. Similarly, hTERT protein was mainly localized in the nuclei of tumor cells, and only a small number of cells showed a positive reaction in the cytoplasm (Figure 2). Stromal cells such as endothelial cells and smooth muscle cells (except lymphocytes) showed no reaction for hTERT. Weak staining ofp65 and hTERT proteins was observed in some epithelial cells in the lower two-thirds of the glands of nonneoplastic mucosa.
Figure 1.
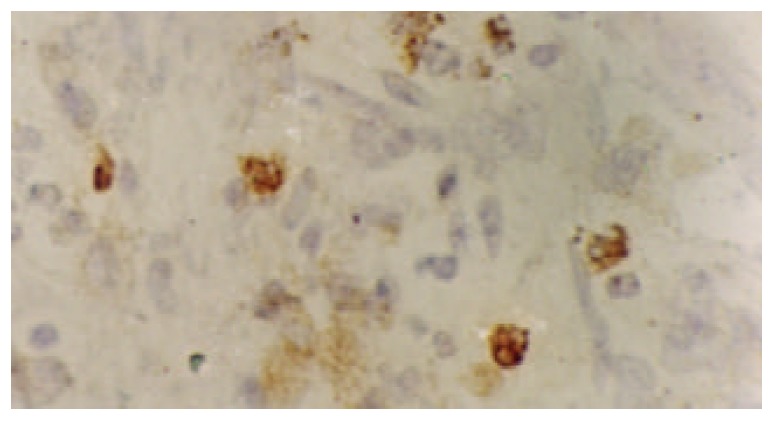
Immunohistochemical detection of NF-κBp65 protein in gastric carcinoma showing cytoplasmic and nuclear staining SP×400.
Figure 2.
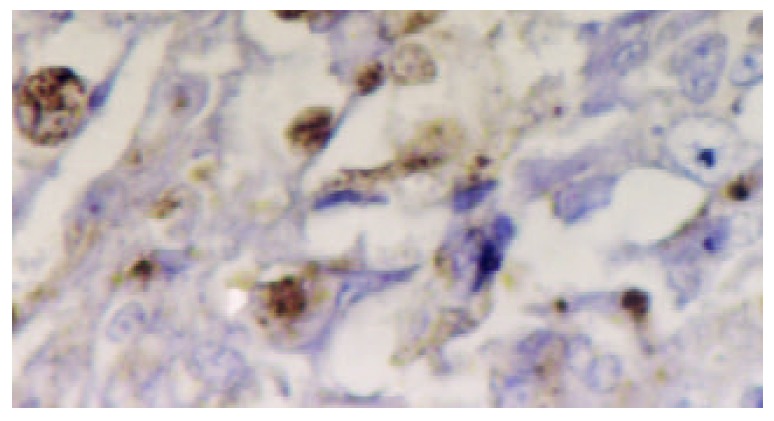
Immunohistochemical detection of hTERT protein in poorly differentiated gastric carcinoma. A positive reaction was shown in the nuclei of cancer cells. SP×400.
In situ hybridization
Revealed that hTERT mRNA was significantly enhanced in the cytoplasm of tumor cells (Figure 3). Only a few signals were seen in nonneoplastic cells, which were slightly increased in the replicating basal layer, and in intestinal metaplastic cells as well as in activated lymphocytes, while the surface epithelia were negative (Figure 4).
Figure 3.
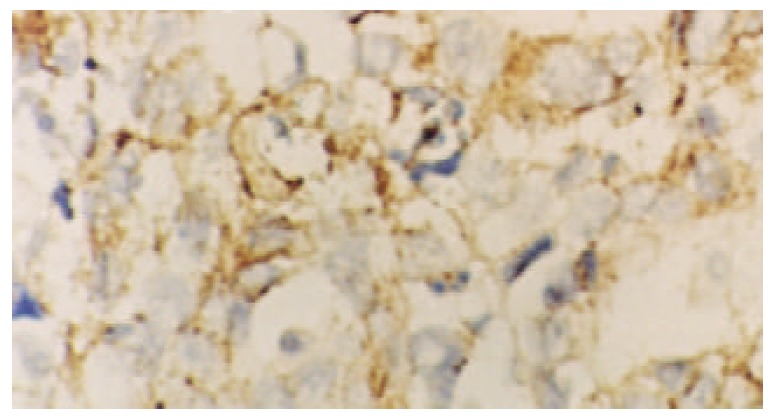
Detection of hTERT mRNA in a mucinous adenocarcinoma. Most tumor cells displayed strong signals that were localized in the cytoplasm. ISH×400.
Figure 4.
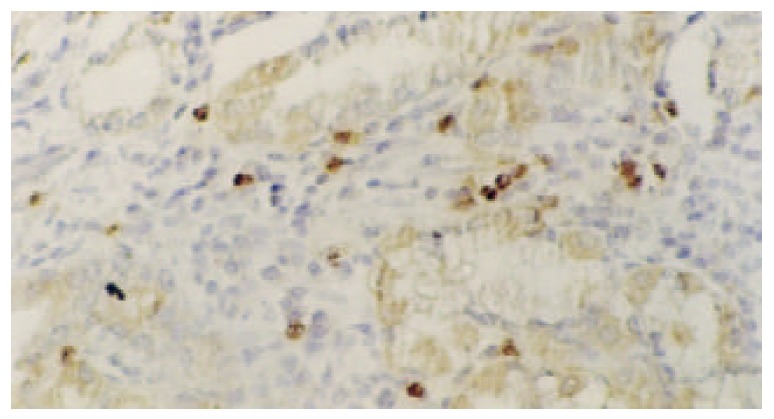
Detection of hTERT mRNA in intestinal metaplasia. The reaction was localized in the cytoplasm of intestinal meta-plastic cells, activated lymphocytes, as well as replicating cells in basal layer, while the surface epithelia were negative. ISH×100.
Expression of NF-κBp65 protein, hTERT mRNA and protein
Expression of p65 protein, hTERT mRNA and protein in gastric precancerous and cancerous tissues was increased compared with normal mucosa (Table 1). The nuclear staining rates of p65 (active form) were higher in gastric cancer than in intestinal metaplasia (P < 0.05), or normal mucosa (P < 0.05). The rates were also significantly higher in dysplasia than in normal mucosa. Similarly, expression of hTERT mRNA and protein was increased in precancerous and cancerous tissues (Table 1).
Table 1.
Expression of NF-κBp65 protein, hTERT mRNA and protein in precancerous lesions and gastric cancer
| Lesion | Cases |
NF-κBp65 |
hTERT mRNA |
hTERT protein |
|||||||||
| + | ++ | +++ | Total (%) | + | ++ | +++ | Total (%) | + | ++ | +++ | Total (%) | ||
| Normal | 10 | 0 | 0 | 0 | 0 (0) | 1 | 0 | 0 | 1 (10.00) | 0 | 0 | 0 | 0 (0) |
| Intestinal metaplasia | 23 | 7 | 1 | 0 | 8 (34.78)b | 6 | 3 | 0 | 9 (39.13)b | 5 | 2 | 0 | 7 (30.43)b |
| Dysplasia | 15 | 7 | 1 | 0 | 8 (53.33)a | 7 | 3 | 0 | 10 (66.67)a | 7 | 2 | 0 | 9 (60.00)a |
| Cancer | 41 | 7 | 14 | 4 | 25 (60.98)a | 6 | 20 | 9 | 35 (85.37)a | 6 | 20 | 6 | 32 (78.05)a |
P < 0.05, vs normal mucosa group;
P < 0.01, vs cancer group.
The relations between clinicopathological features and expression of p65 and hTERT in the 41 patients with gastric carcinoma were further analyzed (Table 2). Nuclear staining rates of p65 and hTERT proteins were both significantly associated with the degree of differentiation, lymph node metastasis, clinical stage and the depth of invasion (P < 0.01-0.05). Furthermore, the nuclear staining of p65 was significantly higher in tumors ≥ 5 cm than in those < 5 cm. With respect to expression of hTERT mRNA, we found that carcinomas at advanced stage showed a significantly higher level than those at early stage. However, there was no significant association between expression of hTERT mRNA and other clinicopathological features.
Table 2.
Correlation between expression of NF-κBP65 protein, hTERT mRNA and protein and clinicopathological features
| Factor | Cases | NF-κB | hTERT | hTERT |
| P65 (%) | mRNA (%) | protein (%) | ||
| Gender | ||||
| Male | 30 | 16 (53.33) | 24 (80.00) | 24 (80.00) |
| Female | 11 | 9 (81.82) | 11 (100.00) | 8 (72.73) |
| Age (years) | ||||
| < 60 | 24 | 14 (58.33) | 21 (87.50) | 20 (83.33) |
| ≥ 60 | 17 | 11 (64.71) | 14 (82.35) | 12 (70.59) |
| Histology (type) | ||||
| Intestinal | 16 | 7 (43.75) | 12 (75.00) | 10 (62.50) |
| Differentiation | 25 | 18 (72.00) | 23 (92.00) | 22 (88.00) |
| Differentiation | ||||
| Well | 13 | 3 (23.08)b | 9 (69.23) | 7 (53.85)a |
| Poor | 28 | 22 (78.57) | 26 (92.86) | 25 (89.29) |
| Lymph node metastasis | ||||
| Negative | 16 | 4 (25.00)b | 12 (75.00) | 9 (56.25)a |
| Positive | 25 | 21 (84.00) | 23 (92.00) | 23 (92.00) |
| Stage | ||||
| Early | 12 | 2 (16.67)b | 7 (58.33)b | 5 (41.67)b |
| Advanced | 29 | 23 (79.31) | 28 (96.55) | 27 (93.10) |
| Tumor size (cm) | ||||
| < 5 | 19 | 6 (31.58)b | 14 (73.68) | 13 (68.42) |
| ≥ 5 | 22 | 19 (86.36) | 21 (95.45) | 19 (86.36) |
| Depth of invasion | ||||
| m, sm | 10 | 2 (20.00)a | 6 (60.00) | 4 (40.00)a |
| ms, ss | 17 | 11 (64.71) | 16 (94.12) | 15 (88.24) |
| se, si | 14 | 12 (85.71) | 13 (92.86) | 13 (92.86) |
P < 0.05,
P < 0.01, From comparison in each group of clinicopatho-logic feature by chi-square test or Fisher’s exact test. m: mucosa, sm: submucosa, mp: muscularis propria, ss: subserosa, se: inva-sion to serosa, si: invasion to other organ. Tumor size was de-fined as the largest size in extension on the gastric mucosa.
Correlation between NF-κB activation and expression of hTERT
There was a positive correlation between NF-κB activation and hTERT mRNA expression in patients with intestinal metaplasia (rs = 0.665), dysplasia (rs = 0.661) and gastric cancer (rs = 0.752). Similarly, hTERT mRNA expression was also positively correlated with hTERT protein in these three groups (P < 0.05), with the rs values of 0.609, 0.750 and 0.730, respectively.
DISCUSSION
The present study reported the detection of NF-κB activation and hTERT gene expression, as well as their correlation, in cancerous and precancerous tissues of stomach.
NF-κB, an important transcription factor, consists of dimeric complexes. We used immunohistochemistry to detect NF-κB activation. The polyclonal antibody we used, could bind to the active and inactive forms of p65, one of NF-κB subunits, as shown in a previous study[29]. We only quantified the nuclear staining (active form) to evaluate NF-κB activation. With such an analysis, nuclear translocation of p65 could be estimated at single-cell level. We found that active NF-κB was not only in the nuclei of tumor cells but also in the nuclei of infiltrating inflammatory cells, especially lymphocytes, which was accordant with the role of NF-κB involved in immune and inflammatory responses[30,31]. Isomoto et al[32] also reported that in Helicobacter pylori-associated gastritis, activated NF-κB was expressed in macrophages, vascular endothelial cells and B lymphocytes in addition to epithelial cells, which might be involved in the inflammation process.
Using in situ hybridization and immunohistochemistry methods, we detected the expression of hTERT at both RNA and protein levels. Importantly, the in situ detection enabled us to differentiate whether telomerase activity was due to proliferative normal cells or lymphocytes. Therefore immunohistochemical detection of hTERT might be a novel tool for the diagnosis of gastric cancer[33]. In this study, we demonstrated that, besides tumor cells, hTERT mRNA and protein were also expressed, albeit weakly in normal gastric fundic mucosa. These epithelial cells were terminally differentiated. It is possible that low levels of hTERT expression might be the characteristics of physically regenerating tissues containing stem cells, and hTERT-positive cells might be competent for regeneration if severe mucosal damage occurred[28]. Although the level of hTERT mRNA correlated significantly with that of hTERT protein in our study, their expression levels were not always coincident in precancerous and cancerous lesions. It is likely that post-translational modifications, such as phosphorylation by akt kinase, might be involved in hTERT expression[28,34].
The present study revealed that NF-κBp65 nuclear staining rates were higher in cancer tissues, followed by dysplasia, intestinal metaplasia and normal mucosa. This is the first report on NF-κBp65 detection in gastric precancerous tissues. As to the expression of hTERT gene, previous studies revealed that carcinomas could express hTERT more frequently than noncancerous tissues[25,35-38]. Our findings support these results. The present study indicated that NF-κB and telomerase were both activated in precancerous lesions, suggesting that NF-κB activation and increased hTERT gene expression emerge at the early stage of gastric carcinogenesis, and that they may be the prerequisites for malignant transformation. Therefore, NF-κB and telomerase might be potentially important targets for the development of anti-tumor therapies for gastric carcinoma[15,39,40].
Sasaki et al[29] reported that NF-κB activation was positively correlated with tumor size, lymphatic invasion, depth of invasion and peritoneal metastasis. Besides that, our study also revealed significantly positive correlations between nuclear staining of NF-κBp65 and differentiation degree and clinical stage. Therefore, NF-κB activation might be associated with tumor growth, invasion and metastasis.
It has been reported that hTERT expression might be downstream of NF-κB activation[26,41]. Yin et al[26] found a potential NF-κB binding site at 350 base pairs upstream from the translational start site of mouse TERT promoters and NF-κB was found to contribute to the activation of TERT expression. In addition, Akiyama et al[41] showed that NF-κB interacted directly with hTERT protein in multiple myeloma cells. In the present study we examined the relationship between nuclear staining of NF-κBp65 and hTERT mRNA at the cellular level by immunohistochemistry and in situ hybridization. We observed that both p65 and hTERT mRNA were overexpressed in gastric cancer with the positive staining rates of 60.98% and 85.37%, respectively. In fact, most specimens showing a high p65 level also showed high hTERT mRNA expression and vice versa. Furthermore, the expression of p65 and hTERT mRNA showed a significant correlation in cancerous and precancerous tissues (intestinal metaplasia and dysplasia). And in nonneoplastic mucosa, the positive staining of p65 and hTERT mRNA were detected simultaneously in proliferative glands and activated lymphocytes. All of these suggested that NF-κBp65 cooperated with hTERT in gastric carcinogenesis. We speculate that NF-κBp65 may induce hTERT promoter activity and upregulate telomerase activity and contribute to the aggressiveness of gastric carcinoma.
However, our study was limited by the high specificity of p65 mAb for the p65 subunit, and thus we could not evaluate other NF-κB dimers. The relationship between other NF-κB subunits and gastric cancer needs further studies.
In conclusion, we demonstrated the presence of activated NF-κBp65 and increased hTERT expression in gastric cancerous and precancerous tissues. The levels of p65 and hTERT expression were higher in gastric cancer than in normal mucosa, and NF-κB activation was associated with tumor growth, invasion and metastasis. Our findings suggest that NF-κBp65 cooperates with hTERT in gastric carcinogenesis.
Footnotes
Edited by Xia HHX and Wang XL
References
- 1.Baeuerle PA, Baltimore D. NF-kappa B: ten years after. Cell. 1996;87:13–20. doi: 10.1016/s0092-8674(00)81318-5. [DOI] [PubMed] [Google Scholar]
- 2.Jia CK, Zheng SS, Li QY, Zhang AB. Immunotolerance of liver allotransplantation induced by intrathymic inoculation of donor soluble liver specific antigen. World J Gastroenterol. 2003;9:759–764. doi: 10.3748/wjg.v9.i4.759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Gong JP, Liu CA, Wu CX, Li SW, Shi YJ, Li XH. Nuclear factor kB activity in patients with acute severe cholangitis. World J Gastroenterol. 2002;8:346–349. doi: 10.3748/wjg.v8.i2.346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Thanos D, Maniatis T. NF-kappa B: a lesson in family values. Cell. 1995;80:529–532. doi: 10.1016/0092-8674(95)90506-5. [DOI] [PubMed] [Google Scholar]
- 5.Pahl HL. Activators and target genes of Rel/NF-kappaB transcription factors. Oncogene. 1999;18:6853–6866. doi: 10.1038/sj.onc.1203239. [DOI] [PubMed] [Google Scholar]
- 6.de Martin R, Schmid JA, Hofer-Warbinek R. The NF-kappaB/Rel family of transcription factors in oncogenic transformation and apoptosis. Mutat Res. 1999;437:231–243. doi: 10.1016/s1383-5742(99)00089-7. [DOI] [PubMed] [Google Scholar]
- 7.Li HL, Chen DD, Li XH, Zhang HW, Lu YQ, Ye CL, Ren XD. Changes of NF-kB, p53, Bcl-2 and caspase in apoptosis induced by JTE-522 in human gastric adenocarcinoma cell line AGS cells: role of reactive oxygen species. World J Gastroenterol. 2002;8:431–435. doi: 10.3748/wjg.v8.i3.431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.La Rosa FA, Pierce JW, Sonenshein GE. Differential regulation of the c-myc oncogene promoter by the NF-kappa B rel family of transcription factors. Mol Cell Biol. 1994;14:1039–1044. doi: 10.1128/mcb.14.2.1039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sonenshein GE. Rel/NF-kappa B transcription factors and the control of apoptosis. Semin Cancer Biol. 1997;8:113–119. doi: 10.1006/scbi.1997.0062. [DOI] [PubMed] [Google Scholar]
- 10.Zhu JW, Yu BM, Ji YB, Zheng MH, Li DH. Upregulation of vascular endothelial growth factor by hydrogen peroxide in human colon cancer. World J Gastroenterol. 2002;8:153–157. doi: 10.3748/wjg.v8.i1.153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wang W, Abbruzzese JL, Evans DB, Larry L, Cleary KR, Chiao PJ. The nuclear factor-kappa B RelA transcription factor is constitutively activated in human pancreatic adenocarcinoma cells. Clin Cancer Res. 1999;5:119–127. [PubMed] [Google Scholar]
- 12.Sovak MA, Bellas RE, Kim DW, Zanieski GJ, Rogers AE, Traish AM, Sonenshein GE. Aberrant nuclear factor-kappaB/Rel expression and the pathogenesis of breast cancer. J Clin Invest. 1997;100:2952–2960. doi: 10.1172/JCI119848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Guo SP, Wang WL, Zhai YQ, Zhao YL. Expression of nuclear factor-kappa B in hepatocellular carcinoma and its relation with the X protein of hepatitis B virus. World J Gastroenterol. 2001;7:340–344. doi: 10.3748/wjg.v7.i3.340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Blackburn EH. Telomerases. Annu Rev Biochem. 1992;61:113–129. doi: 10.1146/annurev.bi.61.070192.000553. [DOI] [PubMed] [Google Scholar]
- 15.Burger AM, Bibby MC, Double JA. Telomerase activity in normal and malignant mammalian tissues: feasibility of telomerase as a target for cancer chemotherapy. Br J Cancer. 1997;75:516–522. doi: 10.1038/bjc.1997.90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lu JP, Mao JQ, Li MS, Lu SL, Hu XQ, Zhu SN, Nomura S. In situ detection of TGF betas, TGF beta receptor II mRNA and telomerase activity in rat cholangiocarcinogenesis. World J Gastroenterol. 2003;9:590–594. doi: 10.3748/wjg.v9.i3.590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Liao C, Zhao MJ, Zhao J, Song H, Pineau P, Marchio A, Dejean A, Tiollais P, Wang HY, Li TP. Mutation analysis of novel human liver-related putative tumor suppressor gene in hepatocellular carcinoma. World J Gastroenterol. 2003;9:89–93. doi: 10.3748/wjg.v9.i1.89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Shen ZY, Xu LY, Chen MH, Shen J, Cai WJ, Zeng Y. Progressive transformation of immortalized esophageal epithelial cells. World J Gastroenterol. 2002;8:976–981. doi: 10.3748/wjg.v8.i6.976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Feng RH, Zhu ZG, Li JF, Liu BY, Yan M, Yin HR, Lin YZ. Inhibition of human telomerase in MKN-45 cell line by antisense hTR expression vector induces cell apoptosis and growth arrest. World J Gastroenterol. 2002;8:436–440. doi: 10.3748/wjg.v8.i3.436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Meyerson M, Counter CM, Eaton EN, Ellisen LW, Steiner P, Caddle SD, Ziaugra L, Beijersbergen RL, Davidoff MJ, Liu Q, et al. hEST2, the putative human telomerase catalytic subunit gene, is up-regulated in tumor cells and during immortalization. Cell. 1997;90:785–795. doi: 10.1016/s0092-8674(00)80538-3. [DOI] [PubMed] [Google Scholar]
- 21.Weinrich SL, Pruzan R, Ma L, Ouellette M, Tesmer VM, Holt SE, Bodnar AG, Lichtsteiner S, Kim NW, Trager JB, et al. Reconstitution of human telomerase with the template RNA component hTR and the catalytic protein subunit hTRT. Nat Genet. 1997;17:498–502. doi: 10.1038/ng1297-498. [DOI] [PubMed] [Google Scholar]
- 22.Nakayama J, Tahara H, Tahara E, Saito M, Ito K, Nakamura H, Nakanishi T, Tahara E, Ide T, Ishikawa F. Telomerase activation by hTRT in human normal fibroblasts and hepatocellular carcinomas. Nat Genet. 1998;18:65–68. doi: 10.1038/ng0198-65. [DOI] [PubMed] [Google Scholar]
- 23.Shao JC, Wu JF, Wang DB, Qin R, Zhang H. Relationship between the expression of human telomerase reverse transcriptase gene and cell cycle regulators in gastric cancer and its significance. World J Gastroenterol. 2003;9:427–431. doi: 10.3748/wjg.v9.i3.427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Pallini R, Pierconti F, Falchetti ML, D'Arcangelo D, Fernandez E, Maira G, D'Ambrosio E, Larocca LM. Evidence for telomerase involvement in the angiogenesis of astrocytic tumors: expression of human telomerase reverse transcriptase messenger RNA by vascular endothelial cells. J Neurosurg. 2001;94:961–971. doi: 10.3171/jns.2001.94.6.0961. [DOI] [PubMed] [Google Scholar]
- 25.Kumaki F, Kawai T, Hiroi S, Shinomiya N, Ozeki Y, Ferrans VJ, Torikata C. Telomerase activity and expression of human telomerase RNA component and human telomerase reverse transcriptase in lung carcinomas. Hum Pathol. 2001;32:188–195. doi: 10.1053/hupa.2001.21567. [DOI] [PubMed] [Google Scholar]
- 26.Yin L, Hubbard AK, Giardina C. NF-kappa B regulates transcription of the mouse telomerase catalytic subunit. J Biol Chem. 2000;275:36671–36675. doi: 10.1074/jbc.M007378200. [DOI] [PubMed] [Google Scholar]
- 27.Japanese Gastric Cancer Association. Japanese Classification of Gastric Carcinoma - 2nd English Edition - Gastric Cancer. 1998;1:10–24. doi: 10.1007/s101209800016. [DOI] [PubMed] [Google Scholar]
- 28.Yasui W, Tahara E, Tahara H, Fujimoto J, Naka K, Nakayama J, Ishikawa F, Ide T, Tahara E. Immunohistochemical detection of human telomerase reverse transcriptase in normal mucosa and precancerous lesions of the stomach. Jpn J Cancer Res. 1999;90:589–595. doi: 10.1111/j.1349-7006.1999.tb00787.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sasaki N, Morisaki T, Hashizume K, Yao T, Tsuneyoshi M, Noshiro H, Nakamura K, Yamanaka T, Uchiyama A, Tanaka M, et al. Nuclear factor-kappaB p65 (RelA) transcription factor is constitutively activated in human gastric carcinoma tissue. Clin Cancer Res. 2001;7:4136–4142. [PubMed] [Google Scholar]
- 30.Cong B, Li SJ, Yao YX, Zhu GJ, Ling YL. Effect of cholecystokinin octapeptide on tumor necrosis factor alpha transcription and nuclear factor-kappaB activity induced by lipopolysaccharide in rat pulmonary interstitial macrophages. World J Gastroenterol. 2002;8:718–723. doi: 10.3748/wjg.v8.i4.718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Meng AH, Ling YL, Zhang XP, Zhang JL. Anti-inflammatory effect of cholecystokinin and its signal transduction mechanism in endotoxic shock rat. World J Gastroenterol. 2002;8:712–717. doi: 10.3748/wjg.v8.i4.712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Isomoto H, Mizuta Y, Miyazaki M, Takeshima F, Omagari K, Murase K, Nishiyama T, Inoue K, Murata I, Kohno S. Implication of NF-kappaB in Helicobacter pylori-associated gastritis. Am J Gastroenterol. 2000;95:2768–2776. doi: 10.1111/j.1572-0241.2000.02304.x. [DOI] [PubMed] [Google Scholar]
- 33.Yasui W, Tahara H, Tahara E, Fujimoto J, Nakayama J, Ishikawa F, Ide T, Tahara E. Expression of telomerase catalytic component, telomerase reverse transcriptase, in human gastric carcinomas. Jpn J Cancer Res. 1998;89:1099–1103. doi: 10.1111/j.1349-7006.1998.tb00502.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kang SS, Kwon T, Kwon DY, Do SI. Akt protein kinase enhances human telomerase activity through phosphorylation of telomerase reverse transcriptase subunit. J Biol Chem. 1999;274:13085–13090. doi: 10.1074/jbc.274.19.13085. [DOI] [PubMed] [Google Scholar]
- 35.Lan J, Xiong YY, Lin YX, Wang BC, Gong LL, Xu HS, Guo GS. Helicobacter pylori infection generated gastric cancer through p53-Rb tumor-suppressor system mutation and telomerase reactivation. World J Gastroenterol. 2003;9:54–58. doi: 10.3748/wjg.v9.i1.54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Yao XX, Yin L, Sun ZC. The expression of hTERT mRNA and cellular immunity in gastric cancer and precancerosis. World J Gastroenterol. 2002;8:586–590. doi: 10.3748/wjg.v8.i4.586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kammori M, Kanauchi H, Nakamura K, Kawahara M, Weber TK, Mafune K, Kaminishi M, Takubo K. Demonstration of human telomerase reverse transcriptase in human colorectal carcinomas by in situ hybridization. Int J Oncol. 2002;20:15–21. [PubMed] [Google Scholar]
- 38.Jong HS, Park YI, Kim S, Sohn JH, Kang SH, Song SH, Bang YJ, Kim NK. Up-regulation of human telomerase catalytic subunit during gastric carcinogenesis. Cancer. 1999;86:559–565. [PubMed] [Google Scholar]
- 39.Ueda M, Kokura S, Imamoto E, Naito Y, Handa O, Takagi T, Yoshida N, Yoshikawa T. Blocking of NF-kappaB activation en-hances the tumor necrosis factor alpha-induced apoptosis of a human gastric cancer cell line. Cancer Lett. 2003;193:177–182. doi: 10.1016/s0304-3835(03)00002-8. [DOI] [PubMed] [Google Scholar]
- 40.Akari H, Bour S, Kao S, Adachi A, Strebel K. The human immunodeficiency virus type 1 accessory protein Vpu induces apoptosis by suppressing the nuclear factor kappaB-dependent expression of antiapoptotic factors. J Exp Med. 2001;194:1299–1311. doi: 10.1084/jem.194.9.1299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Akiyama M, Hideshima T, Hayashi T, Tai YT, Mitsiades CS, Mitsiades N, Chauhan D, Richardson P, Munshi NC, Anderson KC. Nuclear factor-kappaB p65 mediates tumor necrosis factor alpha-induced nuclear translocation of telomerase reverse transcriptase protein. Cancer Res. 2003;63:18–21. [PubMed] [Google Scholar]


