Abstract
AIM: To study the prevalence of Barrett’s esophagus in Chinese and its correlation with gastroesophageal reflux.
METHODS: This study was carried out in a large prospective series of 391 patients who had undergone upper endoscopy. The patients were divided into 3 groups according to the position of squamocolumnar junction (SCJ). Reflux esophagitis (RE) and its degree were recorded. Intestinal metaplasia (IM) in biopsy specimen was typed according to histochemistry and HE and alcian blue (pH2.5) staining separately. Results correlating with clinical, endoscopic, and pathological data were analysed.
RESULTS: The prevalence of IM endoscopically appearing Long-segment Barrett’s Esophagus (LSBE) was 26.53%, Short-segment Barrett’s Esophagus (SSBE) was 33.85% and gastroesophageal junction (GEJ) was 34.00%. IM increased with age of above 40 years old and no difference was found between male and female. Twelve were diagnosed as dysplasia (7 low -grade, 5 high-grade), 16 were diagnosed as cardiac adenocarcinoma and 1 as esophageal adenocarcinoma. The more far away the SCJ moved upward above GEJ, the higher the prevalence and the more severe the RE were.
CONCLUSION: There was no difference of the prevalence of IM in different places of SCJ, and IM increased with age of above 40 years old. It is important to pay attention to dysplasia in the distal esophagus and gastro-esophageal junction, and adenocarcinoma is more common in cardia than in esophagus. BE is a consequence of gastroesophageal reflux disease.
INTRODUCTION
Adenocarcinoma of the esophagus and gastro-esophageal junction (GEJ) is increasing, while the distal gastric cancer has been falling in the past two decades in North America, Europe, Japan and China. In China, the incidence of adenocarcinoma at GEJ is increasing more obviously[1-3].
Adenocarcinomas at the distal esophagus and GEJ differ from those in the rest of the stomach[4]. They share epidemiological characteristics with and often originate from segments of Barrett’s esophagus (BE). It is therefore proposed that both of them be called “esophagocardia adenocarcinoma”[5]. It has been well defined that Barrett’s esophagus is a premalignant condition for esophageal adenocarcinoma and most adenocarcinomas at GEJ[6,7].
In early studies, BE was defined as the presence of specialized IM in a columnar-lined mucosa encompassing more than 3 cm proximal to GEJ or a LSBE(long-segment Barrett’s esophagus)[8]. Any columnar-lined mucosa found less than 3 cm above the GEJ was thought to be a normal variant. However studies in the past years indicate that there is a spectrum of involvement that includes the distal 3 cm of esophagus or a SSBE (short-segment Barrett’s esophagus)[9,10].
Although the cause of BE has not been defined finally, it is considered that BE has a strong correlation with chronic GERD (gastro-esophageal reflux disease)[11]. Ten to twelve percent of patients with BE would receive endoscopy because of refluxing symptoms. Great attention has been paid to the studies of BE by overseas scholars. With the rapid development of China, the western food has become popular and fat persons are increasing, so is the GERD. But few studies on BE have been reported about the correlation between BE and GERD in Chinese. We performed a prospective study in order to determine the prevalence of BE in patients and to evaluate the correlation between BE and gastroesophageal reflux.
MATERIALS AND METHODS
Patients
Consecutive patients undergoing gastroscopy at our hospital from August 1, 2000 to August 30, 2001 were enrolled. Exclusion criteria were history of gastro-esophageal surgery and contraindication to performing biopsies. In each case, the following data were obtained: age, sex, and past medical history of ulcer, gastro-esophageal reflux symptoms (either heartburn or acid regurgitation), drug therapy, and past history of surgery.
The study was approved by the ethics committee of our hospital and all patients gave written informed consent prior to inclusion.
Endoscopic definition and biopsy
Endoscopy was performed by the same expert in a standard procedure, with visualization of the esophagus, stomach and duodenum. The appearance of SCJ was carefully studied in prograde view and after retroversion in the stomach. The position of GEJ was the boundary of tubular esophagus turning into stomach. According to the distance from GEJ to SCJ, the extent of the columnar lining, patients were divided into three groups[12]: Group A, 98 patients, whose SCJ was at least 3 cm above GEJ, suggesting a LSBE; Group B, 127 patients, whose SCJ was less than 3 cm above GEJ, suggesting SSBE; Group C, 150 patients, whose SCJ and GEJ were at nearly same level. Endoscopic esophagitis was graded into Grade I as mucosal hyperaemia, Grade II as non- circumferential mucosal breaks or erosions, Grade III as circumferential erosion or ulcer. Fifteen patients with cardiac adenocarcinoma and one patient with esophageal adenocarcinoma were excluded.
Standard four-quadrant biopsies were performed distal to the SCJ. Separate target biopsies were also taken from any suspicious erosions, nodules or ulcers. Four to six pieces of biopsy specimens were taken from each patient.
Biopsy specimens were fixed in 40 g/L buffered formaldehyde, embedded in paraffin wax, serially sectioned, and then stained with hematoxylin and eosin. BE was defined as the presence of distended, barrel-shaped goblet cells, indicative of intestinal metaplasia[6,13,14](Figure 1). BE was confirmed by further sectioning and staining the biopsies with alcian blue (pH2.5). Each section was reviewed by two experienced gastrointestinal pathologists respectively.
Figure 1.
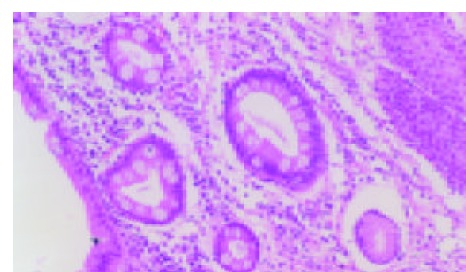
Goblet cells with HE stain (10 × 10).
Statistical analysis
Statistical analyses were performed using the SPSS9.0 analysis system. The χ2 or Fisher’s exact test was used when appropriate. P<0.05 was considered statistically significant.
RESULTS
These patients consisted of 211 males and 180 females with a mean age of 52.41 yr (range 20-82 yr). Reflux was found in 149 cases, pain at superior belly in 89 cases, dysphagia in 26 cases, nausea and vomiting in 18 cases, melena in 13 cases and emaciation in 5 cases.
Clinic features of study population
There were 103 patients with RE (39 grade I, 35 grade II and 29 grade III),69 with Barrett’s esophagus (26 LSBE, 43 SSBE), 51 with IM at GEJ, 12 cases with dysplasia [7 low–grade dysplasia, LGD (Figure 2A,B); 5 high–grade dysplasia, HGD (Figure 2C)], 17 with adenocarcinoma at the gastro-esophageal junction and 1 with adenocarcinoma of esophagus. The age was positively related to the progress from RE→BE→LGD→ HGD→adenocarcinoma (Table 1).
Figure 2.
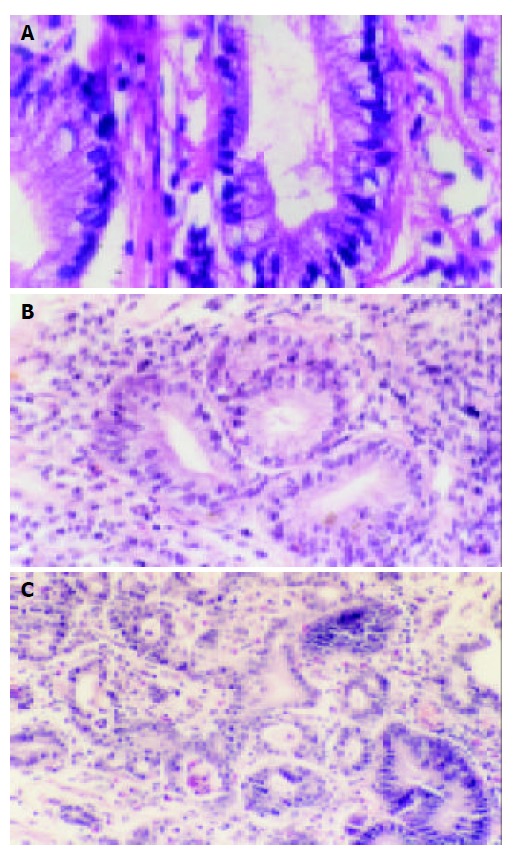
A: Barrett’s esophagus with LGD (10 × 40), B: Barrett’s esophagus with LGD (10 × 10), C: Barrett’s esophagus with HGD and carcinogenesis (top left corner) (10 × 10).
Table 1.
Clinical features of study population
| Number | Average age (yr) | Male:Female | |
| Total | 391 | 52.41 | 211:180 |
| RE I | 39 | 52.12 | 26:13 |
| II | 35 | 53.67 | 26:9 |
| III | 29 | 55.56 | 24:5 |
| IM at GEJ | 51 | 54.37 | 39:12 |
| SSBE | 43 | 54.71 | 34:9 |
| LSBE | 26 | 58.66 | 20:6 |
| LGD | 7 | 59.57 | 5:2 |
| HGD | 5 | 62.00 | 3:2 |
| Cardiac adenocarcinoma | 15 | 62.64 | 13:2 |
LGD: Low grade dysplasia, HGD: High grade dysplasia, IM: Intestinal metaplasia.
Prevalence of IM and dysplasia at different SCJ
There were no significant differences in incidence of IM between three groups and between male and female of each group (Table 2).
Table 2.
Prevalence of IM and dysplasia at different SCJ positions
| Position of SCJ | Number (male/female) | Age (yr) | IM (male/female) | Incidence of IM (%) |
Dysplasia |
|
| LGD | HGD | |||||
| Group A | 98 (61/37) | 58.66 | 26 (20/6) | 26.53 | 2 | 1 |
| Group B | 127 (63/64) | 55.05 | 43 (25/18) | 33.85 | 1 | 2 |
| Group C | 150 (71/79) | 54.37 | 51 (19/32) | 34.00 | 4 | 2 |
| Total | 375 (195/180) | 120 (64/56) | 32.00 | 7 | 5 | |
The incidence between groups and between male and female of each group was compared by χ2 test and P > 0.05.
Association of age and sex with the incidence of IM in different groups
In this study, IM was not found in patients under 30 years old. However, it increased in patients over 30 years old and gradually reached the peak over 40 years old. There were no significant differences of the incidence of IM between males and females in different age groups (P > 0.05)(Table 2).
Relation between RE and different position of SCJ
Comparing the incidence and degree of RE with the different position of SCJ, the positive correlation was found. The longer the SCJ moved upward above GEJ, the higher the prevalence and the more severe the RE were (Table 3).
Table 3.
Different SCJ positions and RE
| Position of SCJ | Number | RE I (%) | RE II (%) | RE III (%) | Total of RE (%) |
| Group A | 98 | 13 (13.26) | 19 (19.39) | 24 (24.48) | 56 (57.14) |
| Group B | 127 | 17 (13.38) | 10 (7.87) | 2 (1.57) | 29 (22.83) |
| Group C | 150 | 9 (6.00) | 6 (4.00) | 3 (2.00) | 18 (12.00) |
| The total | 375 | 39 (10.40) | 35 (9.33) | 29 (7.33) | 103 (27.46) |
Analysis by trend χ2 test, the incidence and degree of RE and the position of SCJ in groups C, B and A had a positive correlation. P < 0.05.
DISCUSSION
The point emphasized in original definition of Barrett’s esophagus was the length replaced by columnar epithelium of normal squamous mucosa. However, the length is not vital in new definition of Barrett’s esophagus, in which what is important is the presence of IM relating to adenocarcinoma[15]. Researches in recent years have implicated the correlation between carcinoma of gastric cardia and Barrett’s esophagus as well as IM at GEJ[16,17]. Some researches discovered that about 35-40% carcinomas of gastric cardia were related with Barrett’s esophagus, even some advanced esophageal adenocarcinomas at GEJ[18,19]. These findings at least illuminate some correlation. The relationship between Barrett’s esophagus and esophageal adenocarcinoma has been confirmed. Whether cardiac carcinoma is a consequence of IM is unclear.
The study population comprised 391 patients who underwent endoscopy. There were 103 patients with RE (39 grade I, 35 grade II and 29 grade III), 120 with Barrett’s esophagus (26 LSBE, 43 SSBE), 51 with IM at GEJ, 12 with dysplasia (LGD 7 cases , HGD 5 cases), 15 with carcinoma of gastric cardia. More lesions were found in males than in females. Furthermore, pathologic changes from RE→Barrett’s esophagus→dysplasia→canceration were related with increased age of patients.
According to the position of SCJ, the patients were divided into 3 groups in this study. The incidence of LSBE in group A was 26.53% (26/98), and the incidence of SSBE in group B was 33.85% (43/127) and IM in group C was 34.00% (51/150). These results were different from those of other scholars. Dias Percira[20] discovered that the incidence of SSBE in Portuguese was 61.3% and the incidence of IM at GEJ was 25%. The incidence of IM at GEJ in Euramerican varied from 5% to 36%[21,22]. Spechler [23] found that the incidence of LSBE was 65.38%, far higher than that in our study. The high incidence of LSBE was directly related to esophageal adenocarcinoma. Nakamura et al[24] found that the incidence of IM at GEJ was 28% in Japanese, which was close to our result. Of course, the exact position of GEJ is very difficult to locate under gastroscope, even with surgical specimens. The generally accepted view is that the position of GEJ is the boundary of tubular esophagus turning into stomach. Moreover, the number of biopsy specimens also works on the detection rate of IM. The more the number of biopsy specimens are, the higher the detection rate of IM is. The numbers of biopsy specimens are directly related to the identification of Barrett’s esophagus, however there are still no criteria as to how many biopsy specimens should be taken from one patient. Many scholars have suggested that more specimens should be taken in suspect lesions and one piece of specimen should be taken at least every 2 cm respectively from each quadrant. Paull et al.[25] found that IM was usually distributed in the proximal mucosa of lesion adjacent to squamoucolumnar junction (SCJ). Specimens adjacent to SCJ are helpful to increase the detection rate of IM. In this study, at least one piece of specimen was taken respectively from each quadrant of distal columnar mucosa adjacent to SCJ and more specimens were taken if there were questionable lesions. So, 4-6 biopsies were taken from each patient. For biopsy specimens, the goblet cell secreting acidic mucus could be dyed blue by AB (pH2.5) staining, which was beneficial to the diagnosis of IM[26].
Increasing researches considered that IM at GEJ had two origins, SSBE and IM of gastric cardia[27]. Whether it is necessary to study SSBE and IM at GEJ separately or whether they are the same lesion is unknown. Besides, there are no exact definitions of IM at distal part of esophagus and at gastric cardia. Researches in recent years showed that the development of IM at GEJ might be related with infection of H pylori[28]. The infection of H pylori in China is common, therefore, it is necessary to do more investigations.
In this research, IM was not found in patients younger than 30 years old. The incidence of IM increased with the age and reached the peak at the age of 50-70 yr, but the incidence of IM had no significant differences between males and females in different age groups and at different SCJ positions (P>0.05). It was reported that attacks of Barrett’s esophagus were seldom found in children and young people, but it would increase with the age. It will take about 20 yr to develop from Barrett’s esophagus to esophageal adenocarcinoma[29].
Researches showed that Barrett’s esophagus was a precancerous lesion of esophageal adenocarcinoma and gastric cardia carcinoma. Pathologic researches discovered that canceration of Barrett’s esophagus would undergo a pathologic process from SIM→LGD→HGD→carcinoma in situ→advanced adenocarcinoma. Some researches discovered that it took 1.5-10 yr to develop into adenocarcinoma from HGD[30,31]. A few patients would remain at the stage of HGD for many years. Part of patients would develop into adenocarcinoma from IM in 3-10 years[30,32]. Seven patients with LGD and 5 with HGD were diagnosed in this study and the follow-up for the patients with dysplasia should be strengthened. The patients with HGD should be followed up every 1-3 mo and multipoint biopsy should be taken to avoid delaying the diagnosis of tumors. We diagnosed 15 cases of cardiac adenocarcinoma and 1 case of esophageal adenocarcinoma, which showed that adenocarcinoma of cardia was more common than that of esophagus in China.
It is recognized by most scholars that RE leads to Barrett’s esophagus. In our study (Table 3), the incidence of RE in group A (57.14%) was higher than that in group B (22.83%) and was higher in group B than in group C (12.00%). Furthermore, the incidence of RE III was also far higher in group A (24.48%) than in group B (1.57%). Dias found that the occurrence of RE in group of SSBE was higher than that in group of GEJ, which was similar to our conclusion[20]. We examined the incidence and degree of RE as well as the SCJ position in groups C, B, A and found that they had a positive correlation (P < 0.05), that is, the longer the SCJ moved upward above GEJ, the higher the prevalence and the more severe of RE were. The result in this study conformed to the viewpoint of most researchers that the squamous epithelium of lower esophagus was replaced by columnar epithelium and then developed into intestinal metaplasia. There is no exact conclusion about whether IM at GEJ has relation to RE or not.
In a word, it is important to pay attention to the diagnosis of dysplasia in the distal esophagus and esophagogastric junctions. Cardia adenocarcinoma is more common than that of esophagus. BE is a consequence of gastroesophageal reflux disease.
Footnotes
Edited by Wang XL and Zhu LH Proofread by Xu FM
References
- 1.Devesa SS, Blot WJ, Fraumeni JF. Changing patterns in the incidence of esophageal and gastric carcinoma in the United States. Cancer. 1998;83:2049–2053. [PubMed] [Google Scholar]
- 2.Blot WJ, Devesa SS, Kneller RW, Fraumeni JF. Rising incidence of adenocarcinoma of the esophagus and gastric cardia. JAMA. 1991;265:1287–1289. [PubMed] [Google Scholar]
- 3.Zhou Q, Wang LD. Biologic characteristic of adenocarcinoma at GEJ. Shijie Huaren Xiaohua Zazhi. 1998;6:636–637. [Google Scholar]
- 4.Ruol A, Parenti A, Zaninotto G, Merigliano S, Costantini M, Cagol M, Alfieri R, Bonavina L, Peracchia A, Ancona E. Intestinal metaplasia is the probable common precursor of adenocarcinoma in barrett esophagus and adenocarcinoma of the gastric cardia. Cancer. 2000;88:2520–2528. doi: 10.1002/1097-0142(20000601)88:11<2520::aid-cncr13>3.0.co;2-l. [DOI] [PubMed] [Google Scholar]
- 5.Rabinovitch PS, Reid BJ, Haggitt RC, Norwood TH, Rubin CE. Progression to cancer in Barrett's esophagus is associated with genomic instability. Lab Invest. 1989;60:65–71. [PubMed] [Google Scholar]
- 6.Weston AP, Krmpotich PT, Cherian R, Dixon A, Topalovski M. Prospective evaluation of intestinal metaplasia and dysplasia within the cardia of patients with Barrett's esophagus. Dig Dis Sci. 1997;42:597–602. doi: 10.1023/a:1018811512939. [DOI] [PubMed] [Google Scholar]
- 7.Cameron AJ, Lomboy CT, Pera M, Carpenter HA. Adenocarcinoma of the esophagogastric junction and Barrett's esophagus. Gastroenterology. 1995;109:1541–1546. doi: 10.1016/0016-5085(95)90642-8. [DOI] [PubMed] [Google Scholar]
- 8.Iascone C, DeMeester TR, Little AG, Skinner DB. Barrett's esophagus. Functional assessment, proposed pathogenesis, and surgical therapy. Arch Surg. 1983;118:543–549. doi: 10.1001/archsurg.1983.01390050027005. [DOI] [PubMed] [Google Scholar]
- 9.Pera M. Trends in incidence and prevalence of specialized intestinal metaplasia, barrett's esophagus, and adenocarcinoma of the gastroesophageal junction. World J Surg. 2003;27:999–1008; discussion 1006-8. doi: 10.1007/s00268-003-7052-2. [DOI] [PubMed] [Google Scholar]
- 10.Weston AP, Krmpotich P, Makdisi WF, Cherian R, Dixon A, McGregor DH, Banerjee SK. Short segment Barrett's esophagus: clinical and histological features, associated endoscopic findings, and association with gastric intestinal metaplasia. Am J Gastroenterol. 1996;91:981–986. [PubMed] [Google Scholar]
- 11.Hirota WK, Loughney TM, Lazas DJ, Maydonovitch CL, Rholl V, Wong RK. Specialized intestinal metaplasia, dysplasia, and cancer of the esophagus and esophagogastric junction: prevalence and clinical data. Gastroenterology. 1999;116:277–285. doi: 10.1016/s0016-5085(99)70123-x. [DOI] [PubMed] [Google Scholar]
- 12.Spechler SJ. Columnar-lined esophagus. Definitions. Chest Surg Clin N Am. 2002;12:1–13, vii. doi: 10.1016/s1052-3359(03)00087-5. [DOI] [PubMed] [Google Scholar]
- 13.Menke-Pluymers MB, Hop WC, Dees J, van Blankenstein M, Tilanus HW. Risk factors for the development of an adenocarcinoma in columnar-lined (Barrett) esophagus. The Rotterdam Esophageal Tumor Study Group. Cancer. 1993;72:1155–1158. doi: 10.1002/1097-0142(19930815)72:4<1155::aid-cncr2820720404>3.0.co;2-c. [DOI] [PubMed] [Google Scholar]
- 14.Rajan E, Burgart LJ, Gostout CJ. Endoscopic and histologic diagnosis of Barrett esophagus. Mayo Clin Proc. 2001;76:217–225. doi: 10.1016/S0025-6196(11)63133-9. [DOI] [PubMed] [Google Scholar]
- 15.Sampliner RE. Practice guidelines on the diagnosis, surveillance, and therapy of Barrett's esophagus. The Practice Parameters Committee of the American College of Gastroenterology. Am J Gastroenterol. 1998;93:1028–1032. doi: 10.1111/j.1572-0241.1998.00362.x. [DOI] [PubMed] [Google Scholar]
- 16.Zaninotto G, Avellini C, Barbazza R, Baruchello G, Battaglia G, Benedetti E, Bernardi A, Boccù C, Bonoldi E, Bottona E, et al. Prevalence of intestinal metaplasia in the distal oesophagus, oesophagogastric junction and gastric cardia in symptomatic patients in north-east Italy: a prospective, descriptive survey. The Italian Ulcer Study Group "GISU". Dig Liver Dis. 2001;33:316–321. doi: 10.1016/s1590-8658(01)80084-0. [DOI] [PubMed] [Google Scholar]
- 17.Hamilton SR, Smith RR, Cameron JL. Prevalence and characteristics of Barrett esophagus in patients with adenocarcinoma of the esophagus or esophagogastric junction. Hum Pathol. 1988;19:942–948. doi: 10.1016/s0046-8177(88)80010-8. [DOI] [PubMed] [Google Scholar]
- 18.Rotterdam H. Pathology of the gastric cardia. Verh Dtsch Ges Pathol. 1999;83:37–42. [PubMed] [Google Scholar]
- 19.Clark GW, Smyrk TC, Burdiles P, Hoeft SF, Peters JH, Kiyabu M, Hinder RA, Bremner CG, DeMeester TR. Is Barrett's metaplasia the source of adenocarcinomas of the cardia? Arch Surg. 1994;129:609–614. doi: 10.1001/archsurg.1994.01420300051007. [DOI] [PubMed] [Google Scholar]
- 20.Pereira AD, Suspiro A, Chaves P, Saraiva A, Glória L, de Almeida JC, Leitão CN, Soares J, Mira FC. Short segments of Barrett's epithelium and intestinal metaplasia in normal appearing oesophagogastric junctions: the same or two different entities? Gut. 1998;42:659–662. doi: 10.1136/gut.42.5.659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Voutilainen M, Färkkilä M, Juhola M, Nuorva K, Mauranen K, Mäntynen T, Kunnamo I, Mecklin JP, Sipponen P. Specialized columnar epithelium of the esophagogastric junction: prevalence and associations. The Central Finland Endoscopy Study Group. Am J Gastroenterol. 1999;94:913–918. doi: 10.1111/j.1572-0241.1999.986_i.x. [DOI] [PubMed] [Google Scholar]
- 22.Hirota WK, Loughney TM, Lazas DJ, Maydonovitch CL, Rholl V, Wong RK. Specialized intestinal metaplasia, dysplasia, and cancer of the esophagus and esophagogastric junction: prevalence and clinical data. Gastroenterology. 1999;116:277–285. doi: 10.1016/s0016-5085(99)70123-x. [DOI] [PubMed] [Google Scholar]
- 23.Spechler SJ, Zeroogian JM, Antonioli DA, Wang HH, Goyal RK. Prevalence of metaplasia at the gastro-oesophageal junction. Lancet. 1994;344:1533–1536. doi: 10.1016/s0140-6736(94)90349-2. [DOI] [PubMed] [Google Scholar]
- 24.Nakamura M, Kawano T, Endo M, Iwai T. Intestinal metaplasia at the esophagogastric junction in Japanese patients without clinical Barrett's esophagus. Am J Gastroenterol. 1999;94:3145–3149. doi: 10.1111/j.1572-0241.1999.01506.x. [DOI] [PubMed] [Google Scholar]
- 25.Paull A, Trier JS, Dalton MD, Camp RC, Loeb P, Goyal RK. The histologic spectrum of Barrett's esophagus. N Engl J Med. 1976;295:476–480. doi: 10.1056/NEJM197608262950904. [DOI] [PubMed] [Google Scholar]
- 26.Fireman Z, Wagner G, Weissman J, Kopelman Y, Wagner Y, Groissman G, Sternberg A. Prevalence of short-segment Barrett's epithelium. Dig Liver Dis. 2001;33:322–325. doi: 10.1016/s1590-8658(01)80085-2. [DOI] [PubMed] [Google Scholar]
- 27.Sharma P, Weston AP, Morales T, Topalovski M, Mayo MS, Sampliner RE. Relative risk of dysplasia for patients with intesti-nal metaplasia in the distal oesophagus and in the gastric cardia. Gut. 2000;46:9–13. doi: 10.1136/gut.46.1.9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hackelsberger A, Günther T, Schultze V, Manes G, Dominguez-Muñoz JE, Roessner A, Malfertheiner P. Intestinal metaplasia at the gastro-oesophageal junction: Helicobacter pylori gastritis or gastro-oesophageal reflux disease? Gut. 1998;43:17–21. doi: 10.1136/gut.43.1.17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cameron AJ. Epidemiologic studies and the development of Barrett's esophagus. Endoscopy. 1993;25:635–636. doi: 10.1055/s-2007-1010420. [DOI] [PubMed] [Google Scholar]
- 30.Hameeteman W, Tytgat GN, Houthoff HJ, van den Tweel JG. Barrett's esophagus: development of dysplasia and adenocarcinoma. Gastroenterology. 1989;96:1249–1256. doi: 10.1016/s0016-5085(89)80011-3. [DOI] [PubMed] [Google Scholar]
- 31.Schnell TG, Sontag SJ, Chejfec G, Aranha G, Metz A, O'Connell S, Seidel UJ, Sonnenberg A. Long-term nonsurgical management of Barrett's esophagus with high-grade dysplasia. Gastroenterology. 2001;120:1607–1619. doi: 10.1053/gast.2001.25065. [DOI] [PubMed] [Google Scholar]
- 32.Sharma P, Morales TG, Bhattacharyya A, Garewal HS, Sampliner RE. Dysplasia in short-segment Barrett's esophagus: a prospective 3-year follow-up. Am J Gastroenterol. 1997;92:2012–2016. [PubMed] [Google Scholar]


